Human Dendritic Cells: Ontogeny and Their Subsets in Health and Disease
Abstract
1. Background
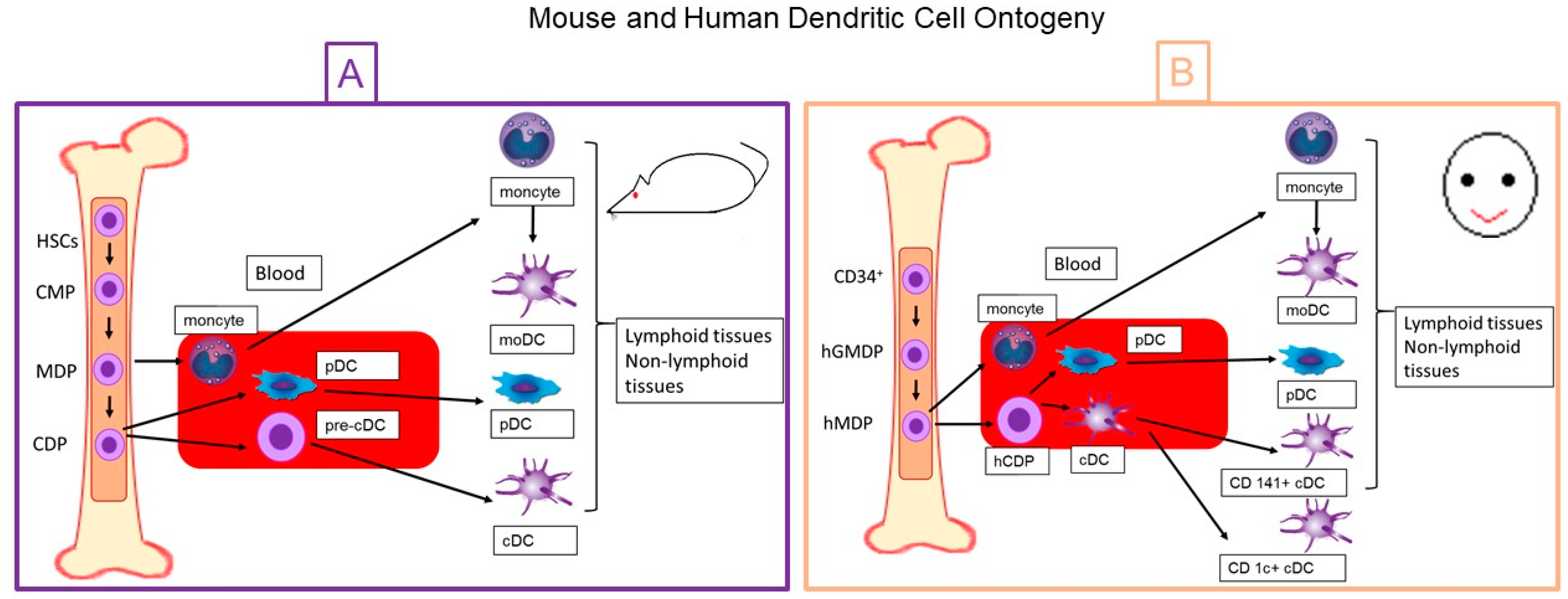
2. Dentritic Cell Ontogeny
2.1. Mouse Dendritic Cells
2.2. Human Dendritic Cells
3. Dendritic Cell Subpopulations
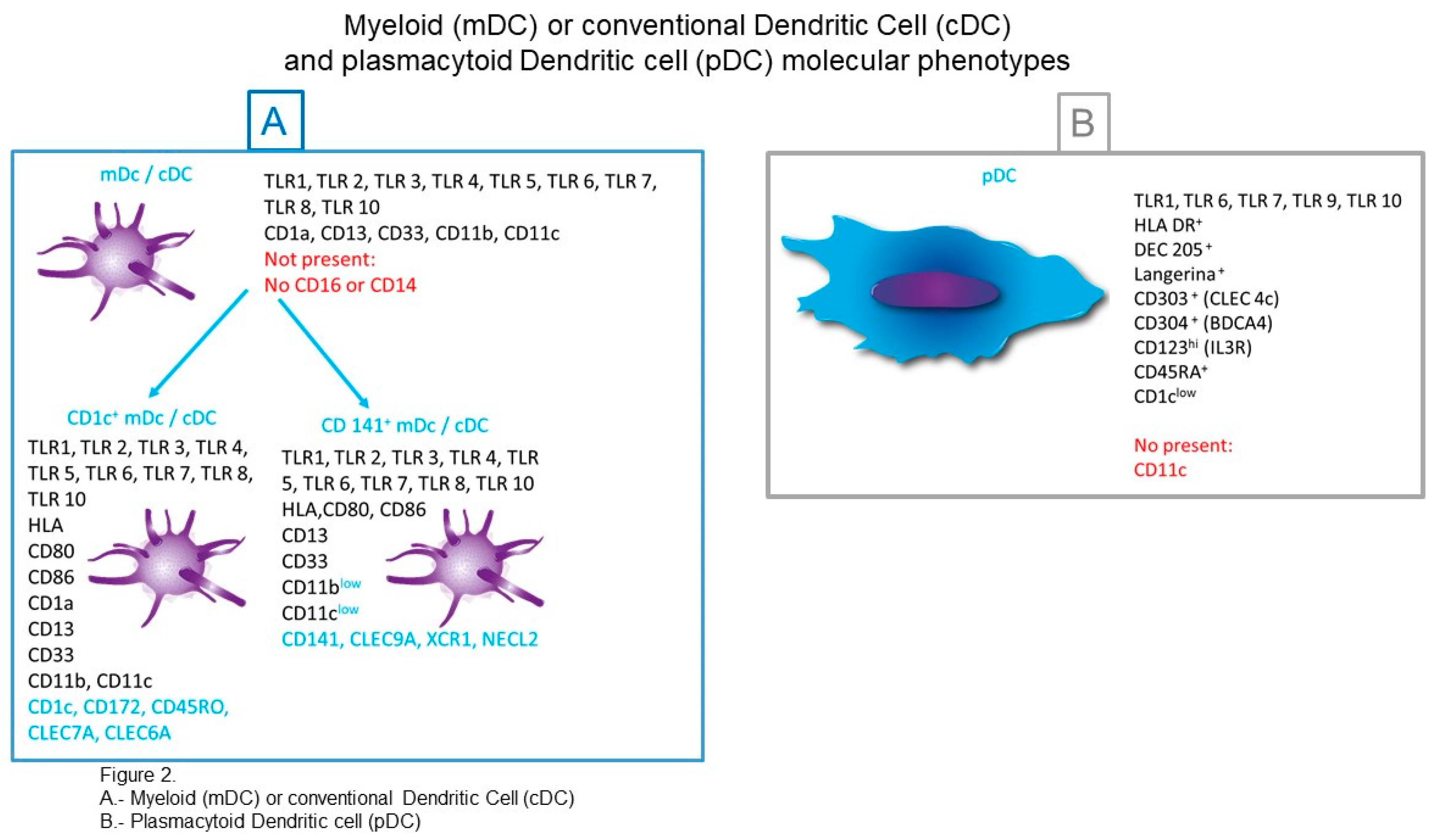
3.1. cDCs (Conventional Dendritic Cells) or mDCs (Myeloid Dendritic Cells)
3.1.1. CD1c+ cDCs
3.1.2. CD141+ cDCs (Conventional Dendritic Cells)
3.2. pDCs (Plasmacytoid DCs)
3.3. Dendritic Cells That Respond to Specific Microorganisms
3.3.1. TNF-α and iNOS Producing Dendritic Cells (Tip-DCs) and Myeloid-Derived Suppressor Cells (MDSC)
3.4. Dendritic Cells CD14+
3.5. Dendritic Cells Derived from Monocytes (moDC)
4. Dendritic Cell Classification Based on Its Anatomical Location
4.1. Blood Dendritic Cells
4.2. Dendritic Cell Migrants and Peripheral Residents in Non-Lymphoid Tissues
4.3. Dendritic Cell Residents of Secondary Lymphoid Tissues
5. Dendritic Cell Maturation
6. Mechanisms Used by Dendritic Cells for Recognition and Antigen Capture
7. Mechanisms Used by Dendritic Cells for Antigenic Presentation
7.1. Mechanisms of Antigen Presentation by Dendritic Cells
7.2. Antigen Presentation by MHC-I
7.3. Cross-Presentation
7.4. Antigen Presentation by MHC-II Molecules
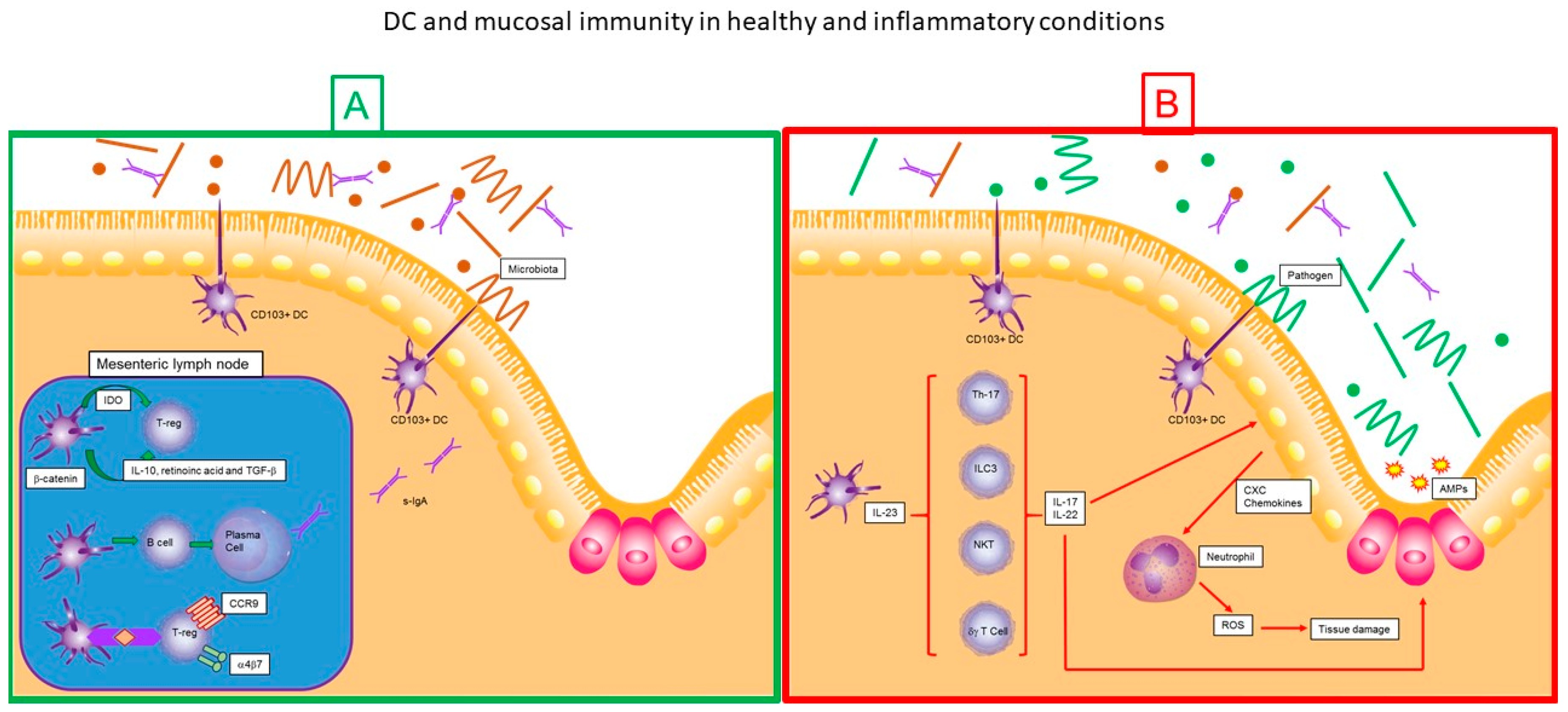
8. Dendritic Cells and Mucosal Immunity
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
List of Abbreviations
| DCs | dendritic cells |
| Mφ | macrophages |
| MHC | major histocompatibility complex |
| Alpha X | complement component 3 receptor 4 subunit |
| PRRs | pattern recognition receptors |
| PAMPs | pathogen-associated molecular patterns |
| HSCs | hematopoietic stem cells |
| CMPs | common myeloid progenitors |
| DCPs | dendritic cell progenitors |
| pDCs | plasmacytoid dendritic cells |
| pre-cDC | precursos of conventional dendritic cells |
| Flt3L | FMS-like tyrosine kinase 3 receptor ligand |
| M-CSF | macrophage-colony stimulating factor |
| GM-CSF | granulocyte macrophage colony stimulating factor |
| hGMDP | progenitor of granulocytes, monocytes and human dendritic cells |
| hMDP | progenitor of dendritic cells and human monocytes |
| hPDC | common progenitor of human dendritic cells |
| GMP | granulocyte and macrophage progenitor |
| cDCs | conventional dendritic cells |
| mDCs | myeloid dendritic cells |
| pDCs | plasmacytoid dendritic cells |
| TLRs | toll-like receptors |
| EpCAM | langerin and epithelial cell adhesion molecule |
| SIGLEC | sialic acid-binding immunoglobulin-like lectin |
| SIRPα | Signal regulatory protein |
| TNF-α | necrosis tumor factor-a |
| IL | Interleukin |
| CLEC | C-type lectin-like receptor |
| CTLR | c-type lectin-like receptor |
| Necl2 | nectin-like protein 2 |
| IFN | Interferon |
| iNOS | inducible nitric oxide synthase |
| NO | nitric oxide |
| CCR7 | CC chemokine receptor 7 |
| intDCs | interstitial dermal cells |
| Ig | immunoglobulin |
| Ag | antigens |
| APC | antigen presenting cell |
| DC-LAMP | membrane protein associated with lysosomes |
| FcγR | receptor of the Fc region of IgG |
| FcεR | receptor of the Fc region of IgE |
| CR | complement receptor |
| DCIR | dendritic cell immunoreceptor |
| ER | endoplasmic reticulum |
| TAP | transporter associated with antigen processing |
| ERAP-1 | endoplasmic reticulum-type 1 associated aminopeptidase |
| IRAP | endosomal aminopeptidase responsive to insulin |
| ERGIC | intermediate compartment AGRE |
| Li | invariable chain |
| CLIP | invariable class II chain |
| pMHCII | major histocompatibility complex II-peptide |
| RILP | lysosomal interaction protein Rab7 |
| MVB | multivesicular endosomes |
| MIS | mucosal immune system |
| MALT | mucosa-associated lymphoid tissue |
| GALT | gut-associated lymphoid tissue |
| NALT | nasopharyngeal-associated lymphoid tissue |
| MLN | mesenteric lymph nodes |
| IDO | indoleamine 2,3-dioxygenase |
| T-reg | regulatory T lymphocytes |
| IBS | irritable bowel syndrome |
| IBD | irritable bowel disease |
| TH17 | T helper 17 |
| NKT | natural killer T cells |
| ILC3s | 3 innate lymphoid cells |
| AMPs | antimicrobial peptides |
| ROS | reactive oxygen species |
References
- Breathnach, A.S. The distribution of Langerhans cells within the human hair follicle, and some observations on its staining properties with gold chloride. J. Anat. 1963, 97, 73–80. [Google Scholar] [PubMed]
- Steinman, R.M.; Cohn, Z.A. Identification of a novel cell type in peripheral lymphoid organs of mice. I. Morphology, quantitation, tissue distribution. J. Exp. Med. 1973, 137, 1142–1162. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M.; Witmer, M.D. Lymphoid dendritic cells are potent stimulators of the primary mixed leukocyte reaction in mice. Proc. Natl. Acad. Sci. USA 1978, 75, 5132–5136. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M. Dendritic cells: From the fabric of immunology. Clin. Investig. Med. 2004, 27, 231–236. [Google Scholar]
- Nussenzweig, M.C.; Steinman, R.M.; Unkeless, J.C.; Witmer, M.D.; Gutchinov, B.; Cohn, Z.A. Studies of the cell surface of mouse dendritic cells and other leukocytes. J. Exp. Med. 1981, 154, 168–187. [Google Scholar] [CrossRef] [PubMed]
- Nussenzweig, M.C.; Steinman, R.M.; Witmer, M.D.; Gutchinov, B. A monoclonal antibody specific for mouse dendritic cells. Proc. Natl. Acad. Sci. USA 1982, 79, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Haniffa, M.; Collin, M.; Ginhoux, F. Ontogeny and Functional Specialization of Dendritic Cells in Human and Mouse. Adv. Immunol. 2013, 120. [Google Scholar] [CrossRef]
- Mildner, A.; Jung, S. Development and function of dendritic cell subsets. Immunity 2014, 40, 642–656. [Google Scholar] [CrossRef] [PubMed]
- Schmid, M.A.W. Characteristics of “Tip-DCs and MDSCs” and their potential role in leishmaniasis. Front. Microbiol. 2012, 3, 74. [Google Scholar] [CrossRef] [PubMed]
- Shortman, K.; Liu, Y.J. Mouse and human dendritic cell subtypes. Nat. Rev. Immunol. 2002, 2, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Shortman, K.; Naik, S.H. Steady-state and inflammatory dendritic-cell development. Nat. Rev. Immunol. 2007, 7, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Shortman, K.; Lahoud, M.H.; Caminschi, I. Improving vaccines by targeting antigens to dendritic cells. Exp. Mol. Med. 2009, 41, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Heath, W.R.; Carbone, F.R. Dendritic cell subsets in primary and secondary T cell responses at body surfaces. Nat. Immunol. 2009, 10, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M.; Idoyaga, J. Features of the dendritic cell lineage. Immunol. Rev. 2010, 234, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Sathe, P.; Helft, J.; Miller, J.; Mortha, A. The dendritic cell lineage: Ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Ann. Rev. Immunol. 2013, 31, 563–604. [Google Scholar] [CrossRef] [PubMed]
- Fogg, D.K.; Sibon, C.; Miled, C.; Jung, S.; Aucouturier, P.; Littman, D.R.; Cumano, A.; Geissmann, F. A clonogenic bone marrow progenitor specific for macrophages and dendritic cells. Science 2006, 311, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Victora, G.D.; Schwickert, T.A.; Guermonprez, P.; Meredith, M.M.; Yao, K.; Randolph, G.J.; Rudensky, A.Y.; Nussenzweig, M. In vivo analysis of dendritic cell development and homeostasis. Science 2009, 324, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Geissmann, F.; Manz, M.G.; Jung, S.; Sieweke, M.H.; Merad, M.; Ley, K. Development of monocytes, macrophages, and dendritic cells. Science 2010, 327, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Naik, S.H.; Sathe, P.; Park, H.-Y.; Metcalf, D.; Proietto, A.I.; Dakic, A.; Carotta, S.; O’Keeffe, M.; Bahlo, M.; Papenfuss, A.; et al. Development of plasmacytoid and conventional dendritic cell subtypes from single precursor cells derived in vitro and in vivo. Nat. Immunol. 2007, 8, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Onai, N.; Obata-Onai, A.; Schmid, M.A.; Manz, M.G. Flt3 in regulation of type I interferon-producing cell and dendritic cell development. Ann. N. Y. Acad. Sci. 2007, 1106, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Onai, N.; Obata-Onai, A.; Schmid, M.A.; Ohteki, T.; Jarrossay, D.; Manz, M.G. Identification of clonogenic common Flt3 M-CSFR plasmacytoid and conventional dendritic cell progenitors in mouse bone marrow. Nat. Immunol. 2007, 8, 1207–1216. [Google Scholar] [CrossRef] [PubMed]
- Ginhoux, F.; Liu, K.; Helft, J.; Bogunovic, M.; Greter, M.; Hashimoto, D.; Price, J.; Yin, N.; Bromberg, J.; Lira, S.; et al. The origin and development of nonlymphoid tissue CD103 DCs. J. Exp. Med. 2009, 206, 3115–3130. [Google Scholar] [CrossRef] [PubMed]
- Onai, N.; Kurabayashi, K.; Hosoi-Amaike, M.; Toyama-Sorimachi, N.; Matsushima, K.; Inaba, K.; Ohteki, T. A clonogenic progenitor with prominent plasmacytoid dendritic cell developmental potential. Immunity 2013, 38, 943–957. [Google Scholar] [CrossRef] [PubMed]
- McKenna, H.J.; Stocking, K.L.; Miller, R.E.; Brasel, K.; De Smedt, T.; Maraskovsky, E.; Maliszewski, C.R.; Lynch, D.H.; Smith, J.; Pulendran, B.; et al. Mice lacking flt3 ligand have deficient hematopoiesis affecting hematopoietic progenitor cells, dendritic cells, and natural killer cells. Blood 2000, 95, 3489–3497. [Google Scholar] [PubMed]
- Karsunky, H.; Merad, M.; Cozzio, A.; Weissman, I.L.; Manz, M.G. Flt3 ligand regulates dendritic cell development from Flt3 lymphoid and myeloid-committed progenitors to Flt3 dendritic cells in vivo. J. Exp. Med. 2003, 198, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Waskow, C.; Liu, K.; Darrasse-Jèze, G.; Guermonprez, P.; Ginhoux, F.; Merad, M.; Shengelia, T.; Yao, K.; Nussenzweig, M. The receptor tyrosine kinase Flt3 is required for dendritic cell development in peripheral lymphoid tissues. Nat. Immunol. 2008, 9, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Kingston, D.; Schmid, M.A.; Onai, N.; Obata-Onai, A.; Baumjohann, D.; Manz, M.G. The concerted action of GM-CSF and Flt3-ligand on in vivo dendritic cell homeostasis. Blood 2009, 114, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Breton, G.; Oliveira, T.Y.K.; Zhou, Y.J.; Aljoufi, A.; Puhr, S.; Cameron, M.J.; Sékaly, R.P.; Nussenzweig, M.C.; Liu, K. Restricted dendritic cell and monocyte progenitors in human cord blood and bone marrow. J. Exp. Med. 2015, 212, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Ziegler-Heitbrock, L.; Ancuta, P.; Crowe, S.; Dalod, M.; Grau, V.; Hart, D.N.; Leenen, P.J.; Liu, Y.J.; MacPherson, G.; Randolph, G.J.; et al. Nomenclature of monocytes and dendritic cells in blood. Blood 2010, 116, e74–e80. [Google Scholar] [CrossRef] [PubMed]
- Collin, M.; Mcgovern, N.; Haniffa, M. Human dendritic cell subsets. Immunology 2013, 140, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Guilliams, M.; Dutertre, C.A.; Scott, C.L.; McGovern, N.; Sichien, D.; Chakarov, S.; Van Gassen, S.; Chen, J.; Poidinger, M.; De Prijck, S.; et al. Unsupervised high-dimensional analysis aligns dendritic cells across tissues and species. Immunity 2016, 45, 669–684. [Google Scholar] [CrossRef] [PubMed]
- Kassianos, A.J.; Hardy, M.Y.; Ju, X.; Vijayan, D.; Ding, Y.; Vulink, A.J.; McDonald, K.J.; Jongbloed, S.L.; Wadley, R.B.; Wells, C.; et al. Human CD1c (BDCA-1)+ myeloid dendritic cells secrete IL-10 and display an immuno-regulatory phenotype and function in response to Escherichia coli. Eur. J. Immunol. 2012, 42, 1512–1522. [Google Scholar] [CrossRef] [PubMed]
- Breton, G.; Lee, J.; Zhou, Y.J.; Schreiber, J.J.; Keler, T.; Puhr, S.; Anandasabapathy, N.; Schlesinger, S.; Caskey, M.; Liu, K.; et al. Circulating precursors of human CD1c+ and CD141+ dendritic cells. J. Exp. Med. 2015, 212, 401–413. [Google Scholar] [CrossRef] [PubMed]
- O’Keeffe, M.; Mok, W.H.; Radford, K.J. Human dendritic cell subsets and function in health and disease. Cell. Mol. Life Sci. 2015, 72, 4309–4325. [Google Scholar] [CrossRef] [PubMed]
- Haniffa, M.; Ginhoux, F.; Wang, X.N.; Bigley, V.; Abel, M.; Dimmick, I.; Bullock, S.; Grisotto, M.; Booth, T.; Taub, P.; et al. Differential rates of replacement of human dermal dendritic cells and macrophages during hematopoietic stem cell transplantation. J. Exp. Med. 2009, 206, 371–385. [Google Scholar] [CrossRef] [PubMed]
- Angel, C.E.; George, E.; Ostrovsky, L.L.; Dunbar, P.R. Comprehensive analysis of MHC-II expression in healthy human skin. Immunol. Cell Biol. 2007, 85, 363–369. [Google Scholar] [CrossRef] [PubMed]
- McLellan, A.D.; Heiser, A.; Sorg, R.V.; Fearnley, D.B.; Hart, D.N. Dermal dendritic cells associated with T lymphocytes in normal human skin display an activated phenotype. J. Investig. Dermatol. 1998, 111, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Zaba, L.C.; Fuentes-Duculan, J.; Steinman, R.M.; Krueger, J.G.; Lowes, M.A. Normal human dermis contains distinct populations of CD11c+BDCA-1+ dendritic cells and CD163+FXIIIA+ macrophages. J. Clin. Investig. 2007, 117, 2517–2525. [Google Scholar] [CrossRef] [PubMed]
- Angel, C.E.; George, E.; Brooks, A.E.; Ostrovsky, L.L.; Brown, T.L.; Dunbar, P.R. Cutting edge: CD1a+ antigen-presenting cells in human dermis respond rapidly to CCR7 ligands. J. Immunol. 2006, 176, 5730–5734. [Google Scholar] [CrossRef] [PubMed]
- Van Rhijn, I.; Ly, D.; Moody, D.B. CD1a, CD1b, and CD1c in immunity against mycobacteria. Adv. Exp. Med. Biol. 2013, 783, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, M.; McAuliffe, B.; Subramani, J.; Basu, S.; Shapiro, L.H. CD13 regulates dendritic cell cross-presentation and T cell responses by inhibiting receptor-mediated antigen uptake. J. Immunol. 2012, 188, 5489–5499. [Google Scholar] [CrossRef] [PubMed]
- Villaseñor-Cardoso, M.I.; Frausto-Del-Río, D.A.; Ortega, E. Aminopeptidase N (CD13) is Involved in Phagocytic Processes in Human Dendritic Cells and Macrophages. Biomed. Res. Int. 2013, 12, 562984. [Google Scholar] [CrossRef] [PubMed]
- Gardai, S.J.; McPhillips, K.A.; Frasch, S.C.; Janssen, W.J.; Starefeldt, A.; Murphy-Ullrich, J.E.; Bratton, D.L.; Oldenborg, P.A.; Michalak, M.; Henson, P.M. Cell-surface calreticulin initiates clearance of viable or apoptotic cells through trans-activation of LRP on the phagocyte. Cell 2005, 123, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Van der Aar, A.M.; Sylva-Steenland, R.M.; Bos, J.D.; Kapsenberg, M.L.; de Jong, E.C.; Teunissen, M.B. Loss of TLR2, TLR4, and TLR5 on Langerhans cells abolishes bacterial recognition. J. Immunol. 2007, 178, 1986–1990. [Google Scholar] [CrossRef] [PubMed]
- Mittag, D.; Proietto, A.I.; Loudovaris, T.; Mannering, S.I.; Vremec, D.; Shortman, K.; Harrison, L.C. Human dendritic cell subsets from spleen and blood are similar in phenotype and function but modified by donor health status. J. Immunol. 2011, 186, 6207–6217. [Google Scholar] [CrossRef] [PubMed]
- Haniffa, M.; Shin, A.; Bigley, V.; McGovern, N.; Teo, P.; See, P.; Wasan, P.S.; Wang, X.N.; Malinarich, F.; Malleret, B.; et al. Human tissues contain CD141hi cross-presenting dendritic cells with functional homology to mouse CD103+ nonlymphoid dendritic cells. Immunity 2012, 37, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Morelli, A.E.; Rubin, J.P.; Erdos, G.; Tkacheva, O.A.; Mathers, A.R.; Zahorchak, A.F.; Thomson, A.W.; Falo, L.D., Jr.; Larregina, A.T. CD4+T cell responses elicited by different subsets of human skin migratory dendritic cells. J. Immunol. 2005, 175, 7905–7915. [Google Scholar] [CrossRef] [PubMed]
- Schlitzer, A.; McGovern, N.; Teo, P.; Zelante, T.; Atarashi, K.; Low, D.; Ho, A.W.; See, P.; Shin, A.; Wasan, P.S.; et al. IRF4 transcription factor-dependent CD11b+ dendritic cells in human and mouse control mucosal IL-17 cytokine responses. Immunity 2013, 38, 970–983. [Google Scholar] [CrossRef] [PubMed]
- Segura, E.; Valladeau-Guilemond, J.; Donnadieu, M.H.; Sastre-Garau, X.; Soumelis, V.; Amigorena, S. Characterization of resident and migratory dendritic cells in human lymph nodes. J. Exp. Med. 2012, 209, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Sancho, D.; Joffre, O.P.; Keller, A.M.; Rogers, N.C.; Martinez, D.; Hernanz-Falcon, P.; Rosewell, I.; Reis e Sousa, C. Identification of a dendritic cell receptor that couples sensing of necrosis to immunity. Nature 2009, 458, 899–903. [Google Scholar] [CrossRef] [PubMed]
- Bachem, A.; Guttler, S.; Hartung, E.; Ebstein, F.; Schaefer, M.; Tannert, A.; Salama, A.; Movassaghi, K.; Opitz, C.; Mages, H.W.; et al. Superior antigen cross-presentation and XCR1 expression define human CD11c+CD141+ cells as homologues of mouse CD8+ dendritic cells. J. Exp. Med. 2010, 207, 1273–1281. [Google Scholar] [CrossRef] [PubMed]
- Galibert, L.; Diemer, G.S.; Liu, Z.; Johnson, R.S.; Smith, J.L.; Walzer, T.; Comeau, M.R.; Rauch, C.T.; Wolfson, M.F.; Sorensen, R.A.; et al. Nectin-like protein 2 defines a subset of T-cell zone dendritic cells and is a ligand for class-I-restricted T-cell-associated molecule. J. Biol. Chem. 2005, 280, 21955–21964. [Google Scholar] [CrossRef] [PubMed]
- Crozat, K.; Guiton, R.; Contreras, V.; Feuillet, V.; Dutertre, C.A.; Ventre, E.; Vu Manh, T.P.; Baranek, T.; Storset, A.K.; Marvel, J.; et al. The XC chemokine receptor 1 is a conserved selective marker of mammalian cells homologous to mouse CD8alpha+ dendritic cells. J. Exp. Med. 2010, 207, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Jongbloed, S.L.; Kassianos, A.J.; McDonald, K.J.; Clark, G.J.; Ju, X.; Angel, C.E.; Chen, C.J.; Dunbar, P.R.; Wadley, R.B.; Jeet, V.; et al. Human CD141+ (BDCA-3)+ dendritic cells (DCs) represent a unique myeloid DC subset that cross-presents necrotic cell antigens. J. Exp. Med. 2010, 207, 1247–1260. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J. IPC: Professional type 1 interferon-producing cells and plasmacytoid dendritic cell precursors. Annu. Rev. Immunol. 2005, 23, 275–306. [Google Scholar] [CrossRef] [PubMed]
- Lutz, M.B. IL-3 in dendritic cell development and function: A comparison with GM-CSF and IL-4. Immunobiology 2004, 209, 79–87. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, K.P.; Munster, D.J.; Clark, G.J.; Dzionek, A.; Schmitz, J.; Hart, D.N. Characterization of human blood dendritic cell subsets. Blood 2002, 100, 4512–4520. [Google Scholar] [CrossRef] [PubMed]
- Omatsu, Y.; Iyoda, T.; Kimura, Y.; Maki, A.; Ishimori, M.; Toyama-Sorimachi, N.; Inaba, K. Development of Murine Plasmacytoid Dendritic Cells Defined by Increased Expression of an Inhibitory NK Receptor, Ly49Q. J. Immunol. 2005, 174, 6657–6662. [Google Scholar] [CrossRef] [PubMed]
- Reizis, B.; Bunin, A.; Ghosh, H.S.; Lewis, K.L.; Sisirak, V. Plasmacytoid dendritic cells: Recent progress and open questions. Annu. Rev. Immunol. 2011, 29, 163–183. [Google Scholar] [CrossRef] [PubMed]
- Dzionek, A.; Fuchs, A.; Schmidt, P.; Cremer, S.; Zysk, M.; Miltenyi, S.; Buck, D.W.; Schmitz, J. BDCA-2, BDCA-3, and BDCA-4: Three markers for distinct subsets of dendritic cells in human peripheral blood. J. Immunol. 2000, 165, 6037–6046. [Google Scholar] [CrossRef] [PubMed]
- Mathan, T.S.; Figdor, C.G.; Buschow, S.I. Human plasmacytoid dendritic cells: From molecules to intercellular communication network. Front. Immunol. 2013, 4, 372. [Google Scholar] [CrossRef] [PubMed]
- Dzionek, A.; Sohma, Y.; Nagafune, J.; Cella, M.; Colonna, M.; Facchetti, F.; Günther, G.; Johnston, I.; Lanzavecchia, A.; Nagasaka, T.; et al. BDCA-2, a novel plasmacytoid dendritic cell-specific type II C-type lectin, mediates antigen capture and is a potent inhibitor of interferon alpha/beta induction. J. Exp. Med. 2001, 194, 1823–1834. [Google Scholar] [CrossRef] [PubMed]
- Swiecki, M.; Colonna, M. The multifaceted biology of plasmacytoid dendritic cells. Nat. Rev. Immunol. 2015, 15, 471–485. [Google Scholar] [CrossRef] [PubMed]
- Lippens, C.; Duraes, F.V.; Dubrot, J.; Brighouse, D.; Lacroix, M.; Irla, M.; Aubry-Lachainaye, J.P.; Reith, W.; Judith, N.; Mandl, J.N.; et al. IDO-orchestrated crosstalk between pDCs and Tregs inhibits autoimmunity. J. Autoimmun. 2016, 75, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Moseman, E.A.; Liang, X.; Dawson, A.J.; Panoskaltsis-Mortari, A.; Krieg, A.M.; Liu, Y.J.; Blazar, B.R.; Chen, W. Human plasmacytoid dendritic cells activated by CpG oligodeoxynucleotides induce the generation of CD4+CD25+ regulatory T cells. J. Immunol. 2004, 173, 4433–4442. [Google Scholar] [CrossRef] [PubMed]
- Chong, S.Z.; Wong, K.L.; Lin, G.; Yang, C.M.; Wong, S.C.; Angeli, V.; Macary, P.A.; Kemeny, D.M. Human CD8 T cells drive Th1 responses through the differentiation of TNF/iNOS-producing dendritic cells. Eur. J. Immunol. 2011, 41, 1639–1651. [Google Scholar] [CrossRef] [PubMed]
- Wilsmann-Theis, D.; Koch, S.; Mindnich, C.; Bonness, S.; Schnautz, S.; von Bubnoff, D.; Bieber, T. Generation and functional analysis of human TNF-a/iNOS-producing dendritic cells (Tip-DC). Allergy 2013, 68, 890–898. [Google Scholar] [CrossRef] [PubMed]
- Serbina, N.V.; Pamer, E.G. Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nat. Immunol. 2006, 7, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Gabrilovich, D.I.; Nagaraj, S. Myeloid-derived suppressor cells as regulators of the immune system. Nat. Rev. Immunol. 2009, 9, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Nestle, F.O.; Zheng, X.G.; Thompson, C.B.; Turka, L.A.; Nickoloff, B.J. Characterization of dermal dendritic cells obtained from normal human skin reveals phenotypic and functionally distinctive subsets. J. Immunol. 1993, 151, 6535–6545. [Google Scholar] [PubMed]
- Klechevsky, E.; Liu, M.; Morita, R.; Banchereau, R.; Thompson-Snipes, L.; Palucka, A.K.; Ueno, H.; Banchereau, J. Understanding human myeloid dendritic cell subsets for the rational design of novel vaccines. Hum. Immunol. 2009, 70, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Klechevsky, E.; Morita, R.; Liu, M.; Cao, Y.; Coquery, S.; Thompson-Snipes, L.; Briere, F.; Chaussabel, D.; Zurawski, G.; Palucka, A.K.; et al. Functional specializations of human epidermal langerhans cells and CD14+ dermal dendritic cells. Immunity 2008, 29, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Angel, C.E.; Chen, C.J.; Horlacher, O.C.; Winkler, S.; John, T.; Browning, J.; MacGregor, D.; Cebon, J.; Dunbar, P.R. Distinctive localization of antigen-presenting cells in human lymph nodes. Blood 2009, 113, 1257–1267. [Google Scholar] [CrossRef] [PubMed]
- Matthews, K.; Chung, N.P.Y.; Klasse, P.J.; Moore, J.P.; Sanders, R.W. Potent induction of antibody-secreting B-cells by human dermal-derived CD14+ dendritic cells triggered by dual Toll-like receptor ligation. J. Immunol. 1950, 189, 5729–5744. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.-C.; Ali, N.; Karagiannis, P.; Di Meglio, P.; Skowera, A.; Napolitano, L.; Barinaga, G.; Grys, K.; Sharif-Paghaleh, E.; Karagiannis, S.N.; et al. Resident CD141 (BDCA3)+ dendritic cells in human skin produce IL-10 and induce regulatory T cells that suppress skin inflammation. J. Exp. Med. 2012, 209, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Chen, Z.; Yang, Y.; Jiang, Z.; Gu, Y.; Liu, Y.; Lin, C.; Pan, Z.; Yu, Y.; Jiang, M.; et al. Human Cd14+ CTLA-4+ regulatory dendritic cells suppress T-cell response by cytotoxic T-lymphocyte antigen-4-dependent IL-10 and indoleamine-2,3-dioxygenase production in hepatocellular carcinoma. Hepatology 2014, 59, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Watchmaker, P.B.; Lahl, K.; Lee, M.; Baumjohann, D.; Morton, J.; Kim, S.J.; Zeng, R.; Dent, A.; Ansel, K.M.; Diamond, B.; et al. Comparative transcriptional and functional profiling defines conserved programs of intestinal DC differentiation in humans and mice. Nat. Immunol. 2013, 15, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Guttman-Yassky, E.; Lowes, M.A.; Fuentes-Duculan, J.; Whynot, J.; Novitskaya, I.; Cardinale, I.; Haider, A.; Khatcherian, A.; Carucci, J.A.; Bergman, R.; et al. Major differences in inflammatory dendritic cells and their products distinguish atopic dermatitis from psoriasis. J. Allergy Clin. Immunol. 2007, 119, 1210–1217. [Google Scholar] [CrossRef] [PubMed]
- Segura, E.; Touzot, M.; Bohineust, A.; Cappuccio, A.; Chiocchia, G.; Hosmalin, A.; Dalod, M.; Soumelis, V.; Amigorena, S. Human inflammatory dendritic cells induce th17 cell differentiation. Immunity 2013, 38, 336–348. [Google Scholar] [CrossRef] [PubMed]
- Banchereau, J.; Klechevsky, E.; Schmitt, N.; Morita, R.; Palucka, K.; Ueno, H. Harnessing human dendritic cell subsets to design novel vaccines. Ann. N. Y. Acad. Sci. 2009, 1174, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Auffray, C.; Sieweke, M.H.; Geiss-Mann, F. Blood monocytes: Development, heterogeneity, and relationship with dendritic cells. Annu. Rev. Immunol. 2009, 27, 669–692. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.K.; Fujita, S.S. Dendritic cells: Nature and classification. Allergol. Int. (Jpn. Soc. Allergol.) 2007, 56, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Mantegazza, A.R.; Savina, A.; Vermeulen, M.; Pérez, L.; Geffner, J.; Hermine, O.; Rosenzweig, S.D.; Faure, F.; Amigorena, S. NADPH oxidase controls phagosomal pH and antigen cross-presentation in human dendritic cells. Blood 2008, 112, 4712–4722. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lu, L.; Yang, Z.; Palaniyandi, S.; Zeng, R.; Gao, L.Y.; Mosser, D.M.; Roopenian, D.C.; Zhu, X. The neonatal FcR-mediated presentation of immune-complexed antigen is associated with endosomal and phagosomal pH and antigen stability in macrophages and dendritic cells. J. Immunol. 2011, 186, 4674–4686. [Google Scholar] [CrossRef] [PubMed]
- Romani, N.N.; Koide, S.S.; Crowley, M.M.; Witmer-Pack, M.M.; Livingstone, A.M.A.; Fathman, C.G.C.; Inaba, K.; Steinman, R.M. Presentation of exogenous protein antigens by dendritic cells to T cell clones. Intact protein is presented best by immature, epidermal Langerhans cells. J. Exp. Med. 1989, 169, 1169–1178. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Palermo, B.; Lenig, D.; Miettinen, M.; Matikainen, S.; Julkunen, I.; Forster, R.; Burgstahler, R.; Lipp, M.; Lanzavecchia, A. Distinct patters and kinetics of chemokine production regulate dendritic cell function. Eur. J. Immunol. 1999, 29, 1617. [Google Scholar] [CrossRef]
- Yanagihara, S.; Komura, E.; Nagafune, J.; Watarai, H.; Yamaguchi, Y. EBI1/CCR7 Is a New Member of Dendritic Cell Chemokine Receptor That Is Up-Regulated upon Maturation. Immunology 1998, 161, 3096. [Google Scholar]
- Dieu, M.-C.; Vanbervliet, B.; Vicari, A.; Bridon, J.-M.; Oldham, E.; Ait-Yahia, S.; Briére, F.; Zlotnik, A.; Lebecque, S.; Caux, C. Selective Recruitment of Immature and Mature Dendritic Cells by Distinct Chemokines Expressed in Different Anatomic Sites. J. Exp. Med. 1998, 188, 373. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, A.L.; Harman, A.; Kim, M.; Nasr, N.; Lai, J. Immunobiology of dendritic cells and the influence of HIV infection. Adv. Exp. Med. Biol. 2013, 762, 1–44. [Google Scholar] [CrossRef] [PubMed]
- Nagae, M.; Ikeda, A.; Hanashima, S.; Kojima, T.; Matsumoto, N.; Yamamoto, K.; Yamaguchi, Y. Crystal structure of human dendritic cell inhibitory receptor C-type lectin domain reveals the binding mode with N-glycan. FEBS Lett. 2016, 590, 1280–1288. [Google Scholar] [CrossRef] [PubMed]
- Bates, E.E.; Fournier, N.; Garcia, E.; Valladeau, J.; Durand, I.; Pin, J.J.; Zurawski, S.M.; Patel, S.; Abrams, J.S.; Lebecque, S.; et al. APCs express DCIR, a novel C-type lectin surface receptor containing an immunoreceptor tyrosine-based inhibitory motif. J. Immunol. 1999, 163, 1973. [Google Scholar] [PubMed]
- Ariizumi, K.; Shen, G.-L.; Shikano, S.; Ritter, I.I.I.R.; Zukas, P.; Edelbaum, D.; Morita, A.; Takashima, A. Cloning of a Second Dendritic Cell-associated C-type Lectin (Dectin-2) and Its Alternatively Spliced Isoforms. J. Biol. Chem. 2000, 275, 11957. [Google Scholar] [CrossRef] [PubMed]
- Colonna, M.; Samaridis, J.; Angman, L. Molecular characterization of two novel C-type lectin-like receptors, one of which is selectively expressed in human dendritic cells. Eur. J. Immunol. 2000, 30, 697–704. [Google Scholar] [CrossRef]
- Geijtenbeek, T.B.; Vliet, S.J.V.; Engering, A.; ‘t Hart, B.A.; Kooyk, Y.V. Self- and nonself recognition by c-type lectins on dendritic cells. Ann. Rev. Immunol. 2004, 22, 33–54. [Google Scholar] [CrossRef] [PubMed]
- Nouri-Shirazi, M.; Banchereau, J.; Fay, J.; Palucka, K. Dendritic cell based tumor vaccines. Immunol. Lett. 2000, 74, 5–10. [Google Scholar] [CrossRef]
- Regnault, A.; Lankar, D.; Lacabanne, V.; Rodriguez, A.; Théry, C.; Rescigno, M.; Saito, T.; Verbeek, S.; Bonnerot, C.; Ricciardi-Castagnoli, P.; et al. Fcgamma receptor-mediated induction of dendritic cell maturation and major histocompatibility complex class I-restricted antigen presentation after immune complex internalization. J. Exp. Med. 1999, 189, 371. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.F.; Lanzavecchia, A.A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor alpha. J. Exp. Med. 1994, 179, 1109–1118. [Google Scholar] [CrossRef] [PubMed]
- Villadangos, J.A.; Schnorrer, P. Intrinsic and cooperative antigen-presenting functions of dendritic-cell subsets in vivo. Nat. Rev. Immunol. 2007, 7, 543–555. [Google Scholar] [CrossRef] [PubMed]
- Wilson, N.S.; Villadangos, J.A. Regulation of antigen presentation and cross-presentation in the dendritic cell network: Facts, hypothesis, and immunological implications. Adv Immunol. 2005, 86, 241–305. [Google Scholar] [CrossRef] [PubMed]
- Veeraswamy, R.K.; Cella, M.; Colonna, M.; Unanue, E.R. Dendritic Cells Process and Present Antigens Across a Range of Maturation States. J. Immunol. 2003, 170, 5367–5372. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Williams, D.B. Assembly of MHC class I molecules within the endoplasmic reticulum. Immunol Res. 2006, 35, 151–162. [Google Scholar] [CrossRef]
- Serwold, T.; González, F.; Kim, J.; Jacob, R.; Shastri, N. ERAAP customizes peptides for MHC class I molecules in the endoplasmic reticulum. Nature 2002, 419, 480–483. [Google Scholar] [CrossRef] [PubMed]
- Van Montfoort, N.; van der Aa, E.; Woltman, A.M. Understanding MHC class I presentation of viral antigens by human dendritic cells as a basis for rational design of therapeutic vaccines. Front. Immunol. 2014, 5, 182. [Google Scholar] [CrossRef] [PubMed]
- Kovacsovics-Banowski, M.; Rock, K.L. A phagosome-to-cytosol pathway for exogenous antigens presented on MHC class I molecules. Science 1995, 267, 243–246. [Google Scholar] [CrossRef]
- Firat, E.; Saveanu, L.; Aichele, P.; Staeheli, P.; Huai, J.; Gaedicke, S.; Nil, A.; Besin, G.; Kanzler, B.; van Endert, P.; et al. The role of endoplasmic reticulum-associated aminopeptidase 1 in immunity to infection and in cross-presentation. J. Immunol. 2007, 178, 2241–2248. [Google Scholar] [CrossRef] [PubMed]
- Saveanu, L.; Caroll, O.; Weimershaus, M.; Guermonprez, P.; Firat, E.; Lindo, V.; Greer, F.; Davoust, J.; Kratzer, R.; Keller, S.R.; et al. IRAP identifies an endosomal compartment required for MHC class I cross-presentation. Science 2009, 325, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Joffre, O.P.; Segura, E.; Savina, A.; Amigorena, S. Cross-presentation by dendritic cells. Nat. Rev. Immunol. 2012, 12, 557–569. [Google Scholar] [CrossRef] [PubMed]
- Burgdorf, S.; Schölz, C.; Kautz, A.; Tampé, R.; Kuts, C. Spatial and mechanistic separation of cross-presentation and endogenous antigen presentation. Nat. Immunol. 2008, 9, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Rock, K.L.; Shen, L. Cross-presentation: Underlying mechanisms and role in immune surveillance. Immunol. Rev. 2005, 207, 166–183. [Google Scholar] [CrossRef] [PubMed]
- Den Haan, J.M.; Bevan, M.J. Constitutive versus Activation-dependent Cross-Presentation of Immune Complexes by CD8+ and CD8− Dendritic Cells In Vivo. J. Exp. Med. 2002, 196, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Sigal, L.J.; Boes, M.; Rock, K.L. Important role of cathepsin S in generating peptides for TAP-independent MHC class I crosspresentation in vivo. Immunity 2004, 21, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Cella, M.; Sallusto, F.; Lanzavecchia, A. Origin, maturation and antigen presenting function of dendritic cells. Curr. Opin. Immunol. 1997, 9, 10. [Google Scholar] [CrossRef]
- Watts, C. The endosome-lysosome pathway and information generation in the immune system. Biochim. Biophys. Acta 2012, 1824, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Ten Broeke, T.; Wubbolts, R.; Stoorvogel, W. MHC class II antigen presentation by dendritic cells regulated through endosomal sorting. Cold Spring Harb. Perspect. Biol. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.A.; Evavold, B.D.; Jensen, P.E. Enhanced dissociation of HLA-DR-bound peptides in the presence of HLA-DM. Science 1996, 274, 618–620. [Google Scholar] [CrossRef] [PubMed]
- Rocha, N.; Kuijl, C.; van der Kant, R.; Janssen, L.; Houben, D.; Janssen, H.; Zwart, W.; Neefjes, J. Cholesterol sensor ORP1L contacts the ER protein VAP to control Rab7-RILP-p150 Glued and late endosome positioning. J. Cell Biol. 2009, 185, 1209–1225. [Google Scholar] [CrossRef] [PubMed]
- Piper, R.C.; Katzmann, D.J. Biogenesis and function of multivesicular bodies. Annu. Rev. Cell Dev. Biol. 2007, 23, 519–547. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Zitvogel, L.; Amigorena, S. Exosomes: Composition, biogenesis and function. Nat. Rev. Immunol. 2002, 2, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Matteoli, G.; Mazzini, E.; Iliev, I.D.; Mileti, E.; Fallarino, F.; Puccetti, P.; Chieppa, M.; Rescigno, M. Gut CD103+ dendritic cells express indoleamine 2,3-dioxygenase which influences T regulatory/T effector cell balance and oral tolerance induction. Gut 2010, 59, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Manicassamy, S.; Reizis, B.; Ravindran, R.; Nakaya, H.; Salazar-Gonzalez, R.M.; Wang, Y.C.; Pulendran, B. Activation of β-catenin in dendritic cells regulates immunity versus tolerance in the intestine. Science 2010, 329, 849–853. [Google Scholar] [CrossRef] [PubMed]
- Mantis, N.J.; Forbes, S.J. Secretory IgA: Arresting microbial pathogens at epithelial borders. Immunol. Investig. 2010, 39, 383–406. [Google Scholar] [CrossRef] [PubMed]
- Johansson-Lindbom, B.; Svensson, M.; Wurbel, M.A.; Malissen, B.; Márquez, G.; Agace, W. Selective generation of gut tropic T cells in gut-associated lymphoid tissue (GALT): Requirement for GALT dendritic cells and adjuvant. J. Exp. Med. 2003, 198, 963–969. [Google Scholar] [CrossRef] [PubMed]
- Mora, J.R.; Bono, M.R.; Manjunath, N.; Weninger, W.; Cavanagh, L.L.; Rosemblatt, M.; Von Andrian, U.H. Selective imprinting of gut-homing T cells by Peyer’s patch dendritic cells. Nature 2003, 424, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Stagg, A.J.; Kamm, M.A.; Knight, S.C. Intestinal dendritic cells increase T cell expression of alpha4beta7 integrin. Eur. J. Immunol. 2002, 32, 1445–1454. [Google Scholar] [CrossRef]
- Frank, D.N.; Amand, A.L.; Feldman, R.A.; Boedeker, E.C.; Harpaz, N.; Pace, N.R. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc. Natl. Acad. Sci. USA 2007, 104, 13780–13785. [Google Scholar] [CrossRef] [PubMed]
- Morgan, X.C.; Tickle, T.L.; Sokol, H.; Gevers, D.; Devaney, K.L.; Ward, D.V.; Reyes, J.A.; Shah, S.A.; LeLeiko, N.; Snapper, S.B.; et al. Dysfunction of the intestinal microbiome in inflammatory bowel disease and treatment. Genome Biol. 2012, 13, R79. [Google Scholar] [CrossRef] [PubMed]
- Gevers, D.; Kugathasan, S.; Denson, L.A.; Vázquez-Baeza, Y.; Van Treuren, W.; Ren, B.; Schwager, E.; Knights, D.; Song, S.J.; Yassour, M.; et al. The treatment-naive microbiome in new-onset Crohn’s disease. Cell Host Microbe 2014, 15, 382–392. [Google Scholar] [CrossRef] [PubMed]
- Arihiro, S.; Ohtani, H.; Suzuki, M.; Murata, M.; Ejima, C.; Oki, M.; Kinouchi, Y.; Fukushima, K.; Sasaki, I.; Makamura, S.; et al. Differential expression of mucosal addressin cell adhesion molecule-1 (MAdCAM-1) in ulcerative colitis and Crohn’s disease. Pathol. Int. 2002, 52, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Briskin, M.; Winsor-Hines, D.; Shyjan, A.; Cochran, N.; Bloom, S.; Wilson, J.; McEvoy, L.M.; Butcher, E.C.; Kassam, N.; Mackay, C.R.; et al. Human mucosal addressin cell adhesion molecule-1 is preferentially expressed in intestinal tract and associated lymphoid tissue. Am. J. Pathol. 1997, 151, 97–110. [Google Scholar] [PubMed]
- Hart, A.L.; Kamm, M.A.; Knight, S.C.; Stagg, A.J. Prospective evaluation of intestinal homing memory T cells in ulcerative colitis. Inflamm. Bowel Dis. 2004, 10, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Hart, A.L.; Kamm, M.A.; Knight, S.C.; Stagg, A.J. Quantitative and functional characteristics of intestinal-homing memory T cells: Analysis of Crohn’s disease patients and healthy controls. Clin. Exp. Immunol. 2004, 135, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Perez-Lopez, A.; Behnsen, J.; Nuccio, S.P.; Raffatellu, M. Mucosal immunity to pathogenic intestinal bacteria. Nat. Rev. Immunol. 2016, 16, 135–148. [Google Scholar] [CrossRef] [PubMed]
- Bol, K.F.; Schreibelt, G.; Gerritsen, W.R.; de Vries, I.J.; Figdor, C.G. Dendritic Cell-Based Immunotherapy: State of the Art and Beyond. Clin. Cancer Res. 2016, 22, 1897–1906. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solano-Gálvez, S.G.; Tovar-Torres, S.M.; Tron-Gómez, M.S.; Weiser-Smeke, A.E.; Álvarez-Hernández, D.A.; Franyuti-Kelly, G.A.; Tapia-Moreno, M.; Ibarra, A.; Gutiérrez-Kobeh, L.; Vázquez-López, R. Human Dendritic Cells: Ontogeny and Their Subsets in Health and Disease. Med. Sci. 2018, 6, 88. https://doi.org/10.3390/medsci6040088
Solano-Gálvez SG, Tovar-Torres SM, Tron-Gómez MS, Weiser-Smeke AE, Álvarez-Hernández DA, Franyuti-Kelly GA, Tapia-Moreno M, Ibarra A, Gutiérrez-Kobeh L, Vázquez-López R. Human Dendritic Cells: Ontogeny and Their Subsets in Health and Disease. Medical Sciences. 2018; 6(4):88. https://doi.org/10.3390/medsci6040088
Chicago/Turabian StyleSolano-Gálvez, Sandra Georgina, Sonia Margarita Tovar-Torres, María Sofía Tron-Gómez, Ariane Estrella Weiser-Smeke, Diego Abelardo Álvarez-Hernández, Giorgio Alberto Franyuti-Kelly, Mijail Tapia-Moreno, Antonio Ibarra, Laila Gutiérrez-Kobeh, and Rosalino Vázquez-López. 2018. "Human Dendritic Cells: Ontogeny and Their Subsets in Health and Disease" Medical Sciences 6, no. 4: 88. https://doi.org/10.3390/medsci6040088
APA StyleSolano-Gálvez, S. G., Tovar-Torres, S. M., Tron-Gómez, M. S., Weiser-Smeke, A. E., Álvarez-Hernández, D. A., Franyuti-Kelly, G. A., Tapia-Moreno, M., Ibarra, A., Gutiérrez-Kobeh, L., & Vázquez-López, R. (2018). Human Dendritic Cells: Ontogeny and Their Subsets in Health and Disease. Medical Sciences, 6(4), 88. https://doi.org/10.3390/medsci6040088





