Heat Tolerance in Curraleiro Pe-Duro, Pantaneiro and Nelore Cattle Using Thermographic Images
Abstract
:1. Introduction
2. Experimental Section
3. Results
| Breed | Body Area | Neck | Rump | Muzzle | Head | Groin | Axilla | HR | RR | RT |
|---|---|---|---|---|---|---|---|---|---|---|
| Pan | 36.67 a | 36.49 a | 36.50 a | 34.19 | 36.50 a | 36.16 a | 36.07 a | 117.52 a | 43.33 b | 39.05 b |
| Nel | 36.45 a | 36.41 a | 36.83 a | 34.31 | 36.35 a | 36.33 a | 36.43 a | 103.91 b | 42.89 b | 39.43 a |
| Curr | 35.34 b | 35.36 b | 35.14 b | 33.5 | 35.38 b | 34.79 b | 35.14 b | 117.37 a | 46.55 a | 38.63 c |
| SEM | 0.15 | 0.15 | 0.19 | 0.33 | 0.22 | 0.16 | 0.16 | 1.15 | 0.56 | 0.03 |
| Pr > F | *** | * | ** | * | ** | *** | * | *** | *** | *** |
| RV | 36.0–60.0 | 26.0–50.0 | 36.7–39.1 |
| Breeds | Body Area | Neck | Rump | Muzzle | Head | Groin | Axilla | HR | RR | RT |
|---|---|---|---|---|---|---|---|---|---|---|
| Morning | ||||||||||
| Pan | 34.50 a | 34.91 a | 33.94 b | 30.95 a | 33.64 a | 34.13 a | 33.11 b | 115.20 a | 39.20 b | 38.82 b |
| Nel | 34.89 a | 34.91 a | 35.19 a | 32.22 a | 34.30 a | 34.69 a | 34.93 a | 103.82 b | 42.04 a | 39.37 a |
| Curr | 32.87 b | 33.22 b | 31.77 c | 29.07 b | 31.64 b | 32.22 b | 32.74 c | 118.48 a | 42.75 a | 38.30 c |
| SEM | 0.15 | 0.14 | 0.21 | 0.33 | 0.23 | 0.17 | 0.16 | 1.69 | 0.66 | 0.05 |
| Pr > F | *** | *** | *** | *** | *** | *** | *** | *** | ** | *** |
| Afternoon | ||||||||||
| Pan | 38.84 a | 38.50 a | 39.05 | 37.65 | 39.57 a | 38.25 a | 38.35 a | 119.55 a | 47.46 ab | 39.29 b |
| Nel | 38.02 b | 37.91 ab | 38.47 | 36.47 | 38.49 b | 37.98 ab | 37.93 ab | 104.00 b | 43.73 b | 39.48 a |
| Curr | 37.81 b | 37.50 b | 38.52 | 37.67 | 38.71 b | 37.36 b | 37.54 b | 116.55 a | 50.35 a | 38.96 c |
| SEM | 0.11 | 0.14 | 0.13 | 0.40 | 0.14 | 0.11 | 0.13 | 1.85 | 0.84 | 0.03 |
| Pr > F | ** | ** | NS | NS | ** | ** | * | *** | ** | *** |
| Time | Mean | |||||||||
| Morning | 34.08 b | 34.20 b | 33.64 b | 30.79 b | 33.25 b | 33.68 b | 33.82 b | 112.50 | 41.33 b | 38.83 b |
| Afternoon | 38.22 a | 37.97 a | 38.68 a | 37.25 a | 38.91 a | 37.86 a | 37.94 a | 113.17 | 47.18 a | 39.24 a |
| SEM | 0.11 | 0.12 | 0.14 | 0.34 | 0.16 | 0.12 | 0.12 | 1.18 | 0.66 | 0.03 |
| Pr > F | *** | *** | *** | *** | *** | *** | *** | NS | *** | *** |
| Soil | Air | RH | WS | BGshade | BGSun | THI | Body Area | Neck | Muzzle | Head | Groin | Rump | Axilla | HR | RR | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Air | 0.78 *** | |||||||||||||||
| RH | −0.73 *** | −0.97 *** | ||||||||||||||
| WS | 0.75 *** | 0.61 *** | −0.58 *** | |||||||||||||
| BGShade | 0.82 *** | 0.99 *** | −0.97 *** | 0.66 *** | ||||||||||||
| BGSun | 0.51 *** | 0.79 *** | −0.82 *** | 0.41 *** | 0.78 *** | |||||||||||
| THI | 0.79 *** | 0.97 *** | −0.90 *** | 0.60 *** | 0.96 *** | 0.74 *** | ||||||||||
| Body Area | 0.84 *** | 0.86 *** | −0.83 *** | 0.60 *** | 0.88 *** | 0.66 *** | 0.84 *** | |||||||||
| Neck | 0.78 *** | 0.82 *** | −0.79 *** | 0.57 *** | 0.83 *** | 0.64 *** | 0.79 *** | 0.93 *** | ||||||||
| Muzzle | 0.65 *** | 0.67 *** | −0.64 *** | 0.49 *** | 0.68 *** | 0.50 *** | 0.65 *** | 0.69 *** | 0.63 *** | |||||||
| Head | 0.82 *** | 0.84 *** | −0.82 *** | 0.59 *** | 0.86 *** | 0.64 *** | 0.82 *** | 0.93 *** | 0.88 *** | 0.68 *** | ||||||
| Groin | 0.77 *** | 0.87 *** | −0.85 *** | 0.53 *** | 0.88 *** | 0.68 *** | 0.84 *** | 0.92 *** | 0.87 *** | 0.64 *** | 0.87 *** | |||||
| Rump | 0.85 *** | 0.86 *** | −0.83 *** | 0.60 *** | 0.88 *** | 0.68 *** | 0.85 *** | 0.95 *** | 0.87 *** | 0.68 *** | 0.91 *** | 0.89 *** | ||||
| Axilla | 0.81 *** | 0.83 *** | −0.80 *** | 0.57 *** | 0.85 *** | 0.64 *** | 0.81 *** | 0.93 *** | 0.88 *** | 0.64 *** | 0.87 *** | 0.89 *** | 0.89 *** | |||
| HR | 0.14 ** | −0.004 NS | 0.04 NS | 0.11 NS | 0.01 NS | −0.21 ** | 0.03 NS | 0.08 NS | 0.04 NS | −0.02 NS | 0.07 NS | 0.06 NS | 0.06 NS | 0.07 NS | ||
| RR | 0.31 *** | 0.31 *** | −0.29 *** | 0.26 *** | 0.32 *** | 0.21 ** | 0.30 *** | 0.40 *** | 0.36 *** | 0.32 *** | 0.40 *** | 0.33 *** | 0.37 *** | 0.38 *** | 0.15 ** | |
| RT | 0.47 *** | 0.57 *** | −0.53 *** | 0.21 ** | 0.57 *** | 0.56 *** | 0.61 *** | 0.58 *** | 0.57 *** | 0.33 *** | 0.53 *** | 0.59 *** | 0.59 *** | 0.53 *** | 0.05 NS | 0.21 ** |
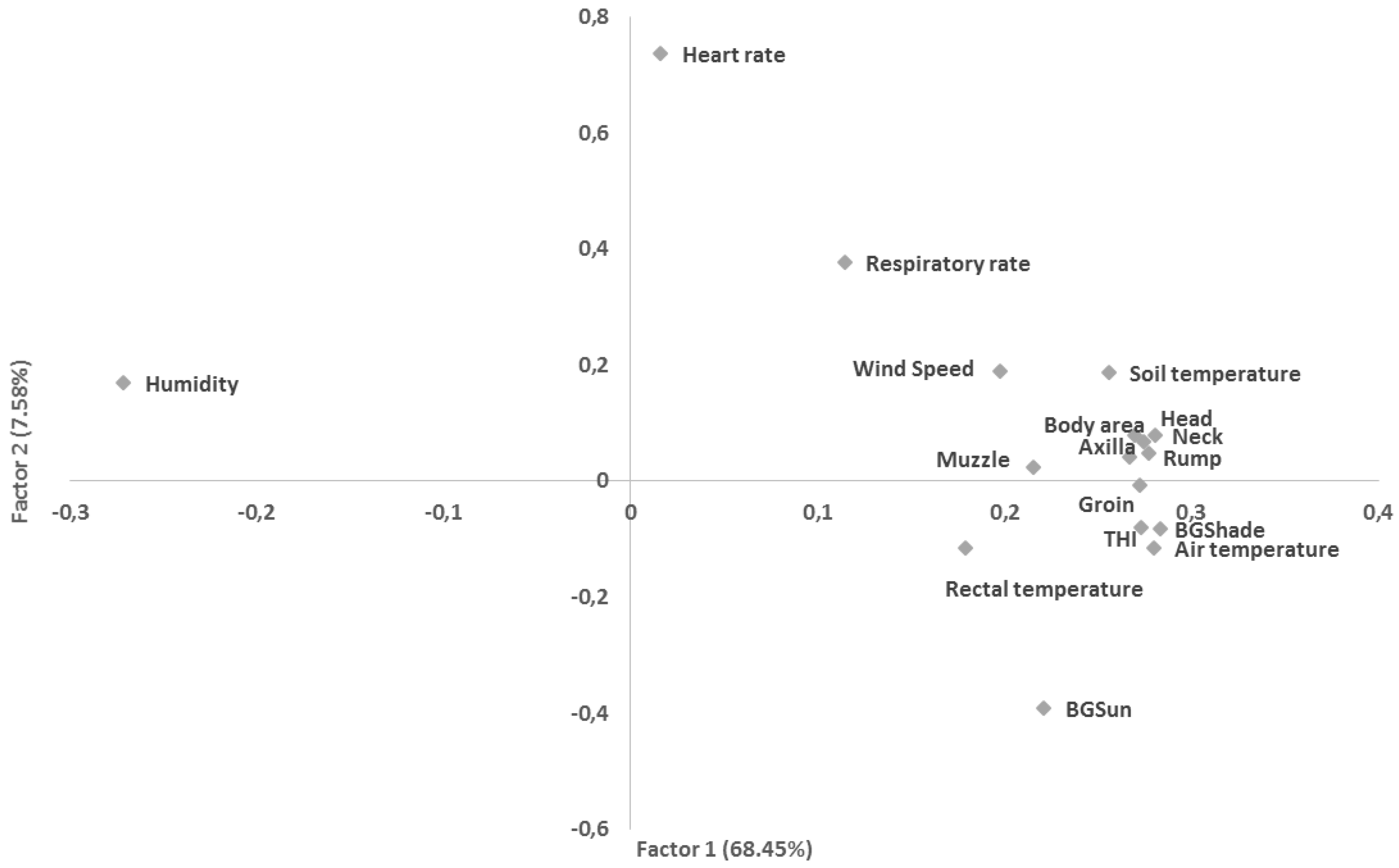
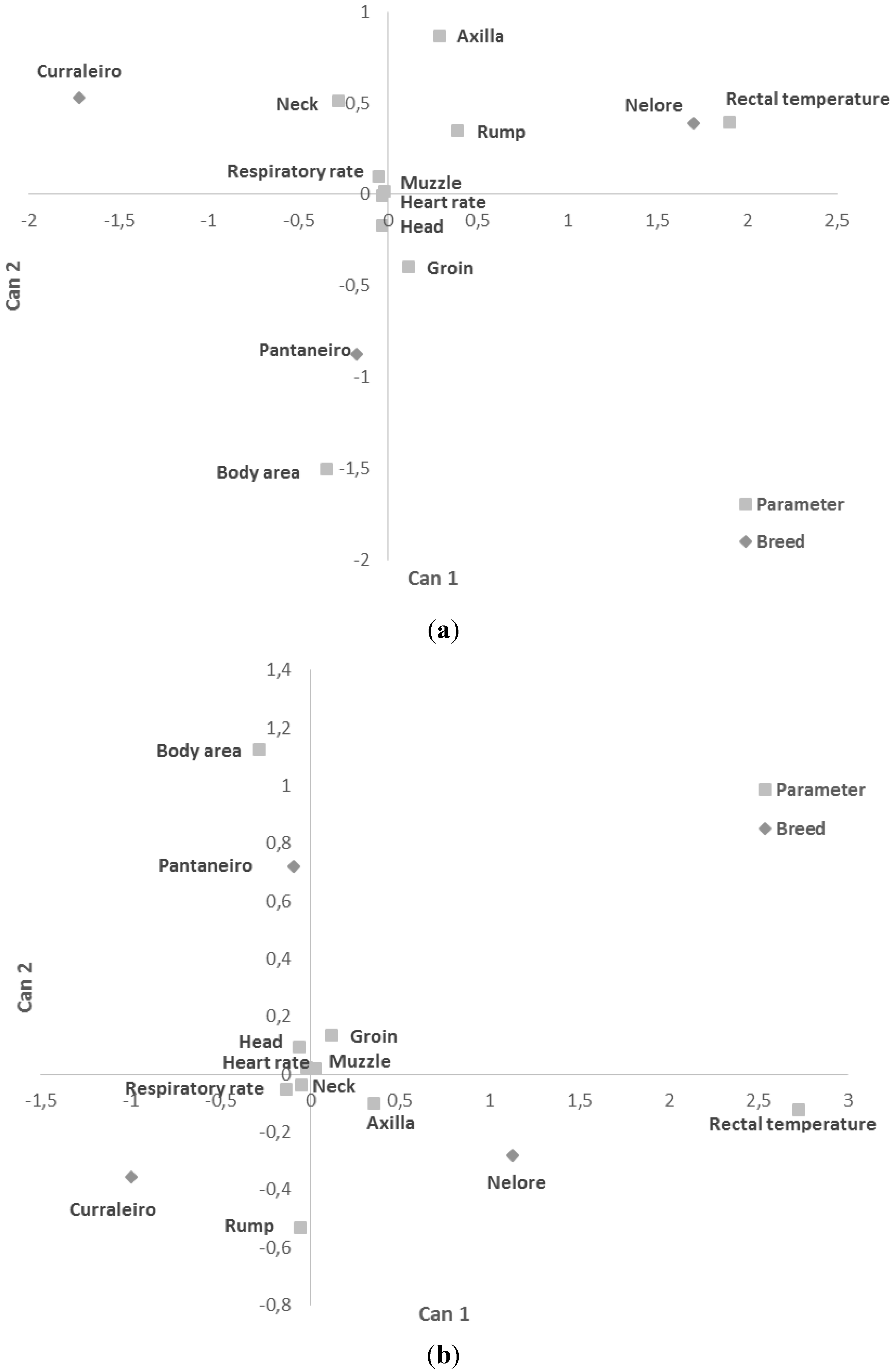
| Group | RR | HR | RT |
|---|---|---|---|
| Morning | |||
| Curraleiro | 78 | 82 | 0 |
| Nelore | 89 | 87 | 29 |
| Pantaneiro | 100 | 76 | 7 |
| Afternoon | |||
| Curraleiro | 96 | 98 | 2 |
| Nelore | 98 | 91 | 40 |
| Pantaneiro | 100 | 98 | 18 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Amaral, B.; Connor, E.; Tao, S.; Hayen, M.; Bubolz, J.; Dahl, G. Heat stress abatement during the dry period influences metabolic gene expression and improves immune status in the transition period of dairy cows. J. Dairy Sci. 2011, 94, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Scholtz, M.; McManus, C.; Leeuw, K.; Louvandini, H.; Seixas, L.; Melo, C.D.; Theunissen, A.; Neser, F. The effect of global warming on beef production in developing countries of the southern hemisphere. Natural Sci. 2013, 5, 106–119. [Google Scholar] [CrossRef]
- Marai, I.; el-Darawany, A.; Fadiel, A.; Abdel-Hafez, M. Physiological traits as affected by heat stress in sheep—A review. Small Rum. Res. 2007, 71, 1–12. [Google Scholar] [CrossRef]
- Delfino, L.J.; Souza, B.B.D.; Silva, R.; Silva, W.W. Efeito do estresse calórico sobre o eritrograma de ruminantes. Agrop. Cient. Semiár. 2012, 8, 01–07. (In Portuguese) [Google Scholar]
- West, J. Effects of heat-stress on production in dairy cattle. J. Dairy Sci. 2003, 86, 2131–2144. [Google Scholar] [CrossRef]
- McManus, C.; Prescott, E.; Paludo, G.; Bianchini, E.; Louvandini, H.; Mariante, A. Heat tolerance in naturalized Brazilian cattle breeds. Livest. Sci. 2009, 120, 256–264. [Google Scholar] [CrossRef]
- Baccari Júnior, F. Métodos e técnicas de avaliação da adaptabilidade dos animais às condições tropicais. Simp. Int. Bioclimatol. Anim. Tróp. 1990, 1, 9–17. (In Portuguese) [Google Scholar]
- Mazza, M.C.M.; Mazza, C.D.S.; Sereno, J.; Santos, S. Pellegrin.etnobiologia e conservação do bovino pantaneiro; Embrapa-CPAP: Corumbá, Brasil, 1994. (In Portuguese) [Google Scholar]
- Pellegrin, A.O.; Sereno, J.R.B.; Mazza, M.C.M.; Leite, R.C. Doenças da reprodução e conservação genética: Levantamento no núcleo de conservação do bovino Pantaneiro. In Comunicado Técnico Embrapa Pantanal; Embrapa Pantanal: Corumbá, Brasil, 1997. [Google Scholar]
- McManus, C.; Paludo, G.; Louvandini, H.; Garcia, J.; Egito, A.; Mariante, A. Heat tolerance in naturalized cattle in Brazil: Physical factors. Arch. Zootec. 2005, 54, 453–458. [Google Scholar]
- Fioravanti, M.C.S.; Juliano, R.S.; Sereno, J.R.B.; Magnabosco, C.U.; Barbosa, V.; Abud, L.J.; Costa, G.L.; Costa, M.F.O. Bovino Curraleiro. In INCT: Informação Genético-Sanitária da Pecuária Brasileira; Brasília, Brasil, 2010. (In Portuguese) [Google Scholar]
- Castanheira, M.; McManus, C.M.; Neto, P.; Costa, M.J.R.P.D.; Mendes, F.D.C.; Sereno, J.R.B.; Bértoli, C.D.; Fioravanti, M.C.S. Maternal offspring behaviour in Curraleiro Pé Duro naturalized cattle in Brazil. Rev. Bras. Zootec. 2013, 42, 584–591. [Google Scholar] [CrossRef]
- Santos, S.; Silva, R.; Comastri-Filho, J.; de Abreu, U.; McManus, C.; Mariante, A.D.S.; Lara, M.; Pellegrin, A.; Ravaglia, E. Desempenho de bezerros Pantaneiros, Nelore e cruzados, criados no Pantanal, Brasil. Arch. Zootec. 2005, 54, 501–508. (In Portuguese) [Google Scholar]
- Bianchini, E.; McManus, C.; Lucci, C.M.; Fernandes, M.C.B.; Prescott, E.; Mariante, A.D.S.; Egito, A.D. Características corporais associadas com a adaptação ao calor em bovinos naturalizados brasileiros. Pesq. Agrop. Bras. 2006, 41, 1413–1448. (In Portuguese) [Google Scholar] [CrossRef]
- Costa, A.N.L.; Feitosa, J.V.; Montezuma, P.A., Jr.; Souza, P.T.; Araújo, A. Rectal temperatures, respiratory rates, production, and reproduction performances of crossbred Girolando cows under heat stress in northeastern Brazil. Int. J. Biometeorol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mazieiro, R.R.D.; Martin, I.; Mattos, M.C.C.; Ferreira, J.C.P. Avaliação das concentrações plasmáticas de cortisol e progesterona em vacas nelore (Bos taurus indicus) submetidas a manejo diário ou manejo semanal. Vet. Zootec. 2012, 19, 366–372. (In Portuguese) [Google Scholar]
- George, W.D.; Godfrey, R.W.; Ketring, R.C.; Vinson, M.C.; Willard, S.T. Relationship among eye and muzzle temperatures measured using digital infrared thermal imaging and vaginal and rectal temperatures in hair sheep and cattle. J. Anim. Sci. 2014, 92, 4949–4955. [Google Scholar] [CrossRef] [PubMed]
- Stewart, M.; Webster, J.R.; Schaefer, A.L.; Cook, N.J.; Scott, S.L. Infrared thermography as a non-invasive tool to study animal welfare. Anim. Welfare 2005, 14, 319–325. [Google Scholar]
- National Research Council. A Guide to Environmental Research on Animals; National Academy of Science: Washington, DC, USA, 1971; p. 374. [Google Scholar]
- Reece, W.O.; Erickson, H.H.; Goff, J.P.; Uemura, E.E. Dukes’ Physiology of Domestic Animals, 13th ed.; Wiley-Blackwell: Oxford, UK, 2015; p. 760. [Google Scholar]
- Paim, T.; Borges, B.O.; Lima, P.M.T.; Gomes, E.F.; Dallago, B.S.L.; Fadel, R.; Menezes, A.M.; Louvandini, H.; Canozzi, M.E.A.; Barcellos, J.O.J.; et al. Thermographic evaluation of climatic conditions on lambs from different genetic groups. Int. J. Biometeorol. 2013, 57, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Nienaber, J.A.; Hahn, G.L. Livestock production system management responses to thermal challenges. Int. J. Biometeorol. 2007, 52, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Abi Saab, S.; Sleiman, F.T. Physiological responses to stress of filial crosses compared to local Awassi sheep. Small Rum. Res. 1995, 16, 55–59. [Google Scholar] [CrossRef]
- Stewart, M.; Webster, J.; Verkerk, G.; Schaefer, A.; Colyn, J.; Stafford, K. Non-invasive measurement of stress in dairy cows using infrared thermography. Physiol. Behav. 2007, 92, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Church, J.S.; Hegadoren, P.R.; Paetkau, M.J.; Miller, C.C.; Regev-Shoshani, G.; Schaefer, A.L.; Schwartzkopf-Genswein, K.S. Influence of environmental factors on infrared eye temperature measurements in cattle. Res. Vet. Sci. 2014, 96, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Radostits, O.M.; Gay, C.C.; Blood, D.C.; Hinchcliff, K.W. Clínica Veterinária: Um Tratado de Doenças dos Bovinos, Ovinos, Suínos, Caprinos e Eqüinos, pp. 677–680, 9th ed.; Guanabara Koogan: Rio de Janeiro, Brazil, 2002; p. 1737. (In Portuguese) [Google Scholar]
- Fitzhugh, H. Animal size and efficiency, with special reference to the breeding female. Anim. Prod. 1978, 27, 393–401. [Google Scholar] [CrossRef]
- Silva, R.G.D.; Maia, A.S.C. Evaporative cooling and cutaneous surface temperature of Holstein cows in tropical conditions. Rev. Bras. Zootec. 2011, 40, 1143–1147. [Google Scholar] [CrossRef]
- McManus, C.M.; Castanheira, M.; Paiva, S.R.; Louvandini, H.; Fioravanti, M.C.; Paludo, G.R.; Bianchini, E.; Corrêa, P.S. Use of multivariate analyses for determining heat tolerance in Brazilian cattle. Trop. Anim. Health Prod. 2011, 43, 623–630. [Google Scholar] [CrossRef] [PubMed]
- McManus, C.M.; Louvandini, H.; Paim, T.P.; Bernal, F.E.M. Factors affecting heat tolerance in crossbred cattle in central Brazil. Cienc. Anim. Bras. 2014, 15, 152–158. [Google Scholar] [CrossRef]
- Swenson, M.J.; Reece, W.O. Fisiologia dos Animais Domésticos, 11th ed.; Guanabara Koogan: Rio de Janeiro, RJ, Brazil, 1996; pp. 532–537. [Google Scholar]
- Grandin, T. Assessment of stress during handling and transport. J. Anim. Sci. 1997, 75, 249–257. [Google Scholar] [PubMed]
- Despopoulos, A.; Silbernagl, S. Color Atlas of Physiology; Thieme: Stuttgart, Germany, 2003. [Google Scholar]
- Knizkova, I.; Kunc, P.; Gürdil, K.A.G.; Pınar, Y.; Selvi, Ç.K. Applications of infrared thermography in animal production. J. Fac. Agric. 2007, 22, 329–336. [Google Scholar]
- Neiva, J.N.M.; Teixeira, M.; Turco, S.H.N.; Oliveira, S.M.P.; Moura, A.A.A.N. Efeito do estresse climático sobre os parâmetros produtivos e fisiológicos de ovinos Santa Inês mantidos em confinamento na região litorânea do nordeste do Brasil. Rev. Bras. Zootec. 2004, 33, 668–678. (In Portuguese) [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardoso, C.C.; Lima, F.G.; Fioravanti, M.C.S.; Egito, A.A.d.; Silva, F.C.d.P.e.; Tanure, C.B.; Peripolli, V.; McManus, C. Heat Tolerance in Curraleiro Pe-Duro, Pantaneiro and Nelore Cattle Using Thermographic Images. Animals 2016, 6, 9. https://doi.org/10.3390/ani6020009
Cardoso CC, Lima FG, Fioravanti MCS, Egito AAd, Silva FCdPe, Tanure CB, Peripolli V, McManus C. Heat Tolerance in Curraleiro Pe-Duro, Pantaneiro and Nelore Cattle Using Thermographic Images. Animals. 2016; 6(2):9. https://doi.org/10.3390/ani6020009
Chicago/Turabian StyleCardoso, Caio Cesar, Flávia Gontijo Lima, Maria Clorinda Soares Fioravanti, Andrea Alves do Egito, Flávia Cristina de Paula e Silva, Candice Bergmann Tanure, Vanessa Peripolli, and Concepta McManus. 2016. "Heat Tolerance in Curraleiro Pe-Duro, Pantaneiro and Nelore Cattle Using Thermographic Images" Animals 6, no. 2: 9. https://doi.org/10.3390/ani6020009
APA StyleCardoso, C. C., Lima, F. G., Fioravanti, M. C. S., Egito, A. A. d., Silva, F. C. d. P. e., Tanure, C. B., Peripolli, V., & McManus, C. (2016). Heat Tolerance in Curraleiro Pe-Duro, Pantaneiro and Nelore Cattle Using Thermographic Images. Animals, 6(2), 9. https://doi.org/10.3390/ani6020009





