Review of Fly-Ash-Based Geopolymers for Soil Stabilisation with Special Reference to Clay
Abstract
1. Introduction
2. Fly-Ash Geopolymers for Soil Stabilisation
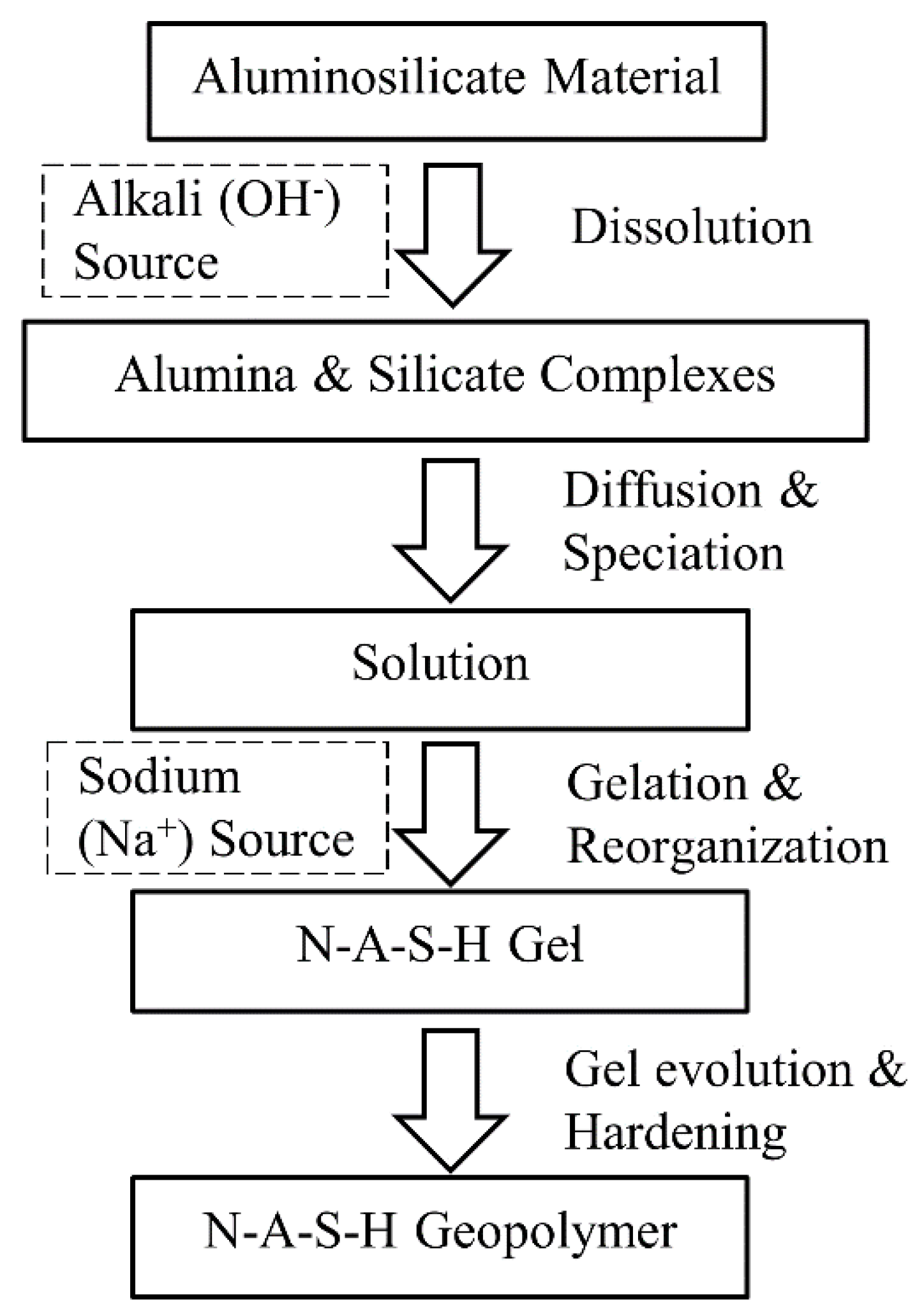
2.1. The N-A-S-H Geopolymer Model for Soil Stabilisation
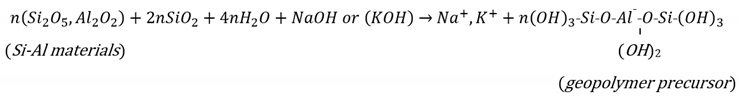
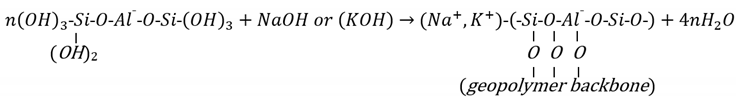
2.1.1. Engineering Properties of the N-A-S-H Geopolymer-Treated Soils
Microstructure
Strength
Durability Characteristics
2.1.2. Factors Affecting Geopolymer Formation of the N-A-S-H Treated Soils
Aluminosilicate and Activator Requirements
Aluminosilicate and Activator Contents
Curing Conditions
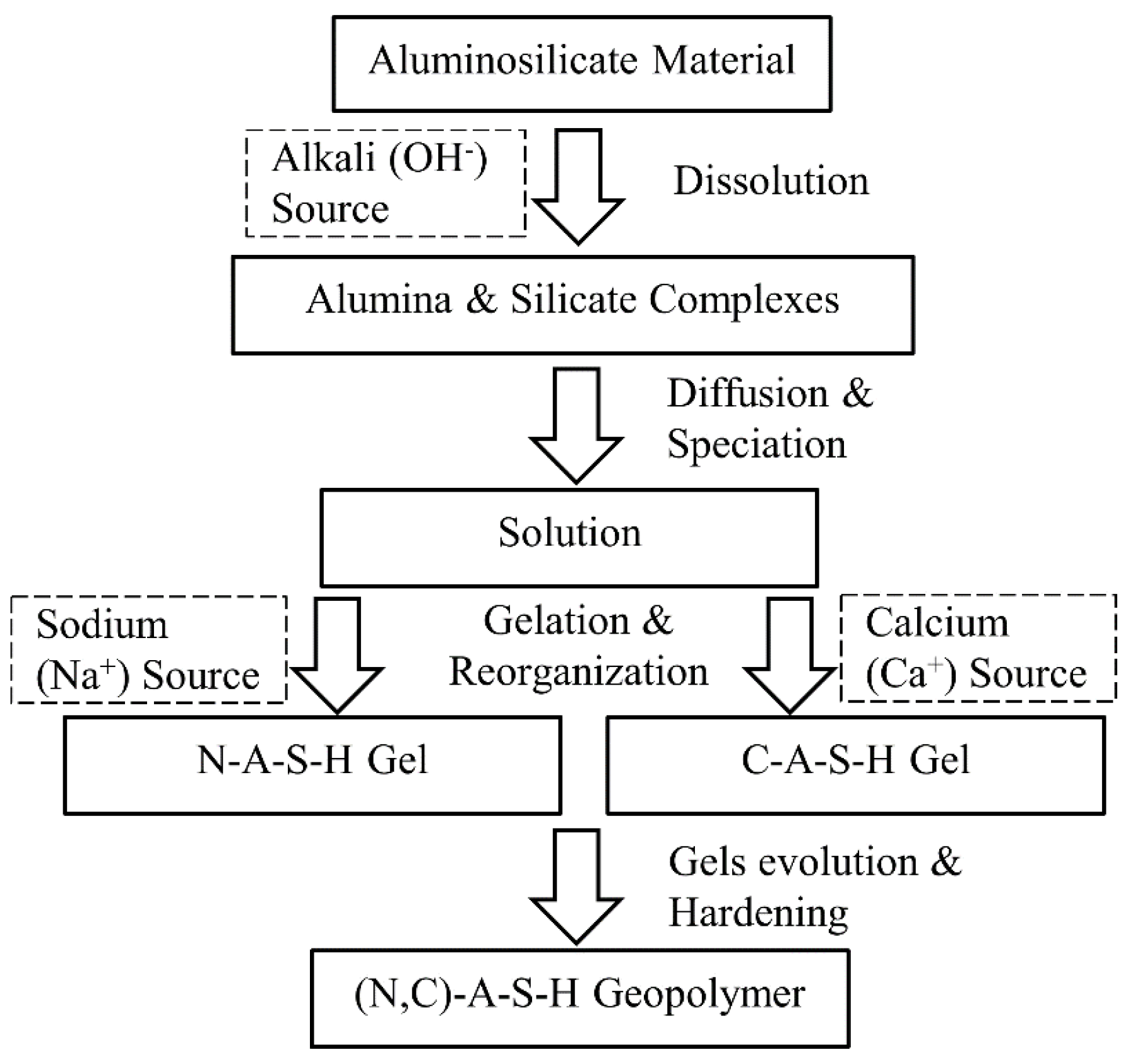
2.2. The (N, C)-A-S-H Geopolymer Model for Soil Stabilisation
3. Utilisation of Geopolymer for Clay Stabilisation
4. Limitations to Broad Utilisation of Geopolymer for Soil Stabilisation
4.1. Curing at Ambient Temperature
4.2. Availability of Practical Procedures
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mitchell, J.K.; Soga, K. Fundamentals of Soil Behavior, 3rd ed.; John Wiley & Sons: New York, NY, USA, 2005. [Google Scholar]
- Bergado, D.; Anderson, L.; Miura, N.; Balasubramaniam, A. Soft Ground Improvement in Lowland and Other Environments; American Society of Civil Engineers: New York, NY, USA, 1996. [Google Scholar]
- Han, J. Principles and Practice of Ground Improvement; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Moseley, M.P.; Kirsch, K. Ground improvement; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Nicholson, P.G. Soil Improvement and Ground Modification Methods; Butterworth-Heinemann: Waltham, MA, USA, 2014. [Google Scholar]
- Kirsch, K. Ground improvement by deep vibratory methods. Noise Control. Eng. J. 2010, 58. [Google Scholar] [CrossRef]
- Karol, R. Chemical Grouting and Soil Stabilization, Revised and Expanded; Informa UK Limited: London, UK, 2003; Volume 12. [Google Scholar]
- Lodeiro, I.G.; Palomo, A.; Fernández-Jiménez, A. An overview of the chemistry of alkali-activated cement-based binders. In Handbook of Alkali-Activated Cements, Mortars and Concretes; Elsevier BV: Amsterdam, The Netherlands, 2015; pp. 19–47. [Google Scholar]
- Provis, J.L.; Bernal, S.A. Geopolymers and related alkali-activated materials. Annu. Rev. Mater. Res. 2014, 44, 299–327. [Google Scholar] [CrossRef]
- Tayibi, H.; Choura, M.; López, F.A.; Alguacil, F.J.; López-Delgado, A. Environmental impact and management of phosphogypsum. J. Environ. Manag. 2009, 90, 2377–2386. [Google Scholar] [CrossRef] [PubMed]
- Sargent, P. The development of alkali-activated mixtures for soil stabilisation. In Handbook of Alkali-Activated Cements, Mortars and Concretes; Elsevier BV: Amsterdam, The Netherlands, 2015; pp. 555–604. [Google Scholar]
- Rios, S.; Ramos, C.; Da Fonseca, A.V.; Cruz, N.; Rodrigues, C. Mechanical and durability properties of a soil stabilised with an alkali-activated cement. Eur. J. Environ. Civ. Eng. 2017, 23, 245–267. [Google Scholar] [CrossRef]
- Cristelo, N.; Glendinning, S.; Pinto, A.T. Deep soft soil improvement by alkaline activation. Proc. Inst. Civil Eng.-Ground Improv. 2011, 164, 73–82. [Google Scholar] [CrossRef]
- Cristelo, N.; Glendinning, S.; Fernandes, L.S.G.; Pinto, A.T. Effects of alkaline-activated fly ash and Portland cement on soft soil stabilisation. Acta Geotech. 2013, 8, 395–405. [Google Scholar] [CrossRef]
- Phummiphan, I.; Horpibulsuk, S.; Sukmak, P.; Chinkulkijniwat, A.; Arulrajah, A.; Shen, S.-L. Stabilisation of marginal lateritic soil using high calcium fly ash-based geopolymer. Road Mater. Pavement Des. 2016, 17, 877–891. [Google Scholar] [CrossRef]
- Phetchuay, C.; Horpibulsuk, S.; Arulrajah, A.; Suksiripattanapong, C.; Udomchai, A. Strength development in soft marine clay stabilized by fly ash and calcium carbide residue based geopolymer. Appl. Clay Sci. 2016, 127, 134–142. [Google Scholar] [CrossRef]
- Abdullah, H.H.; Shahin, M.A.; Walske, M.L.; Karrech, A. Systematic approach to assessing the applicability of fly-ash-based geopolymer for clay stabilization. Can. Geotech. J. 2019, 1–37. [Google Scholar] [CrossRef]
- Das, B.M. Ground Improvement. In Geotechnical Engineering Handbook; Petry, T., Ed.; J. Ross Publishing: Plantation, FL, USA, 2010; p. 9 (1ߝ37). [Google Scholar]
- Pacheco-Torgal, F.; Castro-Gomes, J.; Jalali, S. Alkali-activated binders: A review. Part 2. About materials and binders manufacture. Constr. Build. Mater. 2008, 22, 1315–1322. [Google Scholar] [CrossRef]
- Shi, C.; Fernandez-Jiménez, A.; Palomo, A. New cements for the 21st century: The pursuit of an alternative to Portland cement. Cem. Concr. Res. 2011, 41, 750–763. [Google Scholar] [CrossRef]
- Cristelo, N.; Glendinning, S.; Fernandes, L.S.G.; Pinto, A.T. Effect of calcium content on soil stabilisation with alkaline activation. Constr. Build. Mater. 2012, 29, 167–174. [Google Scholar] [CrossRef]
- Sargent, P.; Hughes, P.N.; Rouainia, M.; White, M.L. The use of alkali activated waste binders in enhancing the mechanical properties and durability of soft alluvial soils. Eng. Geol. 2013, 152, 96–108. [Google Scholar] [CrossRef]
- Zhang, M.; Guo, H.; El-Korchi, T.; Zhang, G.; Tao, M. Experimental feasibility study of geopolymer as the next-generation soil stabilizer. Constr. Build. Mater. 2013, 47, 1468–1478. [Google Scholar] [CrossRef]
- Zhang, M.; Zhao, M.; Zhang, G.; Nowak, P.; Coen, A.; Tao, M. Calcium-free geopolymer as a stabilizer for sulfate-rich soils. Appl. Clay Sci. 2015, 108, 199–207. [Google Scholar] [CrossRef]
- Rios, S.; Cristelo, N.; Da Fonseca, A.V.; Ferreira, C. Structural performance of alkali-activated soil ash versus soil cement. J. Mater. Civ. Eng. 2016, 28, 04015125. [Google Scholar] [CrossRef]
- Rios, S.; Cristelo, N.; Da Fonseca, A.V.; Ferreira, C. Stiffness behavior of soil stabilized with alkali-activated fly ash from small to large strains. Int. J. Géoméch. 2017, 17, 04016087. [Google Scholar] [CrossRef]
- Phummiphan, I.; Horpibulsuk, S.; Rachan, R.; Arulrajah, A.; Shen, S.-L.; Chindaprasirt, P. High calcium fly ash geopolymer stabilized lateritic soil and granulated blast furnace slag blends as a pavement base material. J. Hazard. Mater. 2018, 341, 257–267. [Google Scholar] [CrossRef]
- Liu, Z.; Cai, C.; Liu, F.; Fan, F. Feasibility Study of Loess Stabilization with Fly Ash–Based Geopolymer. J. Mater. Civ. Eng. 2016, 28, 04016003. [Google Scholar] [CrossRef]
- Yip, C.; Lukey, G.; Van Deventer, J. The coexistence of geopolymeric gel and calcium silicate hydrate at the early stage of alkaline activation. Cem. Concr. Res. 2005, 35, 1688–1697. [Google Scholar] [CrossRef]
- Singhi, B.; Laskar, A.I.; Ahmed, M.A. Investigation on soil–geopolymer with slag, fly ash and their blending. Arab. J. Sci. Eng. 2015, 41, 393–400. [Google Scholar] [CrossRef]
- Abdullah, H.; Shahin, M.A.; Sarker, P. Use of fly-ash geopolymer incorporating ground granulated slag for stabilisation of kaolin clay cured at ambient temperature. Geotech. Geol. Eng. 2018, 37, 721–740. [Google Scholar] [CrossRef]
- Abdullah, H.H.; Shahin, M.A.; Walske, M.L. Geo-mechanical behavior of clay soils stabilized at ambient temperature with fly-ash geopolymer-incorporated granulated slag. Soils Found. 2019, 59, 1906–1920. [Google Scholar] [CrossRef]
- Komnitsas, K.; Zaharaki, D. Geopolymerisation: A review and prospects for the minerals industry. Miner. Eng. 2007, 20, 1261–1277. [Google Scholar] [CrossRef]
- Davidovits, J. Geopolymer Chemistry and Applications, 2nd ed.; Institut Géopolymère: Saint-Quentin, France, 2008; pp. 1–585. [Google Scholar]
- Provis, J.L.; van Deventer, J.S.J. Introduction to Geopolymers. In Geopolymers: Structure, Processing, Properties and Industrial Applications; Provis, J., Deventer, J., Eds.; Woodhead Publishing: Abingdon, UK, 2009. [Google Scholar]
- Hardjito, D. Studies on Fly Ash-based Geopolymer Concrete. Ph.D. Thesis, Curtin University of Technology, Perth, Australia, August 2005. [Google Scholar]
- Rios, S.; Cristelo, N.; Miranda, T.; Araújo, N.; Oliveira, J.; Lucas, E. Increasing the reaction kinetics of alkali-activated fly ash binders for stabilisation of a silty sand pavement sub-base. Road Mater. Pavement Des. 2016, 19, 201–222. [Google Scholar] [CrossRef]
- Little, D.; Nair, S. Recommended Practice for Stabilization of Subgqrade Soils and Base Materials; National Cooperative Highway Research Program, Transportation Research Board of the National Academies: Washington, DC, USA, 2009. [Google Scholar]
- Khale, D.; Chaudhary, R. Mechanism of geopolymerization and factors influencing its development: A review. J. Mater. Sci. 2007, 42, 729–746. [Google Scholar] [CrossRef]
- Duxson, P. Geopolymer precursor design. In Geopolymers; Elsevier BV: Amsterdam, The Netherlands, 2009; pp. 37–49. [Google Scholar]
- Tenepalli, J.S.; Neeraja, D.; Sai, T.J. Properties of class F fly ash based Geopolymer mortar produced with alkaline water. J. Build. Eng. 2018, 19, 42–48. [Google Scholar] [CrossRef]
- Van Jaarsveld, J.G.S.; Van Deventer, J.S.J. Effect of the alkali metal activator on the properties of fly ash-based Geopolymers. Ind. Eng. Chem. Res. 1999, 38, 3932–3941. [Google Scholar] [CrossRef]
- Xu, H.; Van Deventer, J. The geopolymerisation of alumino-silicate minerals. Int. J. Miner. Process. 2000, 59, 247–266. [Google Scholar] [CrossRef]
- Provis, J.L. Activating solution chemistry for Geopolymers. In Geopolymers: Structure, Processing, Properties and Industrial Applications; Provis, J., Deventer, J., Eds.; Woodhead Publishing: Abingdon, UK, 2009; pp. 50–71. [Google Scholar]
- Duxson, P.; Fernandez-Jiménez, A.; Provis, J.L.; Lukey, G.C.; Palomo, A.; Van Deventer, J.S.J. Geopolymer technology: the current state of the art. J. Mater. Sci. 2006, 42, 2917–2933. [Google Scholar] [CrossRef]
- Wilkinson, A.; Haque, A.; Kodikara, J. Stabilisation of clayey soils with industrial by-products: Part B. Proc. Inst. Civil Eng.-Ground Improv. 2010, 163, 165–172. [Google Scholar] [CrossRef]
- Markou, I.; Atmatzidis, D.K. Development of a pulverized fly ash suspension grout. Geotech. Geol. Eng. 2002, 20, 123–147. [Google Scholar] [CrossRef]
- Mechanisms of soil-lime stabilization. Available online: http://onlinepubs.trb.org/onlinepubs/hrr/1965/92/92-006.pdf (accessed on 29 May 2020).
- Reactions accompanying stabilization of clay with cement. Available online: http://onlinepubs.trb.org/Onlinepubs/hrr/1963/36/36-008.pdf (accessed on 29 May 2020).
- Corrêa-Silva, M.; Araujo, N.; Cristelo, N.; Miranda, T.; Gomes, A.T.; Coelho, J. Improvement of a clayey soil with alkali activated low-calcium fly ash for transport infrastructures applications. Road Mater. Pavement Des. 2018, 20, 1912–1926. [Google Scholar] [CrossRef]
- Standard Test Methods for Wetting and Drying Compacted Soil-Cement Mixtures. Available online: https://www.astm.org/Standards/D559 (accessed on 29 May 2020).
- Standard Test Methods for Freezing and Thawing Compacted Soil-Cement Mixtures. Available online: https://www.astm.org/Standards/D560 (accessed on 29 May 2020).
- Rethinking Cement. Available online: https://bze.org.au/research/manufacturing-industrial-processes/rethinking-cement/ (accessed on 29 May 2020).
- Palomo, A.; Grutzeck, M.; Blanco, M. Alkali-activated fly ashes. Cem. Concr. Res. 1999, 29, 1323–1329. [Google Scholar] [CrossRef]
- Rovnaník, P. Effect of curing temperature on the development of hard structure of metakaolin-based geopolymer. Constr. Build. Mater. 2010, 24, 1176–1183. [Google Scholar] [CrossRef]
- Sindhunata; Van Deventer, J.S.J.; Lukey, G.C.; Xu, H. Effect of Curing Temperature and Silicate Concentration on Fly-Ash-Based Geopolymerization. Ind. Eng. Chem. Res. 2006, 45, 3559–3568. [Google Scholar] [CrossRef]
- Van Jaarsveld, J.; Van Deventer, J.; Lukey, G. The effect of composition and temperature on the properties of fly ash- and kaolinite-based geopolymers. Chem. Eng. J. 2002, 89, 63–73. [Google Scholar] [CrossRef]
- Temuujin, J.; Williams, R.; Van Riessen, A. Effect of mechanical activation of fly ash on the properties of geopolymer cured at ambient temperature. J. Mater. Process. Technol. 2009, 209, 5276–5280. [Google Scholar] [CrossRef]
- Gianoncelli, A.; Zacco, A.; Struis, R.P.W.J.; Borgese, L.; Depero, L.E.; Bontempi, E. Fly Ash Pollutants, Treatment and Recycling. In Advanced Nanostructured Materials for Environmental Remediation; Springer Science and Business Media LLC: Berlin, Germany, 2013; Volume 4, pp. 103–213. [Google Scholar]
- Deb, P.S.; Nath, P.; Sarker, P. The effects of ground granulated blast-furnace slag blending with fly ash and activator content on the workability and strength properties of geopolymer concrete cured at ambient temperature. Mater. Des. 2014, 62, 32–39. [Google Scholar] [CrossRef]
- Deb, P.S.; Nath, P.; Sarker, P. Properties of fly ash and slag blended geopolymer concrete cured at ambient temperature. In Proceedings of the Seventh International Structural Engineering and Construction Conference, Honolulu, HI, USA, 18–23 June 2013; pp. 1–6. [Google Scholar]
- Granizo, M.L.; Alonso, S.; Palomo, A.; Blanco-Varela, M.T. Alkaline Activation of Metakaolin: Effect of Calcium Hydroxide in the Products of Reaction. J. Am. Ceram. Soc. 2004, 85, 225–231. [Google Scholar] [CrossRef]
- Lodeiro, I.G.; Fernandez-Jiménez, A.; Palomo, A. Variation in hybrid cements over time. Alkaline activation of fly ash–portland cement blends. Cem. Concr. Res. 2013, 52, 112–122. [Google Scholar] [CrossRef]
- Middleton, G.F.; Schneider, L.M. Earth-wall Construction; Commonwealth Scientific and Industrial Research Organisation (Division of Building Construction and Engineering): Canberra, Australia, 1987. [Google Scholar]
- USACE. Engineering and Design: Soil Stabilization for Pavements Mobilization Construction; Engineer Manual No. 1110-3-137; Department of the US Army-Corps of Engineers: Washington, DC, USA, 1984.
| Soil Type | Plasticity Index (%) | Source Material (SM) | Activator Chemicals | SM/(SM + Soil) (%) | Source Material/Soil (%) | Activator/(SM + Soil) (%) | Activator/Source Material (%) | NaOH Molarity or NaOH:H2O | Na2SiO3:NaOH (mass) | Curing Temperature (°C) | Tests Conducted on Stabilised Soils | Reference | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compaction | Peak UCS at 28 days (MPa) | Triaxial | Durability | Microstructure | ||||||||||||
| Sandy Clay | 21.9 | FAF | NaOH + Na2SiO3 | 20–40 | - | 40–50 | - | 10–15 | 2 | ambient | - | 11.4 | - | - | - | [13] |
| Fat Clay | 32.2 | FAC & FAF | NaOH + Na2SiO3 | 10–20 | - | 40 | - | 10 | 2 | 19–23 | - | 1.2–1.8 | - | - | ✓ | [21] |
| Sandy Clay | 21.9 | FAF | NaOH + Na2SiO3 | 20–40 | - | 40–50 | - | 10–15 | 2 | 19–23 | - | 10 | - | - | ✓ | [14] |
| Silty Sand | 12 | FA | NaOH + Na2SiO3 | - | 10 | - | 50 | - | 2 | 20 | - | 0.6 | - | ✓ | - | [22] |
| FA and GGBS | NaOH + Na2SiO3 | 3.5 | ||||||||||||||
| Lean Clay | 15 | MK | NaOH + Na2SiO3 | 3–15 | - | - | - | - | - | 23 | - | 3.8 | - | - | ✓ | [23] |
| Lean Clay | 15 | MK | NaOH + Na2SiO3 | 8,13 | - | - | - | - | - | ambient | - | 4.0 | - | - | ✓ | [24] |
| Clay | 14 | FAC | NaOH + Na2SiO3 | - | 4–20 | - | 45–85 | 12–14.5 | varies | ambient | ✓ | 0.2 | - | - | - | [30] |
| FAC and GGBS | 10.5 | |||||||||||||||
| Silty Clay | 33 | FAF and CCR | NaOH + Na2SiO3 | - | 24–45 | - | 50–200 | 3–18 | 1.5–9 | 25–40 | - | 1.2 | - | - | - | [16] |
| Loess | 13.7 | FAF | NaOH + Na2SiO3 | 10–30 | - | 40 | - | - | - | 23 | - | 4.5 | - | - | ✓ | [28] |
| KOH + Na2SiO3 | 7 | |||||||||||||||
| Silty clayey sand | 6 | FAC | NaOH + Na2SiO3 | - | - | 15.7–19 | - | - | 100:0–50:50 | 27–30 | ✓ | 11 | - | - | - | [15] |
| Silty sand | NP | FAF | NaOH + Na2SiO3 | 15–25 | - | 11–19 | - | 7.5 | 0.5 | 20 | ✓ | 2.3 | ✓ | - | ✓ | [25,26] |
| Silty sand | NP | FAF | NaOH + Na2SiO3 | 10–20 | - | 8–8.8 | - | 5–12.5 | 0.5–1 | 20 | ✓ | 4.2 | ✓ | ✓ | ✓ | [12] |
| Silty clayey sand | 6 | FAC | NaOH + Na2SiO3 | 30 | - | 9.6–17 | - | 5 | 0.66–9 | 27–30 | ✓ | 9 | - | - | ✓ | [27] |
| FAC and GGBFS | 40–60 | 16 | ||||||||||||||
| Clay soils | 26–98 | FAC and GGBFS | NaOH + Na2SiO3 | 10–30 | - | - | 40 | 14 | 2.33 | ambient | ✓ | 3.2 | ✓ | ✓ | ✓ | [17,31,32] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdullah, H.H.; Shahin, M.A.; Walske, M.L. Review of Fly-Ash-Based Geopolymers for Soil Stabilisation with Special Reference to Clay. Geosciences 2020, 10, 249. https://doi.org/10.3390/geosciences10070249
Abdullah HH, Shahin MA, Walske ML. Review of Fly-Ash-Based Geopolymers for Soil Stabilisation with Special Reference to Clay. Geosciences. 2020; 10(7):249. https://doi.org/10.3390/geosciences10070249
Chicago/Turabian StyleAbdullah, Hayder H., Mohamed A. Shahin, and Megan L. Walske. 2020. "Review of Fly-Ash-Based Geopolymers for Soil Stabilisation with Special Reference to Clay" Geosciences 10, no. 7: 249. https://doi.org/10.3390/geosciences10070249
APA StyleAbdullah, H. H., Shahin, M. A., & Walske, M. L. (2020). Review of Fly-Ash-Based Geopolymers for Soil Stabilisation with Special Reference to Clay. Geosciences, 10(7), 249. https://doi.org/10.3390/geosciences10070249





