Atlas of Micromorphological Degradation of Archaeological Birch Bark
Abstract
:1. Introduction
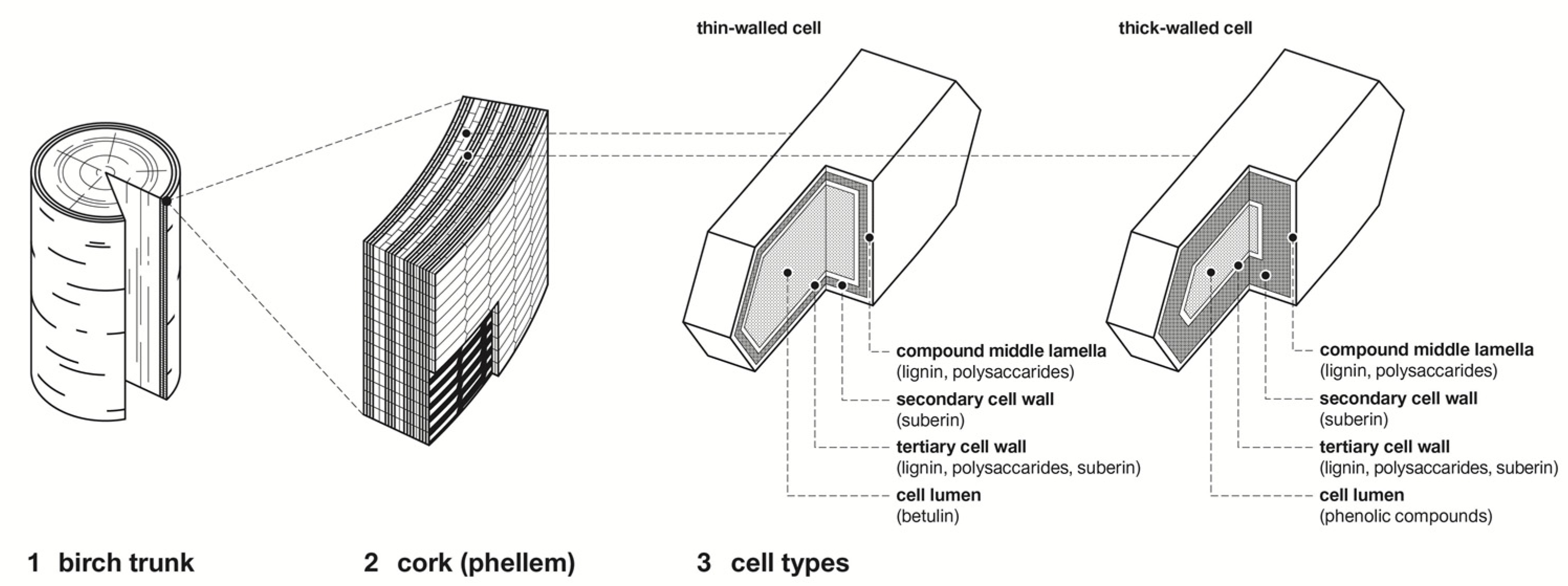
1.1. Morphology of Birch Bark
1.2. Chemical Composition of Birch Bark
1.3. Degradation and Preservation Condition of Archaeological Birch Bark
2. Materials and Methods
2.1. Samples
2.2. Description of Embedding and Sectioning
2.3. LM and TEM
3. Results
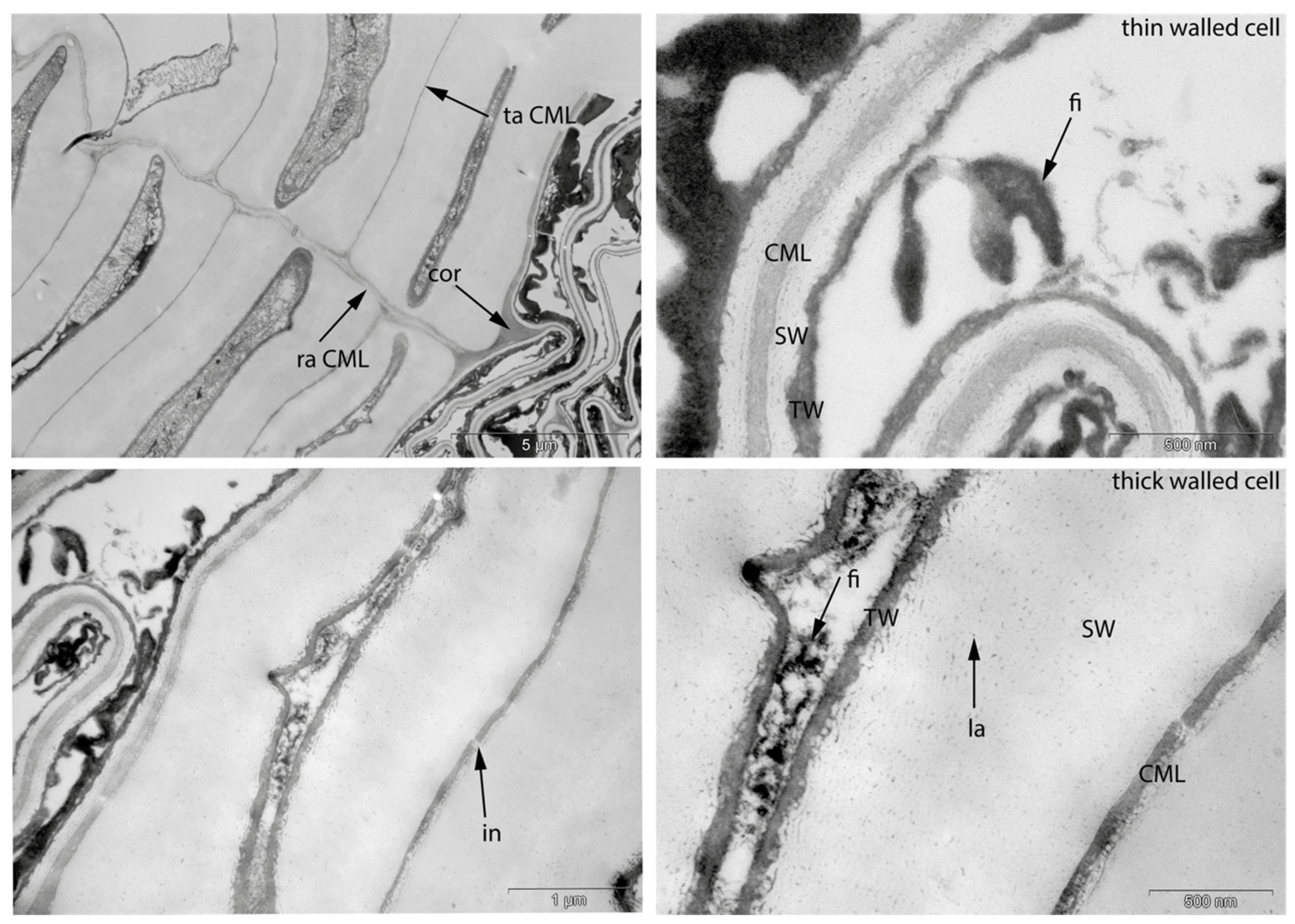
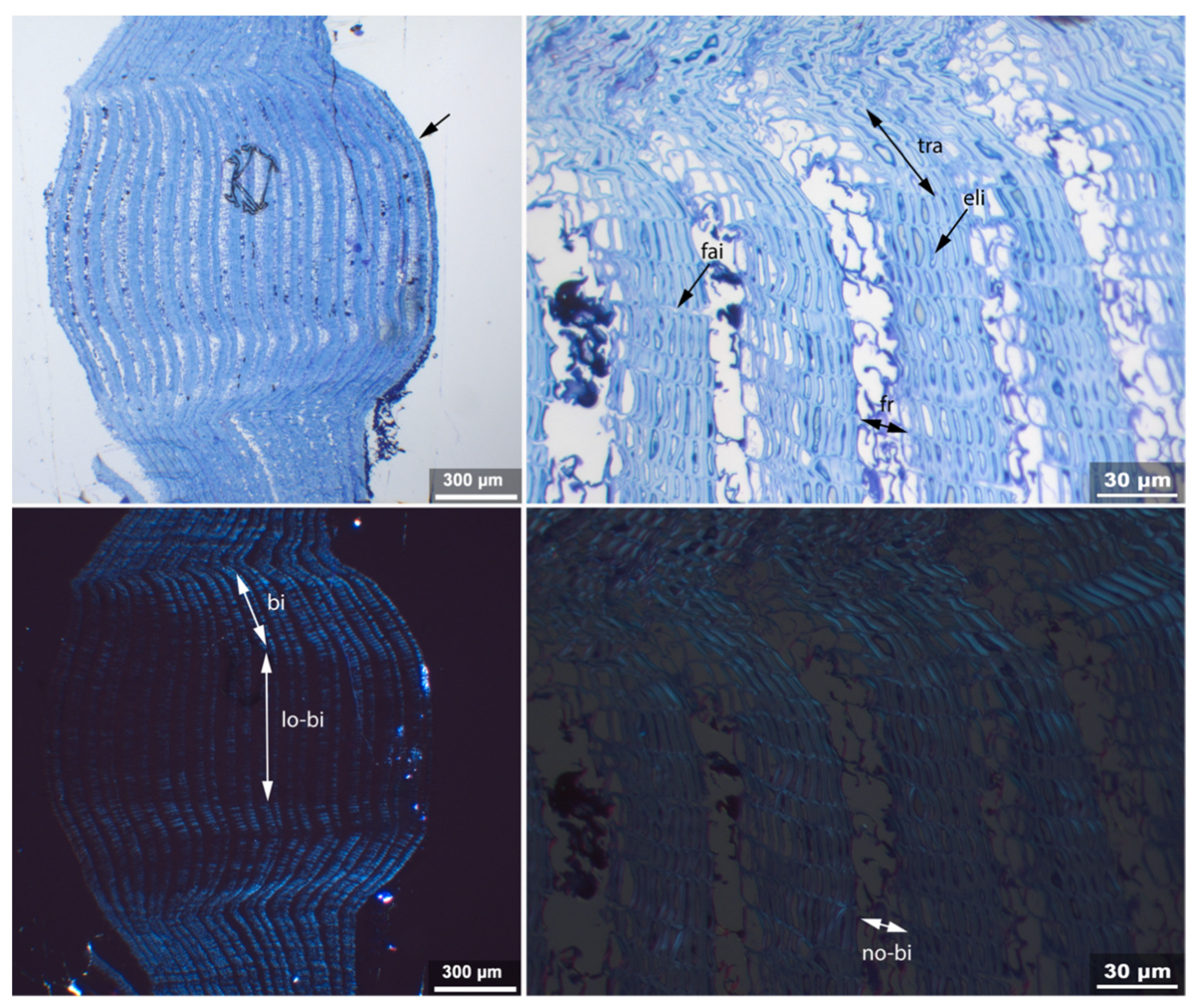
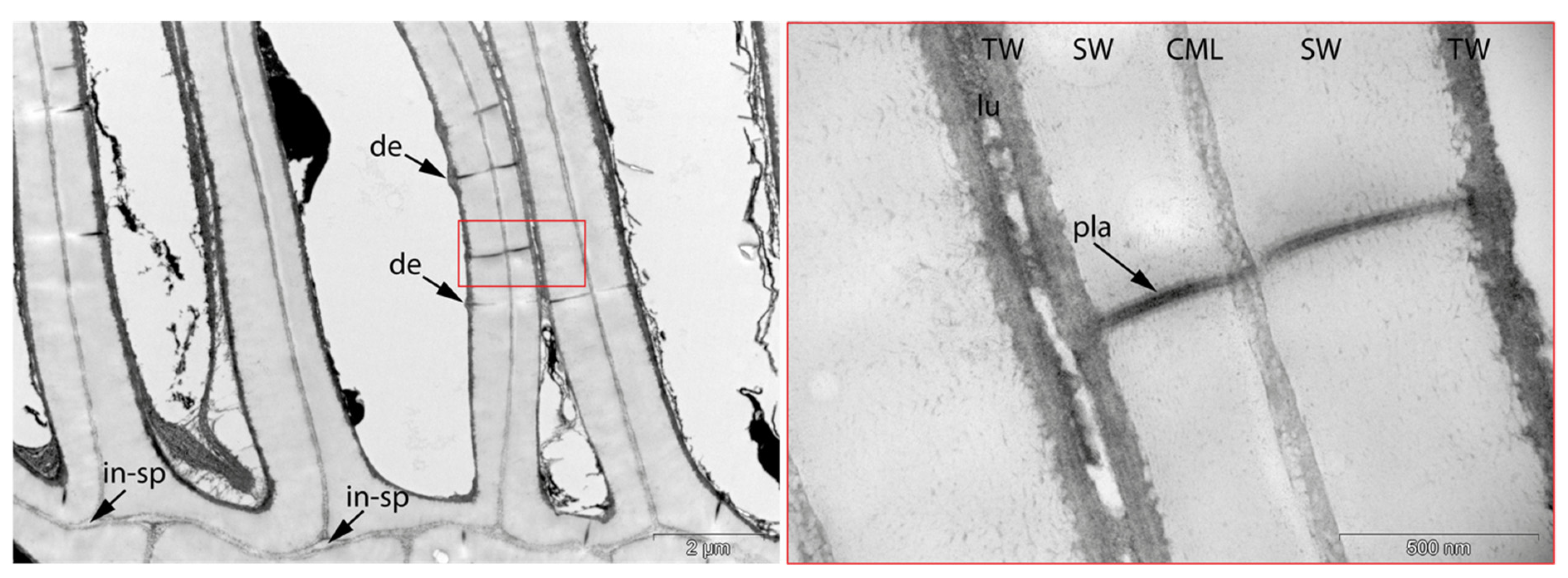
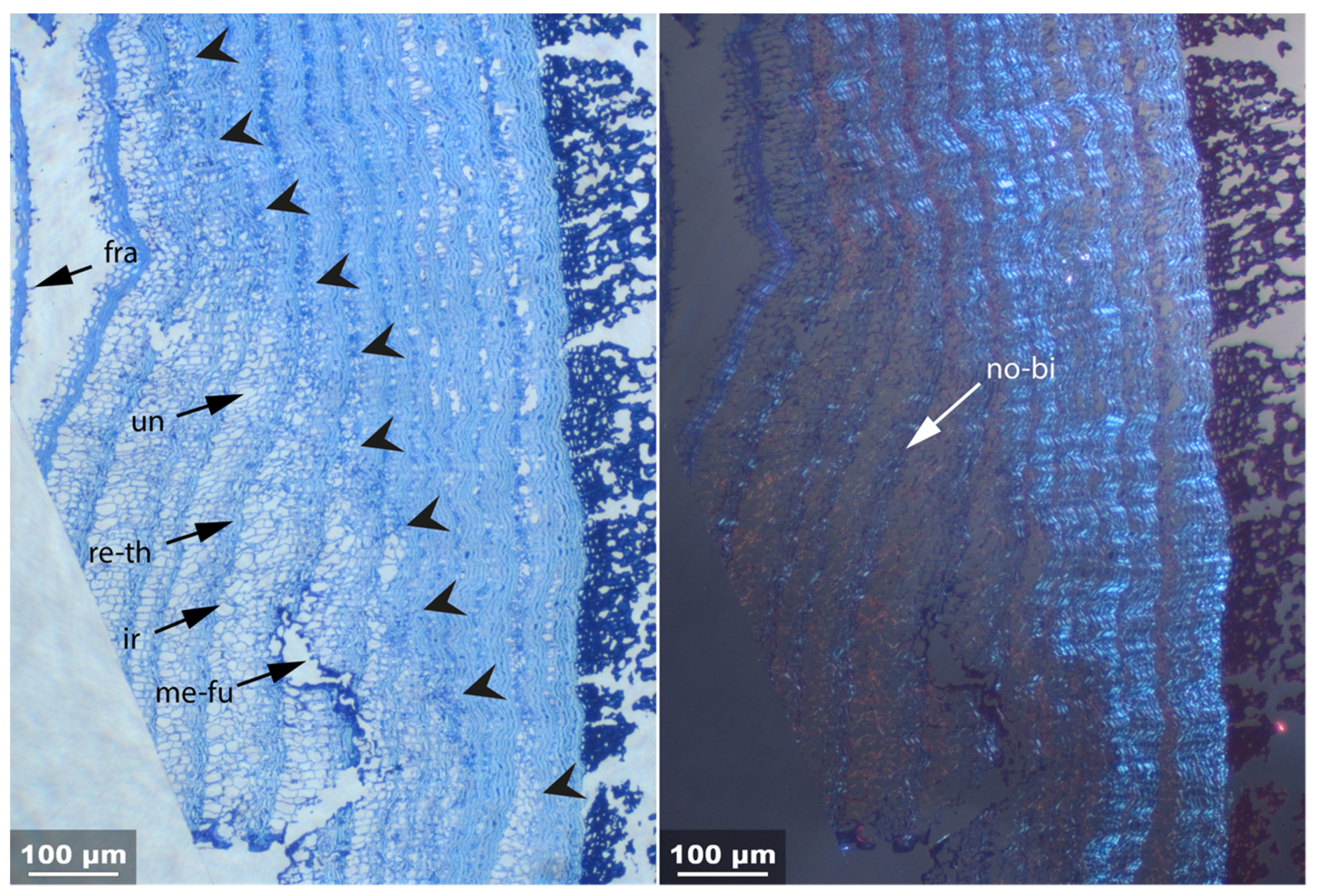
- Fra: Fracture of radial cell walls;
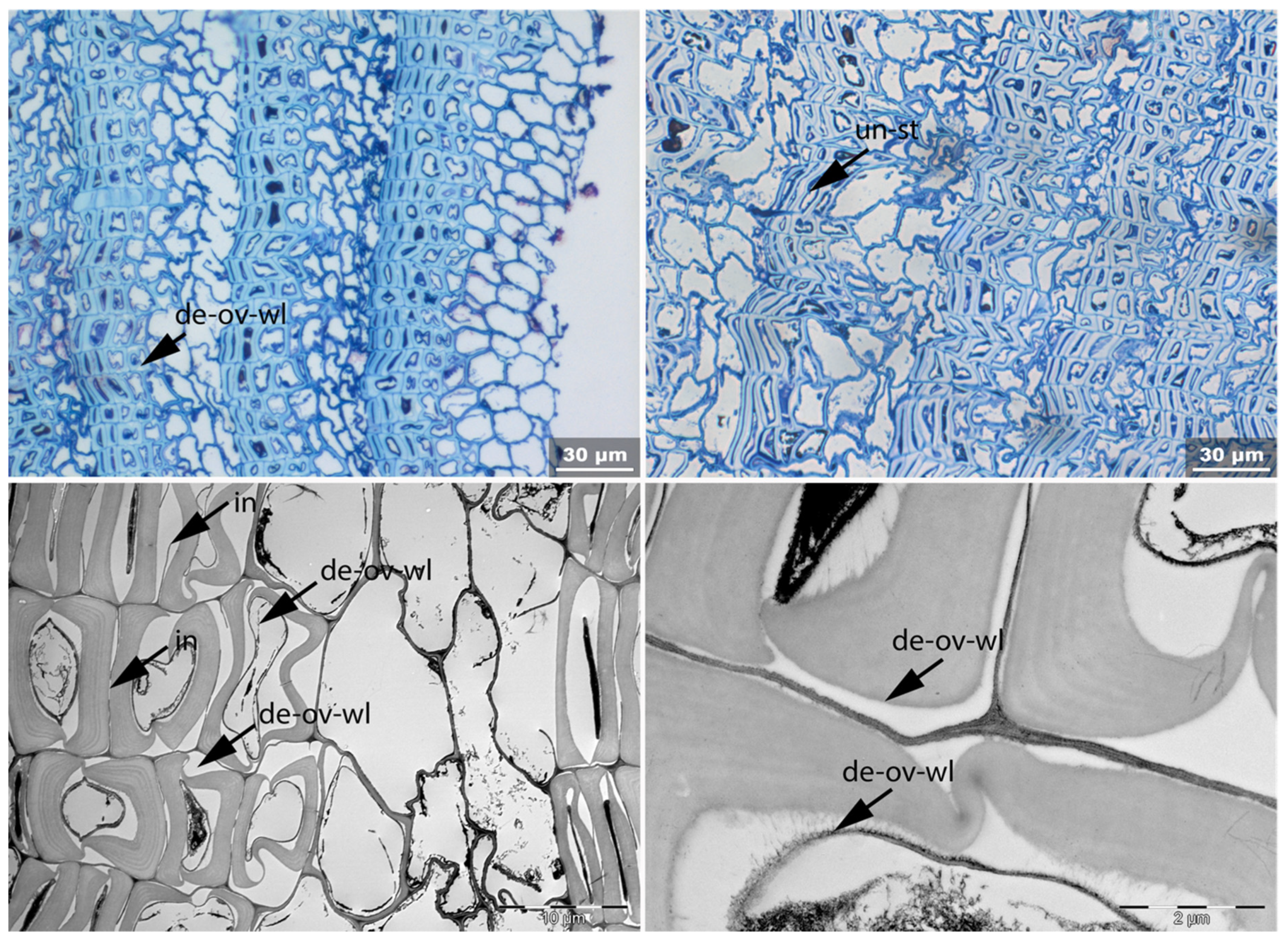
- Un: Unfolding of compressed thin-walled cell; if it is in conjunction with stretching of the thick-walled cells, it is marked as Un-st;
- No-bi: Absence of birefringence of secondary wall of thick-walled cell;
- In-st: Intense toluidine blue staining;
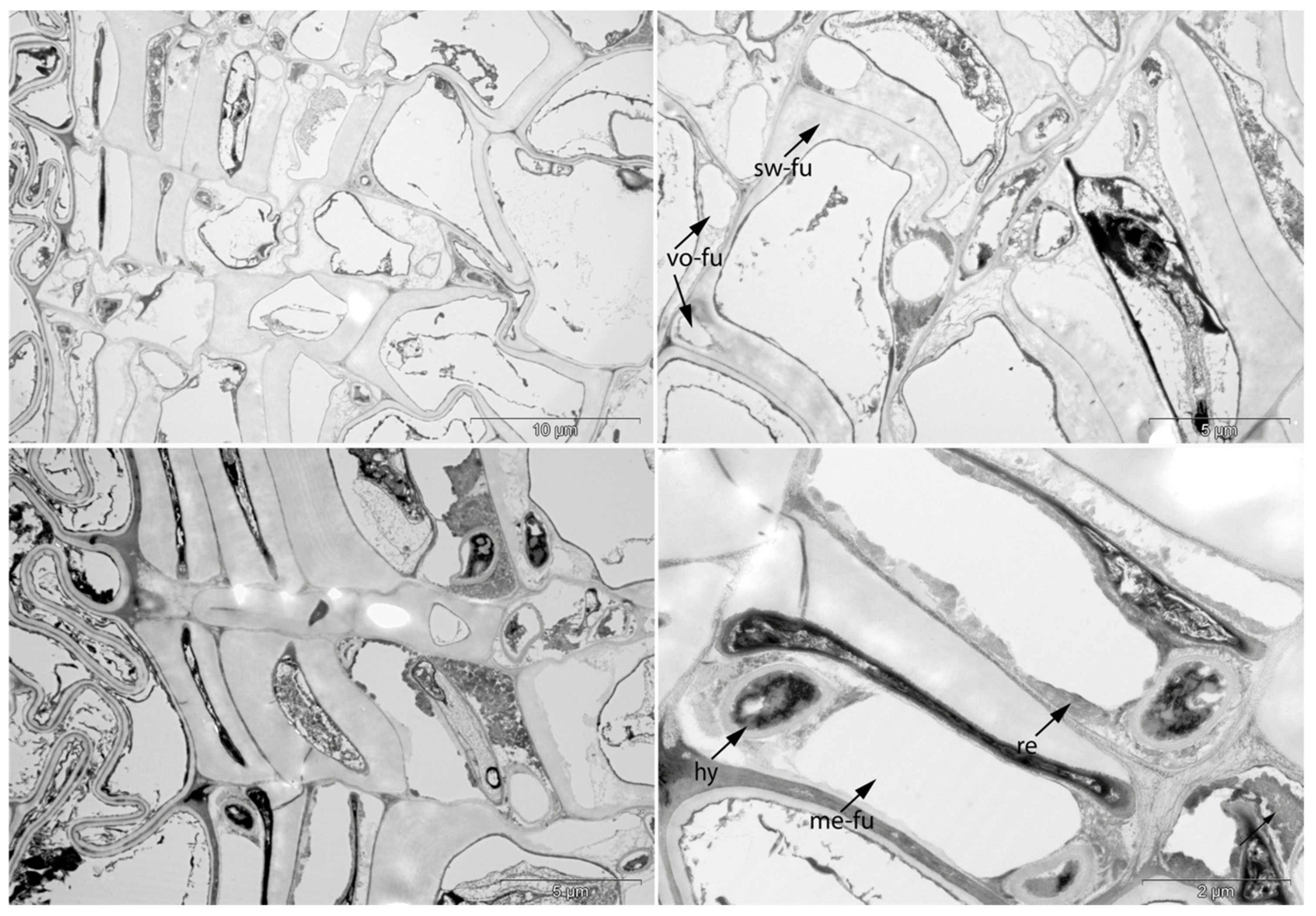
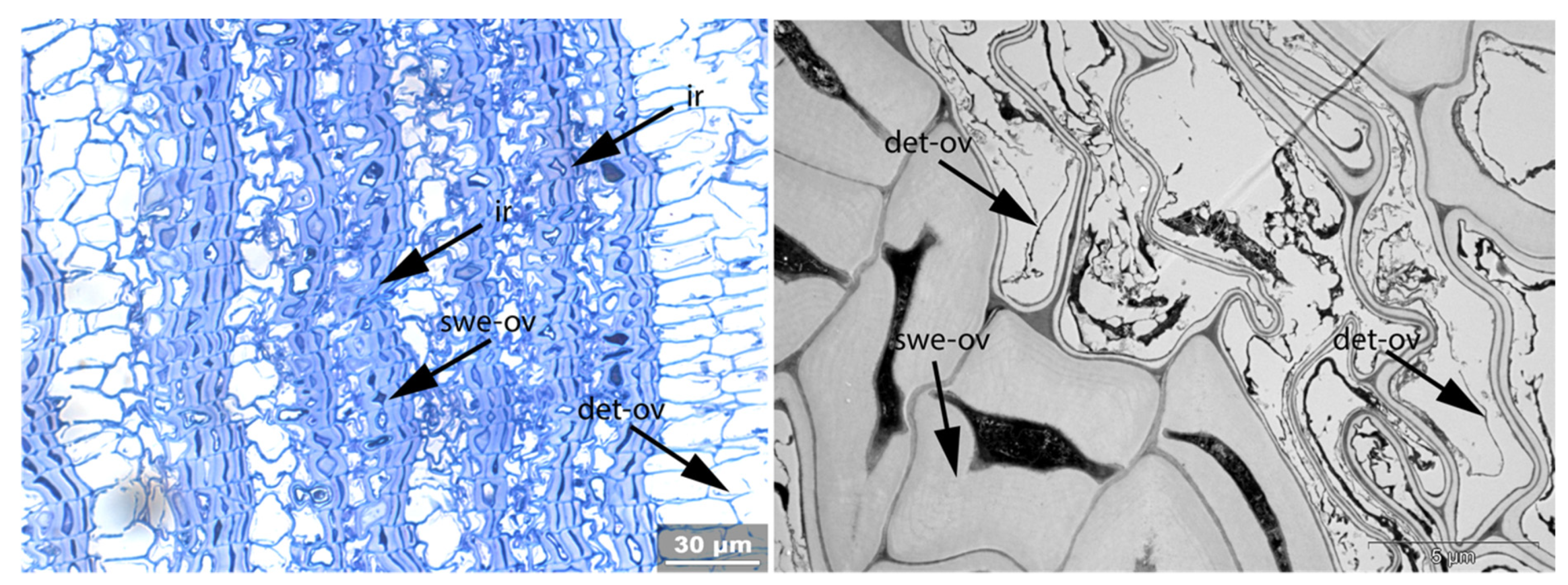
- Swe: Swelling and/or disaggregation of the secondary cell walls;
- Ir: Increased irregularity of cell shape and size leading to irregular cell arrangement;
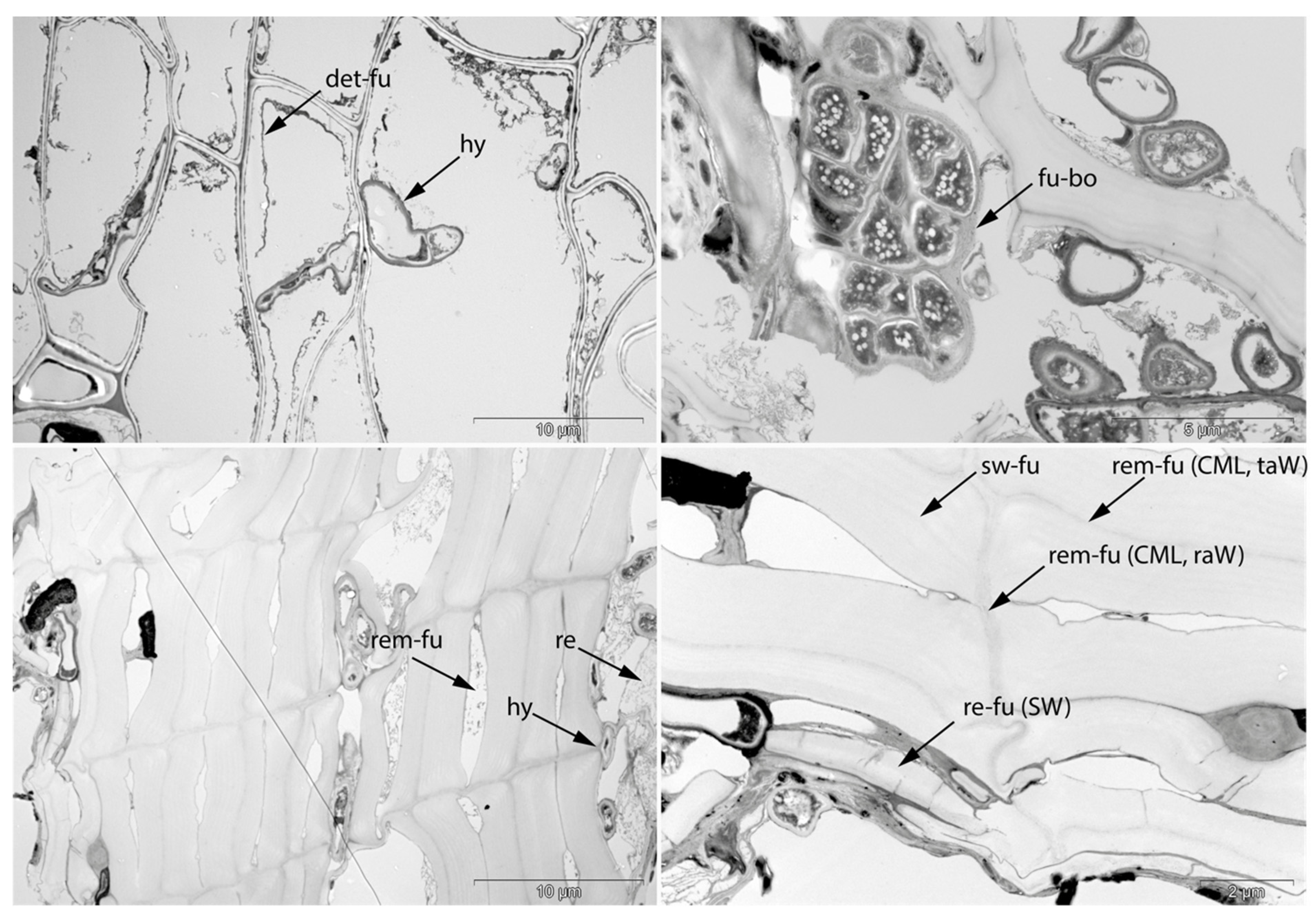
- Det: Detachment, either of cells from each other (Det-cl) or secondary cell walls from the compound middle lamella (Det-wl) or the tertiary wall;
- Rem: Removal of phenolic compounds in the lumen of thick-walled cells or the compound middle lamella or in the secondary cell walls;
- Re-th: Reduced cell wall thickness;
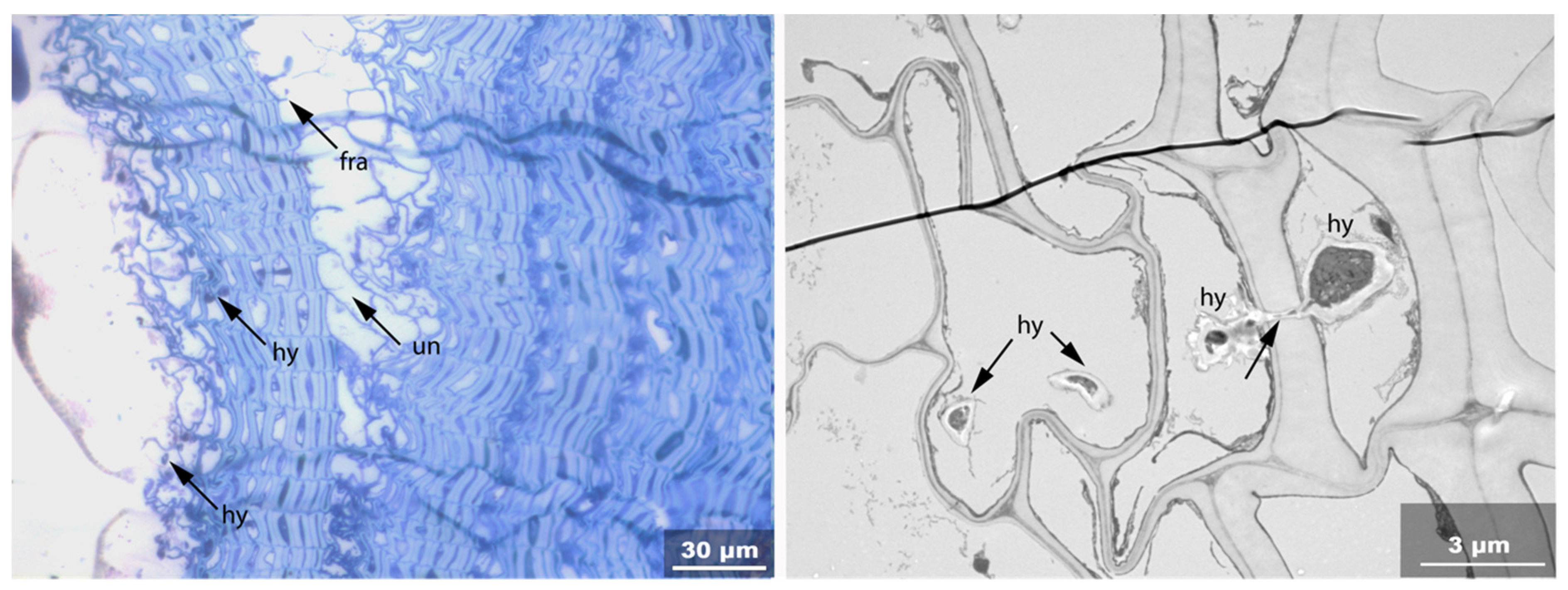
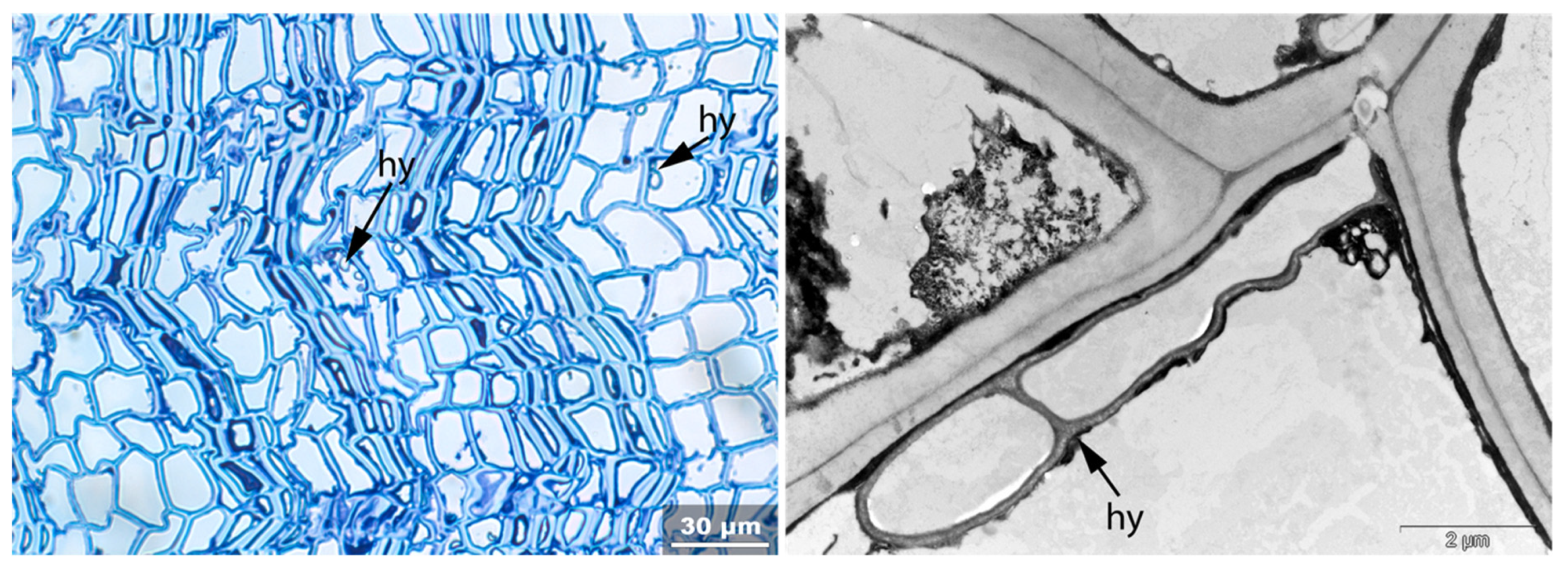
- Hy: Hyphea in thin-walled cell lumen;
- Vo: Circular/oval voids in the corners of secondary thick-walled cell walls with a diameter of about 0.6 µm, or smaller voids in the secondary cell walls with diameter of about 1.2 µm aligned along or in the compound middle lamella; the former are caused by mechanical penetration in the wall by hyphae, the latter by the penetration of bacteria;
- Me: Complete dissolution of cell structure;
- Acc: Accumulation of foreign substance in the cell lumen of thin-walled cells.
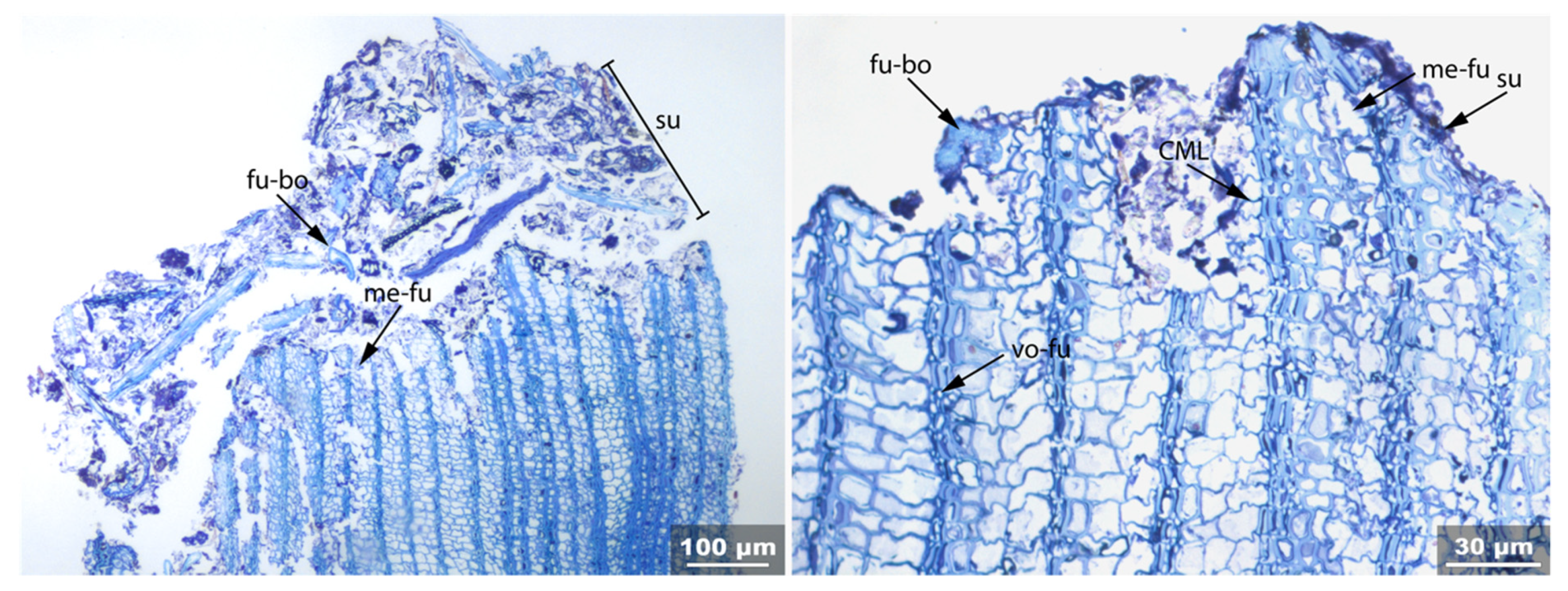
3.1. Reference Material: Contemporary Intact Birch Bark
3.2. Sample 0: Contemporary Birch Bark Degraded in Nature
3.3. Archaeological Samples
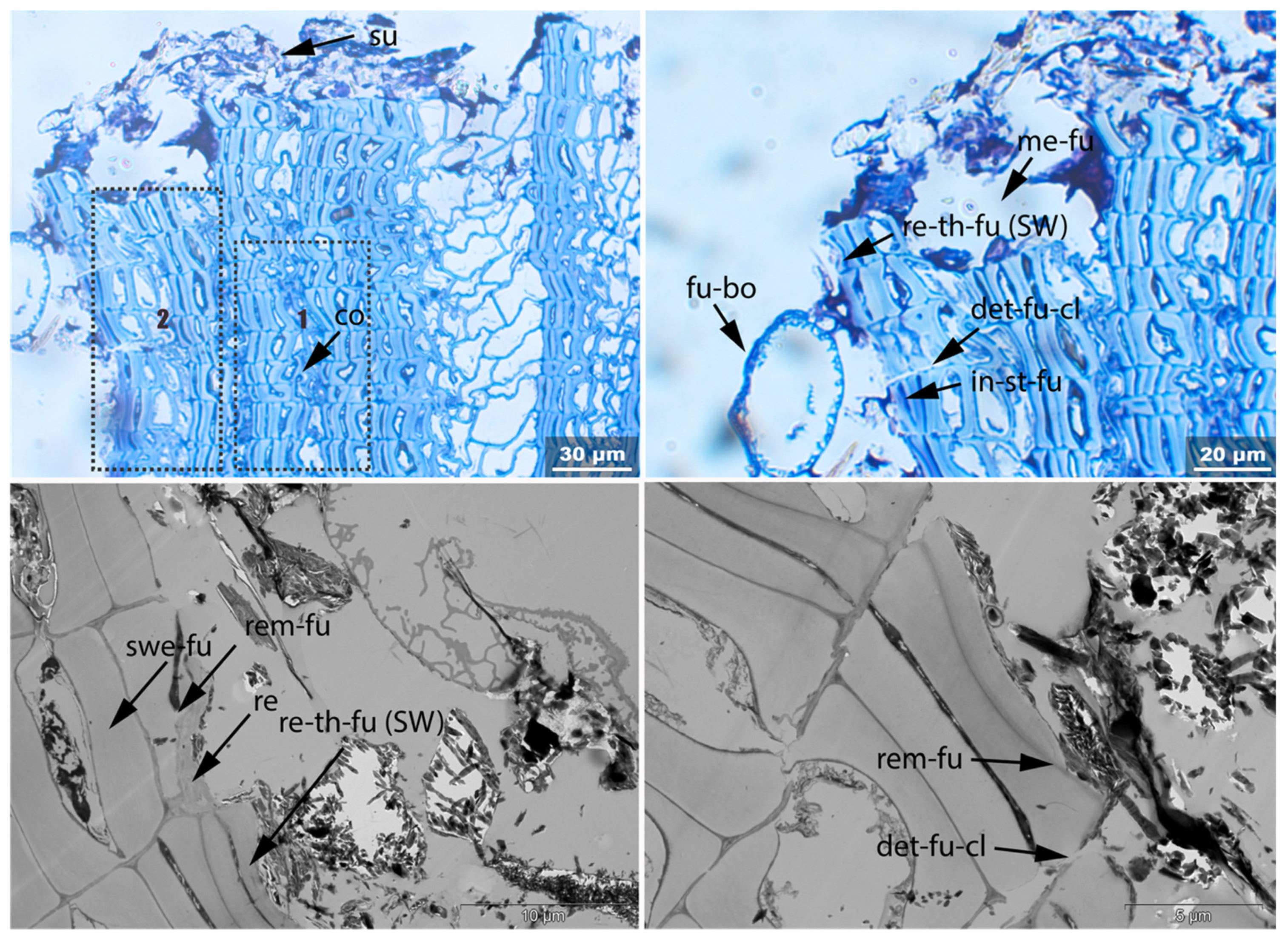
3.3.1. Sample 1
3.3.2. Sample 2
3.3.3. Sample 3
3.3.4. Sample 4
3.3.5. Sample 6
3.3.6. Sample 10
3.3.7. Sample 11
3.3.8. Sample 12
3.3.9. Sample 13
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lüttge, U.; Kluge, M.; Bauer, G. Botanik; Wiley Verlag: Weinheim, Germany, 2005. [Google Scholar]
- Kost, B. Die Gewebe der Gefäßpflanzen. In Strasburger–Lehrbuch der Pflanzenwissenschaften; Kadereit, J.W., Körner, C., Kost, B., Sonnewald, U., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 72–96. [Google Scholar]
- Chang, Y.-P. Anatomy of Common North American Pulpwood Barks; Technical Association of the Pulp and Paper Industry: Tokyo, Japan, 1954. [Google Scholar]
- Jensen, W. The Connection between the Anatomical Structure and Chemical Composition and the Properties of Outer Bark of White Birch. Pappers-och trävarutidskrift för Finl. Suom. Pap.-Ja Puutavaralehti 1949, 15, 113–119. [Google Scholar]
- Jensen, W.A. Botanical Histochemistry: Principles and Practice; Freeman, W.H.: San Francisco, CA, USA, 1962; Volume 12. [Google Scholar]
- Evert, R.F.; Esau, K.; Langenfeld-Heyser, R.; Eichhorn, S.E. Esaus Pflanzenanatomie: Meristeme, Zellen und Gewebe der Pflanzen- ihre Struktur, Funktion und Entwicklung; Walter de Gruyter Verlag: Göttingen, Germany, 2009. [Google Scholar]
- Klügl, J.; Hafner, A.; Di Pietro, G. On the rolling and plasticization of birch bark (in print). In Proceedings of the 14th ICOM-CC Wet Organic Archaeological Materials (WOAM) Working Group Conference, Portsmouth, UK, 20–24 May 2019. [Google Scholar]
- Fortes, M.A.; Rosa, M.E. Growth stresses and strains in cork. Wood Sci. Technol. 1992, 26, 241–258. [Google Scholar] [CrossRef]
- Krahmer, R.L.; Wellons, J.D. Some Anatomical and Chemnical Characteristics of Douglas-Fir Cork. Wood Sci. 1973, 6, 97–105. [Google Scholar]
- Xu, X.; Schneider, E.; Zaremba, C.; Stucky, G.D.; Wudl, F. Modification of the Semitransparent Prunus serrula Bark Film: Making Rubber out of Bark. Chem. Mater. 1998, 10, 3523–3537. [Google Scholar] [CrossRef]
- Groh, B.; Hübner, C.; Lendzian, K. Water and oxygen permeance of phellems isolated from trees: The role of waxes and lenticels. Planta 2002, 215, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Schönherr, J.; Ziegler, H. Water permeability of Betula periderm. Planta 1980, 147, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Shibui, H.; Sano, Y. Structure and formation of phellem of Betula maximowicziana. Int. Assoc. Wood Anat. 2018, 39, 18–36. [Google Scholar] [CrossRef]
- Zajączkowska, U. Cork. eLS 2016, 1–8. [Google Scholar]
- Mauseth, J.D. Botany: An Introduction to Plant Biology; Jones & Bartlett Learning: Burlington, MA, USA, 2019. [Google Scholar]
- Pinto, P.; Sousa, A.; Silvestre, A.; Neto, C.P.; Gandini, A.; Eckerman, C.; Holmbom, B. Quercus suber and Betula pendula outer barks as renewable sources of oleochemicals: A comparative study. Ind. Crop. Prod. 2009, 29, 126–132. [Google Scholar] [CrossRef] [Green Version]
- Ekman, R. The Suberin Monomers and Triterpenoids from the Outer Bark of Betula-verrucosa Ehrh. Holzforschung 1983, 37, 205–211. [Google Scholar] [CrossRef]
- Jensen, W.; Fremer, K.E.; Sierilä, P.; Wartiovaara, V. The Chemistry of Bark. In The Chemistry of Wood; Browning, B.L., Ed.; Interscience Publishers: New York, NY, USA; London, UK, 1963. [Google Scholar]
- Ferreira, J.P.A.; Quilhó, T.; Pereira, H. Characterization of Betula pendula Outer Bark Regarding Cork and Phloem Components at Chemical and Structural Levels in View of Biorefinery Integration. J. Wood Chem. Technol. 2017, 37, 10–25. [Google Scholar] [CrossRef]
- Koljo, B.; Sitte, P. Das Suberin. In Die Chemie der Pflanzenzellwand: Ein Beitrag zur Morphologie, Physik, Chemie und Technologie der Cellulose und ihrer Begleiter; Treiber, E.E., Ed.; Springer: Berlin/Heidelberg, Germany, 1957; pp. 416–432. [Google Scholar]
- Kiyoto, S.; Sugiyama, J. Histochemical structure and tensile properties of birch cork cell walls. Cellulose 2021. [Google Scholar] [CrossRef]
- Graça, J.; Santos, S. Suberin: A Biopolyester of Plants’ Skin. Macromol. Biosci. 2007, 7, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Tse, S.; Dignard, C.; Kata, S.; Henderson, E.J. A study of the light sensitivity of birch bark. Stud. Conserv. 2018, 63, 423–440. [Google Scholar] [CrossRef]
- Monahan, V. Condition of Artifacts from Ice Patches; 2008. [Google Scholar]
- Klügl, J.; Hafner, A.; Di Pietro, G. Towards a description of the degradation of archaeological birch bark. In Proceedings of the ICOM-CC 18th Triennial Conference Preprints, Copenhagen, Denmark, 4–8 September 2017. [Google Scholar]
- Orsini, S.; Ribechini, E.; Modugno, F.; Klügl, J.; Di Pietro, G.; Colombini, M.P. Micromorphological and chemical elucidation of the degradation mechanisms of birch bark archaeological artefacts. Herit. Sci. 2015, 3, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Vasiljeva, N. Softening, Drying and Consolidation of Birch Bark; 2018. [Google Scholar]
- Klügl, J.; Di Pietro, G. The interaction of water with archaeological and ethnographic birch bark and its effects on swelling, shrinkage and deformations. Herit. Sci. 2021, 9, 3. [Google Scholar] [CrossRef]
- Fletcher, L.; Milner, N.; Taylor, M.; Bamforth, M.; Croft, S.; Little, A.; Pomstra, D.; Robson, H.K.; Knight, B. The Use of Birch Bark. In Star Carr Volume 2: Studies in Technology, Subsistence and Environment; Milner, N., Taylor, B., Conneller, C., Eds.; White Rose University Press: York, UK, 2018; pp. 419–534. [Google Scholar]
- Hägele-Masnick, J. The birch bark torches from the Kyffhäuser caves. In Proceedings of the 13th ICOM-CC Group on Wet Organic Archaeological Materials Conference, Florence, Italy; 2016; pp. 392–394. [Google Scholar]
- Larsen, P.K.; Jensen, L.A.; Ryhl-Svendsen, M.; Padfield, T. The microclimate within a Neolithic passage grave. In Proceedings of the ICOM-CC 18th Triennial Conference, Copenhagen, Denmark, 4–8 September 2017; pp. 109–127. [Google Scholar]
- Ward, C.; Giles, D.; Sully, D.; Lee, D.J. The conservation of a group of waterlogged neolithic bark bowls. Stud. Conserv. 1996, 41, 241–249. [Google Scholar]
- Rao, H.; Yang, Y.; Hu, X.; Yu, J.; Jiang, H. Identification of an Ancient Birch Bark Quiver from a Tang Dynasty (A.D. 618–907) Tomb in Xinjiang, Northwest China. Econ. Bot. 2017, 71, 32–44. [Google Scholar] [CrossRef]
- Tintner, J.; Smidt, E.; Aumüller, C.; Martin, P.; Ottner, F.; Wriessnig, K.; Reschreiter, H. Taphonomy of prehistoric bark in a salt environment at the archaeological site in Hallstatt, Upper Austria–An analytical approach based on FTIR spectroscopy. Vib. Spectrosc. 2018, 97. [Google Scholar] [CrossRef]
- Kolattukudy, P.E. Suberin from Plants. In Biopolymers; Steinbüchel, A., Yosiharu, D., Eds.; Wiley: Weinheim, Germany, 2002; Volume 3a, pp. 41–73. [Google Scholar]
- Agrawal, O.P.; Dhawan, S. Studies on fungal resistance of birch-bark. In Proceedings of the ICOM-CC 7th Triennial Meeting, Copenhagen, Denmark, 10–14 September 1984; pp. 1–3. [Google Scholar]
- Ranathunge, K.; Schreiber, L.; Franke, R. Suberin research in the genomics era—New interest for an old polymer. Plant Sci. 2011, 180, 399–413. [Google Scholar] [CrossRef]
- Parameswaran, N.; Wilhelm, G.E. Micromorphology of naturally degraded beech and spruce barks. Eur. J. For. Pathol. 1979, 9, 103–112. [Google Scholar] [CrossRef]
- Rypacek, W. Biologie Holzzerstörender Pilze; VEB Gustav Fischer Verlag: Jena, Germany, 1966. [Google Scholar]
- Zimmermann, W.; Seemüller, E. Degradation of Raspberry Suberin by Fusarium solani f. sp. Pisi and Armillaria mellea. J. Phytopathol. 1984, 110, 192–199. [Google Scholar] [CrossRef]
- Ofong, A.U.; Pearce, R.B. Suberin degradation by Rosellinia desmazieresii. Eur. J. For. Pathol. 1994, 24, 316–322. [Google Scholar] [CrossRef]
- Fernando, G.; Zimmermann, W.; Kolattukudy, P.E. Suberin-grown Fusarium solani f. sp pisi generates a cutinase-like esterase which depolymerizes the aliphatic components of suberin. Physiol. Plant Pathol. 1984, 24, 143–155. [Google Scholar] [CrossRef]
- Swift, J.M. Loss of Suberin from Bark tissue rotted by Armillaria mellea. Nature 1965, 207, 436–437. [Google Scholar] [CrossRef]
- Kontkanen, H.; Westerholm-Parvinen, A.; Saloheimo, M.; Bailey, M.; Ratto, M.; Mattila, I.; Mohsina, M.; Kalkkinen, N.; Nakari-Setala, T.; Buchert, J. Novel Coprinopsis cinerea polyesterase that hydrolyzes cutin and suberin. Appl Env. Microbiol 2009, 75, 2148–2157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, R.; Garcia, H.; Sousa, A.F.; Freire, C.S.R.; Silvestre, A.J.D.; Rebelo, L.P.N.; Pereira, C.S. Isolation of suberin from birch outer bark and cork using ionic liquids: A new source of macromonomers. Ind. Crop. Prod. 2013, 44, 520–527. [Google Scholar] [CrossRef]
- Ferreira, R.; Garcia, H.; Sousa, A.F.; Petkovic, M.; Lamosa, P.; Freire, C.S.R.; Silvestre, A.J.D.; Rebelo, L.P.N.; Pereira, C.S. Suberin isolation from cork using ionic liquids: Characterisation of ensuing products. New J. Chem. 2012, 36, 2014–2024. [Google Scholar] [CrossRef]
- Martins, I.; Hartmann, D.O.; Alves, P.C.; Martins, C.; Garcia, H.; Leclercq, C.C.; Ferreira, R.; He, J.; Renaut, J.; Becker, J.D.; et al. Elucidating how the saprophytic fungus Aspergillus nidulans uses the plant polyester suberin as carbon source. BMC Genom. 2014, 15, 613. [Google Scholar] [CrossRef] [Green Version]
- Ursache, R.; De Jesus Vieira Teixeira, C.; Dénervaud Tendon, V.; Gully, K.; De Bellis, D.; Schmid-Siegert, E.; Grube Andersen, T.; Shekhar, V.; Calderon, S.; Pradervand, S.; et al. GDSL-domain proteins have key roles in suberin polymerization and degradation. Nat. Plants 2021. [Google Scholar] [CrossRef]
- Tibbett, M.; Grantham, K.; Sanders, F.E.; Cairney, J.W.G. Induction of cold active acid phosphomonoesterase activity at low temperature in psychrotrophic ectomycorrhizal Hebeloma spp. Mycol. Res. 1998, 102, 1533–1539. [Google Scholar] [CrossRef]
- Weinstein, R.N.; Montiel, P.O.; Johnstone, K. Influence of growth temperature on lipid and soluble carbohydrate synthesis by fungi isolated from fellfield soil in the maritime Antarctic. Mycologia 2000, 92, 222–229. [Google Scholar] [CrossRef]
- Duarte, A.W.F.; dos Santos, J.A.; Vianna, M.V.; Vieira, J.M.F.; Mallagutti, V.H.; Inforsato, F.J.; Wentzel, L.C.P.; Lario, L.D.; Rodrigues, A.; Pagnocca, F.C.; et al. Cold-adapted enzymes produced by fungi from terrestrial and marine Antarctic environments. Crit. Rev. Biotechnol. 2018, 38, 600–619. [Google Scholar] [CrossRef] [Green Version]
- Robinson, C.H. Cold adaptation in Arctic and Antarctic fungi. New Phytol. 2001, 151, 341–353. [Google Scholar] [CrossRef]
- Björdal, C. Microbial degradation of waterlogged archaeological wood. J. Cult. Herit. 2012, 13, 118–122. [Google Scholar] [CrossRef]
- Björdal, C.; Nilsson, T. Decomposition of waterlogged archaeological wood. In Proceedings of the 8th ICOM-CC WOAM Conference, Stockholm, Sweden, 11–15 June 2001; pp. 235–247. [Google Scholar]
- Pedersen, N.B.; Björdal, C.G.; Jensen, P.; Felby, C. Bacterial Degradation of Archaeological Wood in Anoxic Waterlogged Environments. In Stability of Complex Carbohydrate Structures: Biofuels, Foods, Vaccines and Shipwrecks; Harding, S.E., Ed.; RSC Publishing: Cambridge, MA, USA, 2012; pp. 160–187. [Google Scholar]
- Blanchette, R.A. A review of microbial deterioration found in archaeological wood from different environments. Int. Biodeterior. Biodegrad. 2000, 46, 189–204. [Google Scholar] [CrossRef]
- Blanchette, R.A.; Nilsson, T.; Daniel, G.; Abad, A. Biological Degradation of Wood. In Archaeological Wood; Rowell, R.M., James Barbour, R., Eds.; Advances in Chemistry; American Chemical Society: Washington, DC, USA, 1990; Volume 225, pp. 141–174. [Google Scholar]
- Blanchette, R.; Cease, K.; Abad, A.; Burnes, T.; Obst, J. Ultrastructural characterization of wood from Tertiary fossil forest in the Canadian Arctic. Can. J. Bot. 1991, 69, 560–568. [Google Scholar] [CrossRef]
- Blanchette, R.A. Microbial degradation of wood from aquatic and terrestrial environments. In Cultural Heritage Microbiology: Fundamental Studies in Conservation Science; Mitchell, R., McNamara, C.J., Eds.; ASM Press: Washington, DC, USA, 2010; pp. 179–218. [Google Scholar]
- Blanchette, R.A.; Held, B.W.; Jurgens, J.; Stear, A.; Dupont, C. Fungi attacking historic wood of Fort Conger and the Peary Huts in the High Arctic. PLoS ONE 2021, 16, e0246049. [Google Scholar] [CrossRef]
- Matthiesen, H.; Jensen, J.B.; Gregory, D.; Hollesen, J.; Elberling, B. Degradation of Archaeological Wood Under Freezing and Thawing Conditions—Effects of Permafrost and Climate Change. Archaeometry 2014, 56, 479–495. [Google Scholar] [CrossRef] [Green Version]
- Pedersen, N.B.; Matthiesen, H.; Blanchette, R.; Alfredsen, G.; Held, B.; Westergaard-Nielsen, A.; Hollesen, J. Fungal attack on archaeological wooden artefacts in the Arctic-implications in a changing climate. Sci. Rep. 2020, 10, 8187. [Google Scholar] [CrossRef]
- Hafner, A.; Klügl, J. Neolithisches Bogenfutteral aus Birkenrinde, Holz und Leder. In Schnidejoch und Lötschenpass. Archäologische Forschungen in den Berner Alpen. Schnidejoch et Lötschenpass. Investigations Archéologiques dans les Alpes Bernoises; Archäologischer Dienst Bern: Bern, Switzerland, 2015; Volume 2, pp. 15–18. [Google Scholar]
- Junkmanns, J.; Klügl, J.; Schoch, W.H.; Di Pietro, G.; Hafner, A. Neolithic and Bronze Age archery equipment from alpine ice-patches: A review on components, construction techniques and functionality. J. Neolit. Archaeol. 2019, 21, 283–314. [Google Scholar] [CrossRef]
- Klügl, J. How to conserve a birch bark bow case from an ice patch? In Proceedings of the 12th ICOM-CC Group on Wet Organic Archaeological Materials Conference, Istanbul, Turkey, 13–17 May 2013; pp. 270–278. [Google Scholar]
- Pilø, L.; Finstad, E.; Barrett, J.H. Crossing the ice: An Iron Age to medieval mountain pass at Lendbreen, Norway. Antiquity 2020, 94, 437–454. [Google Scholar] [CrossRef] [Green Version]
- Haeberli, W.; Alean, J. Temperature and accumulation of high altitude firn in the alps. Ann. Glaciol. 1985, 6, 161–163. [Google Scholar] [CrossRef] [Green Version]
- Isaksen, K.; Holmlund, P.; Sollid, J.L.; Harris, C. Three deep Alpine-permafrost boreholes in Svalbard and Scandinavia. Permafr. Periglac. Process. 2001, 12, 13–25. [Google Scholar] [CrossRef]
- Guthruf, K.; Maurer, V.; Ryser, R.; Zeh, M.; Zweifel, N. Zustand der Kleinseen; AWA Amt für Wasser und Abfall, Gewässer-und Bodenschutzlabor GBL: Bern, Switzerland, 2015. [Google Scholar]
- Ghemawat, M.S. Polychromatic staining with toluidine blue O for studying the host-parasite relationships in wheat leaves of Erysiphe graminis f. sp. tritici. Physiol. Plant Pathol. 1977, 11, 251–253. [Google Scholar] [CrossRef]
- Pedersen, N.B. Microscopic and Spectroscopic Characterisation of Waterlogged Archaeological Softwood from Anoxic Environments; University of Copenhagen: Frederiksberg, Denmark, 2015. [Google Scholar]
- Sousa, A.F.; Gandini, A.; Caetano, A.; Maria, T.M.; Freire, C.S.; Neto, C.P.; Silvestre, A.J. Unravelling the distinct crystallinity and thermal properties of suberin compounds from Quercus suber and Betula pendula outer barks. Int. J. Biol. Macromol. 2016, 93, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Daniel, G.; Nilsson, T.; Pettersson, B. Poorly and Non-Lignified Regions in the Middle Lamella Cell Corners of Birch (Betula Verrucosa) and Other Wood Species. IAWA J. 1991, 12, 70–83. [Google Scholar] [CrossRef]
- de Lhoneux, B.; Antoine, R.; Côté, W.A. Ultrastructural implications of gamma-irradiation of wood. Wood Sci. Technol. 1984, 18, 161–176. [Google Scholar] [CrossRef]
- Parameswaran, N.; Wilhelm, G.E.; Liese, W. Ultrastructural aspects of beech bark degradation by fungi. Eur. J. For. Pathol. 1976, 6, 274–286. [Google Scholar] [CrossRef]
- Singh, P.A. A review of microbial decay types found in wooden objects of cultural heritage recovered from buried and waterlogged enviromnents. J. Cult. Herit. 2012, 13S, 16–20. [Google Scholar] [CrossRef]
- Beaulieu, C.; Sidibé, A.; Jabloune, R.; Simao-Beaunoir, A.-M.; Lerat, S.; Monga, E.; Bernards, M.A. Physical, Chemical and Proteomic Evidence of Potato Suberin Degradation by the Plant Pathogenic Bacterium Streptomyces scabiei. Microbes Environ. 2016, 31, 427–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chowdhury, P.; Nag, S.; Ray, A. Degradation of Phenolic Compounds Through UV and Visible-Light-Driven Photocatalysis: Technical and Economic Aspects. Phenolic Compd. Nat. Sources Importance Appl. 2017, 16, 395–417. [Google Scholar]
- Gandini, A.; Pascoal Neto, C.; Silvestre, A.J.D. Suberin: A promising renewable resource for novel macromolecular materials. Prog. Polym. Sci. 2006, 31, 878–892. [Google Scholar] [CrossRef]
- Rosa, M.E.; Pereira, H.; Fortes, M.A. Effects of hot water treatment on the structure and properties of cork. Wood Fiber Sci. 1990, 22, 149–164. [Google Scholar]














| Sample | Object | Location | Date | Burial Conditions | ||
|---|---|---|---|---|---|---|
| Environment | T, RH | O2 | ||||
| Ref | Intact phellem | Valais, CH | Contemporary | tree | Ambient | |
| 0 | Rotten phellem | Valais, CH | Contemporary | In-nature rotten tree | Ambient | |
| 1 | Bow case, lower body | Schnidejoch | ≈2800 BC (2905–2622 cal BC) | Ice patch | 0 °C, 100% (*) | Low |
| 2 | Bow case, lower body | Schnidejoch | ≈2800 BC (2905–2622 cal BC) | Ice patch | 0 °C, 100% | Low |
| 3 | Bow case, lower body | Schnidejoch | ≈2800 BC (2905–2622 cal BC) | Ice patch | 0 °C, 100% | Low |
| 4 | Bow case, probably from lid | Schnidejoch | ≈2800 BC (2881–2579 cal BC) | Ice patch | 0 °C, 100% | Low |
| 5 | Bow case, lower body | Schnidejoch | ≈2800 BC (2905–2622 cal BC) | Ice patch | 0 °C, 100% | Low |
| 6 | Bow case, upper body | Schnidejoch | ≈2800 BC (3011–2668 cal BC) | Ice patch | 0 °C, 100% | Low |
| 7 | Bow case, upper body | Schnidejoch | ≈2800 BC (3011–2668 cal BC) | Ice patch | 0 °C, 100% | Low |
| 8 | Bow case, upper body | Schnidejoch | ≈2800 BC (3011–2668 cal BC) | Ice patch | 0 °C, 100% | Low |
| 9 | Fragment | Lendbreen | ≈500 AD (430–540 cal AD) | Ice patch | −3 °C (**) | Low |
| 10 | Fragment | Lendbreen | ≈900 AD (891–981 cal AD) | Ice patch | −3 °C | Low |
| 11 | Fragment | Lendbreen | ≈1450 AD (1430–1470 cal AD) | Ice patch | −3 °C | Low |
| 12 | Fragment | Lake Moossee | 4500–3800 BC | Waterlogged | ≈4–16 °C (†) | Anoxic |
| 13 | Fragment | Kosackenberg | 2300–1800 BC | Cave | 4–7 °C, ≈95% (‡) | Ambient |
| Fra | Un | No-bi | In-st | Swe | Ir | Det | Rem | Re-th | Hy | Vo | Me | Acc | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ref | x | x | x | ||||||||||
| 0 | x | x | x | x-fu | x-fu | x | x-fu-wl | x-fu | x-fu | x | x-fu | x-fu | |
| 1 | x | x | x-fu | x-fu | x-fu-cl | x-fu | x-fu | x-fu | |||||
| 2 | x | x | x | x-ov | x-fu | x-fu-cl | x-fu | x | |||||
| 3 | x | x | x | x-ov | x | x-ov | x (rarely) | ||||||
| 4 | x | x, x-st | x-ov, x-fu | x | x-ov-wl, x-fu-cl | x-fu | |||||||
| 5 | x | x-ov-wl, x-fu-cl | x-ov | ||||||||||
| 6 | x | x | x | x-fu | x-fu | x-wl-fu | x-fu | x-fu | x | x-fu | x-fu | ||
| 7 | x | x | x | x-fu | x-fu | x-fu-cl, x-fu-wl | x-fu | x | x | x | |||
| 8 | x | x | x | x-ov | x-ov | x | x-ov | x-ov | |||||
| 9 | x | x | x-ov, x-fu | x (rarely) | |||||||||
| 10 | x | x | x-ov | x-fu, x-ov | x-ov, x-fu | x | x-fu | ||||||
| 11 | x | x | x-fu | x-fu | x | x-fu | |||||||
| 12 | x | x | x | x-ov | x-ov | x-fu?-wl, x-fu?-cl | x-fu? | x (rarely) | x-ba | x-ba | |||
| 13 | x | x | x | x-ov | x-fu | x-fu-wl | x-fu | x (rarely) | x |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klügl, J.; Di Pietro, G. Atlas of Micromorphological Degradation of Archaeological Birch Bark. Appl. Sci. 2021, 11, 8721. https://doi.org/10.3390/app11188721
Klügl J, Di Pietro G. Atlas of Micromorphological Degradation of Archaeological Birch Bark. Applied Sciences. 2021; 11(18):8721. https://doi.org/10.3390/app11188721
Chicago/Turabian StyleKlügl, Johanna, and Giovanna Di Pietro. 2021. "Atlas of Micromorphological Degradation of Archaeological Birch Bark" Applied Sciences 11, no. 18: 8721. https://doi.org/10.3390/app11188721
APA StyleKlügl, J., & Di Pietro, G. (2021). Atlas of Micromorphological Degradation of Archaeological Birch Bark. Applied Sciences, 11(18), 8721. https://doi.org/10.3390/app11188721






