Clostridium Bacteria and Autism Spectrum Conditions: A Systematic Review and Hypothetical Contribution of Environmental Glyphosate Levels
Abstract
1. Introduction
2. Materials and Methods
2.1. Systematic Review Question
2.2. Search Strategy
2.3. Selection Criteria
- Only studies published in English.
- Clinical studies in humans.
- Neurological symptoms and/or diagnosed autism must be present.
- Studies that allow access to their specific data.
- In case of duplicate publications, only the most recent data will be preferred.
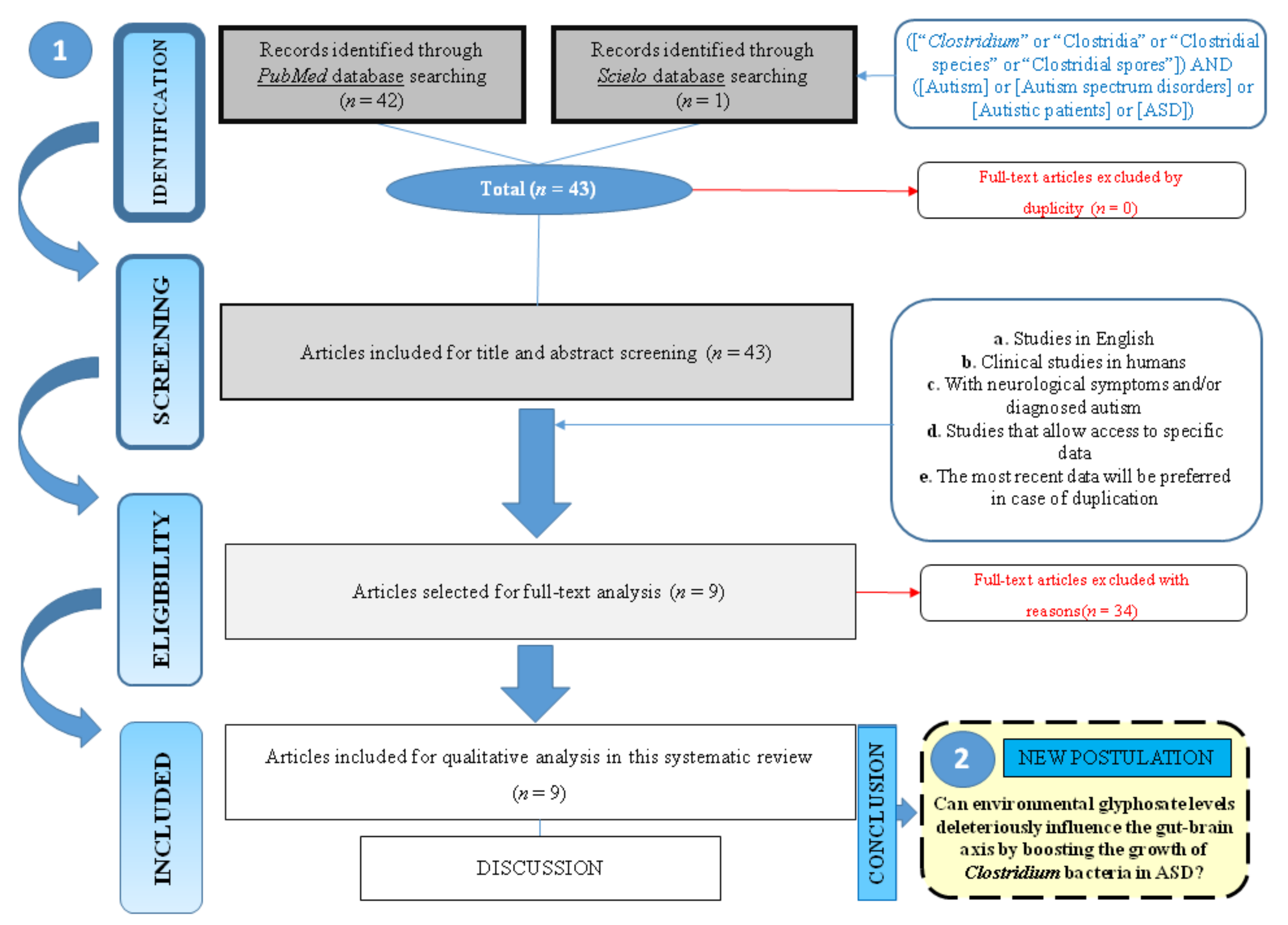
2.4. Selection of Studies
3. Results
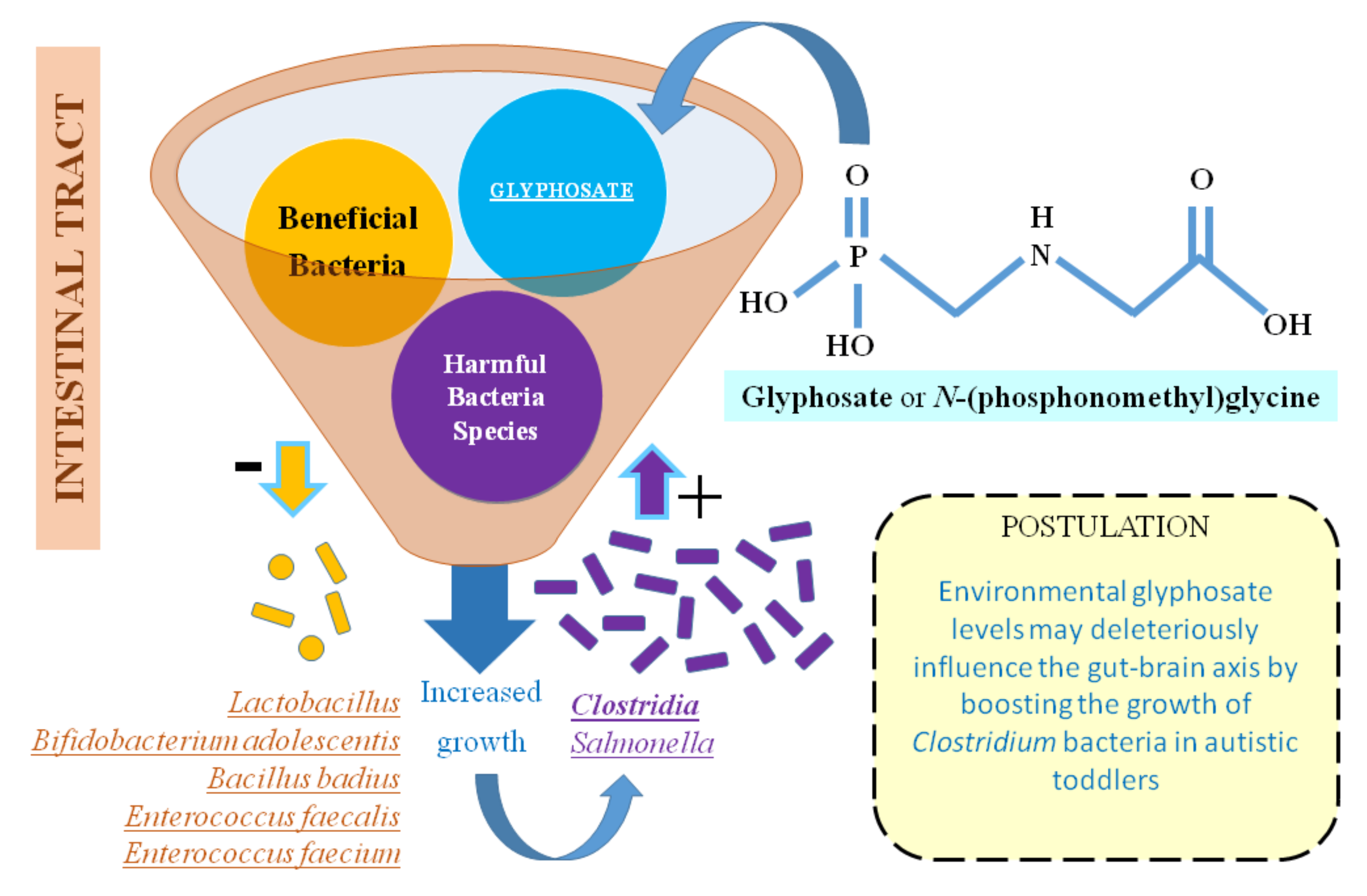
4. Discussion: An Interrelation between Glyphosate, Clostridium Bacteria, and Autism?
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Volkmar, F.; Klin, A.; Cohen, D. Diagnosis and classification of autism and related conditions. In Handbook of Autism and Pervasive Developmental Disorders; Cohen, D., Volkmar, F., Eds.; Wiley: New York, NY, USA, 1997; pp. 5–40. [Google Scholar]
- El-Fishawy, P.; State, M.W. The genetics of autism: Key issues, recent findings, and clinical implications. Psychiatr. Clin. N. Am. 2010, 33, 83–105. [Google Scholar] [CrossRef] [PubMed]
- Zeidán-Chuliá, F.; Gursoy, U.K.; Könönen, E.; Gottfried, C. A dental look at the autistic patient through orofacial pain. Acta Odontol. Scand. 2011, 69, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Argou-Cardozo, I.; Cano Martín, J.A.; Zeidán-Chuliá, F. Dental Amalgam Fillings and the Use of Technological Devices as an Environmental Factor: Updating the Cumulative Mercury Exposure-based Hypothesis of Autism. Eur. J. Dent. 2017, 11, 569–570. [Google Scholar] [PubMed]
- Fombonne, E. The prevalence of autism. JAMA 2003, 289, 87–89. [Google Scholar] [CrossRef] [PubMed]
- Zeidán-Chuliá, F.; Rybarczyk-Filho, J.L.; Salmina, A.B.; de Oliveira, B.H.; Noda, M.; Moreira, J.C. Exploring the multifactorial nature of autism through computational systems biology: Calcium and the Rho GTPase RAC1 under the spotlight. Neuromolecular Med. 2013, 15, 364–383. [Google Scholar] [CrossRef] [PubMed]
- Zeidán-Chuliá, F.; Salmina, A.B.; Noda, M.; Verkhratsky, A. Rho GTPase RAC1 at the Molecular Interface Between Genetic and Environmental Factors of Autism Spectrum Disorders. Neuromolecular Med. 2015, 17, 333–334. [Google Scholar] [CrossRef] [PubMed]
- Wiley, N.C.; Dinan, T.G.; Ross, R.P.; Stanton, C.; Clarke, G.; Cryan, J.F. The microbiota-gut-brain axis as a key regulator of neural function and the stress response: Implications for human and animal health. J. Anim. Sci. 2017, 95, 3225–3246. [Google Scholar] [PubMed]
- Zeidán-Chuliá, F.; Fonseca Moreira, J.C. Clostridium Bacteria and its Impact in Autism Research: Thinking “Outside the Box” of Neuroscience. Commun. Disord. Deaf Stud. Hearing Aids 2013, 1, 101. [Google Scholar] [CrossRef]
- Busch, C.; Aktories, K. Microbial toxins and the glycosylation of rho family GTPases. Curr. Opin. Struct Biol. 2000, 10, 528–535. [Google Scholar] [CrossRef]
- Shaw, W. Elevated Urinary Glyphosate and Clostridia Metabolites with Altered Dopamine Metabolism in Triplets with Autistic Spectrum Disorder or Suspected Seizure Disorder: A Case Study. Integr. Med. (Encinitas) 2017, 16, 50–57. [Google Scholar] [PubMed]
- Finegold, S.M.; Molitoris, D.; Song, Y.; Liu, C.; Vaisanen, M.L.; Bolte, E.; McTeague, M.; Sandler, R.; Wexler, H.; Marlowe, E.M.; et al. Gastrointestinal microflora studies in late-onset autism. Clin. Infect. Dis. 2002, 35, S6–S16. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Liu, C.; Finegold, S.M. Real-time PCR quantitation of clostridia in feces of autistic children. Appl. Environ. Microbiol. 2004, 70, 6459–6465. [Google Scholar] [CrossRef] [PubMed]
- Parracho, H.M.; Bingham, M.O.; Gibson, G.R.; McCartney, A.L. Differences between the gut microflora of children with autistic spectrum disorders and that of healthy children. J. Med. Microbiol. 2005, 54, 987–991. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.R.; Land, M.L.; Kastashov, A.; Saslowsky, T.M.; Lyerly, D.M.; Boone, J.H.; Rufo, P.A. Fecal lactoferrin is a sensitive and specific maker of disease activity in children and young adults with inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 2007, 44, 414e22. [Google Scholar] [CrossRef] [PubMed]
- Martirosian, G.; Ekiel, A.; Aptekorz, M.; Wiechuła, B.; Kazek, B.; Jankowska-Steifer, E.; Jóźwiak, J.; Moskalewski, S. Fecal lactoferrin and Clostridium spp. in stools of autistic children. Anaerobe 2011, 17, 43–45. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, M.; Piccolo, M.; Vannini, L.; Siragusa, S.; De Giacomo, A.; Serrazzanetti, D.I.; Cristofori, F.; Guerzoni, M.E.; Gobbetti, M.; Francavilla, R. Fecal microbiota and metabolome of children with autism and pervasive developmental disorder not otherwise specified. PLoS ONE 2013, 8, e76993. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.; Liu, D.; Wang, Y.; Zeng, T.; Peng, Y. Urinary 3-(3-Hydroxyphenyl)-3-hydroxypropionic Acid, 3-Hydroxyphenylacetic Acid, and 3-Hydroxyhippuric Acid Are Elevated in Children with Autism Spectrum Disorders. Biomed. Res. Int. 2016. [Google Scholar] [CrossRef] [PubMed]
- Iovene, M.R.; Bombace, F.; Maresca, R.; Sapone, A.; Iardino, P.; Picardi, A.; Marotta, R.; Schiraldi, C.; Siniscalco, D.; Serra, N.; et al. Intestinal Dysbiosis and Yeast Isolation in Stool of Subjects with Autism Spectrum Disorders. Mycopathologia 2017, 182, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Kushak, R.I.; Winter, H.S.; Buie, T.M.; Cox, S.B.; Phillips, C.D.; Ward, N.L. Analysis of the Duodenal Microbiome in Autistic Individuals: Association with Carbohydrate Digestion. J. Pediatr. Gastroenterol. Nutr. 2017, 64, e110–e116. [Google Scholar] [CrossRef] [PubMed]
- Finegold, S.M.; Summanen, P.H.; Downes, J.; Corbett, K.; Komoriya, T. Detection of Clostridium perfringens toxin genes in the gut microbiota of autistic children. Anaerobe 2017, 45, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Fang, L.; Lee, M.H. Dysbiosis of gut microbiota in promoting the development of colorectal cancer. Gastroenterol. Rep. (Oxf.) 2018, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Moran-Ramos, S.; López Contreras, B.E.; Canizales-Quinteros, S. Gut Microbiota in Obesity and Metabolic Abnormalities: A Matter of Composition or Functionality? Arch. Med. Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Szablewski, L. Human Gut Microbiota in Health and Alzheimer's Disease. J. Alzheimers Dis. 2018, 62, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Sudo, N.; Chida, Y.; Aiba, Y.; Oyama, N.; Yu, X.N.; Kubo, C.; Koga, Y. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J. Physiol. 2004, 558, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Prince, A.L.; Chu, D.M.; Seferovic, M.D.; Antony, K.M.; Aagaard, K.M. The perinatal microbiome and pregnancy: moving beyond the vaginal microbiome. Cold Spring Harb. Perspect. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A.; Knight, R.; Mazmanian, S.K.; Cryan, J.F.; Tillisch, K. Gut microbes and the brain: paradigm shift in neuroscience. J. Neurosci. 2014, 34, 15490–15496. [Google Scholar] [CrossRef] [PubMed]
- Felice, V.D.; O’Mahony, S.M. The microbiome and disorders of the central nervous system. Pharmacol. Biochem. Behav. 2017, 160, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Tomova, A.; Husarova, V.; Lakatosova, S.; Bakos, J.; Vlkova, B.; Babinska, K.; Ostatnikova, D. Gastrointestinal microbiota in children with autism in Slovakia. Physiol. Behav. 2015, 138, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Popoff, M.R. Multifaceted interactions of bacterial toxins with the gastrointestinal mucosa. Future Microbiol. 2011, 6, 763–797. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, A.; Minden, A.; Yuste, R. Regulation of dendritic spine morphology by the rho family of small GTPases: antagonistic roles of Rac and Rho. Cereb. Cortex. 2000, 10, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Reichova, A.; Zatkova, M.; Bacova, Z.; Bakos, J. Abnormalities in interactions of Rho GTPases with Scaffolding Proteins Contribute to Neurodevelopmental Disorders. J. Neurosci. Res. 2017, 96, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Bakos, J.; Bacova, Z.; Grant, S.G.; Castejon, A.M.; Ostatnikova, D. Are Molecules Involved in Neuritogenesis and Axon Guidance Related to Autism Pathogenesis? Neuromolecular Med. 2015, 17, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Holzman, D.C. Pesticides and autism spectrum disorders: new findings from the CHARGE study. Environ. Health Perspect. 2014, 122, A280. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Marks, A.R.; Bradman, A.; Harley, K.; Barr, D.B.; Johnson, C.; Morga, N.; Jewell, N.P. Organophosphate Pesticide Exposure and Neurodevelopment in Young Mexican-American Children. Environ. Health Perspect. 2007, 115, 792–798. [Google Scholar] [CrossRef] [PubMed]
- Roberts, E.M.; English, P.B.; Grether, J.K.; Windham, G.C.; Somberg, L.; Wolff, C. Maternal Residence Near Agricultural Applications and Autism Spectrum Disorders among Children in the California Central Valle. Environ. Health Perspect. 2007, 115, 1482–1489. [Google Scholar] [PubMed]
- Shelton, J.F.; Hertz-Picciotto, I.; Pessah, I.N. Tipping the Balance of Autism Risk: Potential Mechanisms Linking Pesticides and Autism. Environ. Health Perspect. 2012, 120, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Shelton, J.F.; Geraghty, E.M.; Tancredi, D.J.; Delwiche, L.D.; Schmidt, R.J.; Ritz, B.; Hansen, R.L.; Hertz-Picciotto, I. Neurodevelopmental Disorders and Prenatal Residential Proximity to Agricultural Pesticides: The CHARGE Study. Environ. Health Perspect. 2014, 122, 1103–1109. [Google Scholar] [CrossRef] [PubMed]
- Duke, S.O.; Powles, S.B. Glyphosate: A once-in-a-century herbicide. Pest. Manag. Sci. 2008, 64, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Woodburn, A.T. Glyphosate: Production, pricing and use worldwide. Pest. Manag. Sci. 2000, 56, 309–312. [Google Scholar] [CrossRef]
- Benbrook, C.M. Trends in glyphosate herbicide use in the United States and globally. Environ. Sci. Eur. 2016, 28, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Shehata, A.A.; Schrödl, W.; Aldin, A.A.; Hafez, H.M.; Krüger, M. The effect of glyphosate on potential pathogens and beneficial members of poultry microbiota in vitro. Curr. Microbiol. 2013, 66, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Krüger, M.; Shehata, A.A.; Schrödl, W.; Rodloff, A. Glyphosate suppresses the antagonistic effect of Enterococcus spp. on Clostridium botulinum. Anaerobe 2013, 20, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, W.; Coenen, M.; Schrödl, W.; Shehata, A.A.; Krüger, M. The influence of glyphosate on the microbiota and production of botulinum neurotoxin during ruminal fermentation. Curr. Microbiol. 2015, 70, 374–382. [Google Scholar] [CrossRef] [PubMed]
- You, M.J.; Shin, G.W.; Lee, C.S. Clostridium tertium bacteremia in a patient with glyphosate ingestion. Am. J. Case Rep. 2015, 16, 4–7. [Google Scholar] [PubMed]
- Sandler, R.H.; Finegold, S.M.; Bolte, E.R.; Buchanan, C.P.; Maxwell, A.P.; Väisänen, M.L.; Nelson, M.N.; Wexler, H.M. Short-term benefit from oral vancomycin treatment of regressive-onset autism. J. Child. Neurol. 2000, 15, 429–435. [Google Scholar] [CrossRef] [PubMed]
| TITLE | AUTHOR/YEAR | PMID | MATERIAL | N° PATIENTS/CONTROLS | CONCLUSIONS |
|---|---|---|---|---|---|
| Gastrointestinal microflora studies in late-onset autism | Finegold et al., 2002 | 12173102 | Human (Stools) | 13 ASD/8 controls | Children with autism had nine species of Clostridium not found in controls, with significant alterations in the upper and lower intestinal flora of children with late-onset autism |
| Real-time PCR quantitation of clostridia in feces of autistic children | Song et al., 2004 | 15528506 | Human (Stools) | 15 ASD/8 controls | Analysis of the real-time PCR data showed cell count differences between autistic and control children for Clostridium bolteae |
| Differences between the gut microflora of children with autistic spectrum disorders and that of healthy children | Parracho et al., 2005 | 16157555 | Human (Stools) | 58 ASD/22 controls (2 groups) | The fecal flora of autism spectrum disorder (ASD) patients contained a higher incidence of the Clostridium histolyticum group (Clostridium clusters I and II) of bacteria than that of healthy children |
| Fecal lactoferrin and Clostridium spp. in stools of autistic children | Martirosian et al., 2011 | 21167951 | Human (Stools) | 41 ASD/10 controls | Elevated level of fecal lactoferrin was demonstrated in 24.4% stools, all from boys (31.25%). No toxins were detected. Clostridium spp. was isolated with similar frequency from all samples. Clostridium perfringens were isolated significantly often from the autistic stools |
| Fecal microbiota and metabolome of children with autism and pervasive developmental disorder not otherwise specified | De Angelis et al., 2013 | 24130822 | Human (Stools) | 20 ASD/10 controls | The highest microbial diversity was found in ASD children. Based on 16S-rRNA and culture-dependent data, Faecalibacterium and Ruminococcus were present at the highest level in fecal samples of pervasive developmental disorder not otherwise specified (PDD-NOS) and healthy children. Caloramator, Sarcina and Clostridium genera were the highest in ASD children |
| Urinary 3-(3-Hydroxyphenyl)-3-hydroxypropionic Acid, 3-Hydroxyphenylacetic Acid, and 3-Hydroxyhippuric Acid Are Elevated in Children with Autism Spectrum Disorders | Xiong et al., 2016 | 27123458 | Human (Urine) | 62 ASD/62 controls | Measurement of these three compounds (aromatic metabolites in autism patients are presumably derived from overgrown Clostridium species in gut) are strong predictors of ASDs and support the potential clinical utility for identifying a subgroup of ASD subjects |
| Intestinal Dysbiosis and Yeast Isolation in Stool of Subjects with Autism Spectrum Disorders | Iovene et al., 2017 | 27655151 | Human (Stools) | 47 ASD/33 controls | Significant linear correlation was found between disease severity and calprotectin and Clostridium spp. presence in the stool of subjects with ASD |
| Analysis of the Duodenal Microbiome in Autistic Individuals: Association with Carbohydrate Digestion | Kushak et al., 2017 | 27811623 | Human (Intestinal biopsies) | 21 ASD/19 controls | A positive correlation was found between the abundance of Clostridium species and disaccharidase activity in autistic individuals |
| Detection of Clostridium perfringens toxin genes in the gut microbiota of autistic children | Finegold et al., 2017 | 28215985 | Human (Stools) | 33 ASD/13 controls | The author´s results indicate that autistic subjects with gastrointestinal disease harbor statistically significantly higher counts of C. perfringens and β2-toxin gene-producing C. perfringens in their gut when compared to control children. In addition, the incidence of β2-toxin gene-producing C. perfringens is also significantly higher in these autistic subjects |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Argou-Cardozo, I.; Zeidán-Chuliá, F. Clostridium Bacteria and Autism Spectrum Conditions: A Systematic Review and Hypothetical Contribution of Environmental Glyphosate Levels. Med. Sci. 2018, 6, 29. https://doi.org/10.3390/medsci6020029
Argou-Cardozo I, Zeidán-Chuliá F. Clostridium Bacteria and Autism Spectrum Conditions: A Systematic Review and Hypothetical Contribution of Environmental Glyphosate Levels. Medical Sciences. 2018; 6(2):29. https://doi.org/10.3390/medsci6020029
Chicago/Turabian StyleArgou-Cardozo, Isadora, and Fares Zeidán-Chuliá. 2018. "Clostridium Bacteria and Autism Spectrum Conditions: A Systematic Review and Hypothetical Contribution of Environmental Glyphosate Levels" Medical Sciences 6, no. 2: 29. https://doi.org/10.3390/medsci6020029
APA StyleArgou-Cardozo, I., & Zeidán-Chuliá, F. (2018). Clostridium Bacteria and Autism Spectrum Conditions: A Systematic Review and Hypothetical Contribution of Environmental Glyphosate Levels. Medical Sciences, 6(2), 29. https://doi.org/10.3390/medsci6020029





