The Association between Sleep and Theory of Mind in School Aged Children with ADHD
Abstract
1. Introduction
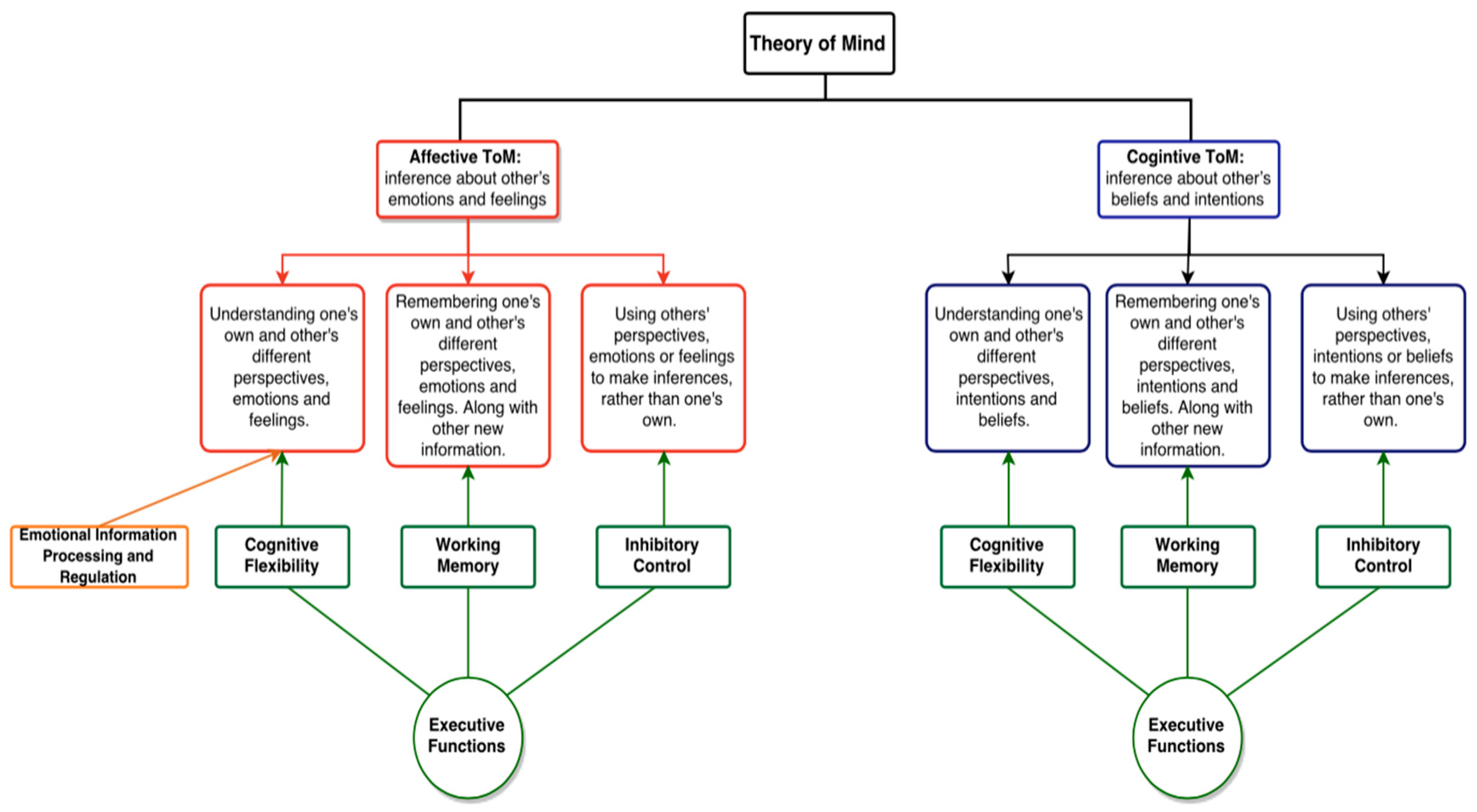
2. Theory of Mind
Executive Functioning and Theory of Mind
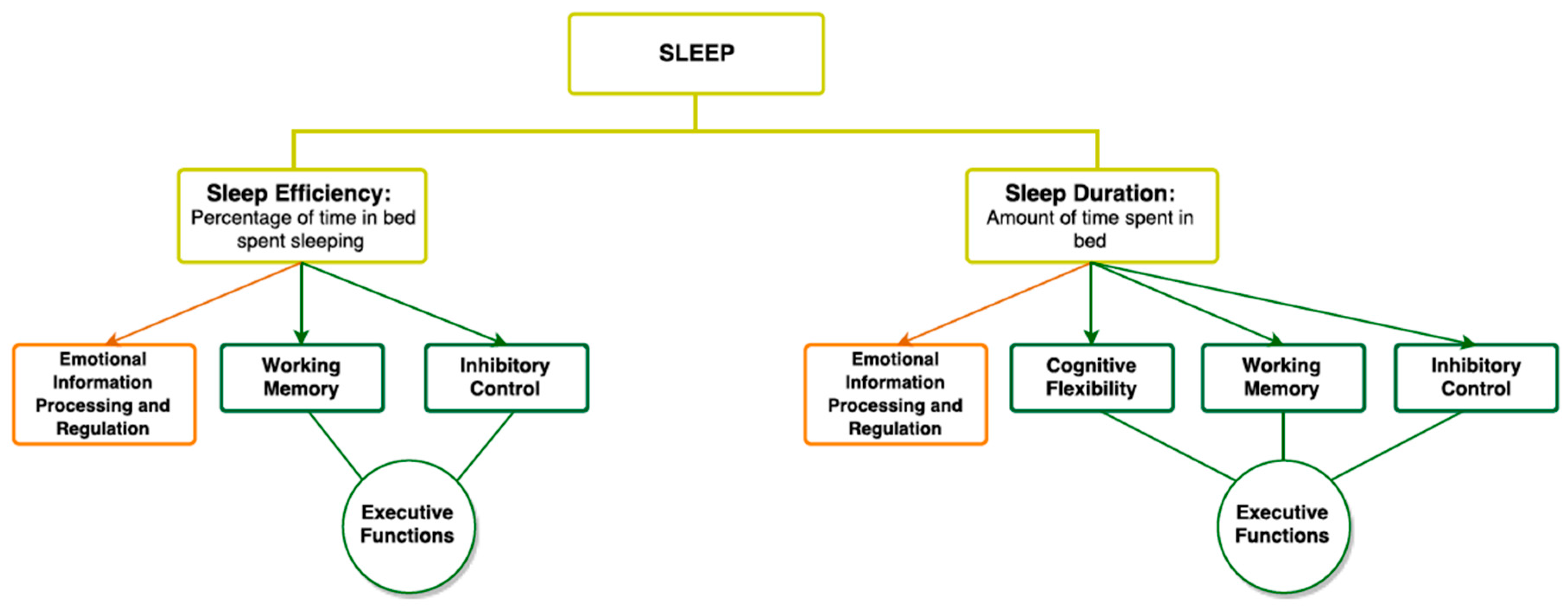
3. Sleep
3.1. Sleep and Executive Functioning
3.2. Sleep and Emotional Information Processing
4. Sleep and ToM: Are They Associated?
5. Attention Deficit/Hyperactivity Disorder
5.1. ADHD, EF, and Social Functioning
5.2. ADHD and ToM
5.3. ADHD and Sleep
6. Conclusions
Author Contributions
Conflicts of Interest
References
- Premack, D.; Woodruff, G. Does the chimpanzee have a theory of mind? Behav. Brain Sci. 1978, 1, 515–526. [Google Scholar] [CrossRef]
- Wellman, H.M. The child’s Theory of Mind; The MIT Press: Cambridge, MA, USA, 1990. [Google Scholar]
- Peterson, C.; Slaughter, V.; Moore, C.; Wellman, H.M. Peer social skills and theory of mind in children with autism, deafness, or typical development. Dev. Psychol. 2016, 52, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.C.; Nixon, C.L.; Wilson, A.; Capage, L. Social interaction skills and theory of mind in young children. Dev. Psychol. 1999, 35, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Imuta, K.; Henry, J.D.; Slaughter, V.; Selcuk, B.; Ruffman, T. Theory of mind and prosocial behavior in childhood: A meta-analytic review. Dev. Psychol. 2016, 52, 1192–1205. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, R.; Watling, D.; Caputi, M. Peer relations and the understanding of faux pas: Longitudinal evidence for bidirectional associations. Child Dev. 2011, 82, 1887–1905. [Google Scholar] [CrossRef] [PubMed]
- Slaughter, V.; Imuta, K.; Peterson, C.C.; Henry, J.D. Meta-analysis of theory of mind and peer popularity in the preschool and early school years. Child Dev. 2015, 86, 1159–1174. [Google Scholar] [CrossRef] [PubMed]
- Bora, E.; Berk, M. Theory of mind in major depressive disorder: A meta-analysis. J. Affect. Disord. 2016, 191, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.; Harkness, K.L.; Sabbagh, M.A.; Jacobson, J.A. Mental state decoding abilities in clinical depression. J. Affect. Disord. 2005, 86, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Hezel, D.M.; McNally, R.J. Theory of mind impairments in social anxiety disorder. Behav. Ther. 2014, 45, 530–540. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S. Theory of mind and autism: A review. Int. Rev. Res. Ment. Retard. 2000, 23, 169–184. [Google Scholar] [CrossRef]
- Corkum, P.; Tannock, R.; Moldofsky, H.; Hogg-Johnson, S.; Humphries, T. Actigraphy and parental ratings of sleep in children with attention-deficit/hyperactivity disorder. Sleep 2001, 24, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Owens, J.A. The ADHD and sleep conundrum: A review. J. Dev. Behav. Pediatr. 2005, 26, 312–322. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, (DSM-5®); American Psychiatric Pub: Washington, DC, USA, 2013. [Google Scholar]
- Visser, S.N.; Danielson, M.L.; Bitsko, R.H.; Holbrook, J.R.; Kogan, M.D.; Ghandour, R.M.; Blumberg, S.J. Trends in the parent-report of health care provider-diagnosed and medicated attention-deficit/hyperactivity disorder: United States, 2003–2011. J. Am. Acad. Child Adolesc. Psychiatry 2014, 53, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Hoza, B.; Mrug, S.; Gerdes, A.C.; Hinshaw, S.P.; Bukowski, W.M.; Gold, J.A.; Arnold, L.E. What aspects of peer relationships are impaired in children with attention-deficit/hyperactivity disorder? J. Consult. Clin. Psychol. 2005, 73, 411. [Google Scholar] [CrossRef] [PubMed]
- Mikami, A.Y. The importance of friendship for youth with attention-deficit/hyperactivity disorder. Clin. Child Fam. Psychol. Rev. 2010, 13, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Braaten, E.B.; Rosén, L.A. Self-regulation of affect in attention deficit-hyperactivity disorder (ADHD) and non-ADHD boys: Differences in empathic responding. J. Consult. Clin. Psychol. 2000, 68, 313. [Google Scholar] [CrossRef] [PubMed]
- Ronk, M.J.; Hund, A.M.; Landau, S. Assessment of social competence of boys with attention-deficit/hyperactivity disorder: Problematic peer entry, host responses, and evaluations. J. Abnorm. Child Psychol. 2011, 39, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Greene, R.W.; Biederman, J.; Faraone, S.V.; Sienna, M.; Garcia-Jetton, J. Adolescent outcome of boys with attention-deficit/hyperactivity disorder and social disability: Results from a 4-year longitudinal follow-up study. J. Consult. Clin. Psychol. 1997, 65, 758. [Google Scholar] [CrossRef] [PubMed]
- Klein, R.G.; Mannuzza, S. Long-term outcome of hyperactive children: A review. J. Am. Acad. Child Adolesc. Psychiatry 1991, 30, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Derefinko, K.J.; Bailey, U.L.; Milich, R.; Lorch, E.P.; Riley, E. The effects of stimulant medication on the online story narrations of children with ADHD. School Ment. Health 2009, 1, 171–182. [Google Scholar] [CrossRef]
- King, S.; Waschbusch, D.A.; Pelham, W.E., Jr.; Frankland, B.W.; Andrade, B.F.; Jacques, S.; Corkum, P.V. Social information processing in elementary-school aged children with ADHD: Medication effects and comparisons with typical children. J. Abnorm. Child Psychol. 2009, 37, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Sodian, B.; Schuwerk, T.; Kristen, S. Implicit and spontaneous theory of mind reasoning in autism spectrum disorders. In Autism Spectrum Disorder-Recent Advances; InTech: Rijeka, Croatia, 2015. [Google Scholar] [CrossRef]
- Schuwerk, T.; Vuori, M.; Sodian, B. Implicit and explicit theory of mind reasoning in autism spectrum disorders: the impact of experience. Autism 2015, 19, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Kovács, Á.M.; Téglás, E.; Endress, A.D. The social sense: Susceptibility to others’ beliefs in human infants and adults. Science 2010, 330, 1830–1834. [Google Scholar] [CrossRef] [PubMed]
- Wimmer, H.; Perner, J. Beliefs about beliefs: Representation and constraining function of wrong beliefs in young children's understanding of deception. Cognition 1983, 13, 103–128. [Google Scholar] [CrossRef]
- Perner, J. Understanding the Representational Mind; The MIT Press: Cambridge, MA, US, 1991; p. 348. [Google Scholar]
- Wellman, H.M.; Cross, D.; Watson, J. Meta-analysis of theory-of-mind development: The truth about false belief. Child Dev. 2001, 72, 655–684. [Google Scholar] [CrossRef] [PubMed]
- Frith, C.D.; Frith, U. Interacting minds—A biological basis. Science (New York, N.Y.) 1999, 286, 1692–1695. [Google Scholar] [CrossRef]
- Hughes, C.; Devine, R.T.; Ensor, R.; Masuo, K.; Ai, M.; Lecce, S. Lost in translation? Comparing british, japanese, and italian children’s theory-of-mind performance. Child Dev. Res. 2014, 1–10. [Google Scholar] [CrossRef]
- Sabbagh, M.A.; Xu, F.; Carlson, S.M.; Moses, L.J.; Lee, K. The development of executive functioning and theory of mind. A comparison of Chinese and U.S. Preschoolers. Psychol. Sci. 2006, 17, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.A. Children’s understanding of second-order mental states. Psychol. Bull. 2009, 135, 749–773. [Google Scholar] [CrossRef] [PubMed]
- Perner, J.; Wimmer, H. “John thinks that mary thinks that…” attribution of second-order beliefs by 5- to 10-year-old children. J. Exp. Child Psychol. 1985, 39, 437–471. [Google Scholar] [CrossRef]
- Happe, F. Theory of mind and the self. Ann. N. Y. Acad. Sci. 2003, 1001, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Segrin, C. Social skills deficits associated with depression. Clin. Psychol. Rev. 2000, 20, 379–403. [Google Scholar] [CrossRef]
- Spitzberg, B.H.; Cupach, W.R. Issues in interpersonal competence research. In Handbook of Interpersonal Competence Research; Springer: New York, NY, USA, 1989; pp. 52–75. [Google Scholar]
- Caputi, M.; Lecce, S.; Pagnin, A.; Banerjee, R. Longitudinal effects of theory of mind on later peer relations: The role of prosocial behavior. Dev. Psychol. 2012, 48, 257–270. [Google Scholar] [CrossRef] [PubMed]
- Baurain, C.; Nader-Grosbois, N. Compétences Sociales et émotionnelles: Enfant Typique et Déficient Intellectuel; Presses Académiques Francophones: Saarbrücken, Germany, 2013. [Google Scholar]
- Riggs, N.R.; Greenberg, M.T.; Kusche, C.A.; Pentz, M.A. The mediational role of neurocognition in the behavioral outcomes of a social-emotional prevention program in elementary school students: Effects of the paths curriculum. Prev. Sci. Off. J. Soc. Prev. Res. 2006, 7, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Harvey, R.J.; Fletcher, J.; French, D.J. Social reasoning: A source of influence on aggression. Clin. Psychol. Rev. 2001, 21, 447–469. [Google Scholar] [CrossRef]
- Renouf, A.; Brendgen, M.; Parent, S.; Vitaro, F.; David Zelazo, P.; Boivin, M.; Dionne, G.; Tremblay, R.E.; Pérusse, D.; Séguin, J.R. Relations between theory of mind and indirect and physical aggression in kindergarten: Evidence of the moderating role of prosocial behaviors. Soc. Dev. 2010, 19, 535–555. [Google Scholar] [CrossRef]
- Kalbe, E.; Schlegel, M.; Sack, A.T.; Nowak, D.A.; Dafotakis, M.; Bangard, C.; Brand, M.; Shamay-Tsoory, S.; Onur, O.A.; Kessler, J. Dissociating cognitive from affective theory of mind: A tms study. Cortex J. Devot. Study Nerv. Syst. Behav. 2010, 46, 769–780. [Google Scholar] [CrossRef] [PubMed]
- Shamay-Tsoory, S.G.; Shur, S.; Barcai-Goodman, L.; Medlovich, S.; Harari, H.; Levkovitz, Y. Dissociation of cognitive from affective components of theory of mind in schizophrenia. Psychiatry Res. 2007, 149, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Abu-Akel, A.; Shamay-Tsoory, S. Neuroanatomical and neurochemical bases of theory of mind. Neuropsychologia 2011, 49, 2971–2984. [Google Scholar] [CrossRef] [PubMed]
- Bottiroli, S.; Cavallini, E.; Ceccato, I.; Vecchi, T.; Lecce, S. Theory of mind in aging: Comparing cognitive and affective components in the faux pas test. Arch. Gerontol. Geriatr. 2016, 62, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Roca, M.; Parr, A.; Thompson, R.; Woolgar, A.; Torralva, T.; Antoun, N.; Manes, F.; Duncan, J. Executive function and fluid intelligence after frontal lobe lesions. Brain J. Neurol. 2010, 133, 234–247. [Google Scholar] [CrossRef] [PubMed]
- Sebastian, C.L.; Fontaine, N.M.; Bird, G.; Blakemore, S.J.; Brito, S.A.; McCrory, E.J.; Viding, E. Neural processing associated with cognitive and affective theory of mind in adolescents and adults. Soc. Cognit. Affect. Neurosci. 2012, 7, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Shamay-Tsoory, S.G.; Aharon-Peretz, J. Dissociable prefrontal networks for cognitive and affective theory of mind: A lesion study. Neuropsychologia 2007, 45, 3054–3067. [Google Scholar] [CrossRef] [PubMed]
- Shamay-Tsoory, S.G.; Tibi-Elhanany, Y.; Aharon-Peretz, J. The ventromedial prefrontal cortex is involved in understanding affective but not cognitive theory of mind stories. Soc. Neurosci. 2006, 1, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Van Overwalle, F.; Baetens, K. Understanding others’ actions and goals by mirror and mentalizing systems: A meta-analysis. NeuroImage 2009, 48, 564–584. [Google Scholar] [CrossRef] [PubMed]
- Frith, C.D.; Frith, U. The neural basis of mentalizing. Neuron 2006, 50, 531–534. [Google Scholar] [CrossRef] [PubMed]
- Schurz, M.; Radua, J.; Aichhorn, M.; Richlan, F.; Perner, J. Fractionating theory of mind: A meta-analysis of functional brain imaging studies. Neurosci. Biobehav. Rev. 2014, 42, 9–34. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Jolliffe, T.; Mortimore, C.; Robertson, M. Another advanced test of theory of mind: Evidence from very high functioning adults with autism or asperger syndrome. J. Child Psychol. Psychiatry Allied Discip. 1997, 38, 813–822. [Google Scholar] [CrossRef]
- Baron-Cohen, S.; Wheelwright, S.; Hill, J.; Raste, Y.; Plumb, I. The “reading the mind in the eyes” test revised version: A study with normal adults, and adults with asperger syndrome or high-functioning autism. J. Child Psychol. Psychiatry Allied Discip. 2001, 42, 241–251. [Google Scholar] [CrossRef]
- Adolphs, R.; Sears, L.; Piven, J. Abnormal processing of social information from faces in autism. J. Cognit. Neurosci. 2001, 13, 232–240. [Google Scholar] [CrossRef]
- Yoo, S.S.; Gujar, N.; Hu, P.; Jolesz, F.A.; Walker, M.P. The human emotional brain without sleep—A prefrontal amygdala disconnect. Curr. Biol. 2007, 17, R877–R878. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.A. Emotion regulation: A theme in search of definition. Monogr.Soc. Res. Child Dev. 1994, 59, 25–52. [Google Scholar] [CrossRef] [PubMed]
- Stone, V.E.; Baron-Cohen, S.; Knight, R.T. Frontal lobe contributions to theory of mind. J. Cognit. Neurosci. 1998, 10, 640–656. [Google Scholar] [CrossRef]
- Carlson, S.M.; Zelazo, P.D.; Faja, S. Executive function. In The Oxford Handbook of Developmental Psychology; Zelazo, P.D., Ed.; Oxford University Press: New York, NY, USA, 2013; Volume 1, pp. 706–743. [Google Scholar]
- Diamond, A. Executive functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [PubMed]
- Friedman, N.P.; Miyake, A. The relations among inhibition and interference control functions: A latent-variable analysis. J. Exp. Psychol. Gen. 2004, 133, 101–135. [Google Scholar] [CrossRef] [PubMed]
- Miyake, A.; Friedman, N.P.; Emerson, M.J.; Witzki, A.H.; Howerter, A.; Wager, T.D. The unity and diversity of executive functions and their contributions to complex “frontal lobe” tasks: A latent variable analysis. Cognit. Psychol. 2000, 41, 49–100. [Google Scholar] [CrossRef] [PubMed]
- Devine, R.T.; Hughes, C. Relations between false belief understanding and executive function in early childhood: A meta-analysis. Child Dev. 2014, 85, 1777–1794. [Google Scholar] [CrossRef] [PubMed]
- Fuster, J.M. The Prefrontal Cortex, 4th ed.; Academic Press: Boston, MA, USA, 2008. [Google Scholar]
- Davidson, M.C.; Amso, D.; Anderson, L.C.; Diamond, A. Development of cognitive control and executive functions from 4 to 13 years: Evidence from manipulations of memory, inhibition, and task switching. Neuropsychologia 2006, 44, 2037–2078. [Google Scholar] [CrossRef] [PubMed]
- Apperly, I.A.; Warren, F.; Andrews, B.J.; Grant, J.; Todd, S. Developmental continuity in theory of mind: Speed and accuracy of belief-desire reasoning in children and adults. Child Dev. 2011, 82, 1691–1703. [Google Scholar] [CrossRef] [PubMed]
- Devine, R.T.; Hughes, C. Silent films and strange stories: Theory of mind, gender, and social experiences in middle childhood. Child Dev. 2013, 84, 989–1003. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.M.; Moses, L.J. Individual differences in inhibitory control and children's theory of mind. Child Dev. 2001, 72, 1032–1053. [Google Scholar] [CrossRef] [PubMed]
- Austin, G.; Groppe, K.; Elsner, B. The reciprocal relationship between executive function and theory of mind in middle childhood: A 1-year longitudinal perspective. Front. Psychol. 2014, 5, 655. [Google Scholar] [CrossRef] [PubMed]
- Bock, A.M.; Gallaway, K.C.; Hund, A.M. Specifying links between executive functioning and theory of mind during middle childhood: Cognitive flexibility predicts social understanding. J. Cognit. Dev. 2015, 16, 509–521. [Google Scholar] [CrossRef]
- Perner, J.; Lang, B. Theory of mind and executive function: Is there a developmental relationship? In Understanding Other Minds: Perspectives from Developmental Cognitive Neuroscience, 2nd ed.; Oxford University Press: New York, NY, USA, 2000; pp. 150–181. [Google Scholar]
- Borbely, A.A. A two process model of sleep regulation. Hum. Neurobiol. 1982, 1, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Hirshkowitz, M.; Whiton, K.; Albert, S.M.; Alessi, C.; Bruni, O.; DonCarlos, L.; Neubauer, D.N. National Sleep Foundation’s sleep time duration recommendations: Methodology and results summary. Sleep Health 2015, 1, 40–43. [Google Scholar] [CrossRef]
- Paruthi, S.; Brooks, L.J.; D’Ambrosio, C.; Hall, W.A.; Kotagal, S.; Lloyd, R.M.; Rosen, C.L. Recommended amount of sleep for pediatric populations: A consensus statement of the American Academy of Sleep Medicine. J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med. 2016, 12, 785. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.S.; Carson, V.; Chaput, J.P.; Connor Gorber, S.; Dinh, T.; Duggan, M.; Janssen, I. Canadian 24-Hour Movement Guidelines for Children and Youth: An Integration of Physical Activity, Sedentary Behaviour, and Sleep 1. Appl. Physiol. Nutr. Metab. 2016, 41, S311–S327. [Google Scholar] [CrossRef] [PubMed]
- Beebe, D.W.; Wells, C.T.; Jeffries, J.; Chini, B.; Kalra, M.; Amin, R. Neuropsychological effects of pediatric obstructive sleep apnea. J. Int. Neuropsychol. Soc. JINS 2004, 10, 962–975. [Google Scholar] [CrossRef] [PubMed]
- Bourke, R.; Anderson, V.; Yang, J.S.; Jackman, A.R.; Killedar, A.; Nixon, G.M.; Davey, M.J.; Walker, A.M.; Trinder, J.; Horne, R.S. Cognitive and academic functions are impaired in children with all severities of sleep-disordered breathing. Sleep Med. 2011, 12, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Dahl, R.E. The impact of inadequate sleep on children’s daytime cognitive function. Semin. Pediatr. Neurol. 1996, 3, 44–50. [Google Scholar] [CrossRef]
- Friedman, N.P.; Corley, R.P.; Hewitt, J.K.; Wright, K.P., Jr. Individual differences in childhood sleep problems predict later cognitive executive control. Sleep 2009, 32, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.M.; Caspi, A.; Moffitt, T.E.; Poulton, R. Sleep problems in childhood predict neuropsychological functioning in adolescence. Pediatrics 2009, 123, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Nelson, T.D.; Nelson, J.M.; Kidwell, K.M.; James, T.D.; Espy, K.A. Preschool sleep problems and differential associations with specific aspects of executive control in early elementary school. Dev. Neuropsychol. 2015, 40, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, J.P.; Soderstrom, M.; Karlsson, A.U.; Lekander, M.; Akerstedt, T.; Lindroth, N.E.; Axelsson, J. Less effective executive functioning after one night’s sleep deprivation. J. Sleep Res. 2005, 14, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Taveras, E.M.; Rifas-Shiman, S.L.; Bub, K.L.; Gillman, M.W.; Oken, E. Prospective study of insufficient sleep and neurobehavioral functioning among school-age children. Acad. Pediatr. 2017, 17, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Owens, J. Insufficient sleep in adolescents and young adults: An update on causes and consequences. Pediatrics 2014, 134, e921–e932. [Google Scholar] [CrossRef] [PubMed]
- Ohayon, M.; Wickwire, E.M.; Hirshkowitz, M.; Albert, S.M.; Avidan, A.; Daly, F.J.; Dauvilliers, Y.; Ferri, R.; Fung, C.; Gozal, D.; et al. National sleep foundation's sleep quality recommendations: First report. Sleep Health 2017, 3, 6–19. [Google Scholar] [CrossRef] [PubMed]
- Durmer, J.S.; Dinges, D.F. Neurocognitive consequences of sleep deprivation. Semin. Neurol. 2005, 25, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Edinger, J.D.; Bonnet, M.H.; Bootzin, R.R.; Doghramji, K.; Dorsey, C.M.; Espie, C.A.; Stepanski, E.J. Derivation of research diagnostic criteria for insomnia: Report of an American Academy of Sleep Medicine Work Group. Sleep 2004, 27, 1567–1596. [Google Scholar] [CrossRef] [PubMed]
- Morin, C.M.; Benca, R. Chronic insomnia. Lancet 2012, 379, 1129–1141. [Google Scholar] [CrossRef]
- Lu, F.M.; Liu, C.H.; Lu, S.L.; Tang, L.R.; Tie, C.L.; Zhang, J.; Yuan, Z. Disrupted topology of frontostriatal circuits is linked to the severity of insomnia. Front. Neurosci. 2017, 11, 214. [Google Scholar] [CrossRef] [PubMed]
- Riley, J.D.; Moore, S.; Cramer, S.C.; Lin, J.J. Caudate atrophy and impaired frontostriatal connections are linked to executive dysfunction in temporal lobe epilepsy. Epilepsy Behav. 2011, 21, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Chuah, Y.M.; Venkatraman, V.; Dinges, D.F.; Chee, M.W. The neural basis of interindividual variability in inhibitory efficiency after sleep deprivation. J. Neuroscience Off. J. Soc. Neurosci. 2006, 26, 7156–7162. [Google Scholar] [CrossRef] [PubMed]
- Drummond, S.P.; Paulus, M.P.; Tapert, S.F. Effects of two nights sleep deprivation and two nights recovery sleep on response inhibition. J. Sleep Res. 2006, 15, 261–265. [Google Scholar] [CrossRef] [PubMed]
- Gruber, R.; Wiebe, S.; Montecalvo, L.; Brunetti, B.; Amsel, R.; Carrier, J. Impact of sleep restriction on neurobehavioral functioning of children with attention deficit hyperactivity disorder. Sleep 2011, 34, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Maski, K.P.; Kothare, S.V. Sleep deprivation and neurobehavioral functioning in children. Int. J. Psychophysiol. Off. J. Int. Organ. Psychophysiol. 2013, 89, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Molfese, D.L.; Ivanenko, A.; Key, A.F.; Roman, A.; Molfese, V.J.; O’Brien, L.M.; Gozal, D.; Kota, S.; Hudac, C.M. A one-hour sleep restriction impacts brain processing in young children across tasks: Evidence from event-related potentials. Dev. Neuropsychol. 2013, 38, 317–336. [Google Scholar] [CrossRef] [PubMed]
- Sadeh, A.; Gruber, R.; Raviv, A. Sleep, neurobehavioral functioning, and behavior problems in school-age children. Child Dev. 2002, 73, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Halperin, J.M.; Sharma, V.; Greenblatt, E.; Schwartz, S.T. Assessment of the continuous performance test: Reliability and validity in a nonreferred sample. Psychol. Assess. A J. Consult. Clin. Psychol. 1991, 3, 603–608. [Google Scholar] [CrossRef]
- Sadeh, A.; Gruber, R.; Raviv, A. The effects of sleep restriction and extension on school-age children: What a difference an hour makes. Child Dev. 2003, 74, 444–455. [Google Scholar] [CrossRef] [PubMed]
- Gruber, R.; Michaelsen, S.; Bergmame, L.; Frenette, S.; Bruni, O.; Fontil, L.; Carrier, J. Short sleep duration is associated with teacher-reported inattention and cognitive problems in healthy school-aged children. Nat. Sci. Sleep 2012, 4, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Kopasz, M.; Loessl, B.; Hornyak, M.; Riemann, D.; Nissen, C.; Piosczyk, H.; Voderholzer, U. Sleep and memory in healthy children and adolescents—A critical review. Sleep Med. Rev. 2010, 14, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Steenari, M.R.; Vuontela, V.; Paavonen, E.J.; Carlson, S.; Fjallberg, M.; Aronen, E. Working memory and sleep in 6- to 13-year-old schoolchildren. J. Am. Acad. Child Adolesc. Psychiatry 2003, 42, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Vriend, J.L.; Davidson, F.D.; Corkum, P.V.; Rusak, B.; Chambers, C.T.; McLaughlin, E.N. Manipulating sleep duration alters emotional functioning and cognitive performance in children. J. Pediatr. Psychol. 2013, 38, 1058–1069. [Google Scholar] [CrossRef] [PubMed]
- Dewald-Kaufmann, J.F.; Oort, F.J.; Meijer, A.M. The effects of sleep extension on sleep and cognitive performance in adolescents with chronic sleep reduction: An experimental study. Sleep Med. 2013, 14, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.C.; Buchsbaum, M.; Bunney, W.E., Jr. Clinical neurochemical implications of sleep deprivation’s effects on the anterior cingulate of depressed responders. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2001, 25, S74–S78. [Google Scholar] [CrossRef]
- Perlman, S.B.; Pelphrey, K.A. Developing connections for affective regulation: Age-related changes in emotional brain connectivity. J. Exp. Child Psychol. 2011, 108, 607–620. [Google Scholar] [CrossRef] [PubMed]
- Berger, R.H.; Miller, A.L.; Seifer, R.; Cares, S.R.; LeBourgeois, M.K. Acute sleep restriction effects on emotion responses in 30- to 36-month-old children. J. Sleep Res. 2012, 21, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Deliens, G.; Gilson, M.; Peigneux, P. Sleep and the processing of emotions. Exp. Brain Res. 2014, 232, 1403–1414. [Google Scholar] [CrossRef] [PubMed]
- Soffer-Dudek, N.; Shahar, G. Daily stress interacts with trait dissociation to predict sleep-related experiences in young adults. J. Abnorm. Psychol. 2011, 120, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Killgore, W.D. Self-reported sleep correlates with prefrontal-amygdala functional connectivity and emotional functioning. Sleep 2013, 36, 1597–1608. [Google Scholar] [CrossRef] [PubMed]
- Weber, M.; Webb, C.A.; Deldonno, S.R.; Kipman, M.; Schwab, Z.J.; Weiner, M.R.; Killgore, W.D. Habitual ‘sleep credit’ is associated with greater grey matter volume of the medial prefrontal cortex, higher emotional intelligence and better mental health. J. Sleep Res. 2013, 22, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Doyle, A.E. Executive functions in attention-deficit/hyperactivity disorder. J. Clin. Psychiatry 2005, 67, 21–26. [Google Scholar]
- Barkley, R.A. Behavioral inhibition, sustained attention, and executive functions: Constructing a unifying theory of ADHD. Psychol. Bull. 1997, 121, 65. [Google Scholar] [CrossRef] [PubMed]
- Willcutt, E.G.; Doyle, A.E.; Nigg, J.T.; Faraone, S.V.; Pennington, B.F. Validity of the executive function theory of attention-deficit/hyperactivity disorder: A meta-analytic review. Biol. Psychiatry 2005, 57, 1336–1346. [Google Scholar] [CrossRef] [PubMed]
- Schoemaker, K.; Bunte, T.; Wiebe, S.A.; Espy, K.A.; Deković, M.; Matthys, W. Executive function deficits in preschool children with ADHD and DBD. J. Child Psychol. Psychiatry 2012, 53, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Durston, S.; Mulder, M.; Casey, B.J.; Ziermans, T.; van Engeland, H. Activation in ventral prefrontal cortex is sensitive to genetic vulnerability for attention-deficit hyperactivity disorder. Biol. Psychiatry 2006, 60, 1062–1070. [Google Scholar] [CrossRef] [PubMed]
- Shaw, P.; Rabin, C. New insights into attention-deficit/hyperactivity disorder using structural neuroimaging. Curr. Psychiatry Rep. 2009, 11, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.P.; Newcorn, J.H.; Fan, J.I.N.; Tang, C.Y.; Halperin, J.M. Brain activation gradients in ventrolateral prefrontal cortex related to persistence of ADHD in adolescent boys. J. Am. Acad. Child Adolesc. Psychiatry 2005, 44, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Loe, I.M.; Feldman, H.M. Academic and educational outcomes of children with ADHD. J. Pediatr. Psychol. 2007, 32, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Biederman, J.; Monuteaux, M.C.; Doyle, A.E.; Seidman, L.J.; Wilens, T.E.; Ferrero, F.; Faraone, S.V. Impact of executive function deficits and attention-deficit/hyperactivity disorder (ADHD) on academic outcomes in children. J. Consult. Clin. Psychol. 2004, 72, 757. [Google Scholar] [CrossRef] [PubMed]
- Savitz, J.B.; Jansen, P. The Stroop Color-Word Interference Test as an indicator of ADHD in poor readers. J. Genet. Psychol. 2003, 164, 319–333. [Google Scholar] [CrossRef] [PubMed]
- Langberg, J.M.; Epstein, J.N.; Urbanowicz, C.; Simon, J.; Graham, A. Efficacy of an organization skills intervention to improve the academic functioning of students with ADHD. School Psychol. Q. 2008, 23, 407–417. [Google Scholar] [CrossRef]
- Gureasko-Moore, S.; Dupaul, G.J.; White, G.P. The effects of self-management in general education classrooms on the organizational skills of adolescents with ADHD. Behav. Modif. 2008, 30, 159–183. [Google Scholar] [CrossRef] [PubMed]
- DuPaul, G.J.; McGoey, K.E.; Eckert, T.L.; VanBrakle, J. Preschool children with attention-deficit/hyperactivity disorder: impairments in behavioral, social, and school functioning. J. Am. Acad. Child Adolesc. Psychiatry 2001, 40, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Frankel, F.; Feinberg, D. Social problems associated with ADHD vs. ODD in children referred for friendship problems. Child Psychiatry Hum. Dev. 2002, 33, 125–146. [Google Scholar] [CrossRef] [PubMed]
- Grygiel, P.; Humenny, G.; Rębisz, S.; Bajcar, E.; Świtaj, P. Peer Rejection and Perceived Quality of Relations With Schoolmates Among Children With ADHD. J. Atten. Disord. 2014. [Google Scholar] [CrossRef] [PubMed]
- Erhardt, D.; Hinshaw, S.P. Initial sociometric impressions of attention-deficit hyperactivity disorder and comparison boys: Predictions from social behaviors and from nonbehavioral variables. J. Consult. Clin. Psychol. 1994, 62, 833. [Google Scholar] [CrossRef] [PubMed]
- Pelham, W.E.; Bender, M.E. Peer relationships in hyperactive children: Description and treatment. Adv. Learn. Behav. Disabil. 1982, 1, 365–436. [Google Scholar]
- Gresham, F.M.; MacMillan, D.L.; Bocian, K.M.; Ward, S.L.; Forness, S.R. Comorbidity of hyperactivity-impulsivity-inattention and conduct problems: Risk factors in social, affective, and academic domains. J. Abnorm. Child Psychol. 1998, 26, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Hoza, B.; Gerdes, A.C.; Hinshaw, S.P.; Arnold, L.E.; Pelham, W.E., Jr.; Molina, B.S.; Odbert, C. Self-perceptions of competence in children with ADHD and comparison children. J. Consult. Clin. Psychol. 2004, 72, 382. [Google Scholar] [CrossRef] [PubMed]
- Mary, A.; Slama, H.; Mousty, P.; Massat, I.; Capiau, T.; Drabs, V.; Peigneux, P. Executive and attentional contributions to Theory of Mind deficit in attention deficit/hyperactivity disorder (ADHD). Child Neuropsychol. 2016, 22, 345–365. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, T.C.; Panayiotou, G.; Spanoudis, G.; Natsopoulos, D. Evidence of poor planning in children with attention deficits. J. Abnorm. Child Psychol. 2005, 33, 611–623. [Google Scholar] [CrossRef] [PubMed]
- Wheeler Maedgen, J.; Carlson, C.L. Social functioning and emotional regulation in the attention deficit hyperactivity disorder subtypes. J. Clin. Child Psychol. 2000, 29, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.M.; Hermens, D.F.; Palmer, D.; Kohn, M.; Clarke, S.; Keage, H.; Gordon, E. Misinterpreting emotional expressions in attention-deficit/hyperactivity disorder: Evidence for a neural marker and stimulant effects. Biol. Psychiatry 2008, 63, 917–926. [Google Scholar] [CrossRef] [PubMed]
- Marsh, A.A.; Blair, R.J.R. Deficits in facial affect recognition among antisocial populations: A meta-analysis. Neurosci. Biobehav. Rev. 2008, 32, 454–465. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, K.L.; Galán, C.A.; Tottenham, N.; Lee, S.S. Impaired social decision-making mediates the association between ADHD and social problems. J. Abnorm. Child Psychol. 2016, 44, 1023–1032. [Google Scholar] [CrossRef] [PubMed]
- Andrade, B.F.; Waschbusch, D.A.; Doucet, A.; King, S.; MacKinnon, M.; McGrath, P.J.; Corkum, P. Social information processing of positive and negative hypothetical events in children with ADHD and conduct problems and controls. J. Atten. Disord. 2012, 16, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Chervin, R.D.; Archbold, K.H.; Dillon, J.E.; Pituch, K.J.; Panahi, P.; Dahl, R.E.; Guilleminault, C. Associations between symptoms of inattention, hyperactivity, restless legs, and periodic leg movements. Sleep 2002, 25, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Picchietti, D.L.; England, S.J.; Walters, A.S.; Willis, K.; Verrico, T. Periodic limb movement disorder and restless legs syndrome in children with attention-deficit hyperactivity disorder. J. Child Neurol. 1998, 13, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Golan, N.; Shahar, E.; Ravid, S.; Pillar, G. Sleep disorders and daytime sleepiness in children with attention-deficit/hyperactive disorder. Sleep 2004, 27, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Konofal, E.; Lecendreux, M.; Cortese, S. Sleep and ADHD. Sleep Med. 2010, 11, 652–658. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tesfaye, R.; Gruber, R. The Association between Sleep and Theory of Mind in School Aged Children with ADHD. Med. Sci. 2017, 5, 18. https://doi.org/10.3390/medsci5030018
Tesfaye R, Gruber R. The Association between Sleep and Theory of Mind in School Aged Children with ADHD. Medical Sciences. 2017; 5(3):18. https://doi.org/10.3390/medsci5030018
Chicago/Turabian StyleTesfaye, Rackeb, and Reut Gruber. 2017. "The Association between Sleep and Theory of Mind in School Aged Children with ADHD" Medical Sciences 5, no. 3: 18. https://doi.org/10.3390/medsci5030018
APA StyleTesfaye, R., & Gruber, R. (2017). The Association between Sleep and Theory of Mind in School Aged Children with ADHD. Medical Sciences, 5(3), 18. https://doi.org/10.3390/medsci5030018




