The Effect of Extensive Human Presence at an Early Age on Stress Responses and Reactivity of Juvenile Ostriches towards Humans
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
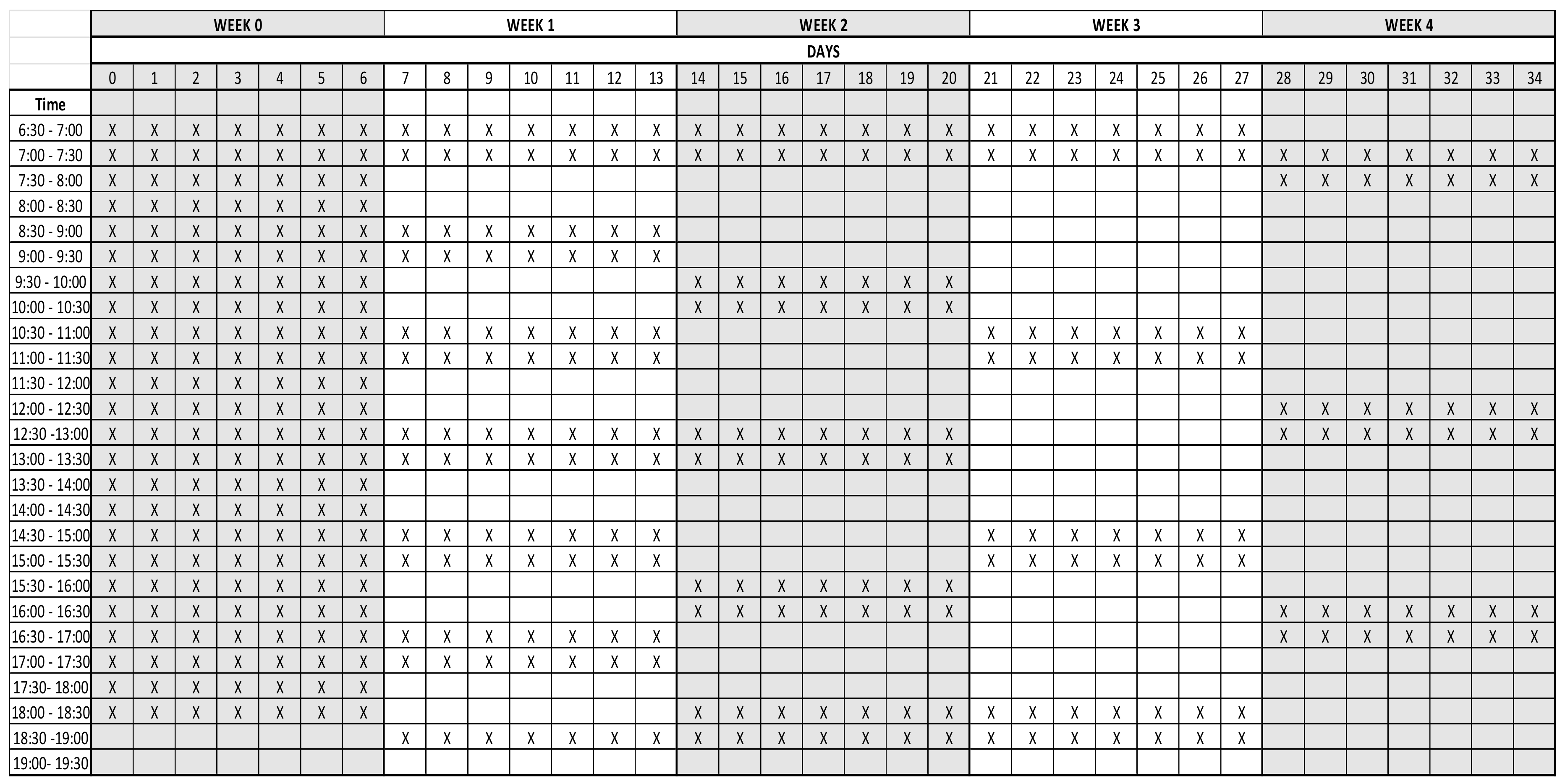
2.2. Treatments
2.3. Physiological Measures of Stress
2.4. Behavioural Responses
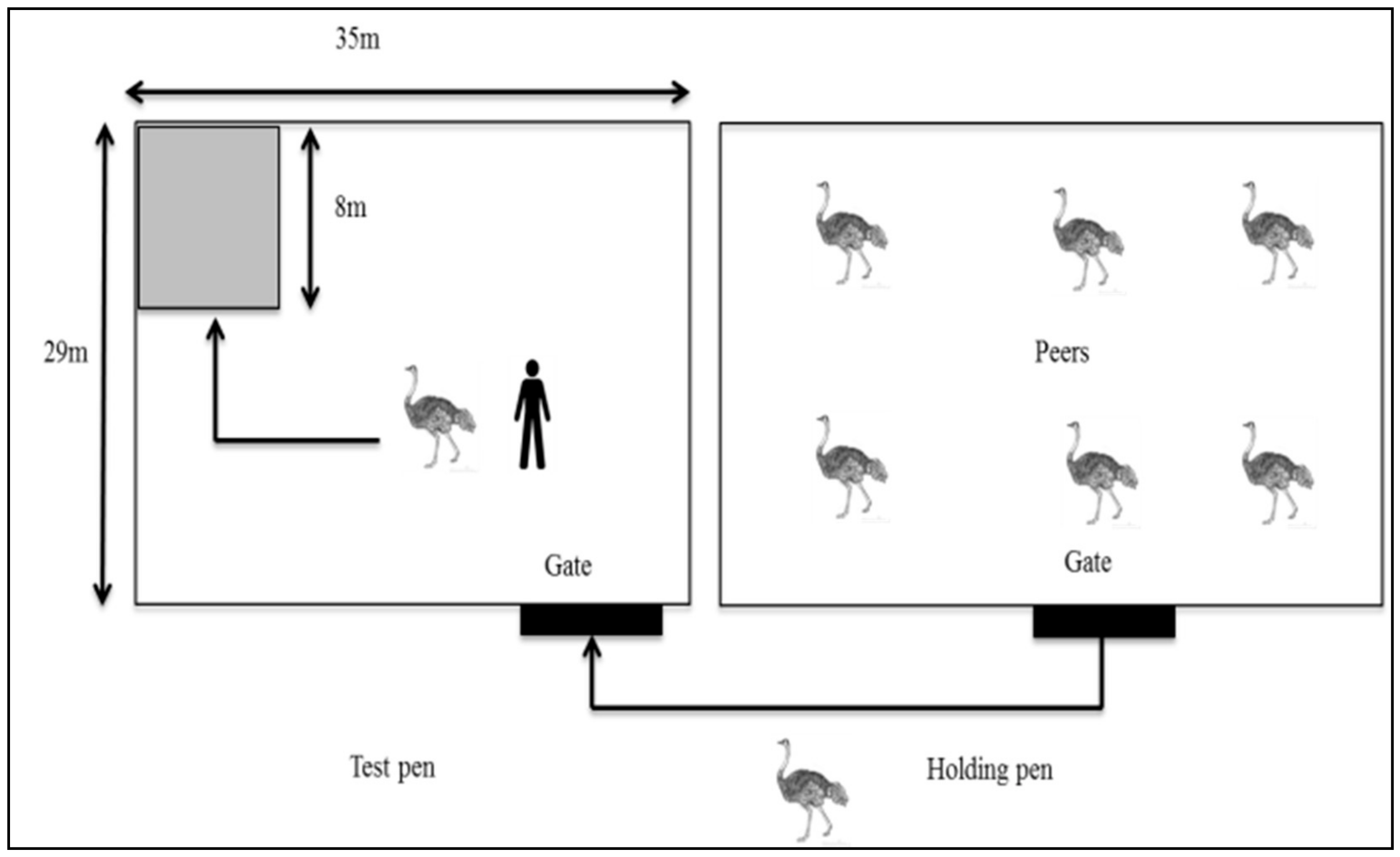
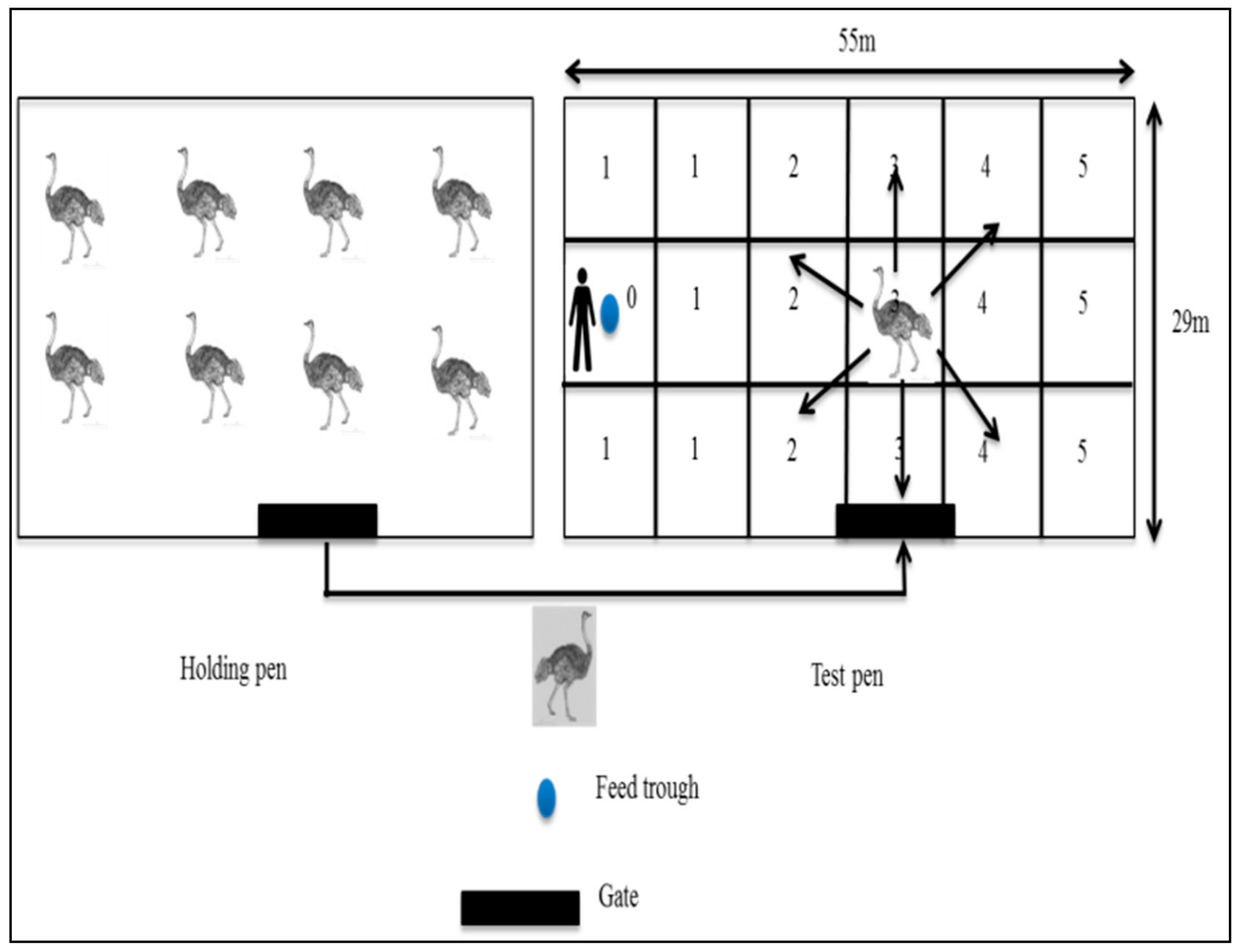
2.4.1. Reactivity Test
2.4.2. Docility Test
2.4.3. Fear Test
2.5. Statistical Analysis
3. Results
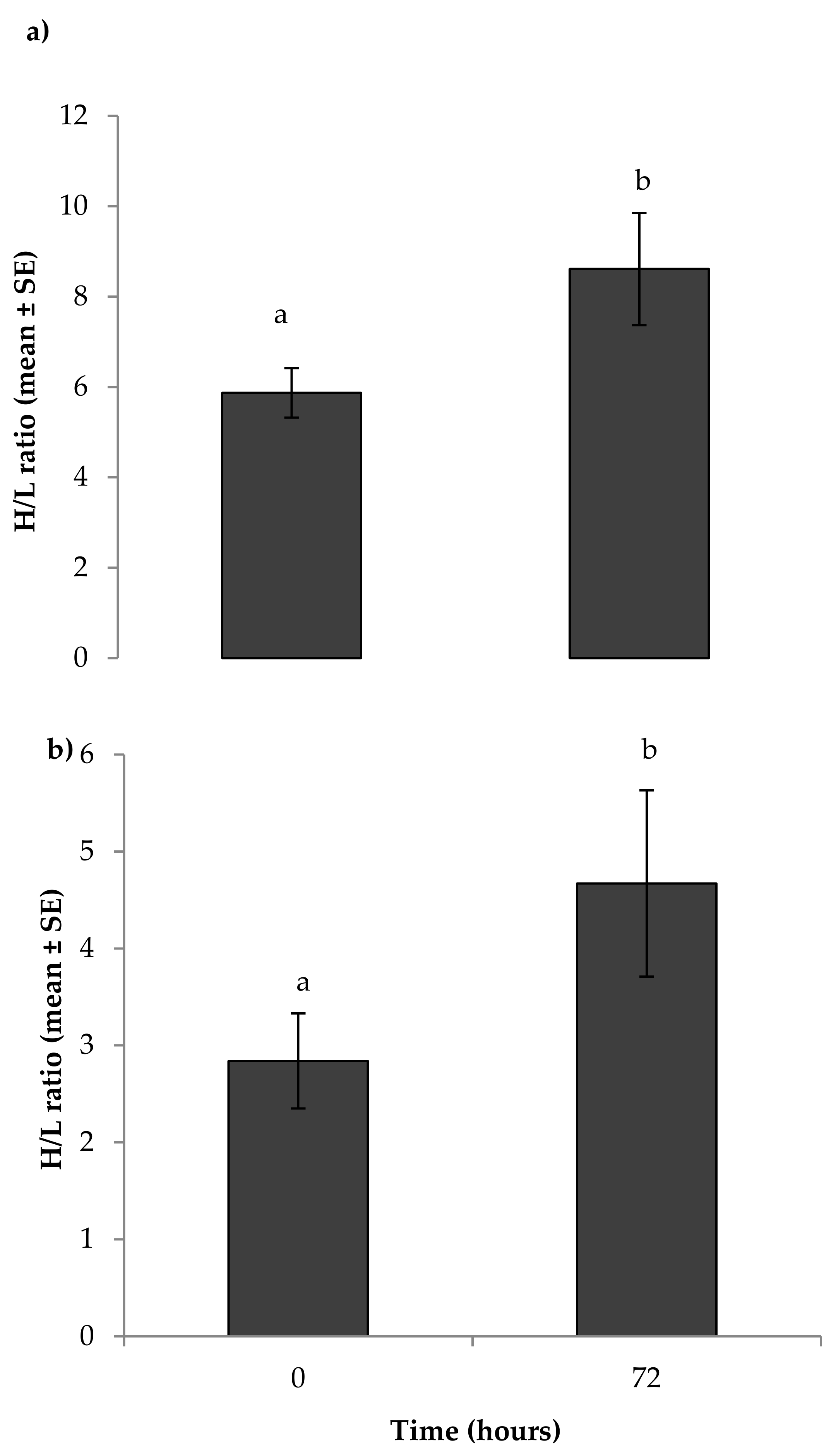
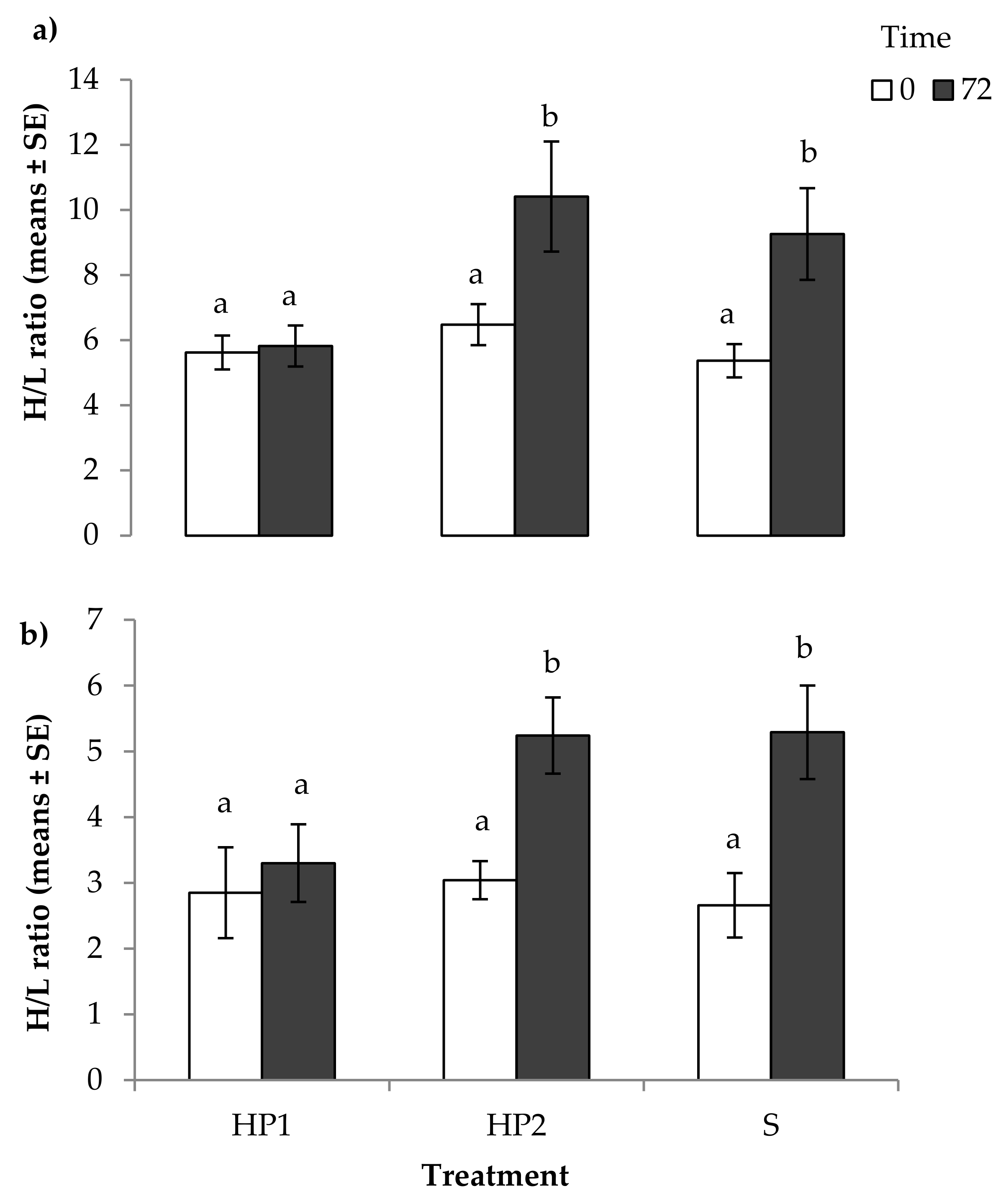
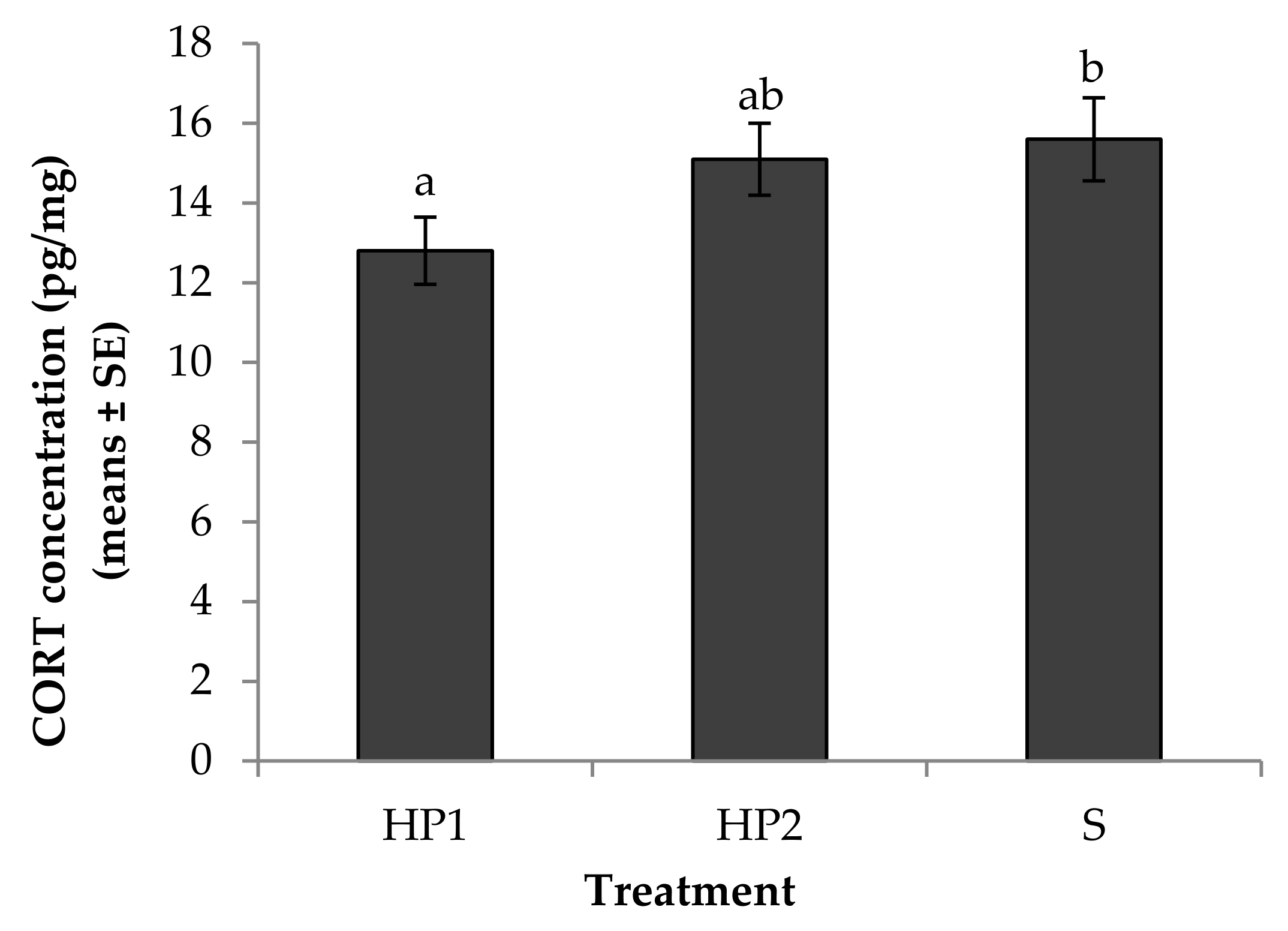
3.1. Physiological Measures of Stress
3.2. Behavioural Responses
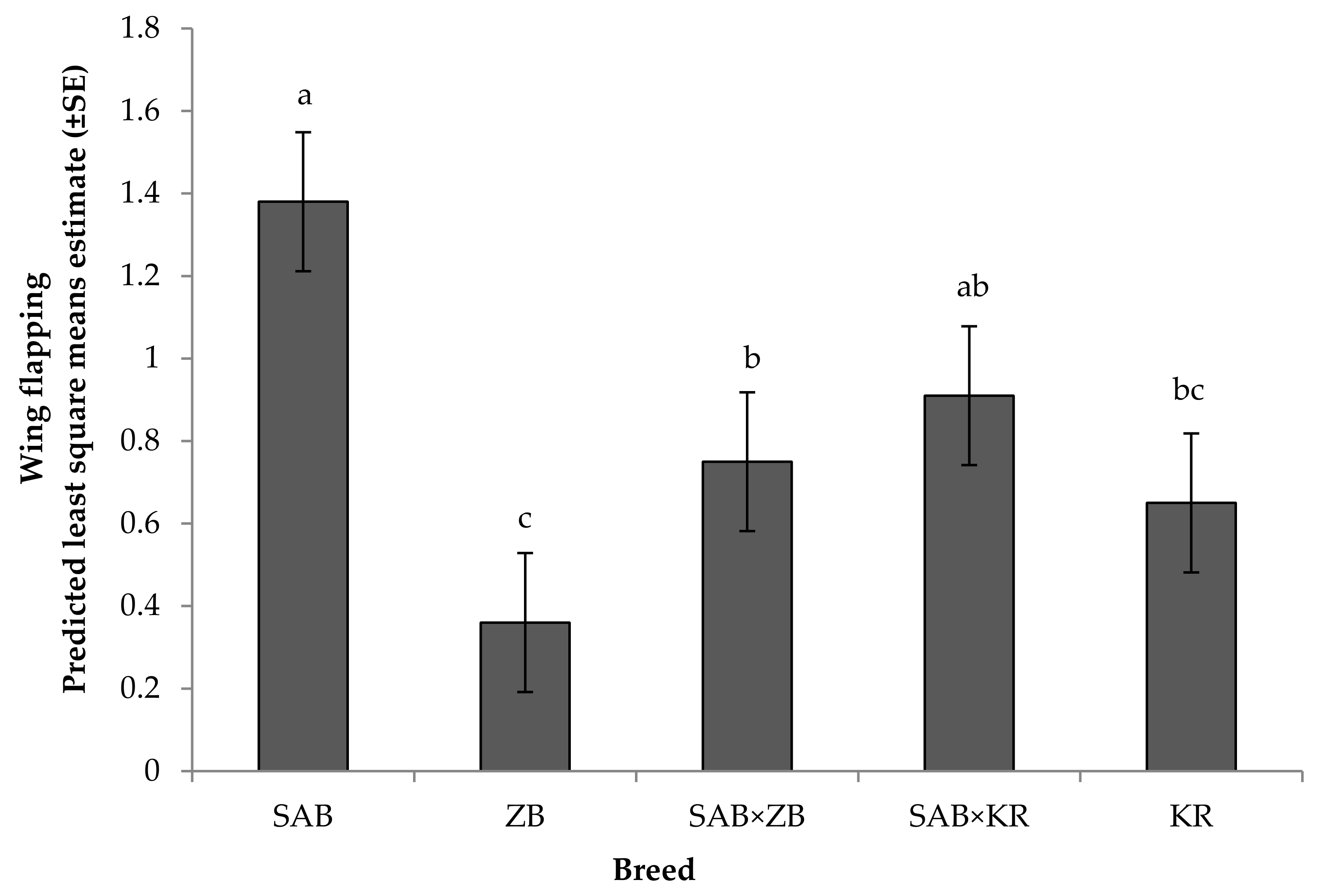
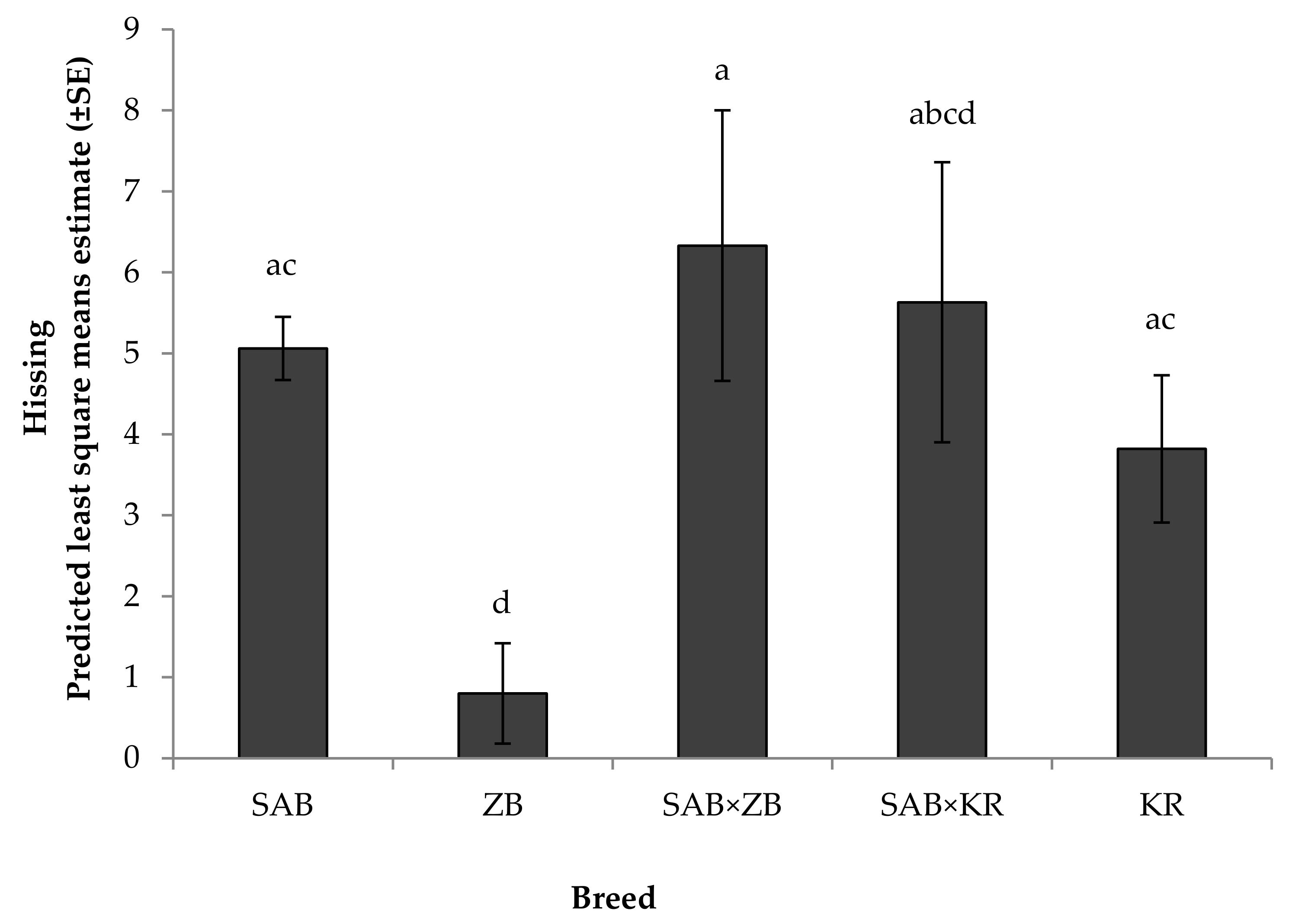
3.2.1. Reactivity Tests towards Humans
3.2.2. Docility and Fear Responses
4. Discussion
4.1. Physiological Measures of Stress
4.2. Behavioural Responses
Reactivity Test
4.3. Docility and Fear Responses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Cloete, S.W.P.; Engelbrecht, A.; Olivier, J.J.; Bunter, K.L. Deriving a preliminary breeding objective for commercial ostriches: An overview. Aust. J. Exp. Agric. 2008, 48, 1247–1256. [Google Scholar] [CrossRef]
- Cloete, S.W.P.; Brand, T.S.; Hoffman, L.; Brand, Z.; Engelbrecht, A.; Bonato, M.; Glatz, P.C.; Malecki, I.A. The development of ratite production through continued research. Worlds. Poult. Sci. J. 2012, 68, 323–334. [Google Scholar] [CrossRef]
- Deeming, D.C. Incubation and Chick Rearing. In The Welfare of Farmed Ratites; Glatz, P., Lunam, C., Malecki, I., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 65–89. [Google Scholar]
- Amado, M.F.; Xavier, D.B.; Boere, V.; Torres-Pereira, C.; McManus, C.; Bernal, F.E.M. Behaviour of captive ostrich chicks from 10 days to 5 months of age. Revista Brasileira de Zootecnia 2011, 40, 1613–1618. [Google Scholar] [CrossRef]
- Lambrechts, H.; Cloete, S.W.P.; Swart, D.; Pfister, A.P. Influence of territorial aggressiveness of ostrich males on egg production of companion female ostriches. Short paper and poster abstracts: 38th Congress of the South African Society of Animal Science. S. Afr. J. Anim. Sci. 2000, 30, 68–69. [Google Scholar]
- Bertenshaw, C.; Rowlinson, P.; Edge, H.; Douglas, S.; Shiel, R. The effect of different degrees of ‘positive’ human–animal interaction during rearing on the welfare and subsequent production of commercial dairy heifers. Appl. Anim. Behav. Sci. 2008, 114, 65–75. [Google Scholar] [CrossRef]
- Waiblinger, S.; Boivin, X.; Pedersen, V.; Tosi, M.V.; Janczak, A.M.; Visser, E.K.; Jones, R.B. Assessing the human-animal relationship in farmed species: A critical review. Appl. Anim. Behav. Sci. 2006, 101, 185–242. [Google Scholar] [CrossRef]
- Lürzel, S.; Barth, K.; Windschnurer, I.; Futschik, A.; Waiblinger, S. The influence of gentle interactions with an experimenter during milking on dairy cows’ avoidance distance and milk yield, flow and composition. Animal 2017, 12, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Tallet, C.; Brajon, S.; Devillers, N.; Lensink, J. Pig human interactions: Creating a positive perception of humans to ensure pig welfare. In Advances in Pig Welfare, 1st ed.; Špinka, M., Ed.; Woodhead Publishing: Duxford, UK, 2018; pp. 381–398. [Google Scholar]
- McCarty, R.; Horwatt, K.; Konarska, M. Chronic stress and sympathetic-adrenal medullary responsiveness. Soc. Sci. Med. 1988, 26, 333–341. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Barnett, J.L.; Hansen, C. The influence of handling by humans on the behaviour, reproduction and corticosteroids of male and female pigs. Appl. Anim. Behav. Sci. 1986, 15, 303–314. [Google Scholar] [CrossRef]
- Breuer, K.; Hemsworth, P.H.; Coleman, G.J. The effect of positive or negative handling on the behavioural and physiological responses of nonlactating heifers. Appl. Anim. Behav. Sci. 2003, 84, 3–22. [Google Scholar] [CrossRef]
- Al-Aqil, A.; Zulkifli, I.; Hair Bejo, M.; Sazili, A.Q.; Rajion, M.A.; Somchit, M.N. Changes in heat shock protein 70, blood parameters and fear-related behavior in broiler chickens as affected by pleasant and unpleasant human contact. Poult. Sci. 2013, 93, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Hemsworth, P.H.; Coleman, G.J. Human–Livestock Interactions: The Stockperson and the Productivity and Welfare of Intensively Farmed Animals, 2nd ed.; CAB International: London, UK, 2011. [Google Scholar]
- Hemsworth, P.H.; Coleman, G.J.; Barnett, J.L.; Jones, R.B. Behavioural responses to humans and the productivity of commercial broiler chickens. Appl. Anim. Behav. Sci. 1994, 41, 101–114. [Google Scholar] [CrossRef]
- Bonato, M.; Malecki, I.A.; Wang, M.D.; Cloete, S.W.P. Extensive human presence at an early age of ostriches improves the docility of birds at a later stage of life. Appl. Anim. Behav. Sci. 2013, 148, 232–239. [Google Scholar] [CrossRef]
- Bunter, K.L.; Cloete, S.W.P. Genetic parameters for egg-, chick- and live-weight traits recorded in farmed ostriches (Struthio camelus). Livest. Prod. Sci. 2004, 91, 9–22. [Google Scholar] [CrossRef]
- Bunter, K.L. The Genetic Analysis of Reproduction and Production Traits Recorded for Farmed Ostriches (Struthio camelus). Ph.D. Thesis, University of New England, Armidale, Australia, 2002. [Google Scholar]
- Shanawany, M.M.; Dingle, J. Ostrich Production System, Part 1–2; Food & Agriculture Organization: Rome, Italy, 1999. [Google Scholar]
- South African Ostrich Business Chamber (SAOBC). Code of Conduct for the Commercial Production of Ostriches, Updated version; South African Business Chamber: Oudtshoorn, South Africa, 2011; pp. 1–38. [Google Scholar]
- Kamau, J.M.; Patrick, B.T.; Mushi, E.Z. The effect of mixing and translocating juvenile ostriches (Struthio camelus) in Botswana on the Heterophil to Lymphocyte ratio. Trop. Anim. Health Prod. 2002, 34, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Bortolotti, G.R.; Marchant, T.A.; Blas, J.; German, T. Corticosterone in feathers is a long-term, integrated measure of avian stress physiology. Funct. Ecol. 2008, 22, 494–500. [Google Scholar] [CrossRef]
- Brand, T.S.; Jordaan, J.W.; Bhiya, C.S.; Aucamp, B.B. Effect of slaughter age and sex on the production output of South African Black ostriches. Poult. Sci. 2013, 51, 510–514. [Google Scholar] [CrossRef] [PubMed]
- Malecki, I.A.; Rybnik-Trzaskowska, P.K. Natural mating and artificial Insemination. In The Welfare of Farmed Ratites; Glatz, P., Lunam, C., Malecki, I., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 45–63. [Google Scholar]
- Bonato, M.; Cherry, M.I.; Cloete, S.W.P. Mate choice, maternal investment and implications for ostrich welfare in a farming environment. Appl. Anim. Behav. Sci. 2015, 171, 1–7. [Google Scholar] [CrossRef]
- Mazurek, M.; McGee, M.; Crowe, M.A.; Prendiville, D.J.; Boivin, X.; Earley, B. Consistency and stability of behavioural fear responses of heifers to different fear-eliciting situations involving animals. Appl. Anim. Behav. Sci. 2011, 131, 21–28. [Google Scholar] [CrossRef]
- Hoffman, L.C.; Lambrechts, H. Bird handling, transportation, lairage and slaughter: Implications for bird welfare and meat quality. In The Welfare of Farmed Ratites; Glatz, P., Lunam, C., Malecki, I., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 195–236. [Google Scholar]
- Hemsworth, P.H.; Rice, M.; Karlen, M.G.; Calleja, L.; Barnett, J.L.; Nash, J.; Coleman, G.J. Human–animal interactions at abattoirs: Relationships between handling and animal stress in sheep and cattle. Appl. Anim. Behav. Sci. 2011, 135, 24–33. [Google Scholar] [CrossRef]
- Bejaei, M.; Cheng, K.M. A survey of current ostrich handling and transport practices in North America with reference to ostrich welfare and transportation guidelines set up in other countries. Poult. Sci. 2014, 93, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Samson, J. Behavioral problems of farmed ostriches in Canada. Can. Vet. J. 1996, 37, 412–414. [Google Scholar] [PubMed]
- Brand, T.S. Ostrich Nutrition Guidelines; Ostrich Manual, Western Cape Department of Agriculture, South African Ostrich Business Chamber: Elsenburg, South Africa, 2014; pp. 13–19. [Google Scholar]
- Kendall, J.A. Strategies for performing multiple comparisons on means. Proc. SAS Users Group Conf. 1993, 18, 1283–1289. [Google Scholar]
- Statistical Analysis System (SAS). Base SAS® 9.3 Procedures Guide: Statistical Procedures, 2nd ed.; SAS Institute Inc.: Cary, NC, USA, 2012. [Google Scholar]
- Hemsworth, P.H. Human–animal interactions in livestock production. Appl. Anim. Behav. Sci. 2003, 81, 185–198. [Google Scholar] [CrossRef]
- Weir, K.A.; Lunam, C.A. Immunohistochemical study of cutaneous nerves in the emu. Cell Tissue Res. 2006, 326, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Diorio, J.; Tannenbaum, B.; Caldji, C.; Francis, D.; Freedman, A.; Sharma, S.; Pearson, D.; Plotsky, P.M.; Meaney, M.J. Maternal care, hippocampal glucocorticoid receptors, and hypothalamic-pituitary-adrenal responses to stress. Science 1997, 277, 1659–1662. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.M.; Molenda-Figueira, H.A.; Sisk, C.L. Back to the future: The organizational–activational hypothesis adapted to puberty and adolescence. Horm. Behav. 2009, 55, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Lürzel, S.; Windschnurer, I.; Futschik, A.; Palme, R.; Waiblinger, S. Effects of gentle interactions on the relationship with humans and on stress-related parameters in group-housed calves. Anim. Welfare 2015, 24, 475–484. [Google Scholar] [CrossRef]
- Zulkifli, I.; Gilbert, J.; Liew, P.K.; Ginsos, J. The effects of regular visual contact with human beings on fear, stress, antibody and growth responses in broiler chickens. Appl. Anim. Behav. Sci. 2002, 79, 103–112. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Coleman, G.J.; Barnett, J.L.; Borg, S. Relationships between human–animal interactions and productivity of commercial dairy cows. J. Anim. Sci. 2000, 80, 2821–2831. [Google Scholar] [CrossRef]
- Tanida, H.; Miura, A.; Tanaka, T.; Yoshimoto, T. The role of handling in communication between humans and weanling pigs. Appl. Anim. Behav. Sci. 1994, 40, 219–228. [Google Scholar] [CrossRef]
- De Oliveira, D.; Paranhos da Costa, M.J.R.; Zupan, M.; Rehn, T.; Keeling, L.J. Early human handling in non-weaned piglets: Effects on behaviour and body weight. Appl. Anim. Behav. Sci. 2015, 164, 56–63. [Google Scholar] [CrossRef]
- Sommavilla, R.; Titto, E.A.L.; Titto, C.G.; Hötzel, M.J. Ninety one-days-old piglets recognize and remember a previous aversive handler. Livest. Sci. 2016, 194, 7–9. [Google Scholar] [CrossRef]
- Jones, R.B.; Faure, J.M. The effects of regular handling on fear in the domestic chick. Behav. Process. 1981, 6, 135–143. [Google Scholar] [CrossRef]
- Davis, H.; Taylor, A. Discrimination between individual humans by domestic fowl (Gallus gallus domesticus). Br. Poult. Sci. 2001, 42, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Tanida, H.; Nagano, Y. The ability of miniature pigs to discriminate between a stranger and their familiar handler. Appl. Anim. Behav. Sci. 1998, 56, 149–159. [Google Scholar] [CrossRef]
- Koba, Y.; Tanida, H. How do miniature pigs discriminate between people? Discrimination between people wearing coveralls of the same colour. Appl. Anim. Behav. Sci. 2001, 73, 45–58. [Google Scholar] [CrossRef]
- Munksgaard, L.; de Passille, A.M.; Rushen, J.; Ladewig, J. Dairy cows’ use of colour cues to discriminate between people. Appl. Anim. Behav. Sci. 1999, 65, 1–11. [Google Scholar] [CrossRef]
- Rushen, J.; de Passille, A.M.B.; Munksgaard, L. Fear of people by cows and effects on milk yield, behaviour and heart rate at milking. J. Dairy Sci. 1999, 82, 720–727. [Google Scholar] [CrossRef]
- Cooper, R.G.; Horbanczuk, J.O.; Villegas-Vizcaino, R.; Sebei, S.K.; Faki Mohammed, A.E.; Mahrose, K.M.A. Wild ostrich (Sturthio camelus) ecology and physiology. Trop. Anim. Health. Prod. 2010, 42, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Burrow, H.M.; Seifert, G.W.; Corbet, N.J. A new technique for measuring temperament in cattle. Proc. Aust. Soc. Anim. Prod. 1988, 17, 154–157. [Google Scholar]
- Coleman, G.J.; Hemsworth, P.H. Training to improve stockperson beliefs and behaviour towards livestock enhances welfare and productivity. Rev. Sci. Tech. 2014, 33, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Lensink, J.; Boissy, A.; Veissier, I. The relationship between farmers’ attitude and behaviour towards calves, and productivity of veal units. Ann. Zootech. 2000, 49, 313–327. [Google Scholar] [CrossRef]
| Effects | Behavioural Responses | ||||
|---|---|---|---|---|---|
| Approach | Touch | Wing Flapping | Excessive Pecking | Hissing | |
| Familiarity: | |||||
| Familiar | 3.95 ± 0.45 | 2.87 ± 0.41 | 0.38 ± 0.20 | 2.23 ± 0.36 | 4.29 ± 1.02 |
| Unfamiliar | 1.99 ± 0.36 | 2.07 ± 0.37 | 0.96 ± 0.21 | 2.30 ± 0.37 | 4.36 ± 1.05 |
| p-value | <0.01 | >0.05 | <0.05 | >0.05 | >0.05 |
| Year: | |||||
| 2013 | 3.34 ± 0.45 | 2.05 ± 0.34 | 0.61 ± 0.19 | 2.10 ± 0.34 | 3.34 ± 0.62 |
| 2015 | 2.59 ± 0.33 | 2.89 ± 0.33 | 0.72 ± 0.16 | 2.42 ± 0.33 | 5.32 ± 0.79 |
| p-value | >0.05 | <0.05 | >0.05 | >0.05 | <0.05 |
| Sex: | |||||
| Male | 2.69 ± 0.30 | 2.50 ± 0.31 | 0.76 ± 0.17 | 2.63 ± 0.33 | 4.42 ± 0.60 |
| Female | 3.25 ± 0.37 | 2.44 ± 0.31 | 0.58 ± 0.17 | 1.90 ± 0.32 | 4.23 ± 0.61 |
| p-value | <0.05 | >0.05 | >0.05 | <0.01 | >0.05 |
| Effect | Sexual Behavioural Responses | ||||
|---|---|---|---|---|---|
| Males | Females | ||||
| Year | Clucking | Stepping | Kantling | Clucking | Crouching |
| 2013 | 6.19 ± 5.38 | 1.69 ± 0.99 | 3.66 ± 0.81 | 6.29 ± 16.47 | 6.60 ± 8.86 |
| 2015 | 12.5 ± 7.20 | 4.54 ± 1.13 | 5.29 ± 0.74 | 11.60 ± 16.83 | 9.29 ± 8.77 |
| p-value | >0.05 | <0.01 | <0.05 | >0.05 | >0.05 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muvhali, P.T.; Bonato, M.; Engelbrecht, A.; Malecki, I.A.; Hough, D.; Robinson, J.E.; Evans, N.P.; Cloete, S.W.P. The Effect of Extensive Human Presence at an Early Age on Stress Responses and Reactivity of Juvenile Ostriches towards Humans. Animals 2018, 8, 175. https://doi.org/10.3390/ani8100175
Muvhali PT, Bonato M, Engelbrecht A, Malecki IA, Hough D, Robinson JE, Evans NP, Cloete SWP. The Effect of Extensive Human Presence at an Early Age on Stress Responses and Reactivity of Juvenile Ostriches towards Humans. Animals. 2018; 8(10):175. https://doi.org/10.3390/ani8100175
Chicago/Turabian StyleMuvhali, Pfunzo T., Maud Bonato, Anel Engelbrecht, Irek A. Malecki, Denise Hough, Jane E. Robinson, Neil P. Evans, and Schalk W. P. Cloete. 2018. "The Effect of Extensive Human Presence at an Early Age on Stress Responses and Reactivity of Juvenile Ostriches towards Humans" Animals 8, no. 10: 175. https://doi.org/10.3390/ani8100175
APA StyleMuvhali, P. T., Bonato, M., Engelbrecht, A., Malecki, I. A., Hough, D., Robinson, J. E., Evans, N. P., & Cloete, S. W. P. (2018). The Effect of Extensive Human Presence at an Early Age on Stress Responses and Reactivity of Juvenile Ostriches towards Humans. Animals, 8(10), 175. https://doi.org/10.3390/ani8100175






