Magnesium: The Forgotten Electrolyte—A Review on Hypomagnesemia
Abstract
1. Introduction
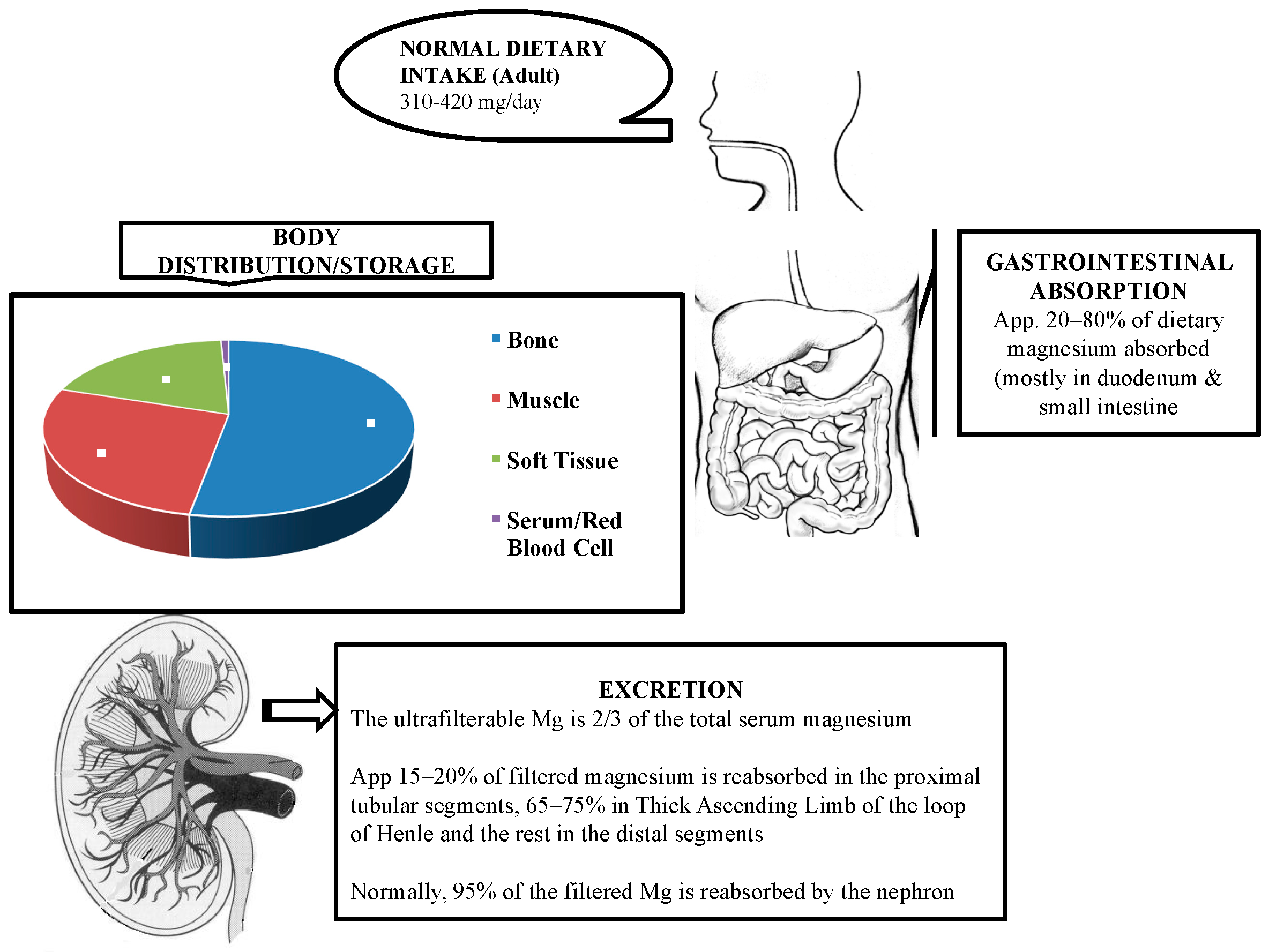
2. Physiology
2.1. Absorption
2.2. Storage and Distribution
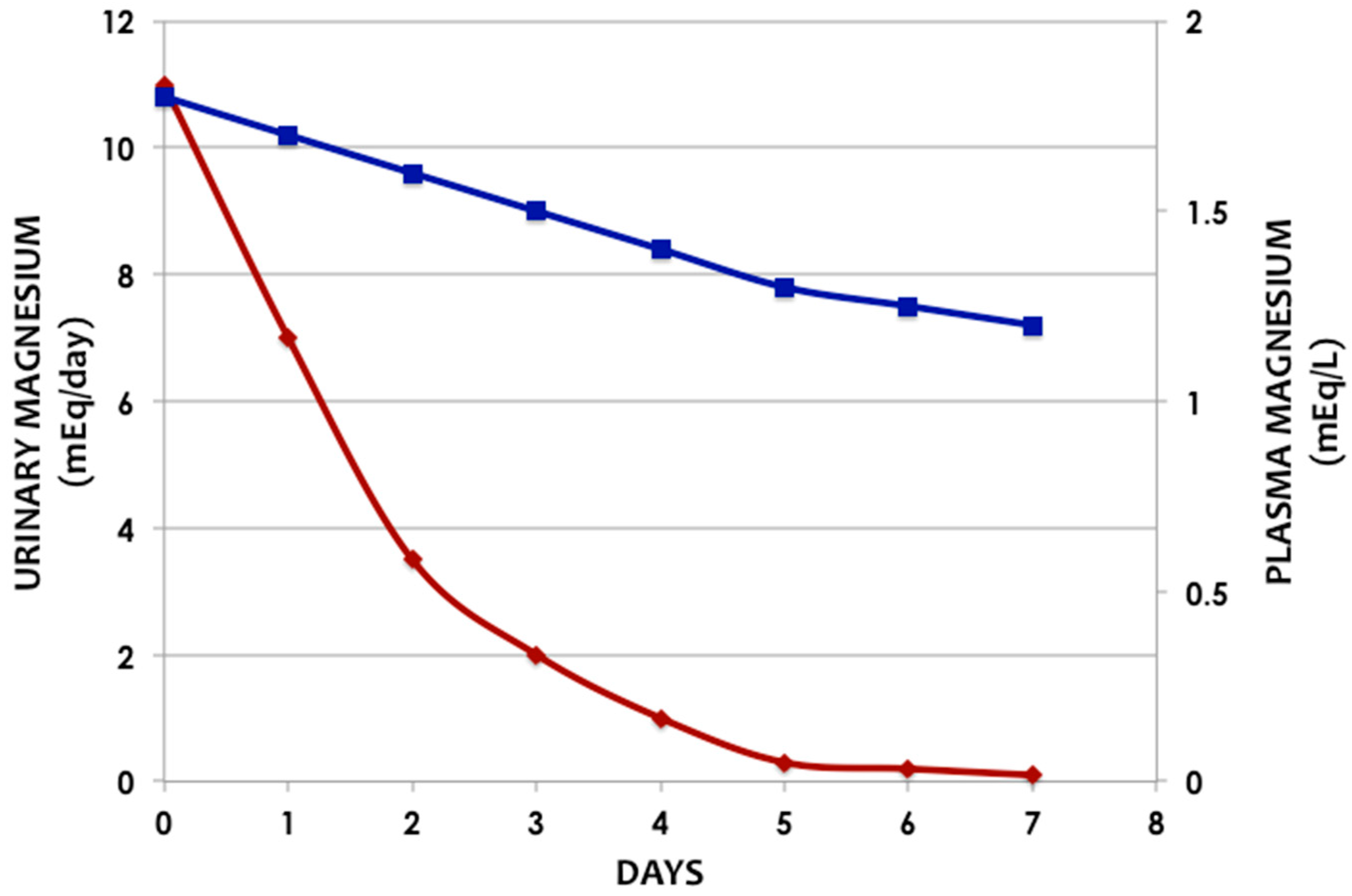
2.3. Excretion/Elimination
2.3.1. Thick Ascending Loop
2.3.2. Distal Convoluted Tubule
3. Regulation
4. Role at Cellular Level
5. Causes of Hypomagnesemia
6. Clinical Presentation/Complications of Hypomagnesemia
7. Neuromuscular/Neurological Manifestations
8. Cardiovascular Manifestations
8.1. Atherosclerotic Vascular Disease/Coronary Artery Disease
8.1.1. Arrhythmias
8.1.2. Hypertension
8.1.3. Congestive Heart Failure
8.2. Endocrine Manifestations
8.2.1. Altered Glucose Homeostasis/Diabetic Complications
8.2.2. Osteoporosis
8.3. Magnesium and Asthma
8.4. Nephrolithiasis
8.5. Magnesium and Pregnancy
8.6. Biochemical Manifestations
8.6.1. Hypokalemia
8.6.2. Hypocalcemia
9. Diagnostic Aspects
9.1. Types of Laboratory Tests
9.2. Serum Magnesium Concentration and Variables
9.3. Serum Magnesium Concentration Reference Interval
9.4. Urinary Magnesium Excretion
10. Treatment
Author Contributions
Funding
Conflicts of Interest
References
- Cotruvo, J.; Bartram, J. (Eds.) Calcium and Magnesium in Drinking-Water: Public Health Significance; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Fulop, T. Hypomagnesemia. Available online: https://emedicine.medscape.com/article/2038394-overview (accessed on 20 January 2019).
- Kayne, L.H.; Lee, D.B. Intestinal magnesium absorption. Miner Electrolyte Metab. 1993, 19, 210–217. [Google Scholar]
- de Baaij, J.H.; Hoenderop, J.G.; Bindels, R.J. Regulation of magnesium balance: Lessons learned from human genetic disease. Clin. Kidney J. 2012, 5, i15–i24. [Google Scholar] [CrossRef] [PubMed]
- Quamme, G.A. Recent developments in intestinal magnesium absorption. Curr. Opin. Gastroenterol. 2008, 24, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Pham, P.C.; Pham, P.A.; Pham, S.V.; Pham, P.T.; Pham, P.M.; Pham, P.T. Hypomagnesemia: A clinical perspective. Int. J. Nephrol. Renovasc. Dis. 2014, 7, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Schlingmann, K.P.; Weber, S.; Peters, M.; Niemann Nejsum, L.; Vitzthum, H.; Klingel, K.; Kratz, M.; Haddad, E.; Ristoff, E.; Dinour, D.; et al. Hypomagnesemia with secondary hypocalcemia is caused by mutations in trpm6, a new member of the trpm gene family. Nat. Genet. 2002, 31, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Fawcett, W.J.; Haxby, E.J.; Male, D.A. Magnesium: Physiology and pharmacology. Br. J. Anaesth. 1999, 83, 302–320. [Google Scholar] [CrossRef]
- Swaminathan, R. Magnesium metabolism and its disorders. Clin. Biochem. Rev. 2003, 24, 47–66. [Google Scholar] [PubMed]
- Hou, J.; Goodenough, D.A. Claudin-16 and claudin-19 function in the thick ascending limb. Curr. Opin. Nephrol. Hypertens 2010, 19, 483–488. [Google Scholar] [CrossRef]
- Al Shibli, A.; Narchi, H. Bartter and gitelman syndromes: Spectrum of clinical manifestations caused by different mutations. World J. Methodol. 2015, 5, 55–61. [Google Scholar] [CrossRef]
- Reinhart, R.A. Magnesium metabolism: A review with special reference to the relationship between intracellular content and serum levels. Arch. Intern. Med. 1988, 148, 2415–2420. [Google Scholar] [CrossRef]
- Shils, M.E. Experimental human magnesium depletion. Medicine (Baltimore) 1969, 48, 61–85. [Google Scholar] [CrossRef]
- de Baaij, J.H.; Hoenderop, J.G.; Bindels, R.J. Magnesium in man: Implications for health and disease. Physiol. Rev. 2015, 95, 1–46. [Google Scholar] [CrossRef]
- Bara, M.; Guiet-Bara, A.; Durlach, J. Regulation of sodium and potassium pathways by magnesium in cell membranes. Magnes. Res. 1993, 6, 167–177. [Google Scholar]
- Moshfegh, A.G.; Goldman, J.; Ahuja, J.; Rhodes, D.; Lacomb, R. What We Eat in America, NHANES 2005–2006: Usual Nutrient Intakes from Food and Water Compared to 1997 Dietary Reference Intakes for Vitamin D, Calcium, Phosphorus, and Magnesium; U.S. Department of Agriculture, Agricultural Research Service: Beltsville, MD, USA, 2009.
- Institute of Medicine (IOM). Food and Nutrition Board. In Dietary Reference Intakes: Calcium, Phosphorus, Magnesium, Vitamin D and Fluoride; National Academy Press: Washington, DC, USA, 1997. [Google Scholar]
- Crook, M.A.; Hally, V.; Panteli, J.V. The importance of the refeeding syndrome. Nutrition 2001, 17, 632–637. [Google Scholar] [CrossRef]
- Agus, Z.S. Hypomagnesemia. J. Am. Soc. Nephrol. 1999, 10, 1616–1622. [Google Scholar]
- Cundy, T.; Dissanayake, A. Severe hypomagnesaemia in long-term users of proton-pump inhibitors. Clin. Endocrinol. 2008, 69, 338–341. [Google Scholar] [CrossRef] [PubMed]
- FDA Drug Safety Communication: Low Magnesium Levels can be Associated with Long-Term Use of Proton Pump Inhibitor Drugs (PPIS). Available online: https://www.fda.gov/Drugs/DrugSafety/ucm245011.htm (accessed on 20 January 2018).
- Cheungpasitporn, W.; Thongprayoon, C.; Kittanamongkolchai, W.; Srivali, N.; Edmonds, P.J.; Ungprasert, P.; O’Corragain, O.A.; Korpaisarn, S.; Erickson, S.B. Proton pump inhibitors linked to hypomagnesemia: A systematic review and meta-analysis of observational studies. Ren. Fail 2015, 37, 1237–1241. [Google Scholar] [CrossRef]
- Efstratiadis, G.; Sarigianni, M.; Gougourelas, I. Hypomagnesemia and cardiovascular system. Hippokratia 2006, 10, 147–152. [Google Scholar] [PubMed]
- Cooper, H.A.; Domanski, M.J.; Rosenberg, Y.; Norman, J.; Scott, J.H.; Assmann, S.F.; McKinlay, S.M.; Hochman, J.S.; Antman, E.M.; MAGIC Trial Investigators. Acute ST-segment elevation myocardial infarction and prior stroke: An analysis from the magnesium in coronaries (MAGIC) trial. Am. Heart. J. 2004, 148, 1012–1019. [Google Scholar] [CrossRef]
- Moens, A.L.; Claeys, M.J.; Timmermans, J.P.; Vrints, C.J. Myocardial ischemia/reperfusion-injury, a clinical view on a complex pathophysiological process. Int. J. Cardiol. 2005, 100, 179–190. [Google Scholar] [CrossRef]
- Hennekens, C.H.; Albert, C.M.; Godfried, S.L.; Gaziano, J.M.; Buring, J.E. Adjunctive drug therapy of acute myocardial infarction--evidence from clinical trials. N. Engl. J. Med. 1996, 335, 1660–1667. [Google Scholar] [CrossRef] [PubMed]
- Agus, Z.S.; Morad, M. Modulation of cardiac ion channels by magnesium. Annu. Rev. Physiol. 1991, 53, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. Dash collaborative research group. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Pham, P.C.; Pham, P.M.; Pham, S.V.; Miller, J.M.; Pham, P.T. Hypomagnesemia in patients with type 2 diabetes. Clin. J. Am. Soc. Nephrol. 2007, 2, 366–373. [Google Scholar] [CrossRef]
- Garnier, A.S.; Duveau, A.; Planchais, M.; Subra, J.F.; Sayegh, J.; Augusto, J.F. Serum magnesium after kidney transplantation: A systematic review. Nutrients 2018, 10, 729. [Google Scholar] [CrossRef]
- Castiglioni, S.; Cazzaniga, A.; Albisetti, W.; Maier, J.A. Magnesium and osteoporosis: Current state of knowledge and future research directions. Nutrients 2013, 5, 3022–3033. [Google Scholar] [CrossRef]
- Kilic, H.; Kanbay, A.; Karalezli, A.; Babaoglu, E.; Hasanoglu, H.C.; Erel, O.; Ates, C. The relationship between hypomagnesemia and pulmonary function tests in patients with chronic asthma. Med. Princ. Pract. 2018, 27, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Haldar, A.K.; Ghosh, I.; Saha, S.K.; Das, A.; Biswas, S. Serum magnesium and stable asthma: Is there a link? Lung India 2010, 27, 205–208. [Google Scholar] [CrossRef]
- Daliparty, V.M.; Manu, M.K.; Mohapatra, A.K. Serum magnesium levels and its correlation with level of control in patients with asthma: A hospital-based, cross-sectional, prospective study. Lung India 2018, 35, 407–410. [Google Scholar]
- Litonjua, A.A. Dietary factors and the development of asthma. Immunol. Allergy Clin. North Am. 2008, 28, 603–629. [Google Scholar] [CrossRef][Green Version]
- Levy, F.L.; Adams-Huet, B.; Pak, C.Y. Ambulatory evaluation of nephrolithiasis: An update of a 1980 protocol. Am. J. Med. 1995, 98, 50–59. [Google Scholar] [CrossRef]
- Altman, D.; Carroli, G.; Duley, L.; Farrell, B.; Moodley, J.; Neilson, J.; Smith, D.; Magpie Trial Collaborative Group. Do women with pre-eclampsia, and their babies, benefit from magnesium sulphate? The magpie trial: A randomised placebo-controlled trial. Lancet 2002, 359, 1877–1890. [Google Scholar]
- Huang, C.L.; Kuo, E. Mechanism of hypokalemia in magnesium deficiency. J. Am. Soc. Nephrol. 2007, 18, 2649–2652. [Google Scholar] [CrossRef]
- Ryan, M.P. Interrelationships of magnesium and potassium homeostasis. Miner Electrolyte Metab. 1993, 19, 290–295. [Google Scholar]
- Elin, R.J. Assessment of magnesium status. Clin. Chem. 1987, 33, 1965–1970. [Google Scholar]
- Jahnen-Dechent, W.; Ketteler, M. Magnesium basics. Clin. Kidney. J. 2012, 5, i3–i14. [Google Scholar] [CrossRef]
- Martin, K.J.; Gonzalez, E.A.; Slatopolsky, E. Clinical consequences and management of hypomagnesemia. J. Am. Soc. Nephrol. 2009, 20, 2291–2295. [Google Scholar] [CrossRef]
- American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American geriatrics society 2015 updated beers criteria for potentially inappropriate medication use in older adults. J. Am. Geriatr. Soc. 2015, 63, 2227–2246. [Google Scholar] [CrossRef]
- USDA National Nutrient Database for Standard Reference, Release 25; U.S. Department of Agriculture, Agricultural Research Service, Nutrient Data Laboratory Home: Beltsville, MD, USA, 2012.
- Coudray, C.; Rambeau, M.; Feillet-Coudray, C.; Gueux, E.; Tressol, J.C.; Mazur, A.; Rayssiguier, Y. Study of magnesium bioavailability from ten organic and inorganic mg salts in mg-depleted rats using a stable isotope approach. Magnes. Res. 2005, 18, 215–223. [Google Scholar]
- Guerrera, M.P.; Volpe, S.L.; Mao, J.J. Therapeutic uses of magnesium. Am. Fam. Physician. 2009, 80, 157–162. [Google Scholar] [PubMed]
- Firoz, M.; Graber, M. Bioavailability of us commercial magnesium preparations. Magnes. Res. 2001, 14, 257–262. [Google Scholar] [PubMed]
- Ranade, V.V.; Somberg, J.C. Bioavailability and pharmacokinetics of magnesium after administration of magnesium salts to humans. Am. J. Ther. 2001, 8, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Drugs. Available online: https://online.epocrates.com/ (accessed on 20 January 2019).
- McCarthy, J.T.; Kumar, R. Divalent cation metabolism: Magnesium. In The Schrier Atlas of Diseases of the Kidney; Wiley-Blackwell: Hoboken, NJ, USA, 1999; Volume 1, pp. 1–11. [Google Scholar]
| Decreased Intake |
| Decreased Dietary consumption |
| Alcohol Dependence |
| Parenteral Nutrition |
| Redistribution from Extracellular to Intracellular Compartment: |
| Refeeding Syndrome |
| Hungry Bone Syndrome |
| Treatment of Diabetic Ketoacidosis |
| Acute Pancreatitis |
| Gastrointestinal Losses: |
| Diarrhea |
| Vomiting |
| Nasogastric suction |
| Fistulas |
| Malabsorption |
| Small bowel bypass surgery |
| Proton Pump Inhibitors |
| Renal Losses: |
| Familial: |
| Bartter syndrome, Gitelman syndrome, Familial hypomagnesemia with hypercalciuria and nephrocalcinosis (FHHNC) |
| Acquired: |
| Medications: Thiazide Diuretic, Aminoglycoside Antibiotics, Amphotericin B, Cisplatin, Pentamidine, Tacrolimus, Cyclosporine |
| Alcohol Dependence, Hypercalcemia |
| Age | Male | Female | Pregnancy | Lactation |
|---|---|---|---|---|
| 19–30 years | 400 | 310 | 350 | 310 |
| 31–50 years | 420 | 320 | 360 | 320 |
| >51 years | 420 | 320 |
| Neuromuscular/Nervous System: |
| Positive Chvostek’s And Trousseau’s Signs, Tremor, Fasciculations, Tetany, Headaches, Seizures, Fatigue, Generalized Fatigue, Asthenia |
| Cardiovascular: |
| Atherosclerotic Vascular Disease/Coronary Artery Disease |
| Arrhythmias: Torsades de pointes, PR prolongation, progressive QRS widening and diminution of T-waves |
| Hypertension |
| Congestive Heart Failure |
| Endocrine: Altered Glucose Homeostasis/Diabetic Complications |
| Osteoporosis |
| Biochemical/Others: |
| Hypokalemia |
| Hypocalcemia |
| Asthma |
| Nephrolithiasis |
| Food | Milligrams per Serving | Percent Daily Value |
|---|---|---|
| Almonds, dry roasted, 1 ounce | 80 | 20 |
| Spinach, boiled, ½ cup | 78 | 20 |
| Cashews, dry roasted, 1 ounce | 74 | 19 |
| Peanuts, oil roasted, ¼ cup | 63 | 16 |
| Cereal, shredded wheat, 2 large biscuits | 61 | 15 |
| Soymilk, plain or vanilla, 1 cup | 61 | 15 |
| Black beans, cooked, ½ cup | 60 | 15 |
| Magnesium Supplement | Elemental Magnesium (Percent) | Bioavailability (as Fractional Absorption of the Administered Dose) | Bioavailability (Relative Comparison) | Tolerability (Diarrhea) |
|---|---|---|---|---|
| Magnesium Oxide | 60 | 4% | Extremely low | ++ |
| Magnesium Carbonate | 45 | * | Extremely low | * |
| Magnesium Hydroxide | 42 | 4% | * | ++ |
| Magnesium Citrate | 16 | 12% | Good | ++ |
| Magnesium Lactate | 12 | 12% | Excellent | + |
| Magnesium Chloride | 12 | 12% | Good | + |
| Magnesium Aspartate | 10 | * | * | * |
| Magnesium Sulfate | 10 | 4% | * | ++ |
| Magnesium Gluconate | 5 | * | Good | ± |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed, F.; Mohammed, A. Magnesium: The Forgotten Electrolyte—A Review on Hypomagnesemia. Med. Sci. 2019, 7, 56. https://doi.org/10.3390/medsci7040056
Ahmed F, Mohammed A. Magnesium: The Forgotten Electrolyte—A Review on Hypomagnesemia. Medical Sciences. 2019; 7(4):56. https://doi.org/10.3390/medsci7040056
Chicago/Turabian StyleAhmed, Faheemuddin, and Abdul Mohammed. 2019. "Magnesium: The Forgotten Electrolyte—A Review on Hypomagnesemia" Medical Sciences 7, no. 4: 56. https://doi.org/10.3390/medsci7040056
APA StyleAhmed, F., & Mohammed, A. (2019). Magnesium: The Forgotten Electrolyte—A Review on Hypomagnesemia. Medical Sciences, 7(4), 56. https://doi.org/10.3390/medsci7040056




