Abstract
(1) Background: With the new edition of the Diagnostic and Statistical Manual of Mental disorders, 5th Edition (DSM-5), numerous parallels have been drawn between recurrent binge eating (RBE) and substance use disorders, with many authors examining RBE or binge eating disorder (BED) as a “food addiction”. The present study aims to clarify the relationship between recurrent binge eating (RBE) and illicit substance use (ISU) through investigating the temporal association between the two problems. (2) Methods: This study was embedded within a larger longitudinal study of non-clinical adult women recruited from Australian tertiary institutions. Participants responded at year 2 and year 4 of follow-up to the Eating Disorder Examination—Questionnaire. ISU was measured using a modified questionnaire taken from the Australian Longitudinal Study on Women’s Health. (3) Results: RBE and ISU co-morbidity was 5.88% in this non-clinical sample, and having one condition increased the likelihood of the other. The two conditions had a different trajectory over two years whereby ISU participants had significant risk of developing RBE in addition to or in place of their ISU but the reverse was not found for RBE participants. (4) Conclusion: This unidirectional relationship suggests that in spite of the similarities of RBE and ISU they may be distinct with respect to their co-morbidity over time.
1. Introduction
1.1. Background
Binge eating disorder (BED) is characterised by recurrent episodes of binge eating (RBE)—defined by an objective overconsumption of food and a sense of loss of control—without the compensatory behaviours which define bulimia nervosa. BED has an estimated lifetime prevalence between 1.9% and 2.8% depending on the population surveyed, making it the most common eating disorder [1,2,3]. It is categorised within the Feeding and Eating Disorder (ED) chapter of the Diagnostic and Statistical Manual of Mental disorders, Fifth Edition (DSM-5), and is thus distinct from substance use disorders (SUDs). However, parallels have been drawn between BED and SUDs by a number of authors, many of them examining BED as a “food addiction” [1,2,3,4]. Criterion A for SUD within the DSM-5 may be divided into groupings of “impaired control, social impairment, risky use and pharmacological criteria” [5]. These are comparable to the BED criteria of “a sense of a lack of control”, eating alone due to embarrassment, ongoing overeating despite negative consequences, and eating large amounts of food when not physically hungry.
BED and SUD also share a number of psychological, neurobiological and genetic correlates. Factors such as neuroticism, impulsivity, sensation seeking and mood dysregulation are associated with both BED and SUD [6,7,8]. Animal models also support the theory that both BED and SUD follow from dysregulation of the same dopaminergic pathways [9,10] and have likewise been able to produce somatic withdrawal symptoms with sucrose cessation [11].
1.2. Co-Morbidity
Literature regarding the co-morbidity shared between RBE/BED and SUD supports the idea that there is an underlying shared pathology between the two conditions. In examining the literature, there are a number of classification issues leading to variability in reported rates of co-morbidity and prior to its recognition in the DSM-5 [5], BED was included as a type of Eating Disorders Not Otherwise Specified (EDNOS) [12]. Complicating matters further, numerous studies either have failed to specify the type of ED they had studied or have classified participants with inconsistent criteria [13]. For many studies RBE has been used to represent BED. For example, in a US national face-to-face survey of 9282 adults, 23% of those who had BED—which was defined as having 3 months or more of RBE—suffered also from a type of SUD [14]. The WHO World Mental Health Surveys support these findings, in which 23.7% of those with BED would have some form of SUD [15]. In exploring the prevalence of SUD, it is noted that the classification of substance abuse is similarly difficult; research in this field varies not only in the scope of substance abuse, ranging from a focus on a single substance to looking at SUDs collectively, but also the severity of substance abuse, studying one-time use as well as physiological dependence [13,16]. Harrop & Marlatt’s review [13] reflects these classification inconsistencies with co-morbidity prevalence ranging from 17–46% depending on ED and SUD types.
Illicit Substance Use (ISU) appears to be more common in ED populations than healthy controls; however, more information is needed to clarify the relationship between different types of illicit drugs and ED subgroups [16]. Cannabis [17,18] and opiate [17] use have found to be increased in those with an ED (subgroups combined) compared to controls. Evidence regarding amphetamine usage is inconsistent; one author reports associations of amphetamine usage with dieting and purging behaviour (without binging) [19,20] whilst another did not find increased use of amphetamines when comparing an ED group with the general population [17]. These findings may suggest that amphetamine usage may be associated with dieting and purging rather than with binging behaviour [16].
1.3. Longitudinal Predictors
On the other hand, longitudinal studies seem to suggest that however many similarities there may be between RBE/BED and ISU, they seem to differ in illness trajectories. A five-year longitudinal study of adolescent girls found that depressive symptoms were predictive of higher future levels of eating pathology and substance abuse (broadly defined and including alcohol use); eating pathology itself also predicted increased future substance abuse, with the inverse not being true [21]. Similarly, an Australian cohort study of adolescents and young adults found that even partial anorexia nervosa (AN)/bulimia nervosa (BN) diagnosis (where a participant satisfied two of four or three criteria for AN/BN) was predictive of amphetamine use [22]. The Growing Up Today Study has found that overeating (without a loss of control (LOC)) and RBE (overeating with a sense of a LOC) were both predictors for ISU; however, overeating alone was a stronger predictor for this outcome [23].
Fewer longitudinal papers have focused on predictors of RBE. Vogeltanze-Holm et al., found that the main factors predicting BED (strictly defined) was ISU in the past 12 months (odds ratio (OR) = 5.77, 95% CI = 1.64, 20.34) and more occasions of alcohol use until intoxication in the past 12 months (OR = 1.38, 95% CI = 1.03, 1.85) [24].
Finally, a five-year longitudinal study documenting the natural history of a variety of behavioural addictions over this period found a central effect of time on the problem behaviours, where the prevalence of the behaviours decreased and often resolved without intervention [25]. Excessive eating (examined over four years only) was found to decrease in prevalence at the same rate as comorbid substance use (broadly defined), with a mean 11.7% (SD = 2.3) suffering from comorbid SUD during the four-year time period.
These findings—in particular, that RBE/BED and ISU/SUD may not mutually predict risk for each other—suggest that perhaps distinct higher-order factors are mediating the relationship between RBE/BED and ISU/SUD rather than being controlled by the same underlying factor [13]. In a review of the phenomenology and treatment of behavioural addictions, Grant et al. hypothesises the opposite [26], claiming that one neurobiological dysfunction could give rise to multiple behavioural symptoms. The support for this theory comes from “consummatory cross-sensitisation” where prolonged intake and sensitisation with one substance can lead to increased consumption of another [4]. As a result of this cross-sensitisation, opiate- and stimulant-dependent individuals may have a cross-substitutability of preference for highly palatable foods, leading to reported cravings and binges [9,27].
Although there have been these studies of outcomes and putative symptoms substitution, it is notable that there have been few studies of the impact of comorbidity on other clinical features such as overall psychological distress, health-related quality of life and/or body weight, and findings have been mixed or inconsistent [28]. This may be of clinical importance if co-morbidity was found to be associated with poorer mental health and/or increased likelihood of obesity.
1.4. Aim and Hypotheses
In this study, we aimed to elucidate the nature of co-morbidity by (1) characterising the extent of the overlap of these two features within a non-clinical population, and (2) examining the trajectories of participants with regard to RBE and ISU over a period of two years’ time. We hypothesised that there would be significant co-morbidity between the two problems and, furthermore, that participants with RBE and those with ISU will have differing illness trajectories without mutual substitution between the two behaviours. We did not have specific hypotheses in regard to examining general psychological distress or health-related quality of life as these have been little studied in regard to the comorbidity of ISU and RBE.
2. Materials and Methods
2.1. Participants
Participants were 794 women initially recruited in 2004/2005 who were assessed repeatedly over a nine-year period (T0–T9). Any one follow-up assessment was not contingent on having competed any other follow-up. They were recruited through advertisements in four regional universities and vocational colleges (including adult students) in the Australian states of Queensland and Victoria for the purpose of a longitudinal study of community (non-clinical) women with and without eating disorder symptoms. Some participants were recruited via email and responded to the questionnaire online, whilst others were directly approached on campus locations and given hard-copy questionnaires and reply-paid envelopes. Due to these recruitment methods, characteristics of non-responders and overall response rate could not be measured.
ISU was only assessed in T2 and T4 of the longitudinal study. As such, the present study comprises of participants who responded in T2 (n = 357) and those who responded in T4 who also responded in T2 (n = 268). Respondents who had no data for measures of binge eating or substance use in T2 (n = 4) or T4 (n = 9) were excluded. Figure 1 shows the participant flow through T1, T2 and T4 of the longitudinal study.
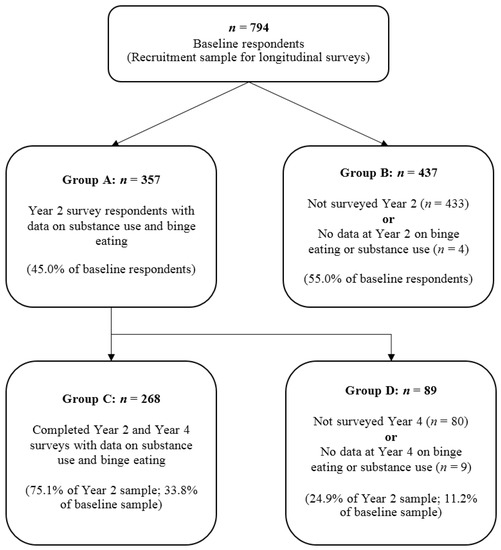
Figure 1.
Participant flow from the beginning of the survey.
2.2. Procedures
The study was approved by the human research ethics committees (HREC) of the universities involved, with the Western Sydney University as lead HREC (Approval number 07/240). All participants completed written informed consent forms and there were no children requiring consent from a parent or guardian.
2.3. Measures
2.3.1. Binge Eating
The Eating Disorder Examination Questionnaire (EDE-Q), a self-report questionnaire based on the Eating Disorder Examination (EDE) interview was used in order to assess eating disorder psychopathology. The EDE-Q has been validated in both community and clinical samples of patients with eating disorders and demonstrates close agreement with the EDE overall [29]. However, with regard to the complex features involved with binge eating behaviours the EDE-Q consistently generated higher levels of disturbance relative to the EDE [30].
Four items in the questionnaire targeted binge eating behaviours. The first two assessed objective binge eating (OBE), asking if the respondent had ever consumed what other people would regard as an unusually large amount of food, with a sensation of loss of control, and if so, how many times that had occurred over the past 28 days. This correlates with the DSM-5 criteria for recurrent binge eating episodes (Criterion A) but does not specify a discrete time period [5]. The second two assessed subjective binge eating (SBE), asking if the respondent had consumed a normal amount of food for the circumstances but had experienced a loss of control, and if yes, the number of times that had occurred. This is consistent with binge eating criteria being considered for the incoming International Classification of Diseases, 11th revision (ICD-11) [31], on the basis that people with subjective episodes have similar levels of impairment and distress related to binge eating as well as other psychopathology as do people with objective episodes [32,33]. As such, binge eating was coded as “present” if they had reported “yes” to either an objective or subjective binge for more than four episodes in the past 28 days and is used as the measure in this study of RBE. At the end of the EDE-Q there was a question asking current weight and height from which body mass index (BMI; kg/m2) scores were derived.
2.3.2. Illicit Substance Use (ISU)
The questionnaire assessed frequency and amount of use of the following illicit substances: cannabis, amphetamines, hallucinogens, barbiturates, “ecstasy/designer drugs/cocaine”, “inhalants” and heroin. Participants were asked if they had used any of the illicit substances listed above in the past year, and if yes, the frequency of their current use over a one-month period. These questions were modified from the Australian Longitudinal Study on Women’s Health (ALSWH), where their frequency of use was categorised into scores of: 1 = less than monthly, 2 = monthly, 3 = weekly, 4 = two to three times per week, and 5 = daily [34]. The ordinal data gathered by this questionnaire allowed the creation of a new variable for the present study measuring overall ISU, which was calculated by taking the sum of the scores for each of the seven drug categories. A score of zero indicated no ISU, whilst the maximum score of thirty-five indicated that the participant was taking illicit drugs in all seven categories every single day of the week. The score is therefore influenced both by the range of illicit drugs consumed, as well as their frequency. For the purpose of this study, ISU was coded as present if the score was greater than 0, i.e., 1 or more.
2.3.3. Psychological Distress
This was assessed with the Kessler-10 item distress scale (K-10) which was designed to detect cases of anxiety and affective disorders in the general population [35]. It is a 10-item instrument with an ordinal 5-point response to each question. It measures the level of distress and severity associated with psychological symptoms of depression and anxiety. The K10 is extensively used internationally, including in the WHO World Mental Health Survey and by government organizations in Australia, Spain, Colombia and Peru [36]. The advantages of the K-10 are its brief nature and its strong psychometric properties. It focuses on the previous 28 days thus is comparable in time-frame to the EDE-Q.
2.3.4. Health-Related Quality of Life (HRQoL)
HRQoL was assessed with the well-validated 12-item Short Form-12 Health Status Questionnaire (SF-12) [37]. The SF-12 measures the impact of physical and mental ill-health on role limitations. It has been used extensively in research assessing impairment associated with physical and mental health conditions, and has robust psychometric properties, including in an Australian population sample [37,38]. It is a 12-item questionnaire that generates two weighted scales, a Physical Component Summary Scale (PCS) and a Mental Component Summary Scale (MCS), with each a mean of 50 and standard deviation of 10 in normative samples. Higher scores indicate higher levels of functioning.
2.4. Statistical Methods
Data were inspected for normality. Descriptive statistics were employed to report frequencies of socio-demographic variables, general symptoms, binge eating and substance use. Between-group differences were compared using ANOVA with post-hoc Tukey analyses for continuous normal data and Kruskal–Wallis and Mann–Whitney U tests for continuous non-normal data. The chi-squared test was utilised to test differences in distribution between categorical groups and ordinal data. Fisher’s exact test was utilised to calculate the p-value for the contingency tables given the small sample size. To determine whether a trajectory based on a transition from year 2 to year 4 was statistically significant, we tested the significance of estimated marginal probability for each trajectory based on the multinomial logistic regression of a multi-category outcome containing all possible combinations of RBE & ISU measured at year 4 conditional on the same outcome at year 2, while controlling for age and mental health-related quality of life both being measured at year 2. While assessing the relationship between a multi-category outcome containing all possible combinations of RBE & ISU measured at year 2 & the same outcome measured at year 4, both the control variables were found to be confounders. Listwise deletion of missing data was applied to the data at year 4 because the percentage missing at time 4 out of a total of 359 cases at time 2 who had any of the four possible RBE & ISU conditions was very low (3.06%), and hence complete case analysis would introduce very little bias. A significance level of p < 0.05 was employed for all tests. Analyses were conducted using IBM SPSS Statistics for Windows, version 22.
3. Results
3.1. Participant Features
Of the 357 participants who completed the follow-up survey at T2 (45.0% of baseline respondents), the median age (at T2) was 25 (Interquartile Range (IQR) = 15), 58.0% were unmarried or separated and 52.9% lived with family, friends or alone. The sample was well educated, with 33.9% achieving at least year 12, and 48.5% attaining a bachelor’s degree or higher. A large minority of the sample studied full-time (41.5%). Participants with symptoms were overrepresented in this study sample; compared to a previous general population study of Australian women, their Mental Health-Related Quality of Life scores (Short-Form 12 Mental Health Component Scores or SF-12 MCS) were lower and their EDE-Q subscale and global scores were higher, although lower than in clinical samples [39]. (See Section 2.3 for descriptors of these assessment measures.) ISU occurred in 20% (n = 72) of participants. Cannabis (n = 59, 82%) was the most frequently used substance followed by ecstasy/designer drugs/cocaine (n = 40, 56%). Other demographic and clinical features of the 357 participants at T2 can be found in Table 1.

Table 1.
Descriptive data of 357 study participants in the present study.
Table 2 compares key characteristics of the four subgroups within the longitudinal study to assess if respondents were significantly different from non-respondents at T2 and T4. These are divided by response status and availability of RBE and ISU data. These groups are also outlined in Figure 1.

Table 2.
Participant characteristics of subgroups within the study.
Year 2 respondents (Group A) were significantly older (MD = 2.23, SE = 0.74) than those who responded at baseline but were lost to follow-up (Group B), but were not significantly different in the other measures of body mass index (BMI (kg/m2)) and RBE characteristics. Participants who followed up at both year 2 and year 4 (Group C) were also significantly older (MD = 3.79, SE = 1.38) than their counterparts who did not respond in year 4 (Group D) and similarly, were not significantly different in BMI, RBE or ISU behaviours.
3.2. Co-Morbid ISU and RBE in the T2 Cohort
At T2, 226 of 357 (63.3%) respondents had neither RBE nor ISU behaviours; 55 (15.4%) had episodes of RBE only; the same number (n = 55, 15.4%) engaged in ISU only, whilst 21 participants (5.88%) in T2 admitted to engaging in both behaviours.
The majority of participants who were identified as having a problem (either RBE or ISU) had one problem only and not the other (55/76, 72.4%) and this finding was not significant (χ2 = 2.32, df = 1; p = 0.09). As shown in Table 3 it was determined that those who had RBE had significantly higher frequency of ISU compared to those without RBE, and similarly, those who had ISU had significantly higher frequency of RBE compared to those without ISU.

Table 3.
Comparative levels of illicit substance use (ISU) and recurrent binge eating (RBE) in participants with and without either problem.
Furthermore, participants with both ISU and RBE had the highest levels of eating disorder symptoms (global and subscale EDE-Q scores) and psychological distress (K-10 scores) and lowest levels of mental health HRQoL. These differences reached significance only for the findings of global EDE-Q scores compared to those with ISU alone, and K-10 scores compared to those with neither problem. Those with neither problem also had significantly lower EDE-Q global scores than all other groups and lower K-10 scores than those with RBE alone. These differences are shown in Table 4.

Table 4.
Comparative clinical features of participants according to their RBE and ISU status.
3.3. Participant Trajectories from T2 to T4
As shown in Table 5, the majority (n = 139, 82.2%) of participants with neither RBE nor ISU in T2 continued to have neither problem in T4, and 12% (n = 21) developed RBE. Almost half (n = 6, 46.2%) of those with both problems in T2 continued to have both problems in T4. Almost half (n = 18, 46.2%) of those with ISU in Year 2 continued to have ISU in year 4 and n = 7 (17.9%) developed an additional problem with RBE and n = 5 (12.8%) transitioned to RBE alone. The majority (n = 26, 57.8%) of those with RBE in T2 had neither problem in T4 and n = 17 (37.8%) continued to have RBE alone.

Table 5.
Longitudinal movement of participants between groups (n = 266).
As shown in Table 6, participants with neither RBE nor ISU at year 2: were significantly more likely (p < 0.001) to remain that way or have RBE only by year 4, were significantly more likely (p < 0.01) to have ISU only by year 4, but were not significantly more likely to have both RBE and ISU by year 4. The most likely trajectory for those who were neither RBE nor ISU at year 2 was to remain that way by year 4. Participants being both RBE and ISU at year 2: were significantly more likely (p < 0.05) to have ISU only or both RBE & ISU by year 4, but were not significantly more likely to have RBE only or neither RBE nor ISU by year 4. The most likely trajectory for those who were both RBE and ISU at year 2 was to remain that way by year 4. Participants being RBE only at year 2 on the contrary: were significantly more likely (p < 0.05) to have RBE only or neither RBE nor ISU by year 4, but were not significantly more likely to have ISU only or both RBE & ISU by year 4. The most likely trajectory for those who were RBE only at year 2 was to have neither RBE nor ISU by year 4. For participants being ISU only at year 2 all transitions to year 4 were statistically significant with the most likely trajectory being both ISU only at year 2 and year 4.

Table 6.
Estimated marginal probability with 95% confidence interval for each trajectory from year 2 to year 4 based on multinomial logistic regression controlling for age and mental health-related quality of life.
4. Discussion
The current study investigated the relationship between RBE and ISU in a sample of Australian non-clinical adult women. The co-occurrence of ISU and RBE was examined cross-sectionally and then longitudinally over two years.
4.1. Comorbid Psychopathology
The hypothesis that RBE and ISU co-occur in the setting of a broader community sample was confirmed. Our study found that those with RBE had a higher frequency of ISU as well as the inverse, i.e., those with ISU had higher frequency of RBE. This co-morbidity might be explained by common neurobiological pathways involved in the two conditions [3,4] or may be a reflection of a self-mediated attempt at regulating negative affect [40], as demonstrated by Killeen et al., who found that past 30 day opiate use was correlated with increased EDE-Q scores [41]. Furthermore, our findings support those of Grilo et al. [42], which found that patients with BED with another concurrent psychiatric disorder had elevated levels of eating disorder psychopathology, although in this study this did not reach significance possibly because of small numbers of those with both problems.
4.2. Participant Trajectories and Between-Group Associations
Results from comparing participant numbers as they moved through from year 2 to year 4 demonstrated that participants with ISU were more likely to develop RBE either in addition to, or in place of their ISU, whereas those with RBE were likely to remain unchanged or spontaneously resolve over time, supporting our hypothesis that the two conditions take unique temporal courses and are differentially predictive for each other. Whist our findings are theoretically supportive of the existing literature in distinguishing RBE/BED and ISU/SUD, there are some differences in results. Measelle et al.’s [21] longitudinal study was similar in part to the present study and focussed on a variety of psychiatric disorders in adolescent girls and the temporal associations between symptom domains; in their study they established that there was a unidirectional relationship between BED and SUD—however, in their case pre-existing eating pathology predicted future growth in substance abuse but not the reverse—the opposite conclusion to this present study. This difference might be because of the shorter duration of this study, that Measelle et al. studied substance use more broadly and included alcohol abuse, and that we were studying subthreshold syndromes. Our findings however support the longitudinal findings of Vogeltanz-Holm et al. [24] that the main predictors for BED are ISU and alcohol intoxication. Furthermore, cessation of drug abuse followed by hyperphagia and weight gain is an established phenomenon in human studies [43] and animal models [44], although whether or not this disordered eating persists and develops into RBE/BED is a matter requiring further investigation.
4.3. Strengths and Limitations
The main strengths of this study include the reasonable sample size (n = 268) for the trajectory analysis and a 75.1% rate of retention of participants over the two-year follow-up period. However, the low numbers of those with both ISU and RBE may have limited finding statistical significance. The longitudinal design of the paper adds robustness to the findings presented in the study. However, the voluntary nature of recruitment and follow-up resulted in only 33.8% of baseline respondents being included for analysis, possibly contributing to elevated findings of eating disorder and ISU. Notably, we did not have a full assessment of the criteria for either BED or SUD, or more detailed assessment of RBE over a longer time frame, and did not assess for legal SUDs. Thus, we turned our focus to ISU and did not include legal substances such as alcohol and tobacco on the presumption that the act of breaching the law and risking the consequences of such more strongly implicates disordered substance use. Another important limitation is the non-inclusion of men as they have significantly higher rates of alcohol and drug use disorders; inclusion might produce altered co-morbidity rates and differing trajectories [45]. Further limitations include the single follow-up, self-report assessments of symptoms and BMI.
4.4. Clinical Implications
The key take-away from this study is that many participants with ISU went on to develop RBE, whilst those with RBE had a tendency for their behaviour to resolve over time. This is a relevant piece of information for clinicians in practice as it suggests that early assessment, monitoring and appropriate early intervention is important for individuals with ISU, and that this occurred despite the low threshold for defining ISU in this research. Finally, despite the stated differences between RBE and ISU, taking an addiction framework towards BED may improve current interventions or instigate the development of new treatments [8].
4.5. Future Directions
Studies investigating predictors for binge eating and its temporal associations are limited and, as such, further mixed-gender longitudinal studies conducted over longer periods of time would be warranted to clarify the associations between RBE and ISU. Other relevant aspects inviting possible future study include the investigation of alcohol or tobacco usage and binge eating behaviour as these commonplace drugs are also frequently consumed in excess. As a final point of interest, within the DSM-5, gambling and other behavioural addictions have been included within the same section as SUDs [46]. Given the co-morbidity between BED and SUD, it would be relevant to also consider the possible neurobiological and symptomatic correlates between full-threshold BED and other addictive disorders. It should be kept in mind as well that BED may be a construct distinct from the entity of “food addiction” which may present more similarities with SUD, and as such, further research is required in this area.
5. Conclusions
In this study, RBE and ISU have been found to be comorbid conditions in a non-clinical sample of young adult women, and furthermore, each condition increased the frequency of episodes of the other. Despite their similarities, the two conditions had a diverse trajectory over time, whereby ISU participants had higher likelihood of later developing RBE co-morbidly or in substitution but the reverse was not found for RBE participants. Further studies are indicated of full-spectrum BED and SUDs.
Acknowledgments
The longitudinal research was funded by a grant from the Australian Rotary Health Research Fund to P.H. and colleagues. H.K.L. was supported by a summer research scholarship from the School of Medicine, Western Sydney University.
Author Contributions
H.K.L. and P.H. contributed to the conception, design and aims of the study. H.K.L., H.M. and P.H. conducted the data analysis. H.K.L. drafted the manuscript. All authors read and approved the final manuscript.
Conflicts of Interest
H.K.L. declares no conflict of interest. P.H. receives sessional fees and lecture fees from the Australian Medical Council, Therapeutic Guidelines publication, and New South Wales Institute of Psychiatry and royalties from Hogrefe and Huber, McGraw Hill Education, and Blackwell Scientific Publications, and she has received research grants from the NHMRC and ARC. She is Deputy Chair of the National Eating Disorders Collaboration steering committee in Australia (2012–2017).
References
- Cassin, S.E.; von Ranson, K.M. Is binge eating experienced as an addiction? Appetite 2007, 49, 687–690. [Google Scholar] [CrossRef] [PubMed]
- Gearhardt, A.N.; White, M.A.; Potenza, M.N. Binge eating disorder and food addiction. Curr. Drug Abuse Rev. 2011, 4, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, L.R.; Odlaug, B.L.; Grant, J.E. The overlap between binge eating disorder and substance use disorders: Diagnosis and neurobiology. J. Behav. Addict. 2013, 2, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.G.; Robbins, T.W. The neurobiological underpinnings of obesity and binge eating: A rationale for adopting the food addiction model. Biol. Psychiatry 2013, 73, 804–810. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Feeding and Eating Disorders. In Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Grant, J.E.; Potenza, M.N.; Weinstein, A.; Gorelick, D.A. Introduction to behavioral addictions. Am. J. Drug Alcohol. Abuse 2010, 36, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Ferriter, C.; Ray, L.A. Binge eating and binge drinking: An integrative review. Eat Behav. 2011, 12, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Schulte, E.M.; Grilo, C.M.; Gearhardt, A.N. Shared and unique mechanisms underlying binge eating disorder and addictive disorders. Clin. Psychol. Rev. 2016, 44, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Avena, N.M.; Rada, P.; Hoebel, B.G. Evidence for sugar addiction: Behavioral and neurochemical effects of intermittent, excessive sugar intake. Neurosci. Biobehav. Rev. 2008, 32, 20–39. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.M.; Kenny, P.J. Dopamine D2 receptors in addiction-like reward dysfunction and compulsive eating in obese rats. Nat. Neurosci. 2010, 13, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Avena, N.M.; Rada, P.; Hoebel, B.G. Sugar and fat bingeing have notable differences in addictive-like behavior. J. Nutr. 2009, 139, 623–628. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Feeding and Eating Disorders. In Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington, DC, USA, 1994. [Google Scholar]
- Harrop, E.N.; Marlatt, G.A. The comorbidity of substance use disorders and eating disorders in women: Prevalence, etiology, and treatment. Addict. Behav. 2010, 35, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.I.; Hiripi, E.; Pope, H.G., Jr.; Kessler, R.C. The prevalence and correlates of eating disorders in the National Comorbidity Survey Replication. Biol. Psychiatry 2007, 61, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; Berglund, P.A.; Chiu, W.T.; Deitz, A.C.; Hudson, J.I.; Shahly, V.; Aguilar-Gaxiola, S.; Alonso, J.; Angermeyer, M.C.; Benjet, C.; et al. The prevalence and correlates of binge eating disorder in the World Health Organization World Mental Health Surveys. Biol. Psychiatry 2013, 73, 904–914. [Google Scholar] [CrossRef] [PubMed]
- Gregorowski, C.; Seedat, S.; Jordaan, G.P. A clinical approach to the assessment and management of co-morbid eating disorders and substance use disorders. BMC Psychiatry 2013, 13, 289. [Google Scholar] [CrossRef] [PubMed]
- Calero-Elvira, A.; Krug, I.; Davis, K.; Lopez, C.; Fernandez-Aranda, F.; Treasure, J. Meta-analysis on drugs in people with eating disorders. Eur. Eat. Disord. Rev. J. Eat. Disord. Assoc. 2009, 17, 243–259. [Google Scholar] [CrossRef] [PubMed]
- Root, T.L.; Pisetsky, E.M.; Thornton, L.; Lichtenstein, P.; Pedersen, N.L.; Bulik, C.M. Patterns of co-morbidity of eating disorders and substance use in Swedish females. Psychol. Med. 2010, 40, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Piran, N.; Robinson, S.R. Associations between disordered eating behaviors and licit and illicit substance use and abuse in a university sample. Addict. Behav. 2006, 31, 1761–1775. [Google Scholar] [CrossRef] [PubMed]
- Piran, N.; Robinson, S.R. Patterns of associations between eating disordered behaviors and substance use in two non-clinical samples: A university and a community based sample. J. Health Psychol. 2011, 16, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Measelle, J.R.; Stice, E.; Hogansen, J.M. Developmental trajectories of co-occurring depressive, eating, antisocial, and substance abuse problems in female adolescents. J. Abnorm. Psychol. 2006, 115, 524–538. [Google Scholar] [CrossRef] [PubMed]
- Patton, G.C.; Coffey, C.; Carlin, J.B.; Sanci, L.; Sawyer, S. Prognosis of adolescent partial syndromes of eating disorder. Br. J. Psychiatry 2008, 192, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Sonneville, K.R.; Horton, N.J.; Micali, N.; Crosby, R.D.; Swanson, S.A.; Solmi, F.; Field, A.E. Longitudinal associations between binge eating and overeating and adverse outcomes among adolescents and young adults: Does loss of control matter? JAMA Pediatr. 2013, 167, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Vogeltanz-Holm, N.D.; Wonderlich, S.A.; Lewis, B.A.; Wilsnack, S.C.; Harris, T.R.; Wilsnack, R.W.; Kristjanson, A.F. Longitudinal predictors of binge eating, intense dieting, and weight concerns in a national sample of women. Behav. Ther. 2000, 31, 221–235. [Google Scholar] [CrossRef]
- Konkoly Thege, B.; Woodin, E.M.; Hodgins, D.C.; Williams, R.J. Natural course of behavioral addictions: A 5-year longitudinal study. BMC Psychiatry 2015, 15, 4. [Google Scholar] [CrossRef] [PubMed]
- Grant, J.E.; Schreiber, L.R.; Odlaug, B.L. Phenomenology and treatment of behavioural addictions. Can J. Psychiatry 2013, 58, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Cowan, J.; Devine, C. Food, eating, and weight concerns of men in recovery from substance addiction. Appetite 2008, 50, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Long, C.G.; Blundell, J.E.; Finlayson, G. A Systematic Review of the Application And Correlates of YFAS-Diagnosed ‘Food Addiction’ in Humans: Are Eating-Related ‘Addictions’ a Cause for Concern or Empty Concepts? Obes. Facts 2015, 8, 386–401. [Google Scholar] [CrossRef] [PubMed]
- Fairburn, C.G.; Beglin, S.J. Assessment of eating disorders: Interview or self-report questionnaire? Int. J. Eat. Disord. 1994, 16, 363–370. [Google Scholar] [PubMed]
- Wilfley, D.E.; Schwartz, M.B.; Spurrell, E.B.; Fairburn, C.G. Assessing the specific psychopathology of binge eating disorder patients: Interview or self-report? Behav. Res. Ther. 1997, 35, 1151–1159. [Google Scholar] [CrossRef]
- Al-Adawi, S.; Bax, B.; Bryant-Waugh, R.; Claudino, A.M.; Hay, P.; Monteleone, P.; Norring, C.; Pike, K.M.; Pilon, D.J.; Herscovici, C.R.; et al. Revision of ICD—Status update on feeding and eating disorders. Adv. Eat. Disord. 2013, 1, 10–20. [Google Scholar] [CrossRef]
- Grilo, C.M.; White, M.A. A controlled evaluation of the distress criterion for binge eating disorder. J. Consult. Clin. Psychol. 2011, 79, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, B.E.; Baker, C.W.; Smith, A.T.; Kelly-Weeder, S. Validity and utility of the current definition of binge eating. Int. J. Eat. Disord. 2009, 42, 674–686. [Google Scholar] [CrossRef] [PubMed]
- Womens Health Australia. The Australian Longitudinal Study on Women’s Health: Data Book, 2nd ed.; Womens Health Australia: Newcastle, UK, 1997. [Google Scholar]
- Andrews, G.; Slade, T. Interpreting scores on the Kessler Psychological Distress Scale (K10). Aust. N. Z. J. Public Health 2001, 25, 494–497. [Google Scholar] [CrossRef] [PubMed]
- Vargas Terrez, B.E.; Villamil Salcedo, V.; Rodríguez Estrada, C.; Pérez Romero, J.; Cortés Sotres, J. Validación de la escala Kessler 10 (K-10) en la detección de depresión y ansiedad en el primer nivel de atención. Propiedades psicométricas. Salud Ment. 2011, 34, 323–331. [Google Scholar]
- Ware, J., Jr.; Kosinski, M.; Keller, S.D. A 12-Item Short-Form Health Survey: Construction of scales and preliminary tests of reliability and validity. Med. Care 1996, 34, 220–233. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, K.; Andrews, G. The SF-12 in the Australian population: Cross-validation of item selection. Aust. N. Z. J. Public Health 2002, 26, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Mond, J.M.; Hay, P.J.; Rodgers, B.; Owen, C.; Beumont, P.J. Validity of the Eating Disorder Examination Questionnaire (EDE-Q) in screening for eating disorders in community samples. Behav. Res. Ther. 2004, 42, 551–567. [Google Scholar] [CrossRef]
- Davis, C.; Carter, J.C. Compulsive overeating as an addiction disorder. A review of theory and evidence. Appetite 2009, 53, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Killeen, T.; Brewerton, T.D.; Campbell, A.; Cohen, L.R.; Hien, D.A. Exploring the relationship between eating disorder symptoms and substance use severity in women with comorbid PTSD and substance use disorders. Am. J. Drug Alcohol. Abuse 2015, 41, 547–552. [Google Scholar] [PubMed]
- Grilo, C.M.; White, M.A.; Masheb, R.M. DSM-IV psychiatric disorder comorbidity and its correlates in binge eating disorder. Int. J. Eat. Disord. 2009, 42, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Edge, P.J.; Gold, M.S. Drug withdrawal and hyperphagia: Lessons from tobacco and other drugs. Curr. Pharm. Des. 2011, 17, 1173–1179. [Google Scholar] [CrossRef] [PubMed]
- Orsini, C.A.; Ginton, G.; Shimp, K.G.; Avena, N.M.; Gold, M.S.; Setlow, B. Food consumption and weight gain after cessation of chronic amphetamine administration. Appetite 2014, 78, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Merikangas, K.R.; McClair, V.L. Epidemiology of substance use disorders. Hum. Genet. 2012, 131, 779–789. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Substance-Related and Addictive Disorders. In Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).