Trace Elements and Persistent Organic Pollutants in Unhatched Loggerhead Turtle Eggs from an Emerging Nesting Site along the Southwestern Coasts of Italy, Western Mediterranean Sea
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
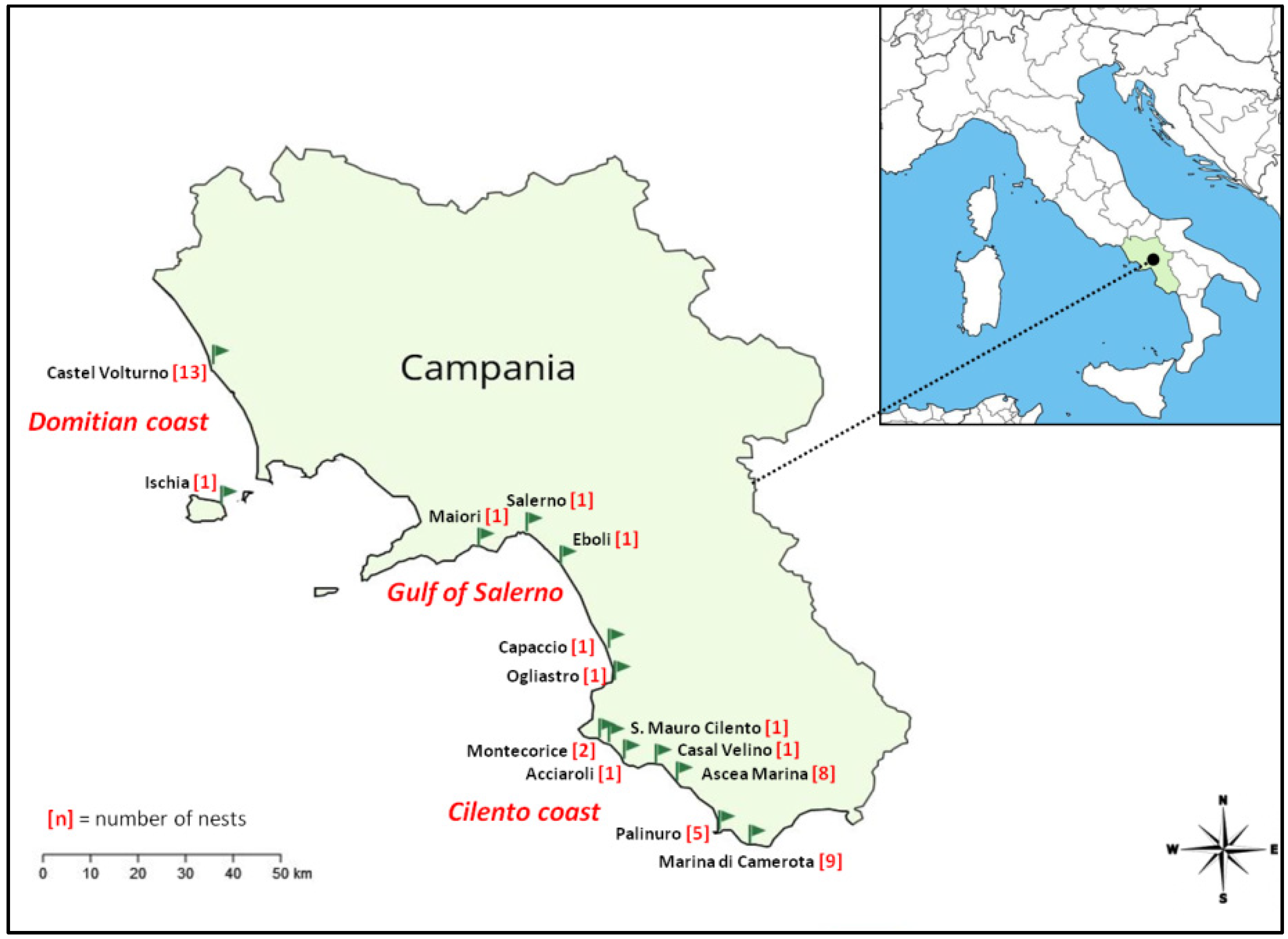
2.1. Sample Collection and Reproductive Parameters
2.2. Sample Preparation
2.3. Trace Element Analysis
2.3.1. Analysis of Metals and Metalloids
2.3.2. Total Mercury Analysis
2.4. PCB Analysis
2.5. OCP Analysis
2.6. Quality Control and Quality Assurance
2.7. Statistical Data Analysis
3. Results and Discussion
3.1. Reproductive Parameters
3.2. Inorganic Pollutants in Eggs
3.3. Organic Pollutants in Eggs
3.4. Correlations with Reproductive Parameters
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Casale, P.; Margaritoulis, D. Sea Turtles in the Mediterranean: Distribution, Threats and Conservation Priorities; IUCN/SSC Marine Turtle Specialist Group: Gland, Switzerland, 2010. [Google Scholar]
- Carreras, C.; Pascual, M.; Tomás, J.; Marco, A.; Hochscheid, S.; Castillo, J.J.; Gozalbes, P.; Parga, M.; Piovano, S.; Cardona, L. Sporadic nesting reveals long distance colonisation in the philopatric loggerhead sea turtle (Caretta caretta). Sci. Rep. 2018, 8, 1435. [Google Scholar] [CrossRef]
- Hochscheid, S.; Maffucci, F.; Abella, E.; Bradai, M.N.; Camedda, A.; Carreras, C.; Claro, F.; de Lucia, G.A.; Jribi, I.; Mancusi, C.; et al. Nesting range expansion of loggerhead turtles in the Mediterranean: Phenology, spatial distribution, and conservation implications. Glob. Ecol. Conserv. 2022, 38, e02194. [Google Scholar] [CrossRef]
- Maffucci, F.; Corrado, R.; Palatella, L.; Borra, M.; Marullo, S.; Hochscheid, S.; Lacorata, G.; Iudicone, D. Seasonal heterogeneity of ocean warming: A mortality sink for ectotherm colonizers. Sci. Rep. 2016, 6, 23983. [Google Scholar] [CrossRef] [PubMed]
- Mancino, C.; Canestrelli, D.; Maiorano, L. Going west: Range expansion for loggerhead sea turtles in the Mediterranean Sea under climate change. Glob. Ecol. Conserv. 2022, 38, e02264. [Google Scholar] [CrossRef]
- Casale, P. Caretta caretta (Mediterranean Subpopulation); IUCN Red List of Threatened Species 2015; e.T83644804A83646294. 2015. Available online: https://www.iucnredlist.org/species/83644804/83646294 (accessed on 31 January 2023).
- Casale, P.; Broderick, A.; Camiñas, J.; Cardona, L.; Carreras, C.; Demetropoulos, A.; Fuller, W.; Godley, B.; Hochscheid, S.; Kaska, Y.; et al. Mediterranean sea turtles: Current knowledge and priorities for conservation and research. Endanger. Species Res. 2018, 36, 229–267. [Google Scholar] [CrossRef]
- Hamann, M.; Godfrey, M.; Seminoff, J.; Arthur, K.; Barata, P.; Bjorndal, K.; Bolten, A.; Broderick, A.; Campbell, L.; Carreras, C.; et al. Global research priorities for sea turtles: Informing management and conservation in the 21st century. Endanger. Species Res. 2010, 11, 245–269. [Google Scholar] [CrossRef]
- Alava, J.J.; Cheung, W.W.L.; Ross, P.S.; Sumaila, U.R. Climate change-contaminant interactions in marine food webs: Toward a conceptual framework. Glob. Chang. Biol. 2017, 23, 3984–4001. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-H.; Chang, C.-P.; Chang, C.-C.; Wang, C.-M.; Lin, C.-F.; Lin, J.-W.; Lin, W.-L.; Liao, H.-J.; Kao, C.-Y.; Fan, P.-S.; et al. Analysis of persistent organochlorine pesticides in shellfish and their risk assessment from aquafarms in Taiwan. Mar. Pollut. Bull. 2021, 172, 112811. [Google Scholar] [CrossRef]
- Renieri, E.A.; Goumenou, M.; Kardonsky, D.A.; Veselov, V.; Alegakis, A.; Buha, A.; Tzatzarakis, M.N.; Nosyrev, A.E.; Rakitskii, V.N.; Kentouri, M.; et al. Indicator PCBs in farmed and wild fish in Greece—Risk assessment for the Greek population. Food Chem. Toxicol. 2019, 127, 260–269. [Google Scholar] [CrossRef]
- Lambiase, S.; Serpe, F.P.; Pilia, M.; Fiorito, F.; Iaccarino, D.; Gallo, P.; Esposito, M. Polychlorinated organic pollutants (PCDD/Fs and DL-PCBs) in loggerhead (Caretta caretta) and green (Chelonia mydas) turtles from Central-Southern Tyrrhenian Sea. Chemosphere 2021, 263, 128226. [Google Scholar] [CrossRef]
- Esposito, M.; Canzanella, S.; Iaccarino, D.; Bruno, T.; Esposito, E.; Di Nocera, F.; Arienzo, M.; Ferrara, L.; Gallo, P. Levels of non-dioxin-like PCBs (NDL-PCBs) in liver of loggerhead turtles (Caretta caretta) from the Tyrrhenian Sea (Southern Italy). Chemosphere 2022, 308, 136393. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.; de Voogt, P. Persistent organic pollutants (POPs): State of the science. Environ. Pollut. 1999, 100, 209–221. [Google Scholar] [CrossRef] [PubMed]
- UNEP (United Nations Environment Programme). Stockholm Convention on Persistent Organic Pollutant; UNEP: Genebra, Switzerland, 2001. [Google Scholar]
- Octaviani, M.; Stemmler, I.; Lammel, G.; Graf, H.F. Atmospheric Transport of Persistent Organic Pollutants to and from the Arctic under Present-Day and Future Climate. Environ. Sci. Technol. 2015, 49, 3593–3602. [Google Scholar] [CrossRef] [PubMed]
- Du Preez, M.; Nel, R.; Bouwman, H. First report of metallic elements in loggerhead and leatherback turtle eggs from the Indian Ocean. Chemosphere 2018, 197, 716–728. [Google Scholar] [CrossRef]
- Casale, P.; Abbate, G.; Freggi, D.; Conte, N.; Oliverio, M.; Argano, R. Foraging ecology of loggerhead sea turtles Caretta caretta in the central Mediterranean Sea: Evidence for a relaxed life history model. Mar. Ecol. Prog. Ser. 2008, 372, 265–276. [Google Scholar] [CrossRef]
- Canzanella, S.; Danese, A.; Mandato, M.; Lucifora, G.; Riverso, C.; Federico, G.; Gallo, P.; Esposito, M. Concentrations of trace elements in tissues of loggerhead turtles (Caretta caretta) from the Tyrrhenian and the Ionian coastlines (Calabria, Italy). Environ. Sci. Pollut. Res. 2021, 28, 26545–26557. [Google Scholar] [CrossRef] [PubMed]
- D’Ilio, S.; Mattei, D.; Blasi, M.; Alimonti, A.; Bogialli, S. The occurrence of chemical elements and POPs in loggerhead turtles (Caretta caretta): An overview. Mar. Pollut. Bull. 2011, 62, 1606–1615. [Google Scholar] [CrossRef]
- Esposito, M.; De Roma, A.; Sansone, D.; Capozzo, D.; Iaccarino, D.; di Nocera, F.; Gallo, P. Non-essential toxic element (Cd, As, Hg and Pb) levels in muscle, liver and kidney of loggerhead sea turtles (Caretta caretta) stranded along the southwestern coasts of Tyrrhenian sea. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2020, 231, 108725. [Google Scholar] [CrossRef]
- Franzellitti, S.; Locatelli, C.; Gerosa, G.; Vallini, C.; Fabbri, E. Heavy metals in tissues of loggerhead turtles (Caretta caretta) from the northwestern Adriatic Sea. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2004, 138, 187–194. [Google Scholar] [CrossRef]
- Keller, J.M.; McClellan-Green, P.D.; Kucklick, J.R.; Keil, D.E.; Peden-Adams, M.M. Effects of Organochlorine Contaminants on Loggerhead Sea Turtle Immunity: Comparison of a Correlative Field Study and In Vitro Exposure Experiments. Environ. Health Perspect. 2006, 114, 70–76. [Google Scholar] [CrossRef]
- Martínez-López, E.; Herrero, D.; López-Berenguer, G.; Peñalver, J. Total Arsenic Concentrations in Sea Turtle Tissues from the Mediterranean Coast of Spain. Bull. Environ. Contam. Toxicol. 2021, 107, 820–826. [Google Scholar] [CrossRef]
- Souza, N.L.N.; Carneiro, M.T.W.D.; Pimentel, E.F.; Frossard, A.; Freire, J.B.; Endringer, D.C.; Júnior, P.D.F. Trace elements influence the hatching success and emergence of Caretta caretta and Chelonia mydas. J. Trace Elem. Med. Biol. 2018, 50, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Yipel, M.; Tekeli, I.O.; Işler, C.T.; Altuğ, M.E. Heavy metal distribution in blood, liver and kidneys of Loggerhead (Caretta caretta) and Green (Chelonia mydas) sea turtles from the Northeast Mediterranean Sea. Mar. Pollut. Bull. 2017, 125, 487–491. [Google Scholar] [CrossRef]
- Alava, J.J.; Keller, J.M.; Kucklick, J.R.; Wyneken, J.; Crowder, L.; Scott, G.I. Loggerhead sea turtle (Caretta caretta) egg yolk concentrations of persistent organic pollutants and lipid increase during the last stage of embryonic development. Sci. Total. Environ. 2006, 367, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Corsolini, S.; Aurigi, S.; Focardi, S. Presence of Polychlorobiphenyls (PCBs) and Coplanar Congeners in the Tissues of the Mediterranean Loggerhead Turtle Caretta caretta. Mar. Pollut. Bull. 2000, 40, 952–960. [Google Scholar] [CrossRef]
- Dennis, M.M.; Poppenga, R.; Conan, A.; Hill, K.; Hargrave, S.; Maroun, V.; Stewart, K.M. Leatherback sea turtle (Dermochelys coriacea) hatch success and essential and nonessential metals in eggs and embryos from nests in St. Kitts (2015). Mar. Pollut. Bull. 2020, 161, 111726. [Google Scholar] [CrossRef]
- Frossard, A.; Vieira, L.V.; Carneiro, M.T.W.D.; Gomes, L.C.; Chippari-Gomes, A.R. Accumulation of trace metals in eggs and hatchlings of Chelonia mydas. J. Trace Elem. Med. Biol. 2020, 62, 126654. [Google Scholar] [CrossRef]
- Muñoz, C.C.; Vermeiren, P. Maternal Transfer of Persistent Organic Pollutants to Sea Turtle Eggs: A Meta-Analysis Addressing Knowledge and Data Gaps Toward an Improved Synthesis of Research Outputs. Environ. Toxicol. Chem. 2020, 39, 9–29. [Google Scholar] [CrossRef]
- Muñoz, C.C.; Vermeiren, P. Profiles of environmental contaminants in hawksbill turtle egg yolks reflect local to distant pollution sources among nesting beaches in the Yucatán Peninsula, Mexico. Mar. Environ. Res. 2018, 135, 43–54. [Google Scholar] [CrossRef]
- Sinaei, M.; Bolouki, M. Metals in Blood and Eggs of Green Sea Turtles (Chelonia mydas) from Nesting Colonies of the Northern Coast of the Sea of Oman. Arch. Environ. Contam. Toxicol. 2017, 73, 552–561. [Google Scholar] [CrossRef]
- Bjorndal, K.A. Nutritional Ecology of Sea Turtles. Copeia 1985, 1985, 736–751. [Google Scholar] [CrossRef]
- Guirlet, E.; Das, K.; Thomé, J.-P.; Girondot, M. Maternal transfer of chlorinated contaminants in the leatherback turtles, Dermochelys coriacea, nesting in French Guiana. Chemosphere 2010, 79, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Van de Merwe, J.P.; Hodge, M.; Olszowy, H.A.; Whittier, J.M.; Lee, S.Y. Using blood samples to estimate persistent organic pollutants and metals in green sea turtles (Chelonia mydas). Mar. Pollut. Bull. 2010, 60, 579–588. [Google Scholar] [CrossRef]
- Hamann, M.; Limpus, C.J.; Owens, D.W. Reproductive Cycles of Males and Females. In The Biology of Sea Turtle; Lutz, P.L., Musick, J.A., Wyneken, J., Eds.; CRC Press: Boca Raton, FL, USA, 2002; Volume 2, pp. 135–161. [Google Scholar]
- Alava, J.J.; Keller, J.M.; Wyneken, J.; Crowder, L.; Scott, G.; Kucklick, J.R. Geographical variation of persistent organic pollutants in eggs of threatened loggerhead sea turtles (Caretta caretta) from southeastern United States. Environ. Toxicol. Chem. 2011, 30, 1677–1688. [Google Scholar] [CrossRef] [PubMed]
- Cammilleri, G.; Calvaruso, E.; Pantano, L.; Cascio, G.L.; Randisi, B.; Macaluso, A.; Vazzana, M.; Caracappa, G.; Giangrosso, G.; Vella, A.; et al. Survey on the presence of non-dioxine-like PCBs (NDL-PCBs) in loggerhead turtles (Caretta caretta) stranded in south Mediterranean coasts (Sicily, Southern Italy). Environ. Toxicol. Chem. 2017, 36, 2997–3002. [Google Scholar] [CrossRef]
- Di Renzo, L.; Ceci, R.; D’Antonio, S.; Di Francesco, G.; Di Giacinto, F.; Ferri, N.; Giansante, C.; Leva, M.; Mariani, G.; Olivieri, V.; et al. Chlorinated Persistent Organic Pollutants (PCDD/Fs and PCBs) in Loggerhead Sea Turtles Stranded along the Central Adriatic Coast. Animals 2022, 12, 3177. [Google Scholar] [CrossRef]
- Febrer-Serra, M.; Renga, E.; Fernández, S.P.; Lassnig, N.; Tejada, S.; Capó, X.; Pinya, S.; Sureda, A. First report of heavy metal presence in muscular tissue of loggerhead turtles Caretta caretta (Linnaeus, 1758) from the Balearic Sea (Balearic Islands, Spain). Environ. Sci. Pollut. Res. 2020, 27, 39651–39656. [Google Scholar] [CrossRef]
- Gómez-Ramírez, P.; Espín, S.; Navas, I.; Martínez-López, E.; Jiménez, P.; María-Mojica, P.; Peñalver, J.; García-Fernández, A.J. Mercury and Organochlorine Pesticides in Tissues of Loggerhead Sea Turtles (Caretta caretta) Stranded Along the Southwestern Mediterranean Coastline (Andalusia, Spain). Bull. Environ. Contam. Toxicol. 2020, 104, 559–567. [Google Scholar] [CrossRef]
- Lazar, B.; Maslov, L.; Romanić, S.H.; Gračan, R.; Krauthacker, B.; Holcer, D.; Tvrtković, N. Accumulation of organochlorine contaminants in loggerhead sea turtles, Caretta caretta, from the eastern Adriatic Sea. Chemosphere 2011, 82, 121–129. [Google Scholar] [CrossRef]
- Maffucci, F.; Caurant, F.; Bustamante, P.; Bentivegna, F. Trace element (Cd, Cu, Hg, Se, Zn) accumulation and tissue distribution in loggerhead turtles (Caretta caretta) from the Western Mediterranean Sea (southern Italy). Chemosphere 2005, 58, 535–542. [Google Scholar] [CrossRef]
- Novillo, O.; Pertusa, J.; Tomás, J. Exploring the presence of pollutants at sea: Monitoring heavy metals and pesticides in loggerhead turtles (Caretta caretta) from the western Mediterranean. Sci. Total. Environ. 2017, 598, 1130–1139. [Google Scholar] [CrossRef] [PubMed]
- Storelli, M.; Barone, G.; Marcotrigiano, G. Polychlorinated biphenyls and other chlorinated organic contaminants in the tissues of Mediterranean loggerhead turtle Caretta caretta. Sci. Total. Environ. 2007, 373, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Storelli, M.; Storelli, A.; D’Addabbo, R.; Marano, C.; Bruno, R.; Marcotrigiano, G. Trace elements in loggerhead turtles (Caretta caretta) from the eastern Mediterranean Sea: Overview and evaluation. Environ. Pollut. 2005, 135, 163–170. [Google Scholar] [CrossRef]
- Kaska, Y.; Furness, R.W. Heavy metals in marine turtle eggs and hatchlings in the Mediterranean. Zool. Middle East 2001, 24, 127–132. [Google Scholar] [CrossRef]
- Godley, B.J.; Thompson, D.R.; Furness, R.W. Do heavy metal concentrations pose a threat to marine turtles from the Mediterranean Sea? Mar. Pollut. Bull. 1999, 38, 497–502. [Google Scholar] [CrossRef]
- Mckenzie, C.; Godley, B.; Furness, R.; Wells, D. Concentrations and patterns of organochlorine contaminants in marine turtles from Mediterranean and Atlantic waters. Mar. Environ. Res. 1999, 47, 117–135. [Google Scholar] [CrossRef]
- Savoca, D.; Arculeo, M.; Arizza, V.; Pace, A.; Melfi, R.; Caracappa, S.; Caracappa, G.; Vullo, C.; Cambera, I.; Visconti, G.; et al. Impact of Heavy Metals in Eggs and Tissues of C. caretta along the Sicilian Coast (Mediterranean Sea). Environments 2022, 9, 88. [Google Scholar] [CrossRef]
- Miller, J.D. Determining clutch size and hatching success. In Research and Management Techniques for the Conservation of Sea Turtles, IUCN/SSC Marine Turtle Specialist Group Publication No. 4; Eckert, K.L., Bjorndal, K.A., Abreu-Grobois, F.A., Donelly, M., Eds.; Consolidated Graphic Communications: Blanchard, PA, USA, 1999; pp. 124–129. [Google Scholar]
- Miller, J.D.; Mortimer, J.A.; Limpus, C.J. A Field Key to the Developmental Stages of Marine Turtles (Cheloniidae) with Notes on the Development of Dermochelys. Chelonian Conserv. Biol. 2017, 16, 111–122. [Google Scholar] [CrossRef]
- Abella, E.; García-Cerdá, R.; Marco, A. Non-invasive monitoring of the fertilization rate in sea turtle nests: Comparison between two techniques. Basic Appl. Herpetol. 2017, 31, 33–34. [Google Scholar] [CrossRef]
- Williamson, S.A.; Evans, R.G.; Robinson, N.J.; Reina, R.D. Hypoxia as a novel method for preventing movement-induced mortality during translocation of turtle eggs. Biol. Conserv. 2017, 216, 86–92. [Google Scholar] [CrossRef]
- Guzman, H.M.; Kaiser, S.; van Hinsberg, V.J. Accumulation of trace elements in leatherback turtle (Dermochelys coriacea) eggs from the south-western Caribbean indicates potential health risks to consumers. Chemosphere 2020, 243, 125424. [Google Scholar] [CrossRef]
- Storelli, M.; Marcotrigiano, G. Heavy metal residues in tissues of marine turtles. Mar. Pollut. Bull. 2003, 46, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Alam, S.K.; Brim, M.S. Organochlorine, PCB, PAH, and metal concentrations in eggs of loggerhead sea turtles (Caretta caretta) from northwest Florida, USA. J. Environ. Sci. Health Part B 2000, 35, 705–724. [Google Scholar] [CrossRef] [PubMed]
- Sakai, H.; Ichihashi, H.; Suganuma, H.; Tatsukawa, R. Heavy metal monitoring in sea turtles using eggs. Mar. Pollut. Bull. 1995, 30, 347–353. [Google Scholar] [CrossRef]
- Bosch, A.C.; O’Neill, B.; Sigge, G.O.; Kerwath, S.E.; Hoffman, L.C. Heavy metals in marine fish meat and consumer health: A review. J. Sci. Food Agric. 2015, 96, 32–48. [Google Scholar] [CrossRef]
- Joseph, J.; Ali, S.; Siang, H.L. Heavy metal compositions in Green turtle (Chelonia mydas) eggs from nesting beaches in Peninsular Malaysia. Asian J. Conserv. Biol. 2014, 3, 83–87. [Google Scholar]
- Ikonomopoulou, M.P.; Olszowy, H.; Limpus, C.; Francis, R.; Whittier, J. Trace element concentrations in nesting flatback turtles (Natator depressus) from Curtis Island, Queensland, Australia. Mar. Environ. Res. 2011, 71, 10–16. [Google Scholar] [CrossRef]
- Lam, J.C.; Tanabe, S.; Chan, S.K.; Lam, M.H.; Martin, M.; Lam, P.K. Levels of trace elements in green turtle eggs collected from Hong Kong: Evidence of risks due to selenium and nickel. Environ. Pollut. 2006, 144, 790–801. [Google Scholar] [CrossRef]
- Kelly, S.M.; Eisenreich, K.M.; Baker, J.E.; Rowe, C.L. Accumulation and maternal transfer of polychlorinated biphenyls in snapping turtles of the upper Hudson River, New York, USA. Environ. Toxicol. Chem. 2008, 27, 2565–2574. [Google Scholar] [CrossRef]
- García-Besné, G.; Valdespino, C.; Rendón-von Osten, J. Comparison of organochlorine pesticides and PCB residues among hawksbill (Eretmochelys imbricata) and green (Chelonia mydas) turtles in the Yucatan Peninsula and their maternal transfer. Mar Pollut Bull. 2015, 91, 139–148. [Google Scholar] [CrossRef]
- Savoca, D.; Arculeo, M.; Vecchioni, L.; Cambera, I.; Visconti, G.; Melfi, R.; Arizza, V.; Piccionello, A.P.; Buscemi, S.; Pace, A. Can phthalates move into the eggs of the loggerhead sea turtle Caretta caretta? The case of the nests on the Linosa Island in the Mediterranean Sea. Mar. Pollut. Bull. 2021, 168, 112395. [Google Scholar] [CrossRef] [PubMed]
- De Solla, S.; Fernie, K.; Ashpole, S. Snapping turtles (Chelydra serpentina) as bioindicators in Canadian Areas of Concern in the Great Lakes Basin. II. Changes in hatching success and hatchling deformities in relation to persistent organic pollutants. Environ. Pollut. 2008, 153, 529–536. [Google Scholar] [CrossRef] [PubMed]
- De Andrés, E.; Gómara, B.; González-Paredes, D.; Ruiz-Martín, J.; Marco, A. Persistent organic pollutant levels in eggs of leatherback turtles (Dermochelys coriacea) point to a decrease in hatching success. Chemosphere 2016, 146, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Marco, A.; da Graça, J.; García-Cerdá, R.; Abella, E.; Freitas, R. Patterns and intensity of ghost crab predation on the nests of an important endangered loggerhead turtle population. J. Exp. Mar. Biol. Ecol. 2015, 468, 74–82. [Google Scholar] [CrossRef]
- Patino-Martinez, J.; Marco, A.; Quiñones, L.; Hawkes, L.A. The potential future influence of sea level rise on leatherback turtle nests. J. Exp. Mar. Biol. Ecol. 2014, 461, 116–123. [Google Scholar] [CrossRef]
- Sarmiento-Ramírez, J.M.; Abella, E.; Martín, M.P.; Tellería, M.T.; López-Jurado, L.F.; Marco, A.; Diéguez-Uribeondo, J. Fusarium solani is responsible for mass mortalities in nests of loggerhead sea turtle, Caretta caretta, in Boavista, Cape Verde. FEMS Microbiol. Lett. 2010, 312, 192–200. [Google Scholar] [CrossRef] [PubMed]
| Nest | Lat. | Long. | Nest Location (Beach) | Clutch Size | Hatching Success % | No Visible Embryo % | Rel. |
|---|---|---|---|---|---|---|---|
| N-01 | 40.134044 | 15.173244 | Ascea (C) | 111 | 57.3 | 34 | No |
| N-02 | 40.98432 | 13.9671 | Castelvolturno (D) | 90 | 74.4 | 12 | Yes |
| N-03 | 40.127743 | 15.178593 | Ascea (C) | 53 | 0 | 87 | No * |
| N-04 | 40.126032 | 15.180173 | Ascea (C) | 120 | 94.1 | 2 | No |
| N-05 | 40.061567 | 15.279357 | Palinuro (C) | 127 | 46.2 | 40 | Yes |
| N-06 | 40.023777 | 15.325655 | Camerota (C) | 98 | 84.5 | 10 | No |
| N-07 | 40.063323 | 15.278012 | Palinuro (C) | 122 | 60.3 | 12 | Yes |
| N-08 | 41.014585 | 13.934977 | Castelvolturno (D) | 88 | 77.3 | 26 | Yes |
| N-09 | 40.987647 | 13.964481 | Castelvolturno (D) | 117 | 56.4 | 0 | Yes |
| N-10 | 40.12566 | 15.18051 | Ascea (C) | 103 | 89.2 | 6 | No |
| N-11 | 40.978316 | 13.972573 | Castelvolturno (D) | 96 | 85.4 | 5 | No |
| N-13 | 40.230402 | 14.959007 | Montecorice (C) | 116 | 77.4 | 16 | Yes |
| N-14 | 40.193241 | 15.018917 | S.Mauro Cilento (C) | 62 | 0 | 10 | Yes * |
| N-15 | 40.01558 | 15.333468 | Camerota (C) | 86 | 90.6 | 3 | Yes |
| N-16 | 40.025687 | 15.323973 | Camerota (C) | 96 | 94.8 | 9 | No |
| N-17 | 40.98723 | 13.96504 | Castelvolturno (D) | 59 | 27.1 | 58 | Yes |
| N-18 | 40.063461 | 15.278016 | Palinuro (C) | 96 | 94.8 | 2 | No |
| N-19 | 40.126477 | 15.179533 | Ascea (C) | 91 | 76.9 | 16 | No |
| N-20 | 40.990556 | 13.961944 | Castelvolturno (D) | 69 | 95.7 | 6 | No |
| N-22 | 40.999299 | 13.95383 | Castelvolturno (D) | 65 | 95.4 | 0 | Yes |
| N-23 | 40.988285 | 13.964209 | Castelvolturno (D) | 54 | 77.8 | 13 | Yes |
| N-24 | 40.005567 | 15.348314 | Camerota (C) | 70 | 31.4 | 39 | Yes |
| N-26 | 40.229115 | 14.961578 | Montecorice (C) | 102 | 84.3 | 8 | Yes |
| N-27 | 40.65988 | 14.79766 | Salerno (S) | 84 | 75 | 6 | Yes |
| N-28 | 40.134726 | 15.172593 | Ascea (C) | 67 | 59.7 | 27 | No |
| N-29 | 40.028213 | 15.321021 | Camerota (C) | 93 | 87.1 | 1 | Yes |
| N-31 | 40.053617 | 15.282841 | Palinuro (C) | 107 | 96.3 | 0 | No |
| N-32 | 40.028996 | 15.320148 | Camerota (C) | 77 | 37.7 | 34 | No |
| N-33 | 40.138943 | 15.168128 | Ascea (C) | 81 | 11.1 | 77 | Yes |
| N-34 | 40.169591 | 15.135516 | Casalvelino (C) | 79 | 91 | 5 | No |
| N-35 | 40.00485 | 15.349346 | Camerota (C) | 74 | 83.8 | 0 | No |
| N-36 | 40.9200191 | 14.0211638 | Castelvolturno (D) | 66 | 71.2 | 17 | Yes |
| N-37 | 40.9190201 | 14.021988 | Castelvolturno (D) | 69 | 94.2 | 3 | No |
| N-38 | 40.98829 | 13.96395 | CastelVolturno (D) | 61 | 52.5 | 28 | No |
| N-39 | 40.427531 | 14.981157 | Capaccio (S) | 55 | 83.6 | 4 | Yes |
| N-40 | 40.189125 | 15.021786 | Acciaroli (C) | 93 | 82.8 | 6 | Yes |
| N-41 | 40.233266 | 14.952506 | Ogliastro (C) | 91 | 86.8 | 1 | Yes |
| N-42 | 41.009253 | 13.941108 | Castelvolturno (D) | 49 | 69.4 | 10 | Yes |
| N-43 | 40.016488 | 15.331952 | Camerota (C) | 83 | 84.3 | 5 | Yes |
| N-46 | 40.497466 | 14.933575 | Eboli (S) | 43 | 78.6 | 12 | Yes |
| N-48 | 40.069724 | 15.274997 | Palinuro (C) | 51 | 88.2 | 0 | Yes |
| N-49 | 40.941694 | 14.0074959 | Castelvolturno (D) | 78 | 74.4 | 3 | Yes |
| N-51 | 40.741606 | 13.864245 | Ischia (I) | 83 | 39.8 | 35 | No |
| N-52 | 40.648808 | 13.63596 | Maiori (S) | 97 | 92.8 | 1 | No |
| N-54 | 40.000393 | 15.367456 | Camerota (C) | 68 | 95.6 | 0 | No |
| N-55 | 40.136656 | 15.170769 | Ascea (C) | 82 | 96.3 | 0 | No |
| Element | GM | AM | SD |
|---|---|---|---|
| As | 1.07 | 1.13 | 0.35 |
| Cd | 0.005 | 0.006 | 0.003 |
| Co | 0.010 | 0.012 | 0.007 |
| Cr | 0.073 | 0.096 | 0.090 |
| Cu | 1.12 | 1.18 | 0.384 |
| Fe | 18.5 | 19.1 | 4.64 |
| Ga | 0.204 | 0.217 | 0.065 |
| Hg | 0.011 | 0.013 | 0.006 |
| Mn | 1.23 | 1.54 | 1.06 |
| Ni | 0.041 | 0.055 | 0.054 |
| Pb | 0.029 | 0.032 | 0.014 |
| Rb | 0.403 | 0.425 | 0.121 |
| Se | 1.07 | 1.12 | 0.33 |
| Sr | 36.6 | 38.7 | 11.4 |
| V | 0.035 | 0.038 | 0.015 |
| Zn | 28.3 | 30.0 | 9.58 |
| PCB | GM | AM | SD |
|---|---|---|---|
| 28 | 0.24 | 0.26 | 0.089 |
| 52 | 0.21 | 0.23 | 0.13 |
| 101 | 0.41 | 0.45 | 0.21 |
| 138 | 10.2 | 11.5 | 6.2 |
| 153 | 19.9 | 22.7 | 13.5 |
| 180 | 9.8 | 11.3 | 7.2 |
| Σ PCBs | 40.9 | 46.4 | 26.9 |
| Hatching Success | Unhatched Eggs | |||
|---|---|---|---|---|
| rs | p | rs | p | |
| Trace element | ||||
| As | −0.035 | 0.82 | 0.15 | 0.33 |
| Cd | −0.0003 | 1.0 | 0.018 | 0.91 |
| Co | 0.27 | 0.082 | −0.15 | 0.34 |
| Cr | 0.25 | 0.097 | −0.15 | 0.32 |
| Cu | −0.29 | 0.054 | −0.16 | 0.30 |
| Fe | 0.25 | 0.092 | −0.13 | 0.38 |
| Ga | 0.12 | 0.42 | −0.067 | 0.66 |
| Hg | 0.19 | 0.20 | −0.21 | 0.16 |
| Mn | −0.037 | 0.81 | 0.079 | 0.61 |
| Ni | 0.26 | 0.083 | −0.20 | 0.20 |
| Pb | 0.023 | 0.88 | 0.011 | 0.94 |
| Rb | −0.083 | 0.59 | 0.12 | 0.43 |
| Se | 0.25 | 0.10 | −0.13 | 0.40 |
| Sr | 0.16 | 0.28 | −0.096 | 0.53 |
| V | −0.0012 | 0.99 | 0.023 | 0.89 |
| Zn | 0.27 | 0.069 | −0.15 | 0.32 |
| PCB | ||||
| PCB-28 | 0.15 | 0.33 | −0.16 | 0.29 |
| PCB-52 | −0.017 | 0.91 | −0.025 | 0.87 |
| PCB-101 | 0.16 | 0.30 | −0.094 | 0.53 |
| PCB-138 | 0.19 | 0.20 | −0.18 | 0.25 |
| PCB-153 | 0.21 | 0.17 | −0.20 | 0.18 |
| PCB-180 | 0.12 | 0.44 | −0.11 | 0.47 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esposito, M.; Canzanella, S.; Iaccarino, D.; Pepe, A.; Di Nocera, F.; Bruno, T.; Marigliano, L.; Sansone, D.; Hochscheid, S.; Gallo, P.; et al. Trace Elements and Persistent Organic Pollutants in Unhatched Loggerhead Turtle Eggs from an Emerging Nesting Site along the Southwestern Coasts of Italy, Western Mediterranean Sea. Animals 2023, 13, 1075. https://doi.org/10.3390/ani13061075
Esposito M, Canzanella S, Iaccarino D, Pepe A, Di Nocera F, Bruno T, Marigliano L, Sansone D, Hochscheid S, Gallo P, et al. Trace Elements and Persistent Organic Pollutants in Unhatched Loggerhead Turtle Eggs from an Emerging Nesting Site along the Southwestern Coasts of Italy, Western Mediterranean Sea. Animals. 2023; 13(6):1075. https://doi.org/10.3390/ani13061075
Chicago/Turabian StyleEsposito, Mauro, Silvia Canzanella, Doriana Iaccarino, Angela Pepe, Fabio Di Nocera, Teresa Bruno, Laura Marigliano, Donato Sansone, Sandra Hochscheid, Pasquale Gallo, and et al. 2023. "Trace Elements and Persistent Organic Pollutants in Unhatched Loggerhead Turtle Eggs from an Emerging Nesting Site along the Southwestern Coasts of Italy, Western Mediterranean Sea" Animals 13, no. 6: 1075. https://doi.org/10.3390/ani13061075
APA StyleEsposito, M., Canzanella, S., Iaccarino, D., Pepe, A., Di Nocera, F., Bruno, T., Marigliano, L., Sansone, D., Hochscheid, S., Gallo, P., & Maffucci, F. (2023). Trace Elements and Persistent Organic Pollutants in Unhatched Loggerhead Turtle Eggs from an Emerging Nesting Site along the Southwestern Coasts of Italy, Western Mediterranean Sea. Animals, 13(6), 1075. https://doi.org/10.3390/ani13061075








