Double Muscling in Cattle: Genes, Husbandry, Carcasses and Meat
Simple Summary
Abstract
1. Introduction
2. Origin of DM Animals: The Unraveling of the Myostatin Gene and Its Expression
3. Consequences of the Inactivation of Myostatin on Conformation and Carcass Quality
3.1. Impact of Double Muscling on Conformation and Carcass Quality
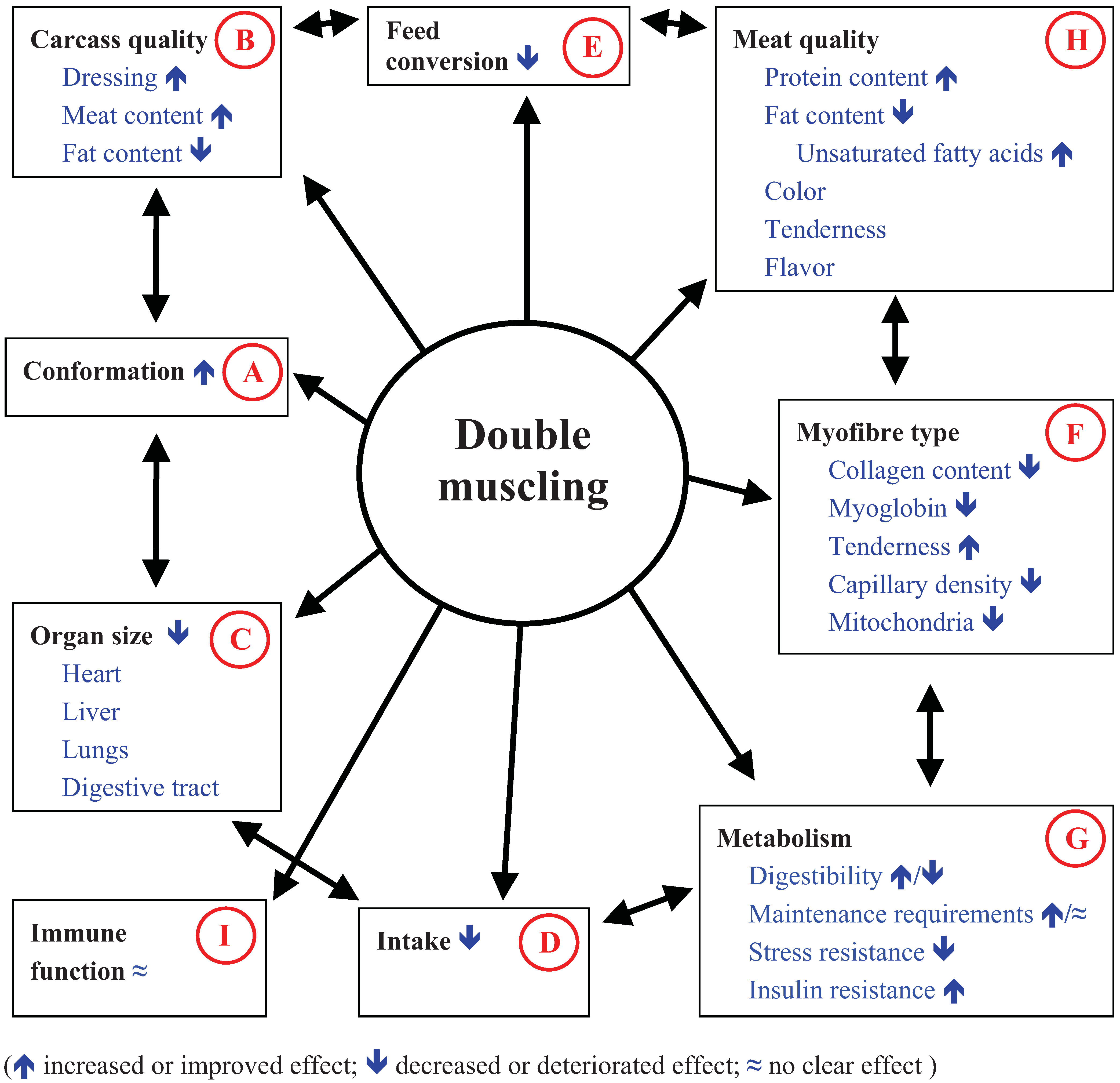
3.2. Impact of Double Muscling on Organ Size
3.3. Consequences of Double Muscling for Locomotion
3.4. Impact of Double Muscling on Reproduction Performance
3.5. Influence of Double Muscling on Histological Aspects of Skeletal Muscle
3.6. Influence of Double Muscling on Metabolism
3.7. Influence of Double Muscling on Stress Susceptibility
3.8. Effect of Double Muscling on Meat Quality
4. Conclusions
Conflict of Interest
References
- Berg, R.T.; Walters, L.E. The meat animal: Changes and challenges. J. Anim. Sci. 1983, 57, 133–146. [Google Scholar]
- Kempster, A.J. Carcass and meat quality research to meet market needs. Anim. Prod. 1989, 48, 483–496. [Google Scholar] [CrossRef]
- Vanek, J.K.; Watts, M.J.; Brester, G.W. Carcass quality and genetic selection in the beef industry. J. Agric. Res. Econ. 2008, 33, 349–363. [Google Scholar]
- Kempster, A.J.; Cuthbertson, A.; Harrington, G. The relationship between conformation and the yield and distribution of lean meat in the carcasses of British pigs, cattle and sheep: A review. Meat Sci. 1982, 6, 37–53. [Google Scholar] [CrossRef]
- Luciano, F.B. The impacts of lean red meat consumption on human health: A review. J. Food 2009, 7, 143–151. [Google Scholar]
- Geay, Y.; Robelin, J.; Vermorel, M.; Beranger, C. Muscle hypertrophy and energy utilization in cattle: The double muscled as an extreme or a deviant animal. In Muscle Hypertrophy of Genetic Origin and Its Use to Improve Beef Production; King, J.W.B., Menissier, F., Eds.; Martinus Nijhoff Publishers: The Hague, The Netherlands, 1982; pp. 74–87. [Google Scholar]
- Vissac, B. L’hypertrophie musculaire d’origine génétique ou caractère culard. Ann. Génét. Sél. Anim. 1972, 4, 87–97. [Google Scholar]
- Guo, K.J.; Liu, F.H.; Lu, L.; Biagini, D.; Lazzaroni, C.; Zoccarato, I. Productive performance of double-muscled Piemontese culled cows in finishing period. Agric. Sci. China 2010, 9, 721–728. [Google Scholar] [CrossRef]
- Kambadur, R.; Sharma, M.; Smith, T.P.L.; Bass, J.J. Mutations in myostatin (GDF8) in double-muscled Belgian Blue and Piedmontese cattle. Genome Res. 1997, 7, 910–915. [Google Scholar]
- Caballero, B.; Sierra, V.; Oliván, M.; Vega-Naredo, I.; Tomás-Zapico, C.; Alvarez-García, O.; Tolivia, D.; Hardeland, R.; Rodríguez-Colunga, M.J.; Coto-Montes, A. Activity of cathepsins during beef aging related to mutations in the myostatin gene. J. Sci. Food Agric. 2007, 87, 192–199. [Google Scholar]
- Marchitelli, C.; Savarese, M.C.; Crisà, A.; Nardone, A.; Marsan, P.A.; Valentini, A. Double muscling in Marchigiana beef breed is caused by a stop codon in the third exon of myostatin gene. Mamm. Genome 2003, 14, 392–395. [Google Scholar] [CrossRef]
- Darwin, C. The Origin of Species by Means of Natural Selection; John Murray: London, UK, 1859. [Google Scholar]
- Arthur, P.F. Double muscling in cattle: A review. Austr. J. Agric. Res. 1995, 46, 1493–1515. [Google Scholar] [CrossRef]
- Kaiser. Uber die sogenannten doppellendigen Rinder. Landw. Jhrb. 1888, 17, 387–403. [Google Scholar]
- Charlier, C.; Coppieters, W.; Farnir, F.; Grobet, L.; Leroy, P.L.; Michaux, C.; Mni, M.; Schwers, A.; Vanmanshoven, P.; Hanset, R.; Georges, M. The mh gene causing double-muscling in cattle maps to bovine Chromosome 2. Mamm. Genome 1995, 6, 788–792. [Google Scholar] [CrossRef]
- Grobet, L.; Royo Martin, L.J.; Poncelet, D.; Pirottin, D.; Brouwers, B.; Riquet, J.; Schoeberlein, A.; Dunner, S.; Menissier, F.; Massabanda, J.; Fries, R.; Hanset, R.; Georges, M. A deletion in the myostatin gene causes double muscled phenotype in cattle. Nat. Genet. 1997, 17, 71–74. [Google Scholar] [CrossRef]
- McPherron, A.C.; Lee, S.J. Double muscling in cattle due to mutations in the myostatin gene. Proc. Natl. Acad. Sci. USA 1997, 94, 12457–12461. [Google Scholar] [CrossRef]
- Chelh, I.; Picard, B.; Hocquette, J.-F.; Cassar-Malek, I. Myostatin inactivation induces a similar muscle molecular signature in double-muscled cattle as in mice. Animal 2010, 5, 278–286. [Google Scholar]
- Gerrard, D.E.; Grant, A.L.; Anderson, D.B.; Lemenager, R.P.; Judge, M.D. In-vivo analysis of serum-borne growth factors in developing co-twinned fetuses. J. Anim. Sci. 1995, 73, 1689–1693. [Google Scholar]
- Lee, S.J.; McPherron, A.C. Regulation of myostatin activity and muscle growth. Proc. Natl. Acad. Sci. USA 2001, 98, 9306–9311. [Google Scholar] [CrossRef]
- Hill, J.J.; Quiu, Y.; Hewick, R.N.; Wolfman, N.M. Regulation of myostatin in vivo by GASP-1: A novel protein with protease inhibitor and follistatin domains. Mol. Endocrinol. 2003, 17, 1144–1154. [Google Scholar] [CrossRef]
- Zimmers, T.A.; Davies, M.V.; Koniaris, L.G.; Haynes, P.; Esquela, A.F.; Tomkinson, K.N.; McPherron, A.C.; Wolfman, N.M.; Lee, S.J. Induction of cachexia in mice by systemically administered myostatin. Science 2002, 296, 1486–1488. [Google Scholar] [CrossRef]
- Joulia-Ekaza, D.; Cabello, G. Myostatin regulation of muscle development: Molecular basis, natural mutations, physiopathological aspects. Exp. Cell Res. 2006, 312, 2401–2414. [Google Scholar] [CrossRef]
- Wolfman, N.M.; McPherron, A.C.; Pappano, W.N.; Davies, M.V.; Song, K.; Tomkinson, K.N.; Wright, J.F.; Zhao, L.; Sebald, S.M.; Greenspan, D.S.; Lee, S.J. Activation of latent myostatin by the BMP-1/tolloid family of metalloproteinases. Proc. Natl. Acad. Sci. USA 2003, 100, 15842–15846. [Google Scholar]
- Rebbapragada, A.; Benchabane, H.; Wrana, J.L.; Celeste, A.J.; Attisano, L. Myostatin signals through a transforming growth factor β-like signaling pathway to block adipogenesis. Mol. Cell. Biol. 2003, 23, 7230–7242. [Google Scholar] [CrossRef]
- Forbes, D.; Jackman, M.; Bishop, A.; Thomas, M.; Kambadur, R.; Sharma, M. Myostatin auto-regulates its expression by feedback loop through Smad7 dependent mechanism. J. Cell. Physiol. 2006, 206, 264–272. [Google Scholar] [CrossRef]
- Bouley, J.; Meunier, B.; Chambon, C.; De Smet, S.; Hocquette, J.F.; Picard, B. Proteomic analysis of bovine skeletal muscle hypertrophy. Proteomics 2005, 5, 490–500. [Google Scholar] [CrossRef]
- Grobet, L.; Poncelet, D.; Royo, L.J.; Brouwers, B.; Pirottin, D.; Michaux, C.; Menissier, F.; Zanotti, M.; Dunner, S.; Georges, M. Molecular definition of an allelic series of mutations disrupting the myostatin function and causing double-muscling in cattle. Mamm. Genome 1998, 9, 210–213. [Google Scholar] [CrossRef]
- Yang, W.; Zhang, Y.; Li, Y.; Wu, Z.; Zhu, D. Myostatin induces cyclin D1 degradation to cause cell cycle arrest through a phosphatidylinositol 3-kinase/AKT/GSK-3β pathway and is antagonized by insulin-like growth factor 1. J. Biol. Chem. 2007, 282, 3799–3808. [Google Scholar]
- Gerrard, D.E.; Grant, A.L. Insulin-like growth factor-II expression in developing skeletal muscle of double muscled and normal cattle. Dom. Anim. Endocrinol. 1994, 11, 339–347. [Google Scholar] [CrossRef]
- Listrat, A.; Belair, L.; Picard, B.; Boulle, N.; Geay, Y.; Djiane, J.; Jammes, H. Insulin-like growth factor II (IGF-II) mRNA expression during skeletal muscle development of double-muscled and normal bovine fetuses. Reprod. Nutr. Dev. 1999, 39, 113–124. [Google Scholar] [CrossRef]
- Kamanga-Sollo, E.; Pampusch, M.S.; White, M.E.; Hathaway, M.R.; Dayton, W.R. Insulin-like growth factor binding protein (IGFBP)-3 and IGFBP-5 mediate TGF-h- and myostatin-induced suppression of proliferation in porcine embryonic myogenic cell cultures. Exp. Cell Res. 2005, 311, 167–176. [Google Scholar] [CrossRef]
- Williams, N.G.; Interlichia, J.P.; Jackson, M.F.; Hwang, D.; Cohen, P.; Rodgers, B.D. Endocrine actions of myostatin: Systemic regulation of the IGF and IGF binding protein axis. Endocrinology 2011, 152, 172–180. [Google Scholar] [CrossRef]
- Ludolph, D.C.; Komeczny, S.F. Transcription factor families: Muscling in on the myogenic program. FASEB J. 1995, 9, 1595–1604. [Google Scholar]
- Arnold, H.H.; Braun, T. Genetics of muscle determination and development. Curr. Top. Dev. Biol. 2000, 48, 129–164. [Google Scholar] [CrossRef]
- Bass, J.; Oldham, J.; Sharma, M.; Kambadur, R. Growth factors controlling muscle development. Dom. Anim. Endocrinol. 1999, 17, 191–197. [Google Scholar] [CrossRef]
- Oldham, J.; Martyn, J.A.K.; Sharma, M.; Jeanplong, F.; Kambadur, R.; Bass, J.J. Molecular expression of myostatin and MyoD is greater in double-muscled than normal-muscled cattle fetuses. Am. J. Physiol. 2001, 280, R1488–R1493. [Google Scholar]
- Langley, B.; Thomas, M.; Bishop, A.; Sharma, M.; Gilmour, S.; Kambadur, R. Myostatin inhibits myoblast differentiation by down-regulating MyoD expression. J. Biol. Chem. 2002, 277, 49831–49840. [Google Scholar]
- Muroya, S.; Watanabe, K.; Hayashi, S.; Miyake, M.; Konashi, S.; Sato, Y.; Takahashi, M.; Kawahata, S.; Yoshikawa, Y.; Aso, H.; Chikuni, K.; Yamaguchi, T. Muscle type-specific effect of myostatin deficiency on myogenic regulatory factor expression in adult double-muscled Japanese Shorthorn cattle. Anim. Sci. J. 2009, 80, 678–685. [Google Scholar] [CrossRef]
- Michel, R.N.; Dunn, S.E.; Chin, E.R. Calcineurin and skeletal muscle growth. Proc. Nutr. Soc. 2004, 63, 341–349. [Google Scholar] [CrossRef]
- Ashmore, C.R.; Parker, W.; Stokes, H.; Doerr, L. Comparative aspects of muscle fiber types in fetuses of the normal and “double-muscled” cattle. Growth 1974, 38, 501–506. [Google Scholar]
- Uytterhaegen, L.; Claeys, E.; Demeyer, D.; Lippens, M.; Fiems, L.O.; Boucqué, C.V.; Van de Voorde, G.; Bastiaens, A. Effects of double-muscling on carcass quality, beef tenderness and myofibrillar protein degradation in Belgian Blue White bulls. Meat Sci. 1994, 38, 255–267. [Google Scholar] [CrossRef]
- Dumont, B.L. Carcass composition and muscle structure in hypertrophied animals. In Muscle Hypertrophy of Genetic Origin and Its Use to Improve Beef Production; King, J.W.B., Menissier, F., Eds.; Martinus Nijhoff Publishers: The Hague, The Netherlands, 1982; pp. 111–133. [Google Scholar]
- Ansay, M.; Hanset, R. Anatomical, physiological and biochemical differences between conventional and double-muscled cattle in the Belgian Blue and White Breed. Livest. Prod. Sci. 1979, 6, 5–13. [Google Scholar] [CrossRef]
- Arthur, P.F.; Makarechian, M.; Price, M.A.; Berg, R.T. Heterosis, maternal and direct effects in double-muscled and normal cattle: II. Carcass traits of young bulls. J. Anim. Sci. 1989, 67, 911–919. [Google Scholar]
- Fiems, L.O.; Van Hoof, J.; Uytterhaegen, L.; Boucqué, C.V.; Demeyer, D.I. Comparative quality of meat from double-muscled and normal beef cattle. In Expression of tissue proteinases and regulation of protein degradation as related to meat quality; Ouali, A., Demeyer, D.I., Smulders, F.J.M., Eds.; Ecceamst: Utrecht, The Netherlands, 1995; pp. 381–393. [Google Scholar]
- Determining the Community Scale for the Classification of Carcasses of Adult Bovine Animals. Council Regulation (EEC) No 1026/91 of 22 April 1991, Amending Regulation Council Regulation (EEC) No 1208/81. Official J. Eur. Comm. 1991, L106, 2–3.
- Vissac, B. Étude du caractère culard. II. Incidence du caractère culard sur la morphologie générale des bovins. Ann. Zootech. 1968, 17, 77–101. [Google Scholar] [CrossRef]
- Vermorel, M.; Ortigues, I.; Vernet, J.; Geay, Y.; Jailler, R.; Jailler, R.; Baumont, R.; Hocquette, J.F.; Deswysen, A.G. Energy metabolism in normal and double-muscled Belgian Blue calves in relation with body composition and organ size. In Energy Metabolism of Farm Animals; Aguilera, J.F., Ed.; Consejo Superior de Investigaciones: Granada, Spain, 1994; pp. 209–211. [Google Scholar]
- Bünger, L.; Ott, G.; Varga, L.; Schlote, W.; Rehfeldt, C.; Renne, U.; Williams, J.L.; Hill, W.G. Marker-assisted introgression of the compact mutant myostatin allele MstnCmpt-dl1Abc into a mouse line with extreme growth effects on body composition and muscularity. Genet. Res. 2004, 84, 161–173. [Google Scholar] [CrossRef]
- Fiems, L.O.; Van Caelenbergh, W.; Vanacker, J.M.; De Campeneere, S.; Seynaeve, M. Prediction of empty body composition of double-muscled beef cows. Livest. Prod. Sci. 2005, 92, 249–259. [Google Scholar] [CrossRef]
- De Campeneere, S.; Fiems, L.O.; De Paepe, M.; Vanacker, J.M.; Boucqué, C.V. Compositional data on Belgian Blue double-muscled bulls. Anim. Res. 2001, 50, 43–55. [Google Scholar]
- Yan, T.; Patterson, D.C.; Mayne, C.S.; Agnew, R.E.; Porter, M.G. Prediction of empty body weight and composition from live weight and other live animal measurements in lactating dairy cows. J. Agric. Sci. Camb. 2009, 147, 241–252. [Google Scholar] [CrossRef]
- Andrew, S.M.; Waldo, D.R.; Erdman, R.A. Direct analysis of body composition of dairy cows at three physiological stages. J. Dairy Sci. 1994, 77, 3022–3033. [Google Scholar] [CrossRef]
- Wright, I.A.; Russel, A.J.F. Partition of fat, body composition and body condition score in mature cows. Anim. Prod. 1984, 38, 23–32. [Google Scholar] [CrossRef]
- Piedrafita, J.; Quintanilla, R.; Sañudo, C.; Olleta, J.L.; Campo, M.M.; Panea, B.; Renand, G.; Turin, F.; Jabet, S.; Osoro, K.; Oliván, M.C.; Noval, G.; Garcı́a, P.; Garcı́a, M.D.; Oliver, M.A.; Gispert, M.; Serra, X.; Espejo, M.; Garcı́a, S.; López, M.; Izquierdo, M. Carcass quality of 10 beef cattle breeds of the Southwest of Europe in their typical production systems. Livest. Prod. Sci. 2003, 82, 1–13. [Google Scholar] [CrossRef]
- Weekly, L.B.; Veit, H.P. Potential morphologic and physiologic factors that may predispose the bovine lung to respiratory diseases. Compend. Contin. Educ. Pract. Vet. 1995, 17, 974–982. [Google Scholar]
- Jones, J.H.; Longworth, K.E.; Lindholm, A.; Conley, K.E.; Karas, R.H.; Kayar, S.R.; Taylor, C.R. Oxygen transport during exercise in large mammals. I. Adaptive variation in oxygen demand. J. Appl. Physiol. 1989, 67, 862–870. [Google Scholar]
- Gustin, P.; Lomba, F.; Bakima, J.; Lekeux, P.; Van de Woestijne, K.P. Partitioning of pulmonary resistance in calves. J. Appl. Physiol. 1987, 62, 1826–1831. [Google Scholar] [CrossRef]
- Gustin, P.; Dhem, A.R.; Lomba, F.; Lekeux, P. Cardio-pulmonary function values in double-muscled cattle during muscular exercise. Vet. Res. Comm. 1988, 12, 407–416. [Google Scholar] [CrossRef]
- Cambier, C.; Clerbaux, T.; Detry, B.; Beerens, D.; Frans, A.; Gustin, P. Blood oxygen binding in double-muscled calves and dairy calves with conventional muscle conformation. Am. J. Vet. Res. 2000, 61, 299–304. [Google Scholar] [CrossRef]
- Roets, E.; Burvenich, C.; Roberts, M. Muscarinic receptor subtypes, β-adrenoceptors and cAMP in the tracheal smooth muscle of conventional and double-muscled calves. Vet. Res. Comm. 1992, 16, 465–476. [Google Scholar] [CrossRef]
- Cusack, P.M.V.; McMeniman, N.; Lean, I.J. The medicine and epidemiology of bovine respiratory disease in feedlots. Aust. Vet. J. 2003, 81, 480–487. [Google Scholar] [CrossRef]
- Fiems, L.; De Campeneere, S.; Cottyn, B.; Boucqué, C. Effect of housing on the performance of double-muscled rearing calves. Ann. Zootech. 1998, 47, 207–214. [Google Scholar] [CrossRef]
- Tyler, H.; Ramsey, H. Hypoxia in neonatal calves: Effect on intestinal transport of immunoglobulins. J. Dairy Sci. 1991, 74, 1952–1956. [Google Scholar]
- Amory, H.; McEntee, K.; Linden, A.S.; Desmecht, D.J.M.; Beduin, J.M.L.; D’Orio, V.; Lekeux, P.M. Comparison of the cardiac pumping capability and cardiac pumping reserve in double-muscled and conventional calves. Can. J. Physiol. Pharmacol. 1993, 71, 946–951. [Google Scholar] [CrossRef]
- Monin, G.; Boccard, R. Caractéristiques physiologiques respiratoires des bovins culards. Ann. Génét. Sél. Anim. 1974, 6, 187–193. [Google Scholar]
- Holmes, J.H.G.; Ashmore, C.R.; Robinson, D.W. Effects of stress on cattle with hereditary muscular hypertrophy. J. Anim. Sci. 1973, 36, 684–694. [Google Scholar]
- Handschin, C.; Chin, S.; Li, P.; Liu, F.; Maratos-Flier, E.; LeBrasseur, N.K.; Yan, Z.; Spiegelman, B.M. Skeletal muscle fiber-type switching, exercise intolerance, and myopathy in PGC-1 α muscle-specific knock-out animals. J. Biol. Chem. 2007, 282, 30014–30021. [Google Scholar]
- Leone, T.C.; Lehman, J.J.; Finck, B.N.; Schaeffer, P.J.; Wende, A.R.; Boudina, S.; Courtois, M.; Wozniak, D.F.; Sambandam, N.; Bernal-Mizrachi, C.; Chen, Z.; Holloszy, J.O.; Medeiros, D.M.; Schmidt, R.E.; Saffitz, J.E.; Abel, E.D.; Semenkovich, C.F.; Kelly, D.P. PGC-1α-deficiency causes multi-system energy metabolic derangements: Muscle dysfunction, abnormal weight control and hepatic steatosis. PLoS Biol. 2005, 3. [Google Scholar] [CrossRef]
- Clerbaux, T.; Gustin, P.; Detry, B.; Cao, M.L.; Frans, A. Comparative study of the oxyhaemoglobin dissociation curve of four mammals: Man, dog, horse and cattle. Comp. Biochem. Physiol. 1993, 106A, 687–694. [Google Scholar]
- Gustin, P.; Clerbaux, T.; Willems, E.; Lekeux, P.; Lomba, F.; Frans, A. Oxygen transport properties of blood in two different bovine breeds. Comp. Biochem. Physiol. 1988, 89A, 553–558. [Google Scholar]
- Clinquart, A.; Van Eenaeme, C.; Mayombo, A.P.; Gauthier, S.; Istasse, L. Plasma hormones and metabolites in cattle in relation to breed (Belgian Blue vs. Holstein) and conformation (double-muscled vs. dual-purpose type). Vet. Res. Comm. 1995, 19, 185–194. [Google Scholar] [CrossRef]
- Fiems, L.O.; Cottyn, B.G.; Boucqué, C.V.; Bogaerts, D.F.; Van Eenaeme, C.; Vanacker, J.M. Effect of beef type, body weight and dietary protein content on voluntary feed intake, digestibility, blood and urine metabolites and nitrogen retention. J. Anim. Physiol. Anim. Nutr. 1997, 77, 1–9. [Google Scholar] [CrossRef]
- Sainz, R.D.; Bentley, B.E. Visceral organ mass and cellularity in growth-restricted and refed beef steers. J. Anim. Sci. 1997, 75, 1229–1236. [Google Scholar]
- Bjercke, R.J.; Goll, D.E.; Robson, R.M.; Dutson, T.R. Relative roles of polysomes and cytoplasmic enzymes in regulating bovine skeletal muscle protein synthesis. J. Anim. Sci. 1984, 59, 684–696. [Google Scholar]
- De Campeneere, S.; Fiems, L.O.; Boucqué, C.V. Energy and protein requirements of Belgian Blue double-muscled bulls. Anim. Feed Sci. Technol. 2001, 90, 153–167. [Google Scholar] [CrossRef]
- Fiems, L.O.; Vanacker, J.M.; De Boever, J.L.; Van Caelenbergh, W.; Aerts, J.M.; De Brabander, D.L. Effect of energy restriction and re-alimentation in Belgian Blue double-muscled beef cows on digestibility and metabolites. J. Anim. Physiol. Anim. Nutr. 2007, 91, 54–61. [Google Scholar] [CrossRef]
- McBride, B.W.; Kelly, J.M. Energy cost of absorption and metabolism in the ruminant gastrointestinal tract and liver: A review. J. Anim. Sci. 1990, 68, 2997–3010. [Google Scholar]
- Vermorel, M.; Bouvier, J.C.; Geay, Y. The effect of genotype (normal and double-muscled Charolais and Friesian) on energy utilization by growing cattle at 2 and 16 months of age. In Energy Metabolism of Farm Animals; Vermorel, M., Ed.; G.de Bussac: Clermont-Ferrand, France, 1976; pp. 217–220. [Google Scholar]
- Fiems, L.O.; Moermans, R.J.; Boucqué, C.V.; Cottyn, B.G.; De Campeneere, S. Voluntary feed intake in dual-purpose and double-muscled Belgian White-Blue bulls. In Regulation of Feed Intake; van der Heide, D., Huisman, E.A., Kanis, E., Osse, J.W.M., Verstegen, M.W.A., Eds.; CAB International: Wallingford, UK, 1999; pp. 21–25. [Google Scholar]
- Haynes, F.E.M.; Greenwood, P.L.; McDonagh, M.B.; Oddy, V.H. Myostatin allelic status interacts with level of nutrition to affect growth, composition, and myofiber characteristics of lambs. J. Anim. Sci. 2012, 90, 456–465. [Google Scholar] [CrossRef]
- Geay, Y.; Robelin, J. Variation of meat production capacity in cattle due to genotype and level of feeding: Genotype-nutrition interaction. Livest. Prod. Sci. 1979, 6, 263–276. [Google Scholar] [CrossRef]
- Fiems, L.O.; De Campeneere, S.; Vanacker, J.M.; De Boever, J.L.; De Brabander, D.L. Are Belgian Blue double-muscled animals deviant for their phosphorus requirements? In Proceedings of the30th Meeting Dutch Speaking Nutrition Researchers, Merelbeke, Belgium, 8 April 2005; pp. 49–50.
- AFRC. A reappraisal of the calcium and phosphorus requirements of sheep and cattle. Nutr. Abst. Rev. B 1991, 61, 573–612.
- NRC, Nutrient Requirements of Dairy Cattle, 7th ed.National Academy Press: Washington, DC, USA, 2001.
- Meschy, F. Recommandations d’apport en phosphore absorbé chez les ruminants. Renc. Rech. Ruminants 2002, 9, 279–285. [Google Scholar]
- Robbins, J.D.; Oltjen, R.R.; Cabell, C.A.; Dolnick, E.H. Influence of varying levels of dietary minerals on the development of urolithiasis, hair growth, and weight gains in rats. J. Nutr. 1965, 85, 355–361. [Google Scholar]
- Ewoldt, J.M.; Jones, M.L.; Miesner, M.D. Surgery of obstructive urolithiasis in ruminants. Vet. Clin. Food Anim. 2008, 24, 455–465. [Google Scholar] [CrossRef]
- Ouhayoun, J.; Arnal, T. Étude du caractère culard. IV. Anatomie microscopique comparée du rein de males Charolais normaux et culards. Ann. Génét. Sél. Anim. 1969, 1, 101–108. [Google Scholar]
- Guyot, H.; Spring, P.; Andrieu, S.; Rollin, F. Comparative responses to sodium selenite and organic selenium supplements in Belgian Blue cows and calves. Livest. Sci. 2007, 111, 259–263. [Google Scholar] [CrossRef]
- NRC, Nutrient Requirements of Beef Cattle, 7th ed.; National Academy Press: Washington, DC, USA, 2000.
- Allen, W.M. New developments in muscle pathology: Nutritional myopathies including “muscular dystrophy” or “white muscle disease”. Vet. Sci. Comm. 1977, 1, 243–250. [Google Scholar] [CrossRef]
- Anderson, P.H.; Berrett, S.; Patterson, D.S.P. The significance of elevated plasma creatinephosphokinase activity in muscle disease of cattle. J. Comp. Path. 1976, 86, 531–538. [Google Scholar] [CrossRef]
- Ashmore, C.R.; Robinson, D.W. Hereditary muscular hypertrophy in the bovine. 1. Histological and biochemical characterization. Proc. Soc. Exp. Biol. Med. 1969, 132, 548–554. [Google Scholar]
- Hendricks, H.B.; Aberle, E.D.; Jones, D.J.; Martin, T.G. Muscle fiber type, rigor development and bone strength in double muscled cattle. J. Anim. Sci. 1973, 37, 1305–1311. [Google Scholar]
- Shahin, K.A.; Berg, R.T.; Price, M.A. Muscle and bone distribution in mature normal and double muscled cows. Livest. Prod. Sci. 1991, 28, 291–303. [Google Scholar] [CrossRef]
- Mendias, C.L.; Marcin, J.E.; Calerdon, D.R.; Faulkner, J.A. Contractile properties of EDL and soleus muscles of myostatin deficient mice. J. Appl. Physiol. 2006, 101, 898–905. [Google Scholar] [CrossRef]
- Amthor, H.; Macharia, R.; Navarrete, R.; Schuelke, M.; Brown, S.C.; Otto, A.; Voit, T.; Muntoni, F.; Vrbova, G.; Partridge, T.; Zammit, P.; Bunger, L.; Patel, K. Lack of myostatin results in excessive muscle growth but impaired force generation. Proc. Natl. Acad. Sci. USA 2007, 104, 1835–1840. [Google Scholar]
- Matsakas, A.; Macharia, R.; Otto, A.; Elashry, M.I.; Mouisel, E.; Romanello, V.; Sartori, R.; Amthor, H.; Sandri, M.; Narkar, V.; Patel, K. Exercise training attenuates the hypermuscular phenotype and restores skeletal muscle function in the myostatin null mouse. Exp. Physiol. 2012, 97, 125–140. [Google Scholar]
- Hamrick, M.W.; Pennington, C.; Byron, C.D. Bone architecture and disc degeneration in the lumbar spine of mice lacking GDF-8 (myostatin). J. Orthop. Res. 2003, 21, 1025–1032. [Google Scholar] [CrossRef]
- Zou, H.; Wieser, R.; Massagué, J.; Niswander, L. Distinct roles of type I bone morphogenetic protein receptors in the formation and differentiation of cartilage. Genes Dev. 1997, 11, 2191–2203. [Google Scholar] [CrossRef]
- Blumer, M.J.F.; Longato, S.; Fritsch, H. Structure, formation and role of cartilage canals in the developing bone. Ann. Anat. 2008, 190, 305–315. [Google Scholar] [CrossRef]
- Merino, R.; Macias, D.; Gañan, Y.; Rodriguez-Leon, J.; Economides, A.N.; Rodriguez-Esteban, C.; Izpisua-Belmonte, J.C.; Hurle, J.M. Control of digit formation by activin signalling. Development 1999, 126, 2161–2170. [Google Scholar]
- Rountree, R.B.; Schoor, M.; Chen, H.; Marks, M.E.; Harley, V.; Mishina, Y.; Kingsley, D.M. BMP receptor signaling is required for postnatal maintenance of articular cartilage. PLoS Biol. 2004, 2, 1815–1827. [Google Scholar]
- Mason, I.L. Symptoms of muscular hypertrophy in heterozygous steers. Anim. Prod. 1963, 5, 57–65. [Google Scholar] [CrossRef]
- Cassar-Malek, I.; Passelaigue, F.; Bernard, C.; Léger, J.; Hocquette, J.F. Target genes of myostatin loss-of-function in muscles of late bovine fetuses. BMC Genomics 2007, 8. [Google Scholar] [CrossRef]
- Nott, C.F.G.; Rollins, W.C.; Tanaka, M. Effects of the m gene for muscular hypertrophy on conformation at one year of age in beef cattle. Ann. Génét. Sél. Anim. 1980, 12, 143–155. [Google Scholar]
- Anderson, D.E.; Desrochers, A.; St. Jean, G. Management of Tendon Disorders in Cattle. Vet. Clin. Food Anim. 2008, 24, 551–566. [Google Scholar] [CrossRef]
- Wechsler, B. Floor quality and space allowance in intensive beef production. Anim. Welfare 2011, 20, 497–503. [Google Scholar]
- Design Recommendations of Beef Cattle Housing; Report of the CIGR Section II; Working Group No. 14; Cattle Housing, 2nd ed. CIGR: East Lansing, MI, USA, 2004.
- Schiavon, S.; Tagliapietra, F.; Dal Maso, M.; Bailoni, L.; Bittante, G. Effects of low-protein diets and rumen-protected conjugated linoleic acid on production and carcass traits of growing double-muscled Piemontese bulls. J. Anim. Sci. 2010, 88, 3372–3383. [Google Scholar] [CrossRef]
- De Campeneere, S.; Fiems, L.O.; De Brabander, D.L. Determination of a standard for physical structure requirement for Belgian Blue bulls. J. Anim. Feed Sci. 2004, 13 Suppl. 1, 623–626. [Google Scholar]
- Short, R.E.; MacNeil, M.D.; Grosz, M.D.; Gerrard, D.E.; Grings, E.E. Pleiotropic effects in Hereford, Limousin, and Piedmontese F2 crossbred calves of genes controlling muscularity including the Piedmontese myostatin allele. J. Anim. Sci. 2002, 80, 1–11. [Google Scholar]
- Vissac, B.; Ménissier, F.; Perreau, B. Étude du caractère culard. VII. Croissance et musculature des femelles, déséquilibre morphologique au vêlage. Ann. Génét. Sél. Anim. 1973, 5, 23–38. [Google Scholar]
- Arthur, P.F.; Makarechian, M.; Price, M.A. Incidence of dystocia and perinatal calf mortality resulting from reciprocal crossing of double-muscled and normal cattle. Can. Vet. J. 1988, 29, 163–167. [Google Scholar]
- Mao, W.H.; Albrecht, E.; Teuscher, F.; Yang, Q.; Zhao, R.Q.; Wegner, J. Growth- and breed-related changes of fetal development in cattle. Asian Aust. J. Anim. Sci. 2008, 21, 640–647. [Google Scholar]
- Fiems, L.O.; De Campeneere, S.; Van Caelenbergh, W.; Boucqué, C.V. Relationship between dam and calf characteristics with regard to dystocia in Belgian Blue double-muscled cows. Anim. Sci. 2001, 72, 389–394. [Google Scholar]
- Fiems, L.O.; De Brabander, D.L. Optimum growth rate of Belgian Blue double-muscled replacement heifers. South Afr. J. Anim. Sci. 2009, 39 Suppl. 1, 6–10. [Google Scholar]
- Fiems, L.O.; De Brabander, D.L. Relation between calf birth weight and dam weight in Belgian Blue double-muscled cattle. In Proceedings of the 60th Annual Meeting of the European Association for Animal Production, Barcelona, Spain, 24–27 August 2009; p. 96.
- Johanson, J.M.; Berger, P.J. Birth weight as a predictor of calving ease and perinatal mortality in Holstein cattle. J. Dairy Sci. 2003, 86, 3745–3755. [Google Scholar] [CrossRef]
- Kolkman, I.; De Vliegher, S.; Hoflack, G.; Van Aert, M.; Laureyns, J.; Lips, D.; de Kruif, A.; Opsomer, G. Protocol of the caesarean section as performed in daily bovine practice in Belgium. Reprod. Dom. Anim. 2007, 42, 583–589. [Google Scholar] [CrossRef]
- Vermorel, M.; Vernet, J.; Saido, S.; Dardillat, C.; Demigne, C.; Davicco, M.J. Energy metabolism and thermoregulation in the newborn calf; Effect of calving conditions. Can. J. Anim. Sci. 1989, 69, 113–122. [Google Scholar] [CrossRef]
- Patterson, D.J.; Bellows, R.A.; Burfening, P.J. Effects of caesarean section, retained placenta and vaginal or uterine prolapse on subsequent fertility in beef cattle. J. Anim. Sci. 1981, 53, 916–921. [Google Scholar]
- Hanzen, C.; Laurent, Y.; Ward, W.R. Comparison of reproductive performance in Belgian dairy and beef cattle. Theriogenology 1994, 41, 1099–1114. [Google Scholar] [CrossRef]
- Frazier, E.L.; Sprott, L.R.; Sanders, J.O.; Dahm, P.F.; Crouch, J.R.; Turner, J.W. Sire marbling score expected progeny difference and weaning weight maternal expected progeny difference associations with age at first calving and calving interval in Angus beef cattle. J. Anim. Sci. 1999, 77, 1322–1328. [Google Scholar]
- McCartney, D.; Basarab, J.A.; Okine, E.K.; Baron, V.S.; Depalme, A.J. Alternative fall and winter feeding systems for spring calving beef cows. Can. J. Anim. Sci. 2004, 84, 511–522. [Google Scholar] [CrossRef]
- Pilarczyk, R.; Wójcik, J. Comparison of body weight and reproduction performance in cows of various beef breeds managed under equal conditions in West Pomerania. Arch. Tierz. 2008, 51, 318–328. [Google Scholar]
- Vermunt, J.J. The caesarean operation in cattle: A review. Iran. J. Vet. Surg. 2008, 3 Suppl. 1, 82–100. [Google Scholar]
- Chupin, D. Analysis of reproduction problems in double muscle females. In Muscle Hypertrophy of Genetic Origin and Its Use to Improve Beef Production; King, J.W.B., Menissier, F., Eds.; Martinus Nijhoff Publishers: The Hague, The Netherlands, 1982; pp. 575–584. [Google Scholar]
- Hoflack, G.; Opsomer, G.; Van Soom, A.; Maes, D.; de Kruif, A.; Duchateau, L. Comparison of sperm quality of Belgian Blue and Holstein Friesian bulls. Theriogenology 2006, 66, 1834–1846. [Google Scholar] [CrossRef]
- Leroy, J.L.M.R.; Opsomer, G.; De Vliegher, S.; Vanholder, T.; Goossens, L.; Geldhof, A.; Bols, P.E.J.; de Kruif, A.; Van Soom, A. Comparison of embryo quality in high-yielding dairy cows, in dairy heifers and in beef cows. Theriogenology 2005, 64, 2022–2036. [Google Scholar] [CrossRef]
- Liang, Y.C.; Yeh, J.Y.; Ou, B.R. Effect of maternal myostatin antibody on offspring growth performance and body composition in mice. J. Exp. Biol. 2007, 210, 477–483. [Google Scholar] [CrossRef]
- Ciarmela, P.; Wiater, E.; Smith, S.M.; Vale, W. Myostatin in rat uterus and myometrial cells. Endocrinology 2009, 150, 906–914. [Google Scholar]
- Wong, C.L.; Huang, Y.Y.; Ho, W.K.; Poon, H.K.; Cheung, P.L.; O, W.S.; Chow, P.H. Growth-differentiation factor-8 (GDF-8) in the uterus: Its identification and functional significance in the golden hamster. Reprod. Biol. Endocrinol. 2009, 7. [Google Scholar] [CrossRef]
- Ethier, J.F.; Findlay, J.K. Roles of activin and its signal transduction mechanisms in reproductive tissues. Reproduction 2001, 121, 667–675. [Google Scholar] [CrossRef]
- Young, J.M.; Henderson, S.; Souza, C.; Ludlow, H.; Groome, N.; McNeilly, A.S. Activin B is produced early in antral follicular development and suppresses thecal androgen production. Reproduction 2012, 143, 637–650. [Google Scholar] [CrossRef]
- Muttukrishna, S.; Tannetta, D.; Groome, N.; Sargent, I. Activin and follistatin in female reproduction. Mol. Cell. Endocrinol. 2004, 225, 45–56. [Google Scholar] [CrossRef]
- Jones, R.L.; Kaitu’u-Lino, T.J.; Nie, G.; Sanchez-Partida, L.G.; Findlay, J.K.; Salamonsen, L.A. Complex expression patterns support potential roles for maternally derived activins in the establishment of pregnancy in mouse. Reproduction 2006, 132, 799–810. [Google Scholar] [CrossRef]
- Wolfenson, D.; Roth, Z.; Meidan, R. Impaired reproduction in heat-stressed cattle: Basic and applied aspects. Anim. Reprod. Sci. 2000, 60, 535–547. [Google Scholar] [CrossRef]
- Halipré, A. Étude du caractere culard. X. Sensibilité des bovins culards au stress thermique. Ann. Génét. Sél. Anim. 1973, 5, 441–449. [Google Scholar]
- Stavaux, D.; Art, T.; Entee, K.M.; Reznick, M.; Lekeux, P. Muscle fibre type and size, and muscle capillary density in young double-muscled Blue Belgian cattle. J. Vet. Med. A 1994, 41, 229–236. [Google Scholar]
- Blazquez, N.B.; Long, S.E.; Mayhew, T.M.; Perry, G.C.; Prescott, N.J.; Wathes, C.M. Rate of discharge and morphology of sweat glands in the perineal, lumbodorsal and scrotal skin of cattle. Res. Vet. Sci. 1994, 57, 277–284. [Google Scholar] [CrossRef]
- Reynolds, L.P.; Ferrell, C.L.; Nienaber, J.A.; Ford, S.P. Effects of chronic environmental heat stress on blood flow and nutrient uptake of the gravid bovine uterus and foetus. J. Agric. Sci. 1985, 104, 289–297. [Google Scholar] [CrossRef]
- Finch, V.A. Body temperature in beef cattle: Its control and relevance to production in the tropics. J. Anim. Sci. 1986, 62, 531–542. [Google Scholar]
- Schafer, K.A. The cell cycle: A review. Vet. Pathol. 1998, 35, 461–478. [Google Scholar] [CrossRef]
- Gagnière, H.; Ménissier, F.; Geay, Y.; Picard, B. Influence of genotype on contractile protein differentiation in different bovine muscles during foetal life. Ann. Zootech. 2000, 49, 405–423. [Google Scholar] [CrossRef]
- Thomas, M.; Langley, B.; Berry, C.; Sharma, M.; Kirk, S.; Bass, J.; Kambadur, R. Myostatin, a negative regulator of muscle growth, functions by inhibiting myoblast proliferation. J. Biol. Chem. 2000, 275, 40235–40243. [Google Scholar]
- Rios, R.; Carneiro, I.; Arce, V.M.; Devesa, J. Myostatin is an inhibitor of myogenic differentiation. Am. J. Physiol. Cell Physiol. 2002, 282, C993–C999. [Google Scholar]
- Amthor, H.; Nicholas, G.; McKinnell, I.; Kemp, C.F.; Sharma, M.; Kambadur, R.; Patel, K. Follistatin complexes myostatin and antagonises myostatin-mediated inhibition of myogenesis. Dev. Biol. 2004, 270, 19–30. [Google Scholar] [CrossRef]
- Hayashi, S.; Miyake, M.; Watanabe, K.; Aso, H.; Hayashi, S.; Ohwada, S.; Yamaguchi, T. Myostatin preferentially down-regulates the expression of fast 2x myosin heavy chain in cattle. Proc. Jpn. Acad. B 2008, 84, 354–362. [Google Scholar] [CrossRef]
- Albrecht, E.; Teuscher, F.; Ender, K.; Wegner, J. Growth- and breed-related changes of muscle bundle structure in cattle. J. Anim. Sci. 2006, 84, 2959–2964. [Google Scholar] [CrossRef]
- Wegner, J.; Albrecht, E.; Fiedler, I.; Teuscher, F.; Papstein, H.J.; Ender, K. Growth- and breed-related changes of muscle fiber characteristics in cattle. J. Anim. Sci. 2000, 78, 1485–1496. [Google Scholar]
- West, R.L. Red to white fiber ratios as an index of double muscling in beef cattle. J. Anim. Sci. 1974, 38, 1165–1175. [Google Scholar]
- Batjoens, P.; Fiems, L.O.; Van Hoof, J.; Van Vooren, T.; Vereecke, D. Myofibre composition and metabolic aspects in different strains of Belgian white-blue bulls and their relation to meat colour. In Proceedings of the37th International Congress of Meat Science and Technology, Kulmbach, Germany, 1–6 September 1991; pp. 324–327.
- Picard, B.; Hocquette, J.F.; Bornes, F.; Brazi, S.; Vermorel, M.; Geay, Y. Muscle metabolism in normal and double muscled calves: Fiber characteristics and glucose transport rate. In Energy Metabolism of Farm Animals; Aguilera, J.F., Ed.; Consejo Superior de Investigaciones: Granada, Spain, 1994; pp. 97–100. [Google Scholar]
- Girgenrath, S.; Song, K.; Whittemore, L.A. Loss of myostatin expression alters fiber-type distribution and expression of myosin heavy chain isoforms in slow- and fast-type skeletal muscle. Muscle Nerve 2005, 31, 34–40. [Google Scholar] [CrossRef]
- Kambadur, R.; Bishop, A.; Salerno, M.S.; McCroskery, S.; Sharma, M. Role of myostatin in muscle growth. In Muscle Development of Livestock Animals; te Pas, M.F.W., Everts, M.E., Haagsman, H.P., Eds.; CAB International: Wallingford, UK, 2004; pp. 297–316. [Google Scholar]
- Deveaux, V.; Picard, B.; Bouley, J.; Cassar-Malek, I. Location of myostatin expression during bovine myogenesis in vivo and in vitro. Reprod. Nut. Dev. 2003, 43, 527–542. [Google Scholar] [CrossRef]
- Deveaux, V.; Cassar-Malek, I.; Picard, B. Comparison of contractile characteristics of muscle from Holstein and double-muscled Belgian Blue foetuses. Comp. Biochem. Physiol. A Physiol. 2001, 131, 21–29. [Google Scholar]
- Gagnière, H.; Picard, B.; Jurie, C.; Geay, Y. Comparative study of metabolic differentiation of foetal muscle in normal and double-muscled cattle. Meat Sci. 1997, 45, 145–152. [Google Scholar] [CrossRef]
- Picard, B.; Gagnière, H.; Robelin, J.; Geay, Y. Comparison of the foetal development of muscle in normal and double-muscled cattle. J. Muscle Res. Cell Motil. 1995, 16, 629–639. [Google Scholar] [CrossRef]
- Picard, B.; Depreux, F.; Geay, Y. Muscle differentiation of normal and double-muscled bovine foetal myoblasts in primary culture. Basic Appl. Myol. 1998, 8, 197–203. [Google Scholar]
- Picard, B.; Lefaucheur, L.; Berri, C.; Duclos, M.J. Muscle fibre ontogenesis in farm animal species. Reprod. Nutr. Dev. 2002, 42, 415–431. [Google Scholar] [CrossRef]
- Naya, F.J.; Mercer, B.; Shelton, J.; Richardson, J.A.; Williams, R.S.; Olson, E.N. Stimulation of slow skeletal muscle fiber gene expression by calcineurin in vivo. J. Biol. Chem. 2000, 275, 4545–4548. [Google Scholar]
- Lin, J.; Wu, H.; Tarr, P.T.; Zhang, C.Y.; Wu, Z.; Boss, O.; Michael, L.F.; Puigserver, P.; Isotani, E.; Olson, E.N.; Lowell, B.B.; Bassel-Duby, R.; Spiegelman, B.M. Transcriptional co-activator PGC-1α drives the formation of slow-twitch muscle fibres. Nature 2002, 418, 797–801. [Google Scholar]
- Puddick, J.; Martinus, R.D. Comparative proteomics of skeletal muscle mitochondria from myostatin-null mice. Cell Biol. Int. Rep. 2011, 18, 35–41. [Google Scholar] [CrossRef]
- Hocquette, J.F.; Ortigues-Marty, I.; Pethick, D.; Herpin, P.; Fernandez, X. Nutritional and hormonal regulation of energy metabolism in skeletal muscles of meat-producing animals. Livest. Prod. Sci. 1998, 56, 115–143. [Google Scholar] [CrossRef]
- Holmes, J.H.G.; Robinson, D.W.; Ashmore, C.R. Blood lactic acid and behaviour in cattle with hereditary muscular hypertrophy. J. Anim. Sci. 1972, 35, 1011–1013. [Google Scholar]
- Garlick, P.J.; Maltin, C.A.; Baillie, A.G.; Delday, M.I.; Grubb, D.A. Fiber-type composition of nine rat muscles. II. Relationship to protein turnover. Am. J. Physiol. Endocrinol. Metab. 1989, 257, E828–E832. [Google Scholar]
- McPherron, A.C.; Lee, S.J. Suppression of body fat accumulation in myostatin-deficient mice. J. Clin. Invest. 2002, 109, 595–601. [Google Scholar]
- Guo, T.; Jou, W.; Chanturiya, T.; Portas, J.; Gavrilova, O.; McPherron, A.C. Myostatin Inhibition in muscle, but not adipose tissue, decreases fat mass and improves insulin sensitivity. PLoS ONE 2009, 4. [Google Scholar] [CrossRef]
- Tu, P.; Bhasin, S.; Hruz, P.W.; Herbst, K.L.; Castellani, L.W.; Hua, N.; Hamilton, J.A.; Guo, W. Genetic disruption of myostatin reduces the development of proatherogenic dyslipidemia and atherogenic lesions in Ldlr null mice. Diabetes 2009, 58, 1739–1748. [Google Scholar] [CrossRef]
- Ferrell, C.L. Contribution of visceral organs to animal energy expenditures. J. Anim. Sci. 1988, 66, 23–34. [Google Scholar]
- Strath, R.A.; Basarab, J.A.; Thompson, J.R.; Berg, R.T. Thyroid hormone concentrations: Some kinetic parameters of triiodothyronine and metabolic rate in “double-muscled” cattle. Can. J. Anim. Sci. 1982, 62, 381–395. [Google Scholar] [CrossRef]
- Hocquette, J.F.; Bas, P.; Bauchart, D.; Vermorel, M.; Geay, Y. Fat partitioning and biochemical characteristics of fatty tissues in relation to plasma metabolites and hormones in normal and double-muscled young growing bulls. Comp. Biochem. Physiol. A 1999, 122, 127–138. [Google Scholar]
- Bossaert, P.; Leroy, J.L.M.R.; De Campeneere, S.; De Vliegher, S.; Opsomer, G. Differences in the glucose-induced insulin response and the peripheral insulin responsiveness between neonatal calves of the Belgian Blue, Holstein-Friesian, and East Flemish breeds. J. Dairy Sci. 2009, 92, 4404–4411. [Google Scholar] [CrossRef]
- Chen, Y.; Ye, J.; Cao, L.; Zhang, Y.; Xia, W.; Zhu, D. Myostatin regulates glucose metabolism via the AMP-activated protein kinase pathway in skeletal muscle cells. Int. J. Biochem. Cell Biol. 2010, 42, 2072–2081. [Google Scholar] [CrossRef]
- Antony, N.; Bass, J.J.; McMahon, C.D.; Mitchell, M.D. Myostatin regulates glucose uptake in BeWo cells. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E1296–E1302. [Google Scholar] [CrossRef]
- Van Eetvelde, M.; Kamal, M.M.; Fiems, L.; Opsomer, G. Pancreatic b-cell function differs between newborn Belgian Blue and Holstein calves. In Proceedings16th Annual Conferenceof theEuropean Society for Domestic Animal Reproduction, Dublin, Ireland, 29 August–1 September 2012.
- Toscano Paganoa, G.T.; Lazzaronia, C.; Pagano, P.G.; Galeazzi, D.; Biagini, D. A study on muscular hypertrophy in cattle: Serum concentrations of IGF-1, cortisol, insulin and testosterone. Livest. Prod. Sci. 2001, 70, 235–239. [Google Scholar] [CrossRef]
- Istasse, L.; Van Eenaeme, C.; Evrard, P.; Gabriel, A.; Baldwin, P.; Maghuin-Rogister, G.; Bienfait, J.M. Animal performance, plasma hormones and metabolites in Holstein and Belgian Blue growing-fattening bulls. J. Anim. Sci. 1990, 68, 2666–2673. [Google Scholar]
- Pajak, B.; Pawlikowska, P.; Cassar-Malek, I.; Picard, B.; Hocquette, J.F.; Orzechowski, A. Abundance of some skeletal muscle mitochondrial proteins is associated with increased blood serum insulin in bovine fetuses. Res. Vet. Sci. 2010, 89, 445–450. [Google Scholar] [CrossRef]
- Ploquin, C.; Chabi, B.; Fouret, G.; Vernus, B.; Feillet-Coudray, C.; Coudray, C.; Bonnieu, A.; Ramonatxo, C. Lack of myostatin alters intermyofibrillar mitochondria activity, unbalances redox status and impairs tolerance to chronic repetitive contractions in muscle. Am. J. Physiol. Endocrinol. Metab. 2012, 302, 1000–1008. [Google Scholar] [CrossRef]
- Lammoglia, M.A.; Bellows, R.A.; Grings, E.E.; Bergman, J.W. Effects of prepartum supplementary fat and muscle hypertrophy genotype on cold tolerance in newborn calves. J. Anim. Sci. 1999, 77, 2227–2233. [Google Scholar]
- Holmes, J.H.G.; Robinson, D.W. Hereditary muscular hypertrophy in the bovine: Metabolic response to nutritional stress. J. Anim. Sci. 1970, 31, 776–780. [Google Scholar]
- Allen, D.L.; McCall, G.E.; Loh, A.S.; Madden, M.C.; Mehan, R.S. Acute daily psychological stress causes increased atrophic gene expression and myostatin-dependent muscle atrophy. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 299, R889–R898. [Google Scholar] [CrossRef]
- Demas, G.E. The energetics of immunity: A neuroendocrine link between energy balance and immune function. Horm. Behav. 2004, 45, 173–180. [Google Scholar] [CrossRef]
- Helterline, D.L.I.; Garikipati, D.; Stenkamp, D.L.; Rodgers, B.D. Embryonic and tissue-specific regulation of myostatin-1 and -2 gene expression in zebrafish. Gen. Comp. Endocrinol. 2007, 151, 90–97. [Google Scholar] [CrossRef]
- Michaux, C.; Brochier, B.; Roupain, J.; Pastoret, P.P.; Hanset, R.; Barta, O. Comparaison de l’immunocompétence des veaux culards et mixtes de race Blanc-Bleu-belge au moyen du test de transformation lymphoblastique. Ann. Med. Vet. 1984, 128, 559–564. [Google Scholar]
- Klont, R.E.; Brocks, L.; Eikelenboom, G. Muscle fibre type and meat quality. Meat Sci. 1998, 49 Suppl. 1, 219–229. [Google Scholar] [CrossRef]
- Guillemin, N.; Cassar-Malek, I.; Hocquette, J.F.; Jurie, C.; Micol, D.; Listrat, A.; Levéziel, H.; Renand, G.; Picard, B. La maîtrise de la tendreté de la viande bovine: Identification de marqueurs biologiques. INRA Prod. Anim. 2009, 22, 331–344. [Google Scholar]
- Strath, R.A.; Thompson, J.R.; Christopherson, R.J. Effect of fasting on plasma glucose kinetics in double-muscled cattle. Can. J. Anim. Sci. 1982, 62, 955–958. [Google Scholar] [CrossRef]
- Mitlöhner, F.M.; Galyean, M.L.; McGlone, J.J. Shade effects on performance, carcass traits, physiology, and behavior of heat-stressed feedlot heifers. J. Anim. Sci. 2002, 80, 2043–2050. [Google Scholar]
- De Smet, S.; Lescouhier, S.; Claeys, E. Two-toned colour in the biceps femoris muscle in relation to post mortem pH and temperature fall in Belgian Blue beef. In Proceedings of the53th International Congress of Meat Science and Technology, Cape Town, South Africa, 10–15 August 2008.
- De Boever, M.; Lescouhier, S.; De Smet, S. Influence of post mortem glycolysis and cooling on colour stability in different muscles of Belgian Blue beef. In Proceedings of the54th International Congress of Meat Science and Technology, Copenhagen, Denmark, 16–21 August 2009; pp. 772–776.
- Sammel, L.M.; Hunt, M.C.; Kropf, D.H.; Hachmeister, K.A.; Kastner, C.L.; Johnson, D.E. Influence of chemical characteristics of beef inside and outside semimembranosus on color traits. J. Food Sci. 2002, 67, 1323–1330. [Google Scholar] [CrossRef]
- Pringle, T.D.; West, R.L.; Williams, S.E.; Johnson, D.D. The Role of the Calpain/Calpastatin System in Muscle Hypertrophy Associated with Double-Muscling in Beef; Annual Report; Department of Animal and Dairy Science, University of Georgia: Athens, GA, USA, 1995; pp. 61–65. [Google Scholar]
- Thompson, J. Managing meat tenderness. Meat Sci. 2002, 62, 295–308. [Google Scholar] [CrossRef]
- Steen, D.; Claeys, E.; Uytterhaegen, L.; De Smet, S.; Demeyer, D. Early post-mortem conditions and the calpain/calpastatin system in relation to tenderness of double-muscled beef. Meat Sci. 1997, 45, 307–319. [Google Scholar] [CrossRef]
- Wheeler, T.L.; Shackelford, S.D.; Casas, E.; Cundiff, L.V.; Koohmaraie, M. The effects of Piedmontese inheritance and myostatin genotype on the palatability of longissimus thoracis, gluteus medius, semimembranosus, and biceps femoris. J. Anim. Sci. 2001, 79, 3069–3074. [Google Scholar]
- Boccard, R.; Dumont, B.L.; Schmitt, O. Caractéristiques différentielles du tissu conjonctif des bovins normaux et “culards”. Gen. Sel. Evol. 1969, 1, 178. [Google Scholar]
- Bailey, A.J.; Enser, M.B.; Dransfield, E.; Restall, D.J.; Avery, N.C. Muscle and adipose tissue from normal and double muscled cattle: Collagen types, muscle fiber diameter, fat cell size and fatty acid composition and organoleptic properties. In Muscle Hypertrophy of Genetic Origin and its Use to Improve Beef Production; King, J.W.B., Menissier, F., Eds.; Martinus Nijhoff Publishers: The Hague, The Netherlands, 1982; pp. 178–203. [Google Scholar]
- Ngapo, T.M.; Berge, P.; Culioli, J.; Dransfield, E.; De Smet, S.; Claeys, E. Perimysial collagen crosslinking and meat tenderness in Belgian Blue double-muscled cattle. Meat Sci. 2002, 61, 91–102. [Google Scholar] [CrossRef]
- Sañudo, C.; Macie, E.S.; Olleta, J.L.; Villarroel, M.; Panea, B.; Albertí, P. The effects of slaughter weight, breed type and ageing time on beef meat quality using two different texture devices. Meat Sci. 2004, 66, 925–932. [Google Scholar] [CrossRef]
- Wiener, P.; Woolliams, J.A.; Frank-Lawale, A.; Ryan, M.; Richardson, R.I.; Nute, G.R.; Wood, J.D.; Homer, D.; Williams, J.L. The effects of a mutation in the myostatin gene on meat and carcass quality. Meat Sci. 2009, 83, 127–134. [Google Scholar] [CrossRef]
- Hornick, J.L.; Van Eenaeme, C.; Clinquart, A.; Diez, M.; Istasse, L. Different periods of feed restriction before compensatory growth in Belgian Blue Bulls: I. Animal performance, nitrogen balance, meat characteristics, and fat composition. J. Anim. Sci. 1998, 76, 249–259. [Google Scholar]
- Fiems, L.O.; De Campeneere, S.; Van Caelenbergh, W.; De Boever, J.L.; Vanacker, J.M. Carcass and meat quality in double-muscled Belgian Blue bulls and cows. Meat Sci. 2002, 63, 345–352. [Google Scholar]
- Picard, B.; Jurie, C.; Garcia-Launay, F.; Meteau, K.; Agabriel, J.; Micol, D. Marqueurs des qualités sensorielles de la viande bovine en race Salers. Renc. Rech. Ruminants 2011, 18, 181–184. [Google Scholar]
- Clements, C.M.; McNally, R.S.; Conti, B.J.; Mak, T.W.; Ting, J.P.Y. DJ-1, a cancer- and Parkinson’s disease-associated protein, stabilizes the antioxidant transcriptional master regulator Nrf2. Proc. Natl. Acad. Sci. USA 2006, 103, 15091–15096. [Google Scholar]
- Jia, X.; Ekman, M.; Grove, H.; Færgestad, E.M.; Aass, L.; Hildrum, K.I.; Hollung, K. Proteome changes in bovine longissimus thoracis muscle during the early postmortem storage period. J. Proteome Res. 2007, 6, 2720–2731. [Google Scholar] [CrossRef]
- Picard, B.; Berri, C.; Lefaucheur, L.; Molette, C.; Sayd, T.; Terlouw, C. Skeletal muscle proteomics in livestock production. Briefings Funct. Genomics 2010, 9, 259–278. [Google Scholar] [CrossRef]
- Jia, X.; Veiseth-Kent, E.; Grove, H.; Kuziora, P.; Aass, L.; Hildrum, K.I.; Hollung, K. Peroxiredoxin-6—A potential protein marker for meat tenderness in bovine longissimus thoracis muscle. J. Anim. Sci. 2009, 87, 2391–2399. [Google Scholar] [CrossRef]
- Chelh, I.; Meunier, B.; Picard, B.; Reecy, M.J.; Chevalier, C.; Hocquette, J.F.; Cassar-Malek, I. Molecular profiles of Quadriceps muscle in myostatin-null mice reveal PI3K and apoptotic pathways as myostatin targets. BMC Genomics 2009, 10. [Google Scholar] [CrossRef]
- Raes, K.; De Smet, S.; Demeyer, D. Effect of double-muscling in Belgian Blue young bulls on the intramuscular fatty acid composition with emphasis on conjugated linoleic acid and polyunsaturated fatty acids. Anim. Sci. 2001, 73, 253–260. [Google Scholar]
- Aldai, N.; Nájera, A.I.; Dugan, M.E.R.; Celaya, R.; Osoro, K. Characterisation of intramuscular, intermuscular and subcutaneous adipose tissues in yearling bulls of different genetic groups. Meat Sci. 2007, 76, 682–691. [Google Scholar] [CrossRef]
- Aldai, N.; Dugan, M.E.R.; Nájera, A.I.; Osoro, K. N-6 and n-3 fatty acids in different beef adipose tissues depending on the presence or absence of the gene responsible for double-muscling. Czech J. Anim. Sci. 2008, 53, 515–522. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fiems, L.O. Double Muscling in Cattle: Genes, Husbandry, Carcasses and Meat. Animals 2012, 2, 472-506. https://doi.org/10.3390/ani2030472
Fiems LO. Double Muscling in Cattle: Genes, Husbandry, Carcasses and Meat. Animals. 2012; 2(3):472-506. https://doi.org/10.3390/ani2030472
Chicago/Turabian StyleFiems, Leo O. 2012. "Double Muscling in Cattle: Genes, Husbandry, Carcasses and Meat" Animals 2, no. 3: 472-506. https://doi.org/10.3390/ani2030472
APA StyleFiems, L. O. (2012). Double Muscling in Cattle: Genes, Husbandry, Carcasses and Meat. Animals, 2(3), 472-506. https://doi.org/10.3390/ani2030472



