The Rise of Heatstroke as a Method of Depopulating Pigs and Poultry: Implications for the US Veterinary Profession
Abstract
Simple Summary
Abstract
1. Introduction
1.1. Depopulation Defined
1.2. Historical Factors Leading to the Development of Depopulation Methods
1.3. Recent Depopulations
2. Description of Heatstroke-Based Depopulation Methods
2.1. Pathophysiology of Heatstroke
2.2. VSD + TH and Burn Injuries
2.3. Affective States and Animal Welfare Implications of VSD Methods
3. Use of Heatstroke-Based Depopulation Methods
3.1. Rationale for Use of Heatstroke-Based Methods
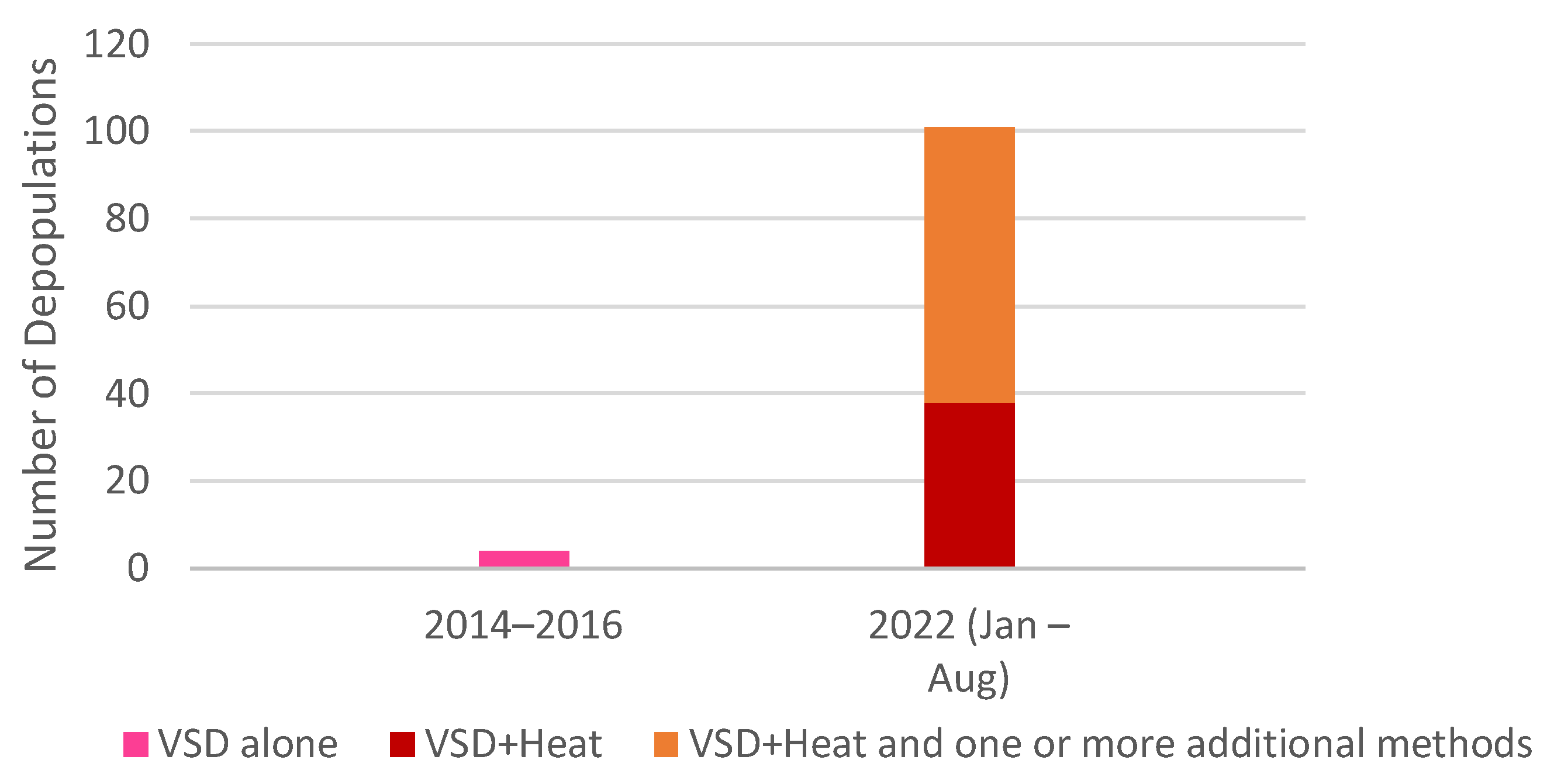
3.2. Rising Use of Heatstroke-Based Methods
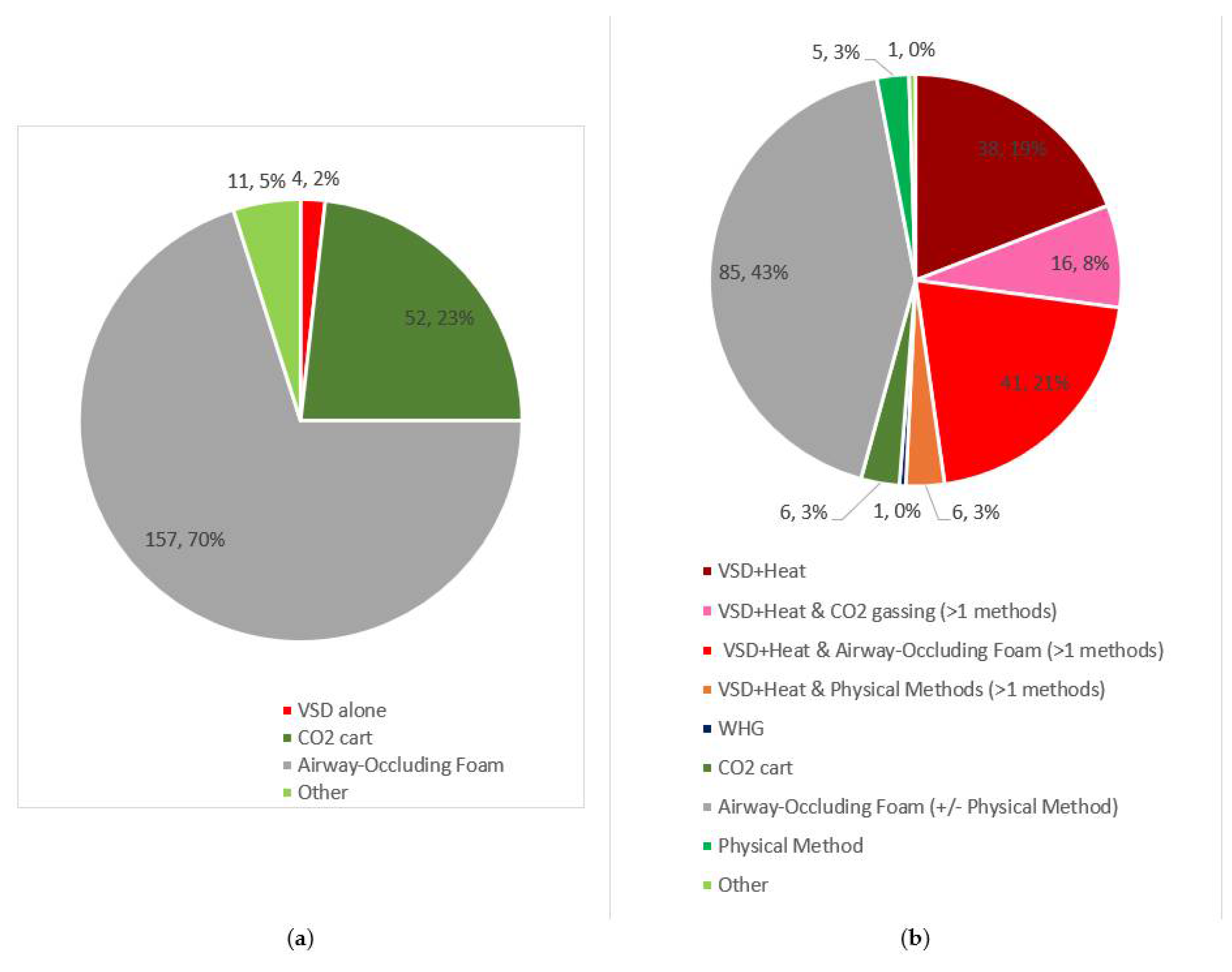
3.2.1. Pigs
3.2.2. Poultry
4. Other Methods of Depopulation
5. The Veterinary Profession’s Ethical Responsibilities Concerning Farm Animal Depopulations
5.1. Specific Opportunities for Veterinary Leadership
5.1.1. Revise the AVMA Guidelines for the Depopulation of Animals
5.1.2. Identify Factors That Increase Animals’ Vulnerability during Emergencies
5.1.3. Engagement with Governmental Agencies and Legislators
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Depopulation Method and Recommended References Containing More Detailed Information | Relevant Species | Regulatory Status [181,182,183,184,185] | Time to LOC † | Intended Mechanism of Killing | Potential Negative Affective States (Including during Handling and Restraint) | Feasibility/Practical Constraints | Biosecurity Considerations | |||
|---|---|---|---|---|---|---|---|---|---|---|
| US (AVMA) * | UK | EU | ||||||||
| Gunshot (free bullet), Captive bolt [41,53,109,123,186,187,188,189,190,191] | Pigs ‡ | Preferred | Allowed | Allowed | Massive diffuse brain damage, via primary injuries (e.g., direct tissue destruction, hemorrhage) and secondary injuries (e.g., cerebral edema). | Anxiety Fear Frustration Helplessness Pain Panic | Ammunition and cartridge supply and storage Interchangeable bolt heads (captive bolts) Requires animal handling/restraint Human safety considerations Specialist training Regular firearm maintenance High staffing requirement Prolonged operational time | Often results in penetrating wound to head, external loss of blood and brain tissue Close human–animal contact Moving equipment between sites | ||
| Electrocution [21,41,53,109,123,192,193,194,195,196,197,198,199,200,201,202,203,204,205,206] | Pigs | Preferred | Allowed | Allowed | Electrocution (one-step head-to-body electrocution or two step electrocution process) | Anxiety Fear Frustration Helplessness Pain Panic | Variable animal handling/restraint Human safety considerations Specialist training Specialized equipment Regular equipment maintenance Variable staffing requirements Variable operational time | With exception of electrocution trailer, close human–animal contact required Moving equipment between sites Movement of sick/injured animals | ||
| Inhaled agents (via mobile containerized gassing units, trailers or modified dumpsters) [53,107,123,207,208,209,210,211,212,213,214,215,216,217,218,219,220,221,222,223,224] | Pigs and Poultry | Preferred | Allowed (only CO2 for pigs) | Allowed (only CO2 for pigs) | Hypercapnic hypoxia/anoxia (CO2, CO2 + N2 or CO2 + Ar); Hypoxia/anoxia (N2 or Ar) | Anxiety Disorientation Dyspnea Fear Frustration Helplessness Nausea Pain Panic | Gas supplies Specialist training Specialist equipment Requires animal handling Human safety considerations Variable staffing requirements Prolonged operational time for the largest facilities | Close human–animal contact required Movement of animals to units Moving equipment between sites | ||
| Inhaled agents (via whole house gassing) [52,53,54,107,127,207,208,209,213,214,215,216,217,218,225,226,227,228,229,230] | Poultry | Preferred | Allowed | Allowed | Hypercapnic hypoxia | Anxiety Disorientation Dyspnea Fear Frustration Helplessness Nausea Pain Panic | Gas supplies Specialist equipment Specialist training Human safety considerations Structures may need to be modified to prevent air leakage | No live animal transport or handling required Moving equipment between sites | ||
| High-expansion § N2-filled foam [53,107,123,231,232,233,234,235,236,237,238,239,240] | Pigs and Poultry | Not mentioned | Allowed for poultry | Allowed for poultry | The bubbles act as a delivery mechanism for the gas, resulting in displacement of air around the animals. As the bubbles pop, the animal is exposed to 100% N2, resulting in death by hypoxia/anoxia. | Anxiety Disorientation Dyspnea Fear Frustration Helplessness Pain Panic | Water and foam concentrate supplies Gas supplies Specialist equipment Specialist training Minimal animal handling Human safety considerations Shed/facility infrastructure limitations on successful operation | No live animal transport or handling required Moving equipment between sites Application in whole-house, in transport vehicles or temporary penning areas | ||
| Low- or medium-expansion § foam (filled with air or inhaled agent, e.g., CO2, Ar, or N2) Includes both water-based foam and compressed air foam [53,59,107,123,231,241,242,243,244,245,246,247,248,249,250] | Pigs and Poultry | Air-filled: preferred/PICC for poultry | Not allowed | Not allowed | The foam is inhaled by the animal resulting in occlusion of airways leading to hypoxia; similar to drowning. With inhaled agent-filled foam, bubble breakdown leads to exposure to gas leading to hypoxia/anoxia +/− hypercapnia | Anxiety Disorientation Dyspnea Fear Frustration Helplessness Nausea Pain Panic | Water and foam concentrate +/− Gas supplies Specialist equipment Specialist training Minimal animal handling and/or restraint Human safety considerations Shed/facility infrastructure limits successful operation | Minimal handling required. Moving equipment between sites | ||
| Sodium Nitrite [251,252,253,254,255,256] | Pigs | PICC | Not allowed | Not allowed | Hypoxemia due to methemoglobinemia | Anxiety Debility Dyspnea (prolonged) Disorientation Fear Frustration Hunger Nausea Pain | Specific storage requirements Short expiration date Poor palatability—compounded formulation or gavaging required Pigs must be trained in advanced for voluntary ingestion Prior food deprivation required Specialist training Greater than 50% of animals have multiple bouts of vomiting Sick pigs may be inappetent, requiring gavage feeding | No live animal transport or handling required for spontaneous ingestion. Close human–animal contact required for gavage administration. Vomitus likely to be present | ||
| VSD + H VSD + TH [1,2,49,50,51,55,106,134,257] | Pigs and Poultry | PICC | Not allowed | Not allowed | Fatal heatstroke/ hyperthermia | Anxiety Debility Dyspnea Disorientation Exhaustion Fear Frustration Helplessness Nausea Malaise Overheating Pain Panic Thirst | Access to equipment (heaters, steam generators) Human safety considerations Specialist training Structure of facility may limit successful operation Retrofitting of barns required (pigs) Minimal live animal handling (poultry) Handling and transport required for pigs Variable operational time | Moving equipment between sites Transport of live pigs to retrofitted barns Vomitus, diarrhea, and respiratory secretions likely to be present | ||
| Controlled demolition [no research could be located] | Poultry | PICC | Not allowed | Not allowed | Trauma Hemorrhage Dehydration Starvation Suffocation Mechanical Asphyxia | Anxiety Chilling Disorientation Dyspnea Fear Frustration Helplessness Hunger Overheating Pain Panic Thirst | Access to demolition equipment Specialist training Cannot be used for caged poultry No live animal handling Human safety considerations High risk of <100% mortality and prolonged time to LOC for some animals Difficult/impossible to check for and euthanize survivors | No live animal transport or handling Difficulty removing carcasses Potential for scavengers to access carcasses Potential environmental contamination Moving equipment between sites | ||
References and Notes
- Baysinger, A.; Senn, M.; Gebhardt, J.; Rademacher, C.; Pairis-Garcia, M. A case study of ventilation shutdown with the addition of high temperature and humidity for depopulation of pigs. J. Am. Vet. Med. Assoc. 2021, 259, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Greenwald, G. Hidden Video and Whistleblower Reveal Gruesome Mass-Extermination Method for Iowa Pigs Amid Pandemic. The Intercept. Available online: https://theintercept.com/2020/05/29/pigs-factory-farms-ventilation-shutdown-coronavirus/ (accessed on 20 September 2022).
- USDA Final Responses to FOIA Request Numbers: APHIS- 02403-F. (February 2022), 2022-APHIS-02981-F (March 2022), 2022-APHIS-03427-F (April 2022), 2022-APHIS-03876-F (May 2022), 2022-APHIS-04627-F (June 2022), 2022-APHIS-04840-F (July 2022), 2022-APHIS-05330-F (August 2022), and 2022-APHIS-05448-F (Records missing from previous requests). Available online: https://awionline.org/sites/default/files/uploads/documents/Bird-Depopulations-Jun-2021-to-Aug-2022.pdf (accessed on 27 December 2022).
- APHIS USDA APHIS|2022 Confirmations of Highly Pathogenic Avian Influenza in Commercial and Backyard Flocks. USDA Animal and Plant Health Inspection Service. Available online: https://www.aphis.usda.gov/aphis/ourfocus/animalhealth/animal-disease-information/avian/avian-influenza/hpai-2022/2022-hpai-commercial-backyard-flocks (accessed on 10 November 2022).
- USDA Final Report for the 2014–2015 Outbreak of Highly Pathogenic Avian Influenza (HPAI) in the United States.; 2016. Available online: https://www.aphis.usda.gov/animal_health/emergency_management/downloads/hpai/2015-hpai-final-report.pdf (accessed on 9 September 2022).
- USDA Final Responses to FOIA Request Number 2017-APHIS-03546-F. Available online: https://awionline.org/sites/default/files/uploads/documents/Bird-Depopulations-Jan-2015-to-Jan-2016.pdf (accessed on 27 December 2022).
- Romanucci, M.; Salda, L.D. Pathophysiology and pathological findings of heatstroke in dogs. Vet. Med. Auckl. NZ 2013, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.F.; Mader, T.L. Managing Heat Stress Episodes in Confined Cattle. Vet. Clin. North Am. Food Anim. Pract. 2018, 34, 325–339. [Google Scholar] [CrossRef] [PubMed]
- McGlone, J.J.; Johnson, A.K.; Sapkota, A.; Kephart, R.K. Transport of market pigs: Improvements in welfare and economics. In Livestock Handling and Transport; Grandin, T., Ed.; CABI: Wallingford, UK, 2014; pp. 298–314. ISBN 978-1-78064-322-9. [Google Scholar]
- Xiong, Y.; Gates, R.; Green-Miller, A. Factors Affecting Trailer Thermal Environment Experienced by Market Pigs Transported in the US. Animals 2018, 8, 203. [Google Scholar] [CrossRef]
- Bender, K.U.S. Vet Expert Shares How to Keep Your Pets Safe During a Heat Wave: “Heat Stroke Is Serious”. Available online: https://people.com/pets/vet-expert-shares-heat-wave-tips-for-pets/ (accessed on 7 September 2022).
- AVMA Hot Cars and Loose Pets. American Veterinary Medical Association. Available online: https://www.avma.org/resources-tools/pet-owners/petcare/hot-cars-and-loose-pets (accessed on 17 October 2022).
- Loeb, J.; Gray, A. Calls for ban on ventilation shutdown in USA. Vet. Rec. 2022, 191, 97. [Google Scholar] [CrossRef] [PubMed]
- Whiting, T.L.; Keane, M.A. Animal protection and mass depopulation. Can. Vet. J. Rev. Vet. Can. 2022, 63, 859–862. [Google Scholar]
- Castillo, D. Is Criticizing Ventilation Shutdown Harmful to Veterinarians? VIN News. Available online: https://news.vin.com/default.aspx?pid=210&Id=10672089&f5=1 (accessed on 17 October 2022).
- Fiala, J. AVMA to Consider Resolution Against Ventilation Shutdown. VIN News. Available online: https://news.vin.com/default.aspx?pid=210&Id=10372360 (accessed on 19 September 2022).
- Letters to the Editor. J. Am. Vet. Med. Assoc. 2021, 259, 1102–1104. [CrossRef]
- AVMA. AVMA Guidelines for the Depopulation of Animals: 2019 Edition. Available online: https://www.avma.org/sites/default/files/resources/AVMA-Guidelines-for-the-Depopulation-of-Animals.pdf (accessed on 10 September 2022).
- AVMA. Pet Ownership and Demographics Sourcebook 2022. Available online: https://www.avma.org/system/files/2022-06/eco-pet-demographic-report-22-low-res.pdf (accessed on 19 September 2022).
- Baysinger, A.; Kogan, L.R. Mental Health Impact of Mass Depopulation of Swine on Veterinarians During COVID-19 Infrastructure Breakdown. Front. Vet. Sci. 2022, 9, 842585. [Google Scholar] [CrossRef]
- Bussolari, C.; Packman, W.; Currin-McCulloch, J.; Strand, E.; Kogan, L. Mass Depopulation of Swine during COVID-19: An Exploration of Swine Veterinarians’ Perspectives. Vet. Sci. 2022, 9, 563. [Google Scholar] [CrossRef]
- Peck, D.F.; Grant, S.; McArthur, W.; Godden, D. Psychological impact of foot-and-mouth disease on farmers. J. Ment. Health 2002, 11, 523–531. [Google Scholar] [CrossRef]
- Van Haaften, E.H.; Olff, M.; Kersten, P.H. The psychological impact of the Foot and Mouth Disease crisis on Dutch dairy farmers. Njas: Wagening. J. Life Sci. 2004, 51, 339–349. [Google Scholar] [CrossRef][Green Version]
- Mort, M.; Convery, I.; Baxter, J.; Bailey, C. Animal Disease and Human Trauma: The Psychosocial Implications of the 2001 UK Foot and Mouth Disease Disaster. J. Appl. Anim. Welf. Sci. 2008, 11, 133–148. [Google Scholar] [CrossRef] [PubMed]
- Chiswell, H. Psychological Morbidity in the Farming Community: A Literature Review. J. Agromed. 2022, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Kollias, N.S. 16 An Overview of the AVMA Working Group on the Psychological Impacts of Humane Endings and Related Guidance Documents. J. Anim. Sci. 2022, 100 (Suppl. S2), 202. [Google Scholar] [CrossRef]
- Anthony, R.; De Paula Vieira, A. One Health Animal Disaster Management: An Ethics of Care Approach. J. Appl. Anim. Welf. Sci. 2022, 25, 180–194. [Google Scholar] [CrossRef] [PubMed]
- Huth, M.; Weich, K.; Grimm, H. Veterinarians between the Frontlines?! The Concept of One Health and Three Frames of Health in Veterinary Medicine. Food Ethics 2019, 3, 91–108. [Google Scholar] [CrossRef]
- Johnson, C. AVMA Humane Endings: An Update; Seventh International Symposium on Animal Mortality Management: Raleigh, NC, USA, 2022. [Google Scholar]
- Steele, J.H. History of Rabies. In The Natural History of Rabies; Baer, G.M., Ed.; Academic Press: New York, NY, USA, 1975; pp. 1–29. ISBN 978-0-12-072401-7. [Google Scholar]
- Kobayashi, M.; Howitt, R.E.; Carpenter, T.E. Model could aid emergency response planning for foot-and-mouth disease outbreaks. Calif. Agric. 2009, 63, 137–142. [Google Scholar] [CrossRef][Green Version]
- Sparrey, J.; Sandercock, D.A.; Sparks, N.H.C.; Sandilands, V. Current and novel methods for killing poultry individually on-farm. World’s Poult. Sci. J. 2014, 70, 737–758. [Google Scholar] [CrossRef]
- U.S. Bureau of the Census. Chapter VI Livestock and Livestock Products. In United States Census of Agriculture: 1950; U.S. Government Printing Office: Washington, DC, USA, 1952; pp. 351–487. [Google Scholar]
- USDA. National Agricultural Statistics Service 2017 Census of Agriculture: United States Summary and State Data; USDA, 2019. Available online: https://www.nass.usda.gov/Publications/AgCensus/2017/Full_Report/Volume_1,_Chapter_1_US/usv1.pdf (accessed on 27 December 2022).
- USDA National Agricultural Statistics Service Chickens and Eggs 2021 Summary; 2022. Available online: https://downloads.usda.library.cornell.edu/usda-esmis/files/1v53jw96n/xk81kp293/cn69n606q/ckegan22.pdf (accessed on 27 December 2022).
- USDA National Agricultural Statistics Service Livestock Slaughter 2021 Summary; 2022. Available online: https://downloads.usda.library.cornell.edu/usda-esmis/files/r207tp32d/pg15cj85z/hd76t466z/lsan0422.pdf (accessed on 27 December 2022).
- Delaware Department of Agriculture Public Records related to HPAI 2022 obtained via Public Records Request by Animal Outlook. Available online: https://awionline.org/sites/default/files/uploads/documents/DE-Public-Records-re-HPAI-Depop-2022.pdf (accessed on 27 December 2022).
- APHIS. Swine 2012 Part III: Changes in the U.S. Swine Industry, 1995−2012. 2017. Available online: https://www.aphis.usda.gov/animal_health/nahms/swine/downloads/swine2012/Swine2012_dr_Trends.pdf (accessed on 27 December 2022).
- USDA Statistical Reporting Service Livestock Slaughter 1969; 1970. Available online: https://downloads.usda.library.cornell.edu/usda-esmis/files/r207tp32d/4f16c698g/k930c1955/LiveSlauSu-04-00-1969.pdf (accessed on 27 December 2022).
- Marchant-Forde, J.N.; Boyle, L.A. COVID-19 Effects on Livestock Production: A One Welfare Issue. Front Vet Sci 2020, 7, 585787. [Google Scholar] [CrossRef]
- Grandin, T. Methods to Prevent Future Severe Animal Welfare Problems Caused by COVID-19 in the Pork Industry. Animals 2021, 11, 830. [Google Scholar] [CrossRef]
- Brosch, C.; Cartanza, G. COVID-19 Acutely Impacted the Delmarva Poultry Industry in Early 2020. Del. J. Public Health 2021, 7, 38–39. [Google Scholar] [CrossRef]
- Hauser, C. Nearly 2 Million Chickens Killed as Poultry Workers Are Sidelined. The New York Times. 28 April 2020. Available online: https://www.nytimes.com/2020/04/28/us/coronavirus-chicken-poultry-farm-workers.html (accessed on 20 September 2022).
- Johnson, A.K.; Rademacher, C.J.; Eggers, J.; Gabler, N.K.; Greiner, L.L.; Kaisand, J.; Karriker, L.A.; Millman, S.T.; Patience, J.F.; Ramirez, B.C.; et al. Innovative strategies for managing swine welfare during the COVID-19 pandemic in Iowa. Transl. Anim. Sci. 2021, 5, txab225. [Google Scholar] [CrossRef] [PubMed]
- Corkery, M.; Yaffe-Bellany, D. Meat Plant Closures Mean Pigs Are Gassed or Shot Instead. The New York Times. 14 May 2020. Available online: https://www.nytimes.com/2020/05/14/business/coronavirus-farmers-killing-pigs.html (accessed on 20 September 2022).
- APHIS. 2016 HPAI Preparedness and Response Plan; 2016. Available online: https://www.aphis.usda.gov/animal_health/downloads/animal_diseases/ai/hpaipreparedness-and-response-plan-2015.pdf (accessed on 26 September 2022).
- World Organisation for Animal Health. Terrestrial Code Chapter 7.6. Killing of Animals for Disease Control Purposes. Available online: https://www.woah.org/fileadmin/Home/eng/Health_standards/tahc/2018/en_chapitre_aw_killing.htm (accessed on 14 September 2022).
- Sifford, R.B.; (APHIS, Riverdale, MD, USA). Personal Communication to Gwendy Reyes-Illg. Available online: https://awionline.org/sites/default/files/uploads/documents/Letter-from-APHIS-to-AWI-Apr-2022.pdf (accessed on 27 December 2022).
- Anderson, K.E.; Livingston, K.; Shah, S.; Martin, M. Evaluating Hen Behavior and Physiological Stressors during VSD for the Development of Humane Methodologies for Mass Depopulation during a Disease Outbreak. Final Report. 2017. Available online: https://www.documentcloud.org/documents/21590311-ncsu-pre-publication-version-from-awi-03302017 (accessed on 27 December 2022).
- Eberle-Krish, K.N.; Martin, M.P.; Malheiros, R.D.; Shah, S.B.; Livingston, K.A.; Anderson, K.E. Evaluation of Ventilation Shutdown in a Multi-level Caged System. J. Appl. Poult. Res. 2018, 27, 555–563. [Google Scholar] [CrossRef]
- Krish, K.N. The Use of Ventilation Shutdown as an Alternative Depopulation Method for Egg Laying and Turkey Breeder Hens in the Event of a Foreign Animal Disease Outbreak (Dissertation). NC State University Libraries. Available online: https://repository.lib.ncsu.edu/handle/1840.20/35787 (accessed on 14 September 2022).
- Sparks, N.H.C.; Sandilands, V.; Raj, A.B.M.; Turney, E.; Pennycott, T.; Voas, A. Use of liquid carbon dioxide for whole-house gassing of poultry and implications for the welfare of the birds. Vet. Rec. 2010, 167, 403–407. [Google Scholar] [CrossRef]
- EFSA Panel on Animal Health and Welfare (AHAW); Nielsen, S.S.; Alvarez, J.; Bicout, D.J.; Calistri, P.; Depner, K.; Drewe, J.A.; Garin-Bastuji, B.; Gonzales Rojas, J.L.; Gortázar Schmidt, C.; et al. Killing for purposes other than slaughter: Poultry. Efsa J. 2019, 17, e05850. [Google Scholar] [CrossRef] [PubMed]
- Beutelschies, S. NVS Development of CO2 Whole House Gassing for Emergency Depopulation of Poultry. YouTube. Available online: https://www.youtube.com/watch?v=TKM_yKFovj4 (accessed on 13 September 2022).
- Zhao, Y.; Xin, H.; Li, L. Modelling and validating the indoor environment and supplemental heat requirement during ventilation shutdown (VSD) for rapid depopulation of hens and turkeys. Biosyst. Eng. 2019, 184, 130–141. [Google Scholar] [CrossRef]
- USDA. HPAI Response: Ventilation Shutdown Plus (+) Policy 2022. Available online: https://www.aphis.usda.gov/animal_health/emergency_management/downloads/hpai/ventilationshutdownpolicy.pdf (accessed on 27 December 2022).
- Kentucky Department of Agriculture Public Records Related to HPAI 2022 Obtained via Public Records Request by Animal Outlook. Available online: https://awionline.org/sites/default/files/uploads/documents/KY-Public-Records-re-HPAI-Depop-2022.pdf (accessed on 27 December 2022).
- Torrella, K. The Horrific Bird Flu That Has Wiped out 36 Million Chickens and Turkeys, Explained. Vox. May 5, 2022. Available online: https://www.vox.com/future-perfect/23053296/bird-flu-chickens-turkeys-cull-depopulation-ventilation-shutdown (accessed on 19 September 2022).
- Kieffer, J.D.; Campler, M.R.; Cheng, T.-Y.; Arruda, A.G.; Youngblood, B.; Moeller, S.J.; Bowman, A.S. Evaluation of a Water-Based Medium-Expansion Foam Depopulation Method in Suckling and Finisher Pigs. Animals 2022, 12, 1041. [Google Scholar] [CrossRef]
- Pork Checkoff PQAPlus Education Handbook Version 4.0. Available online: http://www.porkcdn.com/sites/all/files/documents/PQAPlus/V4.0/Forms/PQAv4e_Handbook.pdf (accessed on 27 December 2022).
- American Association of Swine Veterinarians. Recommendations for the Depopulation of Swine. Available online: https://www.aasv.org/Resources/welfare/depopulation_recommendations.pdf (accessed on 27 December 2022).
- Martin, J.E. Challenges of Animal Welfare Assessment for Controlled Atmosphere Killing Methods. In Proceedings of the UFAW International Conference 2022: Advancing Animal Welfare Science, Edinburgh, UK, 28–29 June 2022; p. T24. [Google Scholar]
- Verhoeven, M.; Gerritzen, M.; Velarde, A.; Hellebrekers, L.; Kemp, B. Time to Loss of Consciousness and Its Relation to Behavior in Slaughter Pigs during Stunning with 80 or 95% Carbon Dioxide. Front. Vet. Sci. 2016, 3, 38. [Google Scholar] [CrossRef]
- Clarkson, J.M.; McKeegan, D.E.F.; Sparrey, J.; Marchesi, F.; Leach, M.C.; Martin, J.E. Determining Candidate Hypobaric Hypoxia Profiles for Humane Killing of Laboratory Mice. Front. Vet. Sci. 2022, 9, 834478. [Google Scholar] [CrossRef]
- Martin, J.E.; Sandilands, V.; Sparrey, J.; Baker, L.; Dixon, L.M.; McKeegan, D.E.F. Welfare assessment of novel on-farm killing methods for poultry. PLoS ONE 2019, 14, e0212872. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.E.; Christensen, K.; Vizzier-Thaxton, Y.; Mitchell, M.A.; McKeegan, D.E.F. Behavioural, brain and cardiac responses to hypobaric hypoxia in broiler chickens. Physiol. Behav. 2016, 163, 25–36. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bruchim, Y.; Horowitz, M.; Aroch, I. Pathophysiology of heatstroke in dogs—Revisited. Temperature 2017, 4, 356–370. [Google Scholar] [CrossRef] [PubMed]
- Bruchim, Y.; Loeb, E.; Saragusty, J.; Aroch, I. Pathological findings in dogs with fatal heatstroke. J. Comp. Pathol. 2009, 140, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Epstein, Y.; Yanovich, R. Heatstroke. N. Engl. J. Med. 2019, 380, 2449–2459. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Rivas, P.A.; Chauhan, S.S.; Ha, M.; Fegan, N.; Dunshea, F.R.; Warner, R.D. Effects of heat stress on animal physiology, metabolism, and meat quality: A review. Meat Sci. 2020, 162, 108025. [Google Scholar] [CrossRef]
- Glazer, J.L. Management of heatstroke and heat exhaustion. Am. Fam. Physician 2005, 71, 2133–2140. [Google Scholar]
- Pearce, S.C.; Mani, V.; Boddicker, R.L.; Johnson, J.S.; Weber, T.E.; Ross, J.W.; Rhoads, R.P.; Baumgard, L.H.; Gabler, N.K. Heat Stress Reduces Intestinal Barrier Integrity and Favors Intestinal Glucose Transport in Growing Pigs. PLoS ONE 2013, 8, e70215. [Google Scholar] [CrossRef]
- Pearce, S.C.; Sanz-Fernandez, M.V.; Hollis, J.H.; Baumgard, L.H.; Gabler, N.K. Short-term exposure to heat stress attenuates appetite and intestinal integrity in growing pigs. J. Anim. Sci. 2014, 92, 5444–5454. [Google Scholar] [CrossRef]
- Alhenaky, A.; Abdelqader, A.; Abuajamieh, M.; Al-Fataftah, A.-R. The effect of heat stress on intestinal integrity and Salmonella invasion in broiler birds. J. Therm. Biol. 2017, 70, 9–14. [Google Scholar] [CrossRef]
- Xie, S.; Nicholson, A.; Woolford, L.; McWhorter, T.J. Physiological, biochemical and histopathological changes associated with heatstroke in the galah ( Eolophus roseicapilla) and rock dove (Columba livia). Avian Pathol. 2019, 48, 57–72. [Google Scholar] [CrossRef]
- Yarmolenko, P.S.; Moon, E.J.; Landon, C.; Manzoor, A.; Hochman, D.W.; Viglianti, B.L.; Dewhirst, M.W. Thresholds for thermal damage to normal tissues: An update. Int. J. Hyperth. 2011, 27, 320–343. [Google Scholar] [CrossRef] [PubMed]
- Lassche, G.; Frenzel, T.; Mignot, M.H.; Jonker, M.A.; van der Hoeven, J.G.; van Herpen, C.M.L.; Scheffer, G.J. Thermal distribution, physiological effects and toxicities of extracorporeally induced whole-body hyperthermia in a pig model. Physiol. Rep. 2020, 8, e14366. [Google Scholar] [CrossRef] [PubMed]
- Gillan, L.A. Blood supply to brains of ungulates with and without a rete mirabile caroticum. J. Comp. Neurol. 1974, 153, 275–290. [Google Scholar] [CrossRef] [PubMed]
- McKechnie, A.E. Regulation of body temperature. In Sturkie’s Avian Physiology; Elsevier: London, UK, 2022; pp. 1231–1264. ISBN 978-0-12-819770-7. [Google Scholar] [CrossRef]
- Porter, W.R.; Witmer, L.M. Avian Cephalic Vascular Anatomy, Sites of Thermal Exchange, and the Rete Ophthalmicum. Anat. Rec. 2016, 299, 1461–1486. [Google Scholar] [CrossRef]
- Richards, S.A. Brain temperature and the cerebral circulation in the chicken. Brain Res. 1970, 23, 265–268. [Google Scholar] [CrossRef]
- Kilgore, D.L.; Birchard, G.F.; Boggs, D.F. Brain temperatures in running quail. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1981, 50, 1277–1281. [Google Scholar] [CrossRef]
- Bech, C.; Midtgård, U. Brain temperature and the rete mirabile ophthalmicum in the Zebra finch (Poephila guttata). J. Comp. Physiol. B 1981, 145, 89–93. [Google Scholar] [CrossRef]
- Andrews, C.J.; Kempf, M.; Kimble, R.; Cuttle, L. Development of a Consistent and Reproducible Porcine Scald Burn Model. PLoS ONE 2016, 11, e0162888. [Google Scholar] [CrossRef]
- Koljonen, V. Hot air sauna burns—Review of their etiology and treatment. J. Burn Care Res. 2009, 30, 705–710. [Google Scholar] [CrossRef]
- Kluger, N.; Laipio, J.; Virolainen, S.; Ranki, A.; Koljonen, V. A Fatal Case of Hot Air Sauna Burn in an Elderly Patient Initially Misdiagnosed as Bullous Pemphigoid. Acta Derm. Venerol. 2011, 91, 732–733. [Google Scholar] [CrossRef]
- Ghods, M.; Corterier, C.; Zindel, K.; Kiene, M.; Rudolf, K.; Steen, M. Hot air sauna burns. Burns 2008, 34, 122–124. [Google Scholar] [CrossRef] [PubMed]
- Koski, A.; Koljonen, V.; Vuola, J. Rhabdomyolysis caused by hot air sauna burn. Burns 2005, 31, 776–779. [Google Scholar] [CrossRef] [PubMed]
- Papp, A. Sauna-related burns: A review of 154 cases treated in Kuopio University Hospital Burn Center 1994-2000. Burn. J. Int. Soc. Burn Inj. 2002, 28, 57–59. [Google Scholar] [CrossRef] [PubMed]
- The North American Sauna Society Sauna Types. The North American Sauna Society. Available online: https://www.saunasociety.org/sauna-types (accessed on 14 September 2022).
- Kudchadkar, S.R.; Hamrick, J.T.; Mai, C.L.; Berkowitz, I.; Tunkel, D. The heat is on... thermal epiglottitis as a late presentation of airway steam injury. J. Emerg. Med. 2014, 46, e43–e46. [Google Scholar] [CrossRef] [PubMed]
- Hathaway, P.B.; Stern, E.J.; Harruff, R.C.; Heimbach, D.M. Steam inhalation causing delayed airway occlusion. Ajr Am. J. Roentgenol. 1996, 166, 322. [Google Scholar] [CrossRef][Green Version]
- Zhai, L.; Adlhart, C.; Spano, F.; Innocenti Malini, R.; Piątek, A.K.; Li, J.; Rossi, R.M. Prediction of Steam Burns Severity using Raman Spectroscopy on ex vivo Porcine Skin. Sci. Rep. 2018, 8, 6946. [Google Scholar] [CrossRef]
- Holm, S.; Engström, O.; Melander, M.; Horvath, M.C.S.; Fredén, F.; Lipcsey, M.; Huss, F. Cutaneous steam burns and steam inhalation injuries: A literature review and a case presentation. Eur. J. Plast. Surg. 2022, 45, 881–896. [Google Scholar] [CrossRef]
- Grandin, T.; (Colorado State U, Ft. Collins, CO, USA). Personal Communication to Indu Mani, 18 September 2022.
- Shamohammadi, H.; Mehrabi, S.; Sadrizadeh, S.; Yaghoubi, M.; Abouali, O. 3D numerical simulation of hot airflow in the human nasal cavity and trachea. Comput. Biol. Med. 2022, 147, 105702. [Google Scholar] [CrossRef]
- Wan, J.; Zhang, G.; Qiu, Y.; Wen, C.; Fu, T. Heat dissipation by blood circulation and airway tissue heat absorption in a canine model of inhalational thermal injury. Burns 2016, 42, 548–555. [Google Scholar] [CrossRef]
- Moritz, A.R.; Henriques, F.C.; McLean, R. The Effects of Inhaled Heat on the Air Passages and Lungs: An Experimental Investigation. Am. J. Pathol. 1945, 21, 311–331. [Google Scholar]
- Ede, T.; Lecorps, B.; von Keyserlingk, M.A.G.; Weary, D.M. Symposium review: Scientific assessment of affective states in dairy cattle. J. Dairy Sci. 2019, 102, 10677–10694. [Google Scholar] [CrossRef] [PubMed]
- Désiré, L.; Boissy, A.; Veissier, I. Emotions in farm animals. Behav. Process. 2002, 60, 165–180. [Google Scholar] [CrossRef]
- Mellor, D.; Beausoleil, N. Extending the “Five Domains” model for animal welfare assessment to incorporate positive welfare states. Anim. Welf. 2015, 24, 241–253. [Google Scholar] [CrossRef]
- McKeegan, D.; Martin, J. Improving welfare in poultry slaughter. In Understanding the Behaviour and Improving the Welfare of Chickens; Nicol, C., Ed.; Burleigh Dodds Series in Agricultural Science; Burleigh Dodds Science Publishing: Philadelphia, PA, USA, 2020; pp. 459–508. ISBN 978-1-78676-422-5. [Google Scholar]
- Martin, J.E.; Christensen, K.; Vizzier-Thaxton, Y.; McKeegan, D.E.F. Effects of analgesic intervention on behavioural responses to Low Atmospheric Pressure Stunning. Appl. Anim. Behav. Sci. 2016, 180, 157–165. [Google Scholar] [CrossRef]
- Forkman, B.; Boissy, A.; Meunier-Salaün, M.-C.; Canali, E.; Jones, R.B. A critical review of fear tests used on cattle, pigs, sheep, poultry and horses. Physiol. Behav. 2007, 92, 340–374. [Google Scholar] [CrossRef]
- Liss, C.; (Animal Welfare Institute, Washington, D.C., USA); Jones, D.; (Animal Welfare Institute, Washington, D.C., USA). Personal Communication to Janet Donlin and AVMA. Available online: https://awionline.org/sites/default/files/uploads/documents/Letter-from%20AWI-to-AVMA-re-VSD-Research-2017.pdf (accessed on 27 December 2022).
- North Carolina State University VSD Videos 5 (5 videos), VSD Videos 6 (7 videos), VSD Videos 8 (the following videos: VSD- Camera 2 VH 1-21-16 (1), VSD- Camera 2 VH 1-21-16 (2), VSD- Camera 2b VH 2-3-16 (1), VSD- Camera 2b VH 2-3-16 (2), VSD- Camera 2b VH 2-3-16 (3), VSD- Camera 2b VH 2-3-16 (4), VSD- Camera 2b VH 2-3-16 (5)) obtained via request by Animal Outlook under North Carolina Public Records Law, G.S. §132-1. Available online: https://drive.google.com/drive/folders/1Ocvpj6kcc1w-oHEw6yQUHs2DkK-WbzT7 (accessed on 27 December 2022).
- McKeegan, D. Mass depopulation. In Advances in Poultry Welfare; Elsevier: Duxford, UK, 2018; pp. 351–372. ISBN 978-0-08-100915-4. [Google Scholar] [CrossRef]
- Mitchell, M.A.; Kettlewell, P.J. Physiological stress and welfare of broiler chickens in transit: Solutions not problems! Poult Sci. 1998, 77, 1803–1814. [Google Scholar] [CrossRef]
- Arruda, A.G.; Beyene, T.J.; Kieffer, J.; Lorbach, J.N.; Moeller, S.; Bowman, A.S. A Systematic Literature Review on Depopulation Methods for Swine. Animals 2020, 10, 2161. [Google Scholar] [CrossRef]
- Unterer, S.; Busch, K. Acute Hemorrhagic Diarrhea Syndrome in Dogs. Vet. Clin. North Am. Small Anim. Pract. 2021, 51, 79–92. [Google Scholar] [CrossRef]
- Stanley, M.; Chippa, V.; Aeddula, N.R.; Quintanilla Rodriguez, B.S.; Adigun, R. Rhabdomyolysis. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Klingler, W.; Velders, M.; Hoppe, K.; Pedro, M.; Schleip, R. Clinical relevance of fascial tissue and dysfunctions. Curr. Pain Headache Rep. 2014, 18, 439. [Google Scholar] [CrossRef]
- Bittner, E.A.; Shank, E.; Woodson, L.; Martyn, J.A.J. Acute and Perioperative Care of the Burn-injured Patient. Anesthesiology 2015, 122, 448–464. [Google Scholar] [CrossRef]
- Hothersall, B.; Caplen, G.; Parker, R.M.A.; Nicol, C.J.; Waterman-Pearson, A.E.; Weeks, C.A.; Murrell, J.C. Thermal nociceptive threshold testing detects altered sensory processing in broiler chickens with spontaneous lameness. PLoS ONE 2014, 9, e97883. [Google Scholar] [CrossRef]
- Hothersall, B.; Caplen, G.; Nicol, C.J.; Taylor, P.M.; Waterman-Pearson, A.E.; Weeks, C.A.; Murrell, J.C. Development of mechanical and thermal nociceptive threshold testing devices in unrestrained birds (broiler chickens). J. Neurosci. Methods 2011, 201, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Crespo, R. Developmental, Metabolic, and Other Noninfectious Disorders. In Diseases of Poultry, 14th ed.; Swayne, D.E., Boulianne, M., Logue, C.M., McDougald, L.R., Nair, V., Suarez, D.L., Wit, S., Grimes, T., Johnson, D., Kromm, M., et al., Eds.; Wiley: Hoboken, NJ, USA, 2020; pp. 1286–1329. ISBN 978-1-119-37116-8. [Google Scholar]
- Polsky, L.; von Keyserlingk, M.A.G. Invited review: Effects of heat stress on dairy cattle welfare. J. Dairy Sci. 2017, 100, 8645–8657. [Google Scholar] [CrossRef]
- Caulfield, M.P.; Cambridge, H.; Foster, S.F.; McGreevy, P.D. Heat stress: A major contributor to poor animal welfare associated with long-haul live export voyages. Vet. J. 2014, 199, 223–228. [Google Scholar] [CrossRef]
- Anderson, C.A. Heat and Violence. Curr Dir Psychol Sci 2001, 10, 33–38. [Google Scholar] [CrossRef]
- Schrøder-Petersen, D.L.; Simonsen, H.B. Tail Biting in Pigs. Vet. J. 2001, 162, 196–210. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.J. Frustration of goal-directed behaviour in swine. Appl. Anim. Behav. Sci. 1999, 64, 19–29. [Google Scholar] [CrossRef]
- Beausoleil, N.; Mellor, D. Introducing breathlessness as a significant animal welfare issue. N. Z. Vet. J. 2015, 63, 44–51. [Google Scholar] [CrossRef]
- EFSA Panel on Animal Health and Welfare (AHAW); Saxmose Nielsen, S.; Alvarez, J.; Bicout, D.J.; Calistri, P.; Depner, K.; Drewe, J.A.; Garin-Bastuji, B.; Gonzales Rojas, J.L.; Gortázar Schmidt, C.; et al. Welfare of pigs during killing for purposes other than slaughter. EFSA J. 2020, 18, e06195. [Google Scholar] [CrossRef]
- Gruber, P. Egg Farmer Recalls Avian Flu Nightmare in Iowa. Lancaster Farming. Available online: https://www.lancasterfarming.com/news/main_edition/egg-farmer-recalls-avian-flu-nightmare-in-iowa/article_7a405e61-4dc7-569f-9ddc-d24a8a46904d.html (accessed on 27 October 2022).
- Zhao, Y.; Richardson, B.; Takle, E.; Chai, L.; Schmitt, D.; Xin, H. Airborne transmission may have played a role in the spread of 2015 highly pathogenic avian influenza outbreaks in the United States. Sci. Rep. 2019, 9, 11755. [Google Scholar] [CrossRef]
- Congressional Research Service Update on the Highly Pathogenic Avian Influenza Outbreak of 2014-2015; 2015. Available online: https://crsreports.congress.gov/product/pdf/R/R44114 (accessed on 20 September 2022).
- Vilsack, T.J.; (USDA, Washington, DC, USA). Personal Communication to Cathy Liss. Available online: https://awionline.org/sites/default/files/uploads/documents/Letter-from-USDA-Sec-to-AWI-Sep-2022.pdf (accessed on 27 December 2022).
- Wernery, U. Infectious Diseases. In Avian Medicine; Elsevier: St. Louis, MO, USA, 2016; pp. 434–521. ISBN 978-0-7234-3832-8. [Google Scholar] [CrossRef]
- Missouri Public Records related to HPAI 2022 obtained via Public Records Request by Animal Outlook. Available online: https://awionline.org/sites/default/files/uploads/documents/MO-Public-Records-re-HPAI-Depop-2022.pdf (accessed on 27 December 2022).
- National Pork Producers Council. The Tragic Impact of COVID-19 on U.S Hog Farmers: The Need to Euthanize. 2020. Available online: https://nppc.org/wp-content/uploads/2020/05/euthanasia-fact-sheet-FINAL-5-8-20.pdf?ftag=MSF0951a18 (accessed on 17 November 2021).
- Bargen, L.L.; Whiting, T.L. Time to critical overcrowding of Manitoba swine barns in the event of restriction on animal movement. Can. Vet. J. Rev. Vet. Can. 2002, 43, 855–862. [Google Scholar]
- Carriquiry, M.; Elobeid, A.; Swenson, D.; Hayes, D. Impacts of African Swine Fever in Iowa and the United States. Center for Agricultural and Rural Development Iowa State University. Available online: https://www.card.iastate.edu/products/publications/pdf/20wp600.pdf (accessed on 27 December 2022).
- Larkin, M. HOD Recommends Depopulation Policy Sent to Expert Panel for Further Consideration. American Veterinary Medical Association. Available online: https://www.avma.org/javma-news/2021-09-15/hod-recommends-depopulation-policy-sent-expert-panel-further-consideration (accessed on 28 November 2022).
- O’Keefe, T. How Ventilation Shutdown Works in Poultry Depopulation. WATT Poultry. Available online: https://www.wattagnet.com/articles/25922-how-ventilation-shutdown-works-in-poultry-depopulation (accessed on 20 September 2022).
- USDA Bird Depopulation Methods 2016–2021. Available online: https://awionline.org/sites/default/files/uploads/documents/Bird-Depopulations-Mar-2017-to-Mar-2021.pdf (accessed on 27 December 2022).
- Stokstad, E. Deadly bird flu establishes a foothold in North America. Science 2022, 377, 912. [Google Scholar] [CrossRef] [PubMed]
- National Association of State Departments of Agriculture NASDA Policy Materials - 2022 NASDA Annual Meeting. Available online: https://www.nasda.org/wp-content/uploads/2022/09/NASDA-Policy-Book-Annual-Meeting-2022.pdf (accessed on 18 December 2022).
- McLaughlin, J. Not Humane, Not AVMA Approved, and Definitely Not Euthanasia: Welfare and Efficacy Issues of Swine Depopulation by Ventilation Shutdown. Drake J. Agric. Law 2022, 27, 159. [Google Scholar]
- Indiana State Board of Animal Health POLICY NUMBER: ANHPD-05-01 Revision 1. Indiana General Assembly. Available online: http://iac.iga.in.gov/iac//20200506-IR-345200247NRA.xml.html (accessed on 27 December 2022).
- Rollin, B.E. Annual Meeting Keynote Address: Animal agriculture and emerging social ethics for animals. J. Anim. Sci. 2004, 82, 955–964. [Google Scholar] [CrossRef]
- Alonso, M.E.; González-Montaña, J.R.; Lomillos, J.M. Consumers’ Concerns and Perceptions of Farm Animal Welfare. Animals 2020, 10, 385. [Google Scholar] [CrossRef]
- Cornish, A.; Raubenheimer, D.; McGreevy, P. What We Know about the Public’s Level of Concern for Farm Animal Welfare in Food Production in Developed Countries. Animals 2016, 6, 74. [Google Scholar] [CrossRef]
- Sinclair, M.; Lee, N.Y.P.; Hötzel, M.J.; de Luna, M.C.T.; Sharma, A.; Idris, M.; Derkley, T.; Li, C.; Islam, M.A.; Iyasere, O.S.; et al. International perceptions of animals and the importance of their welfare. Front. Anim. Sci. 2022, 3, 960379. [Google Scholar] [CrossRef]
- Hernandez, E.; Llonch, P.; Turner, P.V. Applied Animal Ethics in Industrial Food Animal Production: Exploring the Role of the Veterinarian. Animals 2022, 12, 678. [Google Scholar] [CrossRef]
- Kipperman, B.S. The role of the veterinary profession in promoting animal welfare. J. Am. Vet. Med. Assoc. 2015, 246, 502–504. [Google Scholar] [CrossRef]
- Mirabella, L. Nearly 2 Million Chickens at Eastern Shore Farms Set to be Destroyed Because of Coronavirus-Related Plant Shortages. Baltimore Sun. Available online: https://www.baltimoresun.com/coronavirus/bs-md-poultry-producers-coronavirus-20200424-hufdfzs24zhqpeptx4w6yngztm-story.html (accessed on 14 December 2022).
- Veterinarian’s Oath. American Veterinary Medical Association. Available online: https://www.avma.org/resources-tools/avma-policies/veterinarians-oath (accessed on 21 September 2022).
- AVMA; Canadian Veterinary Medical Association. Federation of Veterinarians of Europe Joint Statement on the Roles of Veterinarians in Ensuring Good Animal Welfare. Available online: https://www.avma.org/resources-tools/avma-policies/joint-avma-fve-cvma-roles-veterinarians-promoting-animal-welfare (accessed on 20 October 2022).
- AVMA. AVMA Animal Welfare Principles. American Veterinary Medical Association. Available online: https://www.avma.org/resources-tools/avma-policies/avma-animal-welfare-principles (accessed on 10 September 2022).
- Moses, L.; Malowney, M.J.; Wesley Boyd, J. Ethical conflict and moral distress in veterinary practice: A survey of North American veterinarians. J. Vet. Intern. Med. 2018, 32, 2115–2122. [Google Scholar] [CrossRef]
- Dürnberger, C. Am I actually a veterinarian or an economist? Understanding the moral challenges for farm veterinarians in Germany on the basis of a qualitative online survey. Res. Vet. Sci. 2020, 133, 246–250. [Google Scholar] [CrossRef] [PubMed]
- AVMA Principles of Veterinary Medical Ethics of the AVMA. American Veterinary Medical Association. Available online: https://www.avma.org/resources-tools/avma-policies/principles-veterinary-medical-ethics-avma (accessed on 14 September 2022).
- Whiting, T.L.; Marion, C.R. Perpetration-induced traumatic stress—A risk for veterinarians involved in the destruction of healthy animals. Can. Vet. J. Rev. Vet. Can. 2011, 52, 794–796. [Google Scholar]
- AVMA. AVMA Guidelines for the Euthanasia of Animals: 2020 Edition. Available online: https://www.avma.org/sites/default/files/2020-02/Guidelines-on-Euthanasia-2020.pdf (accessed on 27 December 2022).
- AVMA. AVMA Guidelines for the Humane Slaughter of Animals: 2016 Edition. Available online: https://www.avma.org/sites/default/files/resources/Humane-Slaughter-Guidelines.pdf (accessed on 27 December 2022).
- APHIS Emergency Response for HPAI Outbreaks in Seven States, September 2022: Final Environmental Assessment 2022. Available online: https://downloads.regulations.gov/APHIS-2022-0031-0012/content.pdf (accessed on 7 October 2022).
- Stepien, M.; (APHIS, Riverdale, MD, USA). Personal communication to Marina Bolotnikova, 4 April 2022.
- Environmental Protection Agency Regulatory Definitions of Large CAFOs, Medium CAFO, and Small CAFOs. Available online: https://www3.epa.gov/npdes/pubs/sector_table.pdf (accessed on 27 December 2022).
- USDA. APHIS HPAI Response: Response Goals & Depopulation Policy 2022. Available online: https://www.aphis.usda.gov/animal_health/emergency_management/downloads/hpai/depopulationpolicy.pdf (accessed on 27 December 2022).
- AVMA Pregnant Sow Housing. AVMA. Available online: https://www.avma.org/resources-tools/avma-policies/pregnant-sow-housing (accessed on 27 December 2022).
- AVMA Layer Hen Housing Systems. AVMA. Available online: https://www.avma.org/resources-tools/avma-policies/layer-hen-housing-systems (accessed on 27 December 2022).
- AVMA Veal calf management. AVMA. Available online: https://www.avma.org/resources-tools/avma-policies/veal-calf-management (accessed on 27 December 2022).
- Booker, C.A. Text S.2332 117th Congress (2021–2022): Farm System Reform Act of 2021. Available online: https://www.congress.gov/bill/117th-congress/senate-bill/2332/text (accessed on 23 September 2022).
- AVMA. Emergency Preparedness and Response. 2012
- USDA Highly Pathogenic Avian Influenza H5(EA)—Status of Vaccines for Avian Species’ Immunization in the United States 2015. Available online: https://www.aphis.usda.gov/animal_health/downloads/animal_diseases/ai/hpai-vaccination-technical-doc.pdf (accessed on 27 December 2022).
- USDA Policy and Approach to HPAI Vaccination 2016. Available online: https://www.aphis.usda.gov/animal_health/animal_dis_spec/poultry/downloads/hpai_policy.pdf (accessed on 27 December 2022).
- Polansek, T.U.S. Considers Vaccines to Protect Poultry from Deadly Bird Flu. Reuters. Available online: https://www.reuters.com/world/us/us-considers-vaccines-protect-poultry-deadly-bird-flu-2022-04-04/ (accessed on 15 November 2022).
- Loeb, J. Calls grow for global avian flu jabs. Vet. Rec. 2022, 191, 360–361. [Google Scholar] [CrossRef] [PubMed]
- Torrella, K. America’s Catastrophic, Record-Breaking Bird Flu Outbreak, Explained. Vox. Available online: https://www.vox.com/future-perfect/2022/11/22/23472207/bird-flu-vaccine-turkey-prices-chickens-hens-cull-depopulation (accessed on 1 December 2022).
- Brockotter, F. Unspeakable Damage and Unexplainable Consequences. 9 November 2022, pp. 6–9. Available online: https://kiosk.futurefarming.com/#/main/presenter/504ba7c0-cd5f-469f-a3d8-534f9e056eb3 (accessed on 27 December 2022).
- APHIS HPAI Response: Timeline, Eligibility, and Approval for Restocking 2022. Available online: https://www.aphis.usda.gov/animal_health/emergency_management/downloads/hpai/criteriarestock.pdf (accessed on 10 October 2022).
- United Kingdom. RFI7527_All_Projects_List_amended. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/437140/RFI7527_All_Projects_List_amended.pdf (accessed on 27 December 2022).
- Department for Environment Food & Rural Affairs Contingency Plan for Exotic Notifiable Diseases of Animals in England. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1095427/Contingency_Plan_for_Exotic_Notifiable_Diseases_of_Animals_in_England.pdf (accessed on 28 November 2022).
- Department for Environmental, Food, and Rural Affairs Guidelines for Killing Poultry Using Ventilation Shutdown (VSD) 2009. Available online: https://www.slideshare.net/charmkey5/operating-guidance-vetilation-shutdown-procedure-defra (accessed on 27 December 2022).
- Farm Animal Welfare Committee Opinion on Contingency Planning for Farm Animal Welfare in Disasters and Emergencies. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/324804/FAWC_opinion_on_contingency_planning_for_farm_animal_welfare_in_disasters_and_emergencies.pdf (accessed on 27 December 2022).
- Farm Animal Welfare Council Report on the Welfare of Farmed Animals at Slaughter or Killing Part 2: White Meat Animals. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/325045/FAWC_report_on_the_welfare_of_farmed_animals_at_slaughter_or_killing_part_two_white_meat_species.pdf (accessed on 27 December 2022).
- Booker, C.A. Industrial Agriculture Accountability Act of 2022. Available online: https://www.booker.senate.gov/imo/media/doc/industrial_agriculture_accountability_act.pdf (accessed on 27 December 2022).
- H.R. 9552: To amend the Animal Health Protection Act to Increase Transparency with Respect to Livestock and Poultry Depopulation, and for Other Purposes. (H.R. 9552). GovTrack.us. Available online: https://www.govtrack.us/congress/bills/117/hr9552 (accessed on 15 December 2022).
- Kiezebrink, H. Depopulation with Proven Slaughter Technology. 2022
- Leonidou, E.; Christofi, M.; Vrontis, D.; Thrassou, A. An integrative framework of stakeholder engagement for innovation management and entrepreneurship development. J. Bus. Res. 2020, 119, 245–258. [Google Scholar] [CrossRef]
- Council Regulation (EC) No 1099/2009 of 24 September 2009 on the Protection of Animals at the Time of Killing. EUR-Lex. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32009R1099 (accessed on 27 December 2022).
- The Welfare of Animals at the Time of Killing (England) Regulations 2015. legislation.gov.uk. Available online: https://www.legislation.gov.uk/uksi/2015/1782/contents/made (accessed on 27 December 2022).
- The Welfare of Animals at the Time of Killing Regulations (Northern Ireland) 2014. legislation.gov.uk. Available online: https://www.legislation.gov.uk/nisr/2014/107/contents/made (accessed on 27 December 2022).
- The Welfare of Animals at the Time of Killing (Scotland) Regulations 2012. legislation.gov.uk. Available online: https://www.legislation.gov.uk/ssi/2012/321/contents/made (accessed on 27 December 2022).
- The Welfare of Animals at the Time of Killing (Wales) Regulations 2014. legislation.gov.uk. Available online: https://www.legislation.gov.uk/wsi/2014/951/regulation/3/made (accessed on 27 December 2022).
- McReynolds, S.W.; Sanderson, M.W. Feasibility of depopulation of a large feedlot during a foot-and-mouth disease outbreak. J. Am. Vet. Med. Assoc. 2014, 244, 291–298. [Google Scholar] [CrossRef]
- DeNicola, A.J.; Miller, D.S.; DeNicola, V.L.; Meyer, R.E.; Gambino, J.M. Assessment of humaneness using gunshot targeting the brain and cervical spine for cervid depopulation under field conditions. PLoS ONE 2019, 14, e0213200. [Google Scholar] [CrossRef]
- Derscheid, R.J.; Dewell, R.D.; Dewell, G.A.; Kleinhenz, K.E.; Shearer, L.C.; Gilliam, J.N.; Reynolds, J.P.; Sun, Y.; Shearer, J.K. Validation of a portable pneumatic captive bolt device as a one-step method of euthanasia for use in depopulation of feedlot cattle. J. Am. Vet. Med. Assoc. 2016, 248, 96–104. [Google Scholar] [CrossRef]
- Casey-Trott, T.M.; Millman, S.T.; Turner, P.V.; Nykamp, S.G.; Lawlis, P.C.; Widowski, T.M. Effectiveness of a nonpenetrating captive bolt for euthanasia of 3 kg to 9 kg pigs1. J. Anim. Sci. 2014, 92, 5166–5174. [Google Scholar] [CrossRef]
- Erasmus, M.A.; Lawlis, P.; Duncan, I.J.H.; Widowski, T.M. Using time to insensibility and estimated time of death to evaluate a nonpenetrating captive bolt, cervical dislocation, and blunt trauma for on-farm killing of turkeys. Poult. Sci. 2010, 89, 1345–1354. [Google Scholar] [CrossRef]
- Anderson, K.N.; Deen, J.; Karczewski, J.; Zhitnitskiy, P.E.; Vogel, K.D. History and best practices of captive bolt euthanasia for swine. Transl. Anim. Sci. 2022, 6, txac065. [Google Scholar] [CrossRef]
- Gerritzen, M.A.; Raj, M.A.B. Animal welfare and killing for disease control. In Welfare of Production Animals: Assessment and Management of Risks; Wageningen Acad Publ: Wageningen, The Netherlands, 2009; Volume 5, pp. 191–205. [Google Scholar]
- Vogel, K.D.; Badtram, G.; Claus, J.R.; Grandin, T.; Turpin, S.; Weyker, R.E.; Voogd, E. Head-only followed by cardiac arrest electrical stunning is an effective alternative to head-only electrical stunning in pigs1. J. Anim. Sci. 2011, 89, 1412–1418. [Google Scholar] [CrossRef] [PubMed]
- Dalla Costa, F.A.; Gibson, T.J.; Oliveira, S.E.O.; Gregory, N.G.; Coldebella, A.; Faucitano, L.; Dalla Costa, O.A. On-farm pig dispatch methods and stockpeople attitudes on their use. Livest. Sci. 2019, 221, 1–5. [Google Scholar] [CrossRef]
- Jacobs, L.; Bourassa, D.V.; Boyal, R.S.; Harris, C.E.; Bartenfeld Josselson, L.N.; Campbell, A.; Anderson, G.; Buhr, R.J. Animal welfare assessment of on-farm euthanasia methods for individual, heavy turkeys. Poult. Sci. 2021, 100, 100812. [Google Scholar] [CrossRef] [PubMed]
- Boyal, R.S.; Buhr, R.J.; Harris, C.E.; Jacobs, L.; Bourassa, D.V. Evaluation of mechanical cervical dislocation, captive bolt, carbon dioxide, and electrical methods for individual on-farm euthanasia of broiler breeders. Poult. Sci. 2022, 101, 102000. [Google Scholar] [CrossRef] [PubMed]
- Blackmore, D.K.; Newhook, J.C. Electroencephalographic studies of stunning and slaughter of sheep and calves— Part 3: The duration of insensibility induced by electrical stunning in sheep and calves. Meat Sci. 1982, 7, 19–28. [Google Scholar] [CrossRef]
- Meyer, R. Physiologic Measures of Animal Stress during Transitional States of Consciousness. Animals 2015, 5, 702–716. [Google Scholar] [CrossRef]
- Stegeman, A.; Elbers, A.; de Smit, H.; Moser, H.; Smak, J.; Pluimers, F. The 1997–1998 epidemic of classical swine fever in the Netherlands. Vet. Microbiol. 2000, 73, 183–196. [Google Scholar] [CrossRef]
- Pluimers, F.H.; de Leeuw, P.W.; Smak, J.A.; Elbers, A.R.; Stegeman, J.A. Classical swine fever in The Netherlands 1997-1998: A description of organisation and measures to eradicate the disease. Prev. Vet. Med. 1999, 42, 139–155. [Google Scholar] [CrossRef]
- Gomez, D. International Panel. In Proceedings of the Seventh International Symposium on Animal Mortality Management, Raleigh, NC, USA, 27–30 June 2022. [Google Scholar]
- Thornber, P.M.; Rubira, R.J.; Styles, D.K. Humane killing of animals for disease control purposes. Rev. Sci. Tech. 2014, 33, 303–310. [Google Scholar] [CrossRef]
- Mote, B.; Woiwode, R. Validation of a Mobile Electrocution System for Humane Mass Depopulation of Swine – NPB #20-123. Pork Checkoff Research. Available online: https://porkcheckoff.org/research/validation-mobile-electrocution-system-humane-mass-depopulation-swine/ (accessed on 27 December 2022).
- Kramer, S.A.; Wagner, B.K.; Moeller, S.J.; Bowman, A.S.; Kieffer, J.D.; Gonçalves Arruda, A.; Cressman, M.D.; Pairis-Garcia, M.D. Technical Note: Validation of the effectiveness of electric stunning for euthanasia of mature swine (Sus scrofa domesticus). J. Anim. Sci. 2022, 100, skac072. [Google Scholar] [CrossRef]
- Johnson, C. Application of Electrocution in Suckling Pigs for Mass Depopulation - NPB #20-117. Pork Checkoff Research. Available online: https://live.porkcheckoff.org/research/application-electrocution-suckling-pigs-mass-depopulation/ (accessed on 27 December 2022).
- Husheer, J.; Luepke, M.; Dziallas, P.; Waldmann, K.-H.; von Altrock, A. Electrocution as an alternative euthanasia method to blunt force trauma to the head followed by exsanguination for non-viable piglets. Acta Vet. Scand. 2020, 62, 67. [Google Scholar] [CrossRef]
- Gerritzen, M.; Lambooij, B.; Reimert, H.; Stegeman, A.; Spruijt, B. A note on behaviour of poultry exposed to increasing carbon dioxide concentrations. Appl. Anim. Behav. Sci. 2007, 108, 179–185. [Google Scholar] [CrossRef][Green Version]
- Gerritzen, M.A.; Lambooij, E.; Stegeman, J.A.; Spruijt, B.M. Slaughter of poultry during the epidemic of avian influenza in the Netherlands in 2003. Vet. Rec. 2006, 159, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Krushinskie, E.A.; Smeltzer, M.; Klein, P.; Kiezenbrink, H. Mass Depopulation as an Effective Measure for Disease Control Purposes. In Avian Influenza; Swayne, D.E., Ed.; Blackwell Publishing Ltd.: Oxford, UK, 2008; pp. 309–332. ISBN 978-0-8138-1863-4. [Google Scholar] [CrossRef]
- Stikeleather, L.; Morrow, W.; Meyer, R.; Baird, C.; Halbert, B. Evaluation of CO2 Application Requirements for On-Farm Mass Depopulation of Swine in a Disease Emergency. Agriculture 2013, 3, 599–612. [Google Scholar] [CrossRef]
- Meyer, R.E.; Morrow, W.E.M.; Stikeleather, L.F.; Baird, C.L.; Rice, J.M.; Byrne, H.; Halbert, B.V.; Styles, D.K. Evaluation of carbon dioxide administration for on-site mass depopulation of swine in response to animal health emergencies. J. Am. Vet. Med. Assoc. 2014, 244, 924–933. [Google Scholar] [CrossRef]
- Gent, T.C.; Gebhardt-Henrich, S.; Schild, S.-L.A.; Rahman, A.A.; Toscano, M.J. Evaluation of Poultry Stunning with Low Atmospheric Pressure, Carbon Dioxide or Nitrogen Using a Single Aversion Testing Paradigm. Animals 2020, 10, 1308. [Google Scholar] [CrossRef] [PubMed]
- Sandilands, V.; Raj, A.B.M.; Baker, L.; Sparks, N.H.C. Aversion of chickens to various lethal gas mixtures. Anim. Welf. 2011, 20, 253–262. [Google Scholar]
- Steiner, A.R.; Axiak Flammer, S.; Beausoleil, N.J.; Berg, C.; Bettschart-Wolfensberger, R.; García Pinillos, R.; Golledge, H.D.R.; Marahrens, M.; Meyer, R.; Schnitzer, T.; et al. Humanely Ending the Life of Animals: Research Priorities to Identify Alternatives to Carbon Dioxide. Animals 2019, 9, 911. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.I.; Torrey, S.; Widowski, T.M.; Turner, P.V.; Knezacek, T.D.; Nicholds, J.; Crowe, T.G.; Schwean-Lardner, K. Defining characteristics of immersion carbon dioxide gas for successful euthanasia of neonatal and young broilers. Poult. Sci. 2020, 99, 4408–4416. [Google Scholar] [CrossRef]
- Gerritzen, M.A.; Lambooij, E.; Hillebrand, S.J.; Lankhaar, J.A.; Pieterse, C. Behavioral Responses of Broilers to Different Gaseous Atmospheres. Poult. Sci. 2000, 79, 928–933. [Google Scholar] [CrossRef]
- Gerritzen, M.A.; Lambooij, E.; Reimert, H.G.; Spruijt, B.M.; Stegeman, J.A. Susceptibility of Duck and Turkey to Severe Hypercapnic Hypoxia. Poult. Sci. 2006, 85, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Raj, A.B.M.; Sandilands, V.; Sparks, N.H.C. Review of gaseous methods of killing poultry on-farm for disease control purposes. Vet. Rec. 2006, 159, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Rault, J.-L.; McMunn, K.A.; Marchant-Forde, J.N.; Lay, D.C. Gas alternatives to carbon dioxide for euthanasia: A piglet perspective. J. Anim. Sci. 2013, 91, 1874–1883. [Google Scholar] [CrossRef][Green Version]
- Dalmau, A.; Rodríguez, P.; Llonch, P.; Velarde, A. Stunning pigs with different gas mixtures: Aversion in pigs. Anim. Welf. 2010, 19, 325–333. [Google Scholar]
- Kells, N.; Beausoleil, N.; Johnson, C.; Sutherland, M. Evaluation of Different Gases and Gas Combinations for On-Farm Euthanasia of Pre-Weaned Pigs. Animals 2018, 8, 40. [Google Scholar] [CrossRef] [PubMed]
- Llonch, P.; Dalmau, A.; Rodríguez, P.; Manteca, X.; Velarde, A. Aversion to nitrogen and carbon dioxide mixtures for stunning pigs. Anim. Welf. 2012, 21, 33–39. [Google Scholar] [CrossRef]
- Raj, M.; O’Callaghan, M.; Thompson, K.; Beckett, D.; Morrish, I.; Love, A.; Hickman, G.; Howson, S. Large scale killing of poultry species on farm during outbreaks of diseases: Evaluation and development of a humane containerised gas killing system. World’s Poult. Sci. J. 2008, 64, 227–244. [Google Scholar] [CrossRef]
- Bergen, G.; (Prairie Agricultural Machinery Institute, Humboldt, Saskatchewan, Canada). Personal Communication to Gwendolen Reyes-Illg, 29 September 2022.
- McKeegan, D.E.F.; Sparks, N.H.C.; Sandilands, V.; Demmers, T.G.M.; Boulcott, P.; Wathes, C.M. Physiological responses of laying hens during whole-house killing with carbon dioxide. Br. Poult. Sci. 2011, 52, 645–657. [Google Scholar] [CrossRef]
- Turner, P.V.; Kloeze, H.; Dam, A.; Ward, D.; Leung, N.; Brown, E.E.L.; Whiteman, A.; Chiappetta, M.E.; Hunter, D.B. Mass depopulation of laying hens in whole barns with liquid carbon dioxide: Evaluation of welfare impact. Poult. Sci. 2012, 91, 1558–1568. [Google Scholar] [CrossRef]
- Beutelschies, S. NVS Development of C02 Whole House Gassing for Emergency Depopulation of Poultry. Available online: https://awionline.org/sites/default/files/uploads/documents/NVS-CO2-Whole-House-Gassing-for-Bird-Depop-2016.pdf. (accessed on 27 December 2022).
- Lambooij, E.; Gerritzen, M.A.; Engel, B.; Hillebrand, S.J.W.; Lankhaar, J.; Pieterse, C. Behavioural responses during exposure of broiler chickens to different gas mixtures. Appl. Anim. Behav. Sci. 1999, 62, 255–265. [Google Scholar] [CrossRef]
- McKeegan, D.E.F.; McIntyre, J.; Demmers, T.G.M.; Wathes, C.M.; Jones, R.B. Behavioural responses of broiler chickens during acute exposure to gaseous stimulation. Appl. Anim. Behav. Sci. 2006, 99, 271–286. [Google Scholar] [CrossRef]
- Sifford, R.B.; (APHIS, Riverdale, MD, USA). Personal Communication to Gwendolen Reyes-Illg. Available online: https://awionline.org/sites/default/files/uploads/documents/Letter-from-APHIS-to-AWI-Aug-2022.pdf (accessed on 27 December 2022).
- Lindahl, C.; Sindhøj, E.; Brattlund Hellgren, R.; Berg, C.; Wallenbeck, A. Responses of Pigs to Stunning with Nitrogen Filled High-Expansion Foam. Animals 2020, 10, 2210. [Google Scholar] [CrossRef] [PubMed]
- McKeegan, D.E.F.; Reimert, H.G.M.; Hindle, V.A.; Boulcott, P.; Sparrey, J.M.; Wathes, C.M.; Demmers, T.G.M.; Gerritzen, M.A. Physiological and behavioral responses of poultry exposed to gas-filled high expansion foam. Poult. Sci. 2013, 92, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Gerritzen, M.A.; Sparrey, J. A pilot study to assess whether high expansion CO2-enriched foam is acceptable for on-farm emergency killing of poultry. Animal Welfare 2008, 17, 285–288. [Google Scholar]
- McKeegan, D.; Gerritzen, M.; Sparrey, J. High expansion gas filled foam-a humane agent for emergency killing. Br. Poult. Abstr. 2012, 8, 2–4. [Google Scholar]
- Wallenbeck, A.; Sindhoj, E.; Berg, C.; Lindahl, C. Improved pig welfare at slaughter - pigs’ responses to air- or nitrogen foam. In Proceedings of the 31th Nordic Region Winter Meeting of the International Society for Applied Ethology, Tartu, Estonia, 28–30 January 2020; Soonberg, M., Ed.; Estonian University of Life Sciences: Tartu, Estonia, 2020; p. 21. [Google Scholar]
- Raj, A.B.M.; Smith, C.; Hickman, G. Novel method for killing poultry in houses with dry foam created using nitrogen. Vet. Rec. 2008, 162, 722–723. [Google Scholar] [CrossRef]
- Gerritzen, M.; Reimert, H.; Hindle, V.; McKeegan, D.; Sparrey, J. Welfare Assessment of Gas Filled Foam as an Agent for Killing Poultry, Report 399; Livestock Research Wageningen UR, 2010. Available online: https://www.researchgate.net/publication/275647993_welfare_assessment_of_gas_filled_foam_as_an_agent_for_killing_poultry. (accessed on 27 December 2022).
- Miller, L.; Williams, T.; Pepin, B.; Odland, C. Poster presentation: Nitrogen gas delivered by highly expanding foam for the depopulation of swine. In Proceedings of the 53rd Annual Meeting of the American Association of Swine Veterinarians, Indianapolis, IN, USA, 26 February–1 March 2022. [Google Scholar] [CrossRef]
- Williams, T. Validation and Demonstration of Utilizing High Expansion Nitrogen Foam for Large Scale Depopulation of Swine - NPB #21-069. Pork Checkoff Research. Available online: https://www.porkcheckoff.org/wp-content/uploads/2022/06/21-069-WILLIAMS-final-rpt.pdf (accessed on 27 December 2022).
- Hunt, L. Using Nitrogen Foam for Swine Depopulation. In Proceedings of the Seventh International Symposium on Animal Mortality Management, Raleigh, NC, USA, 27–30 June 2022. [Google Scholar]
- Lorbach, J.N.; Campler, M.R.; Youngblood, B.; Farnell, M.B.; Beyene, T.J.; Kieffer, J.; Moeller, S.J.; Arruda, A.G.; Bowman, A.S. Comparison of Gaseous and Water-Based Medium-Expansion Foam Depopulation Methods in Cull Sows. Animals 2021, 11, 3179. [Google Scholar] [CrossRef]
- Korenyi-Both, J.; Vidaurre, J.; Held, T.; Campler, M.R.; Kieffer, J.; Cheng, T.-Y.; Moeller, S.J.; Bowman, A.S.; Arruda, A.G. Description of electroencephalographic data gathered using water-based medium-expansion foam as a depopulation method for nursery pigs. Sci. Rep. 2022, 12, 16798. [Google Scholar] [CrossRef]
- Arruda, A.G.; Campler, M.R.; Cheng, T.; Youngblood, B.; Capria, V.; Kieffer, J.; Moeller, S.; Bowman, A.S. Reliability of water-based medium-expansion foam as a depopulation method for nursery pigs and cull sows. Transbounding Emerg. Dis. 2022, 69, e2719–e2730. [Google Scholar] [CrossRef]
- Gurung, S.; White, D.; Archer, G.; Styles, D.; Zhao, D.; Farnell, Y.; Byrd, J.; Farnell, M. Carbon Dioxide and Nitrogen Infused Compressed Air Foam for Depopulation of Caged Laying Hens. Animals 2018, 8, 6. [Google Scholar] [CrossRef]
- Benson, E.R.; Alphin, R.L.; Rankin, M.K.; Caputo, M.P.; Hougentogler, D.P.; Johnson, A.L. Mass emergency water-based foam depopulation of poultry. Avian. Dis. 2012, 56, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Dawson, M.D.; Lombardi, M.E.; Benson, E.R.; Alphin, R.L.; Malone, G.W. Using Accelerometers to Determine the Cessation of Activity of Broilers. J. Appl. Poult. Res. 2007, 16, 583–591. [Google Scholar] [CrossRef]
- Benson, E.; Malone, G.W.; Alphin, R.L.; Dawson, M.D.; Pope, C.R.; Van Wicklen, G.L. Foam-based mass emergency depopulation of floor-reared meat-type poultry operations. Poult. Sci. 2007, 86, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Caputo, M.P.; Alphin, R.L.; Pritchett, E.; Hougentogler, D.P.; Johnson, A.L.; Benson, E.R.; Patil, C. Evaluation of the diving reflex in response to nonterminal submersion of White Pekin ducks in water-based foam. Poult. Sci. 2013, 92, 412–417. [Google Scholar] [CrossRef]
- Rankin, M.K.; Alphin, R.L.; Benson, E.R.; Johnson, A.L.; Hougentogler, D.P.; Mohankumar, P. Comparison of water-based foam and carbon dioxide gas emergency depopulation methods of turkeys. Poult. Sci. 2013, 92, 3144–3148. [Google Scholar] [CrossRef]
- Benson, E.R.; Alphin, R.L.; Dawson, M.D.; Malone, G.W. Use of water-based foam to depopulate ducks and other species. Poult. Sci. 2009, 88, 904–910. [Google Scholar] [CrossRef]
- Lower, A. Evaluation of Sodium Nitrite for Mass Euthanasia of Commercial Pigs—NPB #20-118. Pork Checkoff Research. Available online: https://porkcheckoff.org/wp-content/uploads/2021/02/20-118-LOWER-final-rpt.pdf (accessed on 27 December 2022).
- Institute of Medical and Veterinary Science Assessing the Humaneness and Efficacy of a New Feral Pig Bait in Domestic Pigs, Report for the Australian Government Department of the Environment, Water, Heritage and the Arts; Canberra, Australia, 2010. Available online: https://www.dcceew.gov.au/sites/default/files/env/resources/091b0583-f35c-40b3-a530-f2e0c307a20c/files/pigs-imvs-report.pdf (accessed on 27 December 2022).
- Pepin, B. Determine effective oral dosing of sodium nitrite for efficient euthanasia of adult swine using oral drench technique NPB #20-122. Pork Checkoff Research. Available online: https://porkcheckoff.org/wp-content/uploads/2021/02/20-122-PEPIN-final-rpt.pdf (accessed on 27 December 2022).
- Cowled, B.D.; Elsworth, P.; Lapidge, S.J. Additional toxins for feral pig (Sus scrofa) control: Identifying and testing Achilles’ heels. Wildl. Res. 2008, 35, 651. [Google Scholar] [CrossRef]
- Richards, K. International Panel. In Proceedings of the Seventh International Symposium on Animal Mortality Management, Raleigh, NC, USA, 27–30 June 2022. [Google Scholar]
- Control Method: Baiting of Feral Pigs with Sodium Nitrite. Available online: https://pestsmart.org.au/wp-content/uploads/sites/3/2020/07/pig_baiting_sodium_nitrite.pdf (accessed on 11 October 2012).
- Direct Action Everywhere Uncut, Unedited 2.5 Hour Audio of Pigs Being Roasted Alive Via “Ventilation Shutdown.”—YouTube. Available online: https://www.youtube.com/watch?v=ybBTWIUeC6c&t=881s (accessed on 19 September 2022).
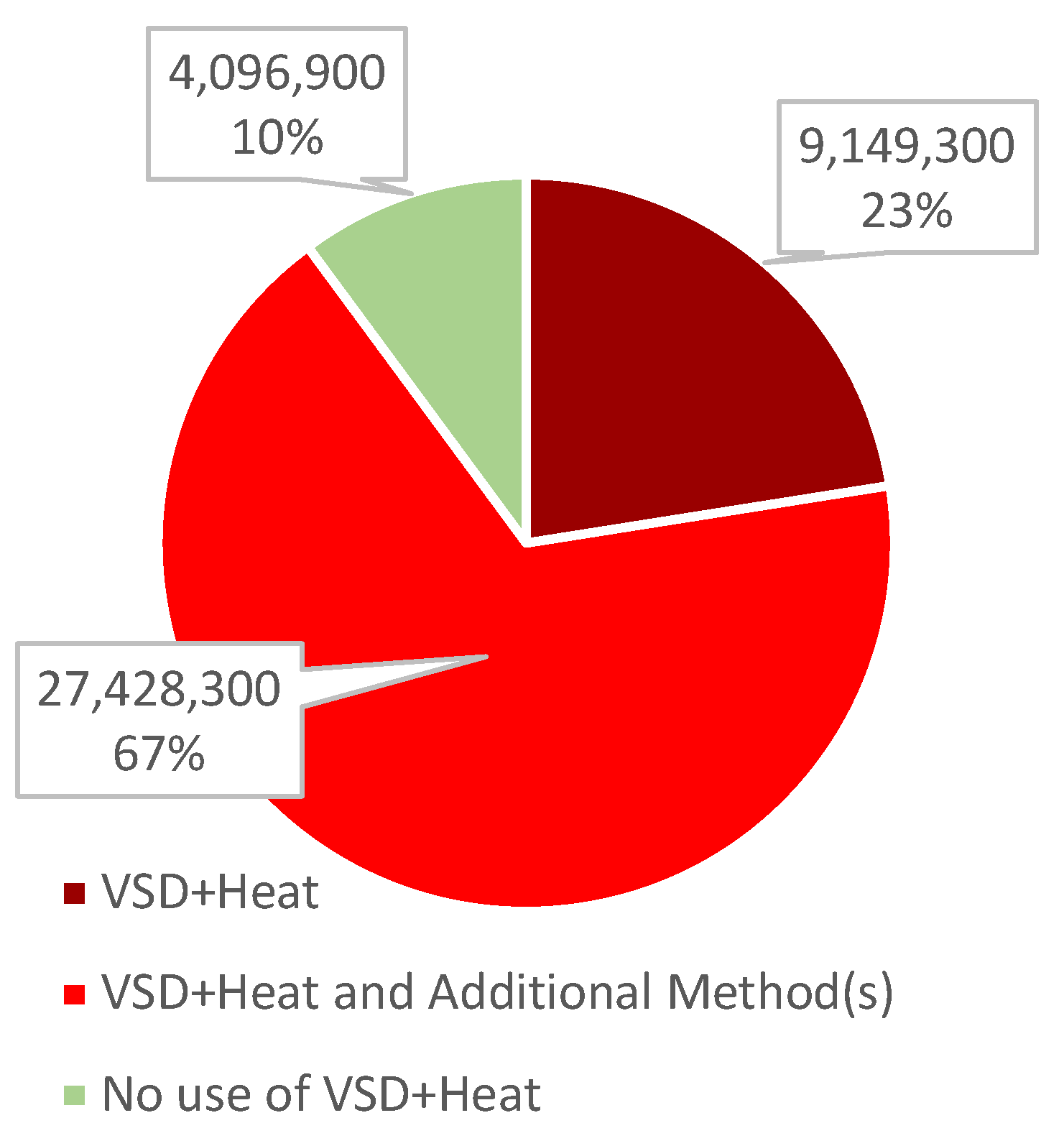
| Location of Farm | Number of Birds on Farm | Time Needed to Depopulate * | Method(s) Used |
|---|---|---|---|
| Iowa | 5,347,500 | 7 days | VSD + Heat CO2 gassing |
| Iowa | 5,011,700 | 16 days | VSD + Heat CO2 gassing |
| Wisconsin | 2,750,700 | 16 days | VSD + Heat |
| Nebraska | 2,118,000 | 18 days | VSD + Heat CO2 gassing |
| Colorado | 1,936,800 | 17 days | VSD + Heat CO2 gassing |
| Nebraska | 1,746,900 | 10 days | VSD + Heat CO2 gassing |
| Utah | 1,501,200 | 18 days | VSD + Heat CO2 gassing |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyes-Illg, G.; Martin, J.E.; Mani, I.; Reynolds, J.; Kipperman, B. The Rise of Heatstroke as a Method of Depopulating Pigs and Poultry: Implications for the US Veterinary Profession. Animals 2023, 13, 140. https://doi.org/10.3390/ani13010140
Reyes-Illg G, Martin JE, Mani I, Reynolds J, Kipperman B. The Rise of Heatstroke as a Method of Depopulating Pigs and Poultry: Implications for the US Veterinary Profession. Animals. 2023; 13(1):140. https://doi.org/10.3390/ani13010140
Chicago/Turabian StyleReyes-Illg, Gwendolen, Jessica E. Martin, Indu Mani, James Reynolds, and Barry Kipperman. 2023. "The Rise of Heatstroke as a Method of Depopulating Pigs and Poultry: Implications for the US Veterinary Profession" Animals 13, no. 1: 140. https://doi.org/10.3390/ani13010140
APA StyleReyes-Illg, G., Martin, J. E., Mani, I., Reynolds, J., & Kipperman, B. (2023). The Rise of Heatstroke as a Method of Depopulating Pigs and Poultry: Implications for the US Veterinary Profession. Animals, 13(1), 140. https://doi.org/10.3390/ani13010140





