Experimental Study on the Surface Properties of Nanoalumina-Filled Epoxy Resin Nanocomposites
Abstract
1. Introduction
2. Materials and Experimental Procedures
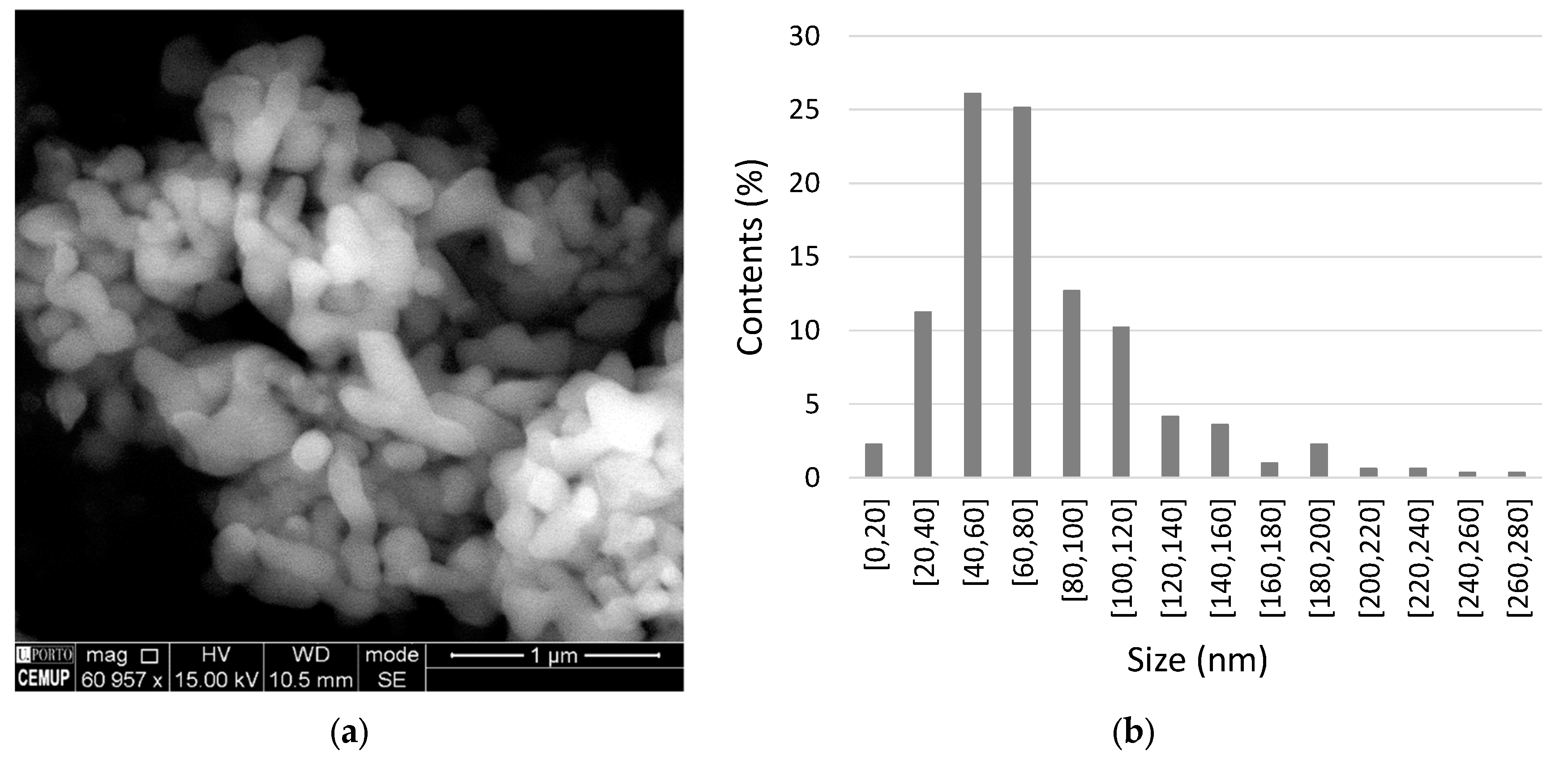
2.1. Raw Materials
2.2. Preparation of the Epoxy Resin Nanocomposites
2.3. Experimental Procedures
3. Results and Discussion
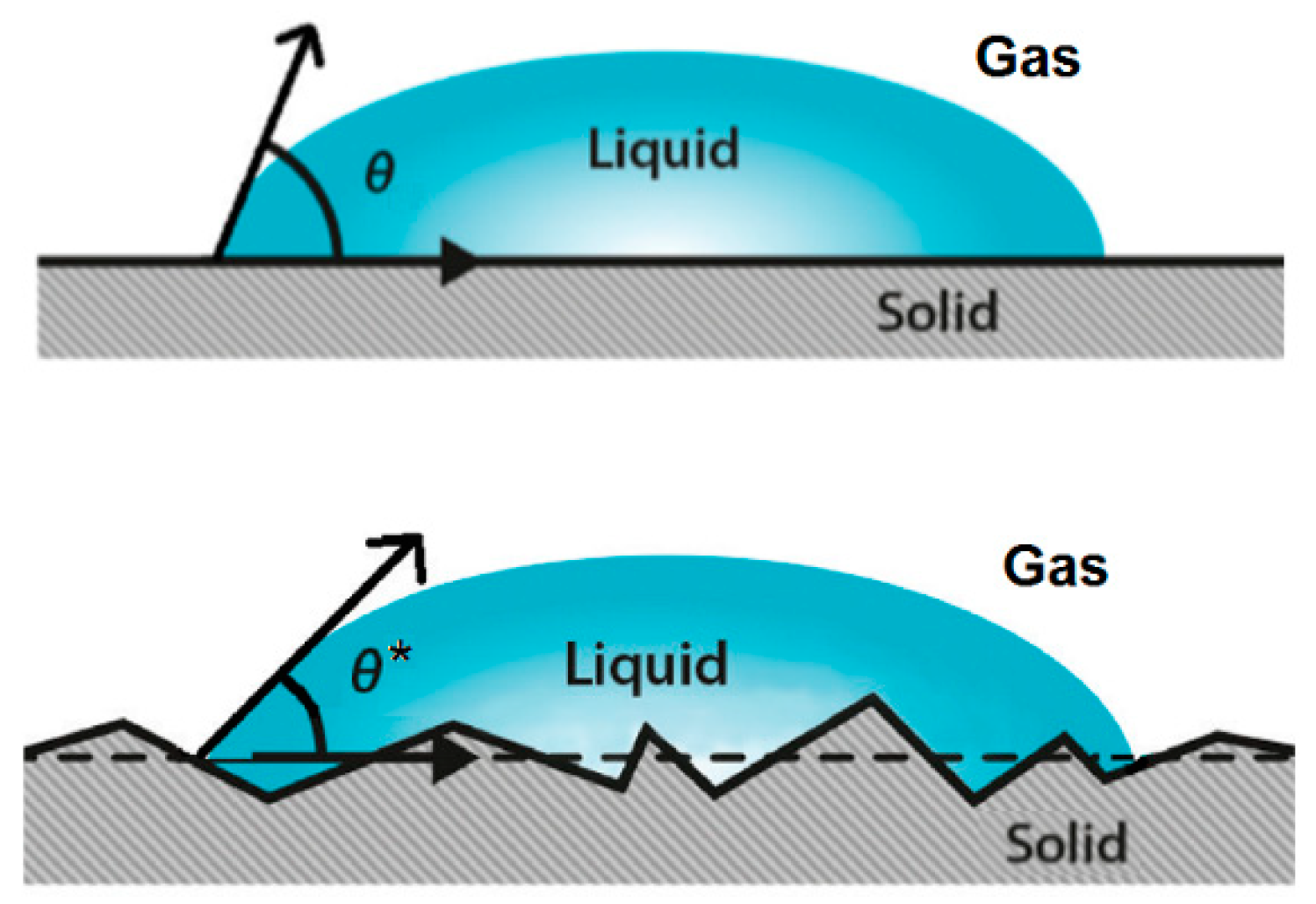
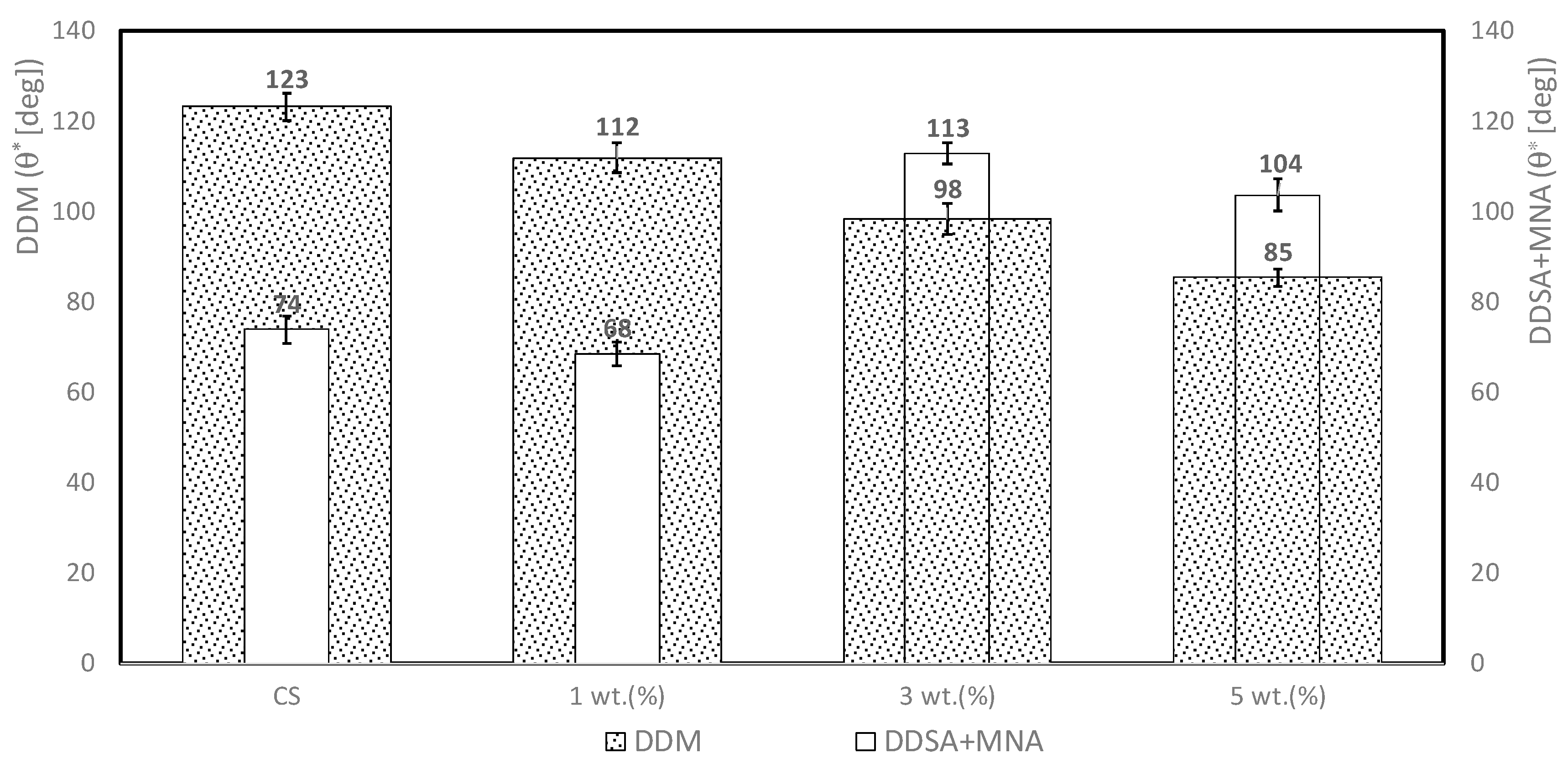
3.1. Contact Angle
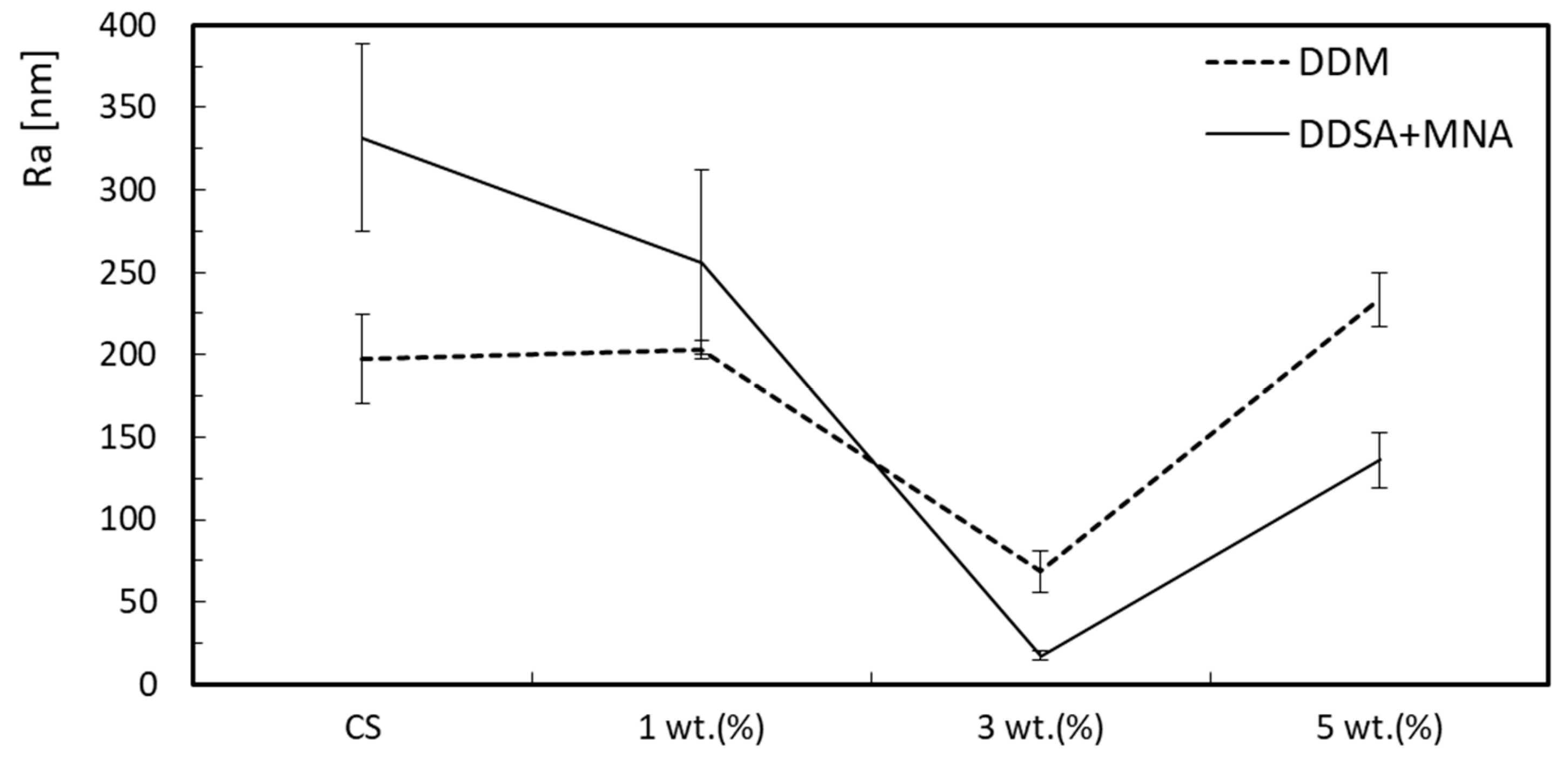
3.2. Surface Roughness
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Amaro, A.M.; Bernardo, L.; Pinto, D.G.; Lopes, S.; Rodrigues, J.; Louro, C.S. Effect of irregular shaped nanoalumina on the enhancement of mechanical properties of epoxy resin nanocomposites using DDM as hardener. Compos. Part B Eng. 2016, 84, 17–24. [Google Scholar] [CrossRef]
- Amaro, A.M.; Bernardo, L.; Pinto, D.G.; Lopes, S.; Rodrigues, J. The influence of curing agents in the impact properties of epoxy resin nanocomposites. Compos. Struct. 2017, 174, 26–32. [Google Scholar] [CrossRef]
- Wetzel, B.; Rosso, P.; Haupert, F.; Friedrich, K. Epoxy nanocomposites—Fracture and toughening mechanisms. Eng. Fract. Mech. 2006, 73, 2375–2398. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, H.; Tang, L.; Liu, G.; Zhang, D. The Effects of Alumina Nanofillers on Mechanical Properties of High-Performance Epoxy Resin. J. Nanosci. Nanotechnol. 2010, 10, 7526–7532. [Google Scholar] [CrossRef] [PubMed]
- Argon, A.S.; Cohen, R.E. Toughenability of polymers. Polymer 2003, 44, 6013–6032. [Google Scholar] [CrossRef]
- Jagtap, R.; Ambre, A. Atomic force microscopy (AFM): Basics and its important applications for polymer characterization: An overview. J. Polym. Mater. 2005, 22, 1–26. [Google Scholar]
- Pinto, D.; Bernardo, L.; Amaro, A.; Lopes, S. Mechanical properties of epoxy nanocomposites using titanium dioxide as reinforcement—A review. Constr. Build. Mater. 2015, 95, 506–524. [Google Scholar] [CrossRef]
- Pinto, D.; Bernardo, L.; Amaro, A.; Lopes, S. Mechanical properties of epoxy nanocomposites using alumina as reinforcement—A review. J. Nano Res. 2015, 30, 9–38. [Google Scholar] [CrossRef]
- Day, R.J.; Lovell, P.A.; Wazzan, A.A. Toughened carbon/epoxy composites made by using core/shell particles. Compos. Sci. Technol. 2001, 61, 41–56. [Google Scholar] [CrossRef]
- Kinloch, A.J.; Maxwell, D.L.; Young, R.J. The fracture of hybrid-particulate composites. J. Mater. Sci. 1985, 20, 4169–4184. [Google Scholar] [CrossRef]
- Maxwell, D.; Young, R.J.; Kinloch, A.J. Hybrid particulate-filled epoxy-polymers. J. Mater. Sci. Lett. 1984, 3, 9–12. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, Z.; Friedrich, K.; Eger, C. Property improvements of in situ epoxy nanocomposites with reduced interparticle distance at high nanosilica content. Acta Mater. 2006, 54, 1833–1842. [Google Scholar] [CrossRef]
- Zhang, H.; Tang, L.C.; Zhang, Z.; Friedrich, K.; Sprenger, S. Fracture behaviours of in situ silica nanoparticle-filled epoxy at different temperatures. Polymer 2008, 49, 3816–3825. [Google Scholar] [CrossRef]
- Johnsen, B.B.; Kinloch, A.J.; Mohammed, R.D.; Taylor, A.C.; Sprenger, S. Toughening mechanisms of nanoparticle-modified epoxy polymers. Polymer 2007, 48, 530–541. [Google Scholar] [CrossRef]
- Bittmann, B.; Englert, M.; Haupert, F.; Schlarb, A.K. Influence of the insertion of TiO2-nanoparticles in epoxy resins on the curing behavior and the resulting mechanical properties. J. Adv. Mater. 2009, 41, 20–27. [Google Scholar]
- Zhang, Z.; Breidt, C.; Chang, L.; Haupert, F.; Friedrich, K. Enhancement of the wear resistance of epoxy: Short carbo fibre, graphite, PTFE and nano-TiO2. Compos. Part A Appl. Sci. Manuf. 2004, 35, 1385–1392. [Google Scholar] [CrossRef]
- Zunjarrao, S.C.; Singh, R.P. Characterization of the fracture behavior of epoxy reinforced with nanometer and micrometer sized aluminum particles. Compos. Sci. Technol. 2006, 66, 2296–2305. [Google Scholar] [CrossRef]
- Naous, W.; Yu, X.Y.; Zhang, Q.X.; Naito, K.; Kagawa, Y. Morphology, Tensile Properties, and Fracture Toughness of Epoxy/Al2O3 Nanocomposites. J. Polym. Sci. Part B Polym. Phys. 2006, 44, 1466–1473. [Google Scholar] [CrossRef]
- Zhao, S.; Schadler, L.S.; Duncan, R.; Hillborg, H.; Auletta, T. Mechanisms leading to improved mechanical performance in nanoscale alumina filled epoxy. Compos. Sci. Technol. 2008, 68, 2965–2975. [Google Scholar] [CrossRef]
- Zalikha, N.Z.; Feisal, M.F.; Ishak, M. Influence of Al2O3 nanoreinforcement on the adhesion and thermomechanical properties for epoxy adhesive. Compos. Part B Eng. 2019, 172, 9–15. [Google Scholar]
- Hill, D.; Barron, A.R.; Alexander, S. Comparison of hydrophobicity and durability of functionalized aluminium oxide nanoparticle coatings with magnetite nanoparticles—Links between morphology and wettability. J. Colloid Interface Sci. 2019, 555, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Zeng, K.Y.; He, C.B. Morphology, tensile and fracture characteristics of epoxy-alumina nanocomposites. Mater. Sci. Eng. 2010, 527, 5670–5676. [Google Scholar] [CrossRef]
- Wetzel, F.; Haupert, M.; Zhang, M.Q. Epoxy nanocomposites with high mechanical and tribological performance. Compos. Sci. Technol. 2003, 63, 2055–2067. [Google Scholar] [CrossRef]
- Shukla, D.K.; Parameswaran, V. Epoxy composites with 200 nm thick alumina platelets as reinforcements. J. Mater. Sci. 2007, 42, 5964–5972. [Google Scholar] [CrossRef]
- Chozhan, C.K.; Chandramohan, A.; Alagar, M. Influence of Multi-Walled Carbon Nanotubes on Mechanical, Thermal and Electrical Behavior of Polybenzoxazine—Epoxy Nanocomposites. Polym. Plast. Technol. Eng. 2015, 54, 68–80. [Google Scholar] [CrossRef]
- George, S.M.; Puglia, D.; Kenny, J.M.; Parameswaranpillai, J.; Thomas, S. Reaction-Induced Phase Separation and Thermomechanical Properties in Epoxidized Styrene-block-butadiene-block-styrene Triblock Copolymer Modified Epoxy/DDM System. Ind. Eng. Chem. Res. 2014, 53, 6941–6950. [Google Scholar] [CrossRef]
- Mandhakini, M.; Lakshmikandhan, T.; Chandramohan, A.; Alagar, M. Effect of Nanoalumina on the Tribology Performance of C4-Ether-Linked Bismaleimide-Toughened Epoxy Nanocomposites. Tribol. Lett. 2014, 54, 67–79. [Google Scholar] [CrossRef]
- Amaro, A.M.; Pinto, D.G.; Bernardo, L.; Lopes, S.; Rodrigues, J.; Louro, C.S. Mechanical Properties of Alumina Nanofilled Polymeric Composites Cured with DDSA and MNA. Fibers Polym. 2018, 19, 460–470. [Google Scholar] [CrossRef]
- Gupta, S.K.; Shukla, D.K.; Ravindra, D.K. Effect of nanoalumina in epoxy adhesive on lap shear strength and fracture toughness of aluminium joints. J. Adhes. 2019, 1–23. [Google Scholar] [CrossRef]
- de Morais, A.B.; Pereira, A.B.; Teixeira, J.P.; Cavaleiro, N.C. Strength of epoxy adhesive-bonded stainless-steel joints. Int. J. Adhes. Adhes. 2007, 27, 679–686. [Google Scholar] [CrossRef]
- Chang, L.; Zhang, Z.; Breidt, C.; Friedrich, K. Tribological properties of epoxy nanocomposites. I. Enhancement of the wear resistance by nano-TiO2 particles. Wear 2005, 258, 141–148. [Google Scholar] [CrossRef]
- Maghsoudian, A.; Salimi, A.; Mirzataheri, M. The effect of nanoalumina silanisation in tetraglycidylether epoxy adhesive. Int. J. Adhes. Adhes. 2019, 92, 119–124. [Google Scholar] [CrossRef]
- Bardella, L. Mechanical Behavior of Glass-Filled Epoxy Resins: Experiments, Homogenization Methods for Syntactic Foams, and Applications. Ph.D. Thesis, University of Brescia, Brescia, Italy, 2000; p. 294. [Google Scholar]
- Belleri, L.B.A. Two features of the uniaxial compression of a glassy epoxy resin: The yield stress rate-dependence and the volumetric instability. Mech. Time Depend. Mater. 2011, 15, 255–275. [Google Scholar]
- Martinet, W.; Van den Plas, D.; Raes, H.; Reekmans, R.; Contreras, R. Bax-induced cell death in Pichia pastoris. Biotechnol. Lett. 1999, 21, 821–829. [Google Scholar] [CrossRef]
- Krüss-Scientific. 2020. Available online: https://www.kruss-scientific.com/products/drop-shape/dsa100/drop-shape-analyzer-dsa100/ (accessed on 3 January 2020).
- Biolin Scientific. 2020. Available online: https://cdn2.hubspot.net/hubfs/516902/Pdf/Attension/Tech%20Notes/AT-TN-07-Surface-roughness-CA-wettability.pdf (accessed on 3 January 2020).
- Tanaka, T.; Lee, J.; Scheller, P.R. Chapter 1.5—Interfacial free energy and wettability. In Treatise on Process Metallurgy, Process Phenomena; Elsevier: Amsterdam, The Netherlands, 2014; Volume 2, pp. 61–77. [Google Scholar]
- Radiom, M.; Yang, C.; Chan, W.K. Characterization of surface tension and contact angle of nanofluids. In Proceedings of the Fourth International Conference on Experimental Mechanics, Singapore, 14 April 2010. [Google Scholar] [CrossRef]
- Syakur, A.; Sutanto, H. Determination of Hydrophobic Contact Angle of Epoxy Resin Compound Silicon Rubber and Silica. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2017; Volume 170, p. 012025. [Google Scholar] [CrossRef]
- Uelzen, T.H.; Muller, J. Wettability enhancement by rough surfaces generated by thin film technology. Thin Solid Film. 2003, 434, 311–315. [Google Scholar] [CrossRef]
- Kubiak, K.J.; Wilson, M.C.T.; Mathia, T.G.; Carval, P.H. Wettability versus roughness of engineering surfaces. Wear 2011, 271, 523–528. [Google Scholar] [CrossRef]
- Lafuma, A.; Quéré, D. Superhydrophobic states. Nat. Mater. 2003, 2, 457–460. [Google Scholar] [CrossRef]
- Bertolino, V.; Cavallaro, G.; Lazzara, G.; Milioto, S.; Parisi, F. Halloysite nanotubes sandwiched between chitosan layers: Novel bionanocomposites with multilayer structures. New J. Chem. 2018, 42, 8384–8390. [Google Scholar] [CrossRef]
- Makaremi, M.; Pasbakhsh, P.; Cavallaro, G.; Lazzara, G.; Kit Aw, Y.; Mae Lee, S.; Milioto, S. Effect of Morphology and Size of Halloysite Nanotubes on Functional Pectin Bionanocomposites for Food Packaging Applications. ACS Appl. Mater. Interfaces 2017, 9, 17476–17488. [Google Scholar] [CrossRef]
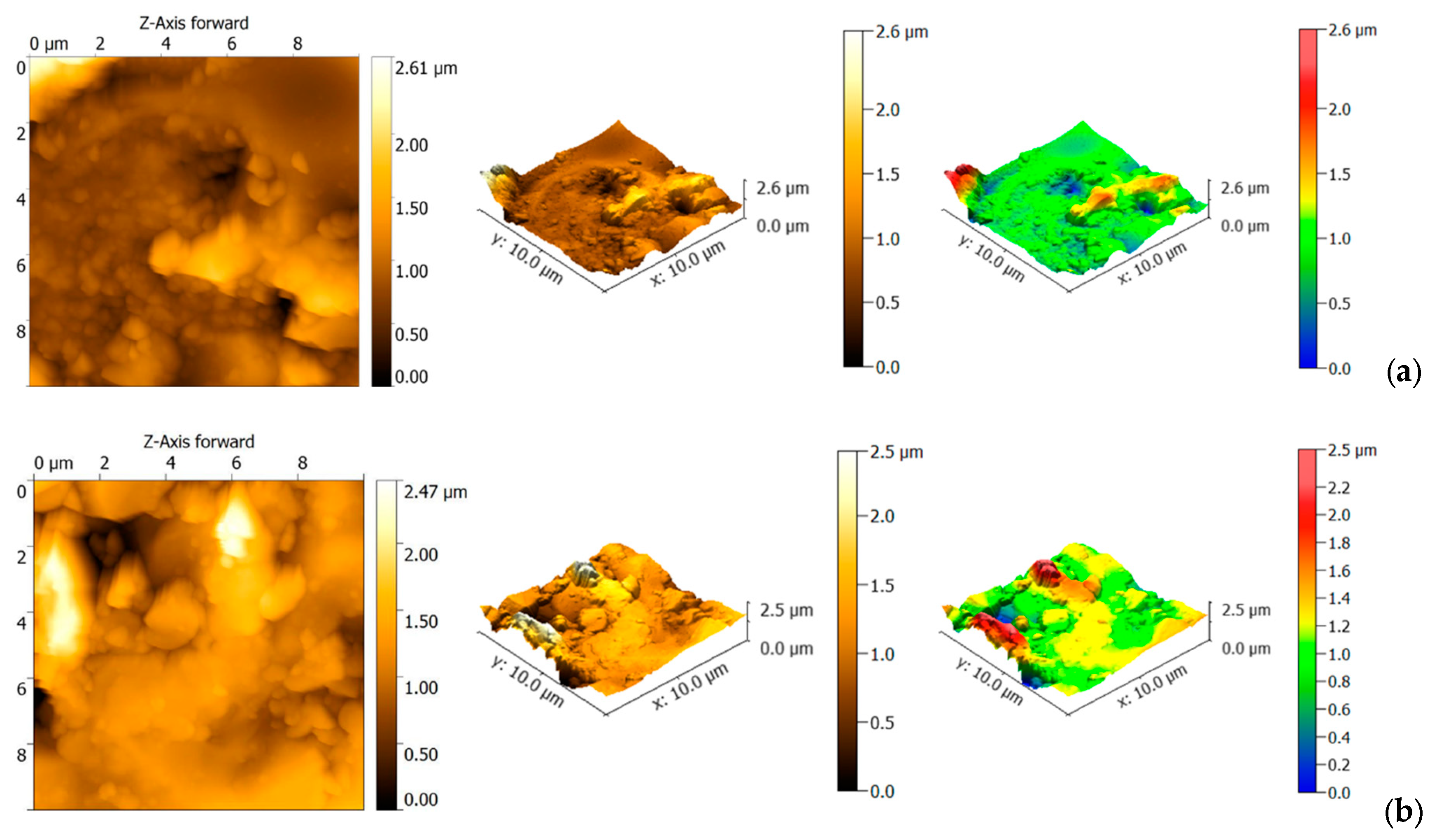
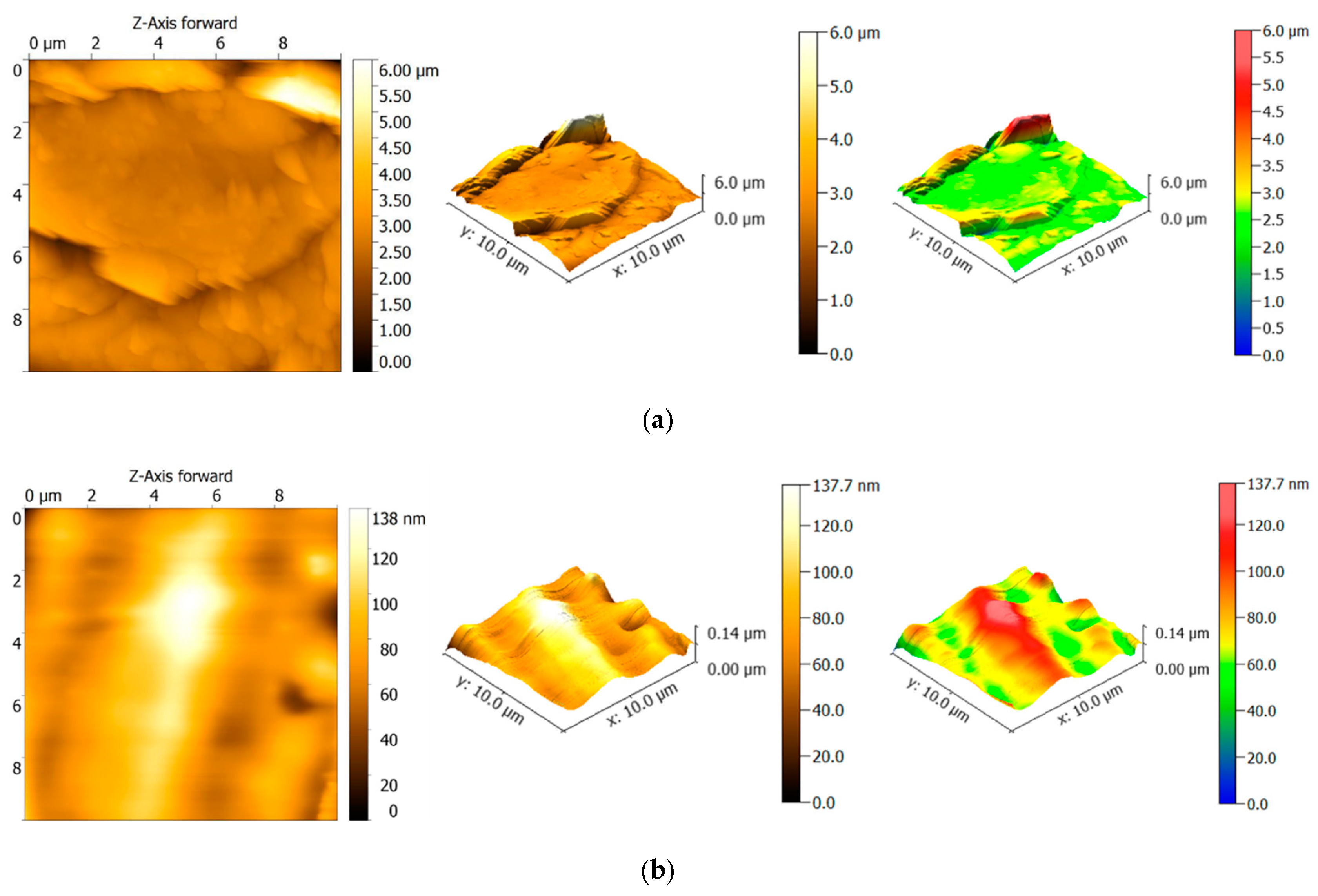
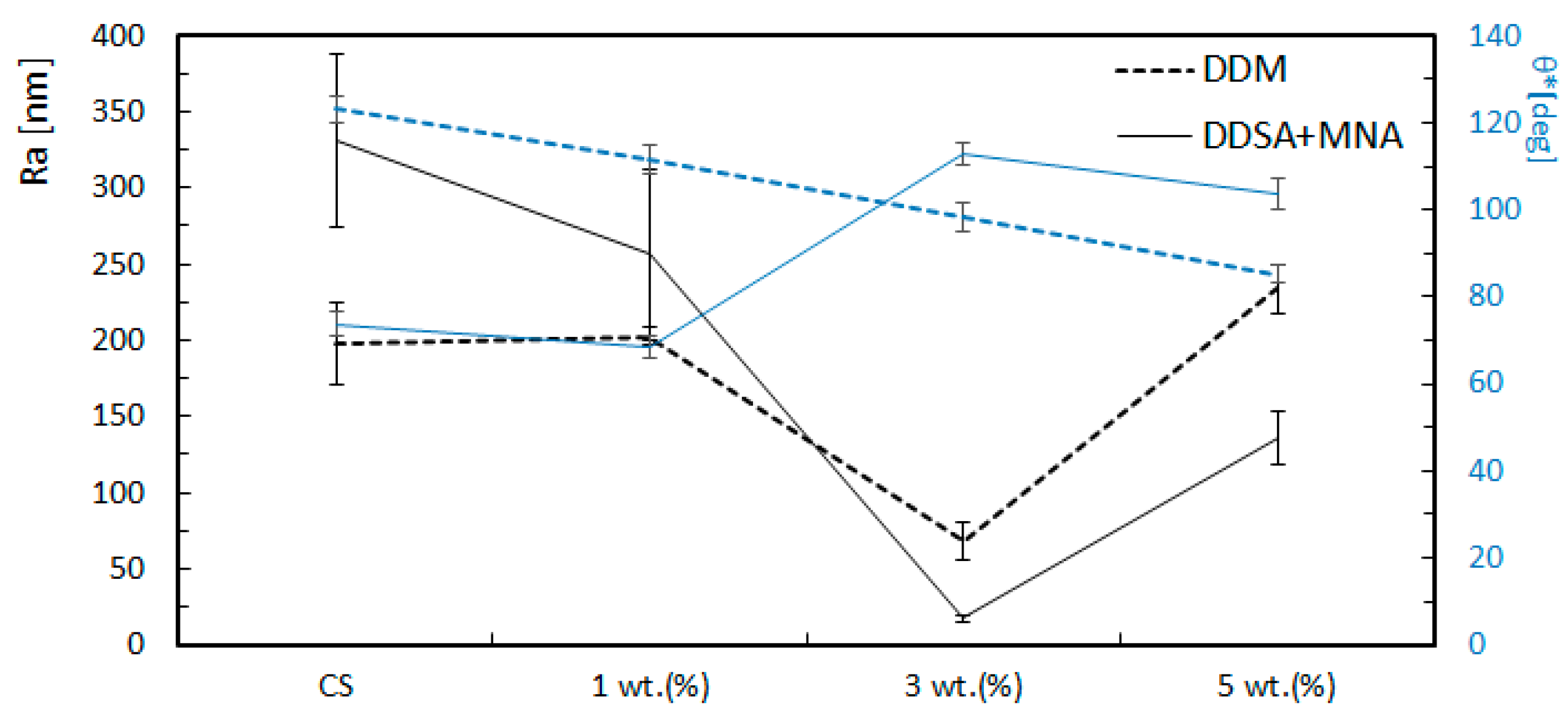
| Sample | nm | St.Dev. nm | Coef. var.% | nm | St.Dev. nm | Coef. var. % | |
|---|---|---|---|---|---|---|---|
| DDM_0 | 0 | 197.6 | 26.8 | 13.6 | 267.7 | 59.0 | 22.0 |
| DDM_1 | 1 | 202.9 | 5.4 | 2.7 | 243.7 | 5.2 | 2.1 |
| DDM_3 | 3 | 68.7 | 12.5 | 18.2 | 95.1 | 16.8 | 17.7 |
| DDM_5 | 5 | 233.6 | 16.3 | 7.0 | 289.8 | 40.4 | 13.9 |
| DDSA+MNA_0 | 0 | 331.6 | 56.7 | 17.1 | 487.1 | 120.8 | 24.8 |
| DDSA+MNA_1 | 1 | 256.2 | 55.7 | 21.7 | 324.3 | 68.8 | 21.2 |
| DDSA+MNA_3 | 3 | 17.4 | 2.7 | 15.5 | 21.6 | 3.6 | 16.7 |
| DDSA+MNA_5 | 5 | 136.0 | 16.9 | 12.4 | 186.5 | 22.4 | 12.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinto, D.; Amaro, A.M.; Bernardo, L. Experimental Study on the Surface Properties of Nanoalumina-Filled Epoxy Resin Nanocomposites. Appl. Sci. 2020, 10, 733. https://doi.org/10.3390/app10030733
Pinto D, Amaro AM, Bernardo L. Experimental Study on the Surface Properties of Nanoalumina-Filled Epoxy Resin Nanocomposites. Applied Sciences. 2020; 10(3):733. https://doi.org/10.3390/app10030733
Chicago/Turabian StylePinto, Deesy, Ana M. Amaro, and Luís Bernardo. 2020. "Experimental Study on the Surface Properties of Nanoalumina-Filled Epoxy Resin Nanocomposites" Applied Sciences 10, no. 3: 733. https://doi.org/10.3390/app10030733
APA StylePinto, D., Amaro, A. M., & Bernardo, L. (2020). Experimental Study on the Surface Properties of Nanoalumina-Filled Epoxy Resin Nanocomposites. Applied Sciences, 10(3), 733. https://doi.org/10.3390/app10030733







