Preliminary Evaluation of an Adaptive Robotic Training Program of the Wrist for Persons with Multiple Sclerosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
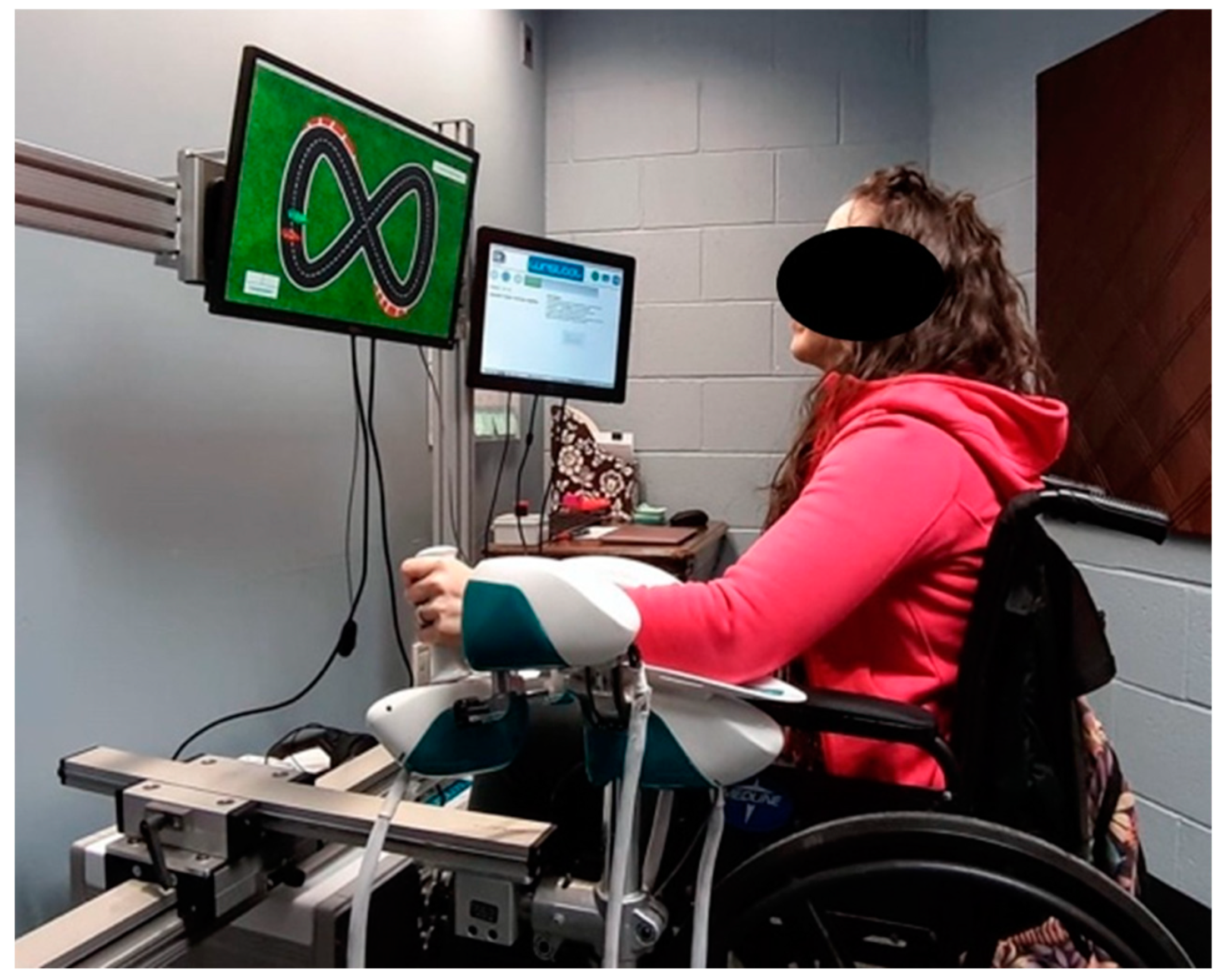
2.2. Experimental Set-Up and Protocol
2.3. Assessment Sessions
- Active and passive wrist ROM in flexion/extension and radial/ulnar deviation. Participants moved actively or were passively moved by the WristBot towards each direction until they reached maximal excursion. The outcome measure was maximal wrist rotation in degrees recorded by encoders along each direction.
- The ability to track a target moving in the space identified by flexion/extension and radial/ulnar deviation. Without any assistive force, participants actively moved the handle of the robot to follow a target over a Lissajous figure (figure-eight shape) for 3 min. The pronation/supination plane was locked during the task. For each participant, the size of the figure never exceeded the 80% of the active ROM previously assessed. The trajectory was described by the following law of motion Equation (1):where min(ROM) is the minimum active ROM among the four directions assessed and T = 30 s (the duration of a complete lap). Outcome measures computed from these data were the Tracking Error (TE) and the Figural Error (FE). TE was defined as the mean angular distance in degrees between end-effector and target position at each time point. This outcome measure evaluated the performance, taking into account the difference in velocity between the end-effector and the target. TE was computed starting from the data recorded by the encoders along the flexion/extension (x [deg]) and radial/ulnar deviation (y [deg]) planes. Robot encoders recorded angular data, i.e., rotations along each degree of freedom. The measure is obtained through Equation (2):where T,i and H,i are the target and the end-effector positions at each time sample and N is the total number of time samples. FE was measured to characterize performance as the trajectory of the participant from the ideal figure-eight shape (target trajectory), regardless of differences of speed between target and end-effector. FE was measured in degrees and computed as shown in Equation (3):where A and n are the time series (A[x(t),y(t)]) and total samples of the target trajectory and B and m are the time series (B[x(t),y(t)]) and total samples of the end-effector trajectory [20].
- Maximum grip force to measure overall grip strength. This was performed twice on each limb and the highest force in kg was recorded (Jamar Smart Digital Hand Dynamometer, Performance Health, Warrenville, IL, USA).
- Grip force endurance test to measure muscular endurance on each limb. Participants gripped the dynamometer at 50% of their maximum grip force and the time to fatigue in seconds (defined as when the participant could no longer hold 50% of their maximum for 2 consecutive seconds) was recorded.
- Maximum isometric wrist force measured in kg was determined for flexion/extension and radial/ulnar deviation using a stationary load cell (Model: BG 500, Mark-10 Corporation, New York, NY, USA).
- 19-Hole Peg Test (9-HPT) was performed twice on each limb. 9HPT is used in clinics to evaluate finger dexterity [21] accordingly to the time taken to accomplish the required tasks. One participant was physically unable to complete the test with the trained limb at T0 due to a disabling tremor and was eliminated from the mean values.
- The Patient Rated Wrist Evaluation (PRWE) was administered, and participants had to answer 15 questions that pertained to pain, ADLs in the affected limb and activities that require the use of both limbs [22]. Higher final scores point out higher levels of wrist disability.
2.4. Training Sessions
3. Data and Statistical Analysis
4. Results
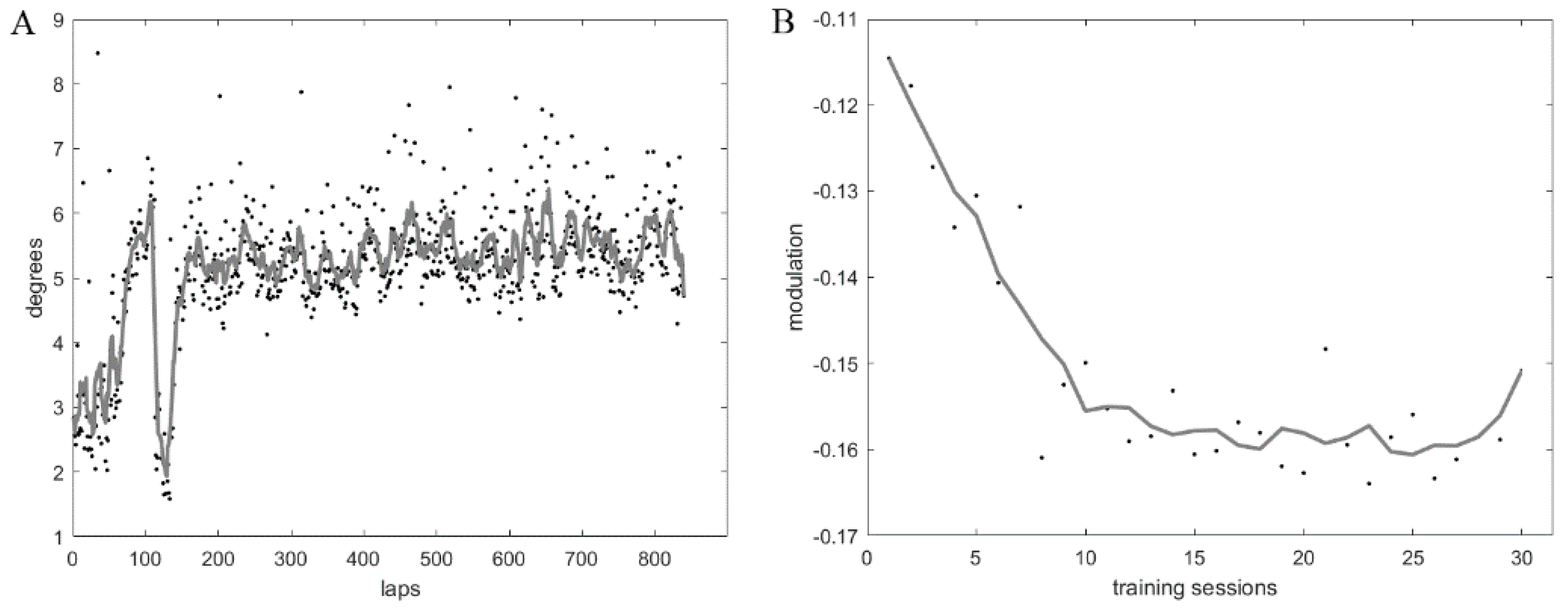
4.1. Training
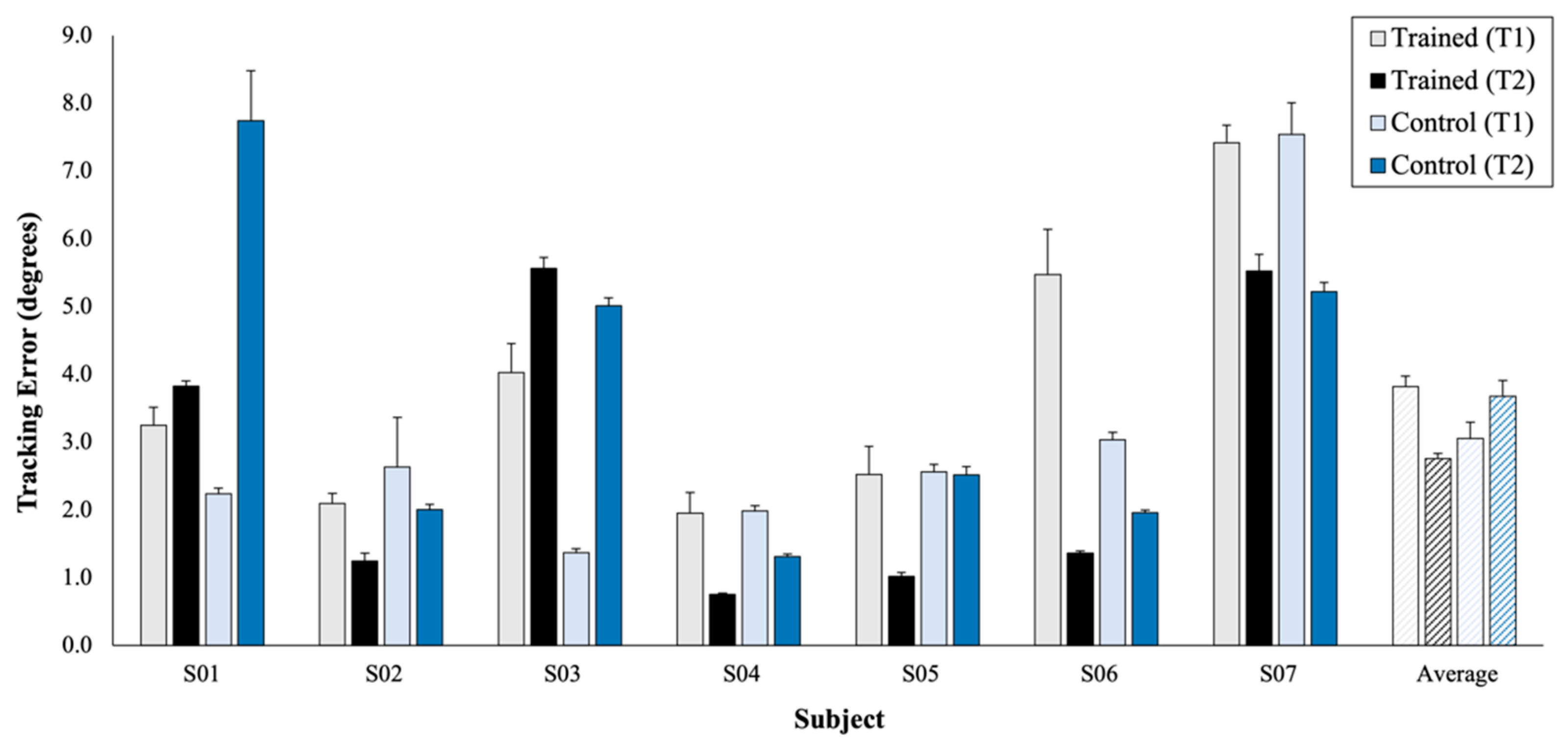
4.2. Assessment: Wrist Kinematics
4.3. Assessment: Wrist Strength
4.4. Assessment: Clinical Outcome Measures
4.5. Correlations
5. Discussion
6. Limitations
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Walton, C.; King, R.; Rechtman, L.; Kaye, W.; Leray, E.; Marrie, R.A.; Robertson, N.; La Rocca, N.; Uitdehaag, B.; van der Mei, I.; et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Mult. Scler. J. 2020, 26, 1816–1821. [Google Scholar] [CrossRef] [PubMed]
- Weiner, H.L.; Stankiewicz, J.M. Multiple Sclerosis: Diagnosis and Therapy; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Burks, J.S.; Johnson, K.P. Multiple Sclerosis: Diagnosis, Medical Management, and Rehabilitation; Demos Medical Publishing Inc.: New York, NY, USA, 2000. [Google Scholar]
- Zhong, J.; Nantes, J.C.; Holmes, S.A.; Gallant, S.; Narayanan, S.; Koski, L. Abnormal functional connectivity and cortical integrity influence dominant hand motor disability in multiple sclerosis: A multimodal analysis: FC, Structural and Behavioural Changes in MS. Hum. Brain Mapp. 2016, 37, 4262–4275. [Google Scholar] [CrossRef] [PubMed]
- Mekki, M.; Delgado, A.D.; Fry, A.; Putrino, D.; Huang, V. Robotic Rehabilitation and Spinal Cord Injury: A Narrative Review. Neurotherapeutics 2018, 15, 604–617. [Google Scholar] [CrossRef] [PubMed]
- Olek, M.J. Multiple Sclerosis: Etiology, Diagnosis, and New Treatment Strategies; Humana Press: Totowa, NJ, USA, 2005. [Google Scholar]
- Vergaro, E.; Squeri, V.; Brichetto, G.; Casadio, M.; Morasso, P.; Solaro, C.; Sanguineti, V. Adaptive robot training for the treatment of incoordination in Multiple Sclerosis. J. Neuroeng. Rehabil. 2010, 7. [Google Scholar] [CrossRef] [PubMed]
- Krebs, H.I.; Volpe, B.T.; Williams, D.; Celestino, J.; Charles, S.K.; Lynch, D.; Hogan, N. Robot-Aided Neurorehabilitation: A Robot for Wrist Rehabilitation. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Lo, H.S.; Xie, S.Q. Exoskeleton robots for upper-limb rehabilitation: State of the art and future prospects. Med. Eng. Phys. 2012, 34, 261–268. [Google Scholar] [CrossRef]
- Vergaro, E.; Casadio, M.; Squeri, V.; Giannoni, P.; Morasso, P.; Sanguineti, V. Self-adaptive robot training of stroke survivors for continuous tracking movements. J. Neuroeng. Rehabil. 2010, 7. [Google Scholar] [CrossRef]
- Colombo, R.; Pisano, F.; Micera, S.; Mazzone, A.; Delconte, C.; Carrozza, M.C.; Dario, P.; Minuco, G. Robotic Techniques for Upper Limb Evaluation and Rehabilitation of Stroke Patients. IEEE Trans. Neural Syst. Rehabil. Eng. 2005, 13, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Carpinella, I.; Cattaneo, D.; Abuarqub, S.; Ferrarin, M. Robot-based rehabilitation of the upper limbs in multiple sclerosis: Feasibility and preliminary results. J. Rehabil. Med. 2009, 41, 966–970. [Google Scholar] [CrossRef]
- Rodgers, H.; Bosomworth, H.; Krebs, H.I.; van Wijck, F.; Howel, D.; Wilson, N.; Aird, L.; Alvarado, N.; Andole, S.; Cohen, D.L.; et al. Robot assisted training for the upper limb after stroke (RATULS): A multicentre randomised controlled trial. Lancet 2019, 394, 51–62. [Google Scholar] [CrossRef]
- Albanese, G.A.; Holmes, M.W.R.; Marini, F.; Morasso, P.; Zenzeri, J. Wrist Position Sense in Two Dimensions: Between-Hand Symmetry and Anisotropic Accuracy Across the Space. Front. Hum. Neurosci. 2021, 15, 662768. [Google Scholar] [CrossRef]
- Albanese, G.A.; Marini, F.; Taglione, E.; Gasparini, C.; Grandi, S.; Pettinelli, F.; Sardelli, C.; Catitti, P.; Sandini, G.; Masia, L.; et al. Assessment of human wrist rigidity and pain in post-traumatic patients. In Proceedings of the 2019 IEEE 16th International Conference on Rehabilitation Robotics (ICORR), Toronto, ON, Canada, 24–28 June 2019; pp. 89–94. [Google Scholar]
- Mugnosso, M.; Marini, F.; Holmes, M.; Morasso, P.; Zenzeri, J. Muscle fatigue assessment during robot-mediated movements. J. Neuroeng. Rehabil. 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Iandolo, R.; Marini, F.; Semprini, M.; Laffranchi, M.; Mugnosso, M.; Cherif, A.; De Michieli, L.; Chiappalone, M.; Zenzeri, J. Perspectives and Challenges in Robotic Neurorehabilitation. Appl. Sci. 2019, 9, 3183. [Google Scholar] [CrossRef]
- Blank, A.A.; French, J.A.; Pehlivan, A.U.; O’Malley, M.K. Current Trends in Robot-Assisted Upper-Limb Stroke Rehabilitation: Promoting Patient Engagement in Therapy. Curr. Phys. Med. Rehabil. Rep. 2014, 2, 184–195. [Google Scholar] [CrossRef]
- Kleim, J.A.; Jones, T.A. Principles of Experience-Dependent Neural Plasticity: Implications for Rehabilitation After Brain Damage. J. Speech Lang. Hear. Res. 2008, 51. [Google Scholar] [CrossRef]
- Conditt, M.A.; Gandolfo, F.; Mussa-Ivaldi, F.A. The Motor System Does Not Learn the Dynamics of the Arm by Rote Memorization of Past Experience. J. Neurophysiol. 1997, 78, 554–560. [Google Scholar] [CrossRef]
- Halabchi, F.; Alizadeh, Z.; Sahraian, M.A.; Abolhasani, M. Exercise prescription for patients with multiple sclerosis; potential benefits and practical recommendations. BMC Neurol. 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Carroll, T.J.; Herbert, R.D.; Munn, J.; Lee, M.; Gandevia, S.C. Contralateral effects of unilateral strength training: Evidence and possible mechanisms. J. Appl. Physiol. 2006, 101, 1514–1522. [Google Scholar] [CrossRef]
- Lamers, I.; Maris, A.; Severijns, D.; Dielkens, W.; Geurts, S.; Van Wijmeersch, B.; Feys, P. Upper Limb Rehabilitation in People With Multiple Sclerosis: A Systematic Review. Neurorehabil. Neural Repair 2016, 30, 773–793. [Google Scholar] [CrossRef]
- Albanese, G.A.; Taglione, E.; Gasparini, C.; Grandi, S.; Pettinelli, F.; Sardelli, C.; Catitti, P.; Sandini, G.; Masia, L.; Zenzeri, J. Efficacy of wrist robot-aided orthopedic rehabilitation: A randomized controlled trial. J. Neuroeng. Rehabil. 2021, 18, 130. [Google Scholar] [CrossRef]
- Casadio, M.; Sanguineti, V. Learning, Retention, and Slacking: A Model of the Dynamics of Recovery in Robot Therapy. IEEE Trans. Neural Syst. Rehabil. Eng. 2012, 20, 286–296. [Google Scholar] [CrossRef]
- Schoenfeld, B.J.; Grgic, J.; Ogborn, D.; Krieger, J.W. Strength and Hypertrophy Adaptations Between Low- vs. High-Load Resistance Training: A Systematic Review and Meta-analysis. J. Strength Cond. Res. 2017, 31, 3508–3523. [Google Scholar] [CrossRef]
- Green, L.A.; Gabriel, D.A. The effect of unilateral training on contralateral limb strength in young, older, and patient populations: A meta-analysis of cross education. Phys. Ther. Rev. 2018, 23, 238–249. [Google Scholar] [CrossRef]
- Carlos, M.-P.; Fernanda, d.S.-T.; Rodrigo, F.-G.; Antonio, d.P.-F.J. Effects of a resistance training program and subsequent detraining on muscle strength and muscle power in multiple sclerosis patients. NeuroRehabilitation 2014, 523–530. [Google Scholar] [CrossRef]
- Sun, Y.; Ledwell, N.M.H.; Boyd, L.A.; Zehr, E.P. Unilateral wrist extension training after stroke improves strength and neural plasticity in both arms. Exp. Brain Res. 2018, 236, 2009–2021. [Google Scholar] [CrossRef] [PubMed]

| Age | MS Phenotype | Dominant Limb | Trained Limb | Years since Diagnosis | EDSS Score | Sex | |
|---|---|---|---|---|---|---|---|
| S01 | 36 | SPMS | R | L | 14 | 7 | Female |
| S02 | 60 | PPMS | R | L | 20 | 7 | Female |
| S03 | 71 | SPMS | R | L | 20 | 3 | Male |
| S04 | 43 | RRMS | R | R | 6 | 6.5 | Female |
| S05 | 61 | SPMS | L | R | 34 | 6 | Female |
| S06 | 27 | RRMS | R | R | 1 ½ | 4 | Male |
| S07 | 30 | RRMS | R | L | 7 | 2 | Female |
| Direction | p-Value | r2 |
|---|---|---|
| Flexion | 0.03 * | 0.64 |
| Extension | 0.02 * | 0.68 |
| Radial Deviation | 0.04 * | 0.59 |
| Ulnar Deviation | 0.08 | 0.49 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mannella, K.; Albanese, G.A.; Ditor, D.; Zenzeri, J.; Holmes, M.W.R. Preliminary Evaluation of an Adaptive Robotic Training Program of the Wrist for Persons with Multiple Sclerosis. Appl. Sci. 2021, 11, 9239. https://doi.org/10.3390/app11199239
Mannella K, Albanese GA, Ditor D, Zenzeri J, Holmes MWR. Preliminary Evaluation of an Adaptive Robotic Training Program of the Wrist for Persons with Multiple Sclerosis. Applied Sciences. 2021; 11(19):9239. https://doi.org/10.3390/app11199239
Chicago/Turabian StyleMannella, Kailynn, Giulia A. Albanese, David Ditor, Jacopo Zenzeri, and Michael W. R. Holmes. 2021. "Preliminary Evaluation of an Adaptive Robotic Training Program of the Wrist for Persons with Multiple Sclerosis" Applied Sciences 11, no. 19: 9239. https://doi.org/10.3390/app11199239
APA StyleMannella, K., Albanese, G. A., Ditor, D., Zenzeri, J., & Holmes, M. W. R. (2021). Preliminary Evaluation of an Adaptive Robotic Training Program of the Wrist for Persons with Multiple Sclerosis. Applied Sciences, 11(19), 9239. https://doi.org/10.3390/app11199239






