1. Introduction
Among livestock production, the swine industry provides one of the most important protein sources for human nutrition [
1]. Even though in recent decades a high degree of improvement has been achieved at the genetic level (prolificity, efficiency, and performance), paradoxically, the weaning phase continues to be a critical period within this system. Indeed, the improvement in a sows’ prolificity has led to a reduction in the birthweight of neonates, also increasing litter heterogeneity with serious consequences during postnatal life [
2]. For decades, several antimicrobial compounds, such as zinc oxide at pharmacological doses and antibiotics as growth promoters, have been widely used as a practical strategy to reduce and control postweaning syndrome in pigs. For instance, zinc oxide possesses a multifactorial mode of action, improving digestion and nutrient digestibility, acting as an antioxidant and immunomodulatory molecule with antibacterial effects and, consequently, impacting positively on intestinal morphology and health [
3]. However, antibiotics and zinc oxide are involved in the continuous increase of antibiotic-resistant bacteria and, even more, zinc oxide is the direct cause of negative effects polluting the environment [
4]. Therefore, accordingly to the worldwide cross-sectorial strategy for “One Health” fixed by the FAO, the OIE, and the WHO, the use of antibiotics should be controlled and restricted. Meanwhile, zinc oxide at pharmacological doses has been completely banned in the European Union since June 2022 [
5].
Different feed additives have been studied and proposed as an alternative to zinc oxide to improve postweaning pigs’ health and reduce the incidence of diarrhea [
3]. Citrus flavonoids possess antioxidant and anti-inflammatory properties, and different studies have evidenced their effects as antimicrobial and immunomodulatory molecules [
6,
7,
8]. Additionally, citrus flavonoids and their metabolites have shown beneficial effects on the intestinal barrier through different mechanisms of action [
8]. Citrus flavonoids, as bitter compounds, modify the gene expression of bitter taste receptors (TAS2R), similar to other compounds with antimicrobial, anti-inflammatory, and antioxidant capacity [
9], and some bitter compounds have shown promising effects to substitute zinc oxide at pharmacological doses [
10]. Therefore, it was hypothesized that citrus flavonoids would become a suitable alternative to the use of therapeutic doses of zinc oxide, also reducing the use of antibiotics in weanling pigs. The objective of this study was to evaluate the effect of supplementing citrus flavonoids and amoxicillin in replacing the use of pharmacological doses of zinc oxide together with several antibiotics in weanling piglets as a first step in the demedicalization process of this productive phase. Accordingly, gene expression, microbiota, and histomorphology in the guts of weanling pigs, along with performance parameters and stress markers, were studied to elucidate the potential application of citrus flavonoids in weanling pigs.
The experiment was planned, designed, and conducted at the end of the year 2018, aiming to deal with the reduction of antimicrobials in swine production (antibiotics and zinc oxide) and facing a new scenario and anticipating the ban on zinc oxide in 2022.
4. Discussion
The main objective of this study was to explore a commercial approach to minimize the use of multiple antimicrobial substances in weanling diets, such as zinc oxide and antibiotics, through supplementation with citrus flavonoids. As the trial was performed under commercial farm conditions, the FLAV treatment also included amoxicillin in addition to citrus flavonoids due to the historical sanitary status of the farm and ZnO treatment included more antibiotic molecules following their conventional strategy to reduce postweaning health disorders. Although the major function of antibiotics is to act as antimicrobial substances against bacteria modifying the normal microbial community of the intestine, their anti-inflammatory effects have been also described [
22]. Consequently, the effects of the ZnO and FLAV groups are discussed. However, the potential effects of all substances included in each strategy should be considered and the effects observed cannot be attributed only to zinc oxide or citrus flavonoid supplementation even if some parts of the discussion are focused on the potential effects of the supplementation of these substances.
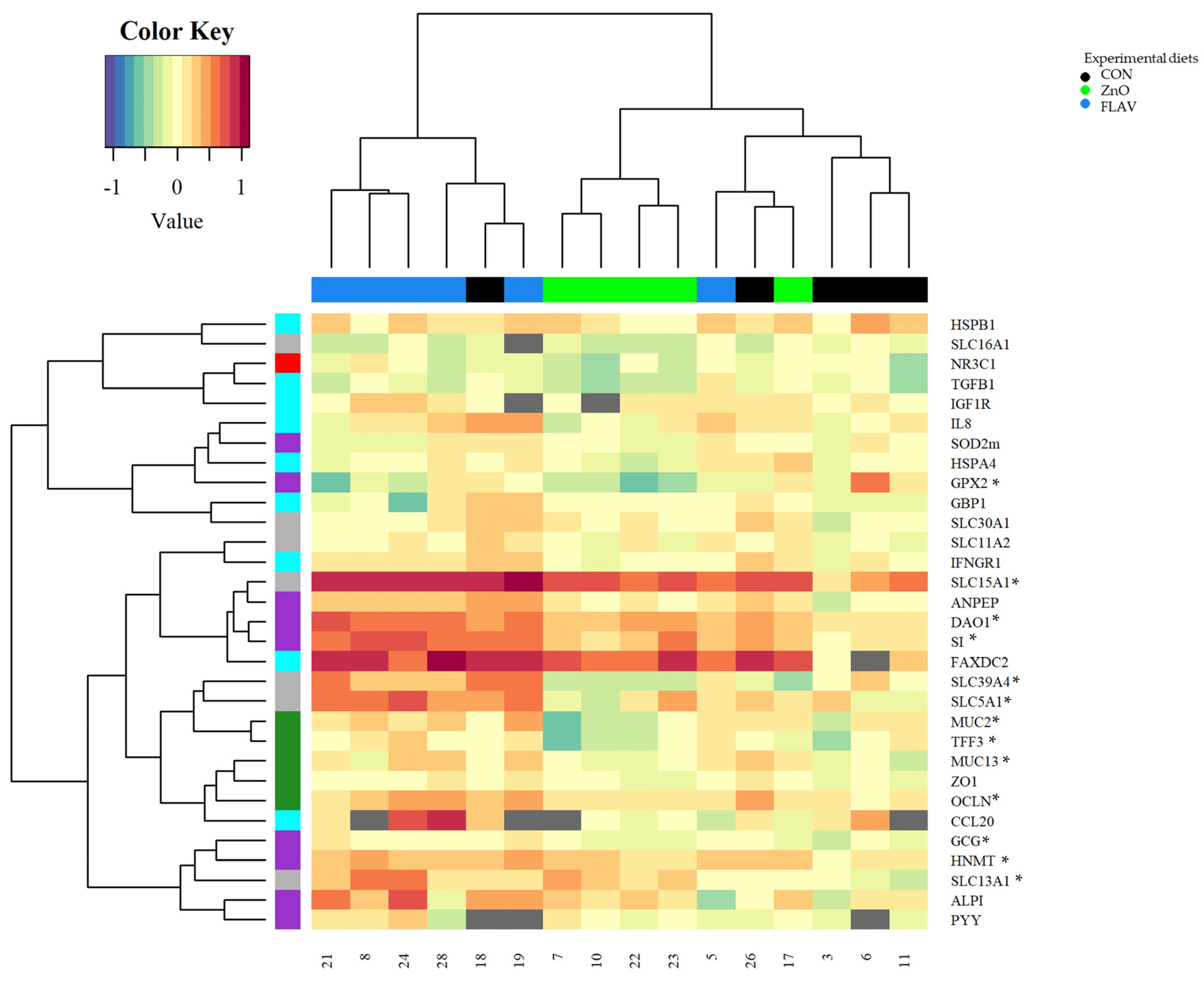
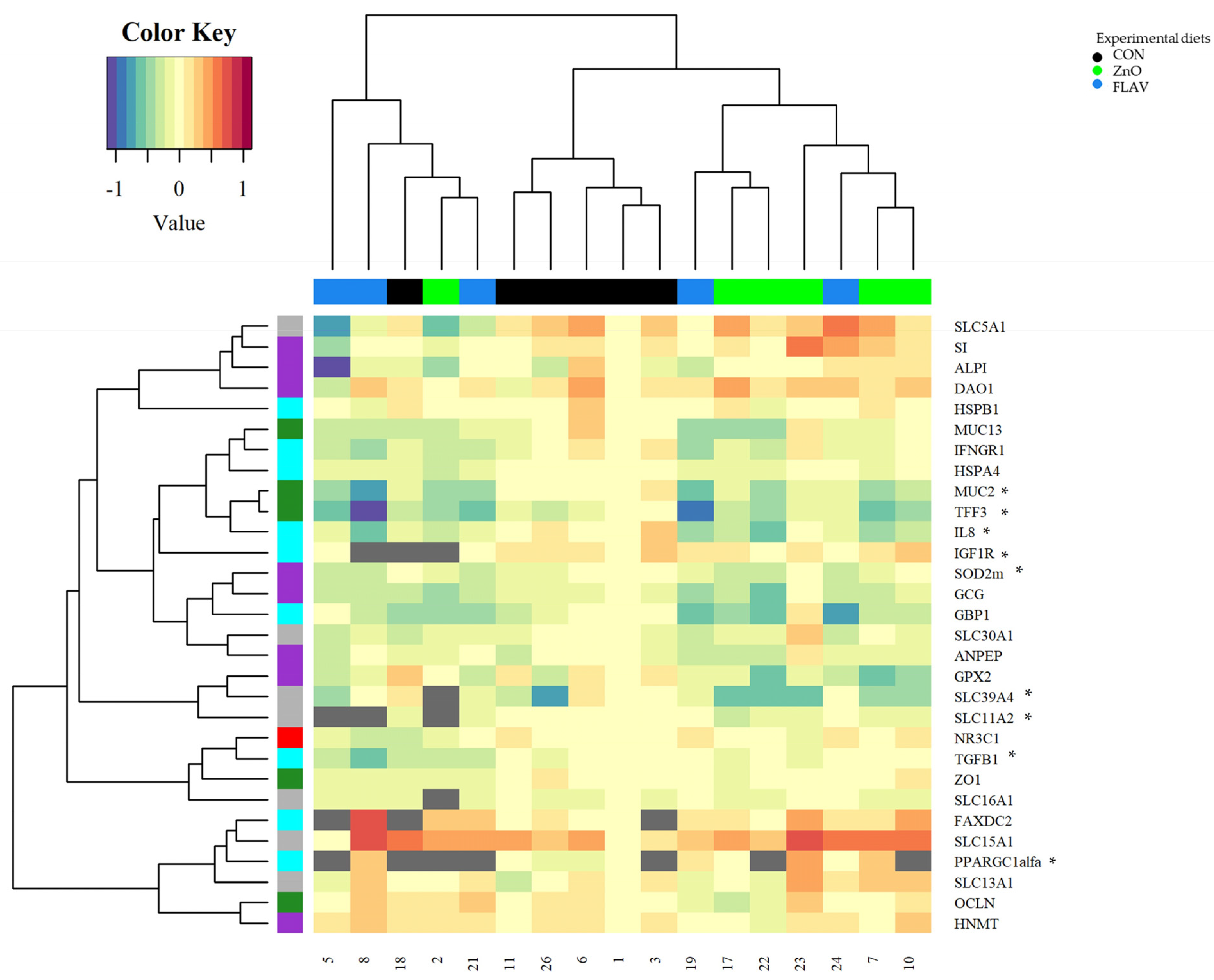
Weaning, in addition to activating stress signaling pathways, can also alter the normal expression of intestinal genes in pigs [
23,
24]. After seven days of feeding, the FLAV treatment increased the jejunum mRNA levels of the genes related to barrier function (MUC13), digestive enzymes (DAO1, GCG, HNMT, and SI), and nutrient transport (SLC13A1 and SLC15A1) compared to those fed the CON diet. Citrus flavonoids, such as naringenin, possess well-demonstrated properties as anti-inflammatory and antioxidant molecules [
8,
25,
26] that would explain the reduction in the inflammatory and antioxidant response in the intestines of these pigs. Interestingly, de Groot et al. [
27] performed a study to establish inflammation patterns in weanling pigs to better understand, in future research, which interventions can positively affect the intestine in this phase. Thus, their results showed that the gene expression of proinflammatory cytokines was increased in the jejunum, ileum, and colon after weaning and during the first 15 days postweaning, for example, the expression of the IL-8 gene. Therefore, the downregulation of the IL-8 gene found in FLAV pigs, but not in the CON group, can be associated with a lower inflammatory environment in the jejunum at day seven. Indeed, citrus flavonoids’ properties improving barrier function have been largely described in humans and animals, mainly attributed to the hesperetin and naringenin flavonoids, which can regulate the expression and secretion of mucins [
8]. For instance, MUC13, a transmembrane mucin glycoprotein, is an important component of intestinal structure and whose unsuitable expression may predispose to infectious and inflammatory diseases [
28]. In the same way, a greater mRNA expression of nutrient transporters and digestive enzymes such as SI may indicate an enhanced intestinal maturity and absorptive function that likely contributed to improved digestion and growth. Interestingly, a downregulation of the Zn transporter gene SLC39A4 was found in ZnO pigs indicating negative feedback to restore zinc homeostasis [
29].
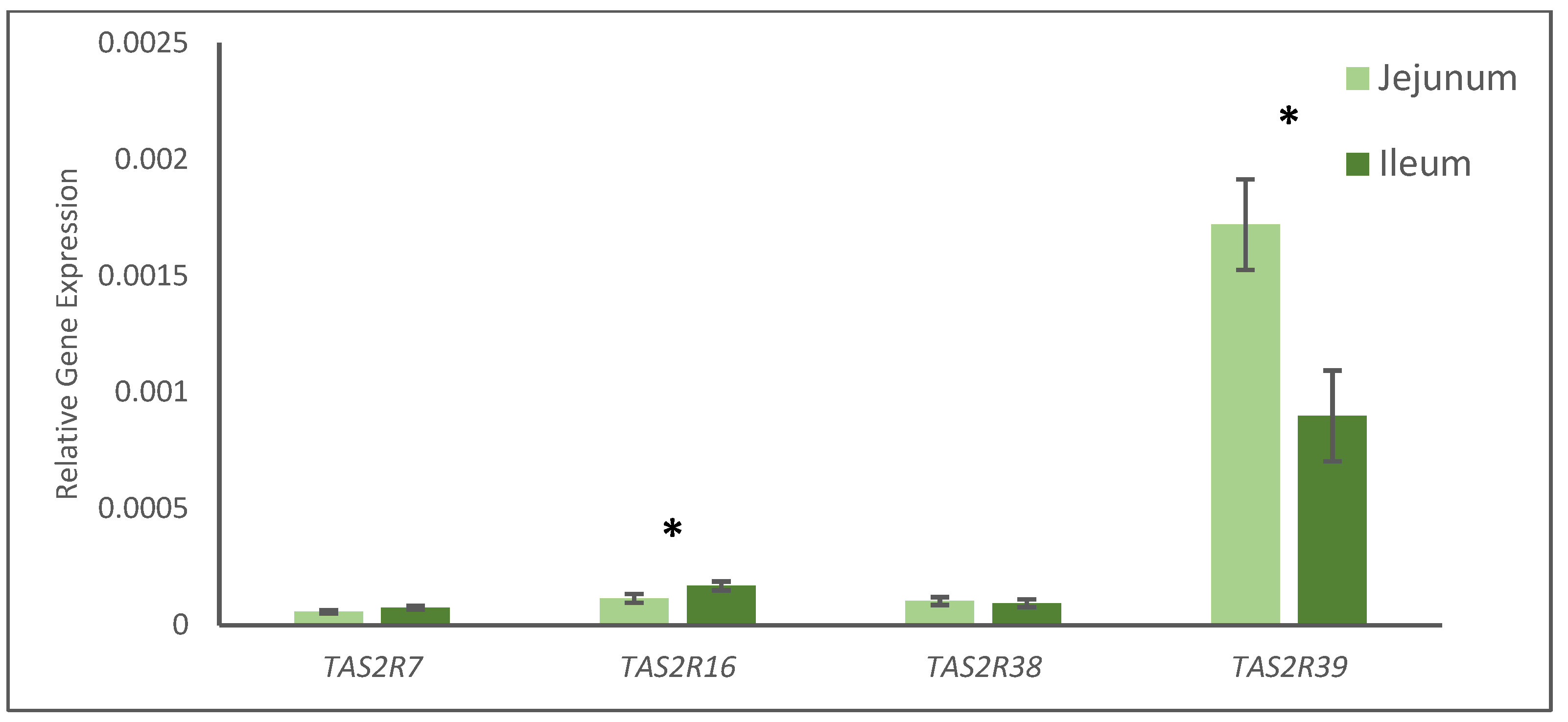
One of the hypotheses of this work was that citrus flavonoids may be a suitable alternative to pharmacological doses of antibiotics and zinc oxide probably led by the effect of bitter compounds on bitter taste receptors (TAS2R). These taste receptors have been described along the gastrointestinal tract constituting the chemosensory system, modulating the proper digestive response (secretion, motility, absorption of nutrients, or aversion), food intake, and behavior [
30,
31,
32,
33]. In the present study, FLAV and ZnO pigs had greater expressed TAS2R39 than CON animals in the intestine at day seven, whereas ZnO increased the gene expression of TAS2R16 as well. Recently, it has been demonstrated that different TAS2Rs differentially modulate food intake in rats through different enteroendocrine secretions [
34]. In accordance with these results, FLAV and ZnO pigs showed a higher ADFI at the end of ST and for the whole study (days 0 to 35). Additionally, activation of TAS2R in the stomachs of rats has been proven to stimulate ghrelin secretion, an orexigenic hormone that enhances gastrointestinal motility [
35] that could explain the increase in the ADFI of pigs when zinc oxide or citrus flavonoids were used in our study. Unfortunately, the gene expression of the TAS2Rs was not analyzed in the stomachs of the pigs, so further research will be needed to elucidate if these receptors are involved in the orexigenic effect observed in FLAV and ZnO animals. Moreover, ZnO treatment also increased gene expression of TAS2R16, whereas FLAV pigs showed numerically higher gene expression than CON, though not statistically different. Paniagua et al. [
9,
36], observed a modulation in the gene expression of TAS2R16 in the rumen epithelium when bulls were supplemented with citrus flavonoids. Therefore, in agreement with these results, bitter substances increased the gene expression of TAS2R16 in the intestine of pigs. Although some antimicrobial molecules, such as chloramphenicol and erythromycin, have been described as bitter molecules activating TAS2R [
37], as far as we know, the antibiotics used in our study have not been confirmed as bitter molecules able to activate TAS2Rs. Consequently, zinc oxide and citrus flavonoid supplementation might explain the activation of TAS2Rs observed in the intestines of the pigs.
No statistical interaction was found for treatment and tissue (jejunum and ileum) when TAS2Rs’ gene expression was analyzed, however, the gene expressions of TAS2R16 and TAS2R39 also showed different expressions depending on the tissue, jejunum, or ileum, whereas TAS2R7 and TAS2R38 were not affected. Colombo et al. [
38], studied the expression of some TAS2Rs in the gastrointestinal tract of five young pigs, and they found differences in different gastrointestinal points (stomach, jejunum, and colon), and concluded that the expression for all TAS2Rs studied was very low. Genes such as TAS2R7 were predominantly found in the stomach, TAS2R16 in the jejunum and TAS2R38 was not frequently expressed. Thus, the results of the present study might be considered in agreement with Colombo et al. [
38], as TAS2R7 evinced low gene expression in the intestines of all the animals; TAS2R16′s expression was found in the jejunum of the animals studied by Colombo et al. [
38], and our results evinced that this receptor responded to the treatment in the tissues studied; probably TAS2R38 is not important in the gastrointestinal tract of the pig, based on the results of both studies; finally, Colombo et al. [
38] did not study the TAS2R39, whereas our results have evinced that this receptor showed a higher degree of gene expression and response to the treatments. As far as we know, this is the first study in pigs where the gene expression of TAS2R39 and its response to bitter molecules have been described. Although much more research is needed to properly elucidate the functions and responses of TAS2R39 in pigs, this receptor might be involved in the multifactorial mechanism of action of zinc oxide and citrus flavonoids observed in pigs at weaning. Furthermore, TAS2R activation is related to the secretion of some enteroendocrine molecules, such as PYY and CKK [
9,
30,
34,
36]. Interestingly, CCK is an intestinal peptide that increases the release of digestive enzymes from the pancreas in the duodenum, facilitating digestion [
30]. Thus, citrus flavonoids might increase digestive enzymes in the jejunum through the activation of TAS2Rs. Additionally, citrus flavonoids exert different positive effects on intestinal health through different biological mechanisms (anti-inflammatory, antioxidant, and barrier function improvement) [
8].
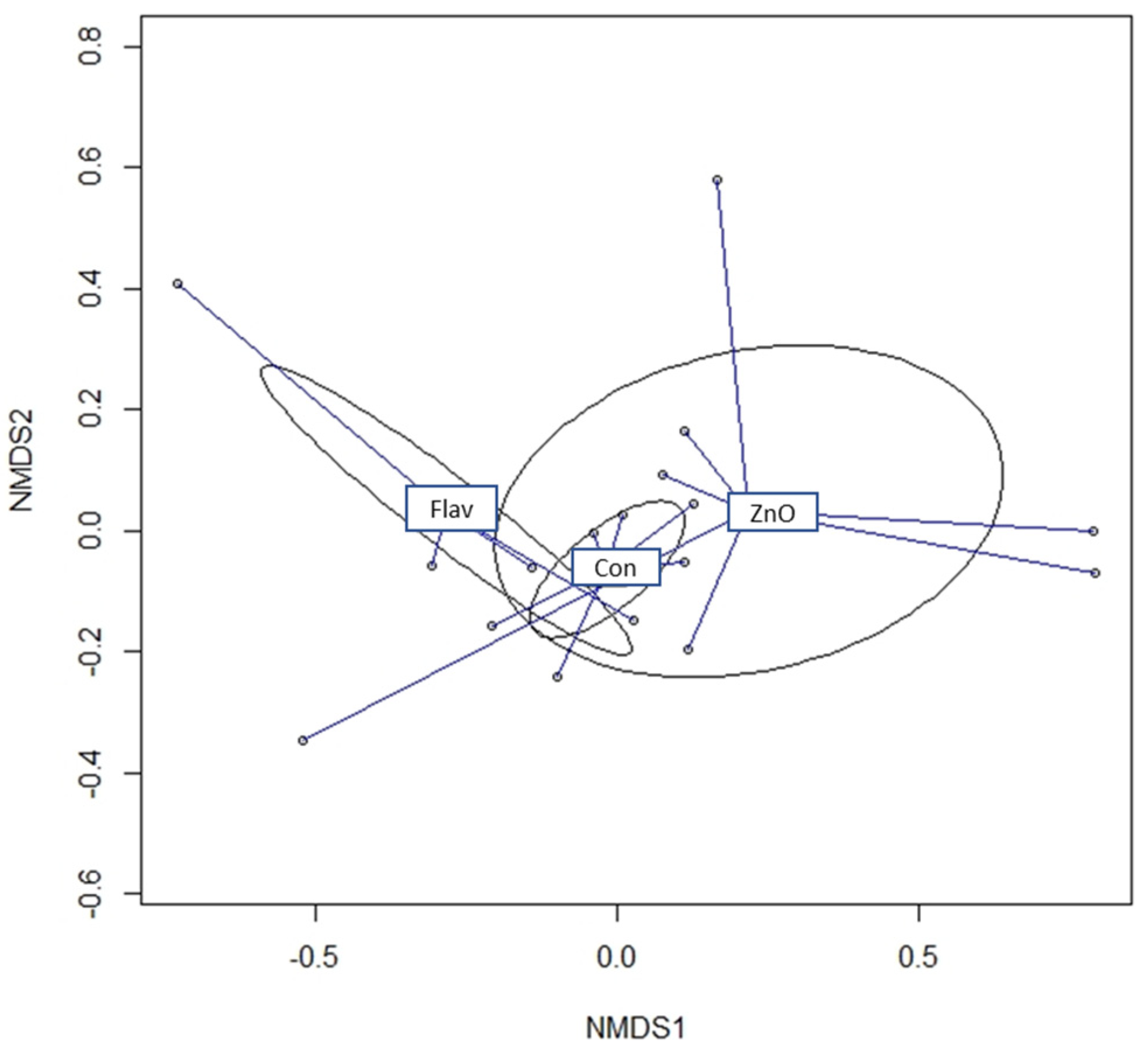
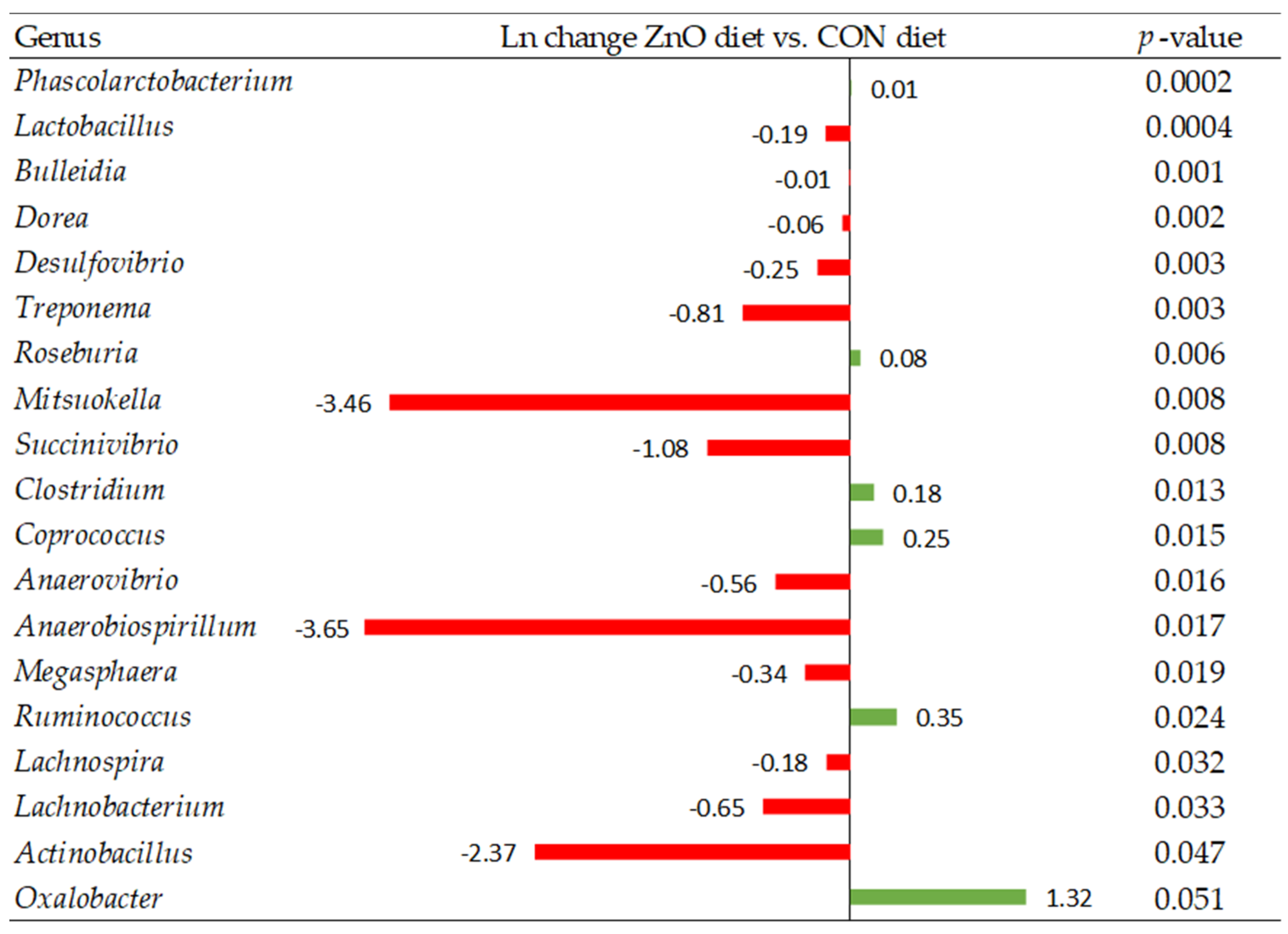
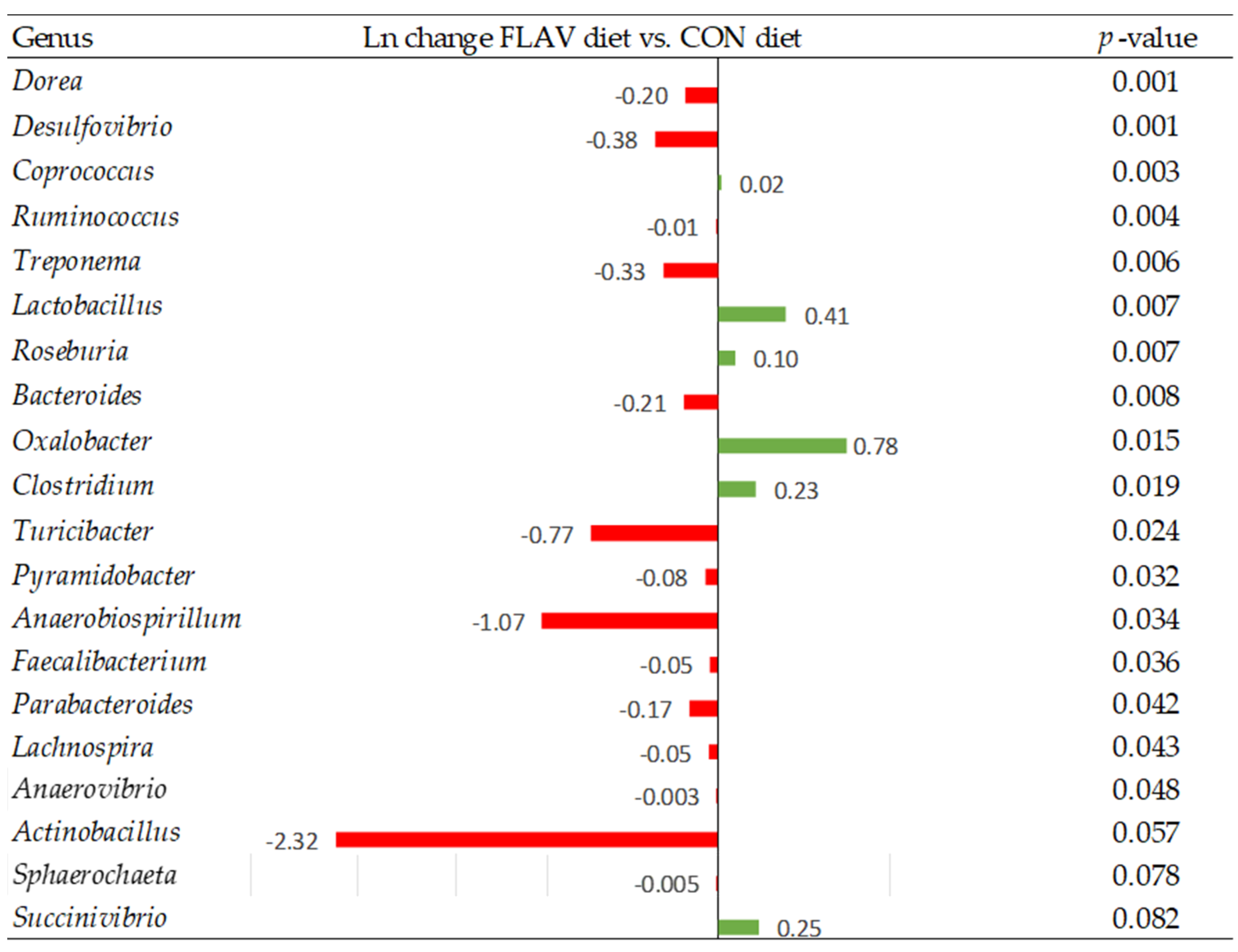
Among all parameters that can influence animal growth performance, intestinal integrity, along with a stable microbiota, is particularly important to enhance feed efficiency and maintain overall health. Curiously, when intestinal morphology was evaluated, no differences were observed between experimental diets. However, taking a deep look into gut health parameters, diets offered to pigs modulate the microbiome community composition. Although alpha diversity indices showed a similar richness within experimental diets, the beta diversity analysis showed dissimilarities among those microbial communities. This finding suggests that the difference is not due to the presence of specific bacteria but to the larger abundance of certain bacteria [
39]. Indeed, pigs fed with the ZnO diet decreased the relative abundance of genera such as
Lactobacillus and
Lachnospira compared to the CON diet. Interestingly, FLAV diets increased the abundance of these same bacteria genera together with
Roseburia compared to the control diet. The importance of the intestinal microbiota for gastrointestinal function and their association with animal health and growth has been previously shown [
39,
40,
41,
42]. Indeed, several bacteria such as species of the
Lactobacillus,
Bifidobacterium,
Lachnospira, or
Roseburia genera are known as beneficial functional microbes which have great potential to contribute to the reduction of the growth of pathogenic bacteria, stimulation of the immune system, feed intake, and feed efficiency [
43]. Furthermore, many members of these bacteria can produce, directly or indirectly, certain metabolites known as postbiotics, such as short-chain fatty acids (SCFA). The SCFAs (i.e., butyrate, propionate, and acetate) are produced by fermenting dietary nondigestible carbohydrates [
44]. It is widely accepted that the role of SCFA as a colonocyte energy supply which instead can exert beneficial effects on intestinal barrier function and reduction of gut inflammation [
45], as well as a crucial physiological effect on several organs, including the brain [
46]. Therefore, these findings support the idea that FLAV treatment might improve pig growth by maintaining a stable and healthy intestinal microbiota able to increase its absorptive capacity.
Moreover, it is worth noting the lower plasma concentration of cortisol found at day seven postweaning in pigs fed the FLAV diet. Cortisol, in addition to being one of the widest biomarkers of stress in pigs, is considered a powerful suppressor of the immune response (production of cytokines and immunoglobulins) and whose secretion predominates under the prolonged exposition to a stressful stimulus [
47]. As mentioned before, the stress in pigs, as a consequence of weaning, can severely impact the body‘s homeostasis and consequently, performance. Therefore, there is a primary interest in minimizing stress levels by improving management activities or through dietary interventions. The present results suggest that the FLAV diet can reduce the stress levels in pigs by approximately twofold compared to the ZnO diet, at least after the first seven days postweaning. Previous studies conducted in beef cattle have shown that supplementing citrus flavonoids can reduce aggressiveness by modulating animal behavior [
9,
37,
48,
49], so citrus flavonoids supplemented in the FLAV treatment might explain this reduction of cortisol in plasma. Nonetheless, further research is needed to corroborate the possible positive effects of citrus flavonoids on animal behavior and stress reduction in pigs, as well as the mechanisms of action associated.
In the present study, pigs fed the FLAV diet showed improved performance during the first 35 days postweaning compared to the CON diet, whereas the ZnO pigs were intermediate. Interestingly, ZnO negatively affected the abundance of some beneficial genera, such as Lactobacillus and Lachnospira, whilst only a few nutrient transporter genes were better expressed in the ilium of these animals compared with the CON piglets. On the contrary, as previously discussed, the FLAV piglets evinced better intestinal maturity together with a greater microbiome gut community, increasing the relative abundance of some genera known as beneficial functional bacteria (Lactobacillus or Roseburia), which might explain the greater performance observed in the FLAV animals at the end of the study. Consequently, although the ZnO group evinced greater ADFI, only numerical improvement was observed for BW and ADG at 35 days postweaning compared with the CON animals, whereas the FLAV piglets achieved greater BW, ADFI, and ADG.