Water Quality Assessment of Surface and Groundwater Sources Using a Water Quality Index Method: A Case Study of a Peri-Urban Town in Southwest, Nigeria
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Physicochemical Measurement
2.3. Water Quality Index (WQI) Determination
2.4. Microbiological Analyses
2.4.1. Enumeration of Total Bacteria Count
2.4.2. Identification of Faecal Coliform and E. coli in Water Samples
Presumptive Test
Confirmatory Test
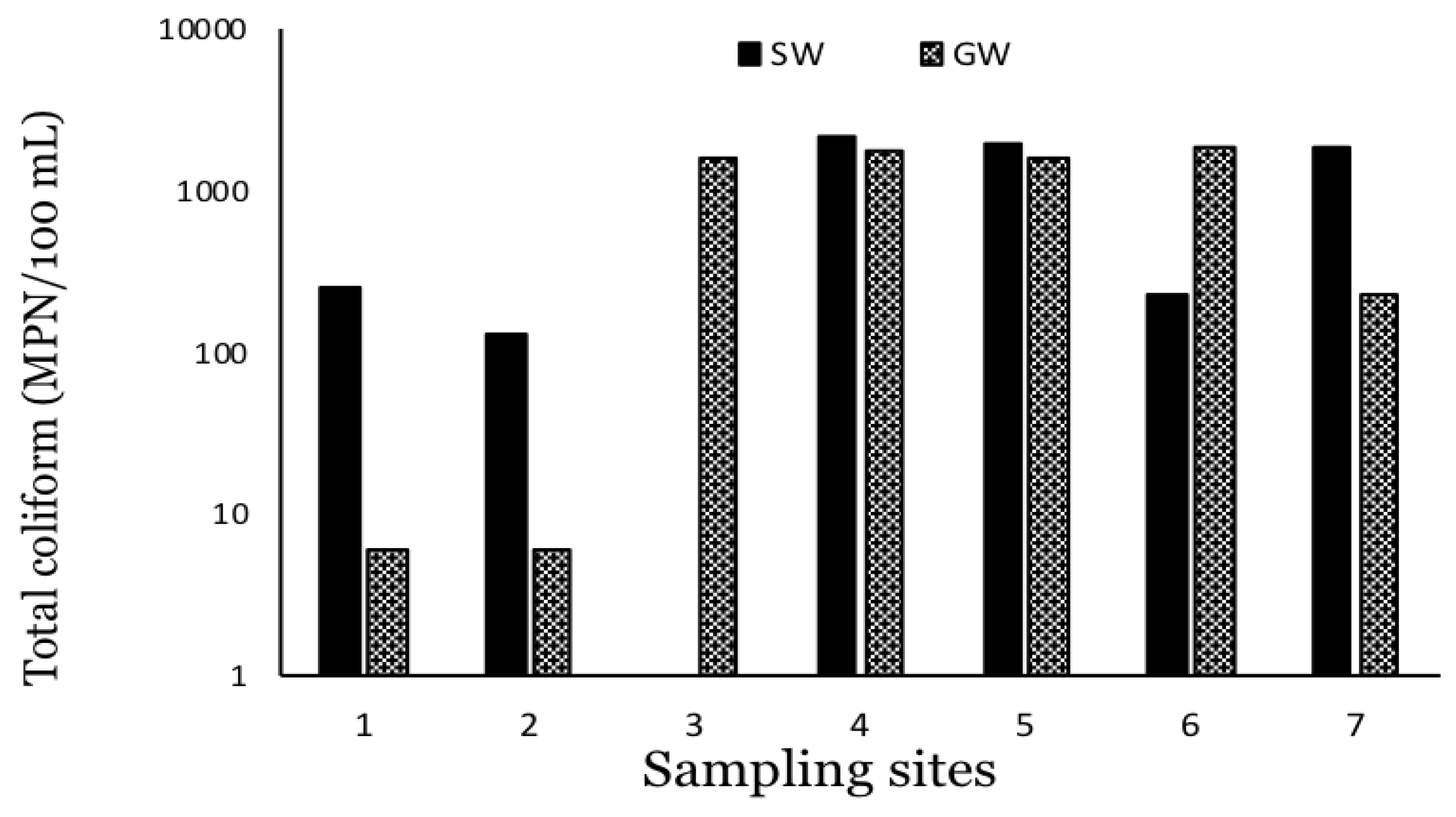
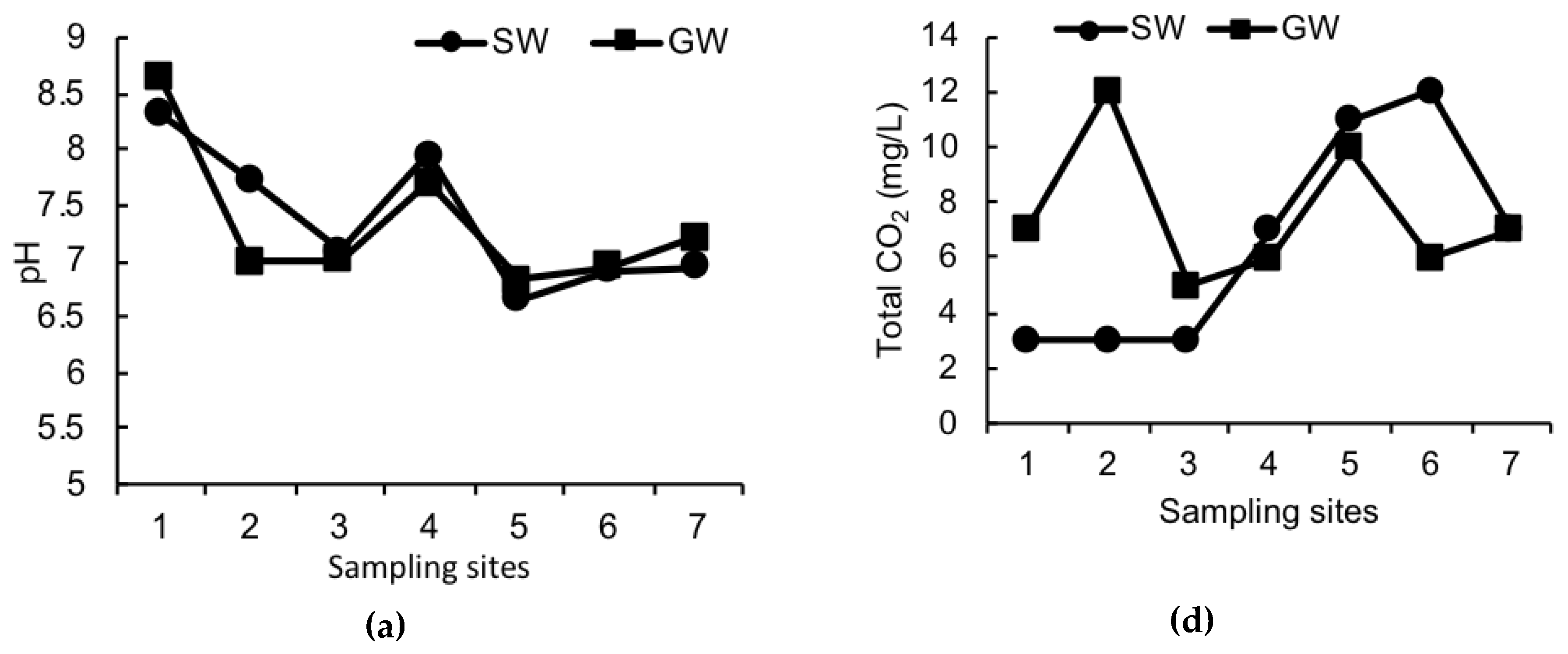
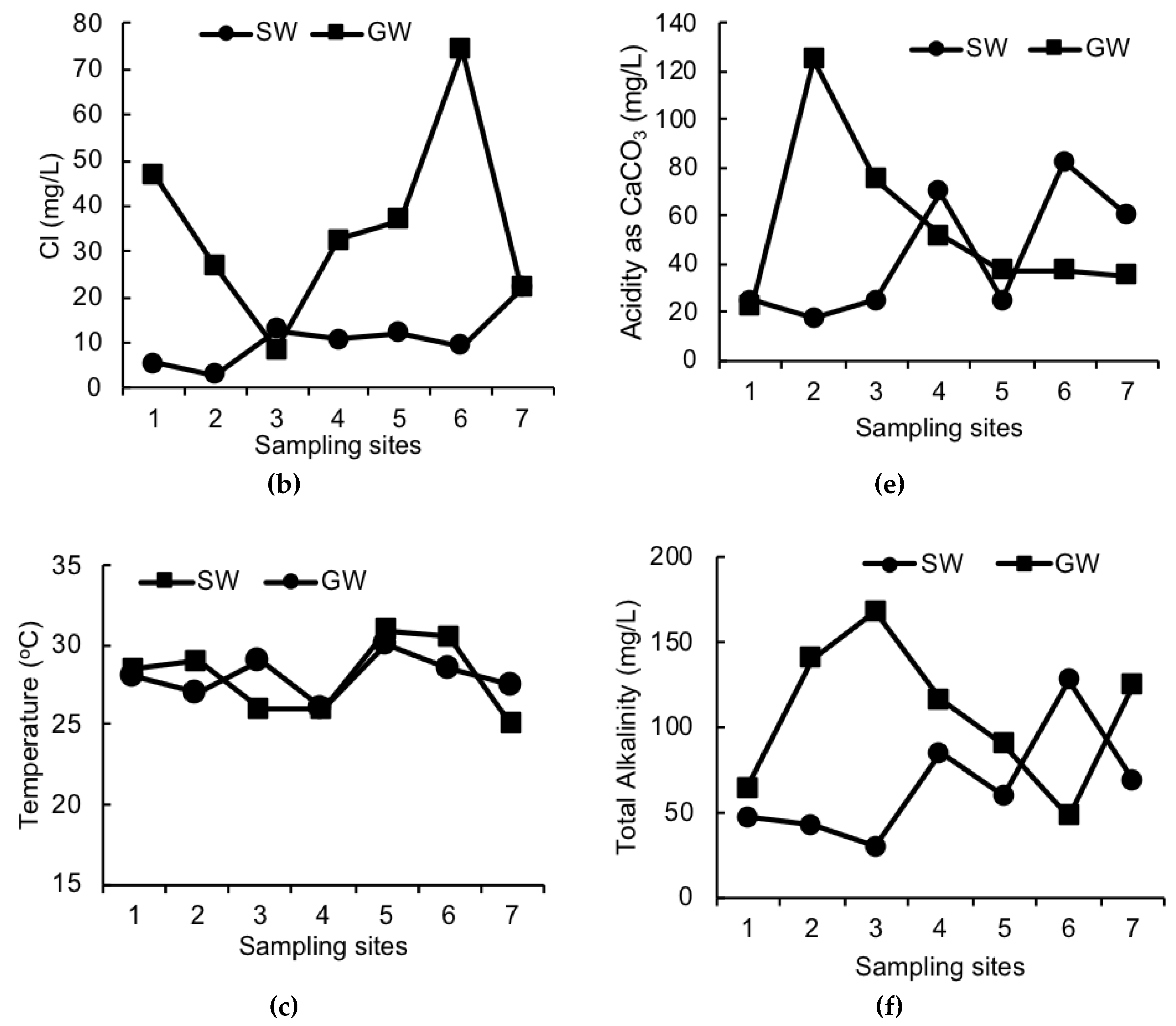
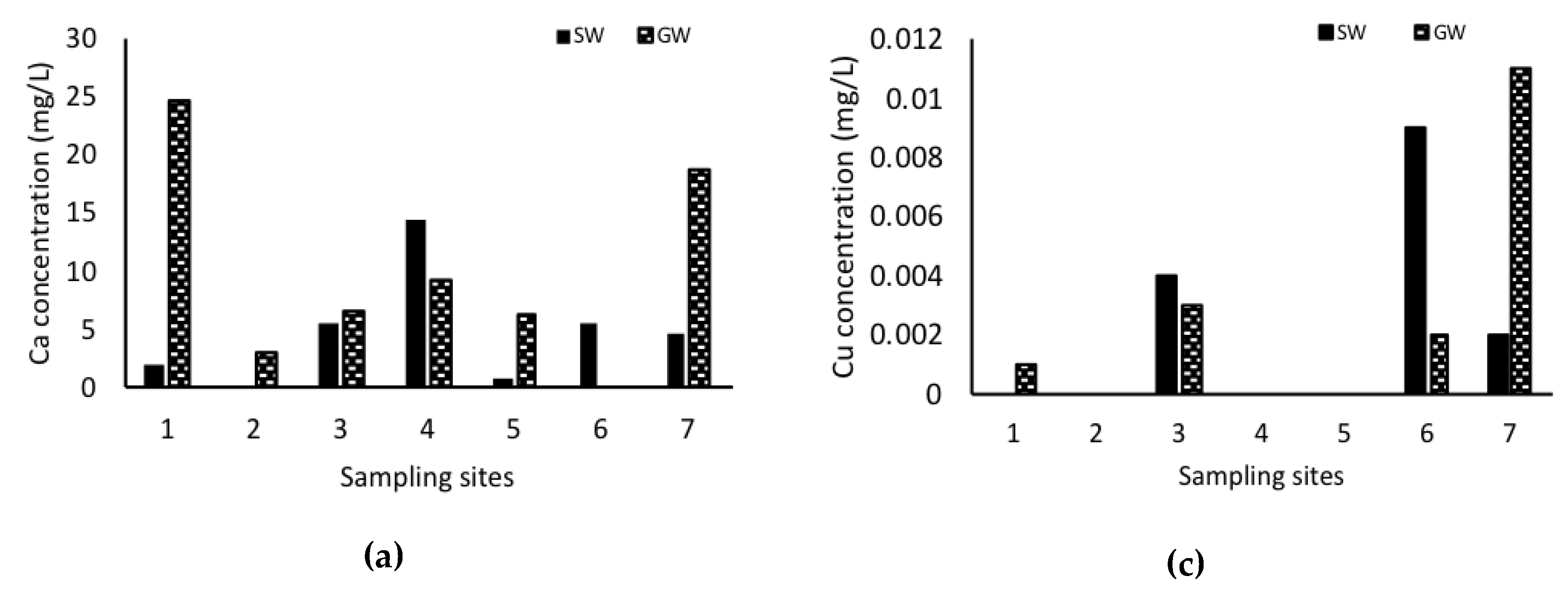
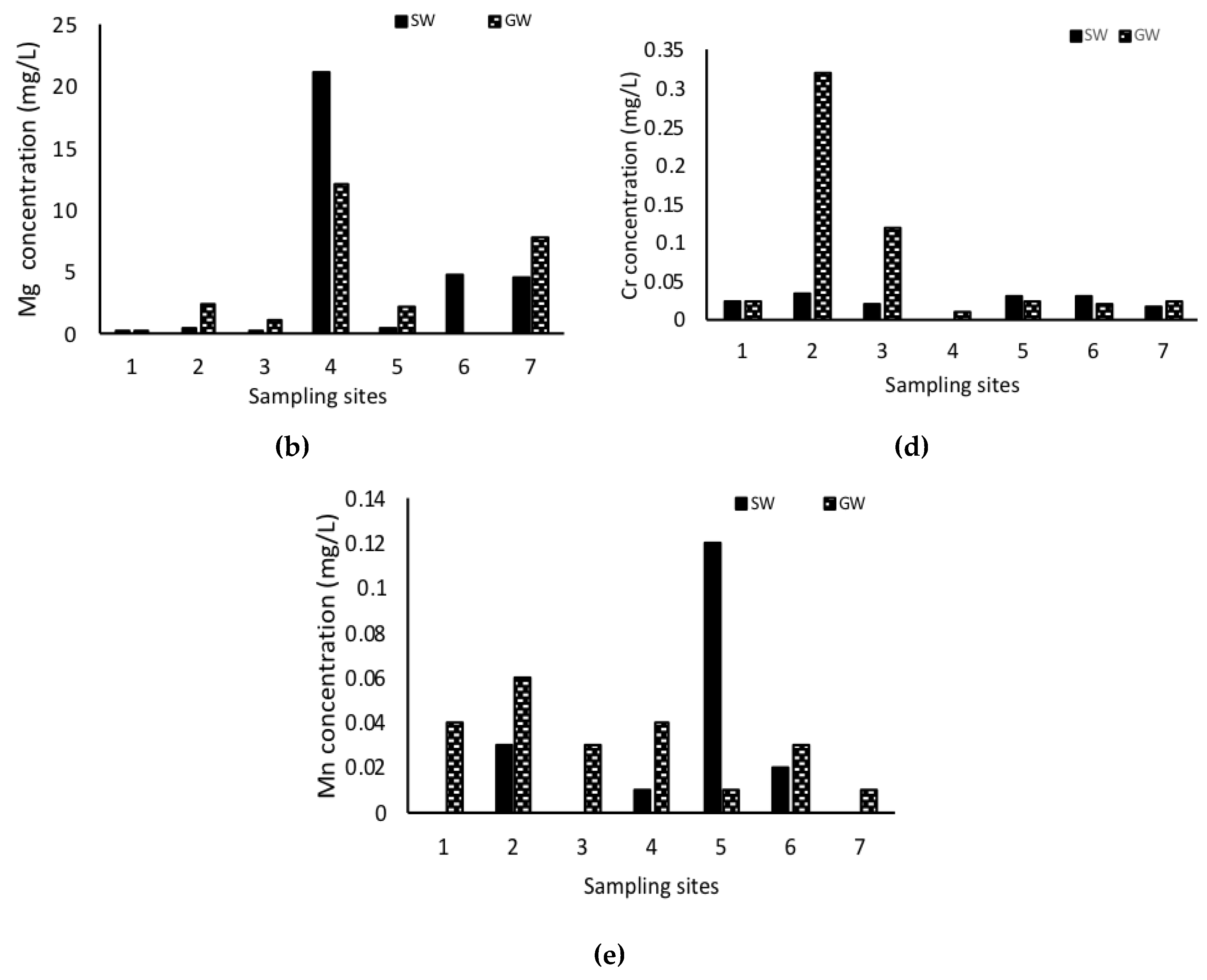
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Edokpayi, J.N.; Rogawski, E.T.; Kahler, D.M.; Hill, C.L.; Reynolds, C.; Nyathi, E.; Smith, J.A.; Odiyo, J.O.; Samie, A.; Bessong, P.; et al. Challenges to Sustainable Safe Drinking Water: A Case Study of Water Quality and Use across Seasons in Rural Communities in Limpopo Province, South Africa. Water 2018, 10, 159. [Google Scholar] [CrossRef]
- Hunter, P.R.; MacDonald, A.M.; Carter, R.C. Water Supply and Health. PLoS Med. 2010, 7, e1000361. [Google Scholar] [CrossRef]
- Edokpayi, J.N.; Odiyo, J.O.; Popoola, E.O.; Msagati, T.M. Evaluation of Microbiological and Physicochemical Parameters of Alternative Source of Drinking Water: A Case Study of Nzhelele River, South Africa. Open Microbiol. J. 2018, 12, 18. [Google Scholar] [CrossRef]
- Ding, Z.; Zhai, Y.; Wu, C.; Wu, H.; Lu, Q.; Lin, J.; He, F. Infectious diarrheal disease caused by contaminated well water in Chinese schools: A systematic review and meta-analysis. J. Epidemiol. 2017, 27, 274–281. [Google Scholar] [CrossRef]
- Diouf, K.; Tabatabai, P.; Rudolph, J.; Marx, M. Diarrhoea prevalence in children under five years of age in rural Burundi: An assessment of social and behavioural factors at the household level. Glob. Health Action 2014, 7, 24895. [Google Scholar] [CrossRef]
- Edokpayi, J.N.; Enitan, A.M.; Mutileni, N.; Odiyo, J.O. Evaluation of water quality and human risk assessment due to heavy metals in groundwater around Muledane area of Vhembe District, Limpopo Province, South Africa. Chem. Cent. J. 2018, 12, 2. [Google Scholar] [CrossRef]
- Edokpayi, J.N.; Odiyo, J.O.; Durowoju, O.S. Impact of wastewater on surface water quality in developing countries: A case study of South Africa. In Water Quality, Chapter: 18; Hlanganani, T., Ed.; IntechOpen: London, UK, 2017; pp. 401–416. [Google Scholar] [CrossRef]
- Odiyo, J.O.; Makungo, R. Chemical and Microbial Quality of Groundwater in Siloam Village, Implications to Human Health and Sources of Contamination. Int. J. Environ. Res. Public Health 2018, 15, 317. [Google Scholar] [CrossRef]
- Durowoju, O.S.; Odiyo, J.O.; Ekosse, G.E. Horizontal variation in trace elements and soil characteristics at Siloam and Tshipise geothermal springs, Limpopo Province, South Africa. Water SA 2016, 42, 694–702. [Google Scholar] [CrossRef]
- Raessler, M. The Arsenic Contamination of Drinking and Groundwaters in Bangladesh: Featuring Biogeochemical Aspects and Implications on Public Health. Arch. Environ. Contam. Toxicol. 2018, 75, 1–7. [Google Scholar] [CrossRef]
- Bai, L.; Wang, Y.; Guo, Y.; Zhou, Y.; Liu, L.; Yan, Z.; Li, F.; Xie, X. Health Risk Assessment Research on Heavy Metals Ingestion Through Groundwater Drinking Pathway for the Residents in Baotou, China. J. Environ. Health 2016, 78, 84–90. [Google Scholar]
- Quintana, G.C.; Mirlean, N. Groundwater Contamination by Mercury from the Aforetime Carroting Practice. Bull. Environ. Contam. Toxicol. 2018, 100, 839–842. [Google Scholar] [CrossRef]
- Navarro, A.; Font, X.; Viladevall, M. Groundwater Contamination by Uranium and Mercury at the Ridaura Aquifer (Girona, NE Spain). Toxics 2016, 4, 16. [Google Scholar] [CrossRef]
- An, X.; Cheng, Y.; Huang, M.; Sun, Y.; Wang, H.; Chen, X.; Wang, J.; Li, D.; Li, C. Treating organic cyanide-containing groundwater by immobilization of a nitrile-degrading bacterium with a biofilm-forming bacterium using fluidized bed reactors. Environ. Pollut. 2018, 237, 908–916. [Google Scholar] [CrossRef]
- Hynds, P.D.; Thomas, M.K.; Pintar, K.D.M. Contamination of Groundwater Systems in the US and Canada by Enteric Pathogens, 1990–2013: A Review and Pooled-Analysis. PLoS ONE 2014, 9, 93301. [Google Scholar] [CrossRef]
- Wallender, E.K.; Ailes, E.C.; Yoder, J.S.; Roberts, V.A.; Brunkard, J.M. Contributing factors to disease outbreaks associated with untreated groundwater. Groundwater 2014, 52, 886–897. [Google Scholar] [CrossRef]
- Hynds, P.D.; Misstear, B.D.; Gill, L.W. Development of a microbial contamination susceptibility model for private domestic groundwater sources. Water Resour. Res. 2012, 48. [Google Scholar] [CrossRef]
- Odiyo, J.O.; Chimuka, L.; Mamali, M.A.; Fatoki, O.S. Trophic status of Vondo and Albasini Dams; impacts on aquatic ecosystems and drinking water. Int. J. Environ. Sci. Technol. 2012, 9, 203–211. [Google Scholar] [CrossRef]
- Bessong, P.O.; Odiyo, J.O.; Musekene, J.N.; Tessema, A. Spatial Distribution of Diarrhoea and Microbial Quality of Domestic Water during an Outbreak of Diarrhoea in the Tshikuwi Community in Venda, South Africa. J. Health Popul. Nutr. 2009, 27, 652–659. [Google Scholar] [CrossRef]
- Obi, C.L.; Potgieter, N.; Bessong, P.O.; Matsaung, G. Assessment of the microbial quality of river water sources in rural Venda communities in South Africa. Water SA 2002, 28, 287–292. [Google Scholar] [CrossRef]
- Wanda, E.M.; Mamba, B.B.; Msagati, T.A. Determination of the water quality index ratings of water in the Mpumalanga and North West provinces, South Africa. Phys. Chem. Earth 2015, 92, 70–79. [Google Scholar] [CrossRef]
- Timmerman, J.G.; Beinat, E.; Termeer, K.; Cofino, W. Analyzing the data-rich but-information-poor syndrome in Dutch water management in historical perspective. Environ. Manag. 2010, 45, 1231–1242. [Google Scholar] [CrossRef]
- Boyacioglu, H. Development of a water quality index based on a European classification scheme. Water SA 2007, 33, 101–106. [Google Scholar] [CrossRef]
- Singaraja, C. Relevance of water quality index for groundwater quality evaluation: Thoothukudi District, Tamil Nadu, India. Appl. Water Sci. 2014, 7, 2157–2173. [Google Scholar] [CrossRef]
- Reza, R.; Singh, G. Heavy metal contamination and its indexing approach for river water. Int. J. Environ. Sci. Technol. 2010, 7, 785–792. [Google Scholar] [CrossRef]
- Rabeiy, R.E. Assessment and modeling of groundwater quality using WQI and GIS in Upper Egypt area. Environ. Sci. Pollut. Res. 2017. [Google Scholar] [CrossRef]
- Ramya Priya, R.; Elango, L. Evaluation of geogenic and anthropogenic impacts on spatio-temporal variation in quality of surface water and groundwater along Cauvery River, India. Environ. Earth Sci. 2018, 77. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, X.; Chen, Y.; Cai, Y.; Deng, J. Assessing river water quality using water quality index in Lake Taihu Basin, China. Sci. Total Environ. 2018, 612, 914–922. [Google Scholar] [CrossRef]
- Sutadian, A.D.; Muttil, N.; Yilmaz, A.G.; Perera, B.J.C. Development of a water quality index for rivers in West Java Province, Indonesia. Ecol. Indic. 2018, 85, 966–982. [Google Scholar] [CrossRef]
- APHA (American Public Health Association). Standard Methods for Examination of Water and Waste Water, 19th ed.; American Public Health Association: Washington, DC, USA, 1995; p. 1467. [Google Scholar]
- WHO. Guidelines for Drinking Water Quality: Training Pack; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Rao, G.S.; Nageswararao, G. Assessment of Groundwater quality using Water Quality Index. Arch. Environ. Sci. 2013, 7, 1–5. [Google Scholar]
- Sharma, P.; Meher, P.K.; Kumar, A.; Gautam, Y.P.; Mishra, K.P. Changes in water quality index of Ganges river at different locations in Allahabad. Sustain. Water Quak. Ecol. 2014, 3–4, 67–76. [Google Scholar] [CrossRef]
- Kawo, N.S.; Karuppannan, S. Groundwater quality assessment using water quality index and GIS technique in Modjo River Basin, central Ethiopia. J. Afr. Earth Sci. 2018, 147, 300–311. [Google Scholar] [CrossRef]
- Chourasia, L.P. Assessment of Groundwater Quality Using Water Quality Index in and Around Korba City, Chhattisgarh, India. Am. J. Softw. Eng. Appl. 2018, 7, 15–21. [Google Scholar] [CrossRef]
- Singh, S.; Hussian, A. Water quality index development for groundwater quality assessment of Greater Noida sub-basin, Uttar Pradesh, India. Cogent. Eng. 2016, 3, 1177155. [Google Scholar] [CrossRef]
- Packiyam, R.; Subha Pachaiyappan, S.; Narayanan, U. Effect of Storage Containers on Coliforms in Household Drinking Water. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 461–477. [Google Scholar] [CrossRef]
- Ezeh, O.K.; Agho, K.E.; Dibley, M.J.; Hall, J.; Page, A.N. The Impact of Water and Sanitation on Childhood Mortality in Nigeria: Evidence from Demographic and Health Surveys, 2003–2013. Int. J. Environ. Res. Public Health 2014, 11, 9256–9272. [Google Scholar] [CrossRef]
- Allam, R.R.; Uthappa, C.K.; Nalini, C.; Udaragudi, P.R.; Tadi, G.P.; Murhekar, M.V. An Outbreak of Cholera due to Contaminated Water, Medak District, Andhra Pradesh, India, 2013. Indian J. Community Med. Off. Publ. Indian Assoc. Prev. Soc. Med. 2015, 40, 283–285. [Google Scholar]
- World Health Organization (WHO). Guidelines for Drinking-Water Quality, 4th ed.; WHO: Geneva, Switzerland, 2011. [Google Scholar]
| Parameters | Assigned Weight | ||||
|---|---|---|---|---|---|
| pH | 4 | 4 | 4 | ND | 4 |
| Ca | 2 | 2 | 2 | 3 | 2 |
| Mg | 2 | 2 | 2 | 3 | 2 |
| Mn | ND | ND | ND | 5 | 5 |
| Cl | 3 | 3 | 1.0 | 5 | 1 |
| Cr | ND | ND | ND | 5 | 5 |
| Cu | ND | ND | ND | ND | 4 |
| Reference | Rao and Nageswararao [32] | Shamar et al [33] | Kawo and Karuppannan [34] | Chourasia [35] | This study |
| Parameter | WHO Standard [31] | Weight (wi) | Relative Weight (Wi) |
|---|---|---|---|
| pH | 6–8.5 | 4 | 0.1739 |
| Ca2+ | 75 | 2 | 0.0870 |
| Mn2+ | 0.4 | 5 | 0.2174 |
| Mg2+ | 50 | 2 | 0.0870 |
| Cu2+ | 2 | 4 | 0.1739 |
| Cr | 0.05 | 5 | 0.2174 |
| Cl− | 250 | 1 | 0.0435 |
| WQI Range | Water Quality |
|---|---|
| <50 | Excellent water |
| 50–100 | Good water |
| 100–200 | Poor water |
| 200–300 | Very poor water |
| >300 | Water unsuitable for drinking purpose |
| Sample Code | SW1 | SW2 | SW3 | SW4 | SW5 | SW6 | SW7 | GW1 | GW2 | GW3 | GW4 | GW5 | GW6 | GW7 | Control |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Faecal coliform | + | + | - | + | + | + | + | + | + | + | + | + | + | + | - |
| E. coli | + | + | - | + | + | + | + | + | + | + | + | + | + | + | - |
| Sampling Site | Water Quality | |
|---|---|---|
| SW1 | 27.44 | Excellent water |
| SW2 | 30.96 | Excellent water |
| SW3 | 25.28 | Excellent water |
| SW4 | 22.36 | Excellent water |
| SW5 | 33.59 | Excellent water |
| SW6 | 29.58 | Excellent water |
| SW7 | 23.34 | Excellent water |
| GW1 | 33.56 | Excellent water |
| GW2 | 157.94 | Poor water |
| GW3 | 69.29 | Good water |
| GW4 | 26.43 | Excellent water |
| GW5 | 26.26 | Excellent water |
| GW6 | 26.73 | Excellent water |
| GW7 | 29.99 | Excellent water |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olasoji, S.O.; Oyewole, N.O.; Abiola, B.; Edokpayi, J.N. Water Quality Assessment of Surface and Groundwater Sources Using a Water Quality Index Method: A Case Study of a Peri-Urban Town in Southwest, Nigeria. Environments 2019, 6, 23. https://doi.org/10.3390/environments6020023
Olasoji SO, Oyewole NO, Abiola B, Edokpayi JN. Water Quality Assessment of Surface and Groundwater Sources Using a Water Quality Index Method: A Case Study of a Peri-Urban Town in Southwest, Nigeria. Environments. 2019; 6(2):23. https://doi.org/10.3390/environments6020023
Chicago/Turabian StyleOlasoji, Samuel O., Nather O. Oyewole, Bayode Abiola, and Joshua N. Edokpayi. 2019. "Water Quality Assessment of Surface and Groundwater Sources Using a Water Quality Index Method: A Case Study of a Peri-Urban Town in Southwest, Nigeria" Environments 6, no. 2: 23. https://doi.org/10.3390/environments6020023
APA StyleOlasoji, S. O., Oyewole, N. O., Abiola, B., & Edokpayi, J. N. (2019). Water Quality Assessment of Surface and Groundwater Sources Using a Water Quality Index Method: A Case Study of a Peri-Urban Town in Southwest, Nigeria. Environments, 6(2), 23. https://doi.org/10.3390/environments6020023





