Wind Farms and Power Lines Have Negative Effects on Territory Occupancy in Eurasian Eagle Owls (Bubo bubo)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
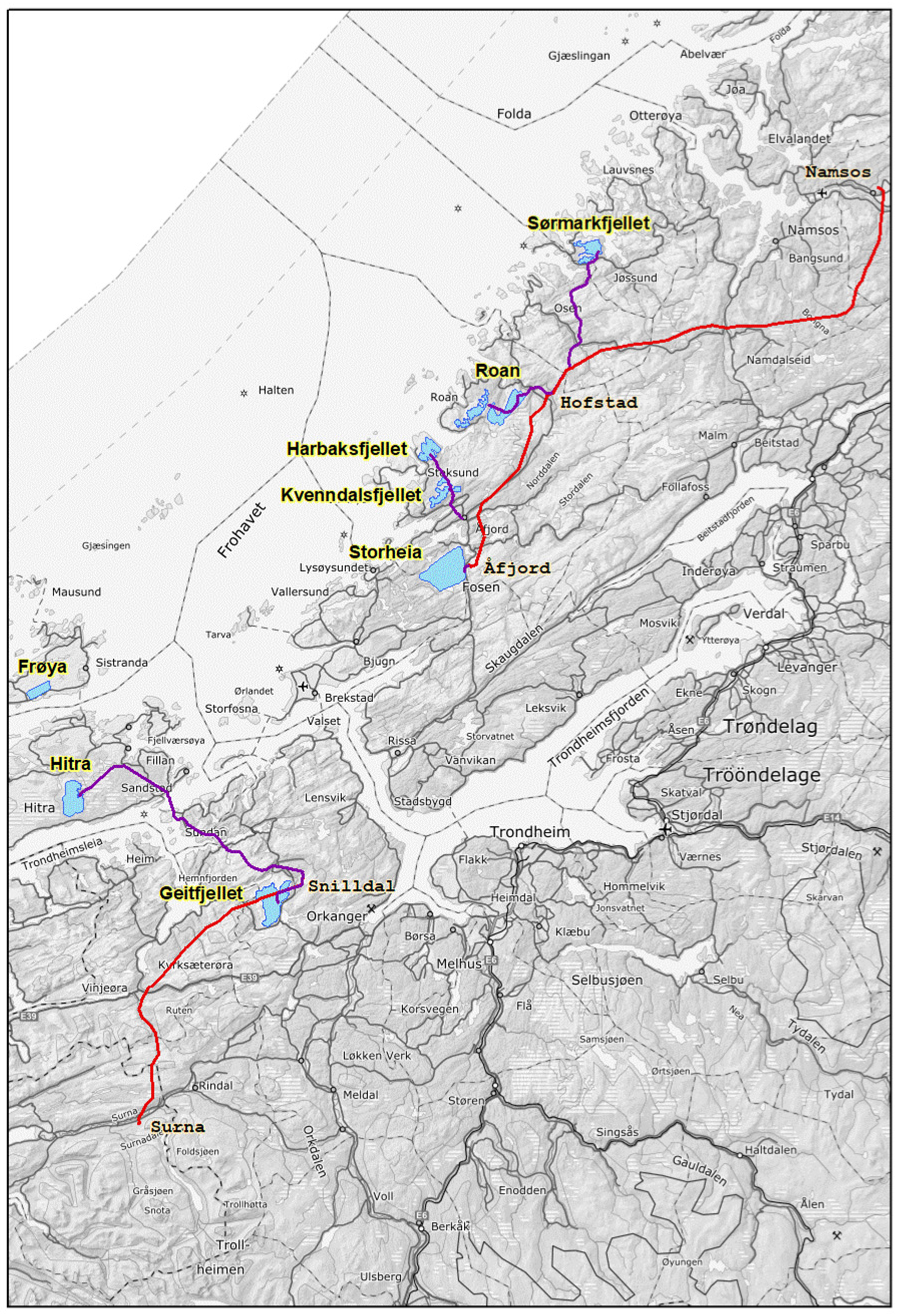
2.1. Study Area
2.2. Construction Disturbance
2.3. Observing Eagle Owls
2.4. Statistics
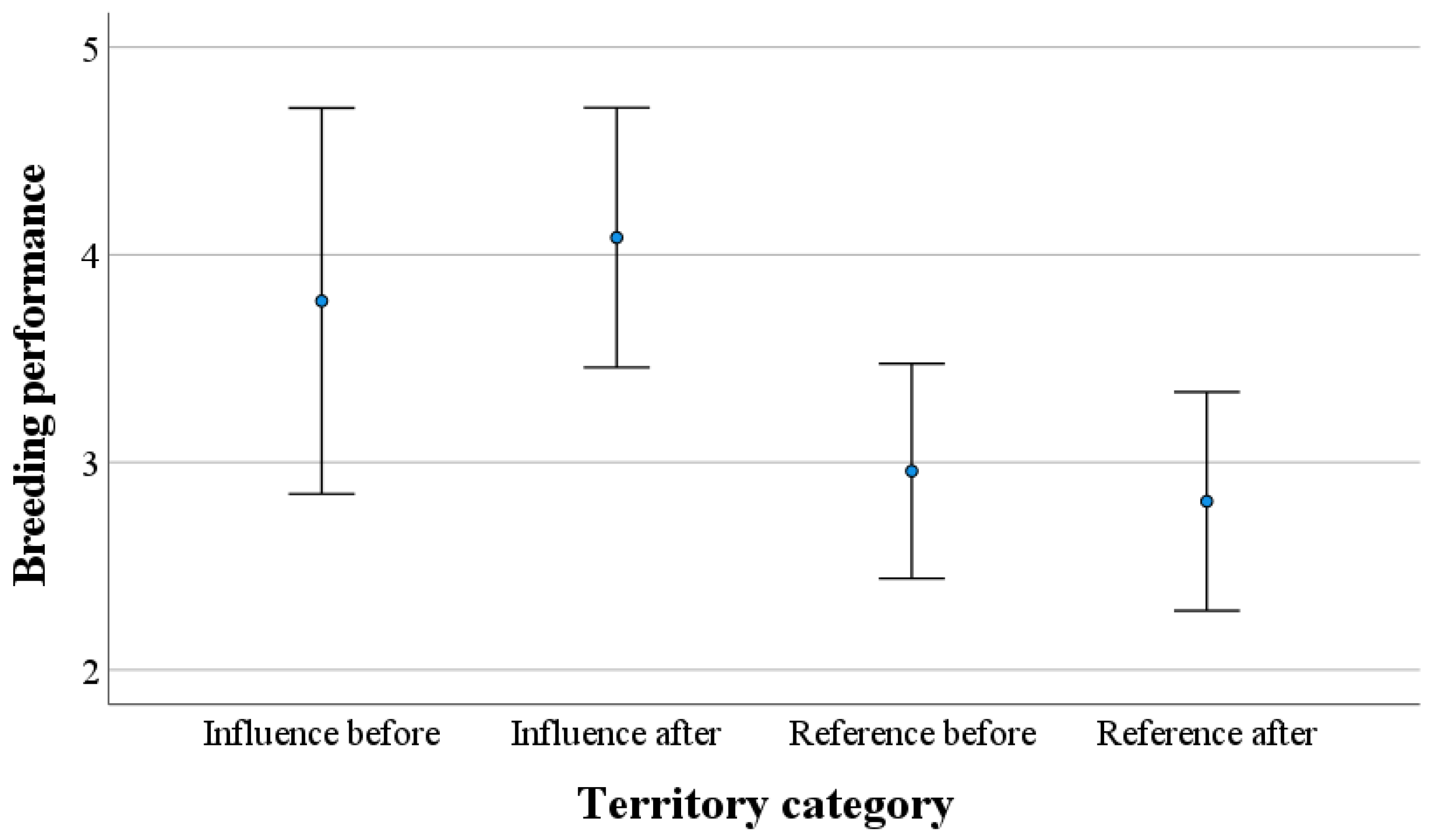
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IPCC. Climate Change 2021. The Physical Science Basis. Summary for Policy Makers; IPCC: Geneva, Switzerland, 2021; pp. 1–40. [Google Scholar]
- IPBES. Global Assessment Report of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services; IPBES Secretariat: Bonn, Germany, 2019; pp. 1–1144. [Google Scholar]
- Stephens, P.A.; Mason, L.R.; Green, R.E.; Gregory, R.D.; Sauer, J.R.; Alison, J.; Aunins, A.; Brotons, L.; Butchart, S.H.M.; Campedelli, T.; et al. Consistent response of bird populations to climate change on two continents. Science 2016, 352, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Spooner, F.E.B.; Pearson, R.G.; Freeman, R. Rapid warming is associated with population decline among terrestrial birds and mammals globally. Glob. Chang. Biol. 2018, 24, 4521–4531. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; McElroy, M.B.; Kiviluoma, J. Global potential for wind-generated electricity. Proc. Natl. Acad. Sci. USA 2009, 106, 10933–10938. [Google Scholar] [CrossRef] [PubMed]
- Fraixedas, S.; Lindén, A.; Meller, K.; Lindström, Å.; Keišs, O.; Kålås, J.A.; Husby, M.; Leivits, A.; Leivits, M.; Lehikoinen, A. Substantial decline of Northern European peatland bird populations: Consequences of drainage. Biol. Conserv. 2017, 214, 223–232. [Google Scholar] [CrossRef]
- Yrjölä, R.A.; Tanskanen, A.; Sarvanne, H.; Vickholm, J.; Lehikoinen, A. Can common forest bird species tolerate disturbances in neighbouring areas? A case study of the Vuosaari Harbour construction in southern Finland. Ornis Fenn. 2018, 95, 49–60. [Google Scholar]
- Garces, A.; Queiroga, F.; Prada, J.; Pires, I. A review of the mortality of wild fauna in Europe in the last century: The consequences of human activity. J. Wildl. Biodivers. 2020, 4, 34–55. [Google Scholar] [CrossRef]
- Domahidi, Z.; Shonfield, J.; Nielsen, S.E.; Spence, J.R.; Bayne, E.M. Spatial distribution of the Boreal Owl and Northern Saw-whet Owl in the Boreal region of Alberta, Canada. Avian Conserv. Ecol. 2019, 14, 12. [Google Scholar] [CrossRef]
- May, R.; Jackson, C.R.; Middel, H.; Stokke, B.G.; Verones, F. Life-cycle impacts of wind energy development on bird diversity in Norway. Environ. Impact Assess. Rev. 2021, 90, 11. [Google Scholar] [CrossRef]
- Stokke, B.G.; Nygård, T.; Falkdalen, U.; Pedersen, H.C.; May, R. Effect of tower base painting on willow ptarmigan collision rates with wind turbines. Ecol. Evol. 2020, 10, 5670–5679. [Google Scholar] [CrossRef]
- May, R.; Nygård, T.; Falkdalen, U.; Åström, J.; Hamre, Ø.; Stokke, B.G. Paint it black: Efficacy of increased wind turbine rotor blade visibility to reduce avian fatalities. Ecol. Evol. 2020, 10, 8927–8935. [Google Scholar] [CrossRef]
- de Lucas, M.; Janss, G.F.E.; Ferrer, M. Birds and Wind Farms: Risk Assessment and Mitigation; Quercus/Libreria Linneo: Madrid, Spain, 2007. [Google Scholar]
- Loss, S.R.; Will, T.; Marra, P. Direct mortality of birds from anthropogenic causes. In Annual Review of Ecology, Evolution, and Systematics; Futuyma, D.J., Ed.; Annual Reviews: Palo Alto, CA, USA, 2015; Volume 46, pp. 99–120. [Google Scholar]
- Beston, J.A.; Diffendorfer, J.E.; Loss, S.R.; Johnson, D.H. Prioritizing avian species for their risk of population-level consequences from wind energy development. PLoS ONE 2016, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Langgemach, T.; Dürr, T. Informationen über Einflüsse der Windenergienutzung auf Vögel.—Stand 10. Mai 2021, Aktualisierungen außer Fundzahlen hervorgehoben; Staatliche Vogelschutzwarte: Nennhausen, Germany, 2021; pp. 1–145. [Google Scholar]
- Pérez-García, J.M.; DeVault, T.L.; Botella, F.; Sánchez-Zapata, J.A. Using risk prediction models and species sensitivity maps for large-scale identification of infrastructure-related wildlife protection areas: The case of bird electrocution. Biol. Conserv. 2017, 210, 334–342. [Google Scholar] [CrossRef]
- Penteriani, V.; Delgado, D.M.M. The Eagle Owl; T & AD Poyser: London, UK, 2019; p. 384. [Google Scholar]
- Martinez, J.A.; Martinez, J.E.; Manosa, S.; Zuberogoitia, I.; Calvo, J.F. How to manage human-induced mortality in the eagle owl Bubo bubo. Bird Conserv. Int. 2006, 16, 265–278. [Google Scholar] [CrossRef]
- Sergio, F.; Marchesi, L.; Pedrini, P.; Ferrer, M.; Penteriani, V. Electrocution alters the distribution and density of a top predator, the eagle owl Bubo bubo. J. Appl. Ecol. 2004, 41, 836–845. [Google Scholar] [CrossRef]
- Bevanger, K.; Overskaug, K. Utility structures as a mortality factor for raptors and owls in Norway. In Holarctic Birds of Prey; Chancellor, R.D., Meyburg, B.U., Ferroro, J.J., Eds.; Adenex-Wwgbp: Merida, Spain, 1998; pp. 381–392. [Google Scholar]
- Rubolini, D.; Bassi, E.; Bogliani, G.; Galeotti, P.; Garavaglia, R. Eagle Owl Bubo bubo and power line interactions in the Italian Alps. Bird Conserv. Int. 2001, 11, 319–324. [Google Scholar] [CrossRef]
- Brumm, H.; Naguib, M. Environmental acoustics and the evolution of bird song. In Advances in the Study of Behavior; Naguib, M., Zuberbuhler, K., Clayton, N.S., Janik, V.M., Eds.; Elsevier Academic Press Inc.: San Diego, CA, USA, 2009; Volume 40, pp. 1–33. [Google Scholar]
- Reijnen, R.; Foppen, R. Effect of road traffic on the breeding site tenacity of male willow warblers (Phylloscopus trochilus). J. Fur Ornithol. 1991, 132, 291–295. [Google Scholar] [CrossRef]
- Foppen, R.; Reijnen, R. The effects of car traffic on breeding bird populations in woodland.2. Breeding dispersal of male Willow warblers (Phylloscopus-trochilus) in relation to the proximity of a highway. J. Appl. Ecol. 1994, 31, 95–101. [Google Scholar] [CrossRef]
- Francis, C.D.; Ortega, C.P.; Cruz, A. Noise Pollution Changes Avian Communities and Species Interactions. Curr. Biol. 2009, 19, 1415–1419. [Google Scholar] [CrossRef]
- Halfwerk, W.; Holleman, L.J.M.; Lessells, C.M.; Slabbekoorn, H. Negative impact of traffic noise on avian reproductive success. J. Appl. Ecol. 2011, 48, 210–219. [Google Scholar] [CrossRef]
- Husby, M.; Hoset, K.; Butler, S. Non-random sampling along rural–urban gradients may reduce reliability of multi-species farmland bird indicators and their trends. Ibis 2021, 163, 579–592. [Google Scholar] [CrossRef]
- Chen, K.; Liu, Q.P.; Liao, G.H.; Yang, Y.; Ren, L.Q.; Yang, H.X.; Chen, X. The Sound Suppression Characteristics of Wing Feather of Owl (Bubo bubo). J. Bionic Eng. 2012, 9, 192–199. [Google Scholar] [CrossRef]
- Mason, J.T.; McClure, C.J.W.; Barber, J.R. Anthropogenic noise impairs owl hunting behavior. Biol. Conserv. 2016, 199, 29–32. [Google Scholar] [CrossRef]
- Delaney, D.K.; Grubb, T.G.; Beier, P.; Pater, L.L.; Reiser, M.H. Effects of helicopter noise on Mexican spotted owls. J. Wildl. Manag. 1999, 63, 60–76. [Google Scholar] [CrossRef]
- Tempel, D.J.; Gutiérrez, R.J. Fecal corticosterone levels in California spotted owls exposed to low-intensity chainsaw sound. Wildl. Soc. Bull. 2003, 31, 698–702. [Google Scholar]
- Swarthout, E.C.H.; Steidl, R.J. Flush responses of Mexican spotted owls to recreationists. J. Wildl. Manag. 2001, 65, 312–317. [Google Scholar] [CrossRef]
- Swarthout, E.C.H.; Steidl, R.J. Experimental effects of hiking on breeding Mexican spotted owls. Conserv. Biol. 2003, 17, 307–315. [Google Scholar] [CrossRef][Green Version]
- Cramp, S. The Birds of the Western Palearctic. Vol. 4: Terns to Woodpeckers; Oxford University Press: Oxford, UK, 1985; Volume 4, p. 960. [Google Scholar]
- Haftorn, S. Norges Fugler; Universitetsforlaget: Oslo, Norway, 1971; p. 862. [Google Scholar]
- Hagen, Y. Rovfuglene og Viltpleien; Universitetsforlaget: Oslo, Norway, 1952; Volume 2, p. 622. [Google Scholar]
- Pearson, M.; Husby, M. Supplementary feeding improves breeding performance in Eurasian Eagle Owl Bubo bubo. Ornis Fenn. 2021, 98, 46–58. [Google Scholar]
- Øien, I.J.; Heggøy, O.; Schimmings, P.; Aarvak, T.; Jacobsen, K.-O.; Oddane, B.; Ranke, P.S.; Steen, O.F. Status for Hubro i Norge. NOF Report 2014-8. 2014, pp. 1–71. Available online: https://www.birdlife.no/prosjekter/rapporter/2014_08_NOF.pdf (accessed on 26 March 2022).
- Stokke, B.G.; Dale, S.; Jacobsen, K.-O.; Lislevand, T.; Solvang, R.; Strøm, H. Fugler Aves—Norge. Norsk Rødliste for Arter; Artsdatabanken: Trondheim, Norway, 2021. [Google Scholar]
- Lukac, G.; Hrsak, V. Influence of visitor numbers on breeding birds in the Paklenica National Park, Croatia. Ekol. Bratisl. 2005, 24, 186–199. [Google Scholar]
- Milchev, B.; Georgiev, V.; Kovachev, A. Breeding failures of the eagle owl Bubo bubo: Pros and cons of nesting in natural and human-made structures in SE Bulgaria. North-West. J. Zool. 2019, 15, 75–83. [Google Scholar]
- Marchesi, L.; Sergio, F.; Pedrini, P. Costs and benefits of breeding in human-altered landscapes for the Eagle Owl Bubo bubo. Ibis 2002, 144, E164–E177. [Google Scholar] [CrossRef]
- Heggøy, O.; Aarvak, T.; Ranke, P.S.; Solheim, R.; Øien, I.J. Home range and excursive post-breeding movements of Eurasian eagle-owls revealed by GPS satellite transmitters. J. Raptor Res. 2021, 55, 619–626. [Google Scholar] [CrossRef]
- Grünkorn, T.; Welcker, J. Raumnutzung und Flugverhalten von Uhus im Umfeld von Windenergieanlagen im Landesteil Schleswig. Eulenwelt 2018, 39–42. [Google Scholar]
- Grünkorn, T.; Welcker, J. Erhebung von Grundlagendaten zur Abschätzung des Kollisionsrisikos von Uhus an Windenergieanlagen im Landesteil Schleswig—Zwischenbericht; BioConsult SH: Husum, Germany, 2018. [Google Scholar]
- Obuch, J.; Bangjord, G. The Eurasian eagle-owl (Bubo bubo) diet in the Trøndelag region (Central Norway). Slovak Raptor J. 2016, 10, 51–64. [Google Scholar] [CrossRef]
- Bangjord, G.; Obuch, J. Næring Hos Hubro i Nærhet til Vindkraft på Frøya i 2021; BirdLife Norge notat 2022-2; BirdLife Norge: Trondheim, Norway, 2021; pp. 1–18. [Google Scholar]
- Husby, M.; Pearson, M.; Dørum, H. Vindkraftverk og Hubro på Innvordfjellet, Flatanger Kommune: Sannsynlige Jaktområder, Mulige Effekter av et Vindkraftverk, og en Vurdering av Avbøtende Tiltak; FoU-rapport nr. 66; Nord Universitet: Bodø, Norway, 2020; pp. 1–23. [Google Scholar]
- Husby, M.; Dørum, H.; Pearson, M. Registreringer av Hubro på og Ved Sørmarkfjellet, Flatanger og Osen Kommuner, i 2019 og 2020; FoU-rapport nr. 70; Nord Universitet: Bodø, Norway, 2021; pp. 1–31. [Google Scholar]
- del Hoyo, J.; Elliott, A.; Sargatal, J. Handbook of the Birds of the World. Vol. 5. Barn-Owls to Hummingbirds; Lynx Edicions: Barcelona, Spain, 1999; p. 759. [Google Scholar]
- Grava, T.; Mathevon, N.; Place, E.; Balluet, P. Individual acoustic monitoring of the European Eagle Owl Bubo bubo. Ibis 2008, 150, 279–287. [Google Scholar] [CrossRef]
- von Lossow, G. Der Uhu Bubo bubo am Mittleren Lech 2003 bis 2009. Ornitol. Anz. 2010, 49, 1–24. [Google Scholar]
- Lengagne, T. Temporal stability in the individual features in the calls of eagle owls (Bubo bubo). Behaviour 2001, 138, 1407–1419. [Google Scholar] [CrossRef]
- Dormann, C.F.; Elith, J.; Bacher, S.; Buchmann, C.; Carl, G.; Carre, G.; Marquez, J.R.G.; Gruber, B.; Lafourcade, B.; Leitao, P.J.; et al. Collinearity: A review of methods to deal with it and a simulation study evaluating their performance. Ecography 2013, 36, 27–46. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference. A Practical Information-Theoretic Approach, 2nd ed.; Springer: New York, NY, USA, 2002; p. 488. [Google Scholar]
- Zuur, A.F.; Ieno, E.N.; Elphick, C.S. A protocol for data exploration to avoid common statistical problems. Methods Ecol. Evol. 2010, 1, 3–14. [Google Scholar] [CrossRef]
- Lo, S.; Andrews, S. To transform or not to transform: Using generalized linear mixed models to analyse reaction time data. Front. Psychol. 2015, 6, 1171. [Google Scholar] [CrossRef]
- Bolker, B.M.; Brooks, M.E.; Clark, C.J.; Geange, S.W.; Poulsen, J.R.; Stevens, M.H.H.; White, J.S.S. Generalized linear mixed models: A practical guide for ecology and evolution. Trends Ecol. Evol. 2009, 24, 127–135. [Google Scholar] [CrossRef]
- Leon-Ortega, M.; Martinez, J.E.; Perez, E.; Lacalle, J.A.; Calvo, J.F. The contribution of non-protected areas to the conservation of Eurasian Eagle-owls in Mediterranean ecosystems. Ecosphere 2017, 8, 11. [Google Scholar] [CrossRef]
- Cramp, S.; Simmons, K.E.L. The Birds of the Western Palearctic. Vol. 2: Hawks to Bustards; Oxford University Press: Oxford, UK, 1980; p. 695. [Google Scholar]
- Illner, H. Comments on the Report “Wind Energy Developments and Natura 2000”, Edited by the European Commission in October 2010; Arbeitsgemeinschaft Biologischer Umweltschutz im Kreis Soest: Bad Sassendorf, Germany, 2011. [Google Scholar]
- Leon-Ortega, M.; Jimenez-Franco, M.V.; Martinez, J.E.; Calvo, J.F. Factors influencing territorial occupancy and reproductive success in a Eurasian Eagle-owl (Bubo bubo) population. PLoS ONE 2017, 12, 15. [Google Scholar] [CrossRef] [PubMed]
- Andreychev, A.V.; Lapshin, A.S.; Kuznetsov, V.A. Breeding success of the Eurasian eagle owl (Bubo bubo) and rodent population dynamics. Biol. Bull. 2016, 43, 851–861. [Google Scholar] [CrossRef]
- Andreychev, A.V.; Lapshin, A.S.; Kuznetsov, V.A. Successful reproduction of eagle owl (Bubo bubo) and dynamics of rodents’ number. Zool. Zhurnal 2016, 95, 204–215. [Google Scholar] [CrossRef]
- Brambilla, M.; Bionda, R. Variation in productivity and territory occupancy in an Eagle Owl Bubo bubo population. Ornis Fenn. 2013, 90, 50–56. [Google Scholar]
- Sergio, F.; Newton, I. Occupancy as a measure of territory quality. J. Anim. Ecol. 2003, 72, 857–865. [Google Scholar] [CrossRef]
- Campioni, L.; Delgado, M.M.; Lourenco, R.; Bastianelli, G.; Fernandez, N.; Penteriani, V. Individual and spatio-temporal variations in the home range behaviour of a long-lived, territorial species. Oecologia 2013, 172, 371–385. [Google Scholar] [CrossRef]
- Campioni, L.; Delgado, M.M.; Penteriani, V. Pattern of repeatability in the movement behaviour of a long-lived territorial species, the eagle owl. J. Zool. 2016, 298, 191–197. [Google Scholar] [CrossRef]
- Penteriani, V.; Gallardo, M.; Roche, P. Landscape structure and food supply affect eagle owl (Bubo bubo) density and breeding performance: A case of intra-population heterogeneity. J. Zool. 2002, 257, 365–372. [Google Scholar] [CrossRef]
- Ogada, D.L.; Kibuthu, P.M. Breeding ecology of Mackinder’s eagle-owls (Bubo capensis mackinderi) In farmlands of central Kenya. J. Raptor Res. 2012, 46, 327–335. [Google Scholar] [CrossRef]
- Pande, S.; Pawashe, A.; Mahajan, M.; Mahabal, A.; Joglekar, C.; Yosef, R. Breeding biology, nesting habitat, and diet of the rock eagle-owl (Bubo bengalensis). J. Raptor Res. 2011, 45, 211–219. [Google Scholar] [CrossRef]
- Bakken, V.; Runde, O.; Tjørve, E. Norsk Ringmerkingsatlas. Vol. 2; Stavanger Museum: Stavanger, Norway, 2006; Volume 2, pp. 1–446. [Google Scholar]
- Tome, D. Post-fledging survival and dynamics of dispersal in Long-eared Owls Asio otus. Bird Stud. 2011, 58, 193–199. [Google Scholar] [CrossRef]
- Naef-Daenzer, B.; Gruebler, M.U. Post-fledging survival of altricial birds: Ecological determinants and adaptation. J. Field Ornithol. 2016, 87, 227–250. [Google Scholar] [CrossRef]
- Husby, M. On the adaptive value of brood reduction in birds: Experiments with the magpie Pica pica. J. Anim. Ecol. 1986, 55, 75–83. [Google Scholar] [CrossRef]
- Husby, M.; Slagsvold, T. Postfledging behavior and survival in male and female magpies Pica pica. Ornis Scand. 1992, 23, 483–490. [Google Scholar] [CrossRef]
- Penteriani, V.; Gallardo, M.; Cazassus, H. Conspecific density biases passive auditory surveys. J. Field Ornithol. 2002, 73, 387–391. [Google Scholar] [CrossRef]
- Bernhoft, A.; Torget, V.; Vikøren, T.; Ørnsrud, R.; Lyche, J.L.; Sandvik, M.; Viljugrein, H.; Tarpai, A.; Mejdell, C.M.; Madslien, K. Miljøgifter i hubro i Norge 1994–2014. Rottegifter, klororganiske forbindelser og toksiske metaller. In Veterinærinstituttet. Report 18—2018; Veterinærinstituttet: Oslo, Norway, 2018. [Google Scholar]
- Pearson, M.J.D. Levels of Perfluoroalkyl and Polyfluoroalkyl Substances (PFASs) in Feathers of Eurasian Eagle-Owls (Bubo Bubo) in Norway; NTNU: Trondheim, Norway, 2017. [Google Scholar]
- Olsson, V. Studies on a population of eagle owls, Bubo bubo (L.) in Southeast Sweden. Swed. Wildl. Res. 1979, 11, 1–93. [Google Scholar]
- LAG VSW. Recommendations for distances of wind turbines to important areas for birds as well as breeding sites of selected bird species (as at April 2015). Ber. Zum Vogelschutz 2015, 51, 15–42. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Husby, M.; Pearson, M. Wind Farms and Power Lines Have Negative Effects on Territory Occupancy in Eurasian Eagle Owls (Bubo bubo). Animals 2022, 12, 1089. https://doi.org/10.3390/ani12091089
Husby M, Pearson M. Wind Farms and Power Lines Have Negative Effects on Territory Occupancy in Eurasian Eagle Owls (Bubo bubo). Animals. 2022; 12(9):1089. https://doi.org/10.3390/ani12091089
Chicago/Turabian StyleHusby, Magne, and Martin Pearson. 2022. "Wind Farms and Power Lines Have Negative Effects on Territory Occupancy in Eurasian Eagle Owls (Bubo bubo)" Animals 12, no. 9: 1089. https://doi.org/10.3390/ani12091089
APA StyleHusby, M., & Pearson, M. (2022). Wind Farms and Power Lines Have Negative Effects on Territory Occupancy in Eurasian Eagle Owls (Bubo bubo). Animals, 12(9), 1089. https://doi.org/10.3390/ani12091089





