The Conformity of Rehabilitation Protocols Used for Different Cartilage Repairs of the Knee Joint—A Review on Rehabilitation Standards in German Speaking Countries
Abstract
1. Introduction
2. Materials and Methods
2.1. Measurement Criteria
2.2. Participants
2.3. Statistical Analysis
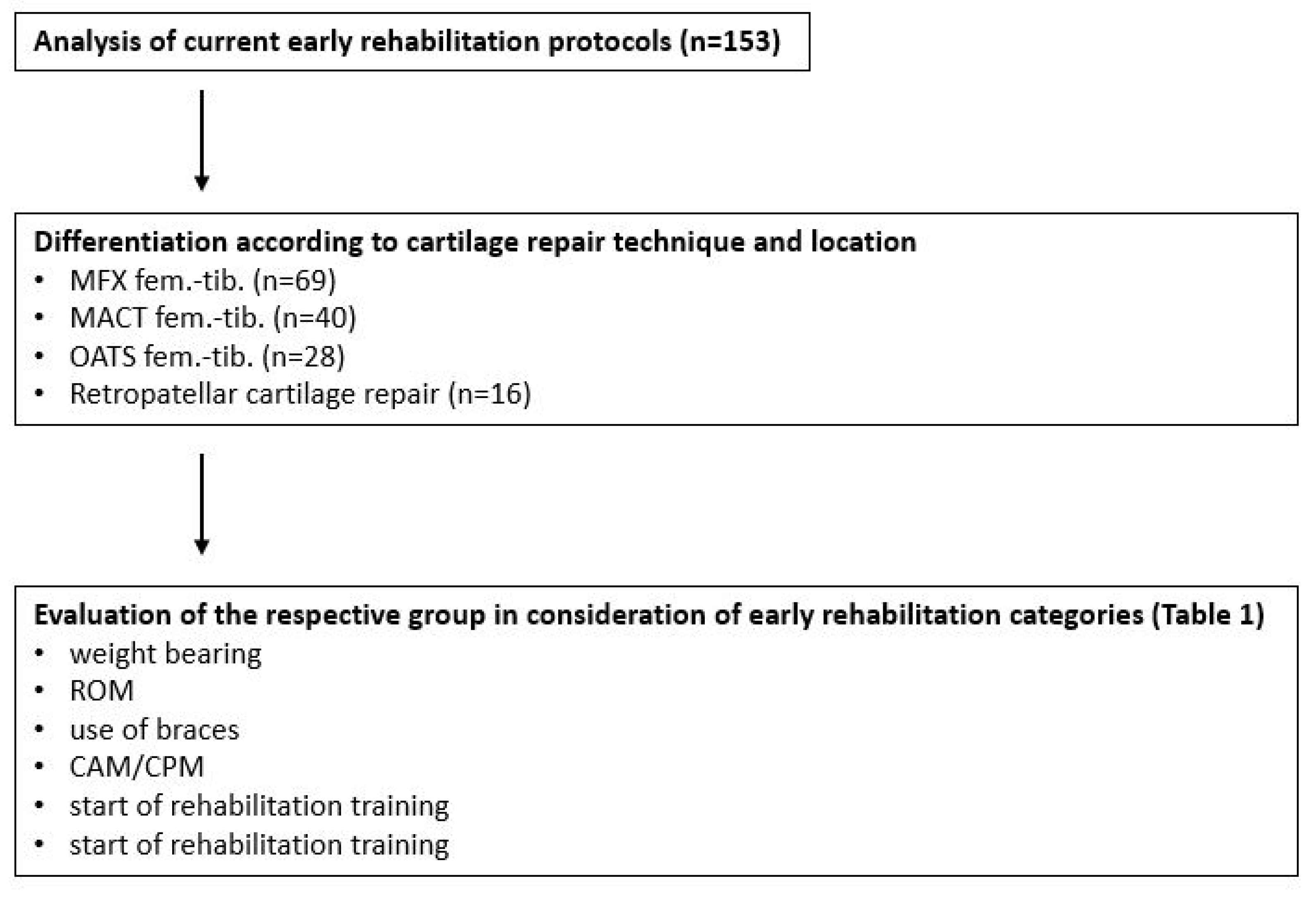
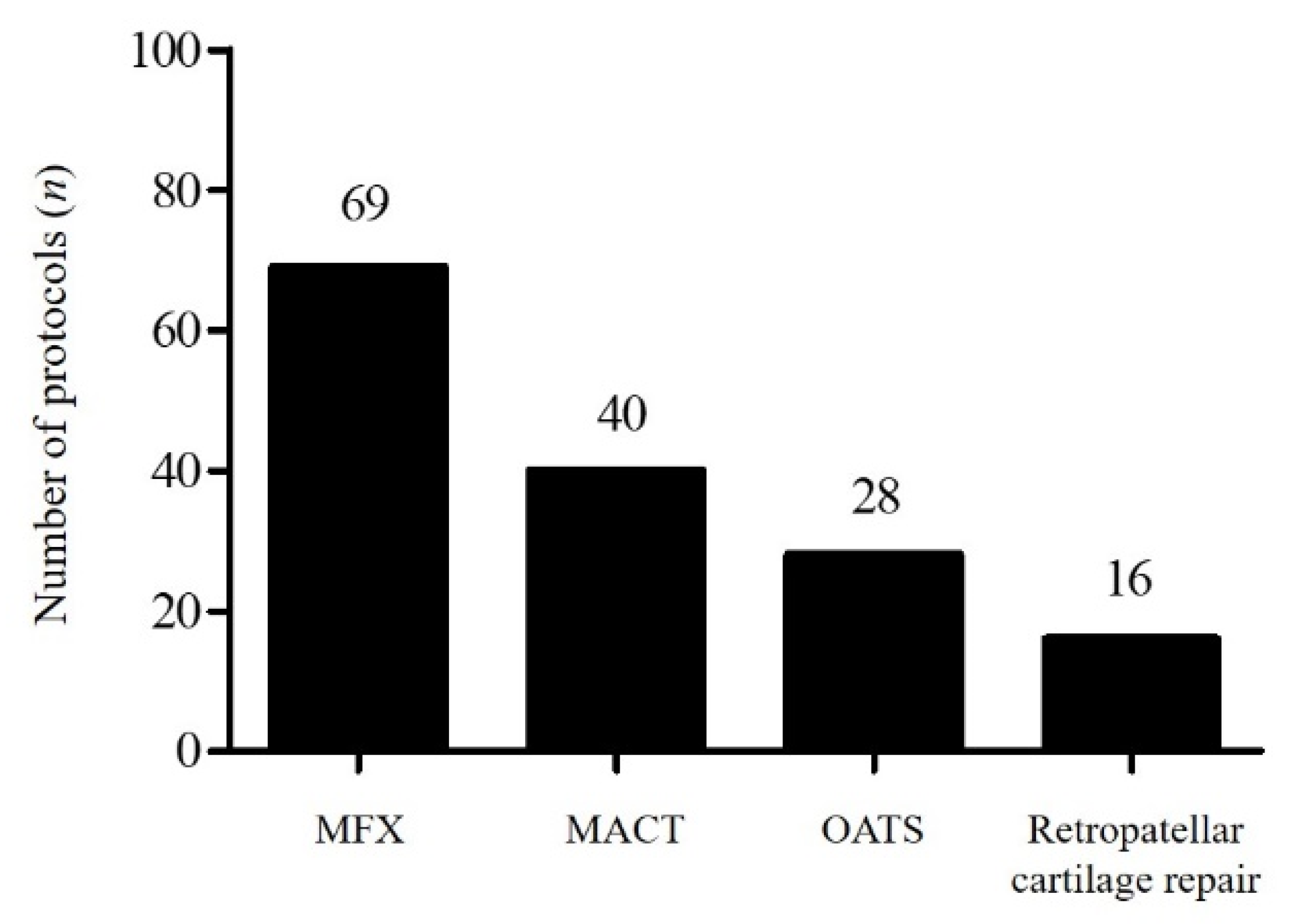
3. Results
3.1. Evaluation Algorithm
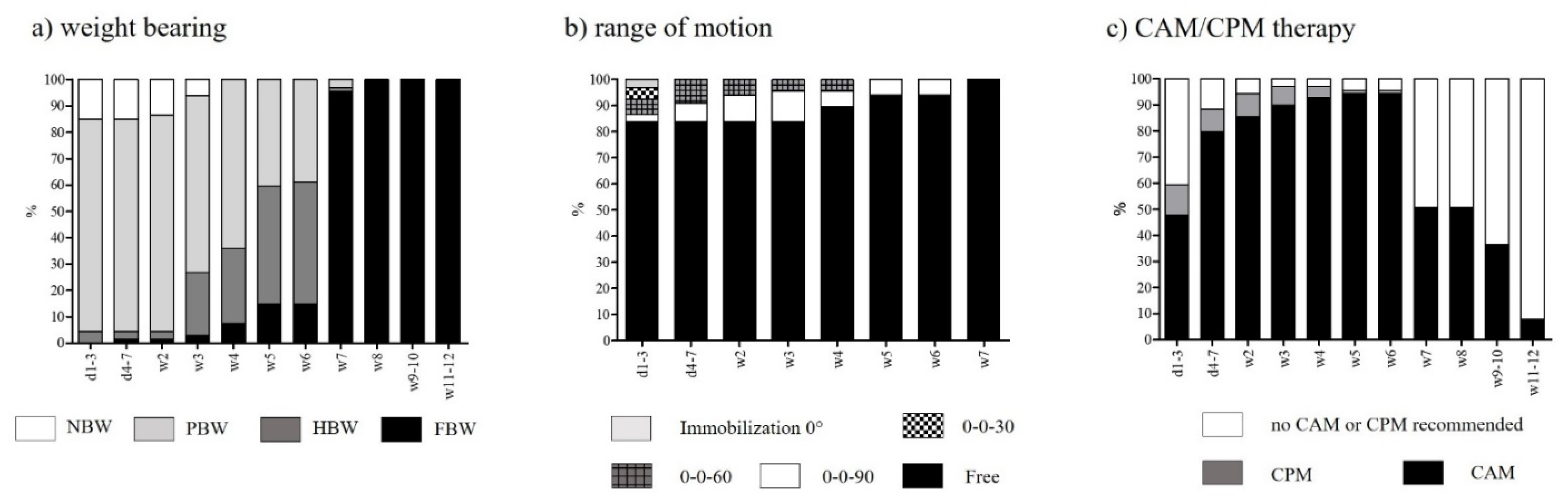
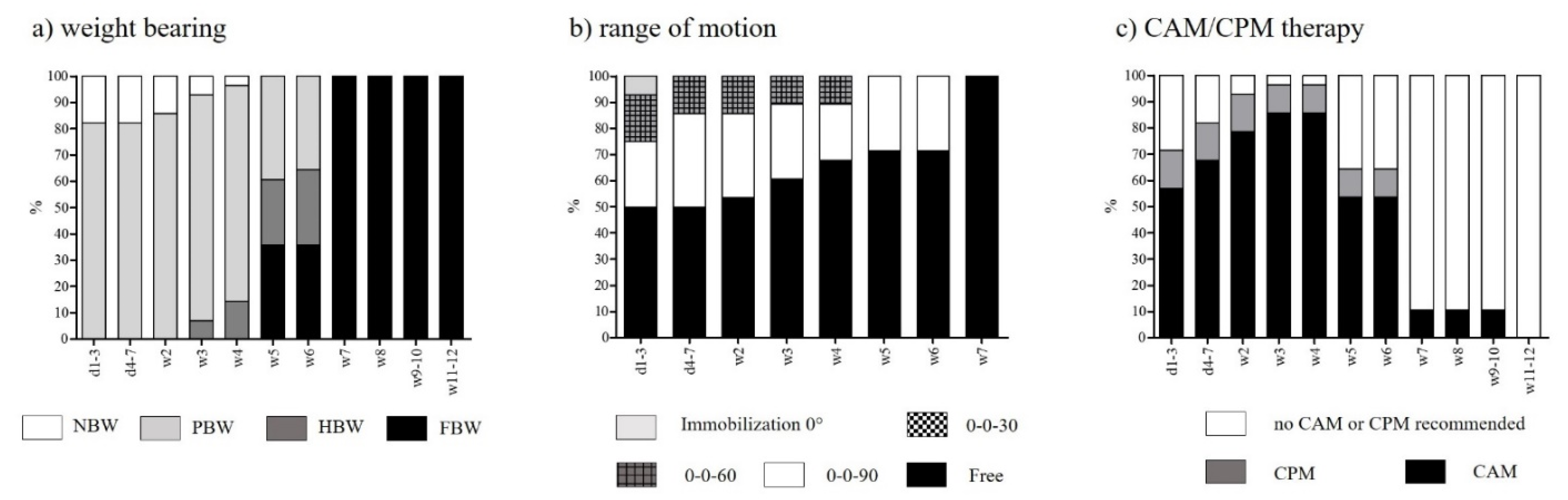
3.2. Rehabilitation after Femorotibial MFX
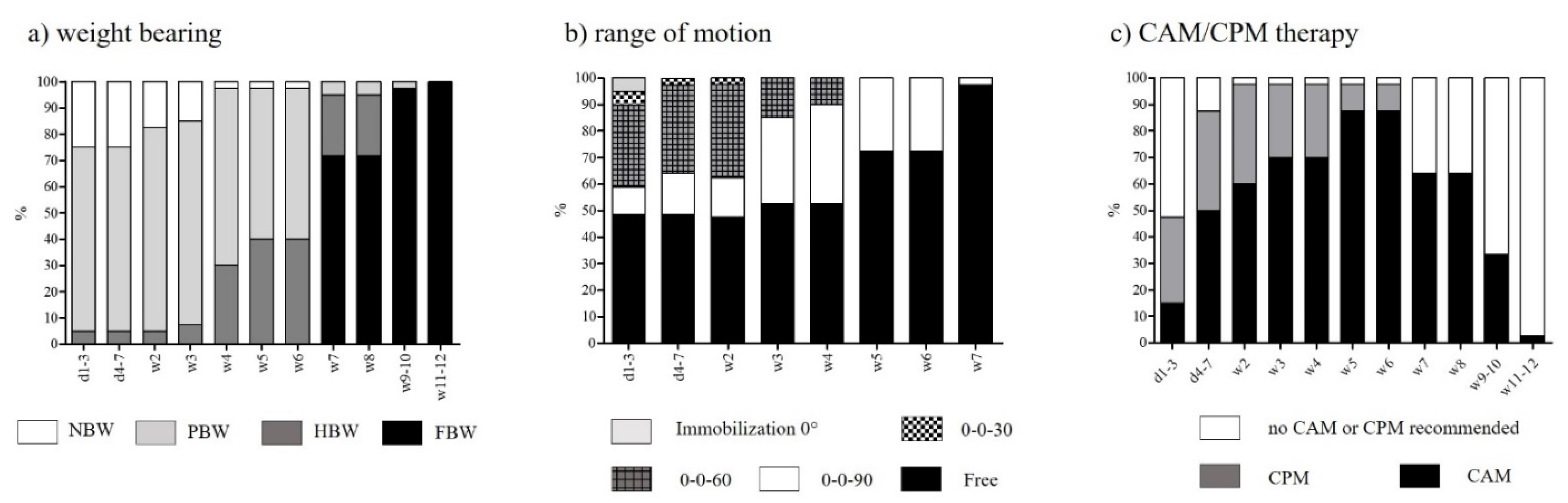
3.3. Rehabilitation after Femorotibial MACT
3.4. Rehabilitation after Femorotibial OATS
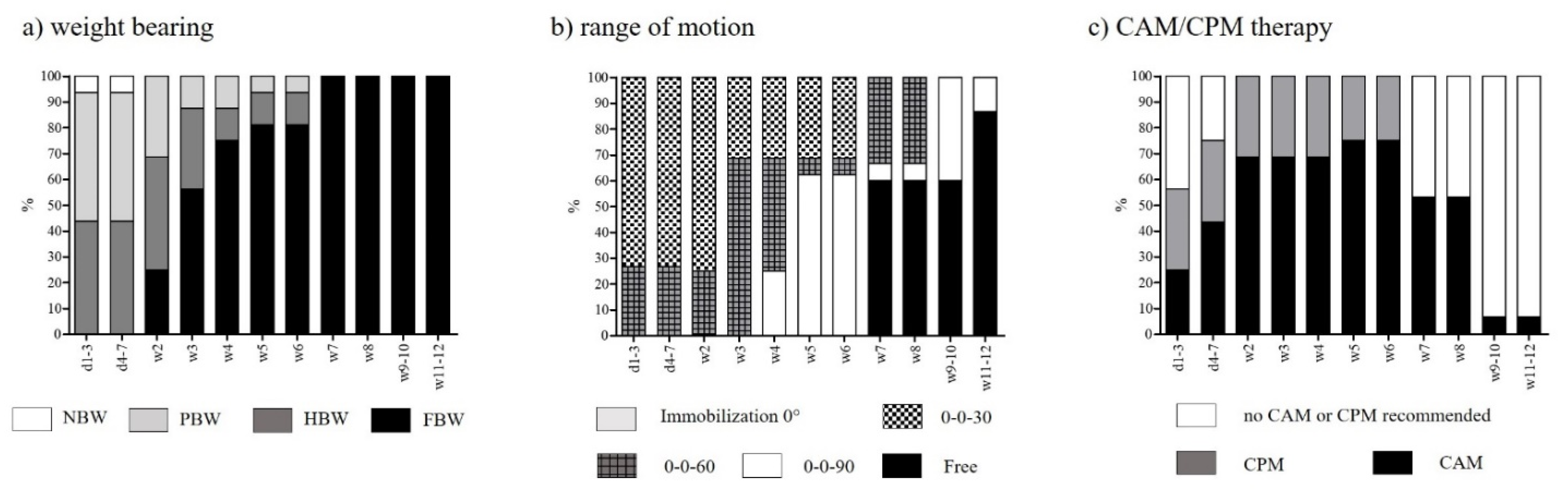
3.5. Rehabilitation after Retropatellar Cartilage Repair
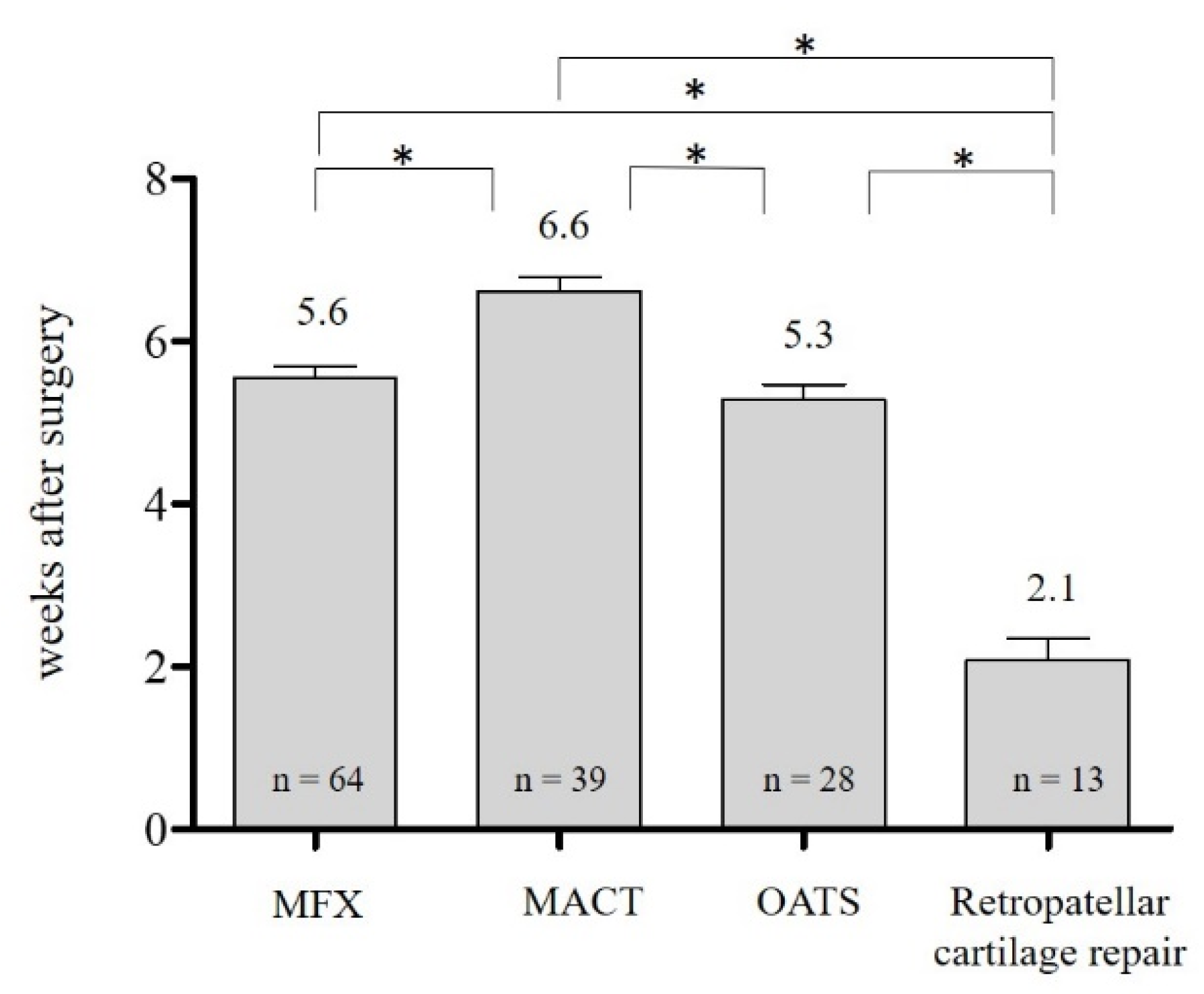
4. Discussion
4.1. Rehabilitation after Femorotibial MFX
4.2. Rehabilitation after Femorotibial MACT
4.3. Rehabilitation after Femorotibial OATS
4.4. Rehabilitation after Retropatellar Cartilage Repair
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Hjelle, K.; Solheim, E.; Strand, T.; Muri, R.; Brittberg, M. Articular cartilage defects in 1000 knee arthroscopies. Arthroscopy 2002, 18, 730–734. [Google Scholar] [CrossRef] [PubMed]
- Widuchowski, W.; Widuchowski, J.; Trzaska, T. Articular cartilage defects: Study of 25,124 knee arthroscopies. Knee 2007, 14, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Heir, S.; Nerhus, T.K.; Rotterud, J.H.; Loken, S.; Ekeland, A.; Engebretsen, L.; Aroen, A. Focal cartilage defects in the knee impair quality of life as much as severe osteoarthritis: A comparison of knee injury and osteoarthritis outcome score in 4 patient categories scheduled for knee surgery. Am. J. Sports Med. 2010, 38, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Niemeyer, P.; Feucht, M.J.; Fritz, J.; Albrecht, D.; Spahn, G.; Angele, P. Cartilage repair surgery for full-thickness defects of the knee in Germany: Indications and epidemiological data from the German Cartilage Registry (KnorpelRegister DGOU). Arch. Orthop. Trauma Surg. 2016, 136, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Zellner, J.; Krutsch, W.; Pfeifer, C.; Koch, M.; Nerlich, M.; Angele, P. Autologous chondrocyte implantation for cartilage repair. Orthop. Res. Rev. 2015, 7, 149–158. [Google Scholar]
- Weber, A.E.; Locker, P.H.; Mayer, E.N.; Cvetanovich, G.L.; Tilton, A.K.; Erickson, B.J.; Yanke, A.B.; Cole, B.J. Clinical Outcomes After Microfracture of the Knee: Midterm Follow-up. Orthop. J. Sports Med. 2018, 6, 1–7. [Google Scholar] [CrossRef]
- Kon, E.; Gobbi, A.; Filardo, G.; Delcogliano, M.; Zaffagnini, S.; Marcacci, M. Arthroscopic second-generation autologous chondrocyte implantation compared with microfracture for chondral lesions of the knee: Prospective nonrandomized study at 5 years. Am. J. Sports Med. 2009, 37, 33–41. [Google Scholar] [CrossRef]
- Na, Y.; Shi, Y.; Liu, W.; Jia, Y.; Kong, L.; Zhang, T.; Han, C.; Ren, Y. Is implantation of autologous chondrocyte superior to microfracture for articular-cartilage defects of the knee? A systematic review of 5-year follow-up data. Int. J. Surg. 2019, 68, 56–62. [Google Scholar] [CrossRef]
- Brittberg, M.; Recker, D.; Ilgenfritz, J.; Saris, D.B.F. Matrix-Applied Characterized Autologous Cultured Chondrocytes Versus Microfracture: Five-Year Follow-up of a Prospective Randomized Trial. Am. J. Sports Med. 2018, 46, 1343–1351. [Google Scholar] [CrossRef]
- Lim, H.-C.; Bae, J.-H.; Song, S.-H.; Park, Y.-E.; Kim, S.-J. Current treatments of isolated articular cartilage lesions of the knee achieve similar outcomes. Clin. Orthop. Relat. Res. 2012, 470, 2261–2267. [Google Scholar] [CrossRef]
- Assenmacher, A.T.; Pareek, A.; Reardon, P.J.; Macalena, J.A.; Stuart, M.J.; Krych, A.J. Long-term Outcomes After Osteochondral Allograft: A Systematic Review at Long-term Follow-up of 12.3 Years. Arthroscopy 2016, 32, 2160–2168. [Google Scholar] [CrossRef]
- Martin, J.A.; Brown, T.; Heiner, A.; Buckwalter, J.A. Post-traumatic osteoarthritis: The role of accelerated chondrocyte senescence. Biorheology 2004, 41, 479–491. [Google Scholar]
- Martin, J.A.; Buckwalter, J.A. Post-traumatic osteoarthritis: The role of stress induced chondrocyte damage. Biorheology 2006, 43, 517–521. [Google Scholar] [PubMed]
- Hambly, K.; Bobic, V.; Wondrasch, B.; van Assche, D.; Marlovits, S. Autologous chondrocyte implantation postoperative care and rehabilitation: Science and practice. Am. J. Sports Med. 2006, 34, 1020–1038. [Google Scholar] [CrossRef]
- Knapik, D.M.; Harris, J.D.; Pangrazzi, G.; Griesser, M.J.; Siston, R.A.; Agarwal, S.; Flanigan, D.C. The basic science of continuous passive motion in promoting knee health: A systematic review of studies in a rabbit model. Arthroscopy 2013, 29, 1722–1731. [Google Scholar] [CrossRef] [PubMed]
- Hinterwimmer, S.; Krammer, M.; Krotz, M.; Glaser, C.; Baumgart, R.; Reiser, M.; Eckstein, F. Cartilage atrophy in the knees of patients after seven weeks of partial load bearing. Arthritis Rheum. 2004, 50, 2516–2520. [Google Scholar] [CrossRef] [PubMed]
- Steadman, J.R.; Rodkey, W.G.; Rodrigo, J.J. Microfracture: Surgical technique and rehabilitation to treat chondral defects. Clin. Orthop. Relat. Res. 2001, 391, 362–369. [Google Scholar] [CrossRef]
- Braun, S.; Steadman, J.R.; Rodkey, W.G.; Briggs, K.K. Microfracture and specific rehabilitation for treating osteoarthritis of the knee. Indications, surgical technique, and rehabilitation protocol. Z. Rheumatol. 2009, 68, 811–818. [Google Scholar]
- Tyler, T.F.; Lung, J.Y. Rehabilitation following osteochondral injury to the knee. Curr Rev Musculoskelet Med 2012, 5, 72–81. [Google Scholar] [CrossRef]
- Erggelet, C. Stimulation Thechniques: Microfracturing, Drilling. In Techniques in Cartilage Repair Surgery; Springer: Berlin/Heidelberg, Germany, 2014; pp. 35–47. [Google Scholar]
- Mithoefer, K.; Williams, R.J., III; Warren, R.F.; Potter, H.G.; Spock, C.R.; Jones, E.C.; Wickiewicz, T.L.; Marx, R.G. The microfracture technique for the treatment of articular cartilage lesions in the knee. A prospective cohort study. J. Bone Jt. Surg. Am. 2005, 87, 1911–1920. [Google Scholar] [CrossRef]
- Salter, R.B.; Simmonds, D.F.; Malcolm, B.W.; Rumble, E.J.; MacMichael, D.; Clements, N.D. The biological effect of continuous passive motion on the healing of full-thickness defects in articular cartilage. An experimental investigation in the rabbit. J. Bone Jt. Surg. Am. 1980, 62, 1232–1251. [Google Scholar] [CrossRef]
- Fazalare, J.A.; Griesser, M.J.; Siston, R.A.; Flanigan, D.C. The use of continuous passive motion following knee cartilage defect surgery: A systematic review. Orthopedics 2010, 33, 878. [Google Scholar] [CrossRef]
- Howard, J.S.; Mattacola, C.G.; Romine, S.E.; Lattermann, C. Continuous Passive Motion, Early Weight Bearing, and Active Motion following Knee Articular Cartilage Repair; Evidence for Clinical Practice. Cartilage 2010, 1, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Rogan, S.; Taeymans, J.; Hirschmuller, A.; Niemeyer, P.; Baur, H. Effect of continuous passive motion for cartilage regenerative surgery—A systematic literature review. Z. Orthop. Unfall. 2013, 151, 468–474. [Google Scholar] [PubMed]
- Hirschmuller, A.; Baur, H.; Braun, S.; Kreuz, P.C.; Sudkamp, N.P.; Niemeyer, P. Rehabilitation after autologous chondrocyte implantation for isolated cartilage defects of the knee. Am. J. Sports Med. 2011, 39, 2686–2696. [Google Scholar] [CrossRef]
- Gillogly, S.D.; Myers, T.H.; Reinold, M.M. Treatment of full-thickness chondral defects in the knee with autologous chondrocyte implantation. J. Orthop. Sports Phys. Ther. 2006, 36, 751–764. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.B. Classical ACI for Chondral and Ostechondral Defects. In Techniques in Cartilage Repair Surgery; Springer: Berlin/Heidelberg, Germany, 2014; pp. 165–174. [Google Scholar]
- Pestka, J.M.; Feucht, M.J.; Porichis, S.; Bode, G.; Sudkamp, N.P.; Niemeyer, P. Return to Sports Activity and Work after Autologous Chondrocyte Implantation of the Knee: Which Factors Influence Outcomes? Am. J. Sports Med. 2016, 44, 370–377. [Google Scholar] [CrossRef]
- Riegger-Krugh, C.L.; McCarthy, E.C.; Robinson, M.S.; Wegzyn, D.A. Autologous chondrocyte implantation: Current surgery and rehabilitation. Med. Sci. Sports Exerc. 2008, 40, 206–214. [Google Scholar] [CrossRef]
- Campbell, A.B.; Pineda, M.; Harris, J.D.; Flanigan, D.C. Return to Sport After Articular Cartilage Repair in Athletes’ Knees: A Systematic Review. Arthroscopy 2016, 32, 651–668.e1. [Google Scholar] [CrossRef]
| Rehabilitation Criterion | Categories |
|---|---|
| Weight-bearing | No body weight (NBW): no loading Partial body weight (PBW): ≤20 kg Half body weight (HBW): >20 kg Full body weight (FBW): no restriction |
| Range of motion (extension/flexion) | Immobilization 0° 0-0-30 0-0-60 0-0-90 0-0-free |
| Use of braces | Yes/No Recommended wearing time (weeks) |
| Continuous active/ passive motion (CAM/CPM) | CAM recommended CPM recommended No CAM/CPM recommended |
| Start of rehabilitation training | Weeks after surgery |
| Start of specific training | Weeks after surgery |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Memmel, C.; Krutsch, W.; Koch, M.; Riedl, M.; Henssler, L.; Zeman, F.; Knorr, C.; Alt, V.; Pfeifer, C. The Conformity of Rehabilitation Protocols Used for Different Cartilage Repairs of the Knee Joint—A Review on Rehabilitation Standards in German Speaking Countries. Appl. Sci. 2021, 11, 8873. https://doi.org/10.3390/app11198873
Memmel C, Krutsch W, Koch M, Riedl M, Henssler L, Zeman F, Knorr C, Alt V, Pfeifer C. The Conformity of Rehabilitation Protocols Used for Different Cartilage Repairs of the Knee Joint—A Review on Rehabilitation Standards in German Speaking Countries. Applied Sciences. 2021; 11(19):8873. https://doi.org/10.3390/app11198873
Chicago/Turabian StyleMemmel, Clemens, Werner Krutsch, Matthias Koch, Moritz Riedl, Leopold Henssler, Florian Zeman, Christian Knorr, Volker Alt, and Christian Pfeifer. 2021. "The Conformity of Rehabilitation Protocols Used for Different Cartilage Repairs of the Knee Joint—A Review on Rehabilitation Standards in German Speaking Countries" Applied Sciences 11, no. 19: 8873. https://doi.org/10.3390/app11198873
APA StyleMemmel, C., Krutsch, W., Koch, M., Riedl, M., Henssler, L., Zeman, F., Knorr, C., Alt, V., & Pfeifer, C. (2021). The Conformity of Rehabilitation Protocols Used for Different Cartilage Repairs of the Knee Joint—A Review on Rehabilitation Standards in German Speaking Countries. Applied Sciences, 11(19), 8873. https://doi.org/10.3390/app11198873







