Application of Calcified Structures in Fish as Indicators of Metal Exposure in Freshwater Ecosystems
Abstract
1. Introduction
2. Materials and Methods
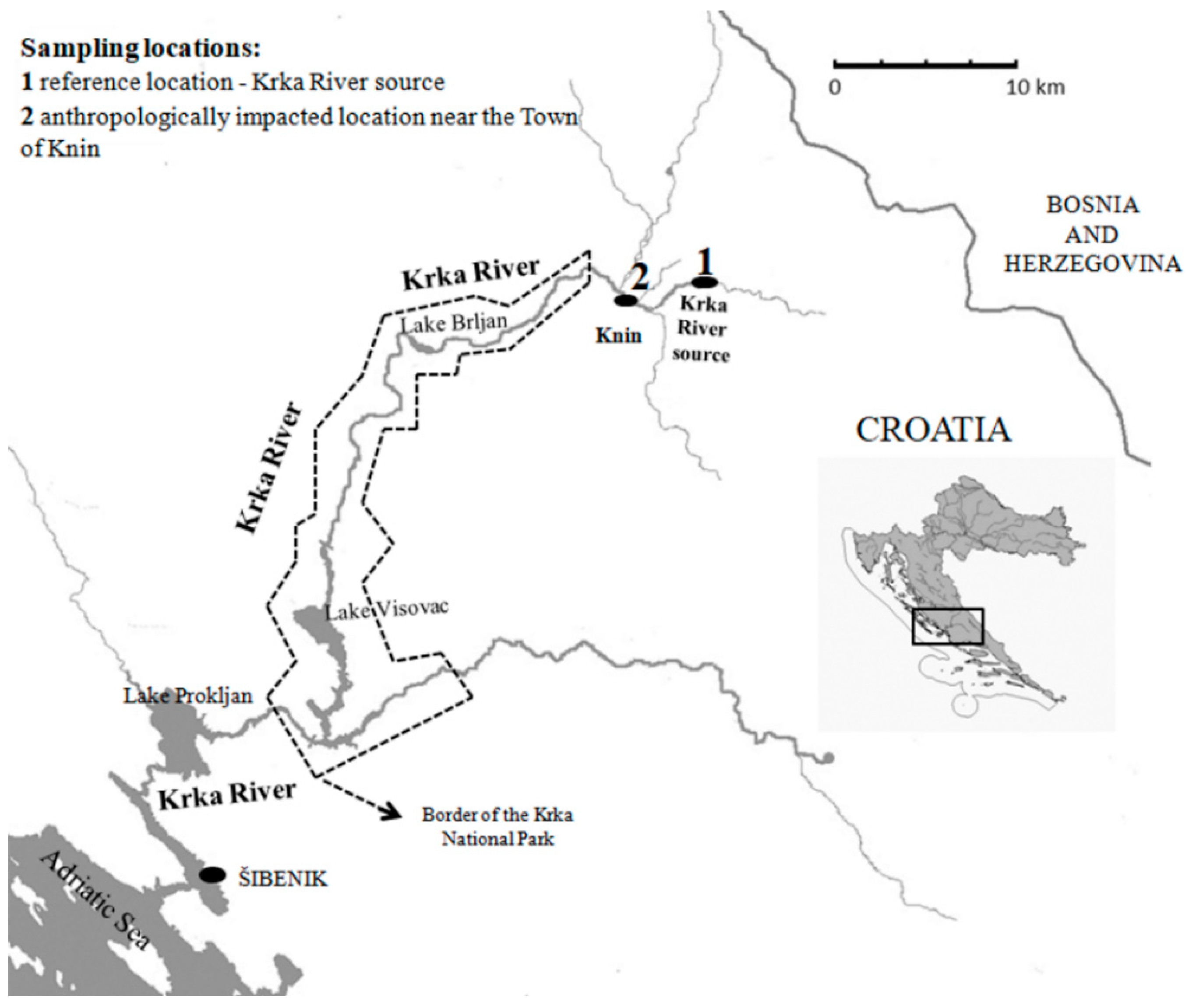
2.1. Study Area and Sampling Procedure
2.2. Dissection of Fish Tissues
2.3. Preparation of Hepatic Cytosolic Fraction
2.4. Acid Digestion of Muscle Tissue and Acanthocephalans
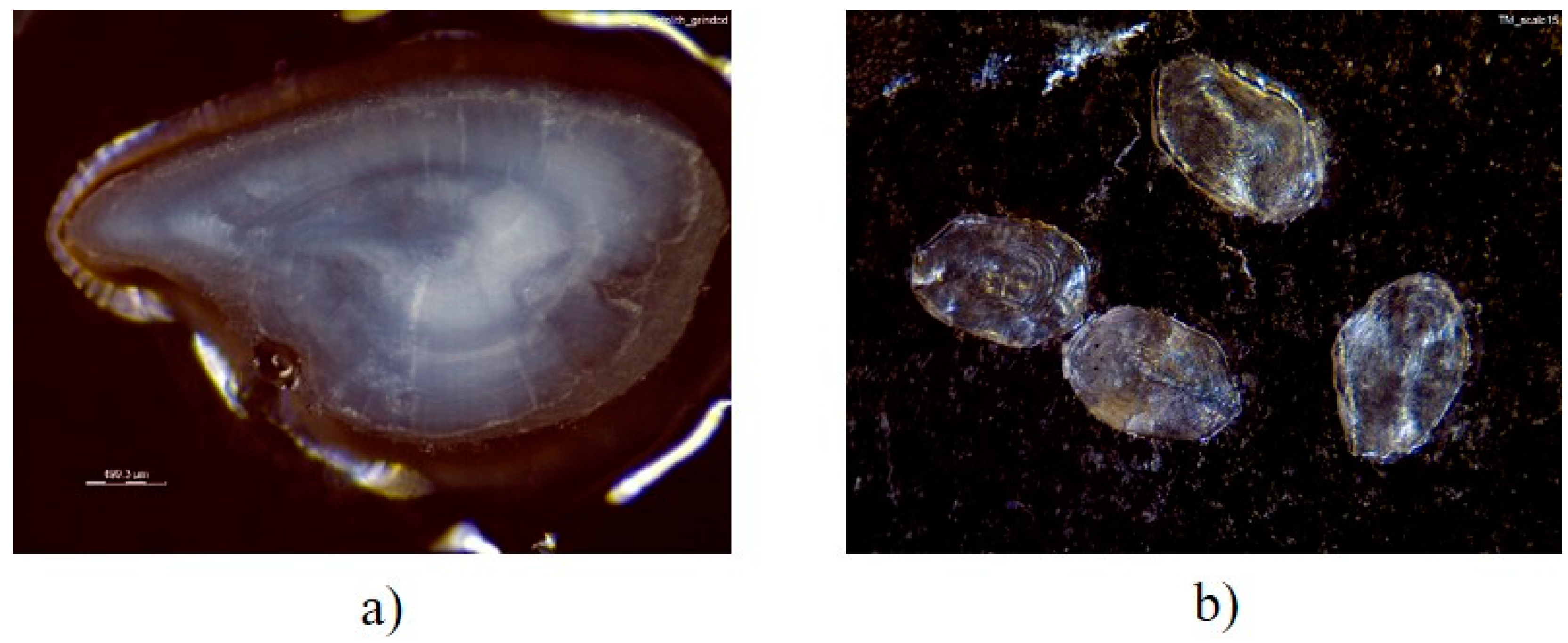
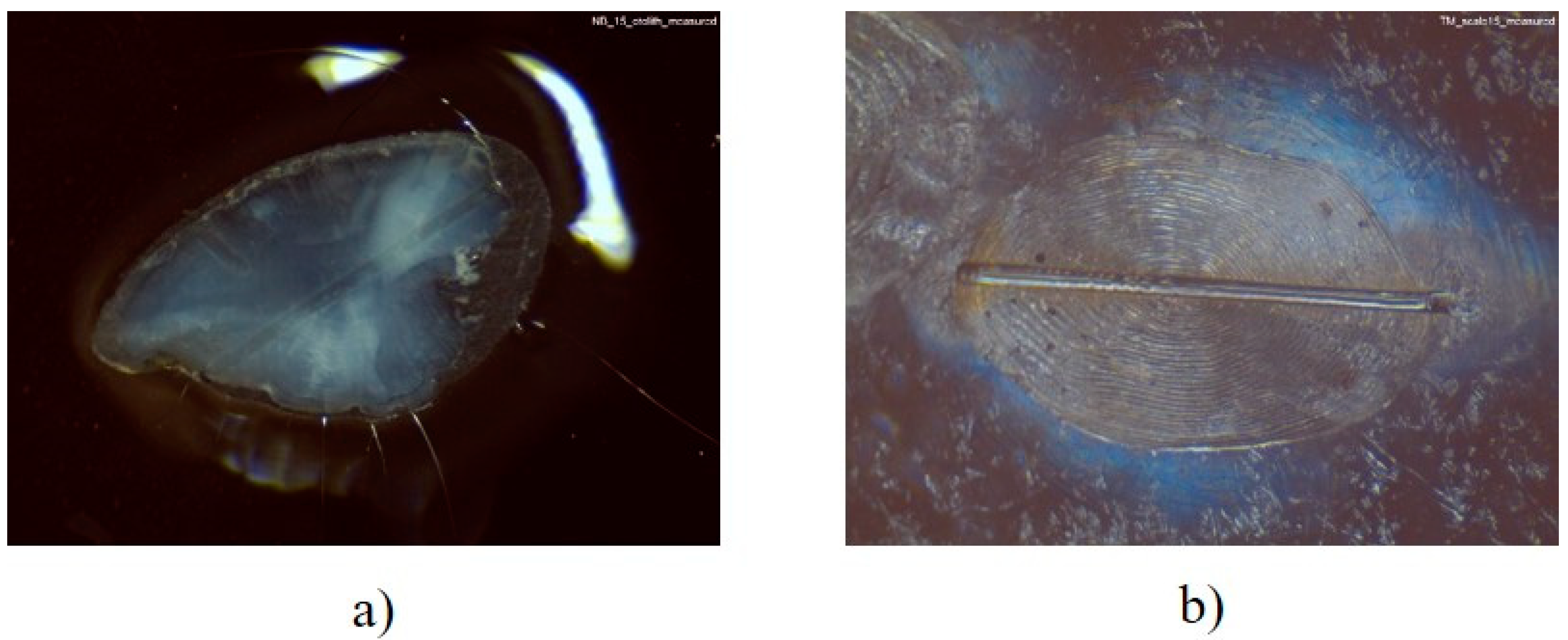
2.5. Calcified Tissue Preparation
2.6. Determination of Metal Content in Water, Fish Soft Tissues and Acanthocephalans
2.7. Determination of Metal Contents in Hard Tissues
2.8. Data Processing and Statistical Analyses
2.8.1. Biological Data
2.8.2. Chemical Data
2.8.3. Statistics
3. Results and Discussion
3.1. Biometric Characteristics of S. trutta Sampled in the Krka River
3.2. Metal Content in the River Water
3.3. Metal Content in Soft Tissues of Fish and Acanthocephalans
3.4. Metal Content in Fish Scales and Otoliths
3.5. Comparison of Metal Accumulation in Soft and Hard Fish Tissues and Acanthocephalans
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boyd, R.S. Heavy Metal Pollutants and Chemical Ecology: Exploring New Frontiers. J. Chem. Ecol. 2010, 36, 46–58. [Google Scholar] [CrossRef]
- Authman, M.M.N.; Zaki, M.S.; Khallaf, E.A.; Abbas, H.H. Use of Fish as Bio-indicator of the Effects of Heavy Metals Pollution. J. Aquac. Res. Dev. 2015, 6, 328–340. [Google Scholar] [CrossRef]
- de Paiva Magalhães, D.; da Costa Marques, M.R.; Baptista, D.F.; Buss, D.F. Metal bioavailability and toxicity in freshwaters. Environ. Chem. Lett. 2015, 13, 69–87. [Google Scholar] [CrossRef]
- Geffen, A.J.; Jarvis, K.; Thorpe, J.P.; Leah, R.T.; Nash, R.D.M. Spatial differences in the trace element concentrations of Irish Sea plaice Pleuronectes platessa and whiting Merlangius merlangus otoliths. J. Sea Res. 2003, 50, 247–254. [Google Scholar] [CrossRef]
- Campana, S.E.; Fowler, A.J.; Jones, C.M. Otolith elemental fingerprinting for stock identification of Atlantic cod (Gadus morhua) using laser ablation ICP MS. Can. J. Fish. Aquat. Sci. 1994, 51, 1942–1950. [Google Scholar] [CrossRef]
- Milton, D.; Halliday, I.; Sellin, M.; Marsh, R.; Staunton-Smith, J.; Woodhead, J. The effect of habitat and environmental history on otolith chemistry of barramundi Lates calcarifer in estuarine populations of a regulated tropical river. Estuar. Coast. Shelf Sci. 2008, 78, 301–315. [Google Scholar] [CrossRef]
- Tabouret, H.; Bareille, G.; Claverie, F.; Pécheyran, C.; Prouzet, P.; Donard, O.F.X. Simultaneous use of strontium:calcium and barium:calcium ratios in otoliths as markers of habitat: Application to the European eel (Anguilla anguilla) in the Adour basin, South West France. Mar. Environ. Res. 2010, 70, 35–45. [Google Scholar] [CrossRef]
- Prohaska, T.; Irrgeher, J.; Zitek, A. Simultaneous multi-element and isotope ratio imaging of fish otoliths by laser ablation split stream ICP-MS/MC ICP-MS. J. Anal. At. Spectrom. 2016, 31, 1612–1621. [Google Scholar] [CrossRef]
- Sawhney, A.K.; Johal, M.S. Potential application of elemental analysis of fish otoliths as pollution indicator. Bull. Environ. Contam. Toxicol. 1999, 63, 698–702. [Google Scholar] [CrossRef]
- Adami, G.; Miletić, M.; Siviero, P.; Barbieri, P.; Reisenhofer, E. Metal contents in tench otoliths: Relationships to the aquatic environment. Ann. Chim. 2001, 91, 401–408. [Google Scholar]
- Saquet, M.; Halden, N.M.; Babaluk, J.; Campbell, J.L.; Nejedly, Z. Micro-PIXE analysis of trace element variation in otoliths from fish collected near acid mine tailings: Potential for monitoring contaminant dispersal. Nucl. Instrum. Methods Phys. Res. B 2002, 189, 196–201. [Google Scholar] [CrossRef]
- Darafsh, F.; Mashinchian, A.; Fatemi, M.; Jamili, S. Study of the application of fish scale as bioindicator of heavy metal pollution (Pb, Zn) in the Cyprinus carpio of the Caspian Sea. Res. J. Environ. Sci. 2008, 2, 438–444. [Google Scholar] [CrossRef][Green Version]
- Ranaldi, M.M.; Gagnon, M.M. Trace metal incorporation in otoliths of black bream (Acanthopagrus butcheri Munro), an indicator of exposure to metal contamination. Water Air Soil Pollut. 2008, 194, 31–43. [Google Scholar] [CrossRef]
- Jarić, I.; Višnjić-Jeftić, Ž.; Cvijanović, G.; Gačić, Z.; Jovanović, L.; Skorić, S.; Lenhardt, M. Determination of differential heavy metal and trace element accumulation in liver, gills, intestine and muscle of sterlet (Acipenser ruthenus) from the Danube River in Serbia by ICP-OES. Microchem. J. 2011, 98, 77–81. [Google Scholar] [CrossRef]
- Krasnići, N.; Dragun, Z.; Erk, M.; Raspor, B. Distribution of selected essential (Co, Cu, Fe, Mn, Mo, Se, and Zn) and nonessential (Cd, Pb) trace elements among protein fractions from hepatic cytosol of European chub (Squalius cephalus L.). Environ. Sci. Pollut. Res. 2013, 20, 2340–2351. [Google Scholar] [CrossRef]
- Dragun, Z.; Filipović Marijić, V.; Krasnići, N.; Ivanković, D.; Valić, D.; Žunić, J.; Kapetanović, D.; Vardić Smrzlić, I.; Redžović, Z.; Grgić, I.; et al. Total and cytosolic concentrations of twenty metals/metalloids in the liver of brown trout Salmo trutta (Linnaeus, 1758) from the karstic Croatian river Krka. Ecotoxicol. Environ. Saf. 2018, 147, 537–549. [Google Scholar] [CrossRef]
- Muhlfeld, C.C.; Marotz, B. Seasonal movement and habitat use by subadult bull trout in the upper Flathead River system, Montana. N. Am. J. Fish. Manag. 2005, 25, 797–810. [Google Scholar] [CrossRef]
- Bath, G.E.; Thorrold, S.R.; Jones, C.M.; Campana, S.E.; Mclaren, J.W.; Lam, J.W.H. Strontium and barium uptake in aragonitic otoliths of marine fish. Geochim. Cosmochim. Acta 2000, 64, 1705–1714. [Google Scholar] [CrossRef]
- Filipović Marijić, V.; Vardić Smrzlić, I.; Raspor, B. Does fish reproduction and metabolic activity influence metal levels in fish intestinal parasites, acanthocephalans, during fish spawning and post-spawning period? Chemosphere 2014, 112, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Filipović Marijić, V.; Vardić Smrzlić, I.; Raspor, B. Effect of acanthocephalan infection on metal, total protein and metallothionein concentrations in European chub from a Sava River section with low metal contamination. Sci. Total Environ. 2013, 463–464, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Sures, B.; Nachev, M.; Selbach, C.; Marcogliese, D.J. Parasite responses to pollution: What we know and where we go in ‘Environmental Parasitology’. Parasites Vectors 2017, 10, 65–83. [Google Scholar] [CrossRef]
- Sures, B. The use of fish parasites as bioindicators of heavy metals in aquatic ecosystems: A review. Aquat. Ecol. 2001, 35, 245–255. [Google Scholar] [CrossRef]
- Sures, B. Environmental parasitology: Relevancy of parasites in monitoring environmental pollution. Trends Parasitol. 2004, 20, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Nachev, M.; Sures, B. Seasonal profile of metal accumulation in the acanthocephalan Pomphorhynchus laevis: A valuable tool to study infection dynamics and implications for metal monitoring. Parasites Vectors 2016, 9, 300–308. [Google Scholar] [CrossRef]
- Thielen, F.; Zimmermann, S.; Baska, F.; Taraschewski, H.; Sures, B. The intestinal parasite Pomphorhynchus laevis (Acanthocephala) from barbel as a bioindicator for metal pollution in the Danube River near Budapest, Hungary. Environ. Pollut. 2004, 129, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Sures, B.; Siddall, R.; Taraschewski, H. Parasites as accumulation indicators of heavy metal pollution. Parasitol. Today 1999, 15, 16–21. [Google Scholar] [CrossRef]
- Campana, S.E.; Chouinard, G.A.; Hanson, J.M.; Frechet, A.; Brattey, J. Otolith elemental fingerprints as biological tracers of fish stocks. Fish. Res. 2000, 46, 343–357. [Google Scholar] [CrossRef]
- Rooker, J.R.; Zdanowicz, V.S.; Secor, D.H. Chemistry of tuna otoliths: Assessment of base composition and postmortem handling effects. Mar. Biol. 2001, 139, 35–43. [Google Scholar]
- Huxham, M.; Kimani, E.; Newton, J.; Augley, J. Stable isotope records from otoliths as tracers of fish migration in a mangrove system. J. Fish. Biol. 2007, 70, 1554–1567. [Google Scholar] [CrossRef]
- Wallace, W.G.; Lee, B.-G.; Luoma, S.N. Subcellular compartmentalization of Cd and Zn in two bivalves. I. Significance of metal-sensitive fractions (MSF) and biologically detoxified metal (BDM). Mar. Ecol. Prog. Ser. 2003, 249, 183–197. [Google Scholar] [CrossRef]
- Rainbow, P.S.; Luoma, S.N.; Wang, W.X. Trophically available metal—a variable feast. Environ. Pollut. 2011, 159, 2347–2349. [Google Scholar] [CrossRef]
- Urien, N.; Cooper, S.; Caron, A.; Sonnenberg, H.; Rozon-Ramilo, L.; Campbell, P.C.G. Subcellular partitioning of metals and metalloids (As, Cd, Cu, Se and Zn) in liver and gonads of wild white suckers (Catostomus commersonii) collected downstream from a mining operation. Aquat. Toxicol. 2018, 202, 105–116. [Google Scholar] [CrossRef]
- Croatian Normative Document HRN EN 14011:2005. Water Quality—Sampling of Fish with Electricity [Kakvoća Vode—Uzorkovanje Riba električnom Strujom, in Croatian]. 2005. Available online: https://standards.iteh.ai/catalog/standards/cen/cd873486-340e-4ee6-ad71-2fbdf24907cc/en-14011-2003 (accessed on 12 December 2021).
- Official Gazette of the Republic of Croatia NN 55/2013. Regulation on the Protection of Animals Used for Scientific Purposes [Pravilnik o Zaštiti životinja Koje se Koriste u Znanstvene Svrhe, in Croatian]. 2013. Available online: https://www.ecolex.org/details/legislation/european-union-protection-of-animals-used-for-scientific-purposes-regulations-2012-si-no-543-of-2012-lex-faoc121063/ (accessed on 12 December 2021).
- Sturgeon, R.E.; Willie, S.N.; Yang, L.; Greenberg, R.; Spatz, R.O.; Chen, Z.; Scriver, C.; Clancy, V.; Lam, J.W.; Thorrold, S. Certification of a fish otolith reference material in support of quality assurance for trace element analysis. J. Anal. At. Spectrom. 2005, 20, 1067–1071. [Google Scholar] [CrossRef]
- Holá, M.; Kalvoda, J.; Bábek, O.; Brzobohatý, R.; Holoubek, I.; Kanický, V.; Škoda, R. LA-ICP-MS heavy metal analyses of fish scales from sediments of the Oxbow Lake Certak of the Morava River (Czech Republic). Environ. Geol. 2010, 58, 141–151. [Google Scholar] [CrossRef]
- Heidinger, R.C.; Crawford, S.D. Effect of temperature and feeding rate on the liver-somatic index of largemouth bass, Micropterus salmoides. J. Fish. Res. Board Can. 1977, 34, 633–638. [Google Scholar] [CrossRef]
- Wootton, R.J. Ecology of Teleost Fishes; Fish and Fisheries Series 1; Chapman and Hall: London, UK; New York, NY, USA, 1990. [Google Scholar]
- Bush, A.O.; Lafferty, K.D.; Lotz, J.M.; Shostak, A.W. Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol. 1997, 83, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Mikac, N.; Filipović Marijić, V.; Mustafić, P.; Lajtner, J. Development of Methodology for Monitoring the Elements of Chemical Status in Biota of Freshwater Ecosystem [Report in Croatian]; Ruđer Bošković Institute: Zagreb, Croatia, 2018. [Google Scholar]
- Linde, A.R.; Sánchez-Galán, S.; Izquierdo, J.I.; Arribas, P.; Marañón, E.; García-Vázquez, E. Brown trout as biomonitor of heavy metal pollution: Effect of age on the reliability of the assessment. Ecotoxicol. Environ. Saf. 1998, 40, 120–125. [Google Scholar] [CrossRef]
- Couture, P.; Rajotte, J.W. Morphometric and metabolic indicators of metal stress in wild yellow perch (Perca flavescens) from Sudbury, Ontario: A review. J. Environ. Monit. 2003, 5, 216–221. [Google Scholar] [CrossRef]
- Zhelev, Z.M.; Mollova, D.; Boyadziev, P. Morphological and hematological parameters of Carassius gibelio (Pisces: Gyprinidae) in conditions of anthropogenic pollution in Southern Bulgaria. Use hematological parameters as biomarkers. Trakia J. Sci. 2016, 14, 1–15. [Google Scholar] [CrossRef]
- Zhelev, Z.M.; Tsonev, S.V.; Boyadziev, P.S. Significant changes in morpho physiological and haematological parameters of Carassius gibelio (Bloch, 1782) (Actinopterygii: Cyprinidae) as response to sporadic effusions of industrial wastewater into the Sazliyka River, Southern Bulgaria. Acta Zool. Bulg. 2018, 70, 547–556. [Google Scholar]
- Mijošek, T.; Filipović Marijić, V.; Dragun, Z.; Ivanković, D.; Krasnići, N.; Redžović, Z.; Veseli, M.; Gottstein, S.; Lajtner, J.; Sertić Perić, M.; et al. Thallium accumulation in different organisms from karst and lowland rivers of Croatia under wastewater impact. Environ. Chem. 2020, 17, 201–212. [Google Scholar] [CrossRef]
- Vardić Smrzlić, I.; Valić, D.; Kapetanović, D.; Dragun, Z.; Gjurčević, E.; Ćetković, H.; Teskeredžić, E. Molecular characterisation and infection dynamics of Dentitruncus truttae from trout (Salmo trutta and Oncorhynchus mykiss) in Krka River, Croatia. Vet. Parasitol. 2013, 197, 604–613. [Google Scholar]
- Paggi, L.; Orecchia, P.; Del Marro, M.; Iori, A.; Manilla, G. Parasites of Salmo trutta from the Tirino River II. Host-Parasite interactions of helminth species. Parassitologia 1978, 20, 161–168. [Google Scholar] [PubMed]
- Dezfuli, B.S.; Giovinazzo, G.; Lui, A.; Giari, L. Inflammatory response to Dentitruncus truttae (Acanthocephala) in the intestine of brown trout. Fish Shellfish Immunol. 2008, 24, 726–733. [Google Scholar] [CrossRef]
- Marcogliese, D.J. Parasites: Small players with crucial roles in the ecological theater. EcoHealth 2004, 1, 151–164. [Google Scholar] [CrossRef]
- Filipović Marijić, V.; Kapetanović, D.; Dragun, Z.; Valić, D.; Krasnići, N.; Redžović, Z.; Grgić, I.; Žunić, J.; Kružlicová, D.; Nemeček, P.; et al. Influence of technological and municipal wastewaters on vulnerable karst riverine system, Krka River in Croatia. Environ. Sci. Pollut. Res. 2018, 25, 4715–4727. [Google Scholar] [CrossRef]
- Sertić Perić, M.; Matoničkin Kepčija, R.; Miliša, M.; Gottstein, S.; Lajtner, J.; Dragun, Z.; Filipović Marijić, V.; Krasnići, N.; Ivanković, D.; Erk, M. Benthos-drift relationships as proxies for the detection of the most suitable bioindicator taxa in flowing waters—a pilot-study within a Mediterranean karst river. Ecotoxicol. Environ. Saf. 2018, 163, 125–135. [Google Scholar] [CrossRef]
- WHO. Manganese in Drinking-water. In Background Document for Development of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Dossi, C.; Ciceri, E.; Giussani, B.; Pozzi, A.; Galgaro, A.; Viero, A.; Vigano, A. Water and snow chemistry of main ions and trace elements in the karst system of Monte Pelmo massif (Dolomites, Eastern Alps, Italy). Mar. Freshw. Res. 2007, 58, 649–656. [Google Scholar] [CrossRef]
- Cukrov, N.; Cmuk, P.; Mlakar, M.; Omanović, D. Spatial distribution of trace metals in the Krka River, Croatia. An example of the self-purification. Chemosphere 2008, 72, 1559–1566. [Google Scholar] [CrossRef]
- Carvalho, M.L.; Santiago, S.; Nunes, M.L. Assessment of the essential element and heavy metal content of edible fish muscle. Anal. Bioanal. Chem. 2005, 382, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Mijošek, T.; Filipović Marijić, V.; Dragun, Z.; Ivanković, D.; Krasnići, N.; Erk, M.; Gottstein, S.; Lajtner, J.; Sertić Perić, M.; Matoničkin Kepčija, R. Comparison of electrochemically determined metallothionein concentrations in wild freshwater salmon fish and gammarids and their relation to total and cytosolic metal levels. Ecol. Indic. 2019, 105, 188–198. [Google Scholar] [CrossRef]
- Mijošek, T.; Filipović Marijić, V.; Dragun, Z.; Krasnići, N.; Ivanković, D.; Erk, M. Evaluation of multi-biomarker response in fish intestine as an initial indication of anthropogenic impact in the aquatic karst environment. Sci. Total Environ. 2019, 660, 1079–1090. [Google Scholar] [CrossRef]
- Clearwater, S.J.; Baskin, S.J.; Wood, C.M.; McDonald, D.G. Gastrointestinal uptake and distribution of copper in rainbow trout. J. Exp. Biol. 2000, 203, 2455–2466. [Google Scholar] [CrossRef]
- Lapointe, D.; Couture, P. Influence of the route of exposure on the accumulation and subcellular distribution of nickel and thallium in juvenile fathead minnows (Pimephales promelas). Arch. Environ. Contam. Toxicol. 2009, 57, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Zitek, A.; Sturm, M.; Waidbacher, H.; Prohaska, T. Discrimination of wild and hatchery trout by natural chronological patterns of elements and isotopes in otoliths using LA-ICP-MS. Fish. Manag. Ecol. 2010, 17, 435–445. [Google Scholar] [CrossRef]
- Wells, B.K.; Rieman, B.E.; Clayton, J.L.; Horan, D.L.; Jones, C.M. Relationships between water, otolith, and scale chemistries of westslope cutthroat trout from the Coeur d”Alene River, Idaho: The potential application of hard-part chemistry to describe movements in freshwater. Trans. Am. Fish. Soc. 2003, 132, 409–424. [Google Scholar] [CrossRef]
- Ramsay, A.L.; Milner, N.J.; Hughes, R.N.; McCarthy, I.D. Comparison of the performance of scale and otolith microchemistry as fisheries research tools in a small upland catchment. Can. J. Fish. Aquat. Sci. 2011, 68, 823–833. [Google Scholar] [CrossRef]
- Kalantzi, I.; Mylona, K.; Pergantis, S.A.; Coli, A.; Panopoulos, S.; Tsapakis, M. Elemental distribution in the different tissues of brood stock from Greek hatcheries. Aquaculture 2019, 503, 175–185. [Google Scholar] [CrossRef]
- Bigi, A.; Foresti, E.; Gregorini, R.; Ripamonti, A.; Roveri, N.; Shah, J.S. The role of magnesium on the structure of biological apatites. Calcif. Tissue Int. 1992, 50, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Kalvoda, J.; Novak, M.; Bábek, O.; Brzobohatý, R.; Holá, M.; Holoubek, I.; Kanický, V.; Škoda, R. Compositional changes in fish scale hydroxylapatite during early diagenesis; an example from an abandoned meander. Biogeochemistry 2009, 94, 197–215. [Google Scholar] [CrossRef]
- Holá, M.; Kalvoda, J.; Nováková, H.; Škoda, R.; Kanický, V. Possibilities of LA-ICP-MS technique for the spatial elemental analysis of the recent fish scales: Line scan vs. depth profiling. Appl. Surf. Sci. 2011, 257, 1932–1940. [Google Scholar] [CrossRef]
- Bostick, W.D.; Stevenson, R.J.; Harris, L.A.; Peery, D.; Hall, J.R.; Shoemaker, J.L.; Jarabek, R.J.; Munday, E.B. Use of Apatite for Chemical Stabilization of Subsurface Contaminants; National Energy Technology Laboratory: Pittsburgh, PA, USA, 2003. [Google Scholar]
- Goto, T.; Sasaki, K. Effects of trace elements in fish bones on crystal characteristics of hydroxyapatite obtained by calcination. Ceram. Int. 2014, 40, 10777–10785. [Google Scholar] [CrossRef]
- Campana, S.E. Chemistry and composition of fish otoliths: Pathways, mechanisms and applications. Mar. Ecol.-Prog. Ser. 1999, 188, 263–297. [Google Scholar] [CrossRef]
- Veinott, G.; Northcote, T.; Rosenau, M.; Evans, R.D. Concentrations of strontium in the pectoral fin rays of the white sturgeon (Acipenser transmontanus) by laser ablation sampling-inductively coupled plasma- mass spectrometry as an indicator of marine migrations. Can. J. Fish. Aquat. Sci. 1999, 56, 1981–1990. [Google Scholar] [CrossRef]
- Clarke, A.D.; Telmer, K.H.; Shrimpton, J.M. Elemental analysis of otoliths, fin rays and scales: A comparison of bony structures to provide population and life-history information for the Arctic grayling (Thymallus arcticus). Ecol. Freshw. Fish. 2007, 16, 354–361. [Google Scholar] [CrossRef]
- Hammond, M.P.; Savage, C. Use of regenerated scales and scale marginal increments as indicators of recent dietary history in fish. Estuar. Coast. 2009, 32, 340–349. [Google Scholar] [CrossRef]
- Adey, E.A.; Black, K.D.; Sawyer, T.; Shimmield, T.M.; Trueman, C.N. Scale microchemistry as a tool to investigate the origin of wild and farmed Salmo salar. Mar. Ecol. Prog. Ser. 2009, 390, 225–235. [Google Scholar] [CrossRef]
| Krka River Source n = 18 | Krka Knin n = 17 | |
|---|---|---|
| Point Sources of Pollution | Reference Site—Unknown Pollution Sources | Contaminated Site—Screw Factory, Industrial and Municipal Wastewaters, Agricultural Runoff |
| Total length (cm) | 20.4 ± 4.2 | 24.4 ± 13.5 |
| Body mass (g) | 114.4 ± 81.2 | 339.4 ± 601.4 |
| HSI (%) | 1.3 ± 0.5 | 1.3 ± 0.6 |
| GSI (%) | 0.37 ± 0.24 | 0.25 ± 0.16 |
| FCI (g cm−3 × 100) | 1.2 ± 0.4 | 1.1 ± 0.3 |
| Sex (M/F/ND *) | 9/9/0 | 5/10/2 |
| Prevalence (number and % of trout infected with parasites) | 17; 94% | 13; 76% |
| Mean intensity of infection (mean ± S.E.) | 40.3 ± 8.9 | 32.1 ± 11.3 |
| Total number of parasite individuals in sampled fish | 685 | 417 |
| Krka River Source n = 3 | Krka Knin n = 3 | |
|---|---|---|
| Ba (µg L−1) | 4.38 ± 0.11 * | 5.69 ± 0.10 * |
| Fe (µg L−1) | 0.340 ± 0.060 * | 11.62 ± 1.89 * |
| Mg (µg L−1) | 11630 ± 130 * | 11100 ± 180 * |
| Mn (µg L−1) | 0.010 ± 0.004 * | 6.73 ± 0.10 * |
| Rb (µg L−1) | 0.280 ± 0.005 * | 0.460 ± 0.001 * |
| Sr (µg L−1) | 88.42 ± 1.72 * | 186.2 ± 1.0 * |
| Tl (µg L−1) | 0.006 ± 0.000 | 0.005 ± 0.000 |
| Zn (µg L−1) | 3.57 ± 0.62 * | 30.03 ± 4.53 * |
| Liver | Muscle | Acanthocephalans | |||||
| µg g−1 w.m. | Krka River Source n = 18 | Krka Knin n = 15 | Krka River Source n = 6 | Krka Knin n = 6 | Krka River Source n = 10 | Krka Knin n = 10 | |
| Ba | 0.311 ± 0.088 | 0.365 ± 0.109 | 0.013 ± 0.001 * | 0.021 ± 0.08 * | 0.325 ± 0.160 * | 0.592 ± 0.255 * | |
| Fe | 27.79 ± 9.25 | 33.60 ± 13.71 | 4.74 ± 1.42 * | 2.98 ± 0.62 * | 13.04 ± 3.75 *** | 50.70 ± 26.05 *** | |
| Mg | 93.02 ± 5.36 | 91.56 ± 8.15 | 300.3 ± 14.2 * | 328.8 ± 17.8 * | 197.9 ± 21.9 | 235.2 ± 61.4 | |
| Mn | 0.716 ± 0.115 | 0.785 ± 0.124 | 0.111 ± 0.024 | 0.170 ± 0.090 | 3.25 ± 0.30 | 4.56 ± 2.02 | |
| Rb | 4.93 ± 2.02 | 3.73 ± 1.79 | 4.12 ± 0.40 | 4.13 ± 1.32 | 2.27 ± 0.37 * | 1.74 ± 0.59 * | |
| Sr | 0.013 ± 0.010 * | 0.037 ± 0.048 * | 0.079 ± 0.089 | 0.189 ± 0.204 | 1.15 ± 0.92 ** | 3.50 ± 2.07 ** | |
| Tl | 0.192 ± 0.099 | 0.164 ± 0.096 | 0.016 ± 0.005 * | 0.009 ± 0.005 * | 1.49 ± 1.22 | 1.07 ± 0.83 | |
| Zn | 19.44 ± 3.92 | 20.72 ± 5.18 | 2.11 ± 0.60 | 2.23 ± 0.84 | 39.95 ± 14.38 * | 128.6 ± 151.9 * | |
| Scales | Otoliths | ||||
| µg g−1 | Krka River Source n = 3 | Krka Knin n = 3 | Krka River Source n = 3 | Krka Knin n = 3 | |
| Ba | n.d. | n.d. | 0.910 ± 0.220 | 0.980 ± 0.100 | |
| Fe | 69.14 ± 27.94 | 76.60 ± 38.12 | n.d. | n.d. | |
| Mg | 4300 ± 469 | 4094 ± 317 | 80.75 ± 2.29 | 77.51 ± 6.17 | |
| Mn | 9.26 ± 3.14 | 20.81 ± 10.16 | 0.860 ± 0.210 | 1.57 ± 0.81 | |
| Rb | n.d. | n.d. | 1.64 ± 0.68 | 1.23 ± 0.46 | |
| Sr | 87.84 ± 22.76 | 205.7 ± 74.4 | 335.0 ± 56.6 | 789.2 ± 563.3 | |
| Tl | n.d. | n.d. | 0.400 ± 0.060 | 0.230 ± 0.020 | |
| Zn | 72.15 ± 25.36 | 113.8 ± 29.8 | 20.97 ± 7.83 | 26.43 ± 6.85 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filipović Marijić, V.; Mijošek, T.; Dragun, Z.; Retzmann, A.; Zitek, A.; Prohaska, T.; Bačić, N.; Redžović, Z.; Grgić, I.; Krasnići, N.; et al. Application of Calcified Structures in Fish as Indicators of Metal Exposure in Freshwater Ecosystems. Environments 2022, 9, 14. https://doi.org/10.3390/environments9020014
Filipović Marijić V, Mijošek T, Dragun Z, Retzmann A, Zitek A, Prohaska T, Bačić N, Redžović Z, Grgić I, Krasnići N, et al. Application of Calcified Structures in Fish as Indicators of Metal Exposure in Freshwater Ecosystems. Environments. 2022; 9(2):14. https://doi.org/10.3390/environments9020014
Chicago/Turabian StyleFilipović Marijić, Vlatka, Tatjana Mijošek, Zrinka Dragun, Anika Retzmann, Andreas Zitek, Thomas Prohaska, Niko Bačić, Zuzana Redžović, Ivana Grgić, Nesrete Krasnići, and et al. 2022. "Application of Calcified Structures in Fish as Indicators of Metal Exposure in Freshwater Ecosystems" Environments 9, no. 2: 14. https://doi.org/10.3390/environments9020014
APA StyleFilipović Marijić, V., Mijošek, T., Dragun, Z., Retzmann, A., Zitek, A., Prohaska, T., Bačić, N., Redžović, Z., Grgić, I., Krasnići, N., Valić, D., Kapetanović, D., Žunić, J., Ivanković, D., Vardić Smrzlić, I., & Erk, M. (2022). Application of Calcified Structures in Fish as Indicators of Metal Exposure in Freshwater Ecosystems. Environments, 9(2), 14. https://doi.org/10.3390/environments9020014









