Bats at an Altitude above 2000 m on Pirin Mountain, Bulgaria
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
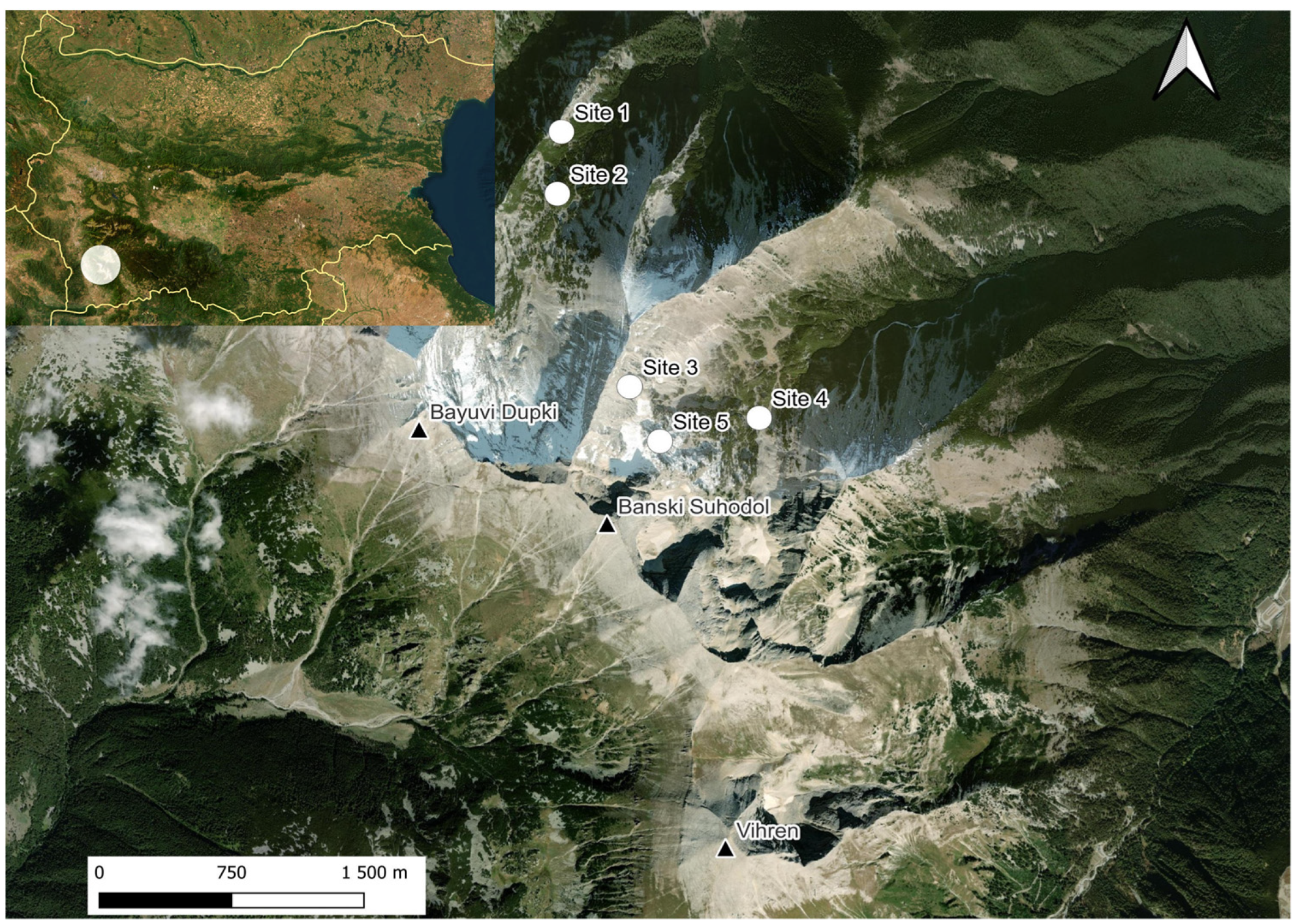
2.1. Study Sites
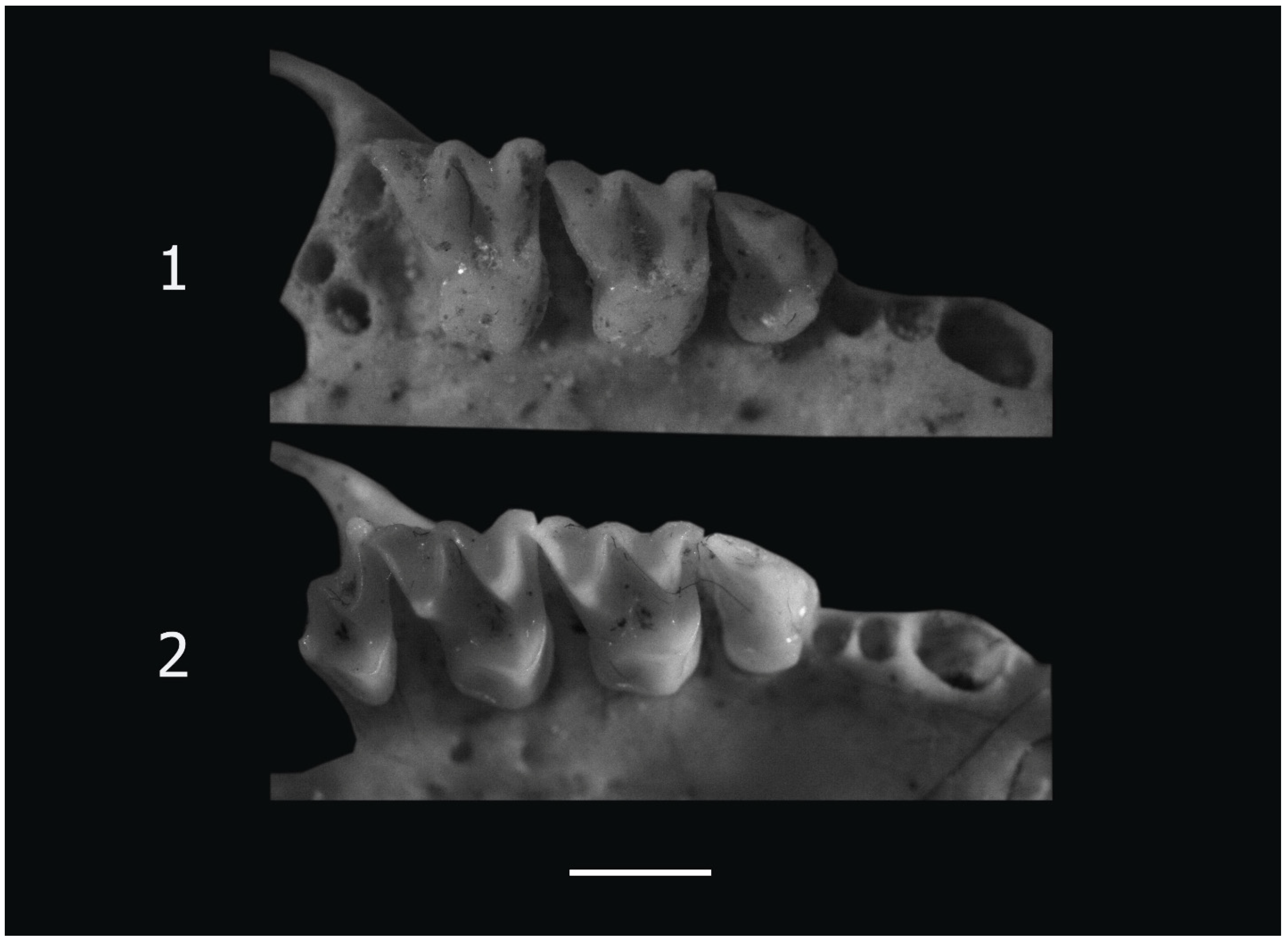
2.2. Skeletal Remains
2.3. Capturing and Bat Identification
2.4. Acoustic Detection
3. Results
3.1. Skeletal Remains
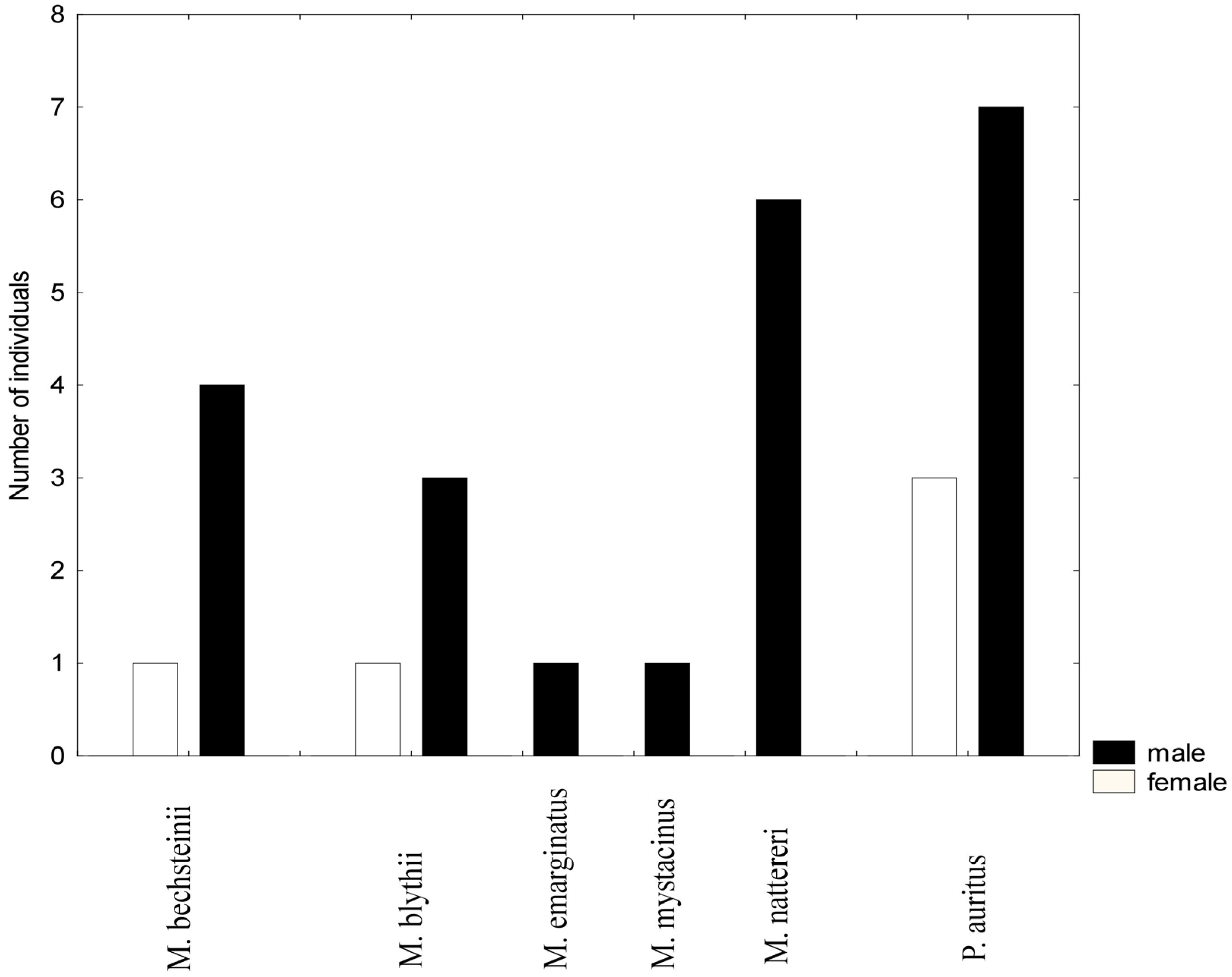
3.2. Mist Netting
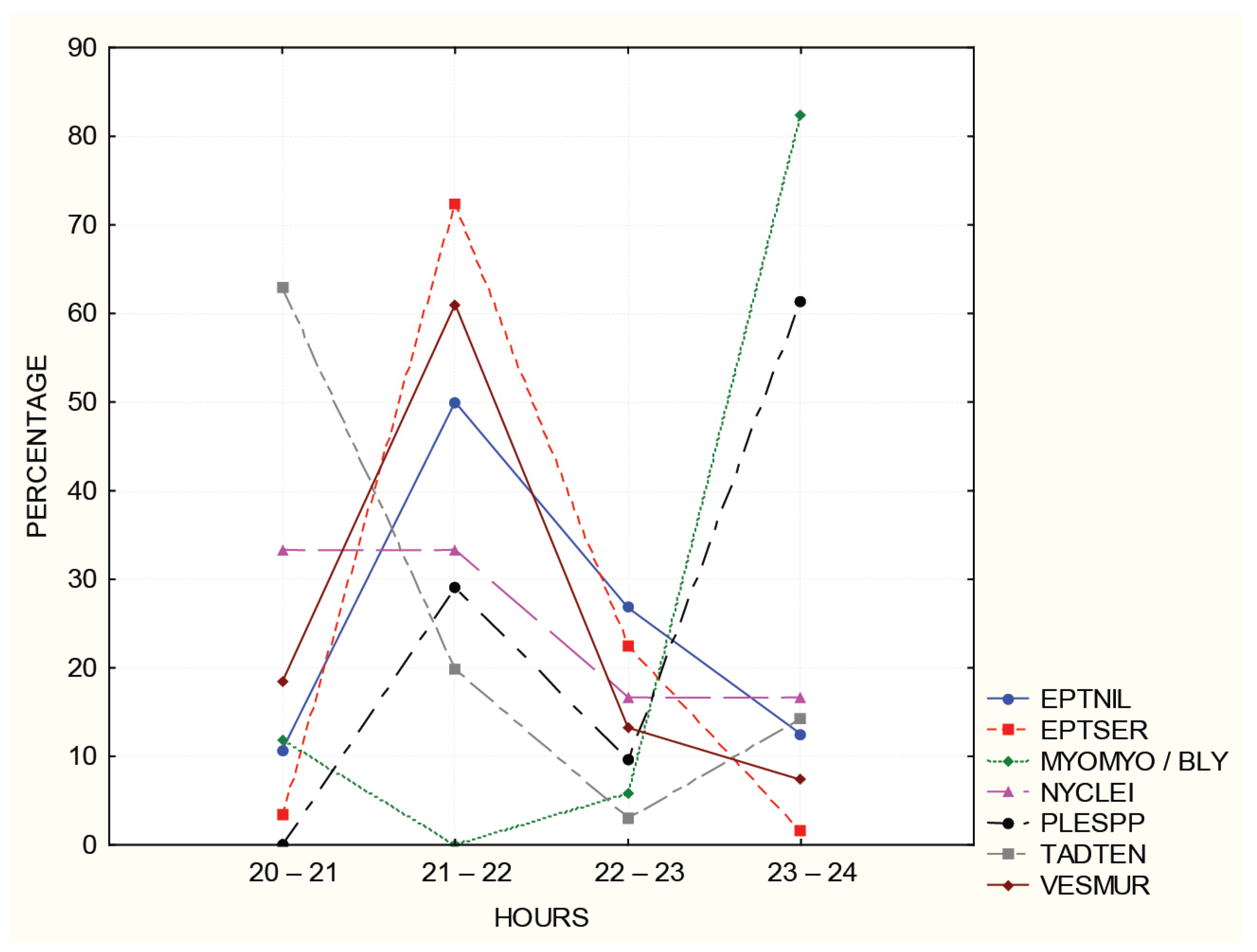
3.3. Acoustic Detecttion
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Popov, V.V. Bats in Bulgaria: Patterns of species distribution richness, rarity and vulnerability derived from distribution models. In Bats; Mikkola, H., Ed.; Intechopen: London, UK, 2018; ISBN 978-1-78923-399-5. [Google Scholar]
- Hanák, V.; Horáček, I. Some comments on the taxonomy of Myotis daubentoni (Kuhl, 1819) (Chiroptera, Mammalia). Myotis 1986, 21–22, 7–19. [Google Scholar]
- Beron, P. Biodiversity of the high mountain terrestrial fauna in Bulgaria. Hist. Nat. Bulg. 1999, 10, 13–33. [Google Scholar]
- Beron, P. On some owls (Aves: Strigidae) in the high parts of Pirin (SW Bulgaria) and their parasitic flies (Diptera: Hippoboscidae). Hist. Nat. Bulg. 2002, 15, 147–149. [Google Scholar]
- Beron, P. Cave Fauna of Bulgaria; East-West Publishing: Varna, Bulgaria, 2015; p. 450. ISBN 978-619-152-712-0. [Google Scholar]
- Beron, P. High—Mountain Terrestrial Fauna in Bulgaria; Prof. Marin Drinov Publishing House of Bulgarian Academy of Sciences: Sofia, Bulgaria, 2022; p. 285. ISBN 978-619-245-258-2. [Google Scholar]
- Benda, P.; Ivanova, T. Long-eared bats, genus Plecotus (Mammalia: Chiroptera), in Bulgaria: A revision of systematic and distributional status. Čas. Nár. Muz. Ř. Přírod. 2003, 172, 157–172. [Google Scholar]
- Benda, P.; Ivanova, T.; Horáček, I.; Hanák, V.; Červený, J.; Gaisler, J.; Gueorguieva, A.; Petrov, B.; Vohralík, V. Bats (Mammalia: Chiroptera) of the Eastern Mediterranean. Part 3. Review of bat distribution in Bulgaria. Acta Soc. Zool. Bohem. 2003, 67, 245–357. [Google Scholar]
- Grunewald, K.; Scheithauer, J. Landscape Development and Climate Change in Southwest Bulgaria (Pirin Mountains), 1st ed.; Springer: Berlin/Heidelberg, Germany, 2011; p. 161. [Google Scholar]
- Barratt, E.M.; Deaville, R.; Burland, T.M.; Bruford, M.W.; Jones, G.; Racey, P.A.; Wayne, R. DNA answers the call of pipistrelle bat species. Nature 1997, 387, 138–139. [Google Scholar] [CrossRef] [PubMed]
- Von Helversen, O.; Heller, K.G.; Mayer, F.; Nemeth, A.; Volleth, M.; Gombkötö, P. Cryptic mammalian species: A new species of whiskered bat (Myotis alcathoe n. sp.) in Europe. Naturwissenschaften 2001, 88, 217–223. [Google Scholar] [CrossRef]
- Mayer, F.; von Helversen, O. Cryptic diversity in European bats. Proc. R. Soc. Lond. B Biol. Sci. 2001, 268, 1825–1832. [Google Scholar] [CrossRef]
- Kiefer, A.; Mayer, F.; Kosuch, J.; von Helversen, O.; Veith, A. Conflicting molecular phylogenies of European long-eared bats (Plecotus) can be explained by cryptic diversity. Mol. Phylogenet. Evol. 2002, 25, 557–566. [Google Scholar] [CrossRef]
- Mucedda, M.; Kiefer, A.; Pidinchedda, E.; Veith, M. A new species of long-eared bat (Chiroptera, Vespertilionidae). Acta Chiropt. 2002, 4, 121–135. [Google Scholar] [CrossRef]
- Spitzenberger, F.; Strelkov, P.; Haring, E. Morphology and mitochondrial DNA sequences show that Plecotus alpinus Kiefer & Veith, 2002 and Plecotus microdontus Spitzenberger, 2002 are synonyms of Plecotus macrobullaris Kuzjakin, 1965. Nat. Croat. 2003, 12, 39–53. [Google Scholar]
- Salicini, I.; Ibáñez, C.; Juste, J. Multilocus phylogeny and species delimitation within the Natterer’s bat species complex in the Western Palearctic. Mol. Phylogenet. Evol. 2011, 61, 888–898. [Google Scholar] [CrossRef] [PubMed]
- Salicini, I.; Ibáñez, C.; Juste, J. Deep differentiation between and within Mediterranean glacial refugia in a flying mammal, the Myotis nattereri bat complex. J. Biogeogr. 2013, 40, 1182–1193. [Google Scholar] [CrossRef]
- Dietz, C.; von Helversen, O. Illustrated Identification Key to the Bats of Europe; Electronic Publication: Tuebingen, Germany; Erlangen, Germany, 2004; p. 35. [Google Scholar]
- Russo, D.; Ancillotto, L.; Hughes, A.C.; Galimberti, A.; Mori, E. Collection of voucher specimens for bat research: Conservation, ethical implications, reduction, and alternatives. Mamm. Rev. 2017, 47, 237–246. [Google Scholar] [CrossRef]
- López-Baucells, A.; Torrent, L.; Rocha, R.; Bobrowiec, P.E.D.; Palmeirim, J.M.; Meyer, C.F.J. Stronger together: Combining automated classifiers with manual post-validation optimizes the workload vs reliability trade-off of species identification in bat acoustic surveys. Ecol. Inform. 2019, 49, 45–53. [Google Scholar] [CrossRef]
- Obrist, M.K.; Boesch, R. BatScope manages acoustic recordings, analyses calls, and classifies bat species automatically. Can. J. Zool. 2018, 96, 939–954. [Google Scholar] [CrossRef]
- Rydell, J.; Nyman, S.; Eklöf, J.; Jones, G.; Russo, D. Testing the performances of automated identification of bat echolocation calls: A request for prudence. Ecol. Indic. 2017, 78, 416–420. [Google Scholar] [CrossRef]
- Russo, D.; Voigt, C. The use of automated identification of bat echolocation calls in acoustic monitoring: A cautionary note for a sound analysis. Ecol. Indic. 2016, 66, 598–602. [Google Scholar] [CrossRef]
- Peshev, T.; Peshev, D.; Popov, V. Fauna Bulgarica. Mammalia; Editio Academica “Marin Drinov”: Sofia, Bulgaria, 2004; p. 632. (In Bulgarian) [Google Scholar]
- Brunet-Rossinni, A.K.; Wilkinson, G.S. Methods for age estimation and the study of senescence in bats. In Ecological and Behavioral Methods for the Study of Bats; Kunz, T.H., Parsons, S., Eds.; The Johns Hopkins University Press: Baltimore, MD, USA, 2009; pp. 315–325. [Google Scholar]
- Pfalzer, G. Inter- und Intraspezifische Variabilität der Sozialrufe Heimischer Fledermausarten (Chiroptera: Vespertilionidae); Mensch & Buch: Berlin, Germany, 2002. [Google Scholar]
- Marckmann, U.; Pfeiffer, B. Bayerisches Landesamt für Umwelt Fledermausschutz in Bayern Bestimmung von Fledermausrufaufnahmen und Kriterien für die Wertung von Akustischen Artnachweisen: Teil 1—Gattungen Nyctalus, Eptesicus, Vespertilio, Pipistrellus (Nyctaloide und Pipistrelloide Arten), Mopsfledermaus, Langohrfledermäuse und Hufeisennasen Bayerns; Fledermausschutz in BayernUmweltSpezial; Bayerisches Landesamt für Umwelt (LfU): Bayern, Germany, 2020; p. 89. [Google Scholar]
- Pfeiffer, B.; Marckmann, U. Bayerisches Landesamt für Umwelt Bestimmung von Fledermausrufaufnahmen und Kriterien für die Wertung von Akustischen Artnachweisen. Teil 2: Myotis; Fledermausschutz in Bayern UmweltSpezial; Bayerisches Landesamt für Umwelt (LfU): Bayern, Germany, 2022; p. 46. Available online: https://www.researchgate.net/publication/366020585_Bayerisches_Landesamt_fur_Umwelt_Bestimmung_von_Fledermausrufaufnahmen_und_Kriterien_fur_die_Wertung_von_akustischen_Artnachweisen_Teil_2_Myotis (accessed on 21 August 2023).
- Zagmajster, M. Display song of party-coloured bat Vespertilio murinus Linnaeus, 1758 (Chiroptera, Mammalia) in southern Slovenia and preliminary study of its variability. Nat. Slov. 2003, 5, 27–41. [Google Scholar] [CrossRef]
- Benda, P.; Tsytsulina, K.A. Taxonomic revision of Myotis mystacinus group (Mammalia: Chiroptera) in the western Palearctic. Acta Soc. Zool. Bohem. 2000, 64, 331–398. [Google Scholar]
- Tsytsulina, K.; Dick, M.H.; Maeda, K.; Masuda, R. Systematics and phylogeography of the steppe whiskered bat Myotis aurascens Kuzyakin, 1935 (Chiroptera, Vespertilionidae). Russ. J. Theriol. 2012, 11, 1–20. [Google Scholar] [CrossRef]
- Çoraman, E.; Dundarova, H.; Dietz, C.; Mayer, F. Patterns of mtDNA introgression suggest population replacement in Palaearctic whiskered bat species. R. Soc. Open Sci. 2020, 7, 191805. [Google Scholar] [CrossRef] [PubMed]
- Alberdi, A.; Aizpurua, O. Plecotus macrobullaris (Chiroptera: Vespertilionidae). Mamm. Species 2018, 50, 26–33. [Google Scholar] [CrossRef]
- Ancillotto, L.; Mori, E.; Bosso, L.; Agnelli, P.; Russo, D. The Balkan long-eared bat (Plecotus kolombatovici) occurs in Italy—First confirmed record and potential distribution. Mamm. Biol. 2019, 96, 61–67. [Google Scholar] [CrossRef]
- Popov, V.; Petrov, B. Agreement of the Conservation of Populations of European Bats (EUROBATS): Report on the Implementation of the Agreement in Bulgaria, September 2003–December 2009; Inf. EUROBATS.MoP6.13; UNEP/EUROBATS Secretariat: Bonn, Germany, 2010. [Google Scholar]
- Spitzenberger, F.; Strelkov, P.; Winkler, H.; Harding, E. A preliminary revision of the genus Plecotus (Chiroptera, Vespertilionidae) based on genetic and morphological results. Zool. Scr. 2006, 35, 187–230. [Google Scholar] [CrossRef]
- Pavlinić, I.; Đaković, M. Identification of four Plecotus species (Chiroptera, Vespertilionidae) in Croatia based on cranial characters. Mammalia 2015, 80, 385–394. [Google Scholar] [CrossRef]
- Bauer, K. Bestimmungsschlüssel österreichischer Säugetiere anhand von Schädelmerkmalen. In Die Säugetiere Österreichs; Spitzeneberger, F., Ed.; Bundesministerium für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft: Graz, Austria, 2001; pp. 863–887. [Google Scholar]
- Blant, M.; Magnin, B.; Ruedi, M. Clé des crânes (Chiroptères). In Mammifères de Suisse—Clés de Détermination; Marchesi, P., Blant, M., Capt, S., Eds.; Fauna Helvetica, CSCF & SSBF: Neuchâtel, Switzerland, 2008; Volume 21, pp. 132–145. [Google Scholar]
- Andriollo, T.; Ruedi, M. Novel molecular tools to identify Plecotus bats in sympatry and a review of their distribution in Switzerland. Rev. Suisse Zool. 2018, 125, 61–72. [Google Scholar]
- Runkel, V.; Gerding, G.; Marckmann, U. The Handbook of Acoustic Bat Detection; Pelagic Publishing Ltd.: London, UK, 2021; ISBN 9781784272210. [Google Scholar]
- Brabant, R.; Laurent, Y.; Dolap, U.; Degraer, S.; Poerink, B.J. Comparing the results of four widely used automated bat identification software programs to identify nine bat species in coastal Western Europe. Belg. J. Zool. 2018, 148, 119–128. [Google Scholar] [CrossRef]
- Győrössy, D.; Győrössy, K.; Estok, P. Comparative analysis of the echolocation calls of the lesser horseshoe bat (Rhinolophus hipposideros) and the Mediterranean horseshoe bat (Rhinolophus euryale) in the Carpathian Basin. North-West. J. Zool. 2020, 16, 204–210. [Google Scholar]
- O’Farrell, M.J.; Gannon, W.L. A Comparison of Acoustic Versus Capture Techniques for the Inventory of Bats. J. Mammal. 1999, 80, 24–30. [Google Scholar] [CrossRef]
- Ochoa, J.; Farrell, M.; Miller, B. Contribution of acoustic methods to the study of insectivorous bat diversity in protected areas from northern Venezuela. Acta Chiropt. 2000, 2, 171–183. [Google Scholar]
- Buresh, I. Über die Chiropterenfauna Bulgariens. BAN 1917, 15, 137–174, (In Bulgarian, German Summary). [Google Scholar]
- Hanák, V.; Josifov, M. Zur Verbreitung der Fledermäuse Bulgariens. Z. Säugetierkunde 1959, 7, 145–151. [Google Scholar]
- Beshkov, V. Bats. In Programme of Biodiversity Conservation. National Strategy of Protection of Bidiversity. Basic Studies; Sakalyan, M., Majni, K., Eds.; NBDCS: Sofia, Bulgaria; BSP: Washington, DC, USA, 1993; Volume 1, pp. 631–644. (In Bulgarian) [Google Scholar]
- Beshkov, V. The Bats of Bulgaria. In Bulgaria’s Biological Diversity: Conservation Status and Needs Assessment; Meine, C., Ed.; Biodiversity Support Program: Washington, DC, USA, 1998; Volumes I and II, pp. 453–466. [Google Scholar]
- Pandurska, R.S. Second record of Tadarida teniotis Rafinesque, 1814 (Chiroptera, Molossidae) from Bulgaria. Acta Zool. Bulg. 1992, 45, 102–103. [Google Scholar]
- Pandurska, R.S.; Beshkov, V.A. Bats (Chiroptera) of high mountains of southern Bulgaria. In Observatoire de Montagne de Moussala, Proceedings of the Symposium International, Borovetz, Sofia, October 1998; Carbonnel, J.P., Stamenov, I.N., Eds.; Institut de Recherche Nucléaire et d’Energie Nucléaire: Sofia, Bulgaria, 1997; pp. 135–140. [Google Scholar]
- Chassovnikarova, T.; Mammals, D.C. Volume Biological Chharacteristics. Pirin National Park Management Plan for the Period 2014–2023. 2014, pp. 632–641. Available online: http://www.pu-pirin.pirin.bg/images/Sreshti/24.07.2015_Obsh_obsajdane/03_Biologichna_h-ka_DNPP.pdf (accessed on 12 January 2022). (In Bulgarian).
- Widerin, K.; Reiter, G. Bat activity and bat migration at the elevation above 3000 m at Hoher Sonnblick massif in the Central Alps, Austria (Chiroptera). Lynx 2018, 49, 223–242. [Google Scholar] [CrossRef][Green Version]
- Widerin, K.; Reiter, G. Bat activity at high altitudes in the Central Alps, Europe. Acta Chiropt. 2017, 19, 379–387. [Google Scholar] [CrossRef]
- Holzhaider, J.; Zhan, A. Bats in the Bavarian Alps: Species composition and utilization of higher altitudes in summer. Mamm. Biol. 2001, 66, 144–154. [Google Scholar]
- Russo, D. Elevation affects the distribution of the two sexes in Daubenton’s bats Myotis daubentonii (Chiroptera: Vespertilionidae) from Italy. Mammalia 2002, 66, 543–551. [Google Scholar] [CrossRef]
- Kañuch, P.; Krištin, A. Altitudinal distribution of bats in the Poľana Mts area (Central Slovakia). Biologia 2006, 61, 605–610. [Google Scholar] [CrossRef]
- Piksa, K.; Bogdanowicz, B.; Tereba, A. Swarming of bats at different elevations in the Carpathian Mountains. Acta Chiropt. 2011, 13, 113–122. [Google Scholar] [CrossRef]
- Grindal, S.D.; Morissette, J.L.; Brigham, R.M. Concentration of bat activity in riparian habitats over an elevational gradient. Can. J. Zool. 1999, 77, 972–977. [Google Scholar] [CrossRef]
- Cryan, P.M.; Bogan, M.A.; Altenbach, J.S. Effect of elevation on distribution of female bats in the Black Hills, South Dakota. J. Mammal. 2000, 81, 719–725. [Google Scholar] [CrossRef]
- Senior, P.; Butlin, R.K.; Altringham, J.D. Sex and segregation in temperate bats. Proc. R. Soc. 2005, 272, 2467–2473. [Google Scholar] [CrossRef] [PubMed]
- Barclay, R.M.R. Population structure of temperate zone insectivorous bats in relation to foraging behaviour and energy demand. J. Anim. Ecol. 1991, 60, 1–13. [Google Scholar] [CrossRef]
- Hamilton, I.M.; Barclay, R.M.R. Patterns of daily torpor and day roost selection by male and female big brown bats (Eptesicus fuscus). Can. J. Zool. 1994, 72, 744–749. [Google Scholar] [CrossRef]
- Davis, W.H.; Hitchcock, H. Biology and migration of the bat, Myotis lucifugus, in New England. J. Mammal. 1965, 46, 296–313. [Google Scholar] [CrossRef]
- Hall, J.S.; Brenner, F.J. Summer netting of bats at a cave in Pennsylvania. J. Mammal. 1968, 49, 779–781. [Google Scholar] [CrossRef]
- Fenton, M.B. Summer activity of Myotis lucifugus (Chiroptera: Vespertilionidae) at hibernacula in Ontario and Quebec. Can. J. Zool. 1969, 47, 597–602. [Google Scholar] [CrossRef]
- Horáček, I.; Zima, J. Net-revealed cave visitation and cave-dwelling in European bats. Folia Zool. 1978, 27, 135–148. [Google Scholar]
- Kiefer, A.; Schreiber, C.; Veith, M. Netzfänge in einem unterirdischen Fledermauquartier in der Eifel (BRD, Rheinland-Pflalz)—Phänologie, Populationsschätzung, Verhalten. Nyctalus 1994, 5, 302–318. [Google Scholar]
- Parsons, K.N.; Jones, G.; Greenaway, F. Swarming activity of temperate zone microchiropteran bats: Effects of sea son, time of night and weather conditions. J. Zool. 2003, 261, 257–264. [Google Scholar] [CrossRef]
- Veith, M.; Kiefer, A.; Johannesen, J.; Seitz, A. The role of swarming sites for maintaining gene flow in the brown long-eared bat (Plecotus auritus). Heredity 2004, 93, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Furmankiewicz, J.; Altringham, J. Genetic structure in a swarming brown long-eared bat (Plecotus auritus) population evidence for mating at swarming sites. Conserv. Genet. 2007, 8, 913–928. [Google Scholar] [CrossRef]
- Furmankiewicz, J. Population size, catchment area and sex-influenced differences in autumn and spring swarming of the brown long-eared bat Plecotus auritus. Can. J. Zool. 2008, 86, 207–216. [Google Scholar] [CrossRef]
- Kerth, G.; Kiefer, A.; Trappmann, C.; Weishaar, M. High gene diversity at swarming sites suggest hot spots for gene flow in the endangered Bechstein’s bat. Conserv. Genet. 2003, 4, 491–499. [Google Scholar] [CrossRef]
- Rivers, N.; Butlin, R.K.; Altringham, J.D. Genetic population structure of Natterer’s bats explained by mating at swarming sites and philopatry. Molec. Ecol. 2005, 14, 4299–4312. [Google Scholar] [CrossRef]
- Piksa, K. Swarming of Myotis mystacinus and other bat species at high elevation in the Tatra Mountains, southern Poland. Acta Chiropt. 2008, 10, 69–79. [Google Scholar] [CrossRef]
- Humphrey, S.R.; Cope, J.B. Population Ecology of the Little Brown Bat, Myotis lucifugus, in Indiana and North-Central Kentucky; Special Publication of The American Society of Mammalogists; The American Society of Mammalogists: Fort Collins, CO, USA, 1976; p. 81. [Google Scholar]
- Furmankiewicz, J.; Górniak, J. Seasonal changes in number and diversity of bat species (Chiroptera) in the Stolec mine (SW Poland). Przyr. Sudet. Zachodnich 2002, 5, 49–70. [Google Scholar]
- Gottfried, I. Use of underground hibernacula by the barbastelle (Barbastella barbastellus) outside the hibernation season. Acta Chiropt. 2009, 11, 363–373. [Google Scholar] [CrossRef]
- Toffoli, R. Elevation record for Myotis daubentonii (Kühl, 1817) in the Italian Western Alps (Mammalia Chiroptera Vespertilionidae). Biodivers. J. 2017, 8, 881–884. [Google Scholar]
- Petrov, P.P. Distribution and status of Myotis bechsteinii in Bulgaria (Chiroptera: Vespertilionidae). Lynx 2006, 37, 179–195. [Google Scholar]
- Hutterer, R.; Ivanova, T.; Meyer-Cords, C.; Rodrigues, L. Bat migrations in Europe. Naturschutz Biol. Vielfalt. 2005, 28, 1–162. [Google Scholar]

| Species | Measurements | N | Min | Mean | Max | SD |
|---|---|---|---|---|---|---|
| Myotis myotis | lmd | 1 | - | 18.1 | - | - |
| hpc | 1 | - | 6.5 | - | - | |
| hmd/m2 | 1 | - | 2.6 | - | - | |
| i-m3 | 1 | - | 11.8 | - | - | |
| Myotis blythii | CBL | 6 | 20 | 20.53 | 21.3 | 0.520 |
| C-M3 | 2 | 9 | - | 9.2 | - | |
| P4-M3 | 6 | 6.3 | 6.58 | 6.7 | 0.147 | |
| M1-M3 | 6 | 5.2 | 5.35 | 5.5 | 0.138 | |
| zg | 6 | 13.6 | 14 | 14.4 | 0.283 | |
| io | 6 | 5.2 | 5.28 | 5.5 | 0.133 | |
| lmd | 3 | 15.8 | 16.2 | 16.3 | 0.265 | |
| hpc | 3 | 5.2 | 5.2 | 5.5 | 0.173 | |
| hmd/m2 | 3 | 2.1 | 2.6 | 2.6 | 0.252 | |
| i-m3 | 1 | - | 10.8 | - | - | |
| p4-m3 | 1 | - | 7.1 | - | - | |
| m1-m3 | 1 | - | 5.7 | - | - | |
| Myotis bechsteinii | CBL | 2 | 16.2 | - | 16.6 | - |
| P4-M3 | 2 | 4.8 | - | 4.9 | - | |
| M1-M3 | 2 | 3.8 | - | 3.95 | - | |
| ZB | 1 | - | 10.5 | - | - | |
| io | 1 | - | 4.4 | - | - | |
| Myotis nattereri | CBL | 3 | 14.4 | 14.58 | 14.8 | - |
| P4-M3 | 3 | 4.4 | 4.5 | 4.6 | - | |
| M1-M3 | 3 | 3.5 | 3.6 | 3.7 | - | |
| ZB | 2 | 9.7 | 9.8 | 9.9 | - | |
| io | 3 | 3.6 | 3.9 | 4.2 | - | |
| Myotis mystacinus | CBL | 2 | 12.6 | - | 12.7 | - |
| P4-M3 | 2 | 3.7 | - | 3.7 | - | |
| M1-M3 | 1 | - | 3.01 | - | - | |
| ZB | 2 | 8 | - | 8 | - | |
| IO | 2 | 3.5 | - | 3.6 | - | |
| lmd | 1 | - | 9.7 | - | - | |
| hpc | 1 | - | 2.7 | - | - | |
| hmd/m2 | 1 | - | 1.1 | - | - | |
| i-m3 | 1 | - | 6.4 | - | - | |
| Myotis brandtii | CBL | 1 | - | 13.6 | - | - |
| P4-M3 | 1 | - | 4.1 | - | - | |
| M1-M3 | 1 | - | 3.3 | - | - | |
| ZB | 1 | - | 7.9 | - | - | |
| IO | 1 | - | 3.7 | - | - | |
| Plecotus auritus | CBL | 10 | 14.7 | 15.19 | 15.5 | 0.260 |
| I1_M3 | 2 | 6.1 | - | 6.4 | - | |
| C-M3 | 2 | 5.3 | - | 5.5 | - | |
| P4-M3 | 10 | 4.1 | 4.25 | 4.4 | 0.097 | |
| M1-M3 | 10 | 3.2 | 3.34 | 3.6 | 0.107 | |
| ZB | 9 | 8.4 | 8.6 | 9 | 0.194 | |
| IO | 11 | 3.5 | 3.60 | 3.7 | 0.072 | |
| LBO | 1 | - | 4.1 | - | - | |
| lmd | 1 | - | 10.6 | - | - | |
| hpc | 1 | - | 3.4 | - | - | |
| hmd/m2 | 1 | - | 1.4 | - | - | |
| i-m3 | 1 | - | 6.9 | - | - |
| Bat Species | Bone Remains | Mist Net | Ultrasound Sequences |
|---|---|---|---|
| Rhinolophus hipposideros | - | - | 1 |
| Barbastella barbastellus | - | - | 10 |
| Myotis myotis | 1 | - | - |
| Myotis blythii | 9 | 12 | |
| Myotis myotis/blythii | 23 | ||
| Myotis emarginatus | - | 4 | 6 |
| Myotis bechsteinii | 2 | 8 | - |
| Myotis nattereri | 3 | 12 | - |
| Myotis brandtii | 1 | - | - |
| Myotis mystacinus | 2 | 3 | - |
| Myotis daubentonii | - | - | 5 |
| Myotis spp. | - | - | 24 |
| Hypsugo savii | - | - | 4 |
| Pipistrellus pipistrellus | - | - | 3 |
| cf. Eptesicus serotinus | - | - | 58 |
| cf. Eptesicus nilssonii | - | - | 56 |
| Vespertilio murinus | - | - | 136 |
| Nyctalus lasiopterus | - | - | 14 |
| cf. Nyctalus leisleri | - | - | 6 |
| Nyctalus noctula | - | - | 3 |
| Plecotus auritus | 12 | 21 | - |
| Plecotus sp. | - | - | 39 |
| Tadarida teniotis | - | - | 272 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dundarova, H.; Popov, V.V. Bats at an Altitude above 2000 m on Pirin Mountain, Bulgaria. Animals 2024, 14, 126. https://doi.org/10.3390/ani14010126
Dundarova H, Popov VV. Bats at an Altitude above 2000 m on Pirin Mountain, Bulgaria. Animals. 2024; 14(1):126. https://doi.org/10.3390/ani14010126
Chicago/Turabian StyleDundarova, Heliana, and Vasil V. Popov. 2024. "Bats at an Altitude above 2000 m on Pirin Mountain, Bulgaria" Animals 14, no. 1: 126. https://doi.org/10.3390/ani14010126
APA StyleDundarova, H., & Popov, V. V. (2024). Bats at an Altitude above 2000 m on Pirin Mountain, Bulgaria. Animals, 14(1), 126. https://doi.org/10.3390/ani14010126







