Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae in Hospitalized Neonatal Foals: Prevalence, Risk Factors for Shedding and Association with Infection
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Equine Study Population, Study Design and Sampling Methods
2.2. Demographic and Medical Data
2.3. ESBL-Producing Enterobacteriaceae (ESBL-E) Isolation and Species Identification
2.4. Molecular Characterization of ESBL-E
2.5. Statistical Analysis
3. Results
3.1. Characterization of Equine Study Population
3.2. Hospital Procedures, Antibiotic Therapy and Outcome
3.3. Prevalence of ESBL-E Shedding among Foals and Mares
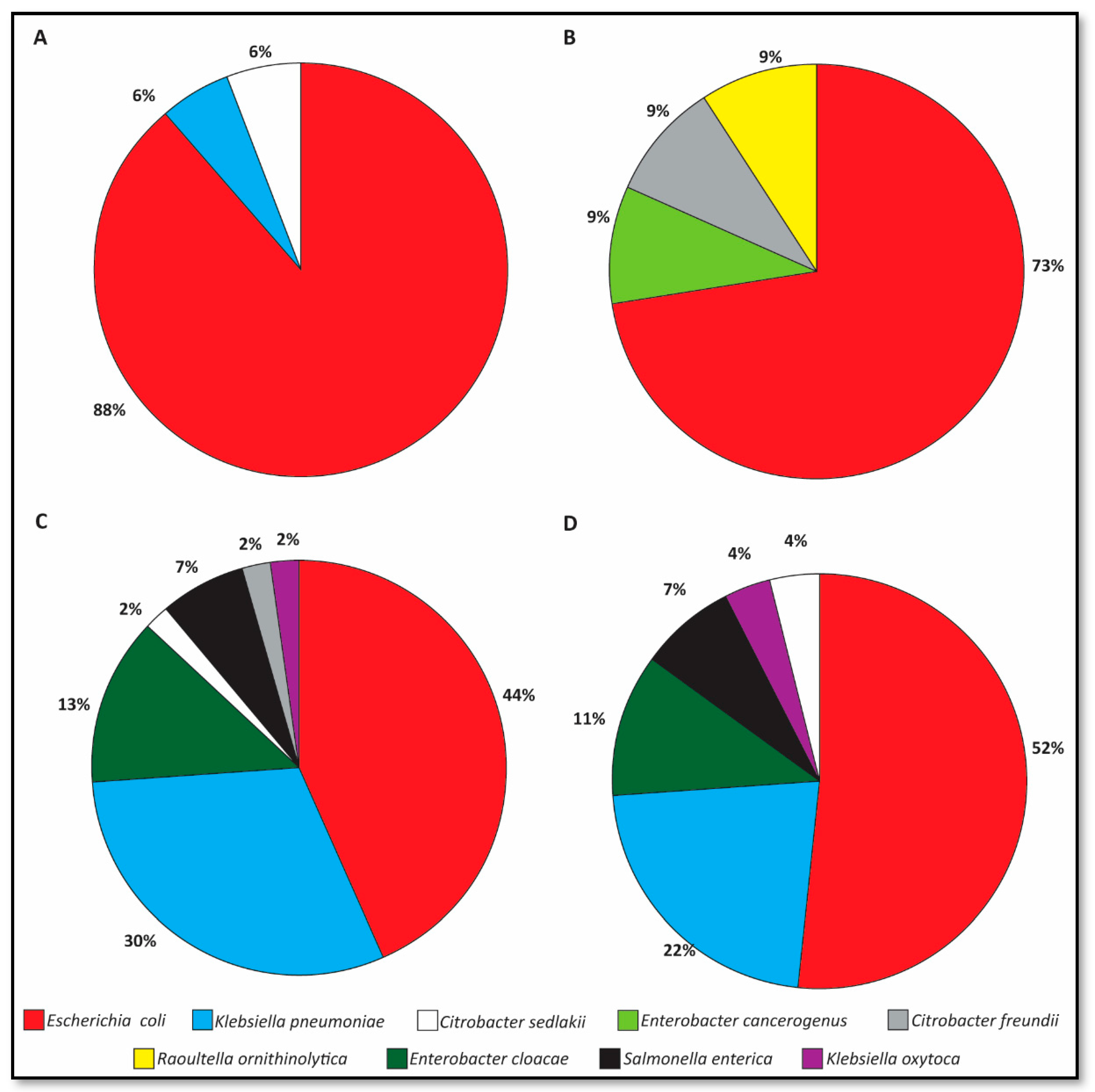
3.4. Species Distribution of ESBL-E Shedding Isolates
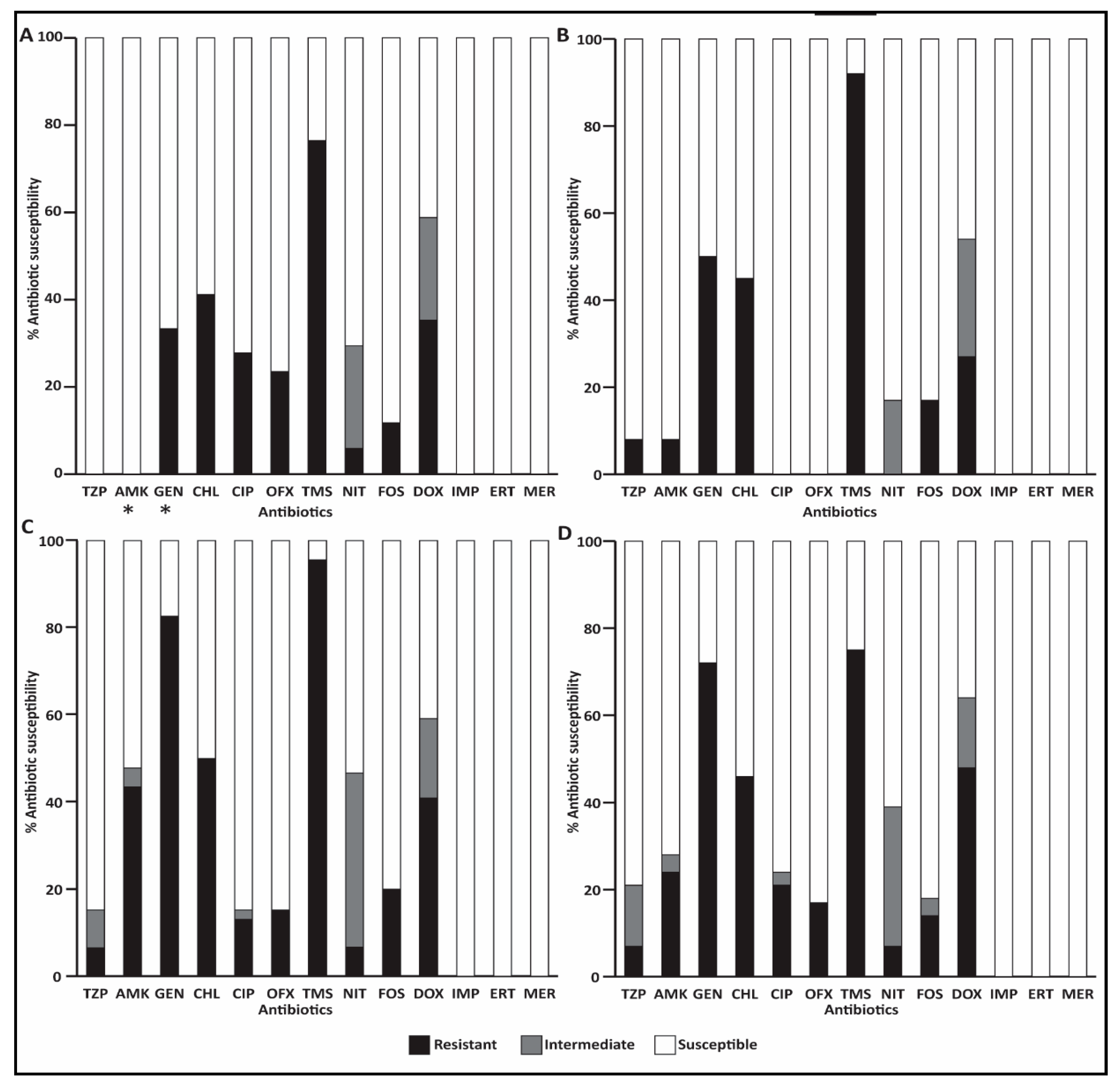
3.5. Antibiotic Susceptibility Profiles of ESBL-E Fecal Isolates
3.5.1. ESBL-E Isolates on Admission
3.5.2. ESBL-E Isolates during Hospitalization
3.5.3. ESBL-E Isolates from Clinical Samples of Infection Sites
3.6. Risk Factor Analysis for ESBL-E Shedding
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Rubin, J.E.; Pitout, J.D.D. Extended-spectrum β-lactamase, carbapenemase and AmpC producing Enterobacteriaceae in companion animals. Vet. Microbiol. 2014, 170, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Bae, I.K.; Lee, S.H. New definitions of extended-spectrum β-lactamase conferring worldwide emerging antibiotic resistance. Med. Res. Rev. 2012, 32, 216–232. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Thungrat, K.; Boothe, D.M. Occurrence of OXA-48 Carbapenemase and Other β-Lactamase Genes in ESBL-Producing Multidrug Resistant Escherichia coli from Dogs and Cats in the United States, 2009–2013. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Denkel, L.A.; Gastmeier, P.; Piening, B. To screen or not to screen mothers of preterm infants for extended-spectrum beta-lactamase-producing Enterobacteriaceae (ESBL-E). J. Perinatol. 2015, 35, 893–894. [Google Scholar] [CrossRef] [PubMed]
- Schwaber, M.J.; Navon-Venezia, S.; Kaye, K.S.; Ben-Ami, R.; Schwartz, D.; Carmeli, Y. Clinical and economic impact of bacteremia with extended- spectrum-beta-lactamase-producing Enterobacteriaceae. Antimicrob. Agents Chemother. 2006, 50, 1257–1262. [Google Scholar] [CrossRef] [PubMed]
- Bert, F.; Larroque, B.; Paugam-Burtz, C.; Dondero, F.; Durand, F.; Marcon, E.; Belghiti, J.; Moreau, R.; Nicolas-Chanoine, M.-H. Pretransplant Fecal Carriage of Extended-Spectrum β-Lactamase–producing Enterobacteriaceae and Infection after Liver Transplant, France. Emerg. Infect. Dis. 2012, 18, 908–916. [Google Scholar] [CrossRef]
- Weese, J.S. Antimicrobial use and antimicrobial resistance in horses. Equine Vet. J. 2015, 47, 747–749. [Google Scholar] [CrossRef]
- Dolejska, M.; Duskova, E.; Rybarikova, J.; Janoszowska, D.; Roubalova, E.; Dibdakova, K.; Maceckova, G.; Kohoutova, L.; Literak, I.; Smola, J.; et al. Plasmids carrying blaCTX-M-1 and qnr genes in Escherichia coli isolates from an equine clinic and a horseback riding centre. J. Antimicrob. Chemother. 2011, 66, 757–764. [Google Scholar] [CrossRef]
- Walther, B.; Klein, K.-S.; Barton, A.-K.; Semmler, T.; Huber, C.; Wolf, S.A.; Tedin, K.; Merle, R.; Mitrach, F.; Guenther, S.; et al. Extended-spectrum beta-lactamase (ESBL)-producing Escherichia coli and Acinetobacter baumannii among horses entering a veterinary teaching hospital: The contemporary “Trojan Horse”. PLoS ONE 2018, 13, e0191873. [Google Scholar] [CrossRef]
- Wohlfender, F.D.; Barrelet, F.E.; Doherr, M.G.; Straub, R.; Meier, H.P. Diseases in neonatal foals. Part 1: the 30 day incidence of disease and the effect of prophylactic antimicrobial drug treatment during the first three days post partum. Equine Vet. J. 2009, 41, 179–185. [Google Scholar] [CrossRef]
- Theelen, M.J.P.; Wilson, W.D.; Edman, J.M.; Magdesian, K.G.; Kass, P.H. Temporal trends in prevalence of bacteria isolated from foals with sepsis: 1979-2010. Equine Vet. J. 2014, 46, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Dyakova, E.; Bisnauthsing, K.N.; Querol-Rubiera, A.; Patel, A.; Ahanonu, C.; Tosas Auguet, O.; Edgeworth, J.D.; Goldenberg, S.D.; Otter, J.A. Efficacy and acceptability of rectal and perineal sampling for identifying gastrointestinal colonization with extended spectrum β-lactamase Enterobacteriaceae. Clin. Microbiol. Infect. 2017, 23, 577.e1–577.e3. [Google Scholar] [CrossRef] [PubMed]
- Brewer, B.D.; Koterba, A.M.; Carter, R.L.; Rowe, E.D. Comparison of empirically developed sepsis score with a computer generated and weighted scoring system for the identification of sepsis in the equine neonate. Equine Vet. J. 1988, 20, 23–24. [Google Scholar] [CrossRef] [PubMed]
- Oreff, G.L.; Tatz, A.J.; Dahan, R.; Segev, G.; Berlin, D.; Kelmer, G. Surgical management and long-term outcome of umbilical infection in 65 foals (2010-2015). Vet. Surg. 2017, 46, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Murk, J.-L.A.N.; Heddema, E.R.; Hess, D.L.J.; Bogaards, J.A.; Vandenbroucke-Grauls, C.M.J.E.; Debets-Ossenkopp, Y.J. Enrichment broth improved detection of extended-spectrum-beta-lactamase-producing bacteria in throat and rectal surveillance cultures of samples from patients in intensive care units. J. Clin. Microbiol. 2009, 47, 1885–1887. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing, 26th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2016. [Google Scholar]
- Falagas, M.E.; Karageorgopoulos, D.E. Pandrug Resistance (PDR), Extensive Drug Resistance (XDR), and Multidrug Resistance (MDR) among Gram-Negative Bacilli: Need for International Harmonization in Terminology. Clin. Infect. Dis. 2008, 46, 1121–1122. [Google Scholar] [CrossRef]
- Woodford, N.; Fagan, E.J.; Ellington, M.J. Multiplex PCR for rapid detection of genes encoding CTX-M extended-spectrum β-lactamases. J. Antimicrob. Chemother. 2006, 57, 154–155. [Google Scholar] [CrossRef]
- Lin, S.-P.; Liu, M.-F.; Lin, C.-F.; Shi, Z.-Y. Phenotypic detection and polymerase chain reaction screening of extended-spectrum β-lactamases produced by Pseudomonas aeruginosa isolates. J. Microbiol. Immunol. Infect. 2012, 45, 200–207. [Google Scholar] [CrossRef]
- Tofteland, S.; Haldorsen, B.; Dahl, K.H.; Simonsen, G.S.; Steinbakk, M.; Walsh, T.R.; Sundsfjord, A.; Norwegian ESBL Study Group. Effects of phenotype and genotype on methods for detection of extended-spectrum-beta-lactamase-producing clinical isolates of Escherichia coli and Klebsiella pneumoniae in Norway. J. Clin. Microbiol. 2007, 45, 199–205. [Google Scholar] [CrossRef]
- Johnson, J.R.; Clermont, O.; Johnston, B.; Clabots, C.; Tchesnokova, V.; Sokurenko, E.; Junka, A.F.; Maczynska, B.; Denamur, E. Rapid and Specific Detection, Molecular Epidemiology, and Experimental Virulence of the O16 Subgroup within Escherichia coli Sequence Type 131. J. Clin. Microbiol. 2014, 52, 1358–1365. [Google Scholar] [CrossRef]
- Versalovic, J.; Koeuth, T.; Lupski, J.R. Distribution of repetitive DNA sequences in eubacteria and application to fingerprinting of bacterial genomes. Nucleic Acids Res. 1991, 19, 6823–6831. [Google Scholar] [CrossRef] [PubMed]
- Heras, J.; Domínguez, C.; Mata, E.; Pascual, V.; Lozano, C.; Torres, C.; Zarazaga, M. GelJ – a tool for analyzing DNA fingerprint gel images. BMC Bioinforma. 2015, 16, 270. [Google Scholar] [CrossRef] [PubMed]
- Herzog, K.A.T.; Schneditz, G.; Leitner, E.; Feierl, G.; Hoffmann, K.M.; Zollner-Schwetz, I.; Krause, R.; Gorkiewicz, G.; Zechner, E.L.; Högenauer, C. Genotypes of Klebsiella oxytoca isolates from patients with nosocomial pneumonia are distinct from those of isolates from patients with antibiotic-associated hemorrhagic colitis. J. Clin. Microbiol. 2014, 52, 1607–1616. [Google Scholar] [CrossRef] [PubMed]
- Wirth, T.; Falush, D.; Lan, R.; Colles, F.; Mensa, P.; Wieler, L.H.; Karch, H.; Reeves, P.R.; Maiden, M.C.J.; Ochman, H.; et al. Sex and virulence in Escherichia coli: an evolutionary perspective. Mol. Microbiol. 2006, 60, 1136–1151. [Google Scholar] [CrossRef] [PubMed]
- Diancourt, L.; Passet, V.; Verhoef, J.; Grimont, P.A.D.; Brisse, S. Multilocus Sequence Typing of Klebsiella pneumoniae Nosocomial Isolates. J. Clin. Microbiol. 2005, 43, 4178–4182. [Google Scholar] [CrossRef]
- Palmer, J. Update on the management of neonatal sepsis in horses. Vet. Clin. North Am. Equine Pract. 2014, 30, 317–336. [Google Scholar] [CrossRef]
- McHugh, M.L. Interrater reliability: the kappa statistic. Biochem. Med. 2012, 22, 276–282. [Google Scholar] [CrossRef]
- Maddox, T.W.; Clegg, P.D.; Diggle, P.J.; Wedley, A.L.; Dawson, S.; Pinchbeck, G.L.; Williams, N.J. Cross-sectional study of antimicrobial-resistant bacteria in horses. Part 1: Prevalence of antimicrobial-resistant Escherichia coli and methicillin-resistant Staphylococcus aureus. Equine Vet. J. 2012, 44, 289–296. [Google Scholar] [CrossRef]
- Maddox, T.W.; Williams, N.J.; Clegg, P.D.; O’Donnell, A.J.; Dawson, S.; Pinchbeck, G.L. Longitudinal study of antimicrobial-resistant commensal Escherichia coli in the faeces of horses in an equine hospital. Prev. Vet. Med. 2011, 100, 134–145. [Google Scholar] [CrossRef]
- Paterson, D.L.; Bonomo, R.A. Extended-Spectrum β-Lactamases: a Clinical Update. Clin. Microbiol. Rev. 2005, 18, 657–686. [Google Scholar] [CrossRef]
- Adler, A.; Sturlesi, N.A.; Fallach, N.; Zilberman-Barzilai, D.; Hussein, O.; Blum, S.E.; Klement, E.; Schwaber, M.J.; Carmeli, Y. Prevalence, Risk Factors, and Transmission Dynamics of Extended-Spectrum-β-Lactamase-Producing Enterobacteriaceae: a National Survey of Cattle Farms in Israel in 2013. J. Clin. Microbiol. 2015, 53, 3515–3521. [Google Scholar] [PubMed]
- Damborg, P.; Marskar, P.; Baptiste, K.E.; Guardabassi, L. Faecal shedding of CTX-M-producing Escherichia coli in horses receiving broad-spectrum antimicrobial prophylaxis after hospital admission. Vet. Microbiol. 2012, 154, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Olivo, G.; Lucas, T.M.; Borges, A.S.; Silva, R.O.S.; Lobato, F.C.F.; Siqueira, A.K.; da Silva Leite, D.; Brandão, P.E.; Gregori, F.; de Oliveira-Filho, J.P.; et al. Enteric Pathogens and Coinfections in Foals with and without Diarrhea. BioMed Res. Int. 2016, 2016, 1512690. [Google Scholar] [CrossRef] [PubMed]
- Barceló Oliver, F.; Russell, T.M.; Uprichard, K.L.; Neil, K.M.; Pollock, P.J. Treatment of septic arthritis of the coxofemoral joint in 12 foals. Vet. Surg. 2017, 46, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Ernst, N.S.; Hernandez, J.A.; MacKay, R.J.; Brown, M.P.; Gaskin, J.M.; Nguyen, A.D.; Giguere, S.; Colahan, P.T.; Troedsson, M.R.; Haines, G.R.; et al. Risk factors associated with fecal Salmonella shedding among hospitalized horses with signs of gastrointestinal tract disease. J. Am. Vet. Med. Assoc. 2004, 225, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Denkel, L.A.; Schwab, F.; Kola, A.; Leistner, R.; Garten, L.; von Weizsäcker, K.; Geffers, C.; Gastmeier, P.; Piening, B. The mother as most important risk factor for colonization of very low birth weight (VLBW) infants with extended-spectrum β-lactamase-producing Enterobacteriaceae (ESBL-E). J. Antimicrob. Chemother. 2014, 69, 2230–2237. [Google Scholar] [CrossRef] [PubMed]
- Callens, B.; Faes, C.; Maes, D.; Catry, B.; Boyen, F.; Francoys, D.; de Jong, E.; Haesebrouck, F.; Dewulf, J. Presence of antimicrobial resistance and antimicrobial use in sows are risk factors for antimicrobial resistance in their offspring. Microb. Drug Resist. 2015, 21, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Simonsen, K.A.; Anderson-Berry, A.L.; Delair, S.F.; Davies, H.D. Early-Onset Neonatal Sepsis. Clin. Microbiol. Rev. 2014, 27, 21–47. [Google Scholar] [CrossRef]
- Li, X.; Xu, X.; Yang, X.; Luo, M.; Liu, P.; Su, K.; Qing, Y.; Chen, S.; Qiu, J.; Li, Y. Risk factors for infection and/or colonisation with extended-spectrum β-lactamase-producing bacteria in the neonatal intensive care unit: a meta-analysis. Int. J. Antimicrob. Agents 2017, 50, 622–628. [Google Scholar] [CrossRef]
| Pathology | No. of Horses (%) | |
|---|---|---|
| On Admission | Developed during Hospitalization | |
| Foals | n = 55 | n = 33 |
| Diarrhea | 13 (24) | 4 (12) |
| Umbilical infection | 13 (24) | 0 |
| Sepsis | 12 (22) | 0 |
| Prematurity | 10 (17) | 0 |
| Septic polyarthritis | 9 (16) | 0 |
| Orthopedic problems (other than septic polyarthritis) | 9 (15) | 1 (3) |
| Perinatal Asphyxia Syndrome (PAS) | 8 (13) | 0 |
| Respiratory problems | 6 (11) | 0 |
| Colic | 6 (10) | 1 (3) |
| Injury | 3 (5) | 0 |
| Neurological signs (other than PAS) | 1 (2) | 4 (12) |
| Uroperitoneum | 1 (2) | 1 (3) |
| Phlebitis | 0 | 1 (3) |
| Uveitis | 0 | 2 (6) |
| Peritonitis | 0 | 1 (3) |
| Other (hernia, guttural pouch tympany and piroplasmosis) | 3 (5) | 0 |
| Mares | n = 55 | n = 33 |
| Colic | 2 (4) | 2 (6) |
| Retained placenta | 2 (4) | 0 |
| Injury | 1 (2) | 0 |
| Orthopedic syndromes | 1 (2) | 0 |
| Placentitis | 1 (2) | 0 |
| Colitis | 0 | 1 (3) |
| on Admission 1 | ≥ 72 h of Hospitalization 2 | |||||
|---|---|---|---|---|---|---|
| Horses | Shedding (%) | Total No. of ESBL-E Isolates | blaESBL Genes (%) 3 | (%)Shedding | Total No. of ESBL-E Isolates | blaESBL Genes (%) 3 |
| Foals | 18/55 (33) (95% CI 21–47) | 18 | BlaCTXM-1: 14/19 (74) BlaCTXM-9: 2/19 (11) | 28/33 (85) 7 (95% CI 70–94) | 46 4 | CTX-M-1: 31/46 (67) CTX-M-2: 1/46 (2) OXA-1: 3/46 (7) |
| Mares | 9/55 (16) (95% CI 8–29) | 115 | BlaCTXM-1: 6/11 (55) | 19/33 (58) 8 (95% CI 40–73) | 27 6 | CTX-M-1: 16/27 (59) CTX-M-2: 1/27 (4) CTX-M-9: 2/27 (7) TEM-163: 2/27 (7) |
| Foal | Age on Admission | ESBL-E Shedding Status | Clinical ESBL-E Infection | |||||
|---|---|---|---|---|---|---|---|---|
| 1st Admission | 1st Hospitalization | 2nd Admission | 2nd Hospitalization | ESBL-E Species | Source | Outcome | ||
| 1 | <12 h | Negative | E. coli ST88 | K. pneumoniae ST1552 | Not sampled | K. pneumoniae ST1552 | abscess 1 | Discharged |
| 2 | <12 h | Negative | E. coli ST38 | E. coli ST86 K. oxytoca ST194 S. enterica | Enterobacter cloacae | E. coli ST746 | umbilicus 2 | Euthanized |
| E. coli ST746 | wound | |||||||
| 3 | <12 h | Negative | E. coli ST746 K. pneumoniae ST37 | No second hospitalization | E. coli ST746 K. pneumoniae ST585 | umbilicus 3 | Euthanized | |
| S. enterica | ||||||||
| 4 | 17 d | E. coli4 | Discharged | No second hospitalization | E. coli ST69 | wound 5 | Discharged | |
| E. coli ST69 | umbilicus | |||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shnaiderman-Torban, A.; Paitan, Y.; Arielly, H.; Kondratyeva, K.; Tirosh-Levy, S.; Abells-Sutton, G.; Navon-Venezia, S.; Steinman, A. Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae in Hospitalized Neonatal Foals: Prevalence, Risk Factors for Shedding and Association with Infection. Animals 2019, 9, 600. https://doi.org/10.3390/ani9090600
Shnaiderman-Torban A, Paitan Y, Arielly H, Kondratyeva K, Tirosh-Levy S, Abells-Sutton G, Navon-Venezia S, Steinman A. Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae in Hospitalized Neonatal Foals: Prevalence, Risk Factors for Shedding and Association with Infection. Animals. 2019; 9(9):600. https://doi.org/10.3390/ani9090600
Chicago/Turabian StyleShnaiderman-Torban, Anat, Yossi Paitan, Haia Arielly, Kira Kondratyeva, Sharon Tirosh-Levy, Gila Abells-Sutton, Shiri Navon-Venezia, and Amir Steinman. 2019. "Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae in Hospitalized Neonatal Foals: Prevalence, Risk Factors for Shedding and Association with Infection" Animals 9, no. 9: 600. https://doi.org/10.3390/ani9090600
APA StyleShnaiderman-Torban, A., Paitan, Y., Arielly, H., Kondratyeva, K., Tirosh-Levy, S., Abells-Sutton, G., Navon-Venezia, S., & Steinman, A. (2019). Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae in Hospitalized Neonatal Foals: Prevalence, Risk Factors for Shedding and Association with Infection. Animals, 9(9), 600. https://doi.org/10.3390/ani9090600






