Barocrinology: The Endocrinology of Obesity from Bench to Bedside
Abstract
1. Introduction
2. Endocrine Causes of Obesity
Syndromic Obesity
3. Endocrine Hormone Levels in Obesity and Weight Loss
4. Endocrine Evaluation of obesity
4.1. Endocrine Condition: Laboratory Evaluation: Do’s and Don’ts
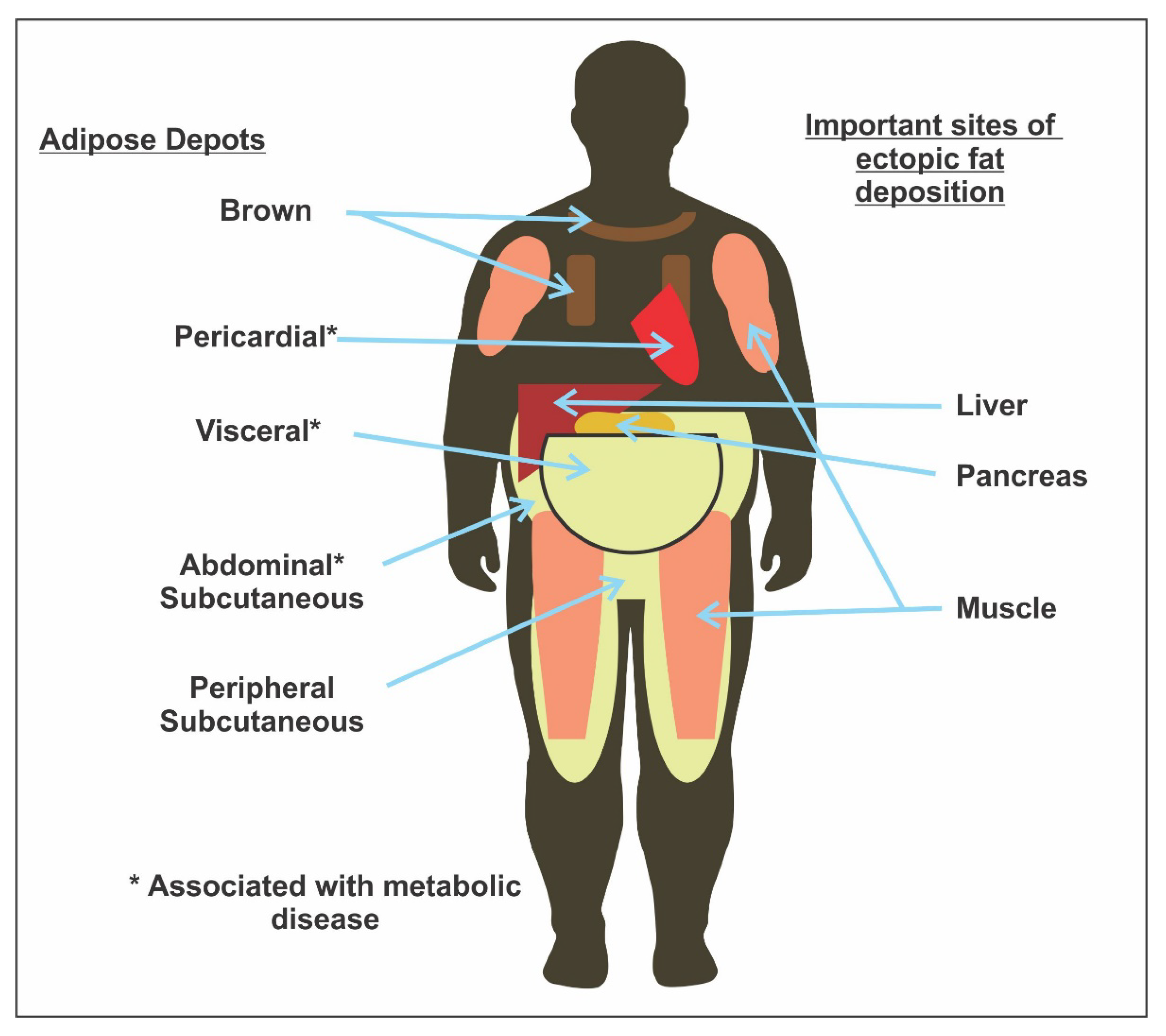
4.2. Anthropometric Evaluation in Endocrinopathy
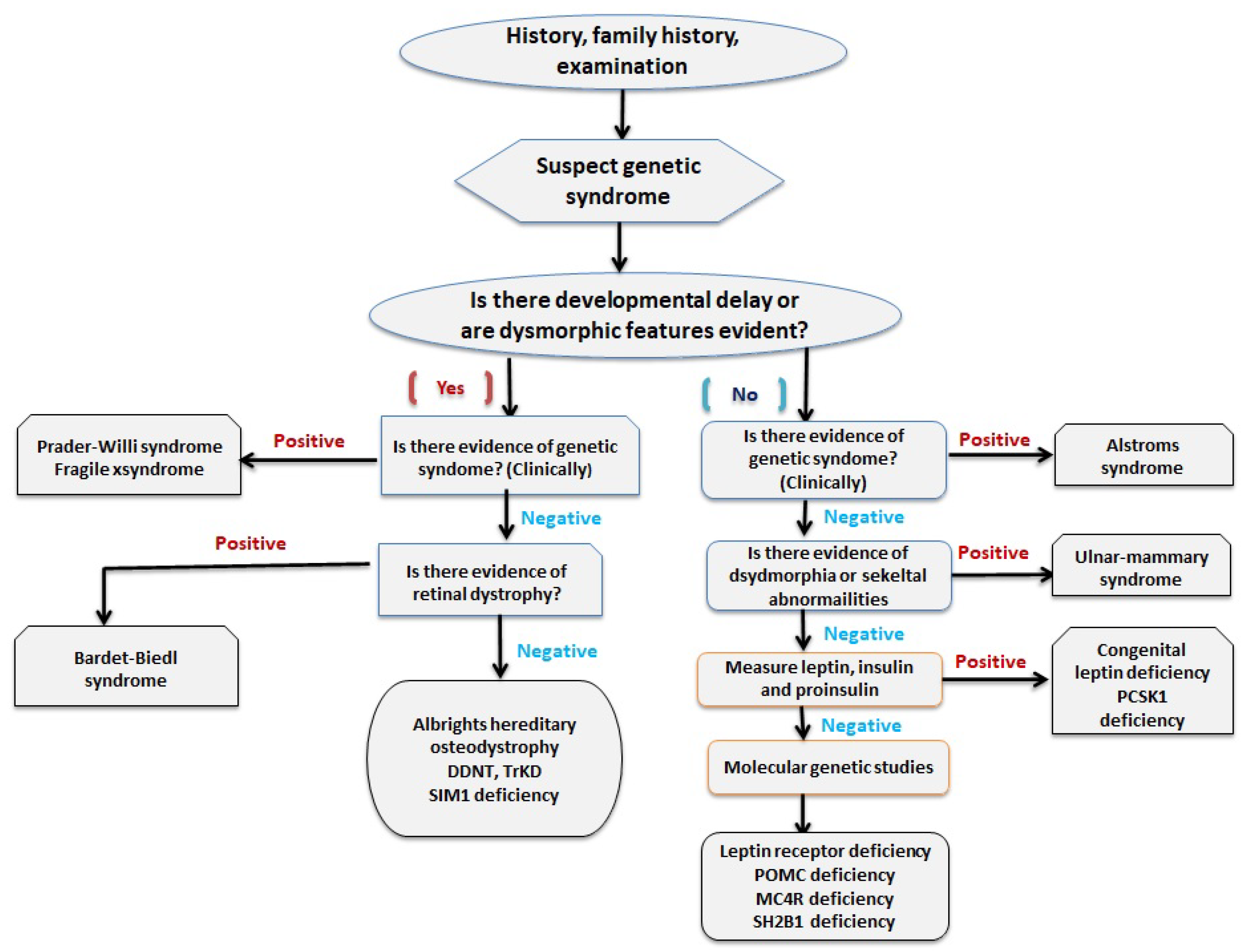
4.3. Approach to Evaluating Syndromic Obesity
5. Endocrine Impact of Obesity Management
5.1. Endocrine Impact of Non-Pharmacological Management of Obesity
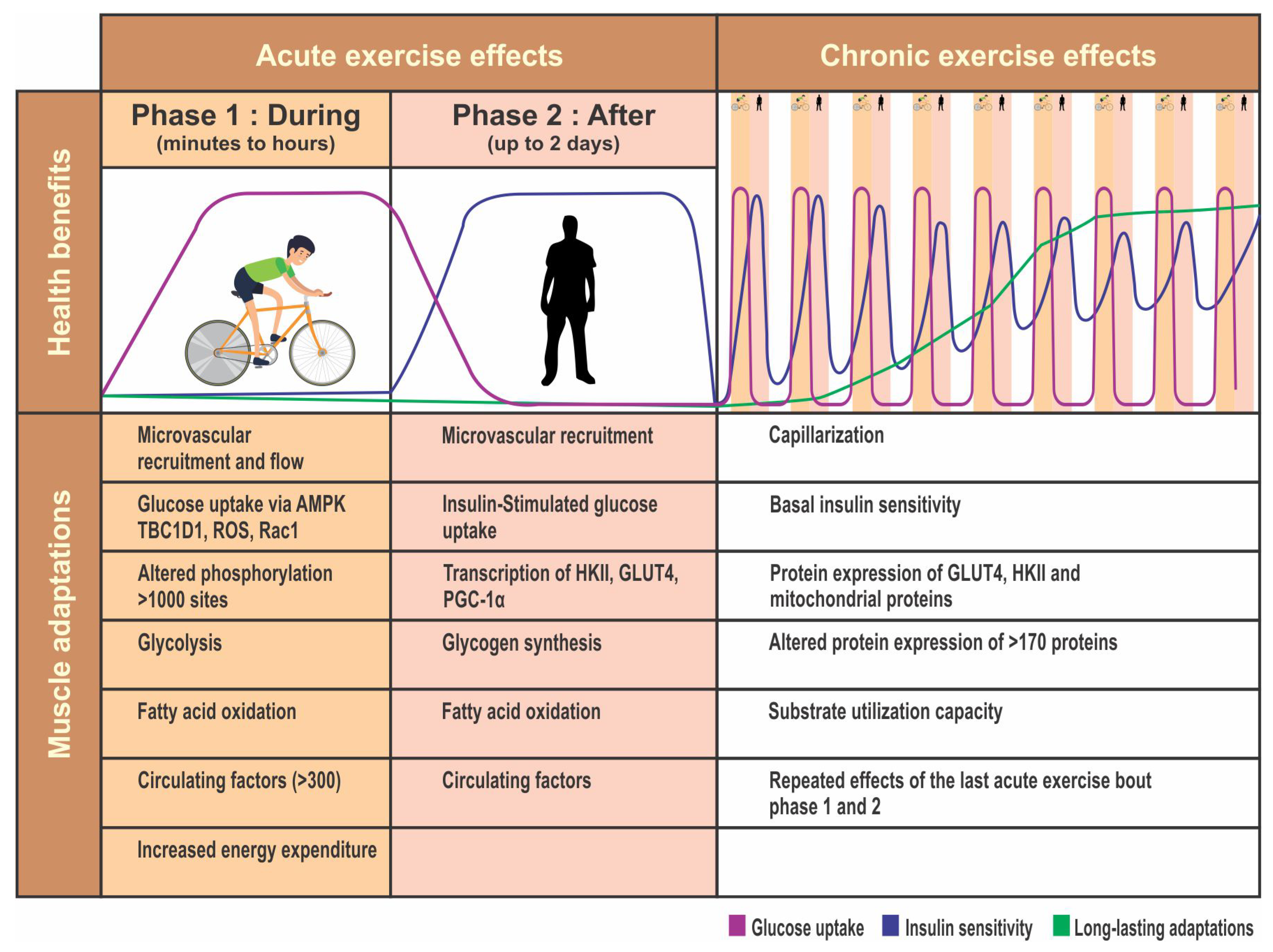
5.1.1. Potential Endocrine/Metabolic Impact of Exercise
5.1.2. Potential Endocrine/Metabolic Impact of Diet
5.1.3. Medical Nutrition Therapy (MNT)
5.2. Endocrine Impact of Pharmacological Management of Obesity
5.2.1. Endocrine Impact of Weight-Reducing Drugs
5.2.2. Weight Impact of Endocrinotropic Drugs
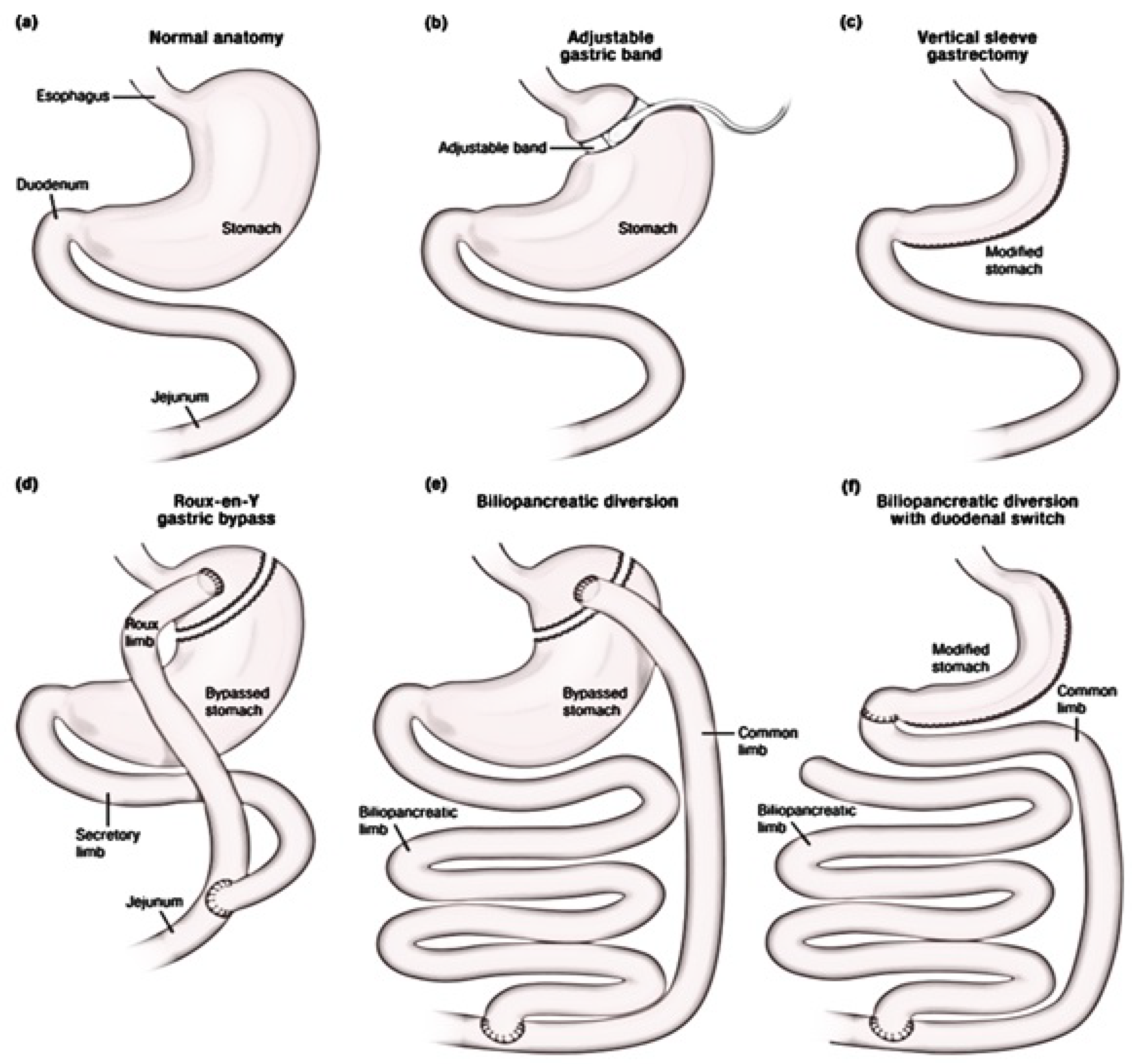
5.3. Endocrine Impact of Bariatric Surgery
5.3.1. Impact of Bariatric Surgery on Islet Function, Insulin Secretion, and Glucose Control
5.3.2. Hormonal Impact of Bariatric Surgery on Hypoglycemia
5.3.3. Impact of Bariatric Surgery on Gut Hormones: Hunger and Satiety
5.3.4. Impact of Bariatric Surgery on Energy Expenditure
5.3.5. Impact of Bariatric Surgery on Somatotropic Axis: Growth Hormone (GH)/Insulin-Growth Factor-1 (IGF-1)
5.3.6. Impact of Bariatric Surgery on Hypothalamic/Pituitary/Gonadal Axes in Women
5.3.7. Impact of Bariatric Surgery on Hypothalamic/Pituitary/Gonadal Axes in Men
5.3.8. Impact of Bariatric Surgery on Bone Metabolism
5.3.9. Impact of Bariatric Surgery on Thyroid Function
5.3.10. Impact of Bariatric Surgery on Adrenal Function
6. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Obesity and Overweight. Published 2016. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 28 February 2020).
- Kalra, S.; Unnikrishnan, A.G. Obesity in India: The weight of the nation. J. Med. Nutr. Nutraceuticals 2012, 1, 37. [Google Scholar] [CrossRef]
- WHO. WHO|Obesity. WHO. Available online: https://www.who.int/topics/obesity/en/ (accessed on 20 January 2020).
- Ordovas, J.M.; Corella, D. Nutritional Genomics. Annu. Rev. Genom. Hum. Genet. 2004, 5, 71–118. [Google Scholar] [CrossRef]
- O’Rahilly, S.; Farooqi, I.S. Human Obesity: A Heritable Neurobehavioral Disorder That Is Highly Sensitive to Environmental Conditions. Diabetes 2008, 57, 2905–2910. [Google Scholar] [CrossRef]
- Pinto, R.M.; Steinmetz, L.S.; Barbosa, J.M.G.; Mendes, A.F.C.S.; Curado, M.P.; Da Cruz, A.D. The Role of Genetics in the Pathophysiology of Obesity: A Systematic Review. Obes. Res. Open J. 2019, 6, 11–17. [Google Scholar] [CrossRef]
- Kapoor, N.; Chapla, A.; Furler, J.; Paul, T.V.; Harrap, S.; Oldenburg, B.; Thomas, N. Genetics of obesity in consanguineous populations—A road map to provide novel insights in the molecular basis and management of obesity. EBioMedicine 2019, 40, 33–34. [Google Scholar] [CrossRef]
- Seetho, I.W.; Wilding, J. How to approach endocrine assessment in severe obesity? Clin. Endocrinol. 2013, 79, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, N.; Chapla, A.; Thomas, N. Genetics of Obesity and Lipodystrophy. In Sadikot’s International Textbook of Diabetes, 1st ed.; Kamlakar, T., Banshi, S., Eds.; JP Medical Ltd.: Gurugram, Haryana, 2019; Available online: https://www.jaypeedigital.com/book/9789352700325/chapter/ch25 (accessed on 1 April 2020).
- NIH. Wilson-Turner Syndrome|Genetic and Rare Diseases Information Center (GARD)—An NCATS Program. Available online: https://rarediseases.info.nih.gov/diseases/5579/wilson-turner-syndrome (accessed on 11 April 2020).
- NIH GHR. Alström Syndrome. In Genetics Home Reference. Available online: https://ghr.nlm.nih.gov/condition/alstrom-syndrome (accessed on 11 April 2020).
- Kokkoris, P.; Pi-Sunyer, F.X. Obesity and endocrine disease. Endocrinol. Metab. Clin. N. Am. 2003, 32, 895–914. [Google Scholar] [CrossRef]
- Pasquali, R.; Casanueva, F.; Haluzik, M.; Van Hulsteijn, L.; LeDoux, S.; Monteiro, M.P.; Salvador, J.; Santini, F.; Toplak, H.; Dekkers, O.M. European Society of Endocrinology Clinical Practice Guideline: Endocrine work-up in obesity. Eur. J. Endocrinol. 2020, 182, G1–G32. [Google Scholar] [CrossRef] [PubMed]
- Sarwer, D.B.; Spitzer, J.C.; Wadden, T.A.; Mitchell, J.E.; Lancaster, K.; Courcoulas, A.; Gourash, W.; Rosen, R.C.; Christian, N.J. Changes in Sexual Functioning and Sex Hormone Levels in Women Following Bariatric Surgery. JAMA Surg. 2014, 149, 26–33. [Google Scholar] [CrossRef]
- Joseph, A.J.; Kapoor, N.; Simon, E.G.; Chacko, A.; Thomas, E.; Eapen, A.; Abraham, D.; Jacob, P.; Paul, T.; Rajaratnam, S.; et al. Endoscopic ultrasonography--a sensitive tool in the preoperative localization of insulinoma. Endocr. Pract. 2013, 19, 602–608. [Google Scholar] [CrossRef]
- Kaufman, J.-M.; Vermeulen, A. The Decline of Androgen Levels in Elderly Men and Its Clinical and Therapeutic Implications. Endocr. Rev. 2005, 26, 833–876. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, P. Discussion About Sexual Health: Is It Age Inappropriate? In Health and Wellbeing in Late Life: Perspectives and Narratives from India; Chatterjee, P., Ed.; Springer: Berlin/Heidelberg, Germany, 2019; pp. 129–146. [Google Scholar]
- Bhasin, S.; Brito, J.P.; Cunningham, G.R.; Hayes, F.J.; Hodis, H.N.; Matsumoto, A.M.; Snyder, P.J.; Swerdloff, R.S.; Wu, F.C.; Yialamas, M.A. Testosterone Therapy in Men With Hypogonadism: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2018, 103, 1715–1744. [Google Scholar] [CrossRef]
- Thomas, N.; Kapoor, N.; Velavan, J.; Vasan, K.S. A Practical Guide to Diabetes Mellitus, 8th ed.; Jaypee Brothers Medical Pub.: New Delhi, India, 2018. [Google Scholar]
- Weir, C.B.; Jan, A. BMI Classification Percentile and Cut off Points. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Kapoor, N.; Furler, J.; Paul, T.V.; Thomas, N.; Oldenburg, B. The BMI–adiposity conundrum in South Asian populations: Need for further research. J. Biosoc. Sci. 2019, 51, 619–621. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, N.; Lotfaliany, M.; Sathish, T.; Thankappan, K.R.; Thomas, N.; Furler, J.; Oldenburg, B.; Tapp, R.J. Prevalence of normal weight obesity and its associated cardio-metabolic risk factors—Results from the baseline data of the Kerala Diabetes Prevention Program (KDPP). PLoS ONE 2020, 15, e0237974. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, N.; Lotfaliany, M.; Sathish, T.; Thankappan, K.R.; Tapp, R.J.; Thomas, N.; Furler, J.; Oldenburg, B. Effect of a Peer-led Lifestyle Intervention on Individuals with Normal Weight Obesity: Insights from the Kerala Diabetes Prevention Program. Clin. Ther. 2020. [Google Scholar] [CrossRef] [PubMed]
- Atri, A.; Jiwanmall, S.A.; Nandyal, M.B.; Kattula, D.; Paravathareddy, S.; Paul, T.V.; Thomas, N.; Kapoor, N. The Prevalence and Predictors of Non-alcoholic Fatty Liver Disease in Morbidly Obese Women—A Cross-sectional Study from Southern India. Eur. Endocrinol. 2020, 16, 152–155. [Google Scholar] [CrossRef]
- Kapoor, N.; Furler, J.; Paul, T.V.; Thomas, N.; Oldenburg, B. Ethnicity-specific cut-offs that predict co-morbidities: The way forward for optimal utility of obesity indicators. J. Biosoc. Sci. 2019, 51, 624–626. [Google Scholar] [CrossRef]
- Kapoor, N.; Furler, J.; Paul, T.V.; Thomas, N.; Oldenburg, B. Normal Weight Obesity: An Underrecognized Problem in Individuals of South Asian Descent. Clin. Ther. 2019, 41, 1638–1642. [Google Scholar] [CrossRef]
- Ramasamy, S.; Joseph, M.; Jiwanmall, S.A.; Kattula, D.; Nandyal, M.B.; Abraham, V.; Samarasam, I.; Paravathareddy, S.; Paul, T.V.; Rajaratnam, S.; et al. Obesity Indicators and Health-related Quality of Life—Insights from a Cohort of Morbidly Obese, Middle-aged South Indian Women. Eur. Endocrinol. 2020, 16, 148–151. [Google Scholar] [CrossRef]
- Chan, D.C.; Barrett, P.H.R.; Watts, G.F.; Burke, V. Waist circumference, waist-to-hip ratio and body mass index as predictors of adipose tissue compartments in men. QJM Int. J. Med. 2003, 96, 441–447. [Google Scholar] [CrossRef]
- Crane, J.D.; McGowan, B.M. Clinical Assessment of the Patient with Overweight or Obesity. In Obesity: Pathogenesis, Diagnosis, and Treatment; Sbraccia, P., Finer, N., Eds.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 1–27. [Google Scholar] [CrossRef]
- Kapoor, N.; Lotfaliany, M.; Sathish, T.; Thankappan, K.R.; Thomas, N.; Furler, J.; Oldenburg, B.; Tapp, R.J. Obesity indicators that best predict type 2 diabetes in an Indian population: Insights from the Kerala Diabetes Prevention Program. J. Nutr. Sci. 2020, 9, e15. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, N.; Jiwanmall, S.A.; Nandyal, M.B.; Kattula, D.; Paravathareddy, S.; Paul, T.V.; Furler, J.; Oldenburg, B.; Thomas, N. Metabolic Score for Visceral Fat (METS-VF) Estimation—A Novel Cost-Effective Obesity Indicator for Visceral Adipose Tissue Estimation. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 3261–3267. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, N.; Inian, S.; Thomas, N. Chapter 28. Obesity with Diabetes. In A Practical Guide to Diabetes Mellitus, 8th ed.; Jaypee Brothers Medical Publishers: New Delhi, India, 2018; Available online: https://www.amazon.co.uk/Practical-Guide-Diabetes-Mellitus/dp/9352701658 (accessed on 1 April 2020).
- Ball, D. Metabolic and endocrine response to exercise: Sympathoadrenal integration with skeletal muscle. J. Endocrinol. 2014, 224, R79–R95. [Google Scholar] [CrossRef] [PubMed]
- Hackney, A.C.; Lane, A.R. Exercise and the Regulation of Endocrine Hormones. Prog. Mol. Biol. Transl. Sci. 2015, 135, 293–311. [Google Scholar] [CrossRef] [PubMed]
- Chycki, J.; Zajac, A.; Michalczyk, M.M.; Maszczyk, A.; Langfort, J. Hormonal and metabolic substrate status in response to exercise in men of different phenotype. Endocr. Connect. 2019, 8, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Edinburgh, R.M.; Bradley, H.E.; Abdullah, N.-F.; Robinson, S.L.; Chrzanowski-Smith, O.J.; Walhin, J.-P.; Joanisse, S.; Manolopoulos, K.N.; Philp, A.; Hengist, A.; et al. Lipid Metabolism Links Nutrient-Exercise Timing to Insulin Sensitivity in Men Classified as Overweight or Obese. J. Clin. Endocrinol. Metab. 2019, 105, 660–676. [Google Scholar] [CrossRef]
- Oh, K.-J.; Lee, D.S.; Kim, W.K.; Han, B.-S.; Lee, S.C.; Bae, K.-H. Metabolic Adaptation in Obesity and Type II Diabetes: Myokines, Adipokines and Hepatokines. Int. J. Mol. Sci. 2016, 18, 8. [Google Scholar] [CrossRef]
- Ranabir, S.; Reetu, K. Stress and hormones. Indian J. Endocrinol. Metab. 2011, 15, 18–22. [Google Scholar] [CrossRef]
- Siervo, M.; Wells, J.C.K.; Cizza, G. The Contribution of Psychosocial Stress to the Obesity Epidemic: An Evolutionary Approach. Horm. Metab. Res. Horm. Stoffwechs. Horm. Metab. 2009, 41, 261–270. [Google Scholar] [CrossRef]
- Redman, L.M.; Ravussin, E. Endocrine alterations in response to calorie restriction in humans. Mol. Cell. Endocrinol. 2009, 299, 129–136. [Google Scholar] [CrossRef]
- Unnikrishnan, A.G.; Bhatt, A.A.; Choudhari, P.K.; Mahajan, R.R.; Sayyad, M.G.; Pratyush, D.D.; Hasan, I.; Javherani, R.S.; Bothale, M.M.; Purandare, V.B. Effect of a low-calorie diet on restoration of normoglycemia in obese subjects with type 2 diabetes. Indian J. Endocrinol. Metab. 2017, 21, 776–780. [Google Scholar] [CrossRef] [PubMed]
- Gupta, L.; Khandelwal, D.; Lal, P.R.; Kalra, S.; Dutta, D. Palaeolithic Diet in Diabesity and Endocrinopathies—A Vegan’s Perspective. Eur. Endocrinol. 2019, 15, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Miller, V.J.; Hyde, P.N.; Dickerson, R.; LaFountain, R.A.; Maresh, C.M.; Kraemer, W.J.; Volek, J.S. The Ketogenic Diet Alters Endocrine Regulation of Energy Metabolism in Ultra-Endurance Athletes. FASEB J. 2017, 31 (Suppl. 1), 1036.3. [Google Scholar] [CrossRef]
- Dwyer, J.T.; Melanson, K.J.; Sriprachy-anunt, U.; Cross, P.; Wilson, M. Dietary Treatment of Obesity. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Wouter, W.H., Dungan, K., Grossman, A., Hershman, J.M., Hofland, H.J., Kaltsas, G., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. Available online: http://www.ncbi.nlm.nih.gov/books/NBK278991/ (accessed on 12 April 2020).
- Lean, M.E.; Leslie, W.S.; Barnes, A.; Brosnahan, N.; Thom, G.; McCombie, L.; Peters, C.; Zhyzhneuskaya, S.; Al-Mrabeh, A.; Hollingsworth, K.G.; et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): An open-label, cluster-randomised trial. Lancet 2018, 391, 541–551. [Google Scholar] [CrossRef]
- Taylor, R.; Leslie, W.S.; Barnes, A.C.; Brosnahan, N.; Thom, G.; McCombie, L.; Sattar, N.; Welsh, P.; Peters, C.; Zhyzhneuskaya, S.; et al. Clinical and metabolic features of the randomised controlled Diabetes Remission Clinical Trial (DiRECT) cohort. Diabetologia 2017, 61, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Mottalib, A.; Salsberg, V.; Yusof, B.-N.M.; Mohamed, W.; Carolan, P.; Pober, D.M.; Mitri, J.; Hamdy, O. Effects of nutrition therapy on HbA1c and cardiovascular disease risk factors in overweight and obese patients with type 2 diabetes. Nutr. J. 2018, 17, 42. [Google Scholar] [CrossRef]
- Fontana, L.; Klein, S.; Holloszy, J.O.; Premachandra, B.N. Effect of Long-Term Calorie Restriction with Adequate Protein and Micronutrients on Thyroid Hormones. J. Clin. Endocrinol. Metab. 2006, 91, 3232–3235. [Google Scholar] [CrossRef]
- Lecoultre, V.; Ravussin, E.; Redman, L.M. The fall in leptin concentration is a major determinant of the metabolic adaptation induced by caloric restriction independently of the changes in leptin circadian rhythms. J. Clin. Endocrinol. Metab. 2011, 96, E1512–E1516. [Google Scholar] [CrossRef]
- Agnihothri, R.V.; Courville, A.B.; Linderman, J.D.; Smith, S.; Brychta, R.; Remaley, A.; Chen, K.Y.; Simchowitz, L.; Celi, F.S. Moderate Weight Loss Is Sufficient to Affect Thyroid Hormone Homeostasis and Inhibit Its Peripheral Conversion. Thyroid 2014, 24, 19–26. [Google Scholar] [CrossRef]
- Srivastava, G.; Apovian, C.M. Current pharmacotherapy for obesity. Nat. Rev. Endocrinol. 2017, 14, 12–24. [Google Scholar] [CrossRef]
- Dar, S.; Tahrani, A.A.; Piya, M.K. The role of GLP-1 receptor agonists as weight loss agents in patients with and without type 2 diabetes. Pr. Diabetes 2015, 32, 297–300. [Google Scholar] [CrossRef]
- Kalra, S.; Priya, G.; Gupta, Y. Glucocrinology. JPMA J. Pak. Med. Assoc. 2018, 68, 963–965. [Google Scholar] [PubMed]
- Kalra, S.; Priya, G. Lipocrinology—The relationship between lipids and endocrine function. Drugs Context 2018, 7, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Verhaegen, A.A.; Van Gaal, L.F. Drugs That Affect Body Weight, Body Fat Distribution, and Metabolism. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Wouter, W.H., Dungan, K., Grossman, A., Hershman, J.M., Hofland, H.J., Kaltsas, G., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. Available online: http://www.ncbi.nlm.nih.gov/books/NBK537590/ (accessed on 7 March 2020).
- Domecq, J.P.; Prutsky, G.; Leppin, A.; Sonbol, M.B.; Altayar, O.; Undavalli, C.; Wang, Z.; Elraiyah, T.; Brito, J.P.; Mauck, K.F.; et al. Drugs Commonly Associated with Weight Change: A Systematic Review and Meta-analysis. J. Clin. Endocrinol. Metab. 2015, 100, 363–370. [Google Scholar] [CrossRef]
- Apovian, C.M.; Aronne, L.J.; Bessesen, D.H.; McDonnell, M.E.; Murad, M.H.; Pagotto, U.; Ryan, D.H.; Still, C.D. Pharmacological Management of Obesity: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2015, 100, 342–362. [Google Scholar] [CrossRef]
- Holst, J.J.; Madsbad, S.; Bojsen-Møller, K.N.; Svane, M.S.; Jørgensen, N.B.; Dirksen, C.; Martinussen, C. Mechanisms in bariatric surgery: Gut hormones, diabetes resolution, and weight loss. Surg. Obes. Relat. Dis. 2018, 14, 708–714. [Google Scholar] [CrossRef]
- Sjöström, L.; Peltonen, M.; Jacobson, P.D.; Ahlin, S.; Anderssonassarsson, J.C.; Anveden, Å.; Bouchard, C.; Carlsson, B.; Karason, K.; Lönroth, H.; et al. Association of Bariatric Surgery with Long-term Remission of Type 2 Diabetes and with Microvascular and Macrovascular Complications. JAMA 2014, 311, 2297–2304. [Google Scholar] [CrossRef]
- Leonetti, F.; Capoccia, D.; Coccia, F.; Casella, G.; Baglio, G.; Paradiso, F.; Abbatini, F.; Iossa, A.; Soricelli, E.; Basso, N. Obesity, Type 2 Diabetes Mellitus, and Other Comorbidities: A prospective cohort study of laparoscopic sleeve gastrectomy vs medical treatment. Arch. Surg. 2012, 147, 694–700. [Google Scholar] [CrossRef]
- Steffen, K.J.; King, W.C.; White, G.E.; Subak, L.L.; Mitchell, J.E.; Courcoulas, A.P.; Flum, D.R.; Strain, G.; Sarwer, D.B.; Kolotkin, R.L.; et al. Sexual functioning of men and women with severe obesity before bariatric surgery. Surg. Obes. Relat. Dis. Off. J. Am. Soc. Bariatr. Surg. 2017, 13, 334–343. [Google Scholar] [CrossRef]
- Moore, R.H.; Sarwer, D.B.; Lavenberg, J.A.; Lane, I.B.; Evans, J.L.; Volger, S.; Wadden, T.A. Relationship between sexual function and quality of life in obese persons seeking weight reduction. Obes. Silver Spring Md. 2013, 21, 1966–1974. [Google Scholar] [CrossRef]
- Escobar-Morreale, H.F.; Santacruz, E.; Luque-Ramírez, M.; Carretero, J.I.B. Prevalence of ‘obesity-associated gonadal dysfunction’ in severely obese men and women and its resolution after bariatric surgery: A systematic review and meta-analysis. Hum. Reprod. Updat. 2017, 23, 390–408. [Google Scholar] [CrossRef] [PubMed]
- Adil, T.; Jain, V.; Rashid, F.; Al-Taan, O.; Whitelaw, D.; Jambulingam, P. Meta-analysis of the effect of bariatric surgery on physical function. Br. J. Surg. 2018, 105, 1107–1118. [Google Scholar] [CrossRef] [PubMed]
- Risstad, H.; Søvik, T.T.; Engström, M.; Aasheim, E.T.; Fagerland, M.W.; Olsén, M.F.; Kristinsson, J.A.; Le Roux, C.W.; Bøhmer, T.; Birkeland, K.I.; et al. Five-Year Outcomes After Laparoscopic Gastric Bypass and Laparoscopic Duodenal Switch in Patients with Body Mass Index of 50 to 60: A randomized clinical trial. JAMA Surg. 2015, 150, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Peterli, R.; Wölnerhanssen, B.K.; Vetter, D.; Nett, P.; Gass, M.; Borbély, Y.; Peters, T.; Schiesser, M.; Schultes, B.; Beglinger, C.; et al. Laparoscopic Sleeve Gastrectomy Versus Roux-Y-Gastric Bypass for Morbid Obesity—3-Year Outcomes of the Prospective Randomized Swiss Multicenter Bypass or Sleeve Study (SM-BOSS). Ann. Surg. 2017, 265, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Salminen, P.; Helmiö, M.; Ovaska, J.; Juuti, A.; Leivonen, M.; Peromaa-Haavisto, P.; Hurme, S.; Soinio, M.; Nuutila, P.; Victorzon, M. Effect of Laparoscopic Sleeve Gastrectomy vs Laparoscopic Roux-en-Y Gastric Bypass on Weight Loss at 5 Years Among Patients with Morbid Obesity: The SLEEVEPASS Randomized Clinical Trial. JAMA 2018, 319, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Hofsø, D.; Nordstrand, N.; Johnson, L.K.; Karlsen, T.I.; Hager, H.; Jenssen, T.; Bollerslev, J.; Godang, K.; Sandbu, R.; Røislien, J.; et al. Obesity-related cardiovascular risk factors after weight loss: A clinical trial comparing gastric bypass surgery and intensive lifestyle intervention. Eur. J. Endocrinol. 2010, 163, 735–745. [Google Scholar] [CrossRef]
- Adams, T.D.; Pendleton, R.C.; Strong, M.B.; Kolotkin, R.L.; Walker, J.M.; Litwin, E.S.; Berjaoui, W.K.; LaMonte, M.J.; Cloward, T.V.; Avelar, E.; et al. Health Outcomes of Gastric Bypass Patients Compared to Nonsurgical, Nonintervened Severely Obese. Obes. Silver Spring Md. 2010, 18, 121–130. [Google Scholar] [CrossRef]
- Heo, Y.-S.; Park, J.-M.; Kim, Y.-J.; Kim, S.-M.; Park, D.J.; Lee, S.-K.; Han, S.-M.; Shim, K.-W.; Lee, Y.-J.; Lee, J.-Y.; et al. Bariatric surgery versus conventional therapy in obese Korea patients: A multicenter retrospective cohort study. J. Korean Surg. Soc. 2012, 83, 335–342. [Google Scholar] [CrossRef]
- Noel, O.F.; Still, C.D.; Argyropoulos, G.; Edwards, M.; Gerhard, G.S. Bile Acids, FXR, and Metabolic Effects of Bariatric Surgery. J. Obes. 2016, 2016, 4390254. [Google Scholar] [CrossRef]
- Tinahones, F.J.; Queipo-Ortuño, M.I.; Clemente-Postigo, M.; Fernnadez-Garcia, D.; Mingrone, G.; Cardona, F. Postprandial hypertriglyceridemia predicts improvement in insulin resistance in obese patients after bariatric surgery. Surg. Obes. Relat. Dis. Off. J. Am. Soc. Bariatr. Surg. 2013, 9, 213–218. [Google Scholar] [CrossRef]
- Douros, J.D.; Tong, J.; D’Alessio, D.A. The Effects of Bariatric Surgery on Islet Function, Insulin Secretion, and Glucose Control. Endocr. Rev. 2019, 40, 1394–1423. [Google Scholar] [CrossRef] [PubMed]
- Cornejo-Pareja, I.; Clemente-Postigo, M.; Tinahones, F.J. Metabolic and Endocrine Consequences of Bariatric Surgery. Front. Endocrinol. 2019, 10, 626. [Google Scholar] [CrossRef] [PubMed]
- Abrahamsson, N.; Börjesson, J.L.; Sundbom, M.; Wiklund, U.; Karlsson, F.A.; Eriksson, J.W. Gastric Bypass Reduces Symptoms and Hormonal Responses in Hypoglycemia. Diabetes 2016, 65, 2667–2675. [Google Scholar] [CrossRef] [PubMed]
- Folli, F.; Sabowitz, B.; Schwesinger, W.H.; Fanti, P.; Guardadomendoza, R.; Muscogiuri, G. Bariatric surgery and bone disease: From clinical perspective to molecular insights. Int. J. Obes. 2012, 36, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Dimitriadis, G.K.; Randeva, M.S.; Miras, A.D. Potential Hormone Mechanisms of Bariatric Surgery. Curr. Obes. Rep. 2017, 6, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, S.D. The Role of Hormonal Factors in Weight Loss and Recidivism after Bariatric Surgery. Gastroenterol. Res. Pr. 2013, 2013, 528450. [Google Scholar] [CrossRef] [PubMed]
- Heshka, S.; Lemos, T.; Astbury, N.M.; Widen, E.; Davidson, L.; Goodpaster, B.H.; Delany, J.P.; Strain, G.W.; Pomp, A.; Courcoulas, A.P.; et al. Resting Energy Expenditure and Organ-Tissue Body Composition 5 Years After Bariatric Surgery. Obes. Surg. 2019, 30, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Brehm, B.J.; Summer, S.; Jenkins, T.; D’Alessio, D.; Inge, T. Thermic effect of food and resting energy expenditure after sleeve gastrectomy for weight loss in adolescent females. Surg. Obes. Relat. Dis. Off. J. Am. Soc. Bariatr. Surg. 2020, 16, 599–606. [Google Scholar] [CrossRef]
- Camastra, S.; Manco, M.; Frascerra, S.; Iaconelli, A.; Mingrone, G.; Ferrannini, E.; Camastra, M.M.S. Daylong pituitary hormones in morbid obesity: Effects of bariatric surgery. Int. J. Obes. 2008, 33, 166–172. [Google Scholar] [CrossRef][Green Version]
- Billeter, A.T.; Fischer, L.; Wekerle, A.-L.; Senft, J.; Müller-Stich, B. Malabsorption as a Therapeutic Approach in Bariatric Surgery. Viszeralmedizin 2014, 30, 198–204. [Google Scholar] [CrossRef]
- Mancini, M.C.; Costa, A.P.; De Melo, M.E.; Cercato, C.; Giannella-Neto, D.; Garrido, A.B., Jr.; Rosberg, S.; Albertsson-Wikland, K.; Villares, S.M.; Halpern, A. Effect of Gastric Bypass on Spontaneous Growth Hormone and Ghrelin Release Profiles*. Obes. Silver Spring Md. 2006, 14, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Mittempergher, F.; Pata, G.; Crea, N.; Di Betta, E.; Vilardi, A.; Chiesa, D.; Nascimbeni, R. Preoperative Prediction of Growth Hormone (GH)/Insulin-Like Growth Factor-1 (IGF-1) Axis Modification and Postoperative Changes in Candidates for Bariatric Surgery. Obes. Surg. 2012, 23, 594–601. [Google Scholar] [CrossRef] [PubMed]
- Engström, B.E.; Burman, P.; Holdstock, C.; Öhrvall, M.; Sundbom, M.; Karlsson, F.A. Effects of gastric bypass on the GH/IGF-I axis in severe obesity—and a comparison with GH deficiency. Eur. J. Endocrinol. 2006, 154, 53–59. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Savastano, S.; Angrisani, L.; Di Somma, C.; Rota, F.; Savanelli, M.C.; Cascella, T.; Orio, F.; Lombardi, G.; Colao, A. Relationship Between Growth Hormone/Insulin-Like Growth Factor-1 Axis Integrity and Voluntary Weight Loss After Gastric Banding Surgery for Severe Obesity. Obes. Surg. 2009, 20, 211–220. [Google Scholar] [CrossRef]
- Di Somma, C.; Angrisani, L.; Rota, F.; Savanelli, M.C.; Cascella, T.; Belfiore, A.; Orio, F.; Lombardi, G.; Colao, A.; Savastano, S. GH and IGF-I deficiency are associated with reduced loss of fat mass after laparoscopic-adjustable silicone gastric banding. Clin. Endocrinol. 2008, 69, 393–399. [Google Scholar] [CrossRef]
- Galli, G.; Pinchera, A.; Piaggi, P.; Fierabracci, P.; Giannetti, M.; Querci, G.; Scartabelli, G.; Manetti, L.; Ceccarini, G.; Martinelli, S.; et al. Serum Insulin-Like Growth Factor-1 Concentrations Are Reduced in Severely Obese Women and Raise After Weight Loss Induced by Laparoscopic Adjustable Gastric Banding. Obes. Surg. 2012, 22, 1276–1280. [Google Scholar] [CrossRef]
- De Marinis, L.; Bianchi, A.; Mancini, A.; Gentilella, R.; Perrelli, M.; Giampietro, A.; Porcelli, T.; Tilaro, L.; Fusco, A.; Valle, D.; et al. Growth Hormone Secretion and Leptin in Morbid Obesity before and after Biliopancreatic Diversion: Relationships with Insulin and Body Composition. J. Clin. Endocrinol. Metab. 2004, 89, 174–180. [Google Scholar] [CrossRef]
- Bonomi, M.; Vezzoli, V.; Krausz, C.; Guizzardi, F.; Vezzani, S.; Simoni, M.; Bassi, I.; Duminuco, P.; Di Iorgi, N.; Giavoli, C.; et al. Characteristics of a nationwide cohort of patients presenting with isolated hypogonadotropic hypogonadism (IHH). Eur. J. Endocrinol. 2018, 178, 23–32. [Google Scholar] [CrossRef]
- Bond, D.S.; Wing, R.R.; Vithiananthan, S.; Sax, H.C.; Roye, G.D.; Ryder, B.A.; Pohl, D.; Giovanni, J. Significant resolution of female sexual dysfunction after bariatric surgery. Surg. Obes. Relat. Dis. Off. J. Am. Soc. Bariatr. Surg. 2011, 7, 1–7. [Google Scholar] [CrossRef]
- Paul, T.V.; Satyaraddi, A.; Cherian, K.E.; Kapoor, N.; Kunjummen, A.T.; Kamath, M.S.; Thomas, N. Body composition, metabolic characteristics, and insulin resistance in obese and nonobese women with polycystic ovary syndrome. J. Hum. Reprod. Sci. 2019, 12, 78–84. [Google Scholar] [CrossRef]
- Escobar-Morreale, H.F.; Botella-Carretero, J.I.; Álvarez-Blasco, F.; Sancho, J.; Millán, J.L.S. The Polycystic Ovary Syndrome Associated with Morbid Obesity May Resolve after Weight Loss Induced by Bariatric Surgery. J. Clin. Endocrinol. Metab. 2005, 90, 6364–6369. [Google Scholar] [CrossRef] [PubMed]
- Pasquali, R.; Diamanti-Kandarakis, E.; Gambineri, A. MANAGEMENT OF ENDOCRINE DISEASE: Secondary polycystic ovary syndrome: Theoretical and practical aspects. Eur. J. Endocrinol. 2016, 175, R157–R169. [Google Scholar] [CrossRef] [PubMed]
- Skubleny, D.; Switzer, N.; Gill, R.S.; Dykstra, M.; Shi, X.; Sagle, M.A.; De Gara, C.; Birch, D.W.; Karmali, S. The Impact of Bariatric Surgery on Polycystic Ovary Syndrome: A Systematic Review and Meta-analysis. Obes. Surg. 2015, 26, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Wing, R.R.; Bond, D.S.; Gendrano, I.N.; Wadden, T.; Bahnson, J.; Lewis, C.E.; Brancati, F.; Schneider, S.; Kitabchi, A.E.; Van Dorsten, B.; et al. Effect of Intensive Lifestyle Intervention on Sexual Dysfunction in Women With Type 2 Diabetes: Results from an ancillary Look AHEAD study. Diabetes Care 2013, 36, 2937–2944. [Google Scholar] [CrossRef]
- Reis, L.O.; Dias, F.G.F. Male Fertility, Obesity, and Bariatric Surgery. Reprod. Sci. 2012, 19, 778–785. [Google Scholar] [CrossRef]
- Kaukua, J.; Pekkarinen, T.; Sane, T.; Mustajoki, P. Sex Hormones and Sexual Function in Obese Men Losing Weight. Obes. Res. 2003, 11, 689–694. [Google Scholar] [CrossRef]
- Eid, G.M.; Cottam, D.R.; Velcu, L.M.; Mattar, S.G.; Korytkowski, M.T.; Gosman, G.; Hindi, P.; Schauer, P.R. Effective treatment of polycystic ovarian syndrome with Roux-en-Y gastric bypass. Surg. Obes. Relat. Dis. Off. J. Am. Soc. Bariatr. Surg. 2005, 1, 77–80. [Google Scholar] [CrossRef]
- Jamal, M.; Gunay, Y.; Capper, A.; Eid, A.; Heitshusen, D.; Samuel, I. Roux-en-Y gastric bypass ameliorates polycystic ovary syndrome and dramatically improves conception rates: A 9-year analysis. Surg. Obes. Relat. Dis. Off. J. Am. Soc. Bariatr. Surg. 2012, 8, 440–444. [Google Scholar] [CrossRef]
- Balen, A.H.; Morley, L.C.; Misso, M.; Franks, S.; Legro, R.S.; Wijeyaratne, C.N.; Stener-Victorin, E.; Fauser, B.C.; Norman, R.J.; Teede, H. The management of anovulatory infertility in women with polycystic ovary syndrome: An analysis of the evidence to support the development of global WHO guidance. Hum. Reprod. Updat. 2016, 22, 687–708. [Google Scholar] [CrossRef]
- Hofstra, J.; Loves, S.; Van Wageningen, B.; Ruinemans-Koerts, J.; Jansen, I.; De Boer, H. High prevalence of hypogonadotropic hypogonadism in men referred for obesity treatment. Neth. J. Med. 2008, 66, 103–109. [Google Scholar]
- Escobar-Morreale, H.F.; Álvarez-Blasco, F.; Botella-Carretero, J.I.; Luque-Ramírez, M. The striking similarities in the metabolic associations of female androgen excess and male androgen deficiency. Hum. Reprod. 2014, 29, 2083–2091. [Google Scholar] [CrossRef] [PubMed]
- Michalakis, K.; Mintziori, G.; Kaprara, A.; Tarlatzis, B.C.; Goulis, D.G. The complex interaction between obesity, metabolic syndrome and reproductive axis: A narrative review. Metabolism 2013, 62, 457–478. [Google Scholar] [CrossRef] [PubMed]
- Reis, L.O.; Favaro, W.J.; Barreiro, G.C.; De Oliveira, L.C.; Chaim, E.A.; Fregonesi, A.; Ferreira, U. Erectile dysfunction and hormonal imbalance in morbidly obese male is reversed after gastric bypass surgery: A prospective randomized controlled trial. Int. J. Androl. 2010, 33, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Mora, M.; Aranda, G.B.; De Hollanda, A.; Flores, L.; Puig-Domingo, M.; Vidal, J. Weight loss is a major contributor to improved sexual function after bariatric surgery. Surg. Endosc. 2013, 27, 3197–3204. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Ambrosi, J.; Rodríguez, A.; Catalán, V.; Frühbeck, G. The Bone-Adipose Axis in Obesity and Weight Loss. Obes. Surg. 2008, 18, 1134–1143. [Google Scholar] [CrossRef]
- Wong, I.P.; Baldock, P.A.; Herzog, H. Gastrointestinal peptides and bone health. Curr. Opin. Endocrinol. Diabetes Obes. 2010, 17, 44–50. [Google Scholar] [CrossRef]
- Nuche-Berenguer, B.; Moreno, P.; Esbrit, P.; Dapía, S.; Caeiro, J.R.; Cancelas, J.; Haro-Mora, J.J.; Villanueva-Peñacarrillo, M.L. Effect of GLP-1 Treatment on Bone Turnover in Normal, Type 2 Diabetic, and Insulin-Resistant States. Calcif. Tissue Int. 2009, 84, 453–461. [Google Scholar] [CrossRef]
- Wucher, H.; Ciangura, C.; Poitou, C.; Czernichow, S. Effects of Weight Loss on Bone Status after Bariatric Surgery: Association between Adipokines and Bone Markers. Obes. Surg. 2008, 18, 58–65. [Google Scholar] [CrossRef]
- Hage, M.P.; Rahme, M. Bone and mineral metabolism in patients undergoing Roux-en-Y gastric bypass. Osteoporos. Int. 2014, 25, 423–439. [Google Scholar] [CrossRef]
- Stein, E.M.; Silverberg, S.J. Bone loss after bariatric surgery: Causes, consequences, and management. Lancet Diabetes Endocrinol. 2014, 2, 165–174. [Google Scholar] [CrossRef]
- Corbeels, K.; Verlinden, L.; Lannoo, M.; Simoens, C.; Matthys, C.; Verstuyf, A.; Meulemans, A.; Carmeliet, G.; Van Der Schueren, B. Thin bones: Vitamin D and calcium handling after bariatric surgery. Bone Rep. 2018, 8, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Zendel, A.; Abu-Ghanem, Y.; Dux, J.; Mor, E.; Zippel, D.; Goitein, D. The Impact of Bariatric Surgery on Thyroid Function and Medication Use in Patients with Hypothyroidism. Obes. Surg. 2017, 27, 2000–2004. [Google Scholar] [CrossRef] [PubMed]
- Rudnicki, Y.; Slavin, M.; Keidar, A.; Kent, I.; Berkovich, L.; Tiomkin, V.; Inbar, R.; Avital, S. The effect of bariatric surgery on hypothyroidism: Sleeve gastrectomy versus gastric bypass. Surg. Obes. Relat. Dis. Off. J. Am. Soc. Bariatr. Surg. 2018, 14, 1297–1303. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Li, Q.; Zhang, M.; Liu, F.; Pan, J.; Tu, Y.; Lu, J.; Zhang, P.; Han, J.; Jia, W.; et al. Decreased Leptin Is Associated with Alterations in Thyroid-Stimulating Hormone Levels after Roux-en-Y Gastric Bypass Surgery in Obese Euthyroid Patients with Type 2 Diabetes. Obes. Facts 2019, 12, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Matute, C.M.; Mora, M.C.F.; Funes, D.R.; Menzo, E.L.; Szomstein, S.; Rosenthal, R.J. Impact of Bariatric Surgery on Thyroid Function and Medication use in Patients with Hypothyroidism. Surg. Obes. Relat. Dis. 2018, 14, S67. [Google Scholar] [CrossRef]
- Valdés, S.; Maldonado-Araque, C.; Lago-Sampedro, A.; Lillo-Muñoz, J.A.; Garcia-Fuentes, E.; Perez-Valero, V.; Gutiérrez-Repiso, C.; Garcia-Escobar, E.; Goday, A.; Urrutia, I.; et al. Reference values for TSH may be inadequate to define hypothyroidism in persons with morbid obesity: Di@bet.es study. Obes. Silver Spring Md. 2017, 25, 788–793. [Google Scholar] [CrossRef]
- Joseph, V.; Kushe, M.; Muniyappa, S.; Rajeswaran, C. Adrenal Insufficiency Post Gastric Bypass Surgery. BioScientifica Epub Ahead of Print 14 October 2016. Available online: https://www.endocrine-abstracts.org/ea/0044/ea0044p195 (accessed on 1 April 2020). [CrossRef]
- Shankaran, V.; Barclay, A.; Cinnadorai, R.; Thida, M.; Srinivasan, B. Adrenal Insufficiency Post Bariatric Surgery. Bio Scientifica Epub Ahead of Print 1 March 2013. Available online: https://www.endocrine-abstracts.org/ea/0031/ea0031p232 (accessed on 1 April 2020). [CrossRef]
- de Heide, L.J.M.; de Boer, H.H.R.; van Borren, M.; Marloes, E.; Edo, A.; Hans de, B. Pharmacokinetics of Glucocorticoid Replacement before and After Bariatric Surgery in Patients with Adrenal Insufficiency. J. Endocr. Soc. 2018, 2, 1338–1344. [Google Scholar] [CrossRef]
| Syndrome | Occurrence | Genetic Mutation | Features |
|---|---|---|---|
| Prader-Willi syndrome | 1:10,000/1:15,000 births | 15q11 SRNPN microdeletion | Newborn: Characteristic facies, small hands and feet, hypopigmentation hypotonia and failure to thrive Childhood: Short stature, hyperphagia, obesity, hypogonadism, delayed motor/cognitive development, sleep disturbances, and behavior abnormalities |
| Bardet–Biedel syndrome | 1:125,000/1:175,000 births | BBS (1–12) chaperonin protein MKKS (Chr20) | Obesity in first year of life, mental retardation, dysmorphic extremities, retinal dystrophy or pigmentary retinopathy, hypogonadism. kidney anomalies |
| Albright’s hereditary osteodystrophy | 1: 20,000/1:1,000,000 births | Autosomal dominant GNAS1 gene (20q13.2) | Short stature, obesity, round face, brachydactyly, subcutaneous calcification, dental and sensorineurat abnormalities Hormonal abnormalities: Generalizd hormonal resistance to PTH, TSH, GHRH and gonadotrophins; functional hypoparathyroidism seen on biochemical examination |
| Fragile X syndrome | 1:2500 births | Unstable expansions of a CGG repeats—FMR1 (fragile X mental retardation) gene- X chromosome | Macroorchidism, large ears, prominent jaw, hyperkinetic behavior, and mental retardation. |
| Wilson–Turner syndrome | Prevalence not known [10] | X-linked mutation | Truncal obesity, intellectual disability, gynecomastia, dysmorphic facial features, hypogonadism, and short stature |
| Cohen syndrome | Diagnosed in fewer than 1000 births worldwide | Chromosome 8q, and a novel gene, COH1 | Dysmorphic extremities, microcephaly, prominent central incisors, retinal dystrophy, and cyclic neutropenia |
| Alstorm syndrome | Little more than 900 cases reported worldwide [11] | 2p14 ALSM1 | Progressive loss of vision and hearing, dilated cardiomyopathy, obesity, type 2 diabetes, and short stature |
| Hormone | Obesity | Proposed Pathophysiology in Obesity | Weight Loss/Fasting |
|---|---|---|---|
| Pituitary | |||
| Prolactin | |||
| Basal prolactin | N | N | |
| Prolactin response to hypoglycemia | N/↓ | N/↓ | |
| Prolactin response to TRH | N/↓ | N | |
| GH/IGF-I | |||
| GH | N/↓ | ↓↑ GHRH, ↑ GH-BP, ↑ insulin, ↓ ghrelin, ↑ somatostatin | ↑ |
| GH production rate | ↓ | ↑ | |
| GH metabolic rate | ↑ | ||
| GH response to GHRH | ↓ | N/↓ | |
| GH response to hypoglycemia | ↓ | N/↓ | |
| IGF-I | N/↓ | I ↑ GH sensitivity t ↑ intrahepatic triglyceride content | |
| Free IGF-I | ↑ | ||
| IGFBP-3 | N/↑ | ||
| HPA axis in obesity | |||
| Basal cortisol (blood and salivary) | N | ↑↑ CRH, ↑ adipose 11-HSD, ↓ CBG Altered suppression tests due to hyperactivity of the HPA axis | |
| Urinary free cortisol | N/↑ | ||
| Basal ACTH | N/↑ | ↑ CRH | |
| Cortisol production rate | ↑ | ||
| Cortisol metabolic rate | ↑ | ||
| 17-OH-corticosteroids | ↑ | ||
| HPA axis response to stimulatory tests in central obesity | |||
| Cortisol after CRH | ↑ | N | |
| ACTH after CRH | ↑ | ↑ | |
| Cortisol after tetracosactide | ↑ | ||
| Cortisol after hypoglycemia | ↑ | ||
| Thyroid | |||
| FT4 | N/slight ↓ | ↑↑ disposal | N |
| T3 | ↑ | Probably related to the amount of food intake and not to obesity itself. | ↓ |
| rT3 | ↓ | ↑ | |
| TSH | N/↑ | ↑↑ leptin and insulin ↑ peripheral T4 disposal | N |
| TSH after TRH | N/↑/↓ | TRH dose decreases | |
| Parathyroid | |||
| PTH | N/↑ | Secondary due to vitamin D deficiency | |
| PTH ↑ calcium ↓ phosphate ↑ | Pseudohypoparathyroidism Type 1a (Albright hereditary osteodystrophy) | ||
| Pancreas/Gut | |||
| Insulin | ↑ | Insulin resistance Insulinoma | ↓ with decreased insulin resistance |
| Ghrelin | ↓ | Lack of ghrelin decrease after meals | ↑ |
| GLP-1 | ↓ | ↑ FFA, microbiota | ↑ |
| Gonads | |||
| Testosterone (male) | ↓ | ↓ SHBG, ↑ aromatase, ↓ GnRH | ↑ testosterone and gonadotrophins, ↓ estradiol |
| Testosterone (female) | ↑ | Insulin resistance (PCOS) ↓ SHBG | ↓ serum total testosterone, androsterone and sulfate dehydroepiandrosterone (DHEA) |
| LH/FSH(male) | ↓ | ↑ oestrogens/androgens | ↑ SHBG, LH, and FSH |
| LH/FSH(female) | ↑ LH | Insulin resistance | ↑ Serum estradiol, FSH and SHBG |
| Adrenal | |||
| Renin | ↑ | ↑ Sympathetic tone | |
| Aldosterone | ↑ | ↑ Adipokines, renin- angiotensin, leptin | |
| Adipocyte | |||
| Leptin | ↑ | ↑ adipose mass, Leptin resistance | ↓ |
| Gland | Prevalence in Obesity | When to Assess | First Diagnostic Procedure | Other Mandatory Work Up in Obesity | Not Recommended in Obesity |
|---|---|---|---|---|---|
| Thyroid (Same TSH and thyroid hormone values should be used in patients with obesity as are used in normal population without obesity) | Severe hypothyroidism is rare but subclinical hypothyroidism is common | Thyroid function should be tested for all patients with obesity | TSH | free T4 and antibodies (anti-TPO) should be measured only if TSH is elevated | Routine FT3 in patients with elevated TSH Routine ultrasound of the thyroid gland (irrespective of thyroid function) |
| Adrenal (same normal values should be applied patients with obesity as are used in normal population without obesity) | Cushing’s disease or Cushing’s syndrome is rare | Central obesity Hypertension Type 2 diabetes Testing for hypercortisolism should be considered in patients going for bariatric surgery | 1 mg ODST | 24 h urine cortisol or late-night salivary cortisol in patients with positive 1 mg overnight dexamethasone suppression test Imaging (find the cause/source) and ACTH in patients with confirmed hypercortisolism | Routine testing for hypercortisolism |
| Drug-induced adrenal dysfunction (e.g., lithium, anti-depressants, antipsychotics, glucocorticoids, etc.) is common | Biochemical testing should be performed in patients with clinical suspicion of hypercortisolism; those undergoing bariatric surgery, or having psychiatric disorders | 8:00 a.m. cortisol | Testing for hypercortisolism in patients using corticosteroids | ||
| Male Gonad (Use age-specific reference ranges for testosterone | Androgen deficiency is common | Severe obesity Symptoms and signs of hypogonadism In elderly male with impaired social and mental health, less energy | LH, FSH, fasting morning testosterone | Total and free testosterone (or calculated), SHBG in patients with clinical features of hypogonadism | Routine biochemical testing for hypogonadism unless key clinical symptoms/signs of hypogonadism and in elderly with impaired social/mental health, less energy |
| Female Gonad | Androgen excess is common | Central obesity Irregular menses Hirsutism Acanthosis nigricans chronic anovulation/infertility | LH, FSH, estradiol, testosterone | Total testosterone, SHBG, Δ 4 androstenedione, 17-hydroxyprogesterone and prolactin in patients with menstrual irregularities (assess in early follicular phase if menstrual cycle is predictable) | Routine testing for gonadal dysfunction |
| Clinical features of PCOS | Total testosterone, free T, Δ 4 androstenedion, SHBG and blood glucose | Ovarian morphology | |||
| Premature ovarian failure is uncommon | Secondary amenorrhea Vasomotor symptoms | LH, FSH, estradiol | Progesterone and prolactin in patients with anovulation | ||
| Physiological ovarian failure in menopause is common | Vaginal mucosal atrophy | LH, FSH, estradiol | |||
| Pituitary | GH deficiency is rare | Hypothalamic or pituitary disease, pituitary or hypothalamic surgery or radiation therapy | IGF1/GH using a dynamic test only in patients with suspected hypopituitarism | Routine testing for IGF1/GH | |
| Hypopituitarism is rare | Suspicion of hypothalamic obesity Surgery or radiotherapy in pituitary region | FT4 TSH LH FSH (testosterone or estradiol) GH IGF-1 PRL ACTH stimulation test GH stimulation test | |||
| Acquired hypothalamic obesity (hypothalamic lesions or, tumors) is rare | Severe hyperphagia Possible multiple endocrine abnormalities | Brain CT/MRI | |||
| Parathyroid | Pseudohypoparathyroidism Type 1a (Albright hereditary osteodystrophy) is rare | Short stature, short fourth metacarpal bones, obesity, sc calcifications, developmental delay | PTH ↑ calcium ↓ phosphate ↑ | Routine testing for hyperparathyroidism or Vitamin D deficiency | |
| Hypothalamus obesity | Hypothalamic obesity associated with Genetic Syndromes is very rare | Hypogonadism (hypogonadism or hypergonadotropic) or variable gonadal function. dysmorphic syndrome, mental and grow retardation | Leptin (leptin resistance); genetic testing | Routine testing of hormones such as leptin and ghrelin in patients with suspicion of syndromic obesity |
| Asian Body Mass Index Classification in Adults [19,20] | Body Mass Index Classification for Adults by World Health Organization [20] | |
|---|---|---|
| Classification | Body mass index (kg/m2) | Body mass index (kg/m2) |
| Underweight | <18.5 | <18.5 |
| Normal weight | 18.5–22.9 | 18.5–24.9 |
| Overweight | 23–24.9 | 25.0–29.9 |
| Obesity class I | 25–29.9 kg/m2 | 30.0–34.9 |
| Obesity class II | 30–34.9 kg/m2 | 35.0–39.9 |
| Obesity class III | ≥35 kg/m2 | ≥40 |
| Non-Nested Models | WC vs. WHR | WC vs. BMI | BMI vs. WHR |
|---|---|---|---|
| p Values | |||
| Retroperitoneal ATM | 0.670 | 0.045 | 0.115 |
| Intraperitoneal ATM | 0.285 | 0.042 | 0.544 |
| Subcutaneous anterior ATM | <0.001 | 0.280 | 0.036 |
| Subcutaneous posterior ATM | <0.001 | 0.759 | <0.001 |
| Drug | Mechanism of Action | Common Adverse Effects | Warnings | Contraindications |
|---|---|---|---|---|
| Phentermine | Sympathomimetic amine | Headache, dizziness, fatigue, dry mouth, constipation, upper respiratory tract-like symptoms and hypoglycaemia in patients with diabetes mellitus | Rare cases of primary pulmonary hypertension or serious regurgitant cardiac valvular disease | Cardiovascular disease, uncontrolled hypertension, agitated states, history of drug use, hyperthyroidism, glaucoma or MAGI use within 14 days |
| Orlistat | Pancreatic and gastric lipase inhibitor | Flatulence, bloating and diarrhoea | Malabsorption of fat-soluble vitamins A, D, E and K | Malabsorption syndrome or cholestasis |
| Phentermine/topiramate ER | Combination of sympathomimetic amine, anorectic and ER antiepileptic drug | Peripheral neuropathy (usually transient), dyspepsia, insomnia, constipation and dry mouth | Teratogenicity (risk of cleft palate and/or cleft lip), suicidal ideation, changes in memory or concentration, metabolic acidosis, hypokalaemia | Glaucoma, hyperthyroidism or MAOI use within 14 days |
| Lorcaserin (currently is banned/debarred due to increased cancer risk) | 5-HT2c receptor agonist | Headache, dizziness, fatigue, dry mouth, constipation, upper respiratory tract-like symptoms and hypoglycaemia in patients with diabetes mellitus | Serotonin syndrome or neuroleptic malignant syndrome; safety unknown with coadministration of serotonin or antidopaminergic agents, cognitive impairment, suicidal ideation and valvular heart disease (though not statistically significant in clinical trials) | Pregnancy |
| Naltrexone SR | Combination opioid antagonist and aminoketone antidepressant | Nausea, constipation, headache, vomiting, dizziness, dry mouth and diarrhoea | Suicidal ideation, decrease in seizure threshold, acute angle glaucoma, hepatotoxicity to naltrexone component and increase in HR and/or BP | Uncontrolled hypertension, seizure disorders, anorexia nervosa or bulimia, chronic opioid use, MAOI use within 14 days, abrupt discontinuation of alcohol or seizure medications |
| Liraglutide (3 mg) | GLP1 analogue | Hypoglycemia, nausea, diarrhea, vomiting, constipation, dyspepsia, decreased appetite, headache, fatigue, dizziness, abdominal pain, and increased lipase | Serious hypoglycemia, thyroid C-cell tumors, acute gallbladder disease, acute pancreatitis, hypersensitivity reactions. | Personal or family history of MTC or MEN syndrome type 2, hypersensitivity to liraglutide, Pregnancy |
| Medications | Weight Profile |
|---|---|
| Antidiabetes | GLP-1 analogs (eg, exenatide, albiglutide, dulaglutide, semaglutide and liraglutide) or SGLT-2 inhibitors (dapagliflozin, empagliflozin and canagliflozin) promote weight loss Basal insulin causes less weight gain than other insulin types |
| Antihypertensives | Angiotensin-converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), and calcium channel blockers should be preferred over β-adrenergic blockers which cause weight gain |
| Antidepressants | Paroxetine, amitryiptyline, mirtazapine, and nortriptyline are linked to weight gain Bupropion causes weight loss |
| Antipsychotics | Clozapine and olanzapine have a greater likelihood for weight gain, while ziprasidone appears to have the lowest risk for weight gain |
| Antiepileptics | Felbamate, topiramate and zonisamide may be preferred over other antiepileptics as they cause weight loss |
| Antihistamines | Choose one with less sedation |
| Antiretroviral | Most antiretrovirals cause weight gain, weight monitoring is important |
| Hormone | Organ/Cell | Mechanism of Action | Levels after Bariatric Procedure | ||
|---|---|---|---|---|---|
| GB | SG | RYGB | |||
| CCK | Duodenum/I cells | Stimulates digestion of fats and proteins | No data | Increase | Increase |
| FGF19 | Ileum | Regulation of glucose and lipid metabolism. Increases energy expenditure | Increase | Increase | Increase |
| FGF21 | Liver | Fatty acid oxidation, improves insulin sensitivity and increases energy expenditure | No data | Decrease | No change |
| Gastrin | Stomach/G cells | Increases HCl production | No change | No change/increase | Decrease |
| Promotes satiety | |||||
| Ghrelin | Stomach/G cells | Increases appetite | No change/increase | Conflicting data | Conflicting data |
| Enhances gastric emptying GI motility and GH secretion | |||||
| GLP-1 | Ileum/L cells | Causes the incretin effect | No change/increase | Increase | Increase |
| Increases insulin sensitivity and production. Delays gastric emptying. Enhances satiety. | |||||
| GLP-2 | Ileum/L cells | Causes gut hypertrophy. Alters GI motility | No change | Increase | Increase |
| Glucagon | Pancreas/A cells | Promotes glucogenolysis and gluconeogenesis | No change | Decrease | Conflicting data |
| Gustducin | Stomach/specialized lining cells | Enhances GLP-1 secretion | No data | No data | Increase |
| Insulin | Pancreas/B cells | Regulates metabolism of carbs, fat and protein. Promotes absorption glucose from the blood | No change/increase | Increase | Increase |
| Obestatin | Stomach/epithelial cells | Promotes satiety | No data | Decrease | Increase |
| Secretin | Duodenum/S cells | Reduces gastric and duodenal motility. Enhances insulin release | No data | No data | No change/decrease |
| VIP | Enteric and parasympathetic nerves | Promotes hormone secretion by the brain, gut and pancreas Increases the secretion of water and electrolytes. Reduces HCl secretion | No data | No data | No data |
| BMI Category | TSH-Cut-Off |
|---|---|
| <20 kg/m2 | 0.6 to 4.8 µUI/mL |
| 20–24.9 kg/m2 | 0.6 to 5.5 µUI/mL |
| 25–29.9 kg/m2 | 0.6 to 5.5 µUI/mL |
| 3–39.9 kg/m2 | 0.5 to 5.9 µUI/mL |
| >40 kg/m2 | 0.7 to 7.5 µUI/mL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalra, S.; Kapoor, N.; Bhattacharya, S.; Aydin, H.; Coetzee, A. Barocrinology: The Endocrinology of Obesity from Bench to Bedside. Med. Sci. 2020, 8, 51. https://doi.org/10.3390/medsci8040051
Kalra S, Kapoor N, Bhattacharya S, Aydin H, Coetzee A. Barocrinology: The Endocrinology of Obesity from Bench to Bedside. Medical Sciences. 2020; 8(4):51. https://doi.org/10.3390/medsci8040051
Chicago/Turabian StyleKalra, Sanjay, Nitin Kapoor, Saptarshi Bhattacharya, Hassan Aydin, and Ankia Coetzee. 2020. "Barocrinology: The Endocrinology of Obesity from Bench to Bedside" Medical Sciences 8, no. 4: 51. https://doi.org/10.3390/medsci8040051
APA StyleKalra, S., Kapoor, N., Bhattacharya, S., Aydin, H., & Coetzee, A. (2020). Barocrinology: The Endocrinology of Obesity from Bench to Bedside. Medical Sciences, 8(4), 51. https://doi.org/10.3390/medsci8040051






