Nanofiltration of Simulated Acid Mine Drainage: Effect of pH and Membrane Charge
Abstract
1. Introduction
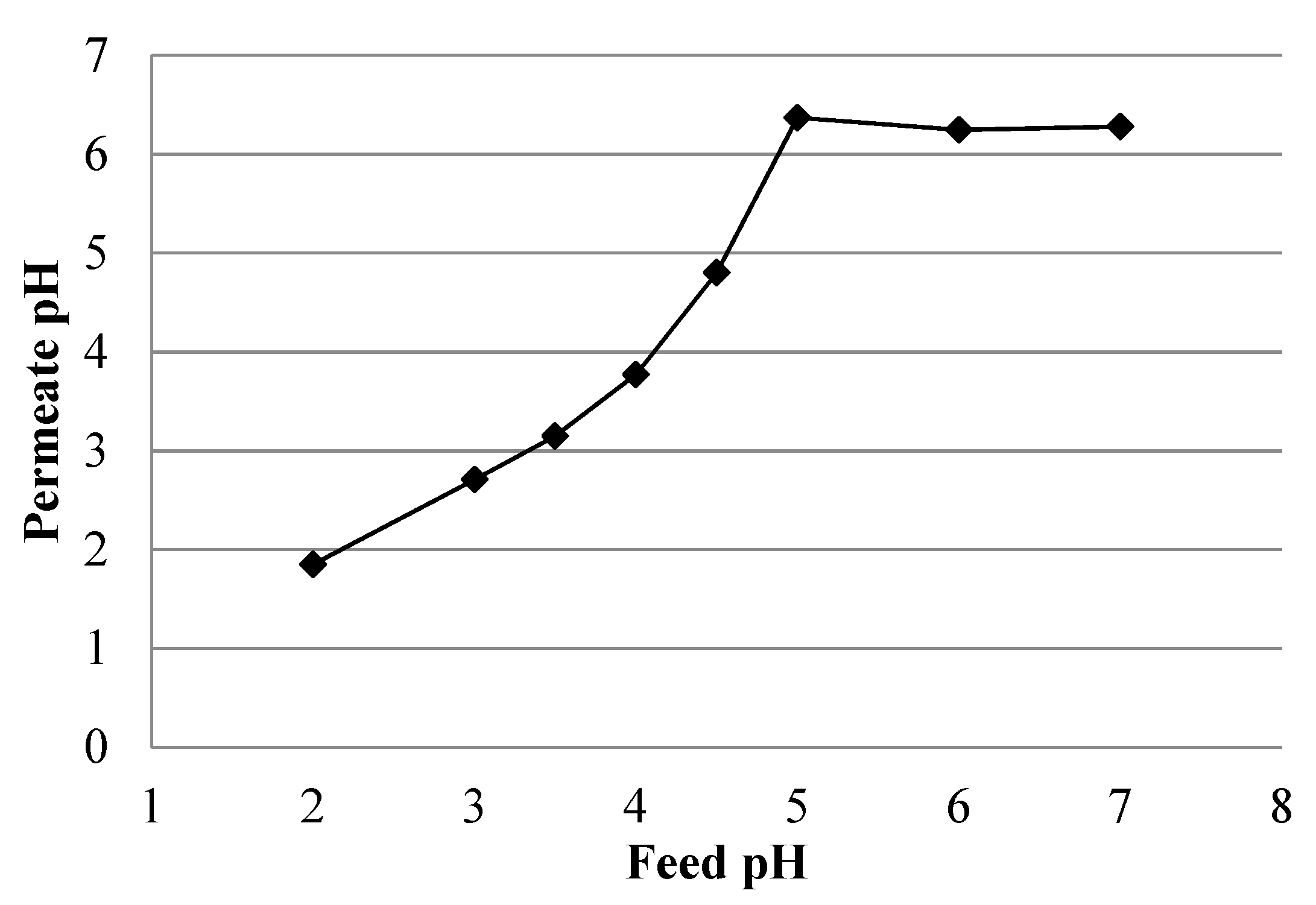
Effect of Feed Solution pH
2. Materials and Methods
2.1. Materials and Apparatus
2.2. Model AMD Solution Design
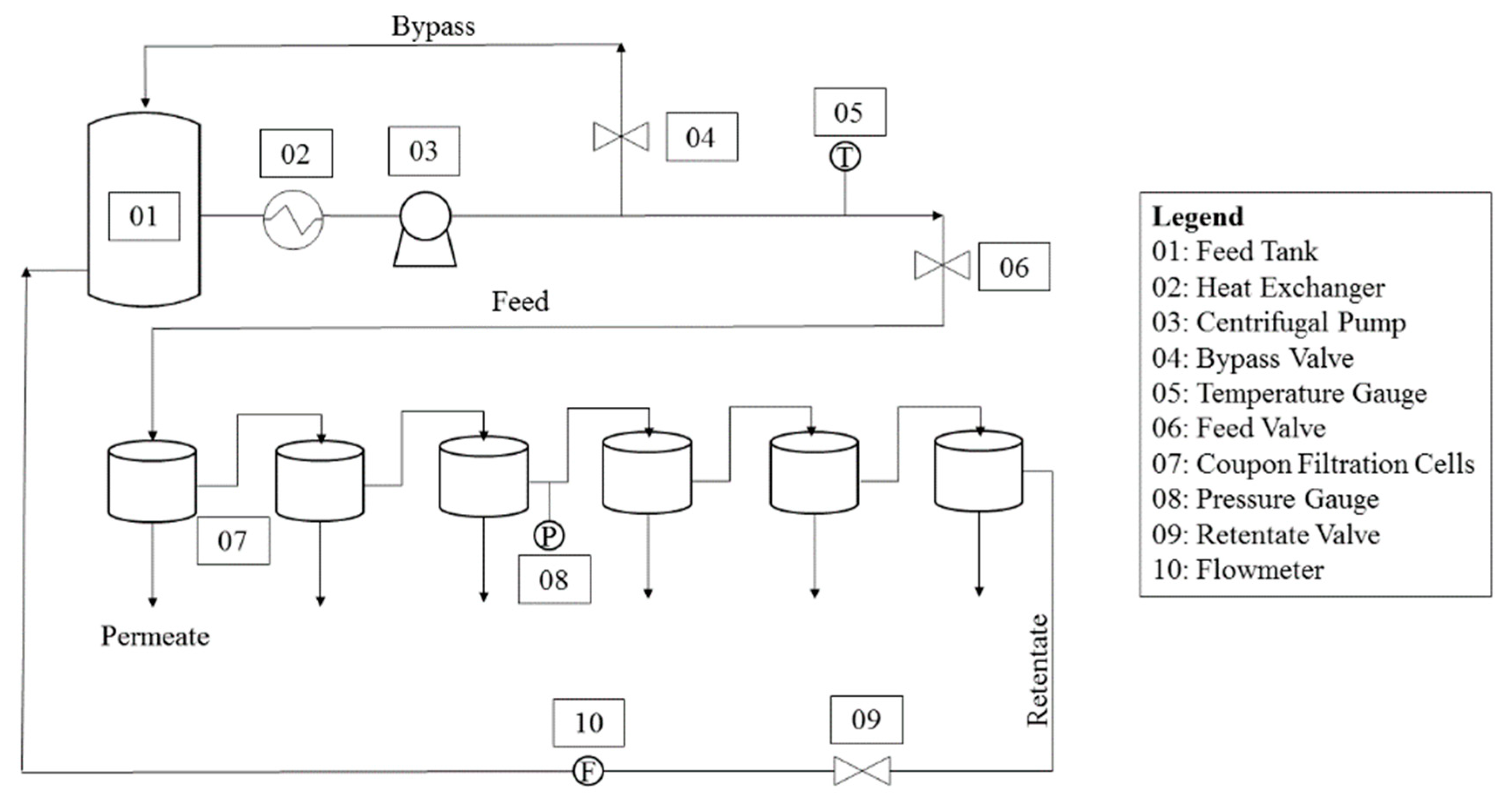
2.3. Membrane Filtration Experiments
3. Analytical methods
3.1. Conductivity—Concentration Calibration Curve
3.2. Spectrophotometer
3.3. Argentometric Titration
3.4. Ion Chromatography (IC)
3.5. Inductively Coupled Plasma-Optical Emission Spectroscopy (ICP-OES)
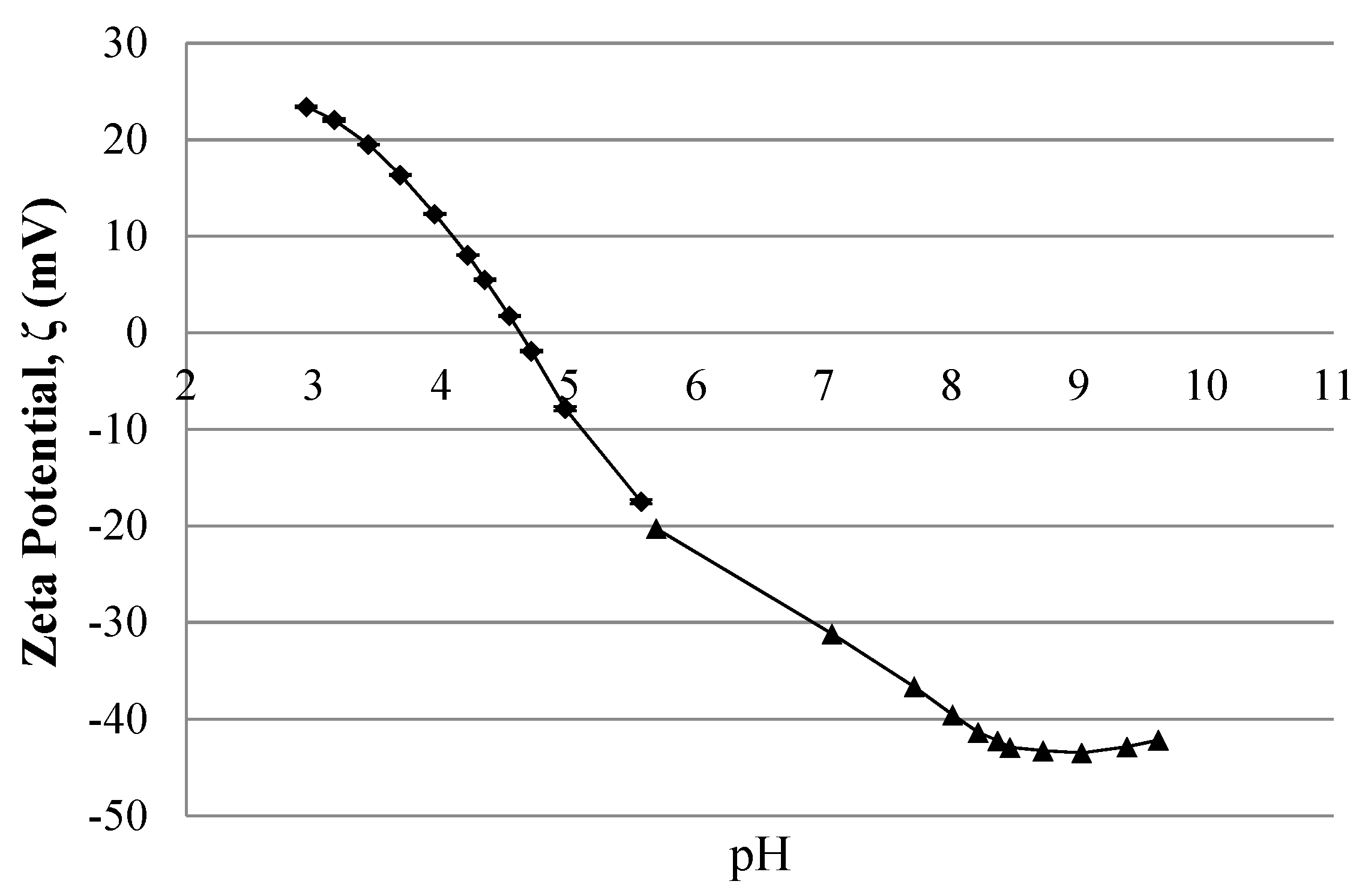
3.6. Electrokinetic Characterization
4. Results and Discussion
4.1. Streaming Potential Data
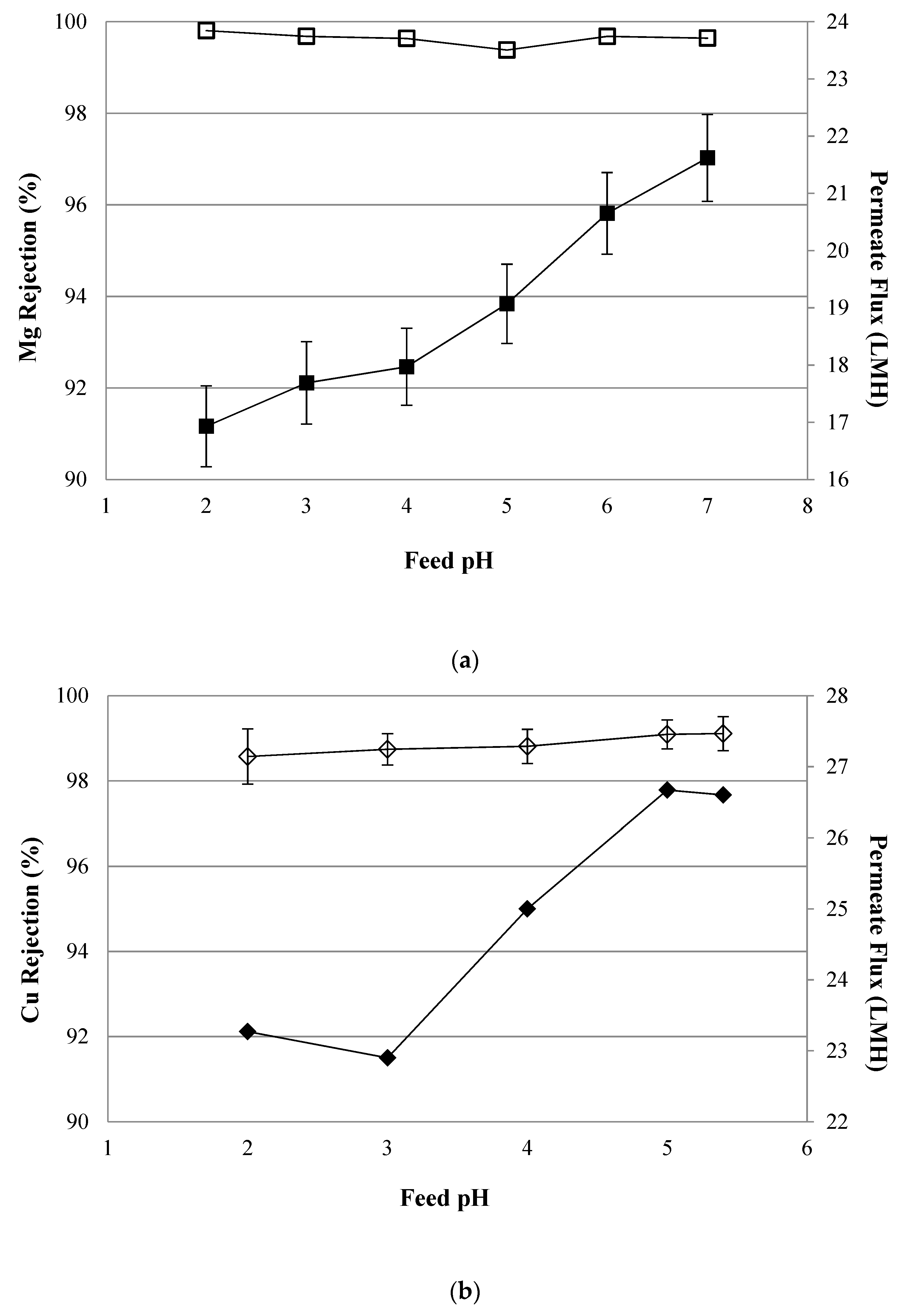
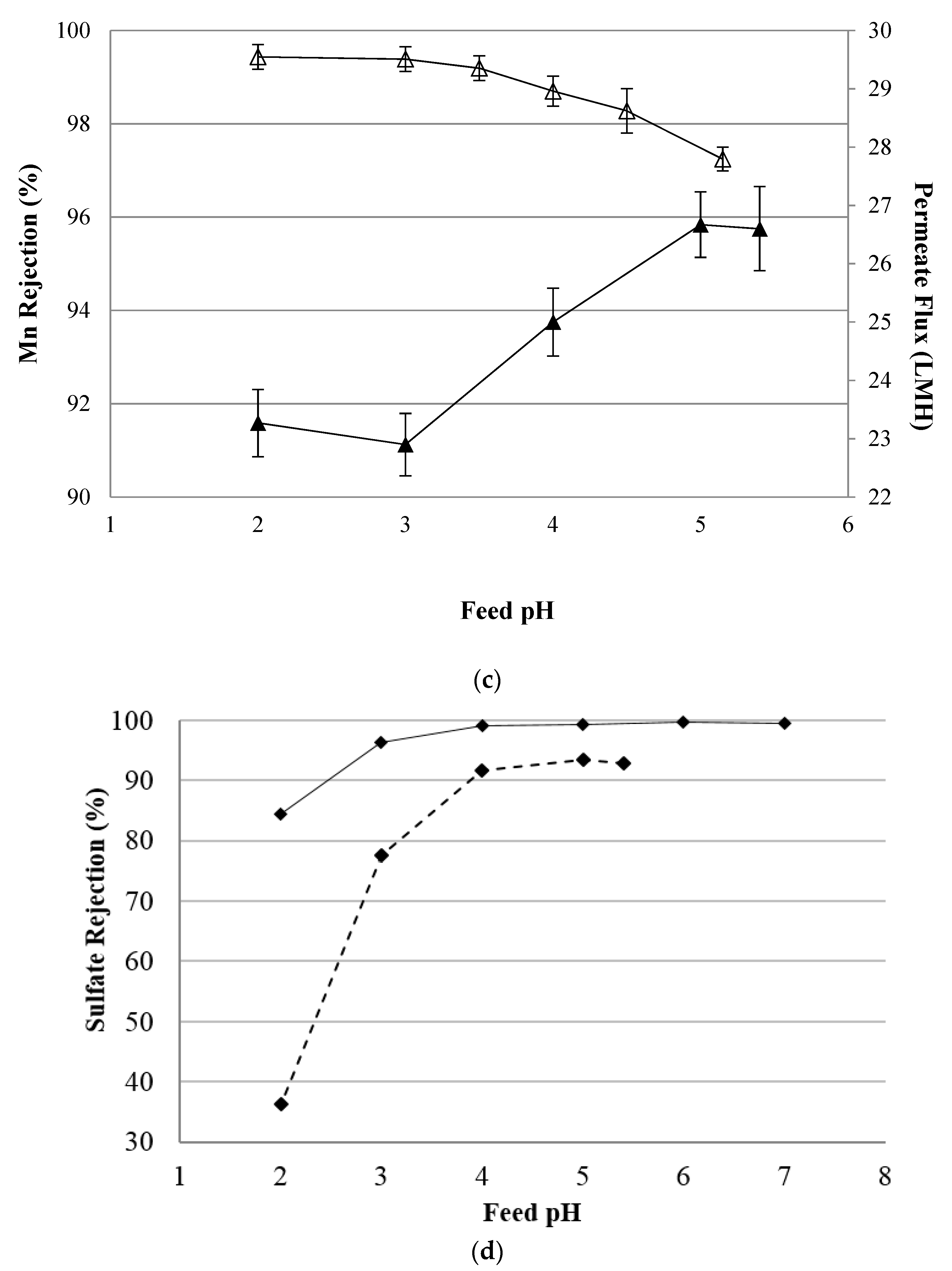
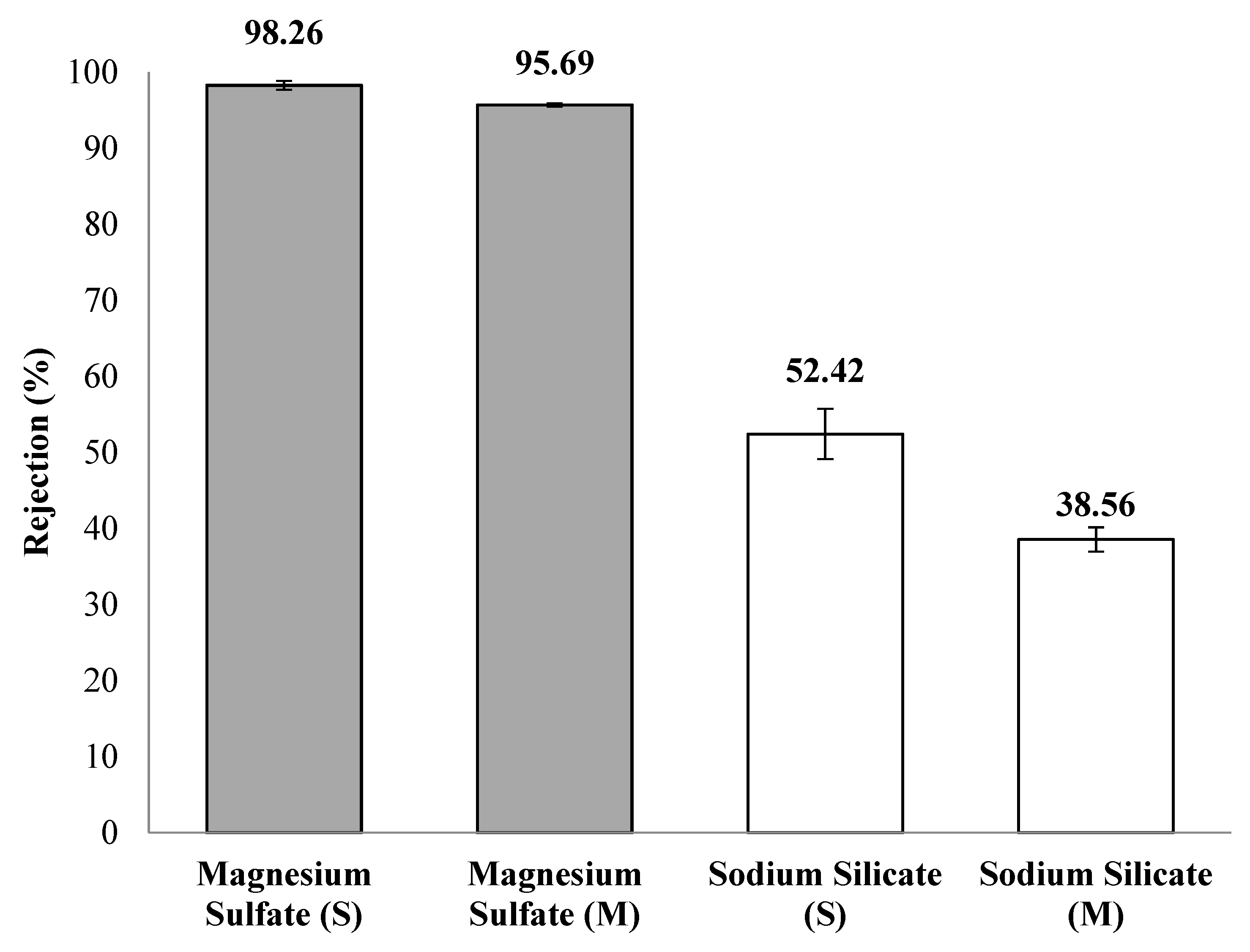
4.2. NF Performance in Single Salt Solutions
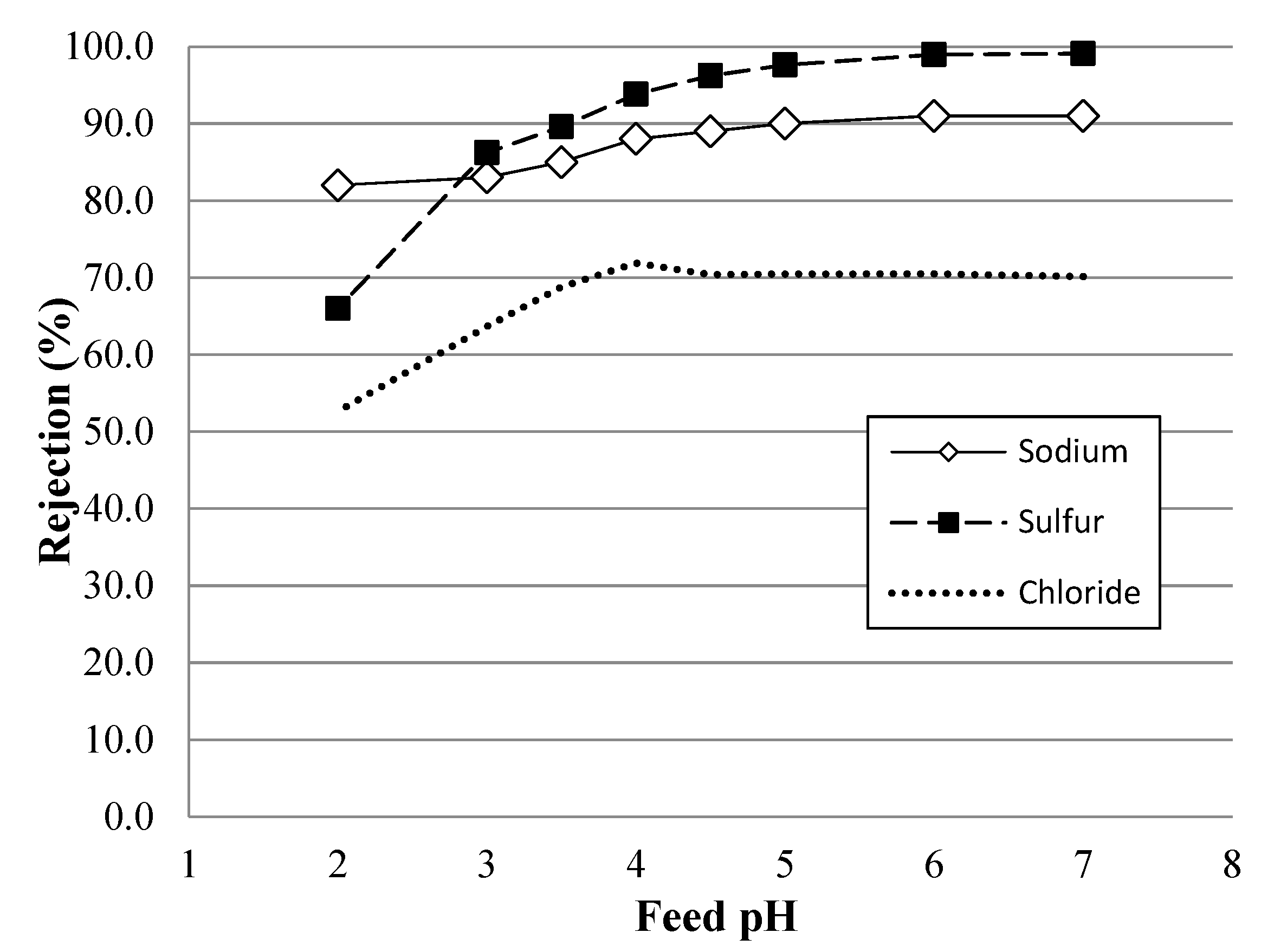
4.3. NF Performance in Mixed-Salt Solutions
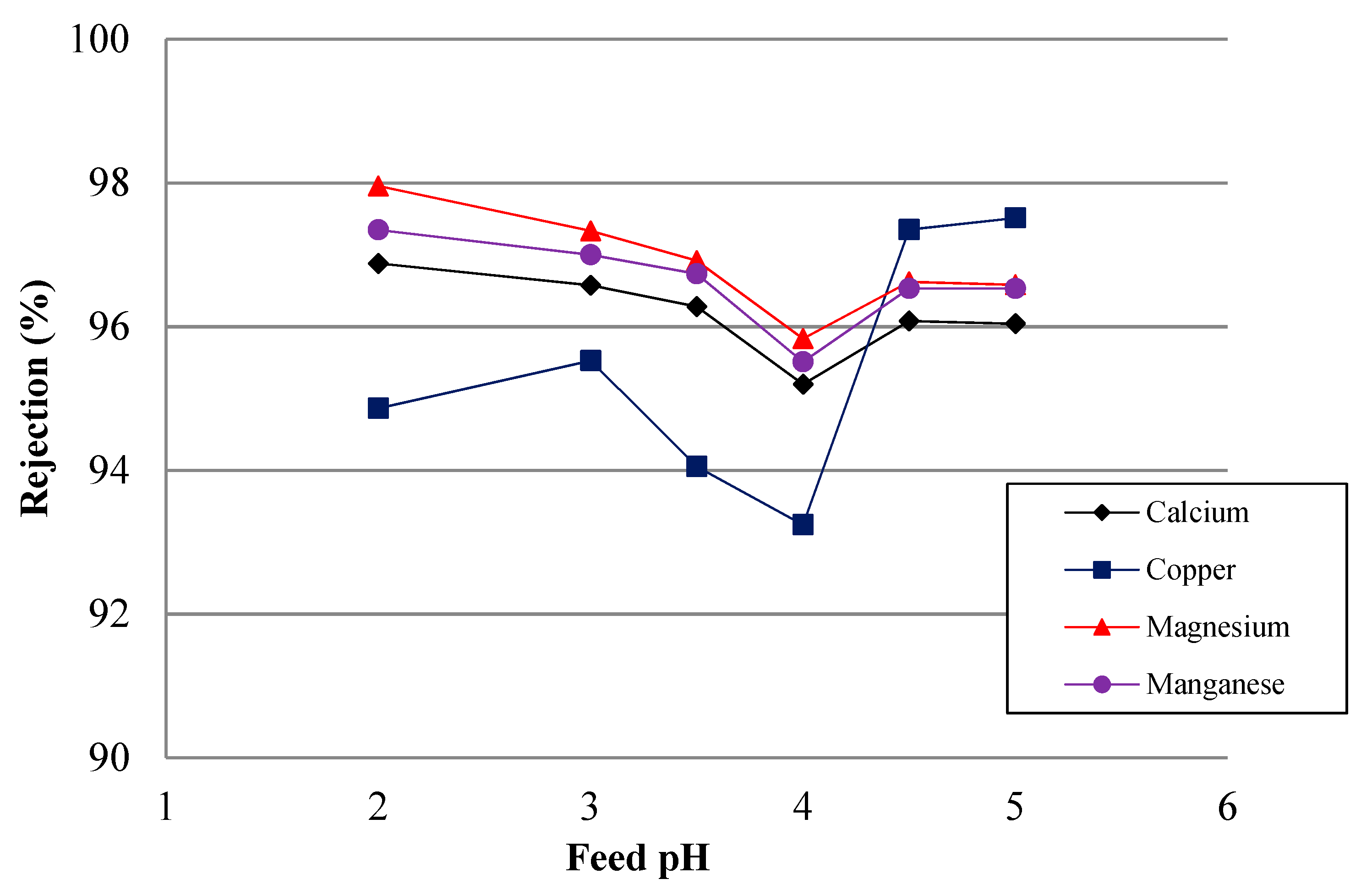
4.4. Simulated AMD Solutions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A. Supporting Information
Concentration Polarization
| Temperature (°C) | Feed Concentration (mg/L) | Feed Pressure (Bar) |
|---|---|---|
| 25 ± 0.5 | 2000 ± 100 of MgSO4 | 10 ± 0.1 |
| 8000 ± 200 of MgSO4 | 25 ± 0.1 |
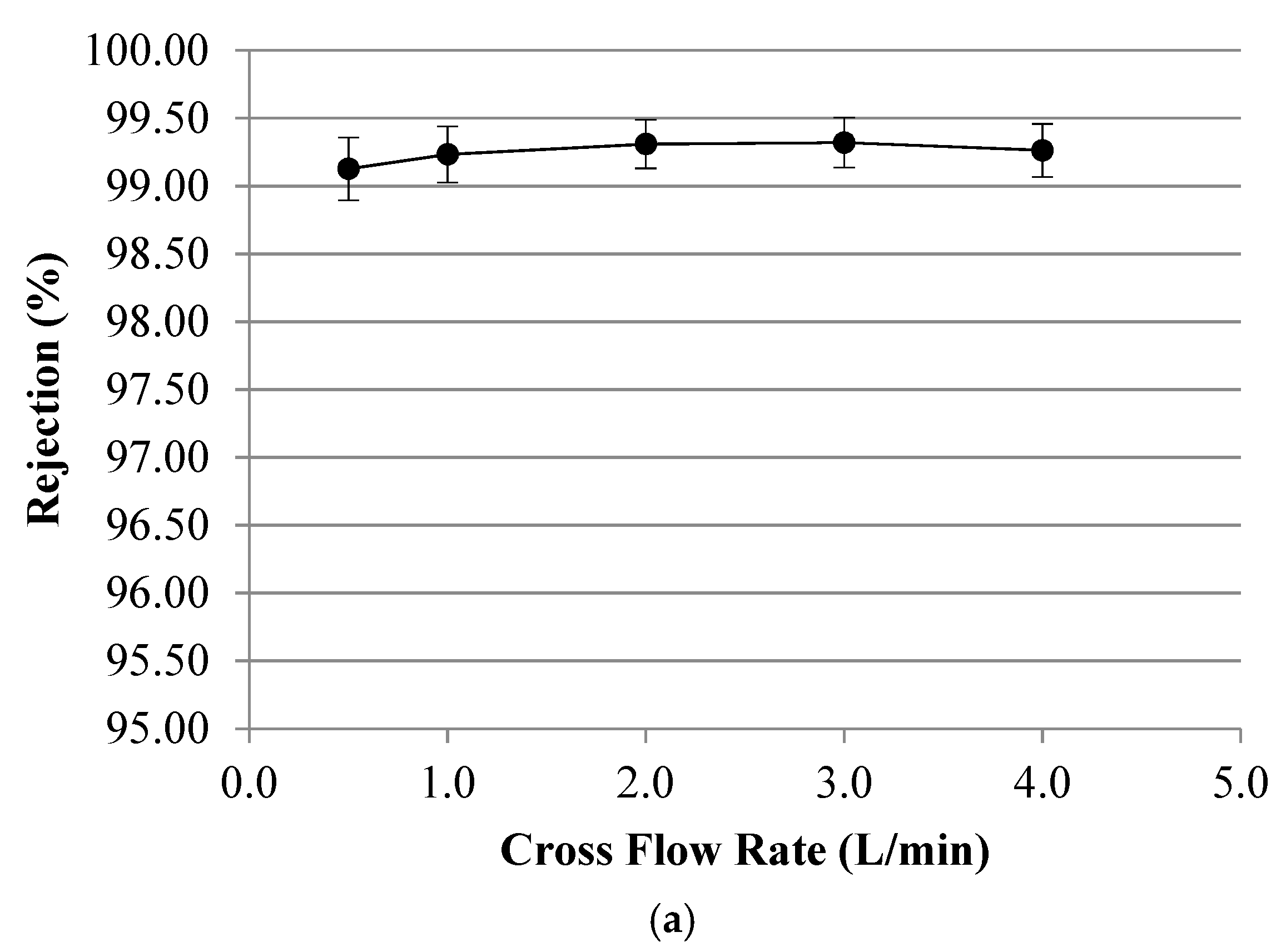
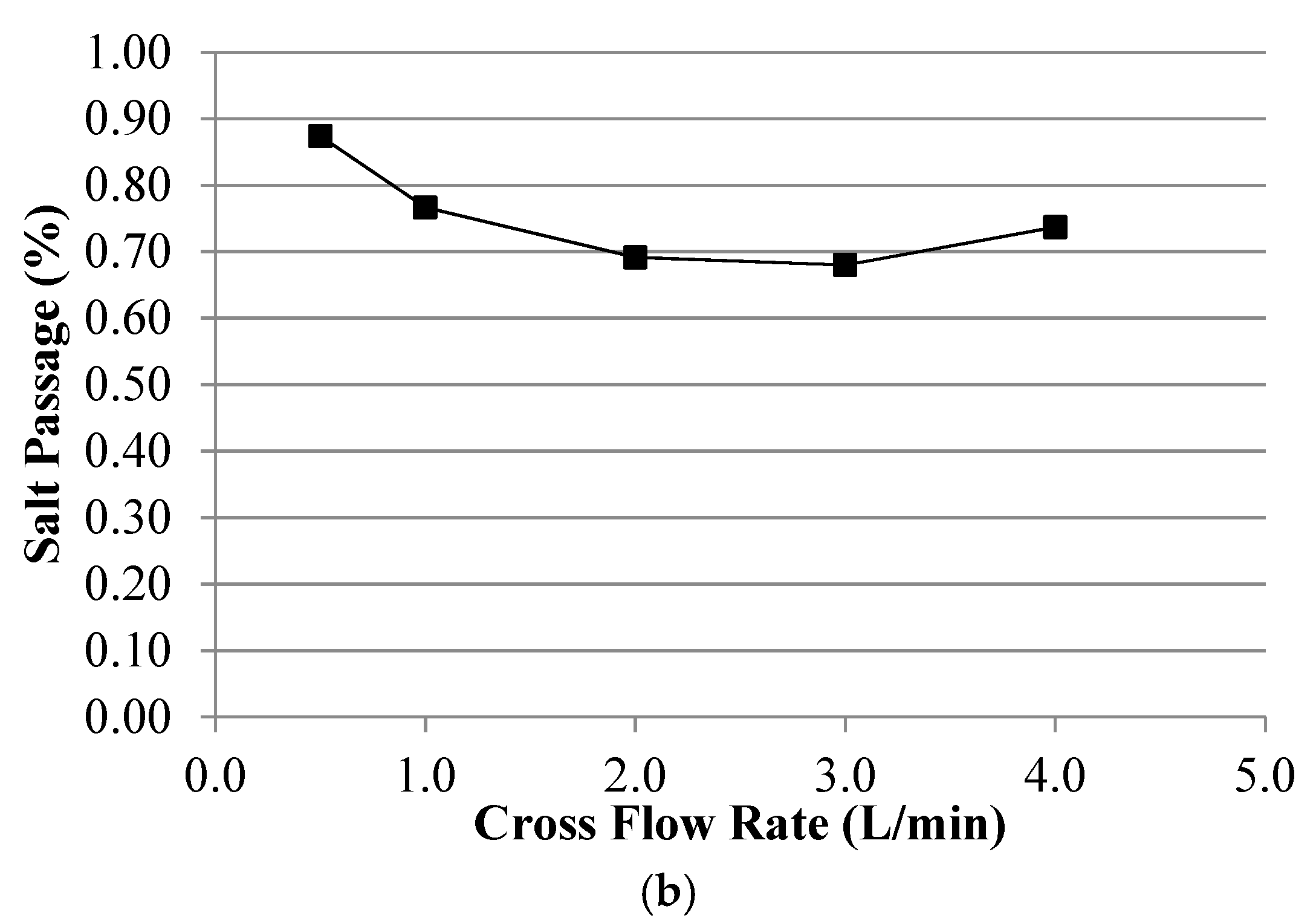
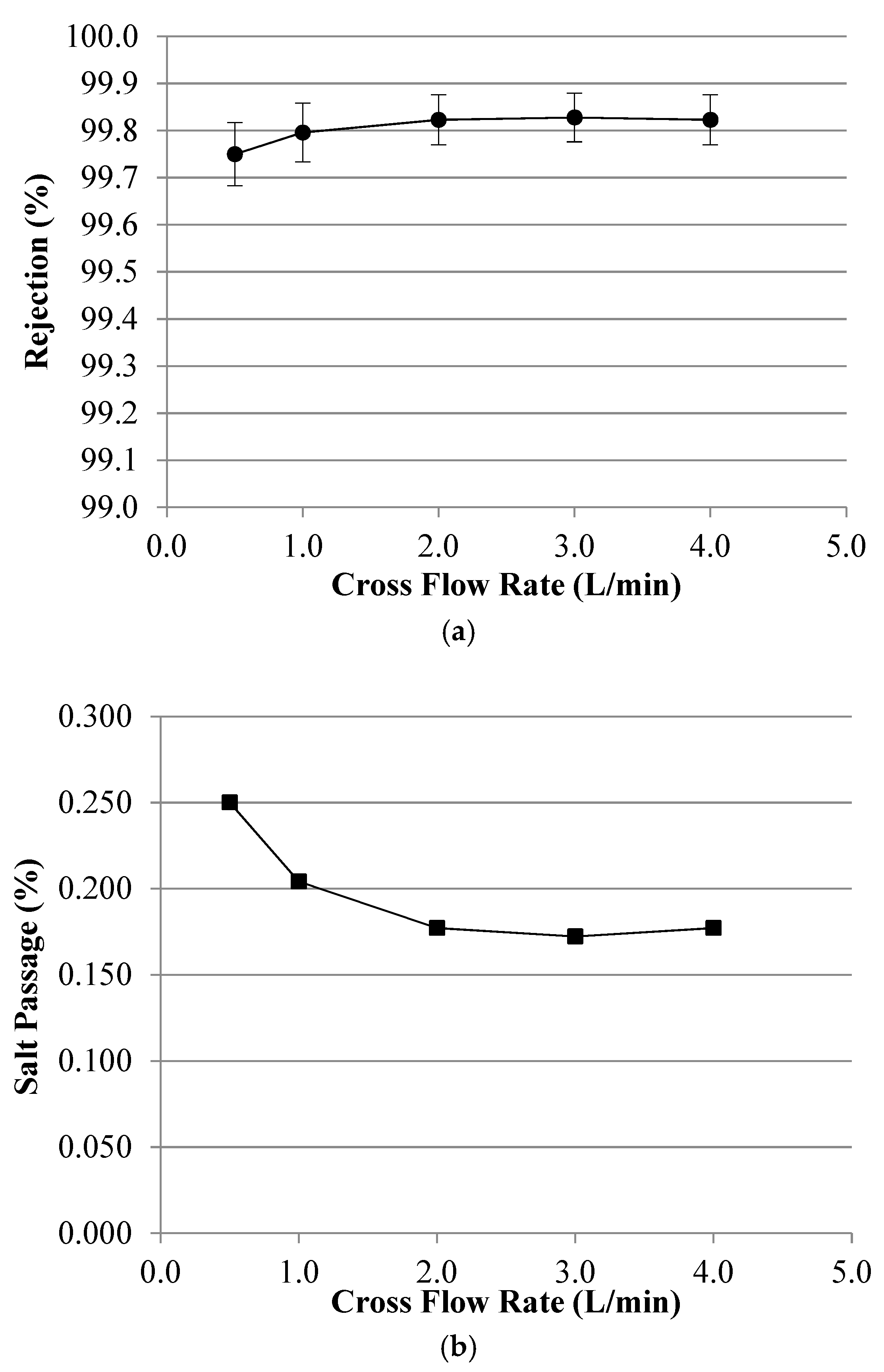
Appendix B. Symbols and Nomenclature
| Symbols | Parameter | Units |
| Rapp | Apparent rejection | % |
| cp | Concentration of solute on the permeate side | mg·L−1 |
| cf | Concentration of solute on the feed side | mg·L−1 |
| Π | Osmotic pressure | bar |
| i | Dimensionless van’t Hoff factor | - |
| M | Molarity | mol·L−1 |
| R | Ideal gas constant | L bar K−1 mol−1 |
| T | Temperature | K |
| t | Time | h |
| ΔΠ | Osmotic pressure difference | bar |
| Πf | Feed osmotic pressure | bar |
| Πp | Permeate osmotic pressure | bar |
| Jv | Permeate flux | L·m−2·h−1 |
| A | Water permeability coefficient | L·m−2·h−1 bar−1 |
| ΔP | Applied pressure | bar |
| Vp | Permeate volume | m3 |
| Am | Membrane area | m2 |
References
- Akcil, A.; Koldas, S. Acid Mine Drainage (AMD): Causes, treatment and case studies. J. Clean. Prod. 2006, 14, 1139–1145. [Google Scholar] [CrossRef]
- Simate, G.S.; Ndlovu, S. Acid mine drainage: Challenges and opportunities. J. Environ. Chem. Eng. 2014, 2, 1785–1803. [Google Scholar]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef]
- Gitari, M.W.; Petrik, L.F.; Etchebers, O.; Key, D.L.; Iwuoha, E.; Okujeni, C. Treatment of Acid Mine Drainage with Fly Ash: Removal of Major Contaminants and Trace Elements. J. Environ. Sci. Heal. Part A 2006, 41, 1729–1747. [Google Scholar] [CrossRef]
- Younger, P.L.; Banwart, S.A.; Hedin, R.S. Mine Water: Hydrology, Pollution, Remediation; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; Volume 9. [Google Scholar]
- Al-Zoubi, H.; Rieger, A.; Steinberger, P.; Pelz, W.; Haseneder, R.; Härtel, G. Optimization Study for Treatment of Acid Mine Drainage Using Membrane Technology. Sep. Sci. Technol. 2010, 45, 2004–2016. [Google Scholar] [CrossRef]
- Watten, B.J.; Sibrell, P.L.; Schwartz, M.F. Acid neutralization within limestone sand reactors receiving coal mine drainage. Environ. Pollut. 2005, 137, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Al-Zoubi, H.; Rieger, A.; Steinberger, P.; Pelz, W.; Haseneder, R.; Härtel, G. Nanofiltration of Acid Mine Drainage. Desalin. Water Treat. 2010, 21, 148–161. [Google Scholar] [CrossRef]
- Sheoran, A.; Sheoran, V.; Choudhary, R. Bioremediation of acid-rock drainage by sulphate-reducing prokaryotes: A review. Miner. Eng. 2010, 23, 1073–1100. [Google Scholar] [CrossRef]
- Da̧browski, A.; Hubicki, Z.; Podkościelny, P.; Robens, E. Selective removal of the heavy metal ions from waters and industrial wastewaters by ion-exchange method. Chemosphere 2004, 56, 91–106. [Google Scholar] [CrossRef]
- Nodwell, M.; Kratochvil, D. Sulphide Precipitation and Ion Exchange Technologies to Treat Acid Mine Drainage. In Proceedings of the 9th International Conference on Acid Rock Drainage, Ottawa, ON, Canada, 20–26 May 2012. [Google Scholar]
- López, J.; Reig, M.; Gibert, O.; Cortina, J. Integration of nanofiltration membranes in recovery options of rare earth elements from acidic mine waters. J. Clean. Prod. 2019, 210, 1249–1260. [Google Scholar] [CrossRef]
- Rieger, A.; Steinberger, P.; Pelz, W.; Haseneder, R.; Härtel, G. Mine water treatment by membrane filtration processes-Experimental investigations on applicability. Desalin. Water Treat. 2009, 6, 54–60. [Google Scholar] [CrossRef]
- Mullett, M.; Fornarelli, R.; Ralph, D. Nanofiltration of Mine Water: Impact of Feed pH and Membrane Charge on Resource Recovery and Water Discharge. Membranes 2014, 4, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Hagmeyer, G.; Gimbel, R. Modelling the rejection of nanofiltration membranes using zeta potential measurements. Sep. Purif. Technol. 1999, 15, 19–30. [Google Scholar] [CrossRef]
- Ferreira Esmi, C.; Schrive, L.; Barre, Y.; Palmeri, J.; Deratani, A. Using nanofiltration in a zero-rejection process: The removal of Ni 2+ and Co 2+ from salty wastewater. Desalin. Water Treat. 2013, 51, 476–484. [Google Scholar] [CrossRef]
- Mänttäri, M.; Pihlajamäki, A.; Nyström, M. Effect of pH on hydrophilicity and charge and their effect on the filtration efficiency of NF membranes at different pH. J. Membr. Sci. 2006, 280, 311–320. [Google Scholar] [CrossRef]
- Tang, B.; Huo, Z.; Wu, P. Study on a novel polyester composite nanofiltration membrane by interfacial polymerization of triethanolamine (TEOA) and trimesoyl chloride (TMC). J. Membr. Sci. 2008, 320, 198–205. [Google Scholar] [CrossRef]
- Lianchao, L.; Baoguo, W.; Huimin, T.; Tianlu, C.; Jiping, X. A novel nanofiltration membrane prepared with PAMAM and TMC by in situ interfacial polymerization on PEK-C ultrafiltration membrane. J. Membr. Sci. 2006, 269, 84–93. [Google Scholar] [CrossRef]
- Werner, C.; Körber, H.; Zimmermann, R.; Dukhin, S.; Jacobasch, H.-J. Extended electrokinetic characterization of flat solid surfaces. J. Colloid Interface Sci. 1998, 208, 329–436. [Google Scholar] [CrossRef]
- Zimmermann, R.; Dukhin, S.; Werner, C. Electrokinetic measurements reveal interfacial charge at polymer films caused by simple electrolyte ions. J. Phys. Chem. B 2001, 105, 8544–8549. [Google Scholar] [CrossRef]
- Bellmann, C.; Klinger, C.; Opfermann, A.; Böhme, F.; Adler, H.-J.P. Evaluation of surface modification by electrokinetic measurements. Prog. Org. Coat. 2002, 44, 93–98. [Google Scholar] [CrossRef]
- Cadotte, J.; Forester, R.; Kim, M.; Peterson, R.; Stocker, T. Nanofiltration membranes broaden the use of membrane separation technology. Desalination 1988, 70, 77–88. [Google Scholar] [CrossRef]
- Childress, A.E.; Elimelech, M. Relating nanofiltration membrane performance to membrane charge (electrokinetik) characteristics. Environ. Sci. Technol. 2000, 34, 3710–3716. [Google Scholar] [CrossRef]
- Luo, J.; Wan, Y. Effects of pH and salt on nanofiltration—A critical review. J. Membr. Sci. 2013, 438, 18–28. [Google Scholar] [CrossRef]
- Childress, A.E.; Elimelech, M. Effect of solution chemistry on the surface charge of polymeric reverse osmosis and nanofiltration membranes. J. Membr. Sci. 1996, 119, 253–268. [Google Scholar] [CrossRef]
- Hoang, T.; Stevens, G.; Kentish, S. The effect of feed pH on the performance of a reverse osmosis membrane. Desalination 2010, 261, 99–103. [Google Scholar] [CrossRef]
- Szymczyk, A.; Fievet, P.; Bandini, S. On the amphoteric behavior of Desal DK nanofiltration membranes at low salt concentrations. J. Membr. Sci. 2010, 355, 60–68. [Google Scholar] [CrossRef]
- Lopez, J.; Reig, M.; Gibert, O.; Valderrama, C.; Cortina, J. Evaluation of NF membranes as treatment technology of acid mine drainage: Metals and sulfate removal. Desalination 2018, 440, 122–134. [Google Scholar] [CrossRef]
- Ku, Y.; Chen, S.; Wang, W. Effect of solution composition on the removal of copper ions by nanofiltration. Sep. Purif. Technol. 2005, 43, 135–142. [Google Scholar] [CrossRef]
- Al-Rashdi, B.; Johnson, D.; Hilal, N. Removal of heavy metal ions by nanofiltration. Desalination 2013, 315, 2–17. [Google Scholar] [CrossRef]
- Donnan, F.G. Theory of membrane equilibria and membrane potentials in the presence of non-dialysing electrolytes. A contribution to physical-chemical physiology. J. Membr. Sci. 1995, 100, 45–55. [Google Scholar] [CrossRef]
- Mazlan, J.; Myneni, S.C.B. Speciation of iron and sulfate in acid waters: Aqueous clusters to mineral precipitates. Environ. Sci. Technol. 2005, 39, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Visser, T.; Modise, S.; Krieg, H.; Keizer, K. The removal of acid sulphate pollution by nanofiltration. Desalination 2001, 140, 79–86. [Google Scholar] [CrossRef]



| Salt | Concentration | pH |
|---|---|---|
| MgSO4 | 4000 ± 100 mg/L MgSO4 | 2.0–7.0 ± 0.1 |
| CuSO4 | 100 ± 10 mg/L Cu | 2.0–5.4 ± 0.1 |
| Mn(NO3)2 | 165 ± 5 mg/L Mn | 2.0–5.1 ± 0.1 |
| Salt | Concentration | pH |
|---|---|---|
| NaSO4 and NaCl | 2000 ± 200 mg/L Na2SO4 and 700 ± 50 mg/L NaCl | 2.0–7.0 ± 0.1 |
| NaSO4 and NaCl | 0.001 ± 0.0001 mol/L Na2SO4 and 0.001 ± 0.0005 mol/L NaCl | 7.0 ± 0.1 |
| 0.005 ± 0.0002 mol/L Na2SO4 and 0.005 ± 0.0017 mol/L NaCl | ||
| 0.010 ± 0.0013 mol/L Na2SO4 and 0.010 ± 0.003 mol/L NaCl | ||
| 0.015 ± 0.0004 mol/L Na2SO4 and 0.015 ± 0.0037 mol/L NaCl | ||
| Na2SiO3 and MgSO4 | 15 ± 1 mg/L Si and 2000 mg/L MgSO4 | 7.0 ± 0.1 |
| Compound | Mullett et al. [14] | Al Zoubi et al. [8] | Target | ||||
|---|---|---|---|---|---|---|---|
| Australian MW (mg/L) | MW B (mg/L) | MW C (mg/L) | MW D (mg/L) | Chilean Copper MW (mg/L) | Brazilian Uranium MW (mg/L) | Model AMD (mg/L) | |
| pH | 4.56 | 5.50 | 4.10 | 2.60 | <2.50 | NA | 2.0–5.0 |
| Calcium, Ca2+ | 480 | 260 | 280 | 270 | 325.9 | 89.9 | 270 |
| Copper, Cu | 410 | 410 | 610 | 590 | 2298 | NA | 390 |
| Magnesium, Mg2+ | 770 | 870 | 900 | 900 | 630.6 | 8.84 | 2300 |
| Manganese, Mn | 440 | 420 | 530 | 500 | 224.5 | 115 | 500 |
| Sodium, Na+ | 2000 | 3000 | 3800 | 3600 | 6.89 | 1.7 | 3600 |
| Sulfate, SO42− | 6900 | 8700 | 10,500 | 10,200 | 14,337 | 1508 | 10,327 |
| Chloride, Cl− | 2300 | 2300 | 3000 | 2900 | NA | 0.21 | 5559 |
| Nitrate, NO3− | NA | NA | NA | NA | NA | NA | 1129 |
| Ion | Concentration (mg/L) | pH |
|---|---|---|
| Calcium, Ca2+ | 250 ± 10 | 2.0–5.0 ± 0.1 |
| Copper, Cu | 370 ± 10 | |
| Magnesium, Mg2+ | 2400 ± 100 | |
| Manganese, Mn | 490 ± 10 | |
| Sodium, Na+ | 3200 ± 100 | |
| Sulfate, SO42− | 8700 ± 200 | |
| Chloride, Cl− | 5000 ± 200 | |
| Nitrate, NO3− | 930 ± 50 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siew, Y.W.; Zedda, K.L.; Velizarov, S. Nanofiltration of Simulated Acid Mine Drainage: Effect of pH and Membrane Charge. Appl. Sci. 2020, 10, 400. https://doi.org/10.3390/app10010400
Siew YW, Zedda KL, Velizarov S. Nanofiltration of Simulated Acid Mine Drainage: Effect of pH and Membrane Charge. Applied Sciences. 2020; 10(1):400. https://doi.org/10.3390/app10010400
Chicago/Turabian StyleSiew, Ye Wee, Karina Listiarini Zedda, and Svetlozar Velizarov. 2020. "Nanofiltration of Simulated Acid Mine Drainage: Effect of pH and Membrane Charge" Applied Sciences 10, no. 1: 400. https://doi.org/10.3390/app10010400
APA StyleSiew, Y. W., Zedda, K. L., & Velizarov, S. (2020). Nanofiltration of Simulated Acid Mine Drainage: Effect of pH and Membrane Charge. Applied Sciences, 10(1), 400. https://doi.org/10.3390/app10010400






