High-Frame-Rate 3-D Vector Flow Imaging in the Frequency Domain
Abstract
1. Introduction
2. Materials and Methods
2.1. High-Volume-Rate Imaging
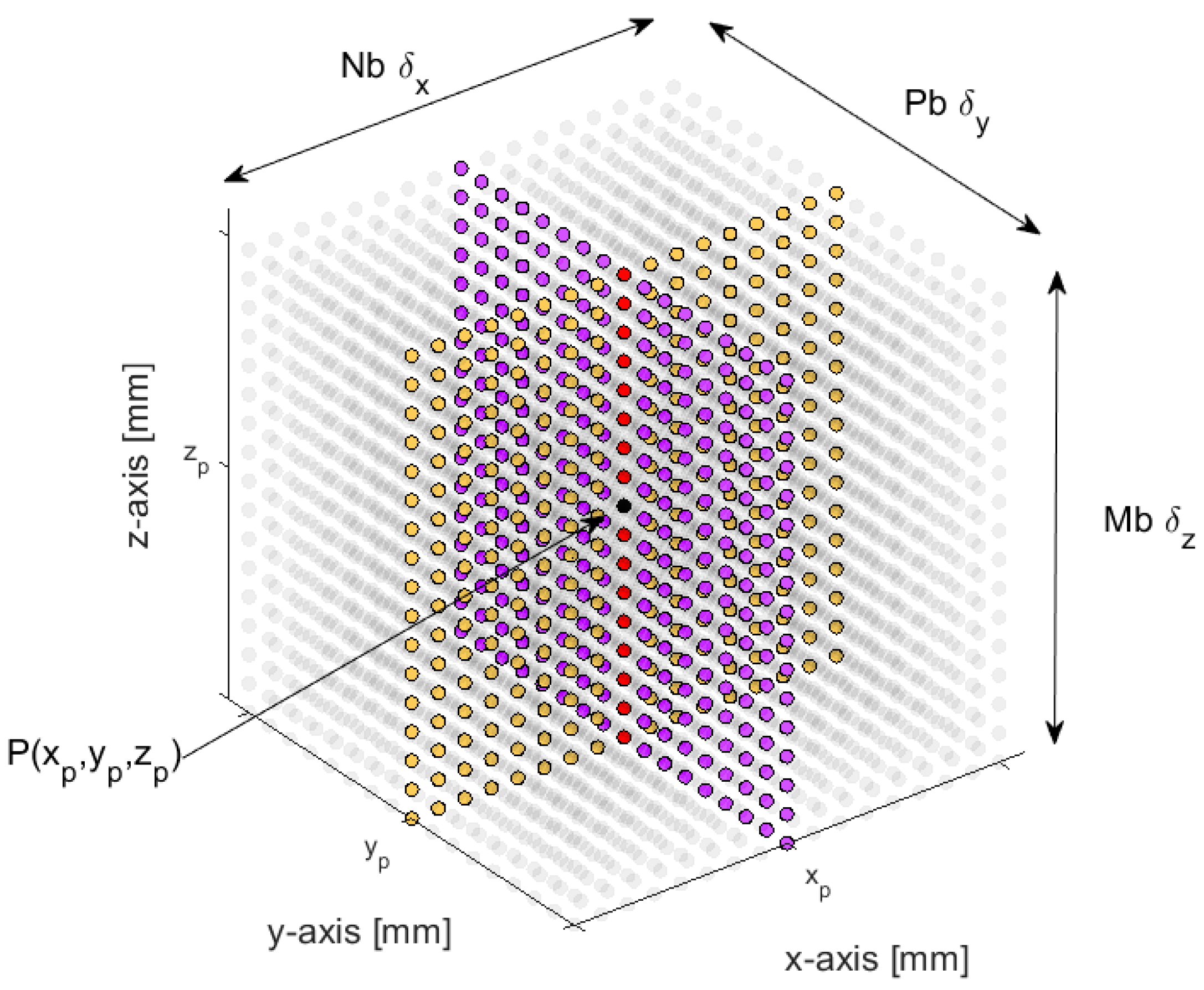
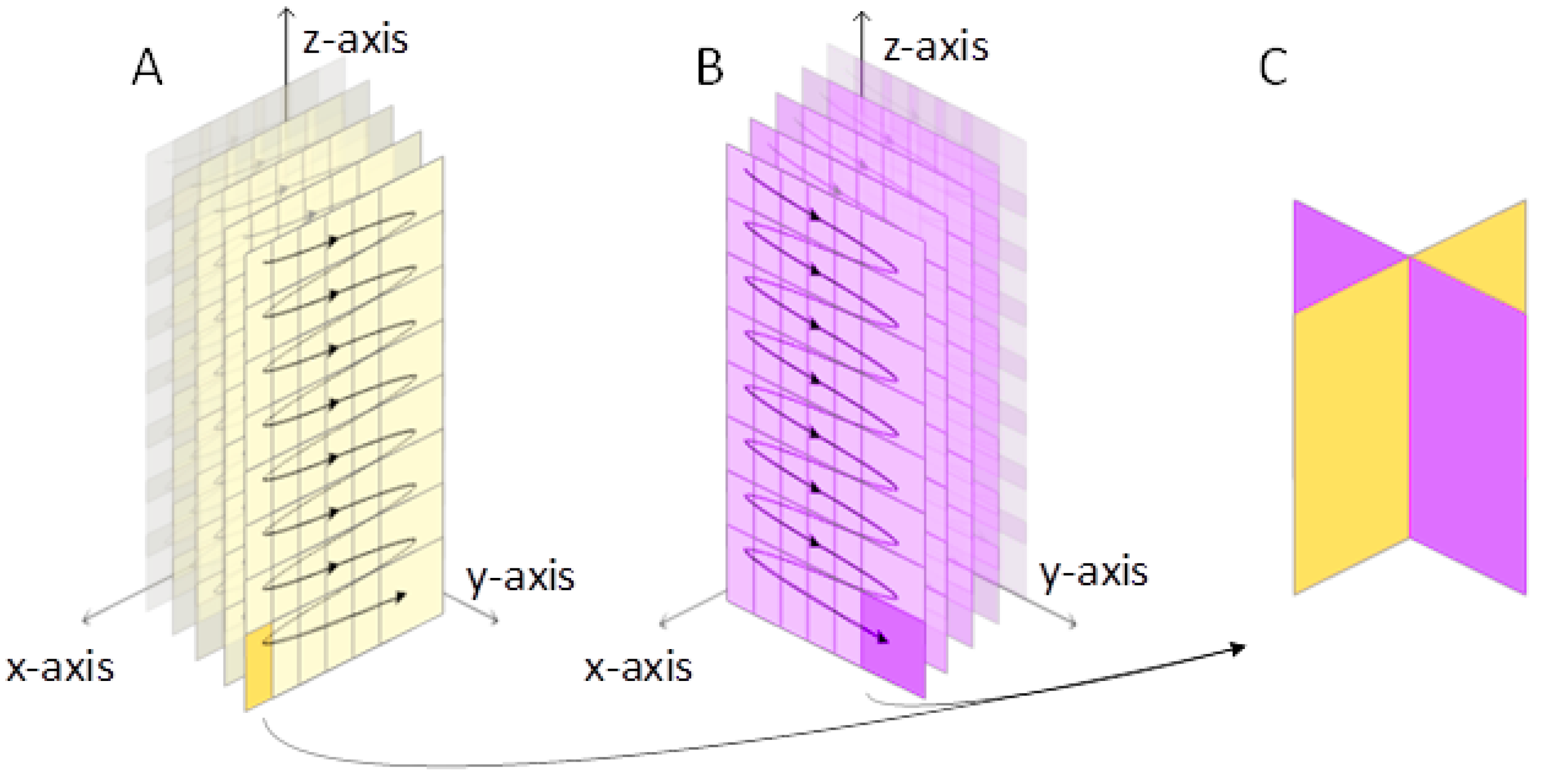
2.2. 3-D Displacement Estimation
2.3. Transmission and Reception Settings
2.4. Simulations
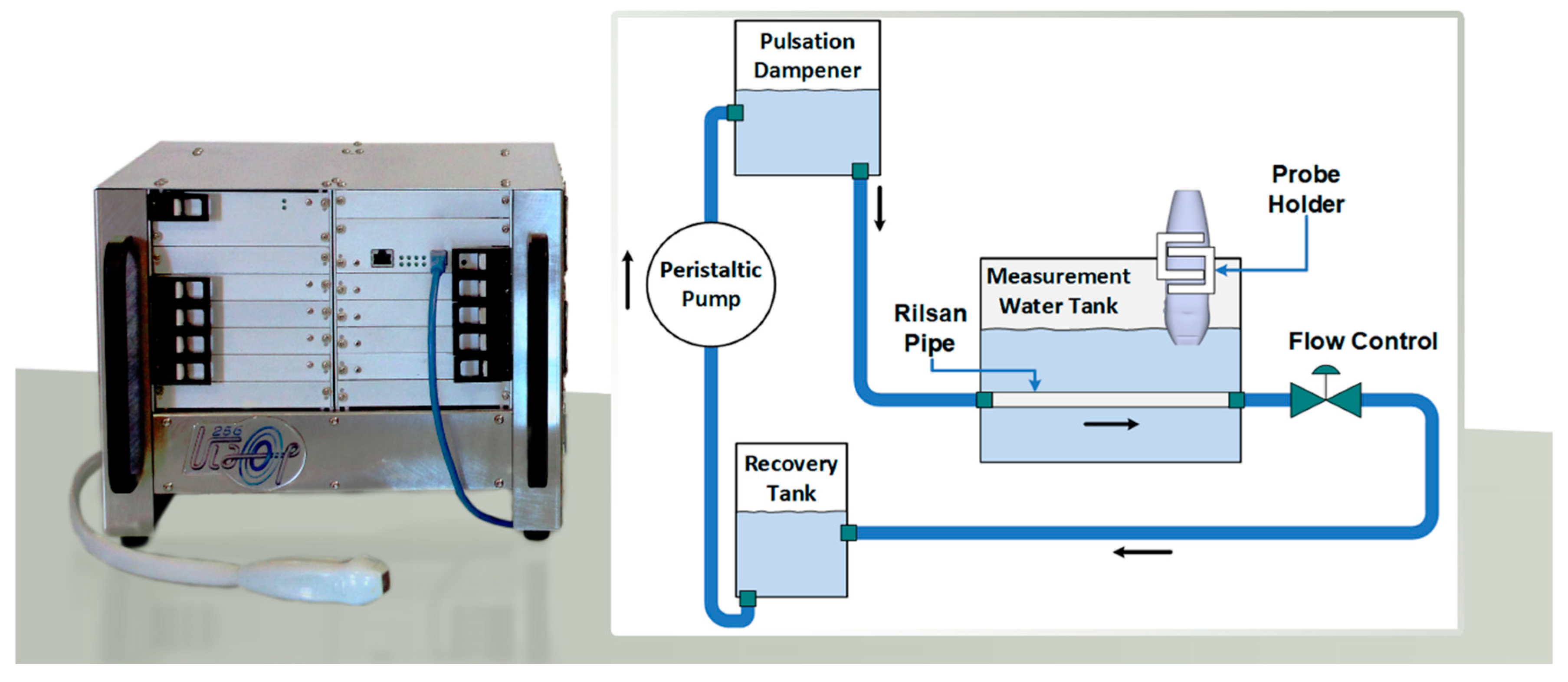
2.5. Experiments
2.6. Performance Metrics
3. Results
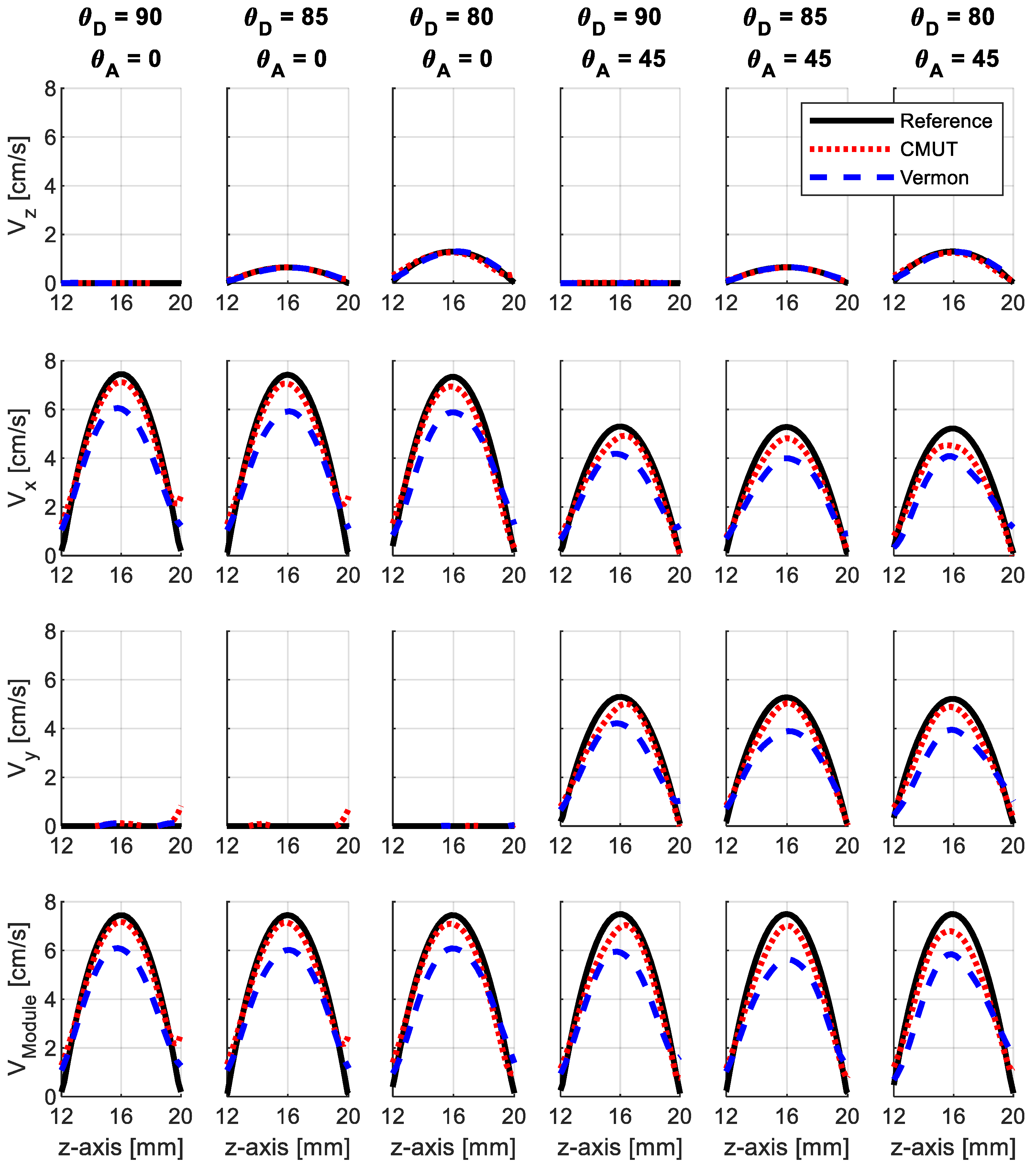
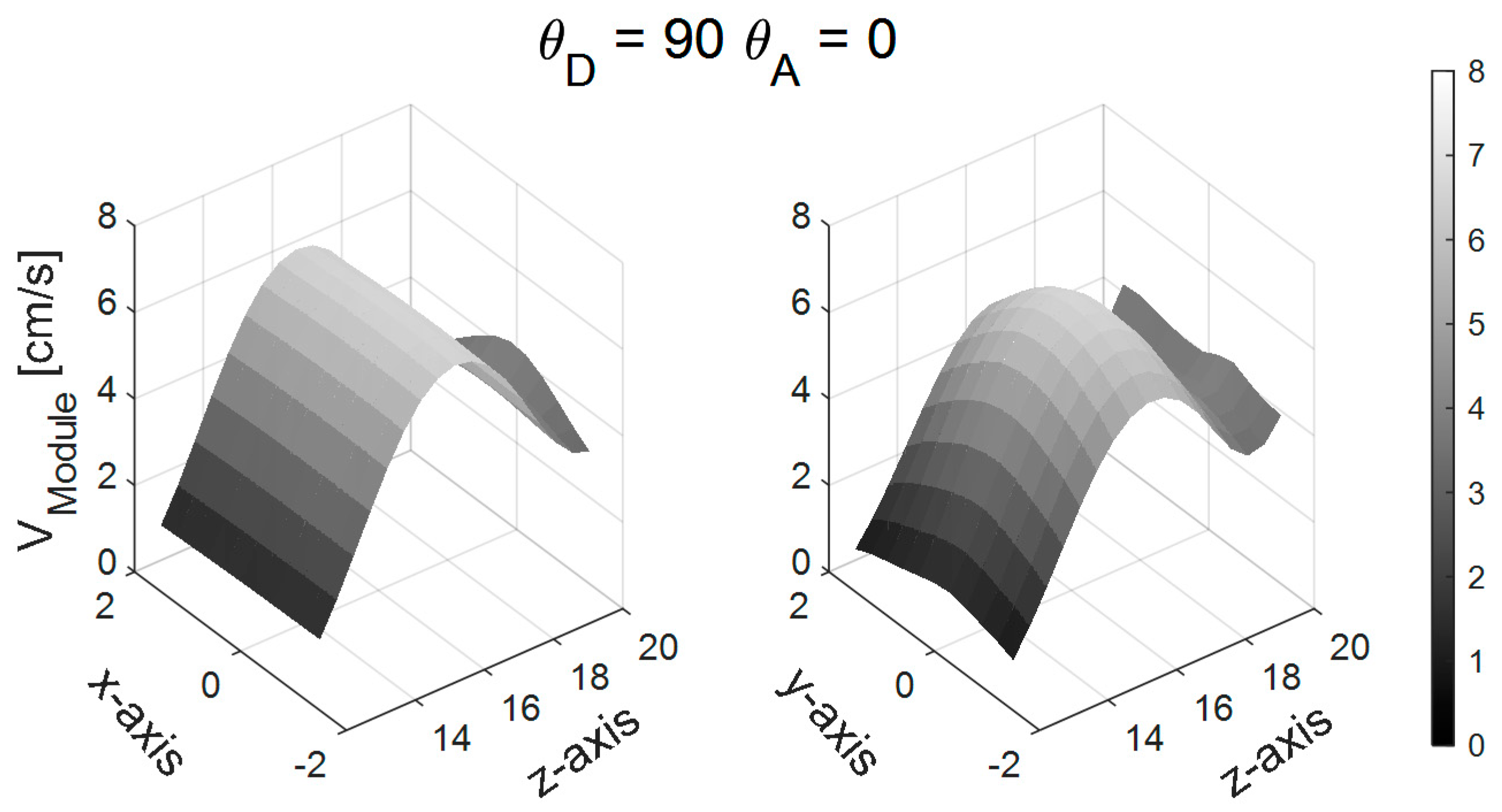
3.1. Simulations
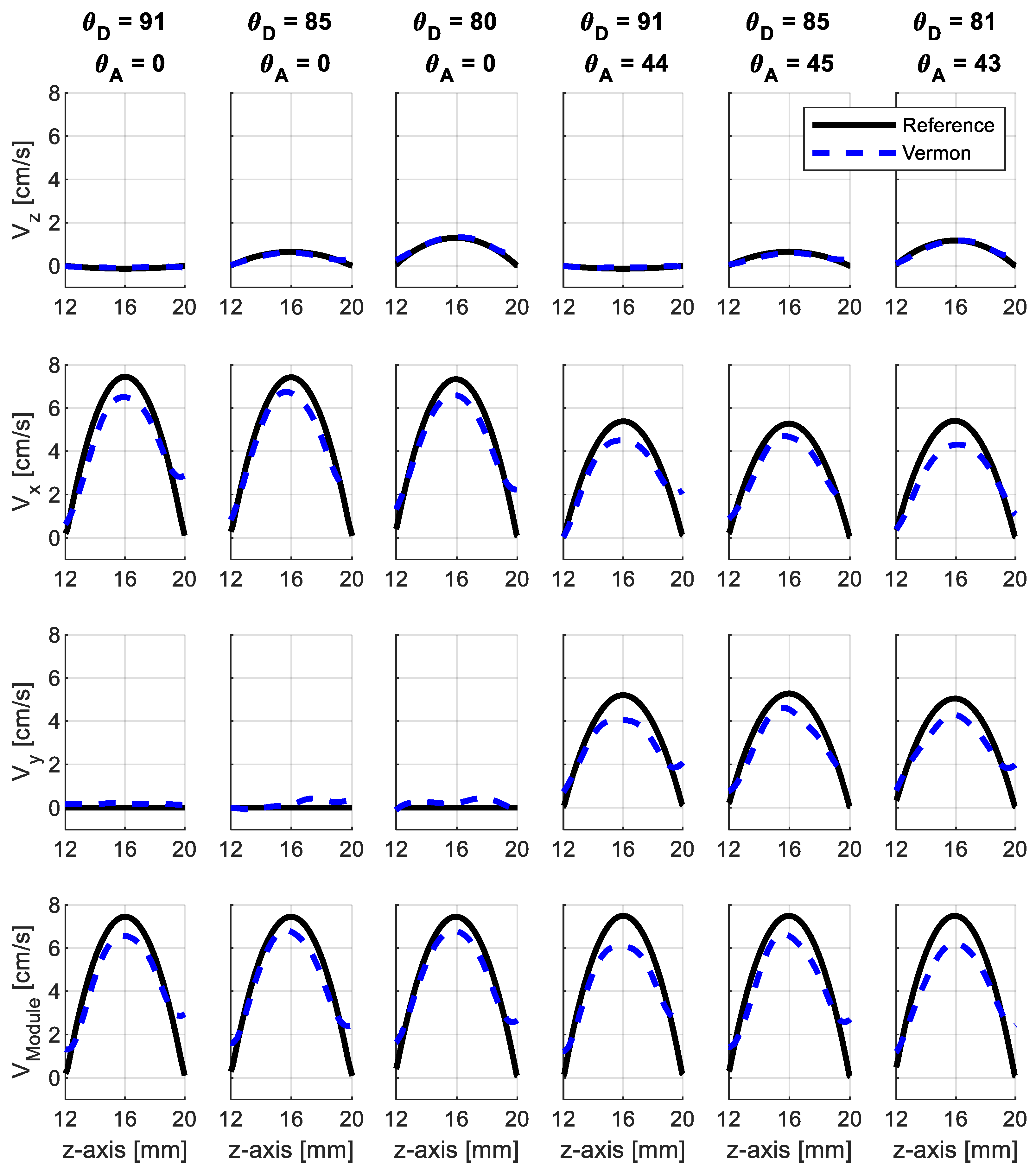
3.2. Experiments
4. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
References
- Evans, D.H.; McDicken, W.N. Doppler Ultrasound: Physics, Instrumentation and Signal Processing, 2nd ed.; Wiley-Blackwell: Hoboken, NJ, USA, 1999; ISBN 0-471-97001-8. [Google Scholar]
- Fox, M.D. Multiple crossed-beam ultrasound Doppler velocimetry. IEEE Trans. Sonics Ultrason. 1978, 25, 281–286. [Google Scholar] [CrossRef]
- Dunmire, B.; Beach, K.W.; Labs, K.-H.; Plett, M.; Strandness, D.E., Jr. Cross-beam vector Doppler ultrasound for angle-independent velocity measurements. Ultrasound Med. Biol. 2000, 26, 1213–1235. [Google Scholar] [CrossRef]
- Capineri, L.; Scabia, M.; Masotti, L. A Doppler system for dynamic vector velocity maps. Ultrasound Med. Biol. 2002, 28, 237–248. [Google Scholar] [CrossRef]
- Tortoli, P.; Dallai, A.; Boni, E.; Francalanci, L.; Ricci, S. An Automatic Angle Tracking Procedure for Feasible Vector Doppler Blood Velocity Measurements. Ultrasound Med. Biol. 2010, 36, 488–496. [Google Scholar] [CrossRef]
- Jensen, J.A.; Munk, P. A new method for estimation of velocity vectors. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 1998, 45, 837–851. [Google Scholar] [CrossRef]
- Udesen, J.; Jensen, J.A. Investigation of transverse oscillation method. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2006, 53, 959–971. [Google Scholar] [CrossRef]
- Lu, J.-Y. 2D and 3D high frame rate imaging with limited diffraction beams. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 1997, 44, 839–856. [Google Scholar] [CrossRef]
- Cheng, J.; Lu, J.-Y. Extended high-frame rate imaging method with limited-diffraction beams. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2006, 53, 880–899. [Google Scholar] [CrossRef]
- Montaldo, G.; Tanter, M.; Bercoff, J.; Benech, N.; Fink, M. Coherent plane-wave compounding for very high frame rate ultrasonography and transient elastography. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2009, 56, 489–506. [Google Scholar] [CrossRef]
- Udesen, J.; Gran, F.; Hansen, K.L.; Jensen, J.A.; Thomsen, C.; Nielsen, M.B. High frame-rate blood vector velocity imaging using plane waves: Simulations and preliminary experiments. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2008, 55, 1729–1743. [Google Scholar] [CrossRef]
- Lenge, M.; Ramalli, A.; Boni, E.; Liebgott, H.; Cachard, C.; Tortoli, P. High-frame-rate 2-D vector blood flow imaging in the frequency domain. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2014, 61, 1504–1514. [Google Scholar] [CrossRef]
- Ekroll, I.K.; Swillens, A.; Segers, P.; Dahl, T.; Torp, H.; Lovstakken, L. Simultaneous quantification of flow and tissue velocities based on multi-angle plane wave imaging. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2013, 60, 727–738. [Google Scholar] [CrossRef]
- Yiu, B.Y.S.; Lai, S.S.M.; Yu, A.C.H. Vector Projectile Imaging: Time-Resolved Dynamic Visualization of Complex Flow Patterns. Ultrasound Med. Biol. 2014, 40, 2295–2309. [Google Scholar] [CrossRef]
- Ricci, S.; Ramalli, A.; Bassi, L.; Boni, E.; Tortoli, P. Real-Time Blood Velocity Vector Measurement Over a 2-D Region. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2018, 65, 201–209. [Google Scholar] [CrossRef]
- Lenge, M.; Ramalli, A.; Tortoli, P.; Cachard, C.; Liebgott, H. Plane-wave transverse oscillation for high-frame-rate 2-D vector flow imaging. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2015, 62, 2126–2137. [Google Scholar] [CrossRef]
- Takahashi, H.; Hasegawa, H. Two dimensional blood velocity estimation using high frame rate echocardiography with transverse oscillation approach. In Proceedings of the 2015 IEEE International Ultrasonics Symposium (IUS), Taipei, Taiwan, 21–24 October 2015; pp. 1–4. [Google Scholar]
- Garcia, D.; del Alamo, J.C.; Tanne, D.; Yotti, R.; Cortina, C.; Bertrand, É.; Antoranz, J.C.; Perez-David, E.; Rieu, R.; Fernandez-Aviles, F.; et al. Two-Dimensional Intraventricular Flow Mapping by Digital Processing Conventional Color-Doppler Echocardiography Images. IEEE Trans. Med. Imaging 2010, 29, 1701–1713. [Google Scholar] [CrossRef]
- Bonnefous, O. Measurement of the complete (3D) velocity vector of blood flows. In Proceedings of the IEEE 1988 Ultrasonics Symposium Proceedings, Chicago, IL, USA, 2–5 October 1988; Volume 2, pp. 795–799. [Google Scholar]
- Newhouse, V.L.; Dickerson, K.S.; Cathignol, D.; Chapelon, J.-Y. Three-dimensional vector flow estimation using two transducers and spectral width. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 1994, 41, 90–95. [Google Scholar] [CrossRef]
- Bruni, G.; Calzolai, M.; Capineri, L.; Fort, A.; Masotti, L.; Rocchi, S.; Scabia, M. Measurement and Imaging of a Velocity Vector Field Based on a Three Transducers Doppler System. In Acoustical Imaging; Tortoli, P., Masotti, L., Eds.; Acoustical Imaging; Springer: Boston, MA, USA, 1996; pp. 431–437. ISBN 978-1-4419-8772-3. [Google Scholar]
- Fenster, A.; Downey, D.B.; Cardinal, H.N. Three-dimensional ultrasound imaging. Phys. Med. Biol. 2001, 46, R67. [Google Scholar] [CrossRef]
- Light, E.D.; Idriss, S.F.; Wolf, P.D.; Smith, S.W. Real-time three-dimensional intracardiac echocardiography. Ultrasound Med. Biol. 2001, 27, 1177–1183. [Google Scholar] [CrossRef]
- Diarra, B.; Robini, M.; Tortoli, P.; Cachard, C.; Liebgott, H. Design of Optimal 2-D Nongrid Sparse Arrays for Medical Ultrasound. IEEE Trans. Biomed. Eng. 2013, 60, 3093–3102. [Google Scholar] [CrossRef]
- Provost, J.; Papadacci, C.; Arango, J.E.; Imbault, M.; Fink, M.; Gennisson, J.-L.; Tanter, M.; Pernot, M. 3D ultrafast ultrasound imaging in vivo. Phys. Med. Biol. 2014, 59, L1–L13. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, M.F.; Christiansen, T.L.; Thomsen, E.V.; Jensen, J.A. 3-D imaging using row-column-addressed arrays with integrated apodization-part i: Apodization design and line element beamforming. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2015, 62, 947–958. [Google Scholar] [CrossRef] [PubMed]
- Ramalli, A.; Boni, E.; Savoia, A.S.; Tortoli, P. Density-tapered spiral arrays for ultrasound 3-D imaging. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2015, 62, 1580–1588. [Google Scholar] [CrossRef] [PubMed]
- Pappalardo, M.; Caliano, G.; Caronti, A.; Savoia, A.S.; Gatta, P.; Longo, C.; Bavaro, V. Bidimensional Ultrasonic Array for Volumetric Imaging. U.S. Patent Application WO2008083876A3, 3 June 2010. [Google Scholar]
- Holbek, S.; Christiansen, T.L.; Stuart, M.B.; Beers, C.; Thomsen, E.V.; Jensen, J.A. 3-D Vector Flow Estimation With Row–Column-Addressed Arrays. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2016, 63, 1799–1814. [Google Scholar] [CrossRef] [PubMed]
- Holbek, S.; Hansen, K.L.; Bouzari, H.; Ewertsen, C.; Stuart, M.B.; Thomsen, C.; Nielsen, M.B.; Jensen, J.A. Common Carotid Artery Flow Measured by 3-D Ultrasonic Vector Flow Imaging and Validated with Magnetic Resonance Imaging. Ultrasound Med. Biol. 2017, 43, 2213–2220. [Google Scholar] [CrossRef]
- Correia, M.; Provost, J.; Tanter, M.; Pernot, M. 4D ultrafast ultrasound flow imaging: In vivo quantification of arterial volumetric flow rate in a single heartbeat. Phys. Med. Biol. 2016, 61, L48. [Google Scholar] [CrossRef]
- Wigen, M.S.; Fadnes, S.; Rodriguez-Molares, A.; Bjåstad, T.; Eriksen, M.; Stensæth, K.H.; Støylen, A.; Lovstakken, L. 4-D Intracardiac Ultrasound Vector Flow Imaging–Feasibility and Comparison to Phase-Contrast MRI. IEEE Trans. Med Imaging 2018, 37, 2619–2629. [Google Scholar] [CrossRef]
- Voorneveld, J.; Saaid, H.; Schinkel, C.; Radeljic, N.; Lippe, B.; Gijsen, F.J.H.; van der Steen, A.F.W.; de Jong, N.; Claessens, T.; Vos, H.J.; et al. 4-D Echo-Particle Image Velocimetry in a Left Ventricular Phantom. Ultrasound Med. Biol. 2020, 46, 805–817. [Google Scholar] [CrossRef]
- Ramalli, A.; Harput, S.; Bezy, S.; Boni, E.; Eckersley, R.J.; Tortoli, P.; D’Hooge, J. High-Frame-Rate Tri-Plane Echocardiography With Spiral Arrays: From Simulation to Real-Time Implementation. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2020, 67, 57–69. [Google Scholar] [CrossRef]
- Harput, S.; Tortoli, P.; Eckersley, R.J.; Dunsby, C.; Tang, M.-X.; Christensen-Jeffries, K.; Ramalli, A.; Brown, J.; Zhu, J.; Zhang, G.; et al. 3-D Super-Resolution Ultrasound Imaging With a 2-D Sparse Array. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2020, 67, 269–277. [Google Scholar] [CrossRef]
- Jensen, J.A. FIELD: A Program for Simulating Ultrasound Systems. Med. Biol. Eng. Comput. 1996, 34, 351–353. [Google Scholar]
- Jensen, J.A.; Svendsen, N.B. Calculation of pressure fields from arbitrarily shaped, apodized, and excited ultrasound transducers. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 1992, 39, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Boni, E.; Bassi, L.; Dallai, A.; Meacci, V.; Ramalli, A.; Scaringella, M.; Guidi, F.; Ricci, S.; Tortoli, P. Architecture of an Ultrasound System for Continuous Real-Time High Frame Rate Imaging. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2017, 64, 1276–1284. [Google Scholar] [CrossRef] [PubMed]
- Savoia, A.S.; Matrone, G.; Bardelli, R.; Bellutti, P.; Quaglia, F.; Caliano, G.; Mazzanti, A.; Tortoli, P.; Mauti, B.; Fanni, L.; et al. A 256-Element Spiral CMUT Array with Integrated Analog Front End and Transmit Beamforming Circuits. In Proceedings of the 2018 IEEE International Ultrasonics Symposium (IUS), Kobe, Japan, 22–25 October 2018; IEEE: Kobe, Japan, 2018; pp. 206–212. [Google Scholar]
- Rossi, S.; Lenge, M.; Dallai, A.; Ramalli, A.; Boni, E. Toward the Real Time Implementation of the 2-D Frequency-Domain Vector Doppler Method. In Applications in Electronics Pervading Industry, Environment and Society; Saponara, S., De Gloria, A., Eds.; Lecture Notes in Electrical Engineering; Springer International Publishing: Cham, Switzerland, 2019; Volume 573, pp. 129–135. ISBN 978-3-030-11972-0. [Google Scholar]
- Roux, E.; Ramalli, A.; Tortoli, P.; Cachard, C.; Robini, M.C.; Liebgott, H. 2-D Ultrasound Sparse Arrays Multidepth Radiation Optimization Using Simulated Annealing and Spiral-Array Inspired Energy Functions. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2016, 63, 2138–2149. [Google Scholar] [CrossRef]
- Roux, E.; Varray, F.; Petrusca, L.; Cachard, C.; Tortoli, P.; Liebgott, H. Experimental 3-D Ultrasound Imaging with 2-D Sparse Arrays using Focused and Diverging Waves. Sci. Rep. 2018, 8, 9108. [Google Scholar] [CrossRef]
- Ramalli, A.; Boni, E.; Dallai, A.; Guidi, F.; Ricci, S.; Tortoli, P. Coded Spectral Doppler Imaging: From Simulation to Real-Time Processing. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 2016, 63, 1815–1824. [Google Scholar] [CrossRef]

| Imaging/Flow/Processing Parameters | Value |
|---|---|
| Central transmission (TX) frequency (f0) | 3.7 *–7 ** MHz |
| Pulse repetition frequency (PRF) | 500 Hz |
| Pixel step (δz × δx × δy) | (0.02 × 0.3 × 0.3) mm |
| Beam-to-flow angles (ϑD, ϑA) | [(90,0),(85,0),(80,0),(90,45),(85,45),(80,45)]° |
| Barycenter depth (z0) | 16 mm |
| Peak velocity () | 7.5 cm/s |
| No. of frequencies $ (kfm · kfn) | [3, 10] |
| z-axis bandwidth (Bz) | 0.74 *–1.4 ** MHz |
| Kernel size (MB × NB × PB) | (1.5 × 2.5 × 2.5) mm |
| CMUT Probe | |||||
| ϑD | ϑA | [%] | [%] | [%] | [%] |
| 90° | 0° | 1.1 ± 0.4 | 4.2 ± 5.3 | −1.4 ± 8.1 | 3.6 ± 5.6 |
| 85° | 0° | 1.1 ± 0.4 | 5.7 ± 5.6 | −0.8 ± 8.3 | 5.1 ± 5.9 |
| 80° | 0° | 1.6 ± 0.4 | 7.9 ± 7.3 | −1.1 ± 8.7 | 7 ± 6.4 |
| 90° | 45° | 1 ± 0.7 | 4.7 ± 10 | 3.6 ± 9.9 | 5.6 ± 10.4 |
| 85° | 45° | 1.2 ± 0.7 | 6.6 ± 9.5 | 5.3 ± 10.3 | 7.8 ± 9.5 |
| 80° | 45° | 1.9 ± 0.7 | 8.5 ± 11.4 | 6.6 ± 11.2 | 9.6 ± 10 |
| Mean of [%] | 6.5 ± 8.0 | ||||
| Vermon Probe | |||||
| ϑD | ϑA | [%] | [%] | [%] | [%] |
| 90° | 0° | 0.7 ± 0.8 | 14 ± 5.9 | −0.1 ± 8.2 | 13.4 ± 5.9 |
| 85° | 0° | 0.8 ± 0.8 | 14.5 ± 5.8 | −0.1 ± 8.7 | 13.7 ± 5.8 |
| 80° | 0° | 1.3 ± 0.9 | 14 ± 7.3 | −0.1 ± 10.1 | 12.6 ± 6.3 |
| 90° | 45° | 0.6 ± 1.2 | 10.8 ± 11.3 | 10.8 ± 10.3 | 15.1 ± 12 |
| 85° | 45° | 0.7 ± 1.2 | 11.4 ± 11.3 | 11.4 ± 11 | 15.6 ± 11.4 |
| 80° | 45° | 1.3 ± 1.2 | 12.5 ± 12.6 | 12.4 ± 12 | 16.3 ± 10.9 |
| Mean of [%] | 14.4 ± 8.7 | ||||
| Vermon Probe | |||||
|---|---|---|---|---|---|
| ϑD | ϑA | [%] | [%] | [%] | [%] |
| 91° | 0° | 0.6 ± 1.3 | 11 ± 11.8 | −2 ± 12.6 | 10.2 ± 9.5 |
| 85° | 0° | 1.1 ± 1.4 | 10.5 ± 13.3 | −2 ± 12.5 | 9.8 ± 10.6 |
| 80° | 0° | 1.4 ± 1.5 | 9.8 ± 14.2 | −2.7 ± 14.1 | 8.8 ± 11.4 |
| 91° | 44° | 0.6 ± 1.4 | 9.3 ± 13.7 | 10.1 ± 13.7 | 13.3 ± 11.6 |
| 85° | 45° | 1.3 ± 1.2 | 8.7 ± 13.2 | 9.6 ± 12.9 | 12.6 ± 10.8 |
| 81° | 43° | 1.9 ± 1.4 | 11.4 ± 14.1 | 10.5 ± 13.6 | 14.6 ± 11.4 |
| Mean of [%] | 11.5 ± 10.9 | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossi, S.; Ramalli, A.; Fool, F.; Tortoli, P. High-Frame-Rate 3-D Vector Flow Imaging in the Frequency Domain. Appl. Sci. 2020, 10, 5365. https://doi.org/10.3390/app10155365
Rossi S, Ramalli A, Fool F, Tortoli P. High-Frame-Rate 3-D Vector Flow Imaging in the Frequency Domain. Applied Sciences. 2020; 10(15):5365. https://doi.org/10.3390/app10155365
Chicago/Turabian StyleRossi, Stefano, Alessandro Ramalli, Fabian Fool, and Piero Tortoli. 2020. "High-Frame-Rate 3-D Vector Flow Imaging in the Frequency Domain" Applied Sciences 10, no. 15: 5365. https://doi.org/10.3390/app10155365
APA StyleRossi, S., Ramalli, A., Fool, F., & Tortoli, P. (2020). High-Frame-Rate 3-D Vector Flow Imaging in the Frequency Domain. Applied Sciences, 10(15), 5365. https://doi.org/10.3390/app10155365








