Recycling and Reuse of Mine Tailings: A Review of Advancements and Their Implications
Abstract
:1. Introduction
2. Methodology
3. Discussion
3.1. Mining and Mineral Processing Wastes
3.2. Recovery of Mine Wastes through Reuse and Recycling
3.2.1. Metal Waste
3.2.2. Gypsum Waste
3.2.3. Metallurgical Waste
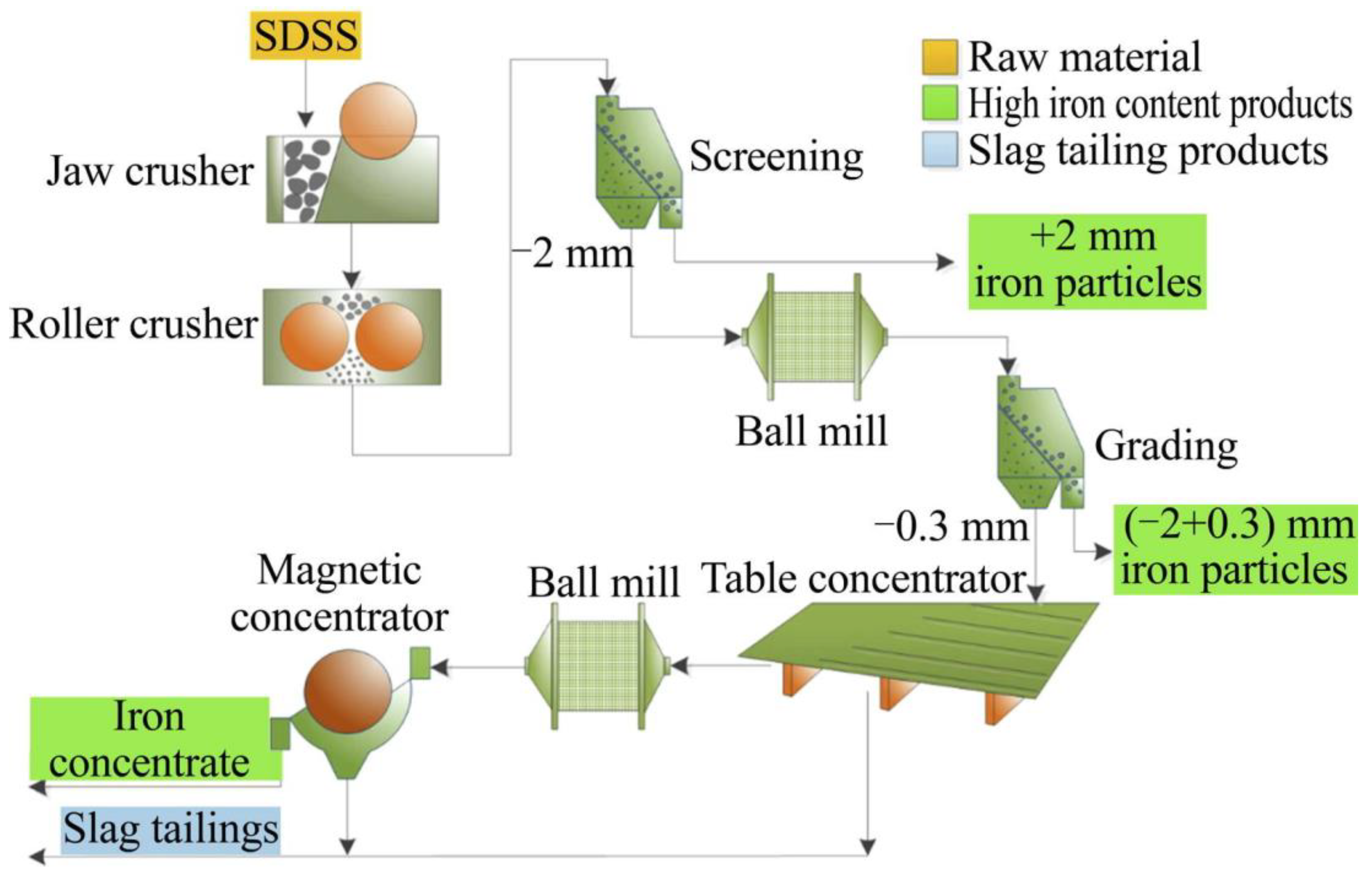
3.2.4. Steel Slag
3.3. Future Perspectives of Mine Tailing Remediation Techniques
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Cadeias, C.; Ávila, P.; Coelho, P.; Teixeira, J.P. Mining Activities: Health Impacts. In Encyclopedia of Environmental Health, 2nd ed.; Nriagu, J.O., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 788–802. [Google Scholar] [CrossRef]
- Hosseinpour, M.; Osanloo, M.; Azimi, Y. Evaluation of positive and negative impacts of mining on sustainable development by a semi-quantitative method. J. Clean. Prod. 2022, 366, 132955. [Google Scholar] [CrossRef]
- Shavina, E.; Prokofev, V. Implementation of environmental principles of sustainable development in the mining region. E3S Web Conf. 2020, 174, 1–6. [Google Scholar] [CrossRef]
- Garbarino, E.; Orveillon, G.; Saveyn, H.G. Management of waste from extractive industries: The new European reference document on the Best Available Techniques. Resour. Policy 2020, 69, 101782. [Google Scholar] [CrossRef]
- Agboola, O.; Babatunde, D.E.; Fay+omi, O.S.I.; Sadiku, E.R.; Popoola, P.; Moropeng, L.; Yahaya, A.; Mamudu, O.A. A review on the impact of mining operation: Monitoring, assessment and management. Results Eng. 2020, 8, 100181. [Google Scholar] [CrossRef]
- Vitti, C.; Arnold, B.J. The Reprocessing and Revalorization of Critical Minerals in Mine Tailings. Mining, Met. Explor. 2022, 39, 1–6. [Google Scholar] [CrossRef]
- Agboola, A.A. Farming Systems in NIGERIA. Agronomy in Nigeria; Wiley and Sons: Hoboken, NJ, USA; University of Ibadan: Ibadan, Nigeria, 2000; pp. 25–34. [Google Scholar]
- Edraki, M.; Baumgartl, T.; Manlapig, E.; Bradshaw, D.; Franks, D.M.; Moran, C.J. Designing mine tailings for better environmental, social and economic outcomes: A review of alternative approaches. J. Clean. Prod. 2014, 84, 411–420. [Google Scholar] [CrossRef]
- Luthra, S.; Mangla, S.K.; Sarkis, J.; Tseng, M.-L. Resources melioration and the circular economy: Sustainability potentials for mineral, mining and extraction sector in emerging economies. Resour. Policy 2022, 77, 1–4. [Google Scholar] [CrossRef]
- Marín, O.A.; Kraslawski, A.; Cisternas, L.A. Estimating processing cost for the recovery of valuable elements from mine tailings using dimensional analysis. Miner. Eng. 2022, 184, 107629. [Google Scholar] [CrossRef]
- Yoshizawa, S.; Tanaka, M.; Shekdar, A.V. Global trends in waste generation. In Recycling, Waste Treatment and Clean Technology; Gaballah, I., Mishar, B., Solozabal, R., Tanaka, M., Eds.; TMS Mineral, Metals and Materials Publishers: Madrid, Spain, 2004; pp. 1541–1552. [Google Scholar]
- Pappu, A.; Saxena, M.; Asolekar, S.R. Solid wastes generation in India and their recycling potential in building materials. Build. Environ. 2007, 42, 2311–2320. [Google Scholar] [CrossRef]
- Galán, J.E. The benefits are at the tail: Uncovering the impact of macroprudential policy on growth-at-risk. J. Financ. Stab. 2020, 100831. [Google Scholar] [CrossRef]
- Jiang, X.; Liu, W.; Xu, H.; Cui, X.; Li, J.; Chen, J.; Zheng, B. Characterizations of heavy metal contamination, microbial com-497 munity, and resistance genes in a tailing of the largest copper mine in China. Environ. Pollut. 2021, 280, 116947. [Google Scholar] [CrossRef] [PubMed]
- Jawadand, S.; Randive, K. A Sustainable Approach to Transforming Mining Waste into Value-Added Products. In Innovations in Sustainable Mining; Springer: Cham, Switzerland, 2021; pp. 1–20. [Google Scholar] [CrossRef]
- Shengo, L.M. Review of Practices in the Managements of Mineral Wastes: The Case of Waste Rocks and Mine Tailings. Wat. Air Soil Poll. 2021, 232, 273. [Google Scholar] [CrossRef]
- Park, I.; Tabelin, C.B.; Jeon, S.; Li, X.; Seno, K.; Ito, M.; Hiroyoshi, N. A review of recent strategies for acid mine drainage prevention and mine tailings recycling. Chemosphere 2019, 219, 588–606. [Google Scholar] [CrossRef] [PubMed]
- Mal, U.; Adhikari, K. Groundwater quality and hydrological stress induced by Lower Gondwana open cast coal mine. J. Earth Syst. Sci. 2021, 130, 32. [Google Scholar] [CrossRef]
- Tatsuhara, T.; Arima, T.; Igarashi, T.; Tabelin, C.B. Combined neutralization–adsorption system for the disposal of hydrothermally altered excavated rock producing acidic leachate with hazardous elements. Eng. Geol. 2012, 139–140, 76–84. [Google Scholar] [CrossRef]
- Lottermoser, B.G.; Ashley, P.M. Mobility and retention of trace elements in hardpan-cemented cassiterite tailings, north Queensland, Australia. Environ. Earth Sci. 2006, 50, 835–846. [Google Scholar] [CrossRef]
- Mackasey, W.O. Abandoned Mines in Canada; Unpublished Report prepared for Mining Watch Canada; WOM Geological Associates, Inc.: Sudbury, OM, Canada, 2000. [Google Scholar]
- Chartrand, M.M.G. Electrochemical remediation of acid mine drainage. J. Appl. Electrochem. 2003, 33, 259–264. [Google Scholar] [CrossRef]
- Wu, P.; Tang, C.; Liu, C.; Zhu, L.; Pei, T.; Feng, L. Geochemical distribution and removal of As, Fe, Mn and Al in a surface water system affected by acid mine drainage at a coalfield in Southwestern China. Environ. Earth Sci. 2008, 57, 1457–1467. [Google Scholar] [CrossRef]
- Motsi, T.; Rowson, N.A.; Simmons, M.J.H. Adsorption of heavy metals from acid mine drainage by natural zeolite. Int. J. Miner. Process. 2009, 92, 42–48. [Google Scholar] [CrossRef]
- Zagury, G.J.; Kulnieks, V.I.; Neculita, C.M. Characterization and reactivity assessment of organic substrates for sul-phate-reducing bacteria in acid mine drainage treatment. Chemosphere 2006, 64, 944–954. [Google Scholar] [CrossRef]
- Zhong, C.M.; Xu, Z.L.; Fang, X.H.; Cheng, L. Treatment of acid mine drainage (AMD) by ultra-low pressure reverse osmosis and nanofiltration. Environ. Eng. Sci. 2007, 24, 1297–1306. [Google Scholar] [CrossRef]
- McLellan, B.; Corder, G.; Giurco, D.; Green, S. Incorporating sustainable development in the design of mineral processing operations—Review and analysis of current approaches. J. Clean. Prod. 2009, 17, 1414–1425. [Google Scholar] [CrossRef]
- Lottermoser, B.G. Mine Wastes: Characterization, Treatment and Environmental Impacts, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2010; 400p. [Google Scholar]
- BRGM. Management of Mining, Quarrying and Ore-Processing Waste in the European Union; 7 Figs., 17 Tables, 7 annexes, 1 CD-ROM (Collected data); BRGM: Orlèans, France, 2001; 79p. [Google Scholar]
- Hann, D. Copper tailings reprocessing. RMZ Mater. Geoenviron. 2021, 67, 1–10. [Google Scholar] [CrossRef]
- Matinde, E.; Simate, G.; Ndlovu, S. Mining and metallurgical wastes: A review of recycling and re-use practices. J. S. Afr. Inst. Min. Met. 2018, 118, 825–844. [Google Scholar] [CrossRef]
- Oluwasola, E.A.; Hainin, M.R.; Aziz, M.M.A. Evaluation of asphalt mixtures incorporating electric arc furnace steel slag and copper mine tailings for road construction. Transp. Geotech. 2015, 2, 47–55. [Google Scholar] [CrossRef]
- Çelik, Ö.; Elbeyli, I.Y.; Piskin, S. Utilization of gold tailings as an additive in Portland cement. Waste Manag. Res. J. Sustain. Circ. Econ. 2006, 24, 215–224. [Google Scholar] [CrossRef]
- Kossoff, D.; Dubbin, W.; Alfredsson, M.; Edwards, S.; Macklin, M.; Hudson-Edwards, K. Mine tailings dams: Characteristics, failure, environmental impacts, and remediation. Appl. Geochem. 2014, 51, 229–245. [Google Scholar] [CrossRef]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef]
- Shamsuddin, M. Physical Chemistry of Metallurgical Processes, 1st ed.; The Minerals, Metals & Materials Society; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016. [Google Scholar]
- Submarine Tailings Disposal Toolkit. Available online: http://www.miningwatch.ca/files/01.STDtoolkit.intr_.pdf (accessed on 12 June 2014).
- MiningWatch Canada Webpage. Troubled Waters: How Mine Waste Dumping is Poisoning Our Oceans, Rivers, and Lakes. Available online: http://www.miningwatch.ca/news/troubled-watershow-mine-waste-dumping-poisoning-our-oceans-rivers-and-lakes (accessed on 12 June 2014).
- Dold, B. Submarine Tailings Disposal (STD)—A Review. Minerals 2014, 4, 642–666. [Google Scholar] [CrossRef]
- Rashad, A.M. Phosphogypsum as a construction material. J. Clean. Prod. 2017, 166, 732–743. [Google Scholar] [CrossRef]
- Zhang, L. Production of bricks from waste materials—A review. Constr. Build. Mater. 2013, 47, 643–655. [Google Scholar] [CrossRef]
- Ahmari, S.; Zhang, L. Production of eco-friendly bricks from copper mine tailings through geopolymerization. Constr. Build. Mater. 2012, 29, 323–331. [Google Scholar] [CrossRef]
- Tang, Z.; Lei, T.; Yu, J.; Shainberg, I.; Mamedov, A.I.; Ben-Hur, M.; Levy, G.J.; Mamedov, A.I. Runoff and Interrill Erosion in Sodic Soils Treated with Dry PAM and Phosphogypsum. Soil Sci. Soc. Am. J. 2006, 70, 679–690. [Google Scholar] [CrossRef]
- Zhang, X.C.; Miller, W.P.; Nearing, M.A.; Norton, L.D. Effects of surface treatment on surface sealing, runoff, and interrill erosion. Trans. ASAE 1998, 41, 989–994. [Google Scholar] [CrossRef]
- Takahashi, T.; Ikeda, Y.; Nakamura, H.; Nanzyo, M. Efficiency of gypsum application to acid Andosols estimated using aluminum release rates and plant root growth. Soil Sci. Plant Nutr. 2006, 52, 584–592. [Google Scholar] [CrossRef]
- Rodríguez-Jordá, M.; Garrido, F.; García-González, M. Potential use of gypsum and lime rich industrial by-products for induced reduction of Pb, Zn and Ni leachability in an acid soil. J. Hazard Mater. 2010, 175, 762–769. [Google Scholar] [CrossRef]
- Delgado, A.; Madrid, A.; Kassem, S.; Andreu, L.; del Campillo, M.C. Phosphorus fertilizer recovery from calcareous soils amended with humic and fulvic acids. Plant Soil 2002, 245, 277–286. [Google Scholar] [CrossRef]
- Gorakhki, M.H.; Bareither, C.A. Sustainable Reuse of Mine Tailings and Waste Rock as Water-Balance Covers. Minerals 2017, 7, 128. [Google Scholar] [CrossRef] [Green Version]
- Lottermoser, B. Recycling, Reuse and Rehabilitation of Mine Wastes. Elements 2011, 7, 405–410. [Google Scholar] [CrossRef]
- Owen, J.; Kemp, D.; Lèbre, É.; Svobodova, K.; Murillo, G.P. Catastrophic tailings dam failures and disaster risk disclosure. Int. J. Disaster Risk Reduct. 2019, 42, 101361. [Google Scholar] [CrossRef]
- Kumar, U.; Singh, D.N. E-waste management through regulations. Int. J. Eng. Invent. 2013, 3, 6–14. [Google Scholar] [CrossRef]
- Azcue, J.M. Environmental Impacts of Mining Activities: Emphasis on Mitigation and Remedial Measures; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar] [CrossRef]
- Cobîrzan, N.; Muntean, R.; Thalmaier, G.; Felseghi, R.-A. Recycling of Mining Waste in the Production of Masonry Units. Materials 2022, 15, 594. [Google Scholar] [CrossRef] [PubMed]
- Aznar-Sánchez, J.A.; García-Gómez, J.J.; Velasco-Muñoz, J.F.; Carretero-Gómez, A. Mining Waste and Its Sustainable Management: Advances in Worldwide Research. Minerals 2018, 8, 284. [Google Scholar] [CrossRef]
- Wang, H.-G.; Liu, W.; Jia, N.; Zhang, M.; Guo, M. Facile synthesis of metal-doped Ni-Zn ferrite from treated Zn-containing electric arc furnace dust. Ceram. Int. 2017, 43, 1980–1987. [Google Scholar] [CrossRef]
- Ndlovu, S.; Simate, G.S.; Matinde, E. Waste Production and Utilization in the Metal Extraction Industry, 1st ed.; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Mackay, I.; Mendez, E.; Molina, I.; Videla, A.R.; Cilliers, J.J.; Brito-Parada, P.R. Dynamic froth stability of copper flotation tailings. Miner. Eng. 2018, 124, 103–107. [Google Scholar] [CrossRef]
- Kalisz, S.; Kibort, K.; Mioduska, J.; Lieder, M.; Małachowska, A. Waste management in the mining industry of metals ores, coal, oil and natural gas—A review. J. Environ. Manag. 2021, 304, 114239. [Google Scholar] [CrossRef]
- Rao, G.V.; Markandeya, R.; Kumar, R. Modeling and Optimisation of Multigravity Separator for Recovery of Iron Values from Sub Grade Iron Ore Using Three Level Three Factor Box Behnken Design. Int. J. Miner. Process. Extr. Met. 2017, 2, 46. [Google Scholar] [CrossRef]
- Duarte, J.C.; Estrada, P.; Beaumont, H.; Sitima, M.; Pereira, P. Biotreatment of tailings for metal recovery. Mine Water Environ. 1990, 9, 193–206. [Google Scholar] [CrossRef]
- Stankovic, S.; Moric, I.; Pavic, A.; Vojnovic, S.; Vasiljevic, B.; Cvetkovic, V. Bioleaching of copper from old flotation tailings samples (Copper Mine Bor, Serbia). J. Serbian Chem. Soc. 2015, 80, 391–405. [Google Scholar] [CrossRef]
- Pulungan, L.; Pramusanto, P.; Hermana, F.A. The research of gold processing from tailings of iron sand processing from South Kalimantan by using amalgamation methods in West Java. J. Phys. Conf. Ser. 2019, 1375, 12047. [Google Scholar] [CrossRef]
- Cánovas, C.R.; Macías, F.; Pérez-López, R.; Basallote, M.D.; Millán-Becerro, R. Valorization of wastes from the fertilizer industry: Current status and future trends. J. Clean. Prod. 2018, 174, 678–690. [Google Scholar] [CrossRef]
- Garg, M.; Singh, M.; Kumar, R. Some aspects of the durability of a phosphogypsum-lime-fly ash binder. Constr. Build. Mater. 1996, 10, 273–279. [Google Scholar] [CrossRef]
- Lin, X.; Peng, Z.; Yan, J.; Li, Z.; Hwang, J.-Y.; Zhang, Y.; Li, G.; Jiang, T. Pyrometallurgical recycling of electric arc furnace dust. J. Clean. Prod. 2017, 149, 1079–1100. [Google Scholar] [CrossRef]
- de Buzin, P.J.W.K.; Heck, N.C.; Vilela, A.C.F. EAF dust: An overview on the influences of physical, chemical and mineral features in its recycling and waste incorporation routes. J. Mater. Res. Technol. 2017, 6, 194–202. [Google Scholar] [CrossRef]
- Rodríguez, O.; Alguacil, F.J.; Baquero, E.E.; García-Díaz, I.; Fernández, P.; Sotillo, B.; López, F.A. Recovery of niobium and tantalum by solvent extraction from Sn–Ta–Nb mining tailings. RSC Adv. 2020, 10, 21406–21412. [Google Scholar] [CrossRef] [PubMed]
- Hansen, H.; Rojo, A.; Ottosen, L. Electrodialytic Remediation of Copper Mine Tailings. Procedia Eng. 2012, 44, 2053–2055. [Google Scholar] [CrossRef]
- Bisio, G. Energy recovery from molten slag and exploitation of the recovered energy. Energy 1997, 22, 501–509. [Google Scholar] [CrossRef]
- Barati, M.; Esfahani, S.; Utigard, T. Energy recovery from high temperature slags. Energy 2011, 36, 5440–5449. [Google Scholar] [CrossRef]
- Duan, W.; Yu, Q.; Wang, Z. Comprehensive Analysis of the Coal Particle in Molten Blast Furnace Slag To Recover Waste Heat. Energy Fuels 2017, 31, 8813–8819. [Google Scholar] [CrossRef]
- Šajn, R.; Ristović, I.; Čeplak, B. Mining and Metallurgical Waste as Potential Secondary Sources of Metals—A Case Study for the West Balkan Region. Minerals 2022, 12, 547. [Google Scholar] [CrossRef]
- Whitworth, A.J.; Vaughan, J.; Southam, G.; van der Ent, A.; Nkrumah, P.N.; Ma, X.; Parbhakar-Fox, A. Review on metal extraction technologies suitable for critical metal recovery from mining and processing wastes. Miner. Eng. 2022, 182, 107537. [Google Scholar] [CrossRef]
- Kim, S.-K.; Yang, D.-H.; Rao, S.; Nam, C.-W.; Rhee, K.-I.; Sohn, J.-S. A new approach to the recycling of gold mine tailings using red mud and waste limestone as melting fluxes. Geosystem Eng. 2012, 15, 44–49. [Google Scholar] [CrossRef]
- Lemougna, P.N.; Yliniemi, J.; Ismailov, A.; Levänen, E.; Tanskanen, P.; Kinnunen, P.; Röning, J.; Illikainen, M. Recycling lithium mine tailings in the production of low temperature (700–900 °C) ceramics: Effect of ladle slag and sodium compounds on the processing and final properties. Constr. Build. Mater. 2019, 221, 332–344. [Google Scholar] [CrossRef]
- Choi, Y.W.; Kim, Y.J.; Choi, O.; Lee, K.M.; Lachemi, M. Utilization of tailings from tungsten mine waste as a substitution material for cement. Constr. Build. Mater. 2009, 23, 2481–2486. [Google Scholar] [CrossRef]
- Qiu, G.; Luo, Z.; Shi, Z.; Ni, M. Utilization of coal gangue and copper tailings as clay for cement clinker calcinations. J. Wuhan Univ. Technol. Sci. Ed. 2011, 26, 1205–1210. [Google Scholar] [CrossRef]
- Prahallada, M.C.; Shanthappa, B.C. Use of copper ore tailings—As an excellent pozzolana in the preparation of concrete. Int. J. Adv. Eng. 2014, 3, 1–10. [Google Scholar]
- Onuaguluchi, O.; Eren, Ö. Reusing copper tailings in concrete: Corrosion performance and socioeconomic implications for the Lefke-Xeros area of Cyprus. J. Clean. Prod. 2016, 112, 420–429. [Google Scholar] [CrossRef]
- Gorai, B.; Jana, R. Premchand Characteristics and utilisation of copper slag—A review. Resour. Conserv. Recycl. 2003, 39, 299–313. [Google Scholar] [CrossRef]
- da Silva, F.; Araújo, F.; Teixeira, M.; Gomes, R.; von Krüger, F. Study of the recovery and recycling of tailings from the concentration of iron ore for the production of ceramic. Ceram. Int. 2014, 40, 16085–16089. [Google Scholar] [CrossRef]
- Shewalul, Y.W. Experimental study of the effect of waste steel scrap as reinforcing material on the mechanical properties of concrete. Case Stud. Constr. Mater. 2021, 14, e00490. [Google Scholar] [CrossRef]
- Rouaiguia, I.; Bounouala, M.; Abdelmalek, C.; Idres, A.; Benselhoub, A. Optical sorting technology for waste management from the Boukhadra iron ore mine (NE Algeria). REM—Int. Eng. J. 2022, 75, 55–65. [Google Scholar] [CrossRef]
- Instituto Brasileiro de Mineração (IBRAM). Gestão e Manejo de Rejeitos da Mineração, 1st ed.; Instituto Brasileiro de Mineração, IBRAM: Brasilia, Brazil, 2016; p. 128. [Google Scholar]
- Mueller, H.; Maithy, S.; Prajapati, S.; Bhatta, A.D.; Shrestha, B.L. Greenbrick Making Manual; Hillside Press: Kathmandu, Nepal, 2008; Available online: http://www.ecobrick.in/resource_data/KBAS100046.pdf. (accessed on 1 November 2021).
- Bennet, J.M.; Sudhakar, M.; Natarajan, C. Development of coal ash—GGBS based geopolymer bricks. EJOSAT 2013, 2, 133–139. [Google Scholar]
- Kuranchie, F.A.; Shukla, S.; Habibi, D.; Mohyeddin, A. Utilisation of iron ore tailings as aggregates in concrete. Cogent Eng. 2015, 2, 1–11. [Google Scholar] [CrossRef]
- Ugama, T.I.; Ejeh, S.P.; Amartey, D.Y. Effect of iron ore tailing on the properties of concrete. Civ. Environ. Res. 2014, 6, 7–13. [Google Scholar]
- Utilization of iron ore tailings as replacement to fine aggregates in cement concrete pavements. Int. J. Res. Eng. Technol. 2014, 3, 369–376. [CrossRef]
- Niu, H.; Helser, J.; Corfe, I.J.; Kuva, J.; Butcher, A.R.; Cappuyns, V.; Kinnunen, P.; Illikainen, M. Incorporation of bioleached sulfidic mine tailings in one-part alkali-activated blast furnace slag mortar. Constr. Build. Mater. 2022, 333, 127195. [Google Scholar] [CrossRef]
- Simate, G.S.; Ndlovu, S. Acid mine drainage: Challenges and opportunities. J. Environ. Chem. Eng. 2014, 2, 1785–1803. [Google Scholar] [CrossRef]
- Bian, X.; Zeng, L.; Ji, F.; Xie, M.; Hong, Z. Plasticity role in strength behavior of cement-phosphogypsum stabilized soils. J. Rock Mech. Geotech. Eng. 2022. [Google Scholar] [CrossRef]
- Kovler, K. Radiological constraints of using building materials and industrial by-products in construction. Constr. Build. Mater. 2009, 23, 246–253. [Google Scholar] [CrossRef]
- Vásconez-Maza, M.D.; Bueso, M.C.; Mulas, J.; Faz, Á.; Martínez-Segura, M.A. Characterising an abandoned phosphogypsum deposit by combining radiological, geophysical, geochemical, and statistical techniques. CATENA 2022, 216, 106401. [Google Scholar] [CrossRef]
- Nyirenda, R. The processing of steelmaking flue-dust: A review. Miner. Eng. 1991, 4, 1003–1025. [Google Scholar] [CrossRef]
- Beukes, J.P.; van Zyl, P.G.; RAS, M. Treatment of Cr (VI)-containing wastes in the South African ferrochrome industry—A review of currently applied methods. J. S. Afr. Inst. Min. Metall. 2012, 112, 347–352. [Google Scholar]
- Saxena, S.; Jotshi, C. Management and combustion of hazardous wastes. Prog. Energy Combust. Sci. 1996, 22, 401–425. [Google Scholar] [CrossRef]
- Reuter, M.; Xiao, Y.; Boin, U. Recycling and environmental issues of metallurgical slags and salt fluxes. In VII International Conference on Molten Slags, Fluxes and Salts; Southern African Institute of Mining and Metallurgy: Johannesburg, South Africa, 2004; Volume 2004, pp. 349–356. [Google Scholar]
- Durinck, D.; Engström, F.; Arnout, S.; Heulens, J.; Jones, P.T.; Björkman, B.; Blanpain, B.; Wollants, P. Hot stage processing of metallurgical slags. Resour. Conserv. Recycl. 2008, 52, 1121–1131. [Google Scholar] [CrossRef]
- Quijorna, N.; Miguel, G.S.; Andrés, A. Incorporation of Waelz Slag into Commercial Ceramic Bricks: A Practical Example of Industrial Ecology. Ind. Eng. Chem. Res. 2011, 50, 5806–5814. [Google Scholar] [CrossRef]
- Karayannis, V.; Ntampegliotis, K.; Lamprakopoulos, S.; Papapolymerou, G.; Spiliotis, X. Novel sintered ceramic materials in-693 corporated with EAF carbon steel slag. Mater. Res. Express. 2017, 4, 015505. [Google Scholar] [CrossRef]
- Estokova, A.; Singovszka, E. Assessment of risk from irradiation originating from mortars with mineral waste addition. Indoor Built Environ. 2021, 31, 219–229. [Google Scholar] [CrossRef]
- Dubey, S.S.A.; Kushwah, S.S. Utilisation of Iron and Steel Slag in Building Construction. Proceedings of the International Conference on Sustainable Materials and Structures for Civil Infrastructures (SMSCI2019). AIP Conf. Proc. 2019, 2158, 20032. [Google Scholar]
- Rosales, J.; Agrela, F.; Entrenas, J.A.; Cabrera, M. Potential of Stainless-Steel Slag Waste in Manufacturing Self-Compacting Concrete. Materials 2020, 13, 2049. [Google Scholar] [CrossRef]
- Fahad, M.B.; Abdulkarem, A.M.; Hamed, T.H. A review on wastes as sustainable construction materials. IOP Conf. Ser. Earth Environ. Sci. 2021, 779, 12014. [Google Scholar] [CrossRef]
- Fisher, L.V.; Barron, A.R. The recycling and reuse of steelmaking slags—A review. Resour. Conserv. Recycl. 2019, 146, 244–255. [Google Scholar] [CrossRef]
- Bodor, M.; Santos, R.M.; Cristea, G.; Salman, M.; Cizer, Ö.; Iacobescu, R.I.; Chiang, Y.W.; Van Balen, K.; Vlad, M.; Van Gerven, T. Laboratory investigation of carbonated BOF slag used as partial replacement of natural aggregate in cement mortars. Cem. Concr. Compos. 2016, 65, 55–66. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Gao, P.; Han, Y. Resource utilization of slag from desulphurization and slag skimming: A comprehensive recycling process of all components. Int. J. Min. Sci. Technol. 2022, 32, 585–593. [Google Scholar] [CrossRef]
- Peng, Y.; Liu, Z.; Liu, X.; Sheng, M.; Li, H.; Xu, X.; Ai, L.; Yan, Q.; Yang, Y. Preparation of composite micro-slag based on the application of tailings slag in cement and concrete. Constr. Build. Mater. 2022, 322, 126515. [Google Scholar] [CrossRef]
- Bian, Z.; Miao, X.; Lei, S.; Chen, S.-E.; Wang, W.; Struthers, S. The Challenges of Reusing Mining and Mineral-Processing Wastes. Science 2012, 337, 702–703. [Google Scholar] [CrossRef] [PubMed]
- Fox, P.; Gilmour, S.; Parbhakar-Fox, A.; Olin, P. Geometallurgical Characterization of Non-Ferrous Historical Slag in Western Tasmania: Identifying Reprocessing Options. Minerals 2019, 9, 415. [Google Scholar] [CrossRef]
- Kouziyev, D.; Krivenko, A.; Chezganova, D.; Valeriy, B. Sensing of Dynamic Loads in the Open-Cast Mine Combine. E3S Web Conf. 2019, 105, 3014. [Google Scholar] [CrossRef]
- Bardovsky, A.D.; Gorbatyuk, S.M.; Albul, S.V.; Gorbatyuk, N.V. Optimization of Process Flow Diagrams for Processing of Mineral Wastes. Metallurgist 2021, 65, 465–472. [Google Scholar] [CrossRef]
- Qaidi, S.; Tayeh, B.A.; Zeyad, A.M.; de Azevedo, A.R.; Ahmed, H.U.; Emad, W. Recycling of mine tailings for the geopolymers production: A systematic review. Case Stud. Constr. Mater. 2022, 16, e00933. [Google Scholar] [CrossRef]
- Figueiredo, R.A.; Silveira, A.B.; Melo, E.L.; Costa, G.Q.; Brandão, P.R.; Aguilar, M.T.; Henriques, A.B.; Mazzinghy, D.B. Mechanical and chemical analysis of one-part geopolymers synthesised with iron ore tailings from Brazil. J. Mater. Res. Technol. 2021, 14, 2650–2657. [Google Scholar] [CrossRef]
- Sheoran, V.; Choudhary, R. Phytostabilization of mine tailings. In Phytorestoration of Abandoned Mining and Oil Drilling Sites., 1st ed.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 307–324. [Google Scholar]
- Shettima, A.U.; Hussin, M.W.; Ahmad, Y.; Mirza, J. Evaluation of iron ore tailings as replacement for fine aggregate in concrete. Constr. Build. Mater. 2016, 120, 72–79. [Google Scholar] [CrossRef]
- Samir, M.; Alama, F.; Buysse, P.; Van Nylen, T.; Ostanin, O. Disposal of Mining Waste: Classification and International Recycling Experience. E3S Web Conf. 2018, 41, 2012. [Google Scholar] [CrossRef]
- Tsukerman, V.A.; Ivanov, S.V. Use of Mineral Waste of Industrial Enterprises in the Arctic Zone of the Russian Federation. IOP Conf. Series: Earth Environ. Sci. 2022, 988, 32001. [Google Scholar] [CrossRef]
- Li, C.; Sun, H.; Bai, J.; Li, L. Innovative methodology for comprehensive utilization of iron ore tailings: Part 1. The recovery of iron from iron ore tailings using magnetic separation after magnetizing roasting. J. Hazard Mater. 2010, 174, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Wang, L.; Wu, A.; Kabwe, E.; Chen, X.; Yan, R. Copper recycle from sulfide tailings using combined leaching of ammonia solution and alkaline bacteria. J. Clean. Prod. 2018, 189, 746–753. [Google Scholar] [CrossRef]
- Yin, Z.; Sun, W.; Hu, Y.; Zhang, C.; Guan, Q.; Wu, K. Evaluation of the possibility of copper recovery from tailings by flotation through bench-scale, commissioning, and industrial tests. J. Clean. Prod. 2017, 171, 1039–1048. [Google Scholar] [CrossRef]
- Tyulenev, M.; Lesin, Y.; Litvin, O.; Maliukhina, E.; Abay, A. Increasing the Reliability of the Work of Artificial Filtering Arrays for the Purification of Quarry Waste Water. E3S Web Conf. 2017, 21, 2019. [Google Scholar] [CrossRef]
- Rivas, D.F.; Boffito, D.C.; Faria-Albanese, J.; Glassey, J.; Afraz, N.; Akse, H.; Boodhoo, K.; Bos, R.; Cantin, J.; Chiang, Y.W.; et al. Process intensification education contributes to sustainable development goals. Part 1. Educ. Chem. Eng. 2020, 32, 1–14. [Google Scholar] [CrossRef]
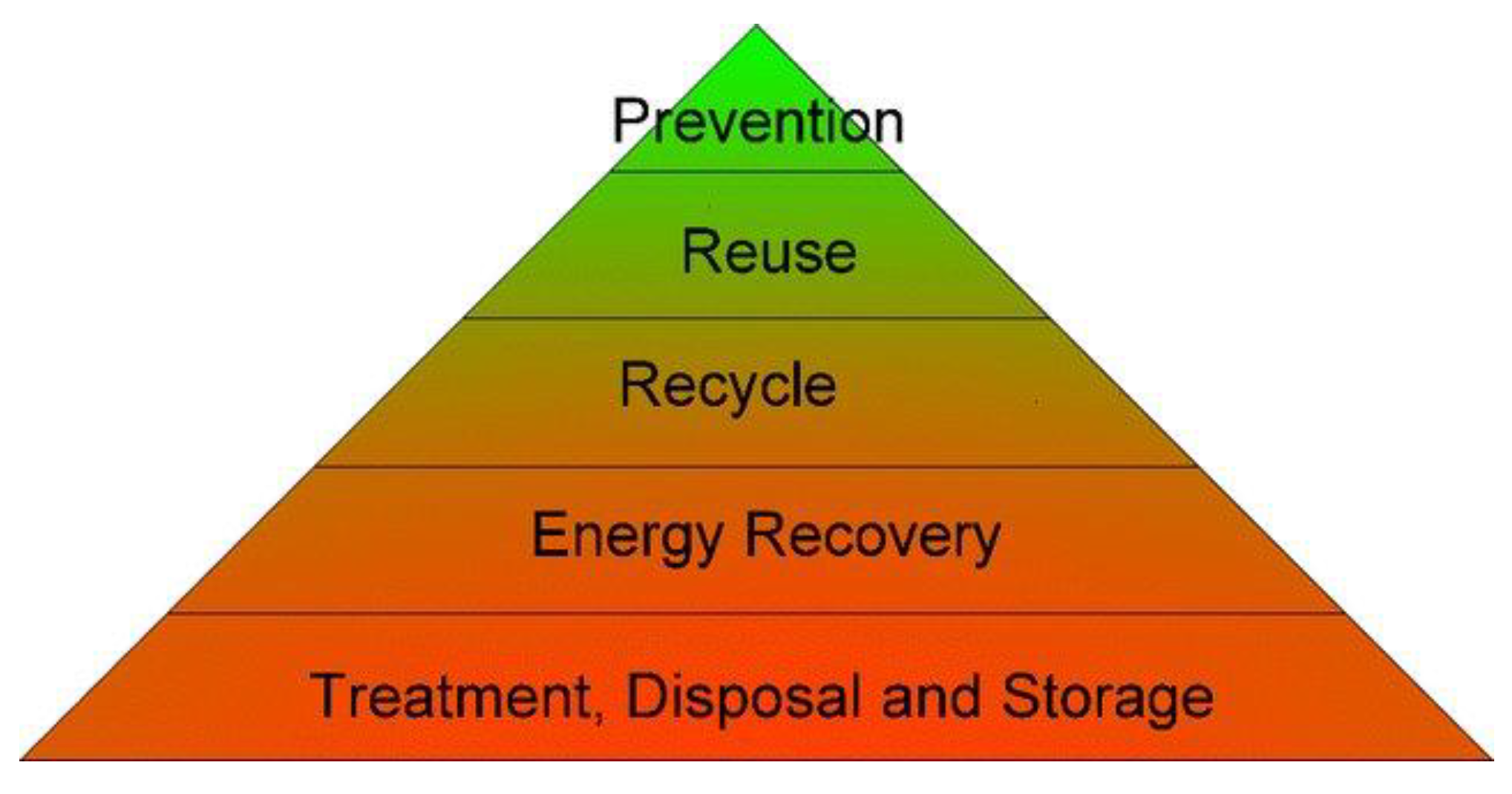


| Types of Mining Waste | Physical Classification of Residues | Environmental Classification | Disposal Options |
|---|---|---|---|
Rock waste (sterile)/Processing waste
| Solid form
| Chemical and mineralogical composition Physical properties Volume and surface occupied Waste disposal method | Tailings dams Exhausted mine pits, In piles, by dry stacking (suitable for areas of high seismic activity, for cold climates) Disposal in paste Underground backfilling Submarine tailing disposal (STD) |
| Types of Tailings Identified | Application | Number of Articles Analyzed |
|---|---|---|
| Iron ore; Copper; Platinum Group Metals; AMD; Zinc; Phosphogypsum; Slag; Red mud; Electric oven powders; Limestone powder; Fly ash and sewage sludge; Clay-based residues; Gold tailings; Marble; Coal combustion | Construction materials | 25 |
| Manganese; Phosphogypsum; Platinum Group Metals; Combustion coal; Mine drainage sludge; Limestone powder; Phytoremediated tailings | Agricultural applications | 7 |
| AMD-causing tailings | Geopolymers | 3 |
| Chromium ore tailings | Automobile catalytic converters; electronic materials; jewelry | 3 |
| Sand-based tailings; Platinum Group Metals; Coal combustion; Copper slag | Landfills and source of rare earth elements | 3 |
| Type of Waste | Main Recycling/Reuse Processes | Advantages | Limitations | Citing Articles |
|---|---|---|---|---|
| Metal waste | Flotation | Large-scale use; effective application in fine minerals; application in non-magnetic ores. | Low recovery when mixed with mud. | Wang et al., 2017 [55]; Ndlovu et al., 2017 [56]; Mackay et al., 2018 [57]; Shengo, 2021 [16]; Kalisz et al., 2022 [58]. |
| Gravity Separation | No use of chemical products; Relatively little environmental impact except for the disposal of sludge; Operational simplicity; Lower cost than flotation; Application in materials with larger particle size. | Considerable loss of tailings when the method is dense type. | Wang et al., 2017 [55]; Ndlovu et al., 2017 [56]; Rao et al., 2017 [59]. | |
| Magnetic Separation | Low operational cost; Simplicity of equipment; A small amount in the release of waste that can affect the environment. | Application only in waste with the presence of magnetic materials. | Wang et al., 2017 [55]; Ndlovu et al., 2017 [56]. | |
| Solvent Extraction | Economically and operationally feasible to execute in a short time; obtaining elements with high purity; effective in the selective extraction of heavy metals from industrial waste. | Cost, degradation, volatility of solvents. | Ndlovu et al., 2017 [56]. | |
| Biolixiviation (bioleaching) | Microorganisms are used to obtain metals from low-grade ores; High technological potential; Recent technology. | Slow rate; Climate dependent; Containment requirements. | Duarte et al., 1990 [60]; Stanković et al., 2015 [61]. | |
| Amalgamation | An efficient process for extracting larger particle size metals; Simple and inexpensive process. | Limitation in recovering fine-grained materials. | Pulungan et al., 2019 [62]. | |
| Gypsum waste | Solvent Extraction | Good selectivity; Obtaining elements with high purity. | - | Cánovas et al., 2018 [63]; Garg et al., 1996 [64]. |
| Acid Leaching | Low energy input; Low investment. | Difficult separation of impurities; Presence of a high volume of acid. | Cánovas et al., 2018 [63]. | |
| Metallurgical waste | Pyrometallurgical Process | Ability to receive zinc-based metallurgical powders. | High thermal energy requirements; Additional steps to recover volatile metals from flue gas. | Matinde et al., 2018 [30]; Ndlovu et al., 2017 [56]; Lin et al., 2017 [65]. |
| Hydrometallurgical process | Increasing use in recent years; Flexible and economical; Few environmental problems. | Chemical consumption; Separations challenges. | Matinde et al., 2018 [31]; Buzin et al., 2017 [66]; Ndlovu et al., 2017 [56]; Rodríguez et al. 2020 [67]. | |
| Electrometallurgical process | Emerging technology; Smaller scale use. | Materials of construction requirements. | Hansen et al., 2012 [68]. | |
| Steel slag | Dry Granulation | More used; More effective; Less environmental pollution. | Lower product value. | Bisio, 1997 [69]; Barati et al., 2011 [70]. |
| Air Blast Granulation | Metals recovered with higher heterogeneity. | Higher energy consumption. | Bisio, 1997 [69]; Barati et al., 2011 [70]. | |
| Granulation with Liquid Slag Impact | Reduction of energy intensity in the metal production process. | The release of toxic gases; Little possibility of using vitreous slag in materials such as cement. | Barati et al., 2011 [70]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araujo, F.S.M.; Taborda-Llano, I.; Nunes, E.B.; Santos, R.M. Recycling and Reuse of Mine Tailings: A Review of Advancements and Their Implications. Geosciences 2022, 12, 319. https://doi.org/10.3390/geosciences12090319
Araujo FSM, Taborda-Llano I, Nunes EB, Santos RM. Recycling and Reuse of Mine Tailings: A Review of Advancements and Their Implications. Geosciences. 2022; 12(9):319. https://doi.org/10.3390/geosciences12090319
Chicago/Turabian StyleAraujo, Francisco S. M., Isabella Taborda-Llano, Everton Barbosa Nunes, and Rafael M. Santos. 2022. "Recycling and Reuse of Mine Tailings: A Review of Advancements and Their Implications" Geosciences 12, no. 9: 319. https://doi.org/10.3390/geosciences12090319
APA StyleAraujo, F. S. M., Taborda-Llano, I., Nunes, E. B., & Santos, R. M. (2022). Recycling and Reuse of Mine Tailings: A Review of Advancements and Their Implications. Geosciences, 12(9), 319. https://doi.org/10.3390/geosciences12090319









