Coagulation Traits of Sheep and Goat Milk
Abstract
:Simple Summary
Abstract
1. Introduction
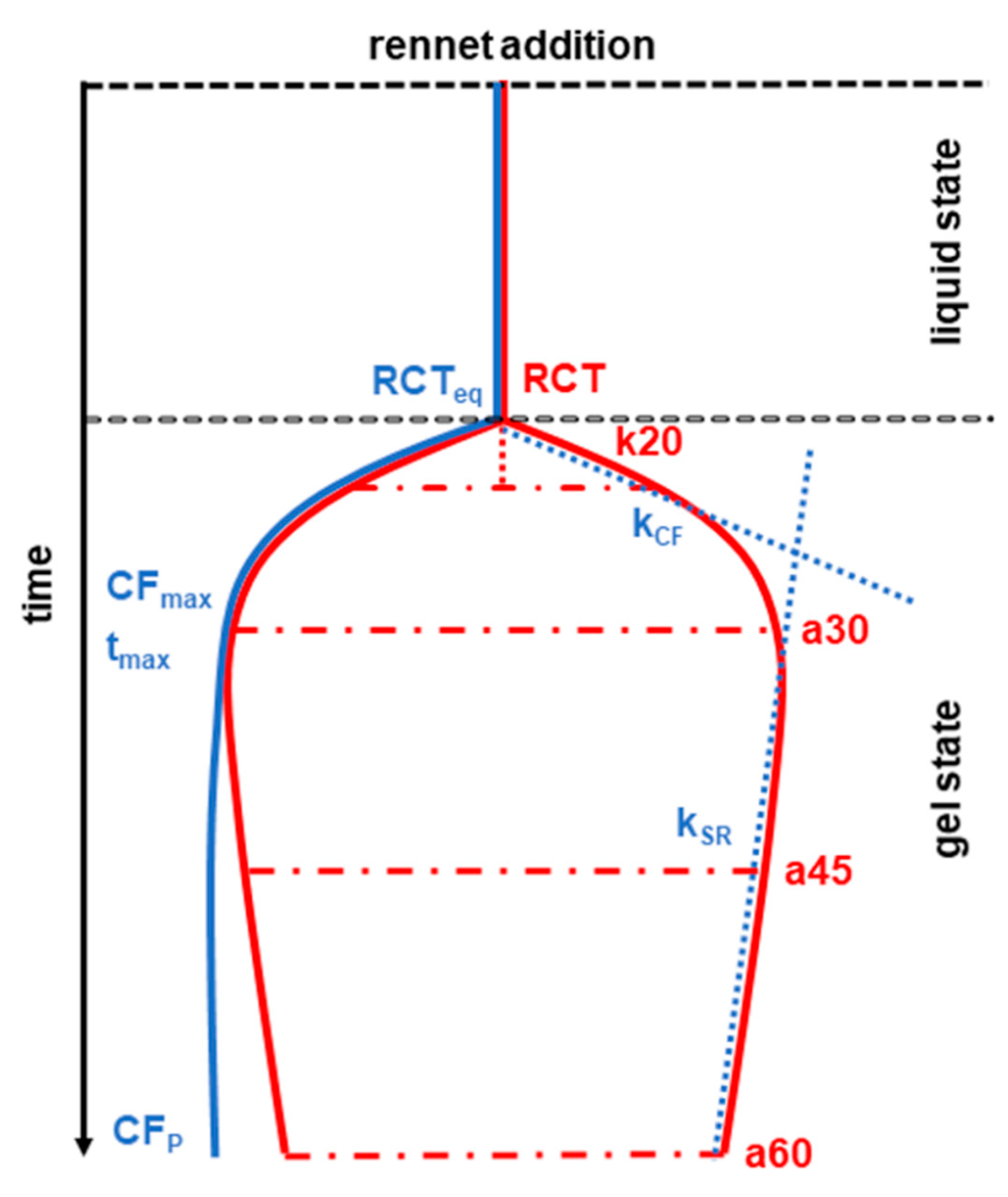
2. Sheep Milk
3. Goat Milk
4. Conclusions
- The differences among small ruminant breeds and farming techniques are still marked. Indeed, differently than cattle dairy sector, in which intensive farming and Holstein–Friesian cows are prevailing worldwide, sheep and goat milk productions are made from numerous breeds, both specialized and local ones, in large intensive or small extensive farms. In this scenario, a general application of results achieved from a specific breed and farming method could be very limited.
- The influence of individual samples on coagulation ability of bulk milk destined to dairy plants.
- The actual utilization of faster and cheaper methods to achieve coagulation traits and the introduction of these traits in genetic schemes and milk payment systems at the cheese industry level [42].
Funding
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Statistical Database of the Food and Agriculture Organization of the United Nations. Available online: http://www.faostat.fao.org (accessed on 27 June 2019).
- Boyazoglu, J.; Morand-Fehr, P. Mediterranean dairy sheep and goat products and their quality. A critical review. Small Rumin. Res. 2001, 40, 1–11. [Google Scholar] [CrossRef]
- García, V.; Rovira, S.; Boutoial, K.; López, M.B. Improvements in goat milk quality: A review. Small Rumin. Res. 2014, 121, 51–57. [Google Scholar] [CrossRef]
- Pirisi, A.; Lauret, A.; Dubeuf, J.P. Basic and incentive payments for goat and sheep milk in relation to quality. Small Rumin. Res. 2007, 68, 167–178. [Google Scholar] [CrossRef]
- Emmons, D.B.; Modler, H.W. Invited review: A commentary on predictive cheese yield formulas. J. Dairy Sci. 2010, 93, 5517–5537. [Google Scholar] [CrossRef] [PubMed]
- Lucey, J.A. Rennet-induced coagulation of milk. In Encyclopedia of Dairy Sciences, 2nd ed.; Fuquay, J.W., Fox, P.F., McSweeney, P.L.H., Eds.; Academic Press: San Diego, CA, USA, 2011; Volume 1, pp. 579–584. [Google Scholar]
- O’Callaghan, D.J.; O’Donnell, C.P.; Payne, F.A. Review of systems for monitoring curd setting during cheesemaking. Int. J. Dairy Technol. 2002, 55, 65–67. [Google Scholar] [CrossRef]
- Mc Mahon, D.J.; Brown, R.J. Evaluation of Formagraph for comparing rennet solutions. J. Dairy Sci. 1982, 65, 1639–1642. [Google Scholar] [CrossRef]
- Bittante, G. Modeling rennet coagulation and curd firmness of milk. J. Dairy Sci. 2011, 94, 5821–5832. [Google Scholar] [CrossRef]
- Bittante, G.; Contiero, B.; Cecchinato, A. Prolonged observation and modelling of milk coagulation, curd firming, and syneresis. Int. Dairy J. 2013, 29, 115–123. [Google Scholar] [CrossRef]
- Bittante, G.; Cipolat-Gotet, C.; Pazzola, M.; Dettori, M.L.; Vacca, G.M.; Cecchinato, A. Genetic analysis of coagulation properties, curd firming modeling, milk yield, composition and acidity in Sarda dairy sheep. J. Dairy Sci. 2017, 100, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Dal Zotto, R.; De Marchi, M.; Cecchinato, A.; Penasa, M.; Cassandro, M.; Carnier, P.; Gallo, L.; Bittante, G. Reproducibility and repeatability of measures of milk coagulation properties and predictive ability of mid-infrared reflectance spectroscopy. J. Dairy Sci. 2008, 91, 4103–4112. [Google Scholar] [CrossRef]
- Cipolat-Gotet, C.; Cecchinato, A.; De Marchi, M.; Penasa, M.; Bittante, G. Comparison between mechanical and near-infrared methods for assessing coagulation properties of bovine milk. J. Dairy Sci. 2012, 95, 6806–6819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Visentin, G.; McDermott, A.; McParland, S.; Berry, D.P.; Kenny, O.A.; Brodkorb, A.; Fenelon, M.A.; De Marchi, M. Prediction of bovine milk technological traits from mid-infrared spectroscopy analysis in dairy cows. J. Dairy Sci. 2015, 98, 6620–6629. [Google Scholar] [CrossRef] [Green Version]
- Cipolat-Gotet, C.; Cecchinato, A.; Stocco, G.; Bittante, G. The 9-MilCA method as a rapid, partly automated protocol for simultaneously recording milk coagulation, curd firming, syneresis, cheese yield, and curd nutrients recovery or whey loss. J. Dairy Sci. 2016, 99, 1065–1082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ambrosoli, R.; Di Stasio, L.; Mazzocco, P. Content of αS1-casein and coagulation properties in goat milk. J. Dairy Sci. 1988, 71, 24. [Google Scholar] [CrossRef]
- Barłowska, J.; Szwajkowska, M.; Litwinczuk, Z.; Król, J. Nutritional value and technological suitability of milk from various animal species used for dairy production. Compr. Rev. Food Sci. Food Saf. 2011, 10, 291–302. [Google Scholar] [CrossRef]
- Balthazar, C.F.; Pimentel, T.C.; Ferrão, L.L.; Almada, C.N.; Santillo, A.; Albenzio, M.; Mollakhalili, N.; Mortazavian, A.M.; Nascimento, J.S.; Silva, M.C.; et al. Sheep milk: Physicochemical characteristics and relevance for functional food development. Compr. Rev. Food Sci. Food Saf. 2017, 16, 247–262. [Google Scholar] [CrossRef]
- Scopus. Abstract and Citation Database of Peer-Reviewed Literature: Scientific Journals, Books and Conference Proceedings. Available online: https://www.scopus.com (accessed on 30 June 2019).
- Bittante, G.; Pellattiero, E.; Malchiodi, F.; Cipolat-Gotet, C.; Pazzola, M.; Vacca, G.M.; Schiavon, S.; Cecchinato, A. Quality traits and modelling of coagulation, curd firming and syneresis of sheep milk of Alpine breeds fed diets supplemented with rumen protected conjugated fatty acid. J. Dairy Sci. 2014, 97, 4018–4028. [Google Scholar] [CrossRef]
- Pazzola, M.; Dettori, M.L.; Cipolat-Gotet, C.; Cecchinato, A.; Bittante, G.; Vacca, G.M. Phenotypic factors affecting coagulation properties of milk from Sarda ewes. J. Dairy Sci. 2014, 97, 7247–7257. [Google Scholar] [CrossRef]
- Bittante, G.; Penasa, M.; Cecchinato, A. Invited review: Genetics and modeling of milk coagulation properties. J. Dairy Sci. 2012, 95, 6843–6870. [Google Scholar] [CrossRef]
- Ikonen, T.; Ahlfors, K.; Kempe, R.; Ojala, M.; Ruottinen, O. Genetic parameters for the milk coagulation properties and prevalence of noncoagulating milk in Finnish dairy cows. J. Dairy Sci. 1999, 82, 205–214. [Google Scholar] [CrossRef]
- Saha, S.; Amalfitano, N.; Sturaro, E.; Schiavon, S.; Tagliapietra, F.; Bittante, G.; Carafa, I.; Franciosi, E.; Gallo, L. Effects of summer transhumance of dairy cows to alpine pastures on body condition, milk yield and composition, and cheese making efficiency. Animals 2019, 9, 192. [Google Scholar] [CrossRef] [PubMed]
- Caballero-Villalobos, J.; Perea, J.M.; Angón, E.; Arias, R.; Garzón, A. Coagulation efficiency and its determinant factors: A case study for Manchega ewe milk in the region of Castilla-La Mancha, Spain. J. Dairy Sci. 2018, 101, 3878–3886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manuelian, C.L.; Penasa, M.; Giangolini, G.; Boselli, C.; Currò, S.; De Marchi, M. Short communication: Fourier-transform mid-infrared spectroscopy to predict coagulation and acidity traits of sheep bulk milk. J. Dairy Sci. 2019, 102, 1927–1932. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vacca, G.M.; Pazzola, M.; Dettori, M.L.; Pira, E.; Malchiodi, F.; Cipolat-Gotet, C.; Cecchinato, A.; Bittante, G. Modeling of coagulation, curd firming, and syneresis of milk from Sarda ewes. J. Dairy Sci. 2015, 98, 2245–2259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noce, A.; Pazzola, M.; Dettori, M.L.; Amills, M.; Castelló, A.; Cecchinato, A.; Bittante, G.; Vacca, G.M. Variations at regulatory regions of the milk protein genes are associated with milk traits and coagulation properties in the Sarda sheep. Anim. Genet. 2016, 47, 717–726. [Google Scholar] [CrossRef]
- Carta, A.; Casu, S.; Salaris, S. Invited review: Current state of genetic improvement in dairy sheep. J. Dairy Sci. 2009, 92, 5814–5833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manca, M.G.; Serdino, J.; Gaspa, G.; Urgeghe, P.; Ibba, I.; Contu, M.; Fresi, P.; Macciotta, N.P.P. Derivation of multivariate indices of milk composition, coagulation properties, and individual cheese yield in dairy sheep. J. Dairy Sci. 2016, 99, 4547–4557. [Google Scholar] [CrossRef]
- Pazzola, M.; Cipolat-Gotet, C.; Bittante, G.; Cecchinato, A.; Dettori, M.L.; Vacca, G.M. Phenotypic and genetic relationships between indicators of the mammary gland health status and milk composition, coagulation, and curd firming in dairy sheep. J. Dairy Sci. 2018, 101, 3164–3175. [Google Scholar] [CrossRef]
- Ferragina, A.; Cipolat-Gotet, C.; Cecchinato, A.; Pazzola, M.; Dettori, M.L.; Vacca, G.M.; Bittante, G. Prediction and repeatability of milk coagulation properties and curd-firming modeling parameters of ovine milk using Fourier-transform infrared spectroscopy and Bayesian models. J. Dairy Sci. 2017, 100, 3526–3538. [Google Scholar] [CrossRef]
- Vacca, G.M.; Cipolat-Gotet, C.; Paschino, P.; Casu, S.; Usai, M.G.; Bittante, G.; Pazzola, M. Variation of milk technological properties in sheep milk: Relationships among composition, coagulation and cheese-making traits. Int. Dairy J. 2019, 97, 5–14. [Google Scholar] [CrossRef]
- Othmane, M.H.; Carriedo, J.A.; de la Fuente Crespo, L.F.; San Primitivo, F. An individual laboratory cheese-making method for selection in dairy ewes. Small Rumin. Res. 2002, 45, 67–73. [Google Scholar] [CrossRef]
- Puledda, A.; Gaspa, G.; Manca, M.G.; Serdino, J.; Urgeghe, P.P.; Dimauro, C.; Negrini, R.; Macciotta, N.P.P. Estimates of heritability and genetic correlations for milk coagulation properties and individual laboratory cheese yield in Sarda ewes. Animal 2016, 11, 920–928. [Google Scholar] [CrossRef] [PubMed]
- Clark, S.; Mora García, M.B. A 100-Year Review: Advances in goat milk research. J. Dairy Sci. 2017, 100, 10026–10044. [Google Scholar] [CrossRef] [PubMed]
- Vacca, G.M.; Stocco, G.; Dettori, M.L.; Pira, E.; Bittante, G.; Pazzola, M. Milk yield, quality, and coagulation properties of 6 breeds of goats: Environmental and individual variability. J. Dairy Sci. 2018, 101, 7236–7247. [Google Scholar] [CrossRef] [PubMed]
- Stocco, G.; Pazzola, M.; Dettori, M.L.; Paschino, P.; Bittante, G.; Vacca, G.M. Effect of composition on coagulation, curd firming, and syneresis of goat milk. J. Dairy Sci. 2018, 101, 9693–9702. [Google Scholar] [CrossRef] [PubMed]
- Pazzola, M.; Stocco, G.; Dettori, M.L.; Cipolat-Gotet, C.; Bittante, G.; Vacca, G.M. Modeling of coagulation, curd firming, and syneresis of goat milk. J. Dairy Sci. 2018, 101, 7027–7039. [Google Scholar] [CrossRef] [PubMed]
- Vacca, G.M.; Stocco, G.; Dettori, M.L.; Summer, A.; Cipolat-Gotet, C.; Bittante, G.; Pazzola, M. Cheese yield, cheesemaking efficiency, and daily production of 6 breeds of goats. J. Dairy Sci. 2018, 101, 7236–7247. [Google Scholar] [CrossRef] [PubMed]
- Pazzola, M.; Stocco, G.; Dettori, M.L.; Bittante, G.; Vacca, G.M. Effect of goat milk composition on cheesemaking traits and daily cheese production. J. Dairy Sci. 2019, 102, 3947–3955. [Google Scholar] [CrossRef]
- Bittante, G.; Cecchinato, A.; Cologna, N.; Penasa, M.; Tiezzi, F.; De Marchi, M. Factors affecting the incidence of first-quality wheels of Trentingrana cheese. J. Dairy Sci. 2011, 94, 3700–3707. [Google Scholar] [CrossRef] [Green Version]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pazzola, M. Coagulation Traits of Sheep and Goat Milk. Animals 2019, 9, 540. https://doi.org/10.3390/ani9080540
Pazzola M. Coagulation Traits of Sheep and Goat Milk. Animals. 2019; 9(8):540. https://doi.org/10.3390/ani9080540
Chicago/Turabian StylePazzola, Michele. 2019. "Coagulation Traits of Sheep and Goat Milk" Animals 9, no. 8: 540. https://doi.org/10.3390/ani9080540
APA StylePazzola, M. (2019). Coagulation Traits of Sheep and Goat Milk. Animals, 9(8), 540. https://doi.org/10.3390/ani9080540




