Narcolepsy in Children and Adults: A Guide to Improved Recognition, Diagnosis and Management
Abstract
1. Introduction
2. Inadequate and Delayed Diagnosis
3. Approach to Symptom Recognition
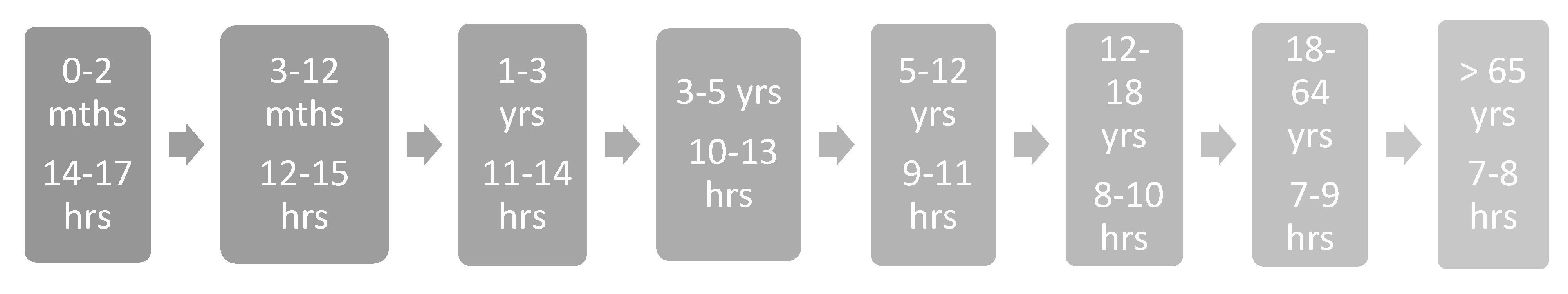
3.1. Pediatrics
3.2. Adults
4. Approach to Diagnosis
4.1. Sleep Testing
- ✓
- Prepare patient for length of study and description of the overnight PSG and the MSLT.
- ✓
- Evaluate medications to determine if any influence sleep latency or are REM suppressants.
- ✓
- Is it safe to temporarily discontinue these medications?
- ○
- If so, wean medication with plans to be off medication for at least 5 half-lives or 2 weeks if half-life unknown.
- ✓
- Complete sleep diaries and/or actigraphy to document usual sleep wake patterns
- ○
- Consider optimizing sleep schedule first if shift work, delayed sleep phase or chronic insufficient sleep is present.
- ✓
- Ensure PSG demonstrates at least 6 h of total sleep time prior to MSLT.
- ✓
- Evaluate PSG for additional supportive evidence of narcolepsy.
- ○
- Sleep fragmentation, RBD, REMWA, increased PLMs, SOREMP.
- ✓
- Consider Tanner Staging and age of patient when evaluating average sleep latency.
- ○
- If study is borderline and history is convincing, consider repeat study in 6 months versus CSF hypocretin (orexin), if suspicious on NT1.
- ✓
- Attempt to keep patient awake between naps during MSLT.
- ○
- If unable to do so, consider continuous recording during MSLT to evaluate sleep wake pattern.*
4.2. Human Leukocyte Antigen (HLA) Testing
4.3. Cerebrospinal fluid (CSF) Testing
- Non-diagnostic PSG/MSLT testing in a patient with cataplexy and EDS
- Non-diagnostic PSG/MSLT testing in a patient without cataplexy, EDS and HLA+
- Non-diagnostic PSG/MSLT testing in a patient with EDS, HLA+, +/− cataplexy, who is unable to discontinue REM suppressing/sleep influencing medications due to safety/medical concerns
- Pediatric patients at extreme ages (i.e less than 5 years old)
- Pediatric patients with abnormal SL based on tanner stage, but non-diagnostic based on criteria
5. Approach to Treatment
5.1. Pharmacologic Strategies
5.2. Non-Pharmacologic Strategies
6. Conclusions
Funding
Conflicts of Interest
References
- American Academy of Sleep Medicine. International Classification of Sleep Disorders, 3rd ed.; American Academy of Sleep Medicine: Darien, CT, USA, 2014. [Google Scholar]
- Moscovitch, A.; Partinen, M.; Guilleminault, C. The positive diagnosis of narcolepsy and narcolepsy’s borderland. Neurology 1993, 43, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Dye, T.J.; Gurbani, N.; Simakajornboon, N. Epidemiology and pathophysiology of childhood narcolepsy. Paediatr. Respir. Rev. 2018, 25, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Scheer, D.; Schwartz, S.W.; Parr, M.; Zgibor, J.; Sanchez-Anguiano, A.; Rajaram, L. Prevalence and incidence of narcolepsy in a US health care claims database, 2008–2010. Sleep 2019, 42. [Google Scholar] [CrossRef] [PubMed]
- Prevalence of Diagnosed Pediatric Narcolepsy in the United States. In Sleep; Oxford Univ Press Inc. Journals Dept.: Cary, NC, USA, 2019.
- Rocca, F.; Pizza, F.; Ricci, E.; Plazzi, G. Narcolepsy during Childhood: An Update. Neuropediatrics 2015, 46, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Okun, M.L.; Lin, L.; Pelin, Z.; Hong, S.; Mignot, E. Clinical aspects of narcolepsy-cataplexy across ethnic groups. Sleep 2002, 25, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Dauvilliers, Y.; Montplaisir, J.; Molinari, N.; Carlander, B.; Ondze, B.; Besset, A.; Billiard, M. Age at onset of narcolepsy in two large populations of patients in France and Quebec. Neurology 2001, 57, 2029–2033. [Google Scholar] [CrossRef]
- Thorpy, M.; Morse, A.M. Reducing the Clinical and Socioeconomic Burden of Narcolepsy by Earlier Diagnosis and Effective Treatment. Sleep Med. Clin. 2017, 12, 61–71. [Google Scholar] [CrossRef]
- Thorpy, M.; Cronin, S.; Temple, H. Age of onset and time to diagnosis of narcolepsy. Neurology 1999, 52, A110. [Google Scholar]
- Morse, A.M.; Sanjeev, K. Narcolepsy and Psychiatric Disorders: Comorbidities or Shared Pathophysiology? Med. Sci. 2018, 6, 16. [Google Scholar] [CrossRef]
- Ohayon, M.; Thorpy, M.; Black, J.; Williams, M.J.; Pasta, D.J.; Hyman, D.; Villa, K.F. Misdiagnoses and Comorbidities among Participants in the Nexus Narcolepsy Registry. Neurology 2019, 92, 6–37. [Google Scholar]
- Rosenberg, R.; Kim, A.Y. The AWAKEN Survey: Knowledge of Narcolepsy among Physicians and the General Population. Postgrad. Med. 2014, 126, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Johns, M. The assessment of sleepiness in children and adolescents. Sleep Biol. Rhythm. 2015, 13 (Suppl. 1), 97. [Google Scholar]
- Johns, M.W. A New Method for Measuring Daytime Sleepiness: The Epworth Sleepiness Scale. Sleep 1991, 14, 540–545. [Google Scholar] [CrossRef]
- Anic-Labat, S.; Guilleminault, C.; Kraemer, H.C.; Meehan, J.; Arrigoni, J.; Mignot, E. Validation of a cataplexy questionnaire in 983 sleep-disorders patients. Sleep 1999, 22, 77–87. [Google Scholar] [PubMed]
- Sturzenegger, C.; Baumann, C.R.; Lammers, G.J.; Kallweit, U.; van der Zande, W.L.; Bassetti, C.L. Swiss Narcolepsy Scale: A simple screening tool for hypocretin-deficient narcolepsy with cataplexy. Clin. Transl. Neurosci. 2018, 2, 2514183X18794175. [Google Scholar] [CrossRef]
- Hirshkowitz, M.; Whiton, K.; Albert, S.M.; Alessi, C.; Bruni, O.; DonCarlos, L.; Hazen, N.; Herman, J.; Katz, E.S.; Kheirandish-Gozal, L.; et al. National Sleep Foundation’s sleep time duration recommendations: Methodology and results summary. Sleep Health 2015, 1, 40–43. [Google Scholar] [CrossRef]
- Aran, A.; Lin, L.; Nevsimalova, S.; Plazzi, G.; Hong, S.C.; Weiner, K.; Zeitzer, J.; Mignot, E. Elevated Anti-Streptococcal Antibodies in Patients with Recent Narcolepsy Onset. Sleep 2009, 32, 979–983. [Google Scholar] [CrossRef]
- Han, F.; Lin, L.; Warby, S.C.; Faraco, J.; Li, J.; Dong, S.X.; An, P.; Zhao, L.; Wang, L.H.; Li, Q.Y.; et al. Narcolepsy onset is seasonal and increased following the 2009 H1N1 pandemic in china. Ann. Neurol. 2011, 70, 410–417. [Google Scholar] [CrossRef]
- Postiglione, E.; Antelmi, E.; Pizza, F.; Lecendreux, M.; Dauvilliers, Y.; Plazzi, G. The clinical spectrum of childhood narcolepsy. Sleep Med. Rev. 2018, 38, 70–85. [Google Scholar] [CrossRef]
- Piško, J.; Pastorek, L.; Bušková, J.; Sonka, K.; Nevsimalova, S. Nightmares in narcolepsy: Underinvestigated symptom? Sleep Med. 2014, 15, 967–972. [Google Scholar] [CrossRef]
- Poli, F.; Pizza, F.; Mignot, E.; Ferri, R.; Pagotto, U.; Taheri, S.; Finotti, E.; Bernardi, F.; Pirazzoli, P.; Cicognani, A.; et al. High Prevalence of Precocious Puberty and Obesity in Childhood Narcolepsy with Cataplexy. Sleep 2013, 36, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Morandin, M. Understanding Automatic Behavior in Narcolepsy: New Insights Using a Phenomenological Approach. Open Sleep J. 2013, 6, 1–7. [Google Scholar] [CrossRef]
- Littner, M.R.; Kushida, C.; Wise, M.; Davila, D.G.; Morgenthaler, T.; Lee-Chiong, T.; Hirshkowitz, M.; Loube, D.L.; Bailey, D.; Berry, R.B.; et al. Practice Parameters for Clinical Use of the Multiple Sleep Latency Test and the Maintenance of Wakefulness Test. Sleep 2005, 28, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Andlauer, O.; Moore, H.; Jouhier, L.; Drake, C.; Peppard, P.E.; Han, F.; Hong, S.-C.; Poli, F.; Plazzi, G.; O’Hara, R.; et al. Nocturnal rapid eye movement sleep latency for identifying patients with narcolepsy/hypocretin deficiency. JAMA Neurol. 2013, 70, 891–902. [Google Scholar] [CrossRef]
- Aldrich, M.S.; Chervin, R.D.; Malow, B.A. Value of the multiple sleep latency test (MSLT) for the diagnosis of narcolepsy. Sleep 1997, 20, 620–629. [Google Scholar] [PubMed]
- Carskadon, M. Sleeping and Waking Disorders: Indications and Techniques; Addison Wesley: Menlo Park, CA, USA, 1982. [Google Scholar]
- Ollila, H.M.; Ravel, J.-M.; Han, F.; Faraco, J.; Lin, L.; Zheng, X.; Plazzi, G.; Dauvilliers, Y.; Pizza, F.; Hong, S.-C.; et al. HLA-DPB1 and HLA Class I Confer Risk of and Protection from Narcolepsy. Am. J. Hum. Genet. 2015, 96, 852. [Google Scholar] [CrossRef]
- Pelin, Z.; Guilleminault, C.; Risch, N.; Mignot, E. F.C. Grumet US Modafinil in Narcolepsy Multicenter Study Group HLA-DQB1*0602 homozygosity increases relative risk for narcolepsy but not disease severity in two ethnic groups. Tissue Antigens 1998, 51, 96–100. [Google Scholar] [CrossRef]
- Mignot, E.; Lin, L.; Rogers, W.; Honda, Y.; Qiu, X.; Lin, X.; Okun, M.; Hohjoh, H.; Miki, T.; Hsu, S.H.; et al. Complex HLA-DR and -DQ Interactions Confer Risk of Narcolepsy-Cataplexy in Three Ethnic Groups. Am. J. Hum. Genet. 2001, 68, 686–699. [Google Scholar] [CrossRef]
- Luca, G.; Haba-Rubio, J.; Dauvilliers, Y.; Lammers, G.-J.; Overeem, S.; Donjacour, C.E.; Mayer, G.; Javidi, S.; Iranzo, A.; Santamaria, J.; et al. Clinical, polysomnographic and genome-wide association analyses of narcolepsy with cataplexy: A European Narcolepsy Network study. J. Sleep Res. 2013, 22, 482–495. [Google Scholar] [CrossRef]
- Nevšímalová, S.; Mignot, E.; Šonka, K.; Arrigoni, J.L. Familial aspects of narcolepsy-cataplexy in the Czech Republic. Sleep 1997, 20, 1021–1026. [Google Scholar] [CrossRef][Green Version]
- Mignot, E.; Lammers, G.J.; Ripley, B.; Okun, M.; Nevsimalova, S.; Overeem, S.; Vankova, J.; Black, J.; Harsh, J.; Bassetti, C.; et al. The role of cerebrospinal fluid hypocretin measurement in the diagnosis of narcolepsy and other hypersomnias. Arch. Neurol. 2002, 59, 1553–1562. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.K.; Muehlbach, M.J.; Schweitzer, P.K. Hypnotics and caffeine as countermeasures for shiftwork-related sleepiness and sleep disturbance. J. Sleep Res. 1995, 4, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.W.; Fredrickson, P.A.; Lin, S.C. Narcolepsy update. In Mayo Clinic Proceedings; Elsevier: Amsterdam, The Netherlands, 1990; Volume 65, pp. 991–998. [Google Scholar]
- Agudelo, H.A.M.; Correa, U.J.; Sierra, J.C.; Pandi-Perumal, S.R.; Schenck, C.H. Cognitive behavioral treatment for narcolepsy: Can it complement pharmacotherapy? Sleep Sci. 2014, 7, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Mullington, J.; Broughton, R. Scheduled Naps in the Management of Daytime Sleepiness in Narcolepsy-Cataplexy. Sleep 1993, 16, 444–456. [Google Scholar] [CrossRef] [PubMed]
- Rosekind, M.R.; Smith, R.M.; Miller, D.L.; Co, E.L.; Gregory, K.B.; Webbon, L.L.; Gander, P.H.; Lebacqz, J.V. Alertness management: Strategic naps in operational settings. J. Sleep Res. 1995, 4, 62–66. [Google Scholar] [CrossRef]
- Alaia, S.L. Life effects of narcolepsy: Measures of negative impact, social support, and psychological weil-being. Loss Grief Care 1992, 5, 1–22. [Google Scholar]
- Neikrug, A.B.; Crawford, M.R.; Ong, J.C. Behavioral sleep medicine services for hypersomnia disorders: A survey study. Behav. Sleep Med. 2017, 15, 158–171. [Google Scholar] [CrossRef]
- Husain, A.M.; Yancy, W.S., Jr.; Carwile, S.T.; Miller, P.P.; Westman, E.C. Diet therapy for narcolepsy. Neurology 2004, 62, 2300–2302. [Google Scholar] [CrossRef]
- Cohen, F.L.; Nehring, W.M.; Cloninger, L. Symptom Description and Management in Narcolepsy. Holist. Nurs. Pract. 1996, 10, 44–53. [Google Scholar] [CrossRef]
- Brown, T.M.; Swick, T.J.; Alessi, C.; Aurora, R.N.; Boehlecke, B.; Chesson, A.L.; Friedman, L.; Maganti, R.; Owens, J.; Pancer, J.; et al. Practice Parameters for the Treatment of Narcolepsy and other Hypersomnias of Central Origin. Sleep 2007, 30, 1705–1711. [Google Scholar]
- Xyrem (Sodium Oxybate) Oral Solution CIII; Jazz Pharmaceuticals, Inc.: Palo Alto, CA, USA, 2018.
- Barateau, L.; Dauvilliers, Y. Recent advances in treatment for narcolepsy. Ther. Adv. Neurol. Disord. 2019, 12, 1756286419875622. [Google Scholar] [CrossRef] [PubMed]
- Thorpy, M.J.; Bogan, R.K. Update on the Pharmacologic Management of Narcolepsy: Mechanisms of Action and Clinical Implications. Sleep Med. 2019. [Google Scholar] [CrossRef]
- Flygare, J. Succeeding in School and in the Workplace with Narcolepsy; Springer Science and Business Media LLC: Berlin, Gremany, 2016; pp. 395–405. [Google Scholar]
- US Equal Employment Opportunity Commission. 2010; Procedures for providing reasonable accommodation for individuals with disabilities. Available online: https://www.eeoc.gov/eeoc/internal/reasonable_accommodation.cfm (accessed on 26 November 2019).
- Black, J.; Reaven, N.L.; Funk, S.E.; McGaughey, K.; Ohayon, M.; Guilleminault, C.; Ruoff, C.; Mignot, E. The Burden of Narcolepsy Disease (BOND) study: Health-care utilization and cost findings. Sleep Med. 2014, 15, 522–529. [Google Scholar] [CrossRef] [PubMed]
| Type | Description | Example |
|---|---|---|
| Negative | Transient loss of antigravity muscle tone, frequently evoked by emotion. Near continuous hypotonia without emotional stimulus | Generalized collapse to the ground with preserved awareness, knee buckling, loss of tone in hands, head drop. General floppiness, abnormal/semi-ataxic gait. |
| Active | Hyperkinetic features that may be enhanced by emotional stimuli. Complex Movement disorder. | Perioral/tongue movements, facial grimacing, eyebrow raising. Tic like stereotyped motor movements |
| Mixed | “Cataplectic Facies” | Facial hypotonia with ptosis, mouth opening and tongue protrusion. |
| Symptoms Treated | Drug | FDA Approval (Ages) |
|---|---|---|
| Excessive Daytime Sleepiness | Modafinil | Yes (18 years and older) |
| Armodafinil | Yes (18 years and older) | |
| Sodium Oxybate | Yes (ages 7 years and older) | |
| Methylphenidate | Yes (ages 6 years and older) | |
| Dextroamphetamine | Yes (ages 6 years and older) | |
| Solriamfetol | Yes (18 years and older) | |
| Pitolisant | Yes (18 years and older) | |
| Cataplexy | Sodium Oxybate | Yes (ages 7 years and older) |
| Venlafaxine | No | |
| TCA * (e.g., protryptiline, clormipramine) | No | |
| SSRI* (e.g fluoxetine) | No | |
| Atomoxetine ** | No | |
| EDS + Cataplexy | Sodium Oxybate | Yes (ages 7 years and older) |
| Behavioral Strategy | Description |
|---|---|
| Strategic Caffeine | Plan use of caffeine intake to promote performance and alertness [35] |
| Sleep Hygiene | Sleep related behaviors to enhance and achieve age appropriate number of hours of sleep [36] |
| Sleep Scheduling | Regular sleep–wake schedule [36] |
| Cognitive Behavioral Therapy | Systematic application of techniques needed to evaluate and improve behavior [37] |
| Scheduled napping | Nap that is scheduled during individuals typical height of sleep inertia [38] |
| Strategic napping | Planned nap of specific duration to promote performance and alertness [39] |
| Support Groups | In person or online social communities for support [40] |
| Exercise | Any cardiovascular activity for physical engagement [41] |
| Mindfulness | Meditation and self-awareness [41] |
| Yoga | breath control, simple meditation, adoption of specific postures for health/relaxation [41] |
| Diet | Small, frequent meals to mitigate post-prandial. Low carbohydrate, ketogenic diet [42] |
| Temperature Manipulations | Cold temperature environments and avoidance of hot environments [43] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morse, A.M. Narcolepsy in Children and Adults: A Guide to Improved Recognition, Diagnosis and Management. Med. Sci. 2019, 7, 106. https://doi.org/10.3390/medsci7120106
Morse AM. Narcolepsy in Children and Adults: A Guide to Improved Recognition, Diagnosis and Management. Medical Sciences. 2019; 7(12):106. https://doi.org/10.3390/medsci7120106
Chicago/Turabian StyleMorse, Anne Marie. 2019. "Narcolepsy in Children and Adults: A Guide to Improved Recognition, Diagnosis and Management" Medical Sciences 7, no. 12: 106. https://doi.org/10.3390/medsci7120106
APA StyleMorse, A. M. (2019). Narcolepsy in Children and Adults: A Guide to Improved Recognition, Diagnosis and Management. Medical Sciences, 7(12), 106. https://doi.org/10.3390/medsci7120106




