Using the Horse Grimace Scale (HGS) to Assess Pain Associated with Acute Laminitis in Horses (Equus caballus)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Obel Grade Scoring
2.3. Horse Grimace Scale (HGS) Recordings
2.3.1. Image Collection
2.3.2. Image Selection and Scoring
2.3.3. Video Processing and Scoring
2.4. Statistical Data Analysis
3. Results
3.1. Obel Grade
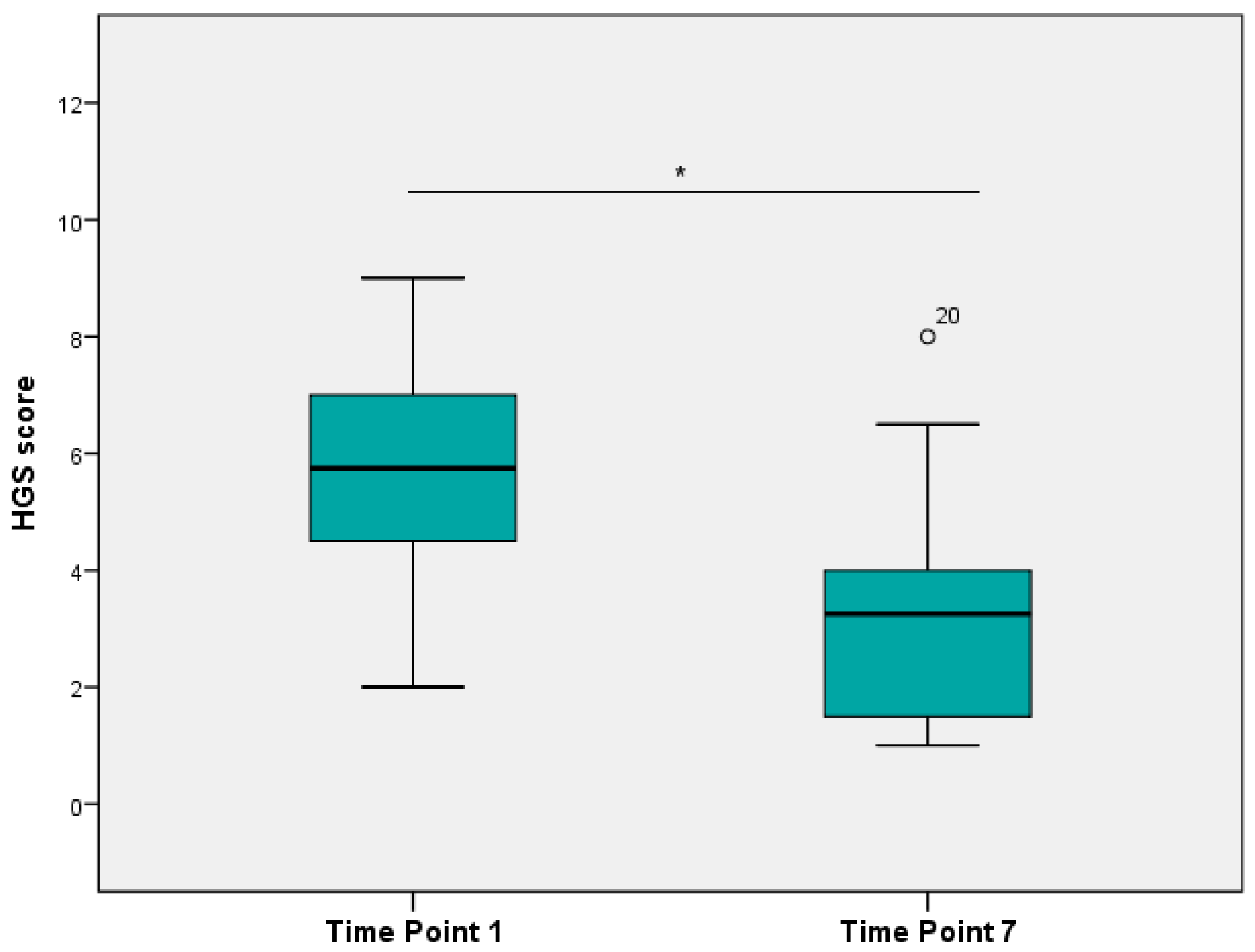
3.2. Scoring Images with HGS
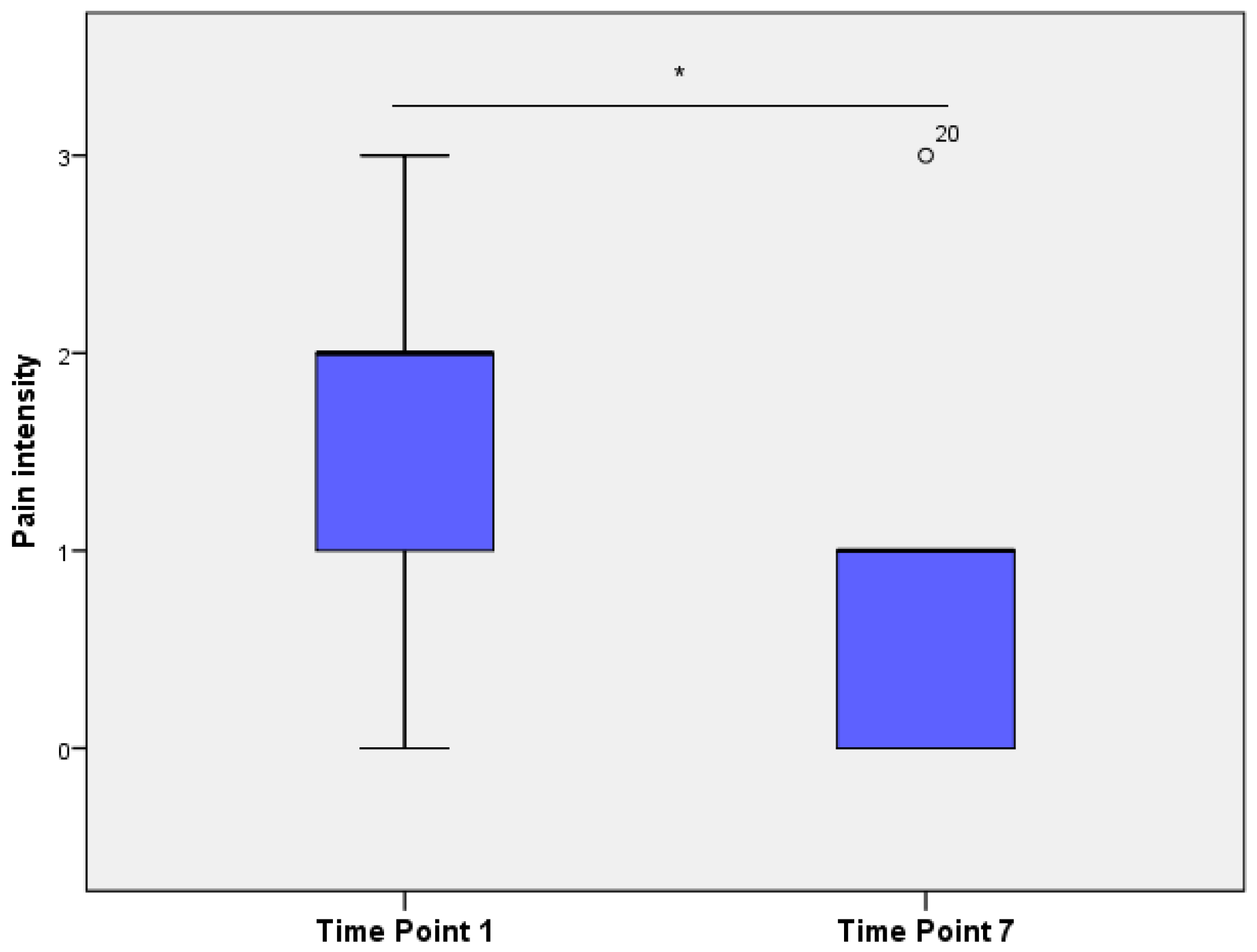
3.3. Scoring Videos with HGS
3.4. Relationship between Obel Grade and HGS
3.5. Comparison of HGS Scores from Images and Video
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Collins, S.N.; Pollitt, C.; Wylie, C.E.; Matiasek, K. Laminitic pain: Parallels with pain states in humans and other species. Vet. Clin. N. Am. Equine Pract. 2010, 3, 643–671. [Google Scholar] [CrossRef] [PubMed]
- Wylie, C.E.; Collins, S.N.; Verheyen, K.L.P.; Newton, J.R. A cohort study of equine laminitis in Great Britain 2009–2011: Estimation of disease frequency and description of clinical signs in 577 cases. Equine Vet. J. 2013, 45, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.M. Vision 20/20-Conquer laminitis by 2020. J. Equine Vet. Sci. 2010, 30, 74–76. [Google Scholar] [CrossRef]
- Obel, N. Studies on the Histopathology of Acute Laminitis; Botryckeri: Uppsala, Sweden, 1948. [Google Scholar]
- Menzies-Gow, N.J.; Stevens, K.B.; Sepulveda, M.F.; Jarvis, N.; Marr, C.M. Repeatability and reproducibility of the Obel grading system for equine laminitis. Vet. Rec. 2010, 167, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Viñuela-Fernández, I.; Jones, E.; Chase-Topping, M.E.; Price, J. Comparison of subjective scoring systems used to evaluate equine laminitis. Vet. J. 2011, 188, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Owens, J.G.; Kamerling, S.G.; Stanton, S.R.; Keowen, M.L. Effects of ketoprofen and phenylbutazone on chronic hoof pain and lameness in the horse. Equine Vet. J. 1995, 27, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Langford, D.J.; Bailey, A.L.; Chanda, M.L.; Clarke, S.E.; Drummond, T.E.; Echols, S.; Glick, S.; Ingrao, J.; Klassen-Ross, T.; Lacroix-Fralish, M.L.; et al. Coding of facial expressions of pain in the laboratory mouse. Nat. Methods 2010, 7, 447–449. [Google Scholar] [CrossRef] [PubMed]
- Leach, M.C.; Klaus, K.; Miller, A.L.; Scotto di Perrotolo, M.; Sotocinal, S.G.; Flecknell, P.A. The assessment of post-vasectomy pain in mice using behaviour and the Mouse Grimace Scale. PLoS ONE 2012, 7, e35656. [Google Scholar] [CrossRef] [PubMed]
- Sotocinal, S.G.; Sorge, R.E.; Zaloum, A.; Tuttle, A.H.; Martin, L.J.; Wieskopf, J.S.; Mapplebeck, J.C.S.; Wei, P.; Zhan, S.; Zhang, S.; et al. The Rat Grimace Scale: A partially automated method for quantifying pain in the laboratory rat via facial expressions. Mol. Pain 2011, 7, 55. [Google Scholar] [CrossRef] [PubMed]
- Keating, S.C.J.; Thomas, A.A.; Flecknell, P.A.; Leach, M.C. Evaluation of EMLA cream for preventing pain during tattooing of rabbits: Changes in physiological, behavioural and facial expression responses. PLoS ONE 2012, 7, e44437. [Google Scholar] [CrossRef] [PubMed]
- Gleerup, K.B.; Forkman, B.; Lindegaard, C.; Andersen, P.H. An equine pain face. Vet. Anaesth. Analg. 2015, 42, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Dalla Costa, E.; Minero, M.; Lebelt, D.; Stucke, D.; Canali, E.; Leach, M.C. Development of the Horse Grimace Scale (HGS) as a pain assessment tool in horses undergoing routine castration. PLoS ONE 2014, 9, e92281. [Google Scholar]
- Altman, D. Practical Statistics for Medical Research; Chapman & Hall/CRC Texts in Statistical Science: London, UK, 1991. [Google Scholar]
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.C. Facial expression of pain: An evolutionary account. Behav. Brain Sci. 2002, 25, 439–488. [Google Scholar] [CrossRef] [PubMed]
- Leach, M.C.; Coulter, C.A.; Richardson, C.A.; Flecknell, P.A. Are we looking in the wrong place? Implications for behavioural-based pain assessment in rabbits (Oryctolagus cuniculi) and beyond? PLoS ONE 2011, 6, e13347. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.L.; Leach, M.C. The mouse grimace scale: A clinically useful tool? PLoS ONE 2015, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Leach, M.; Allweiler, S.; Richardson, C.; Rough, J.; Narbe, R.; Flecknell, P. Behavioural effects of ovariohysterectomy and oral administration of meloxicam in laboratory housed rabbits. Res. Vet. Sci. 2009, 87, 336–347. [Google Scholar] [CrossRef] [PubMed]
- Minero, M.; Dalla Costa, E.; Dai, F.; Murray, L.A.M.; Canali, E.; Wemelsfelder, F. Use of qualitative behaviour assessment as an indicator of welfare in donkeys. Appl. Anim. Behav. Sci. 2016, 174, 147–153. [Google Scholar] [CrossRef]
- Wemelsfelder, F. How animals communicate quality of life: The qualitative assessment of behaviour. Anim. Welf. 2007, 16, 25–31. [Google Scholar]
- Bartlett, M.S.; Littlewort, G.C.; Frank, M.G.; Lee, K. Automatic decoding of facial movements reveals deceptive pain expressions. Curr. Biol. 2014, 24, 738–743. [Google Scholar] [CrossRef] [PubMed]
- Desrosiers, A.; Harrison, D.; Letham, A. Use of facial expressions for pain assessment in infants during acute painful procedures. Pediatr. Pain Lett. 2015, 17, 1–8. [Google Scholar]


| Obel Grade | Description |
|---|---|
| 0 | No gait abnormalities |
| 1 | The horse exhibits a normal gait at a walk. The trot shows a shortened stride with an audible cadence abnormality, but shows even head and neck lifting for each foot. |
| 2 | The walk is stilted, but shows no abnormal head or neck lifting. The trot shows obvious lameness with uneven head and neck lifting. A forefoot can be lifted off the ground easily. |
| 3 | The lameness is obvious at a walk and trot. The horse resists attempts to have a forefoot lifted and is reluctant to move. |
| 4 | The horse experiences difficulty bearing weight at rest or is very reluctant to move. |
| Horse ID | Breed | Gender | Age |
|---|---|---|---|
| 1 | pony | mare | 4 |
| 2 | Welsh Cob D | gelding | 8 |
| 3 | Icelandic Horse | mare | 4 |
| 4 | Shetland Pony | gelding | 13 |
| 5 | pony | gelding | 17 |
| 6 | Haflinger | mare | 15 |
| 7 | pony | mare | 15 |
| 8 | Haflinger | mare | 6 |
| 9 | Icelandic Horse | mare | 6 |
| 10 | pony | gelding | 6 |
| Facial Action Unit (FAU) | Description |
|---|---|
| Stiffly backwards ears | The ears are held stiffly and turned backwards; movements are limited also in presence of environmental stimuli |
| Orbital tightening | The eyelid is half-closed or closed, the orbit is contracted, eyes are not focused on the environment |
| Tension above the eye area | Increased muscle tension in the area above the eyes, the underlying bone structure becomes clearly visible |
| Prominent strained chewing muscles | Increased tension of the chewing muscles, that becomes prominent and clearly recognizable |
| Mouth strained and pronounced chin | Strained mouth, the corner of the lips is shortened, the lower lip is tense, the chin is contract and becomes more pronounced (crescent-shaped) |
| Strained nostrils and flattening of the profile | The nostrils are dilated and strained, the profile changes and you can see two bulges (one at the nostrils and upper lip) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dalla Costa, E.; Stucke, D.; Dai, F.; Minero, M.; Leach, M.C.; Lebelt, D. Using the Horse Grimace Scale (HGS) to Assess Pain Associated with Acute Laminitis in Horses (Equus caballus). Animals 2016, 6, 47. https://doi.org/10.3390/ani6080047
Dalla Costa E, Stucke D, Dai F, Minero M, Leach MC, Lebelt D. Using the Horse Grimace Scale (HGS) to Assess Pain Associated with Acute Laminitis in Horses (Equus caballus). Animals. 2016; 6(8):47. https://doi.org/10.3390/ani6080047
Chicago/Turabian StyleDalla Costa, Emanuela, Diana Stucke, Francesca Dai, Michela Minero, Matthew C. Leach, and Dirk Lebelt. 2016. "Using the Horse Grimace Scale (HGS) to Assess Pain Associated with Acute Laminitis in Horses (Equus caballus)" Animals 6, no. 8: 47. https://doi.org/10.3390/ani6080047
APA StyleDalla Costa, E., Stucke, D., Dai, F., Minero, M., Leach, M. C., & Lebelt, D. (2016). Using the Horse Grimace Scale (HGS) to Assess Pain Associated with Acute Laminitis in Horses (Equus caballus). Animals, 6(8), 47. https://doi.org/10.3390/ani6080047






