Process of Formaldehyde and Volatile Organic Compounds’ Removal fromWaste Gases
Abstract
1. Introduction
2. Production Processes of Formaldehyde
2.1. Formaldehyde Production Using Metal Oxide Catalyst Processes
2.2. Formaldehyde Production Using Silver Catalyst Processes
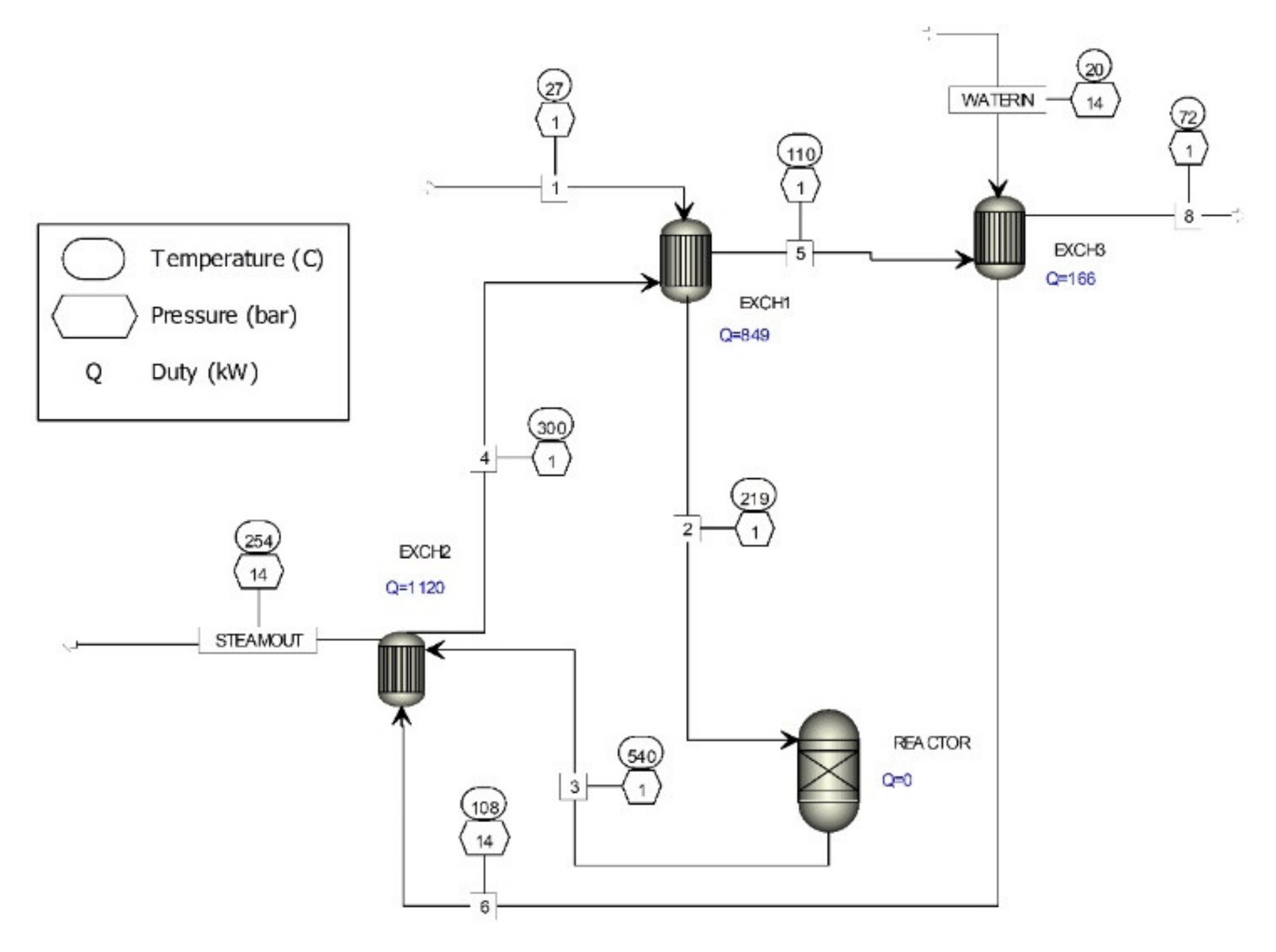
3. Off-Gas Purification Process
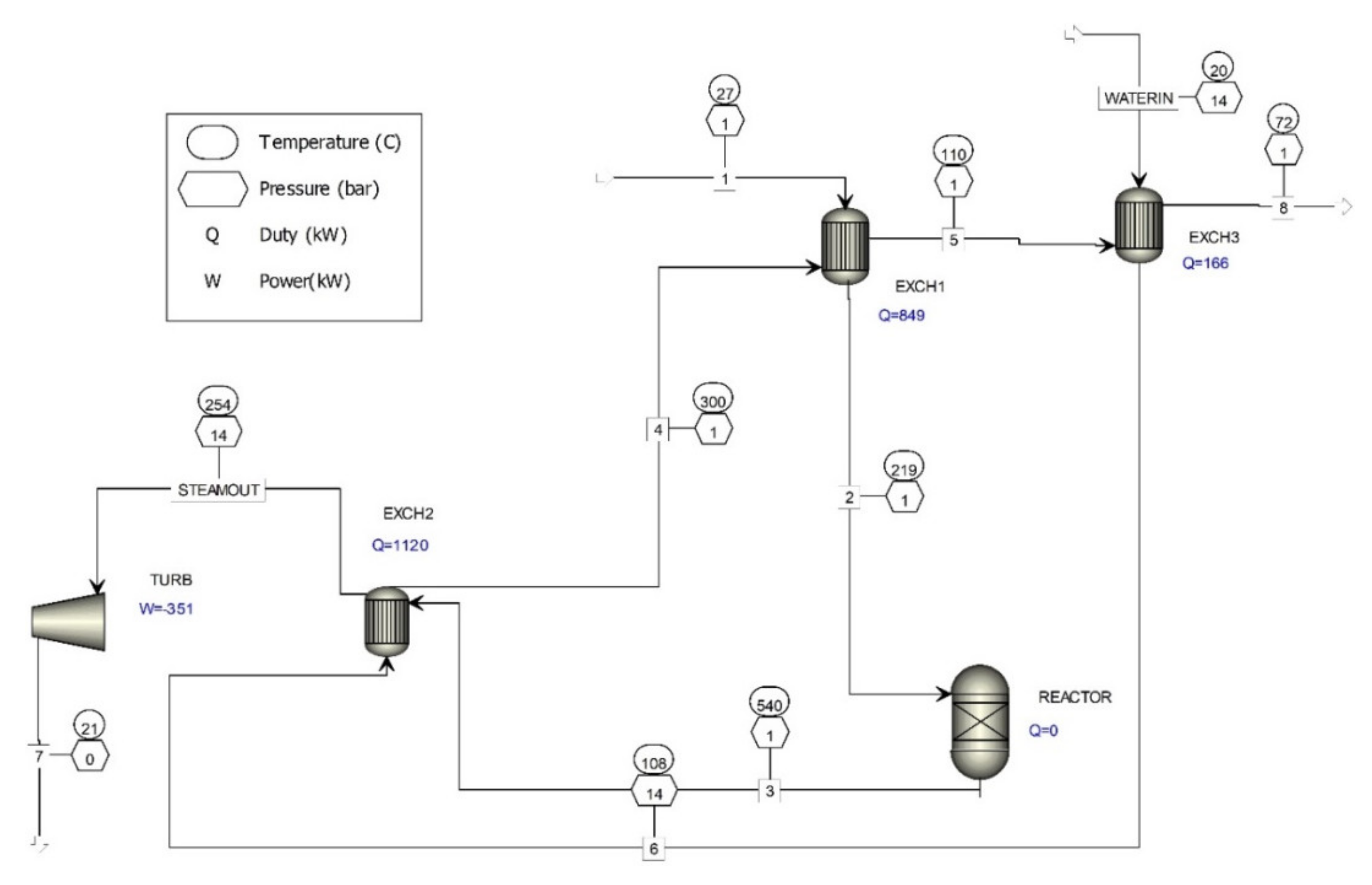
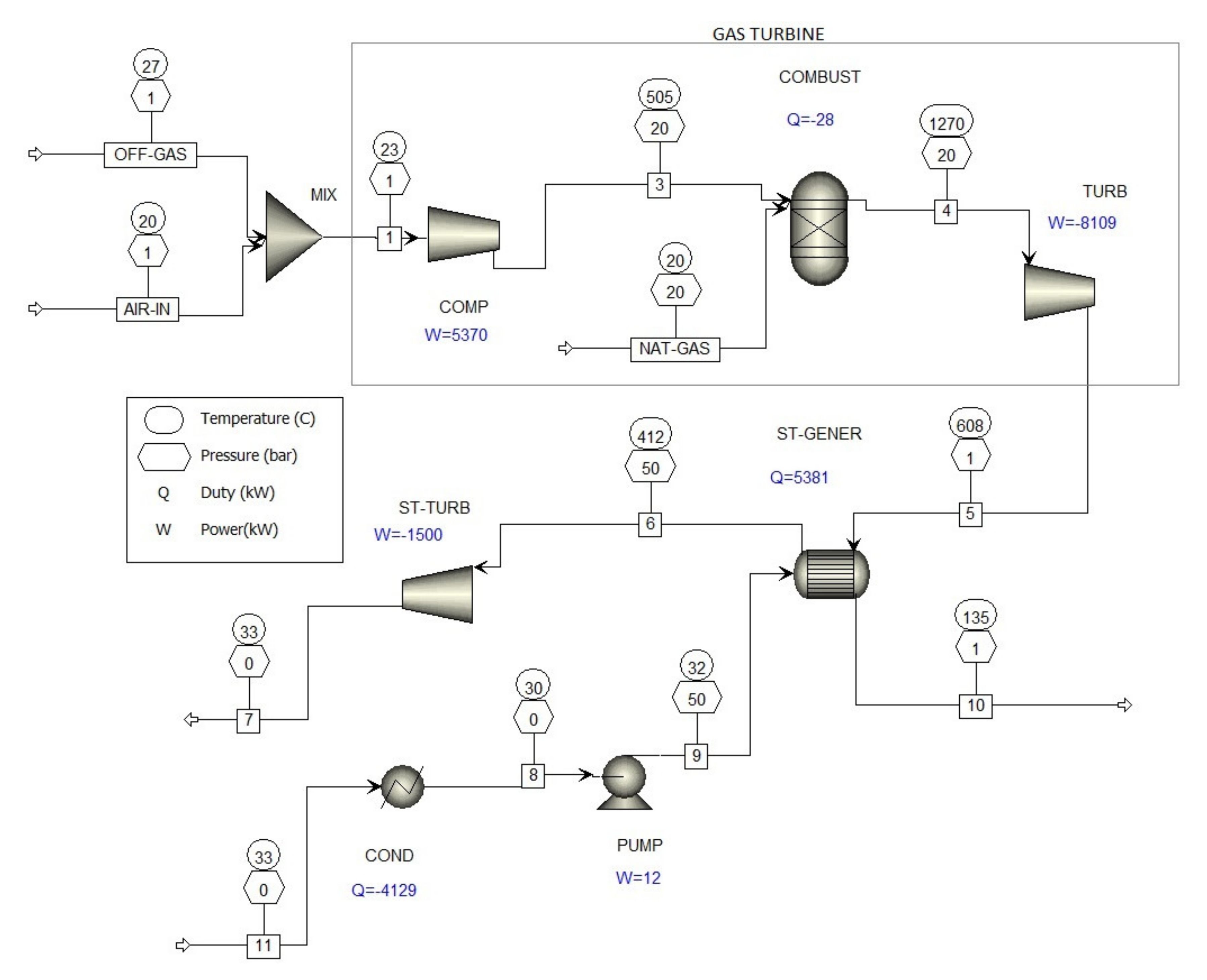
Energy Production
4. Upgrading the Waste Gas Purification Process
4.1. EU Directives on Industrial Emissions
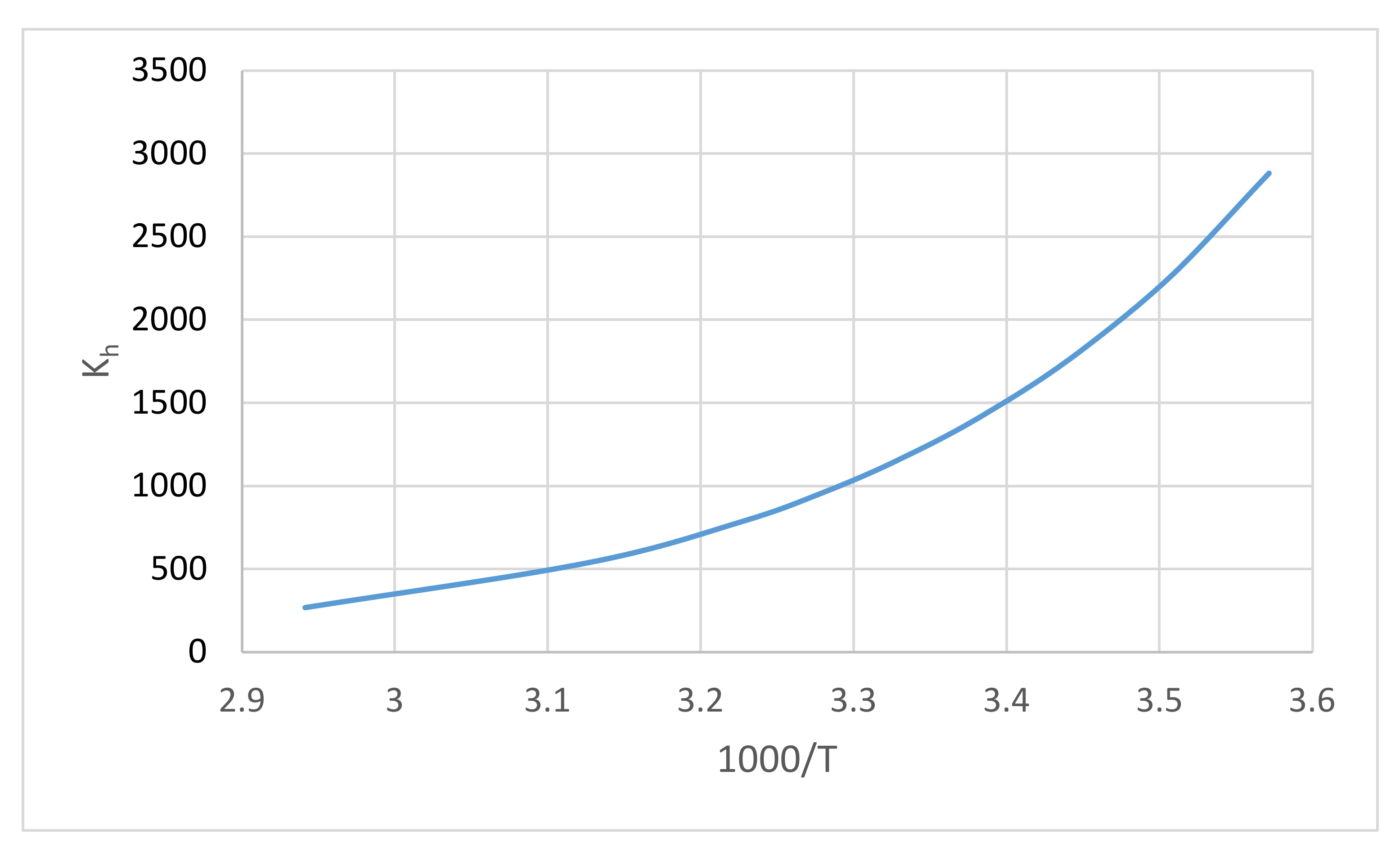
4.2. Kinetics and Chemical Equilibrium in the Hydration of Formaldehyde
- Vapor pressure of pure components, water, formaldehyde, and methylene glycol;
- Constants of the chemical reaction equilibrium for the formation of methylene glycol in the vapor phase and the formation of polyoxymethylene glycols in the liquid phase; and
- UNIFAC parameters for size and surface, and binary parameters for interactions between all groups.
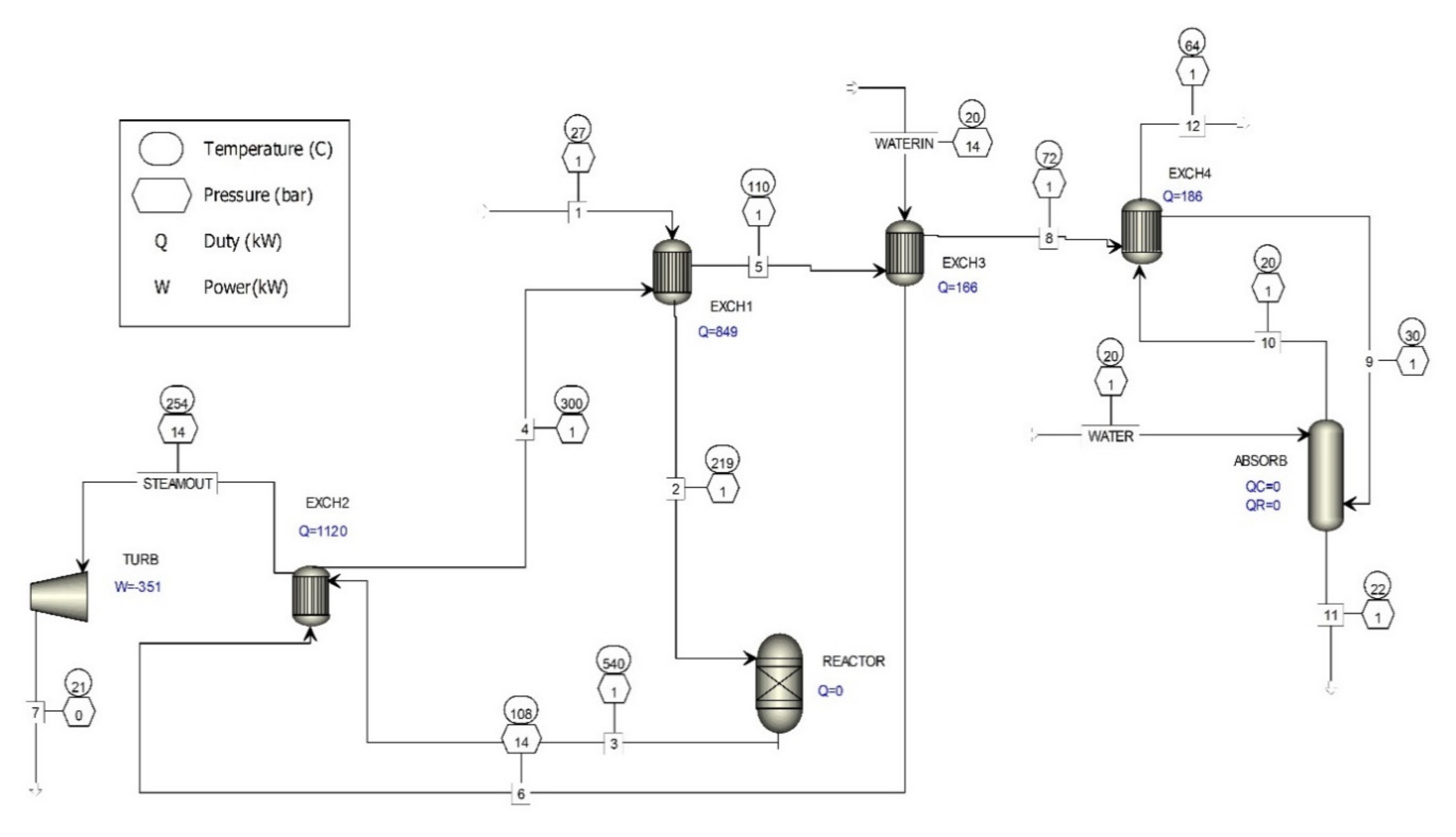
4.3. The Waste Gas Purification Process with Absorbers
5. Co-Incineration of Waste Industrial Gases
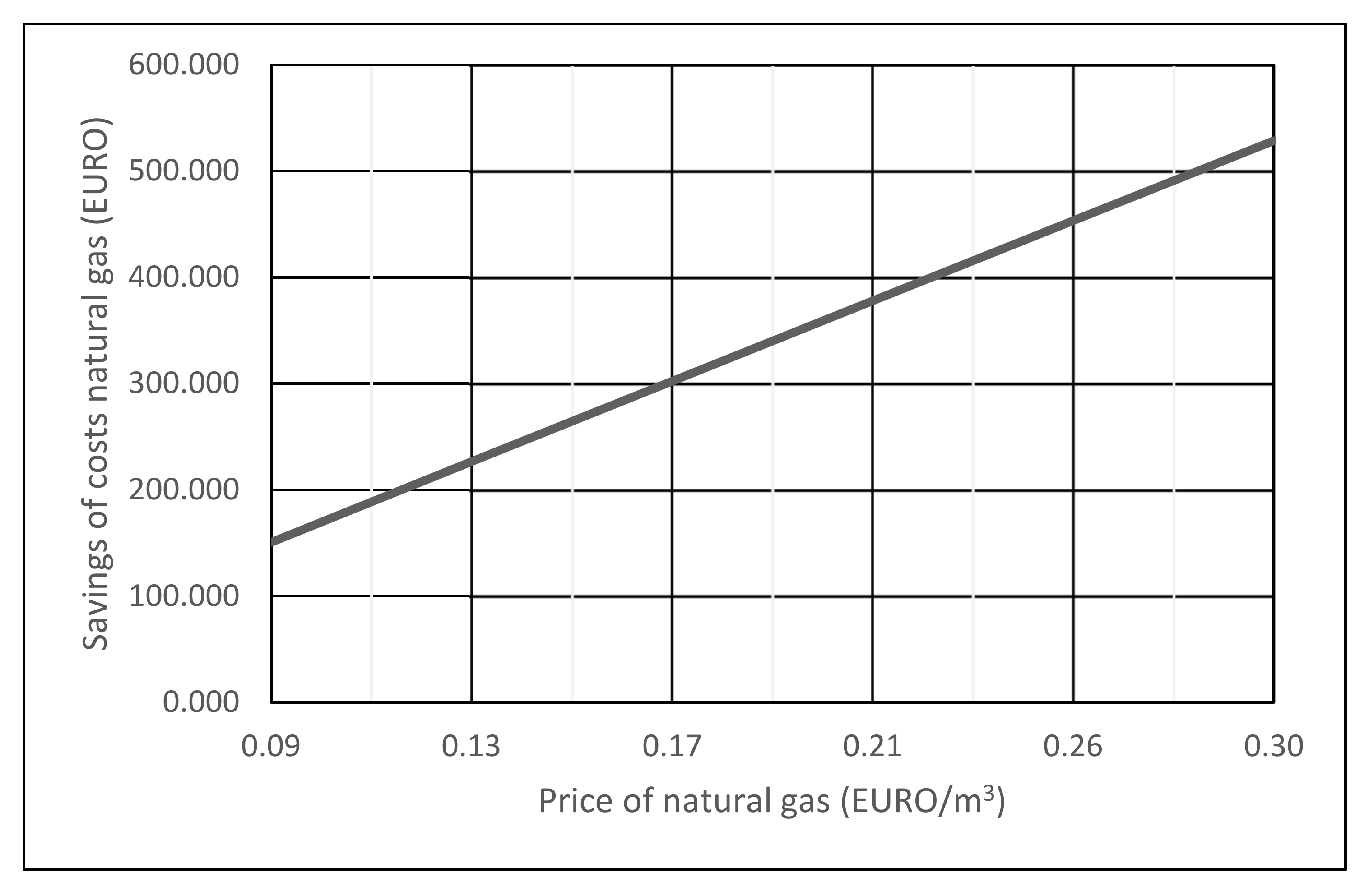
6. Economics of Co-Incineration of Waste Gases
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| K | mass-transfer coefficient, m/s |
| Kh | chemical equilibrium constant of the formaldehyde hydration, dimensionless number |
| kh | reaction rate constant of hydration, s−1 |
| kd | reaction rate constant of dehydration, s−1 |
| LP | liquid phase |
| m | equilibrium constant in gas–liquid system |
| ps | pure component vapor pressure, kPa |
| VP | vapor phase |
| βg | mass-transfer coefficients in a gas phase, m/s |
| βl | mass-transfer coefficients in a liquid phase, m/s |
References
- Walker, J.F. Formaldehyde, 3rd ed.; Robert. E. Krieger Publishing Co.: Huntington, NY, USA, 1975. [Google Scholar]
- Aspen, J.; Gao, S.; Hawari, Y.; Lam, E.; Larsson, T. Optimization of Formaldehyde Plant, Feasibility Studies KET050; Haldor Topsøes: Lyngby, Denmark, 2015. [Google Scholar]
- Shawabkeh, R. Production of Formaldehyde from Methanol; Integrated Final Report; King Fahd University of Petroleum & Minerals: Dhahran, Saudi Arabia, 2012. [Google Scholar]
- Bahmanpour, A.M.; Hoadley, A.; Tanksale, A. Critical review and exergy analysis of formaldehyde production processes. Rev. Chem. Eng. 2014. [Google Scholar] [CrossRef]
- Lunev, N.K.; Shmyrko, Y.I.; Pavlenko, N.V.; Norton, B. Selective formation of formaldehyde from carbon dioxide and hydrogen over PtCu/SiO2. Proc. Appl. Organomet. Chem. 2001, 15, 148–150. [Google Scholar]
- Qian, M.; Liauw, M.A.; Emig, G. Formaldehyde synthesis from methanol over silver catalysts. Appl. Catal. A Gen. 2003. [Google Scholar] [CrossRef]
- Millar, G.J.; Collins, M. Industrial Production of Formaldehyde Using Polycrystalline Silver Catalyst. Ind. Eng. Chem. Res. 2017, 56, 9247–9265. [Google Scholar] [CrossRef]
- Directive Council. European Commission Establishing Best Available Techniques (BAT) Conclusions, under Directive 2010/75/EU of the European Parliament and of the Council, for the production of large volume organic chemicals. Off. J. Eur. Union 2004, 334, 17–119. Available online: http://eur-lex.europa.eu/pri/en/oj/dat/2003/l_285/l_28520031101en00330037.pdf (accessed on 10 December 2019).
- Zhou, X.; Liu, Y.; Song, C.; Wang, X.; Wang, F.; Liu, J. A novel method to determine the formaldehyde emission characteristic parameters of building materials at multiple temperatures. Build. Environ. 2019. [Google Scholar] [CrossRef]
- Chen, W.; Mendell, M.; Li, N.; Kumagai, K. Formaldehyde emissions from seams and cut edges of laminate flooring: Implications for emission testing protocols and exposure estimation. Build. Environ. 2018. [Google Scholar] [CrossRef]
- Li, B.; Cheng, Z.; Yao, R.; Wang, H.; Yu, W.; Bu, Z.; Xiong, J.; Zhang, T.; Essah, E.; Luo, Z.; et al. An investigation of formaldehyde concentration in residences and the development of a model for the prediction of its emission rates. Build. Environ. 2019. [Google Scholar] [CrossRef]
- Kuramshina, K.S.; Pavlova, K.A. Physical and chemical analysis of formaldehyde absorption process. J. Eng. Appl. Sci. 2016, 11, 9655–9663. [Google Scholar]
- Winkelman, J.G.M.; Voorwinde, O.K.; Ottens, M.; Beenackers, A.A.C.M.; Janssen, L.P.B.M. Kinetics and chemical equilibrium of the hydration of formaldehyde. Chem. Eng. Sci. 2002. [Google Scholar] [CrossRef]
- Albert, M.; Garcia, B.G.; Kreiter, C.; Maurer, G. Vapor-liquid and chemical equilibria of formaldehyde-water mixtures. AIChE J. 1999. [Google Scholar] [CrossRef]
- Maurer, G. Vapor-Liquid Equilibrium of Formaldehyde- and Water-Containing Multicomponent Mixtures. AIChE J. 1986, 32, 932–948. [Google Scholar] [CrossRef]
- Hahnenstein, I.; Albert, M.; Hasse, H.; Kreiter, C.G.; Maurer, G. NMR Spectroscopic and Densimetric Study of Reaction Kinetics of Formaldehyde Polymer Formation in Water, Deuterium Oxide, and Methanol. Ind. Eng. Chem. Res. 1995. [Google Scholar] [CrossRef]
- Hahnenstein, I.; Hasse, H.; Kreiter, C.G.; Maurer, G. 1H- and 13C-NMR-Spectroscopic Study of Chemical Equilibria in Solutions of Formaldehyde in Water, Deuterium Oxide, and Methanol. Ind. Eng. Chem. Res. 1994. [Google Scholar] [CrossRef]
- Hahnenstein, I.; Hasse, H.; Liu, Y.Q.; Maurer, G. Thermodynamic Properties of Formaldehyde Containing Mixtures for Separation Process Design. In AIChE Symposium Series; American Institute of Chemical Engineers: New York, NY, USA, 1994; p. 141. [Google Scholar]
- Fredenslund, A. Vapor—Liquid Equilibria Using UNIFAC, a Group Contribution Method; Elsevier: Amsterdam, The Netherlands, 1977. [Google Scholar]
- Kogan, L.V. State of the Vapor Phase above Solutions of Formaldehyde in Water and Methanol. Zhur. Prikl. Khim. 1979, 52, 2576–2578. [Google Scholar]
- Winkelman, J.G.M.; Beenackers, A.A.C.M. Correlations for the density and viscosity of aqueous formaldehyde solutions. Ind. Eng. Chem. Res. 2000. [Google Scholar] [CrossRef]
- Seyedan, B.; Dhar, P.L.; Gaur, R.R.; Bindra, G.S. Computer simulation of a combined cycle power plant. Heat Recover. Syst. CHP 1995. [Google Scholar] [CrossRef]
- Colmenar-Santos, A.; Gómez-Camazón, D.; Rosales-Asensio, E.; Blanes-Peiró, J.J. Technological improvements in energetic efficiency and sustainability in existing combined-cycle gas turbine (CCGT) power plants. Appl. Energy 2018. [Google Scholar] [CrossRef]
| N | (A) | kg/hr | Weight% | N | (B) | kg/hr | Weight% |
|---|---|---|---|---|---|---|---|
| 1 | N2 | 13,492 | 89.4667 | 1 | N2 | 13,492 | 89.94667 |
| 2 | O2 | 916 | 6.10667 | 2 | O2 | 594 | 3.96000 |
| 3 | CO | 221 | 1.47333 | 3 | CO | 0.284 | 0.00189 |
| 4 | Dimethyl ether | 82 | 0.54667 | 4 | Dimethyl ether | 0.583 | 0.00389 |
| 5 | Formaldehyde | 3.5 | 0.02333 | 5 | Formaldehyde | 0.033 | 0.00022 |
| 6 | Methanol | 15.5 | 0.10333 | 6 | Methanol | 0.10 | 0.00067 |
| 7 | Water | 270 | 1.80000 | 7 | Water | 400 | 2.66667 |
| 8 | CO2 | 0 | 0% | 8 | CO2 | 513 | 3.42000 |
| 15,000 | 100 | 15,000 | 100 |
| Component | A | B | C |
|---|---|---|---|
| Formaldehyde | 4.4625 | −2204.13 | −30.15 |
| Water | 6.2886 | −3816.44 | −46.13 |
| Methylene glycol | 19.5527 | −6189.19 | −9.15 |
| Stream Name | Units | 8 | 9 | 10 | 11 | 12 | WATER |
|---|---|---|---|---|---|---|---|
| Phase | VP | VP | VP | LP | VP | LP | |
| Temperature | C | 72.30551 | 30 | 20.30396 | 21.85142 | 64.02441 | 20 |
| Pressure | bar | 1 | 1 | 1 | 1 | 1 | 1 |
| Mass Vapor Fraction | 1 | 1 | 0.999996 | 0 | 1 | 0 | |
| Mass Liquid Fraction | 0 | 0 | 4.33 × 10−6 | 1 | 0 | 1 | |
| Mass Flows | kg/hr | 15,000 | 15,000 | 14,641.68 | 80,358.31 | 14,641.68 | 80,000 |
| Water | kg/hr | 400.0001 | 399.9996 | 223.4809 | 80,176.42 | 223.4811 | 80,000 |
| CO2 | kg/hr | 513.0002 | 513.0002 | 441.0303 | 71.99611 | 441.0303 | 0 |
| N2 | kg/hr | 13,492.01 | 13,492.01 | 13,388.47 | 103.5946 | 13,388.47 | 0 |
| Methanol | kg/hr | 0.1 | 0.1 | 6.52 × 10−6 | 0.099935 | 6.52 × 10−6 | 0 |
| Dimetyle | kg/hr | 0.58 | 0.58 | 0.022383 | 0.557619 | 0.022383 | 0 |
| CO | kg/hr | 0.28 | 0.28 | 0.277746 | 0.002256 | 0.277746 | 0 |
| O2 | kg/hr | 594.0003 | 594.0003 | 588.3943 | 5.609134 | 588.3943 | 0 |
| Formald | kg/hr | 0.032856 | 0.032041 | 0.005188 | 0.026854 | 0.00557 | 0 |
| Methylen | kg/hr | 0.00023 | 0.001533 | 0.000642 | 0.000891 | 3.17 × 10−6 | 0 |
| Volume Flow | m3/s | 4.26106 | 3.739237 | 3.515835 | 0.022417 | 4.039626 | 0.022271 |
| Stream Name | Units | 3 | 4 | 5 | 10 | 11 | OFF-GAS | AIR-IN | NAT-GAS |
|---|---|---|---|---|---|---|---|---|---|
| Phase | VP | VP | VP | VP | VP | VP | VP | VP | |
| Temperature | C | 504.91 | 1270 | 607.882 | 135 | 328.048 | 27 | 20 | 20 |
| Pressure | bar | 20 | 20 | 1.05 | 1.05 | 0.05 | 1.28 | 1 | 20 |
| Mass Vapor Fraction | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | |
| Mass Liquid Fraction | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Mass Flows | kg/hr | 35,772.3 | 36,349.8 | 36,349.8 | 36,349.8 | 6120 | 15,000 | 20,772.3 | 577.539 |
| Water | kg/hr | 270 | 1691.95 | 1691.95 | 1691.95 | 6120 | 270 | 0 | 0 |
| CO2 | kg/hr | 0 | 2215.27 | 2215.27 | 2215.27 | 0 | 0 | 0 | 0 |
| N2 | kg/hr | 29,426.1 | 29,426.1 | 29,426.1 | 29,426.1 | 0 | 13,492 | 15,934.1 | 0 |
| Methanol | kg/hr | 15.5 | 6.21 × 10−18 | 6.21 × 10−18 | 6.21 × 10−18 | 0 | 15.5 | 0 | 0 |
| Dimetyle | kg/hr | 82 | 1.42 × 10−40 | 1.42 × 10−40 | 1.42 × 10−40 | 0 | 82 | 0 | 0 |
| CO | kg/hr | 221 | 0.0109448 | 0.0109448 | 0.0109448 | 0 | 221 | 0 | 0 |
| O2 | kg/hr | 5754.22 | 3016.54 | 3016.54 | 3016.54 | 0 | 916 | 4838.22 | 0 |
| Formald | kg/hr | 3.5 | 3.42 × 10−9 | 3.42 × 10−9 | 3.42 × 10−9 | 0 | 3.5 | 0 | 0 |
| Methylen | kg/hr | 0 | 8.00 × 10−14 | 8.00 × 10−14 | 8.00 × 10−14 | 0 | 0 | 0 | 0 |
| Methane | kg/hr | 0 | 6.748.00 × 10−21 | 6.74 × 10−21 | 6.74 × 10−21 | 0 | 0 | 0 | 577.539 |
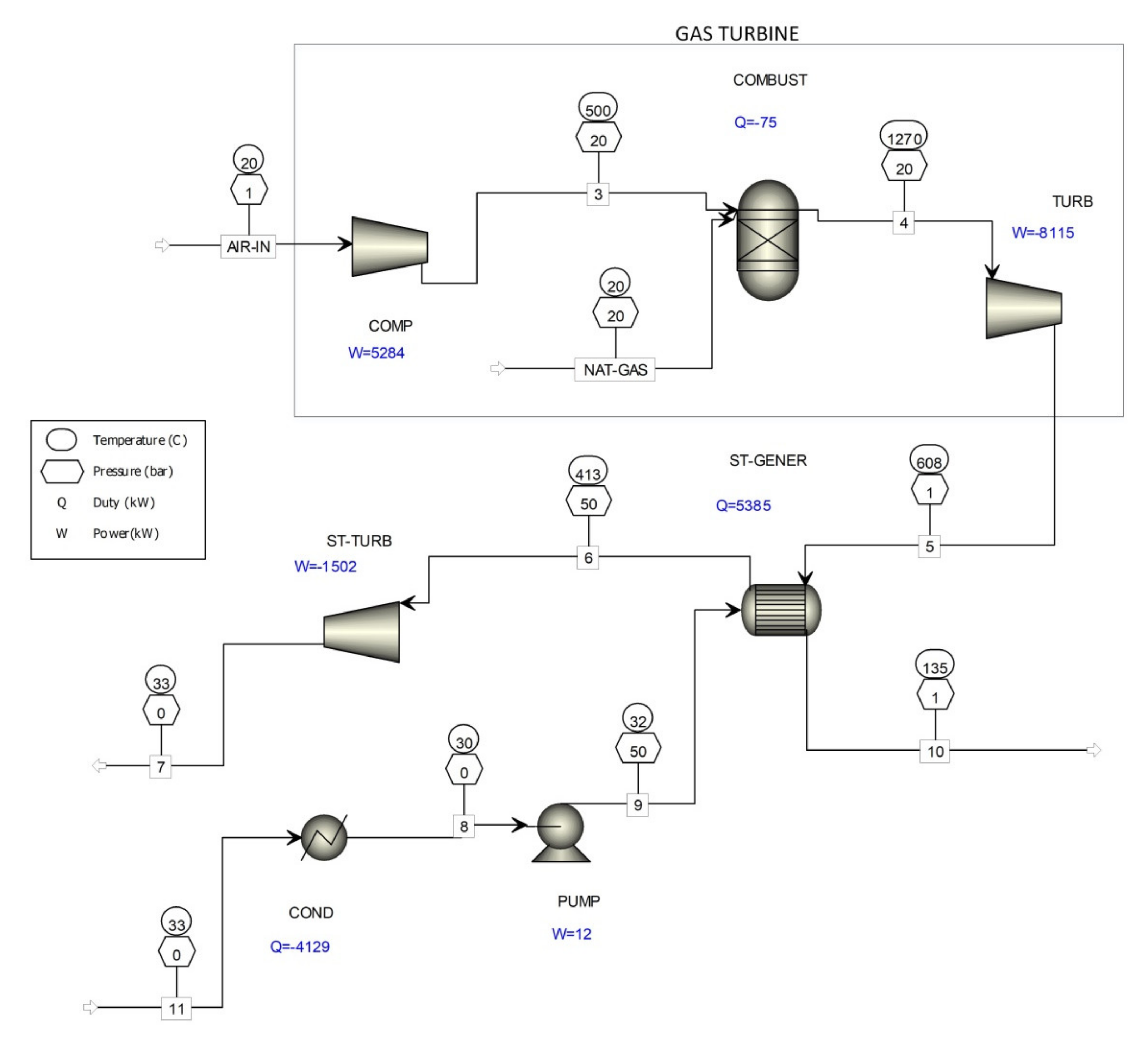
| Stream Name | Units | 3 | 4 | 5 | 10 | 11 | AIR-IN | NAT-GAS |
|---|---|---|---|---|---|---|---|---|
| Phase | VP | VP | VP | VP | VP | VP | VP | |
| Temperature | C | 499.72 | 1270 | 607.52 | 135 | 328.048 | 20 | 20 |
| Pressure | bar | 20 | 20 | 1.05 | 1.05 | 0.05 | 1 | 20 |
| Mass Vapor Fraction | 1 | 1 | 1 | 1 | 1 | 1 | 1 | |
| Mass Liquid Fraction | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Mass Flows | kg/hr | 35,936.1 | 36,652.2 | 36,652.2 | 36,652.2 | 6120 | 35,936.1 | 716.149 |
| Water | kg/hr | 0 | 1608.4 | 1608.4 | 1608.4 | 6120 | 0 | 0 |
| CO2 | kg/hr | 0 | 1964.6 | 1964.6 | 1964.6 | 0 | 0 | 0 |
| N2 | kg/hr | 27,565.9 | 27,565.9 | 27,565.9 | 27,565.9 | 0 | 27,565.9 | 0 |
| O2 | kg/hr | 8370.12 | 5513.27 | 5513.27 | 5513.27 | 0 | 8370.12 | 0 |
| Methane | kg/hr | 0 | 5.95 × 10−18 | 5.95 × 10−18 | 5.95 × 10−18 | 0 | 0 | 716.149 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mursics, J.; Urbancl, D.; Goricanec, D. Process of Formaldehyde and Volatile Organic Compounds’ Removal fromWaste Gases. Appl. Sci. 2020, 10, 4702. https://doi.org/10.3390/app10144702
Mursics J, Urbancl D, Goricanec D. Process of Formaldehyde and Volatile Organic Compounds’ Removal fromWaste Gases. Applied Sciences. 2020; 10(14):4702. https://doi.org/10.3390/app10144702
Chicago/Turabian StyleMursics, Jozsef, Danijela Urbancl, and Darko Goricanec. 2020. "Process of Formaldehyde and Volatile Organic Compounds’ Removal fromWaste Gases" Applied Sciences 10, no. 14: 4702. https://doi.org/10.3390/app10144702
APA StyleMursics, J., Urbancl, D., & Goricanec, D. (2020). Process of Formaldehyde and Volatile Organic Compounds’ Removal fromWaste Gases. Applied Sciences, 10(14), 4702. https://doi.org/10.3390/app10144702




