Drug-Dosing Adjustment in Dogs and Cats with Chronic Kidney Disease
Abstract
Simple Summary
Abstract
1. Introduction: Chronic Kidney Disease and Implications in Drug Dosing
2. Evaluation of Renal Function
3. Mechanisms Underlying Altered Renal Drug Handling in CKD
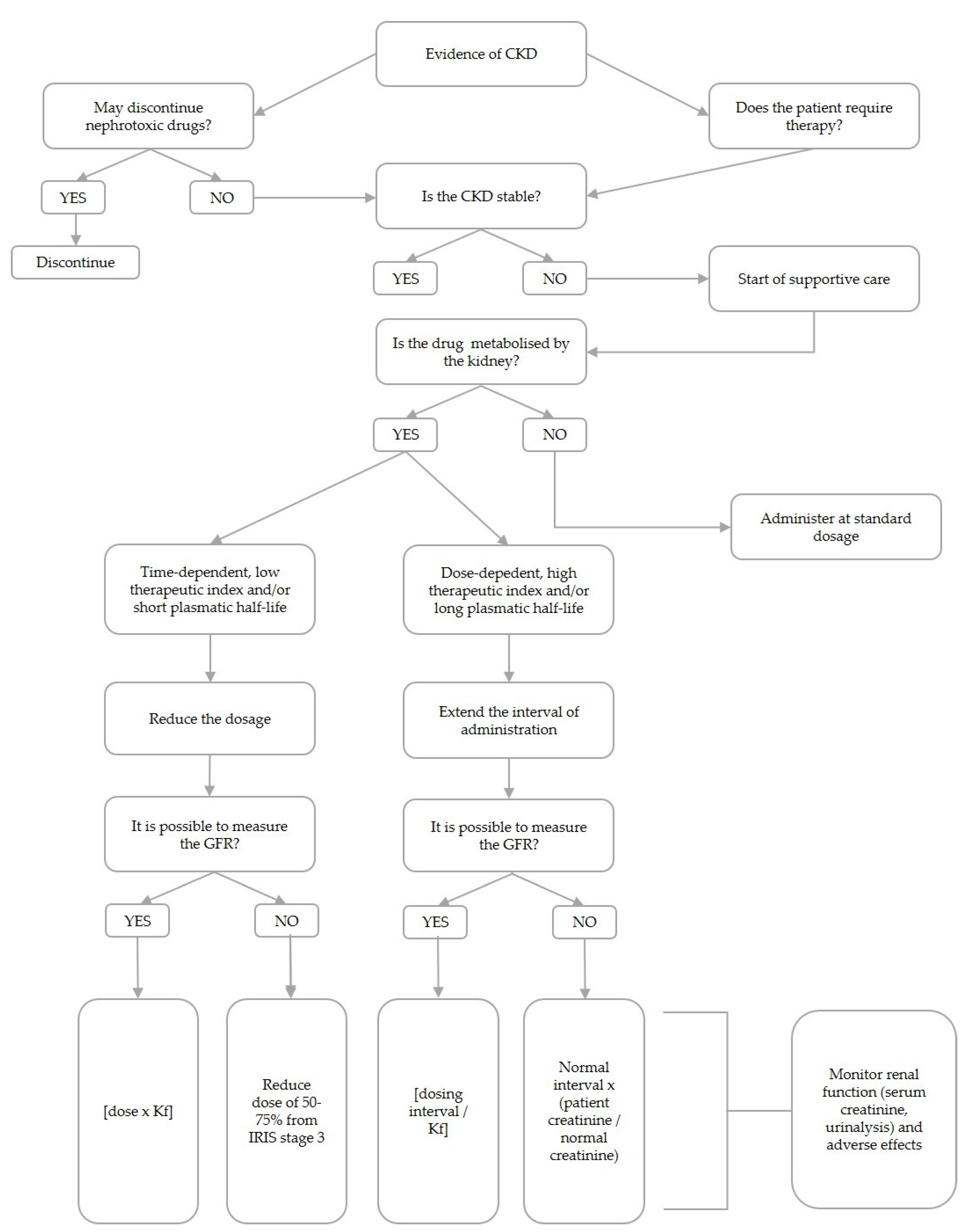
4. Rational Design of Dosing Regimens in CKD Patients
5. Antimicrobial Agents
5.1. Aminoglycosides
5.2. Penicillins
5.3. Cephalosporins
5.4. Sulfonamides
5.5. Tetracyclines
5.6. Fluoroquinolones
6. Anti-Inflammatory Drugs
6.1. Non-Steroidal Anti-Inflammatory Drugs
6.2. Corticosteroids
7. Antiproteinuric and Antihypertensive Drugs
7.1. Renin-Angiotensin-Aldosterone System (RAAS) Inhibitors
7.2. Amlodipine
7.3. Other Cardiovascular Drugs
8. Antithrombotic Drugs
9. Gastroprotectants, Antiemetics and Appetite Stimulants
Mirtazapine
10. Antileishmanial Drugs
11. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Polzin, D.J. Chronic Kidney Disease in Small Animals. Vet. Clin. N. Am. Small Anim. Pract. 2011, 41, 15–30. [Google Scholar] [CrossRef]
- Brown, S.A. Management of Chronic Kidney Disease. In BSAVA Manual of Canine and Feline Nephrology and Urology; British Small Animal Veterinary Association, Ed.; British Small Animal Veterinary Association: Gloucs, UK, 2007; ISBN 978-0905214931. [Google Scholar]
- Polzin, D.J. Chronic Kidney Disease. In Nephrology and Urology of Small Animals; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2011; ISBN 978-0-8138-1717-0. [Google Scholar]
- Marino, C.L.; Lascelles, B.D.X.; Vaden, S.L.; Gruen, M.E.; Marks, S.L. Prevalence and Classification of Chronic Kidney Disease in Cats Randomly Selected from Four Age Groups and in Cats Recruited for Degenerative Joint Disease Studies. J. Feline Med. Surg. 2014, 16, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Roudebush, P.; Polzin, D.J.; Adams, L.G.; Towell, T.L.; Forrester, S.D. An Evidence-Based Review of Therapies for Canine Chronic Kidney Disease. J. Small Anim. Pract. 2010, 51, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Polzin, D.J. Evidence-Based Step-Wise Approach to Managing Chronic Kidney Disease in Dogs and Cats: Managing Chronic Kidney Disease. J. Vet. Emerg. Crit. Care 2013, 23, 205–215. [Google Scholar] [CrossRef]
- Sparkes, A.H.; Caney, S.; Chalhoub, S.; Elliott, J.; Finch, N.; Gajanayake, I.; Langston, C.; Lefebvre, H.P.; White, J.; Quimby, J. ISFM Consensus Guidelines on the Diagnosis and Management of Feline Chronic Kidney Disease. J. Feline Med. Surg. 2016, 18, 219–239. [Google Scholar] [CrossRef]
- Goggs, R.; Bacek, L.; Bianco, D.; Koenigshof, A.; Li, R.H.L. Consensus on the Rational Use of Antithrombotics in Veterinary Critical Care (CURATIVE): Domain 2-Defining Rational Therapeutic Usage. J. Vet. Emerg. Crit. Care 2019, 29, 49–59. [Google Scholar] [CrossRef]
- Tett, S.E.; Kirkpatrick, C.M.J.; Gross, A.S.; McLachlan, A.J. Principles and Clinical Application of Assessing Alterations in Renal Elimination Pathways. Clin. Pharmacokinet. 2003, 42, 1193–1211. [Google Scholar] [CrossRef]
- Lea-Henry, T.N.; Carland, J.E.; Stocker, S.L.; Sevastos, J.; Roberts, D.M. Clinical Pharmacokinetics in Kidney Disease: Fundamental Principles. CJASN 2018, 13, 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- Verbeeck, R.K.; Musuamba, F.T. Pharmacokinetics and Dosage Adjustment in Patients with Renal Dysfunction. Eur. J. Clin. Pharm. 2009, 65, 757–773. [Google Scholar] [CrossRef]
- Riviere, J.E.; Vaden, S.L. Drug Therapy during Renal Disease and Renal Failure. In Canine and Feline Nephrology and Urology; Williams and Wilkins: Philadelphia, PA, USA, 1995; ISBN 0683066668. [Google Scholar]
- Albarellos, G.A.; Landoni, M.F. Current Concepts on the Use of Antimicrobials in Cats. Vet. J. 2009, 180, 304–316. [Google Scholar] [CrossRef]
- Linnetz, E.H.; Graves, T.K. Glomerular Filtration Rate in General Small Animal Practice. Compend. Contin. Educ. Vet. 2010, 32, E1–E5. [Google Scholar]
- Lefebvre, H.P. Renal Function Testing. In Nephrology and Urology of Small Animals; Wiley-Blackwell: Hoboken, NJ, USA, 2011; ISBN 978-0-8138-1717-0. [Google Scholar]
- Pressler, B.M. Clinical Approach to Advanced Renal Function Testing in Dogs and Cats. Clin. Lab. Med. 2015, 35, 487–502. [Google Scholar] [CrossRef] [PubMed]
- McKenna, M.; Pelligand, L.; Elliott, J.; Walker, D.; Jepson, R. Clinical Utility of Estimation of Glomerular Filtration Rate in Dogs. J. Vet. Intern. Med. 2020, 34, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Pocar, P.; Scarpa, P.; Berrini, A.; Cagnardi, P.; Rizzi, R.; Borromeo, V. Diagnostic Potential of Simplified Methods for Measuring Glomerular Filtration Rate to Detect Chronic Kidney Disease in Dogs. J. Vet. Intern. Med. 2019, 33, 2105–2116. [Google Scholar] [CrossRef] [PubMed]
- Von Hendy-Willson, V.E.; Pressler, B.M. An Overview of Glomerular Filtration Rate Testing in Dogs and Cats. Vet. J. 2011, 188, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.A.; Finco, D.R.; Boudinot, F.D.; Wright, J.; Taver, S.L.; Cooper, T. Evaluation of a Single Injection Method, Using Iohexol, for Estimating Glomerular Filtration Rate in Cats and Dogs. Am. J. Vet. Res. 1996, 57, 105–110. [Google Scholar] [PubMed]
- Bexfield, N.H.; Heiene, R.; Gerritsen, R.J.; Risøen, U.; Eliassen, K.A.; Herrtage, M.E.; Michell, A.R. Glomerular Filtration Rate Estimated by 3-Sample Plasma Clearance of Iohexol in 118 Healthy Dogs. J. Vet. Intern. Med. 2008, 22, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Heiene, R.; Reynolds, B.S.; Bexfield, N.H.; Larsen, S.; Gerritsen, R.J. Estimation of Glomerular Filtration Rate via 2- and 4-Sample Plasma Clearance of Iohexol and Creatinine in Clinically Normal Cats. Am. J. Vet. Res. 2009, 70, 176–185. [Google Scholar] [CrossRef]
- Heiene, R.; Moe, L. Pharmacokineatic Aspects of Measurement of Glomerular Filtration Rate in the Dog: A Review. J. Vet. Intern. Med. 1998, 12, 401–414. [Google Scholar] [CrossRef]
- Sargent, H.J.; Elliott, J.; Jepson, R.E. The New Age of Renal Biomarkers: Does SDMA Solve All of Our Problems? J. Small Anim. Pr. 2021, 62, 71–81. [Google Scholar] [CrossRef]
- IRIS Kidney—Guidelines—IRIS Staging of CKD. Available online: http://www.iris-kidney.com/guidelines/staging.html (accessed on 10 October 2021).
- Finco, D.R.; Brown, S.A.; Crowell, W.A.; Barsanti, J.A. Exogenous Creatinine Clearance as a Measure of Glomerular Filtration Rate in Dogs with Reduced Renal Mass. Am. J. Vet. Res. 1991, 52, 1029–1032. [Google Scholar]
- Finch, N. Measurement of Glomerular Filtration Rate in Cats: Methods and Advantages over Routine Markers of Renal Function. J. Feline Med. Surg. 2014, 16, 736–748. [Google Scholar] [CrossRef] [PubMed]
- Nabity, M.B.; Lees, G.E.; Boggess, M.M.; Yerramilli, M.; Obare, E.; Yerramilli, M.; Rakitin, A.; Aguiar, J.; Relford, R. Symmetric Dimethylarginine Assay Validation, Stability, and Evaluation as a Marker for the Early Detection of Chronic Kidney Disease in Dogs. J. Vet. Intern. Med. 2015, 29, 1036–1044. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.P.; Lefebvre, H.P.; Watson, A.D.J. Creatinine in the Dog: A Review. Vet. Clin. Pathol. 2003, 32, 162–179. [Google Scholar] [CrossRef]
- Barsanti, J.A. Urinary Disorders. In Small Animal Clinical Diagnosis by Laboratory Methods; Elsevier: Amsterdam, The Netherlands, 2012; ISBN 978-1-4377-0657-4. [Google Scholar]
- Wamsley, H.; Alleman, R. Complete Urinalysis. In BSAVA Manual of Canine and Feline Nephrology and Urology; British Small Animal Veterinary Association: Gloucester, UK, 2017; ISBN 978-1-905319-94-7. [Google Scholar]
- Adams, L.G.; Polzin, D.J.; Osborne, C.A.; O’Brien, T.D. Effects of Dietary Protein and Calorie Restriction in Clinically Normal Cats and in Cats with Surgically Induced Chronic Renal Failure. Am. J. Vet. Res. 1993, 54, 1653–1662. [Google Scholar]
- Kielstein, J.T.; Salpeter, S.R.; Bode-Boeger, S.M.; Cooke, J.P.; Fliser, D. Symmetric Dimethylarginine (SDMA) as Endogenous Marker of Renal Function—A Meta-Analysis. Nephrol. Dial. Transpl. 2006, 21, 2446–2451. [Google Scholar] [CrossRef]
- Bedford, M.T.; Clarke, S.G. Protein Arginine Methylation in Mammals: Who, What, and Why. Mol. Cell 2009, 33, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.A.; Yerramilli, M.; Obare, E.; Yerramilli, M.; Melendez, L.D.; Jewell, D.E. Relationship between Lean Body Mass and Serum Renal Biomarkers in Healthy Dogs. J. Vet. Intern. Med. 2015, 29, 808–814. [Google Scholar] [CrossRef]
- Yerramilli, M.; Farace, G.; Quinn, J.; Yerramilli, M. Kidney Disease and the Nexus of Chronic Kidney Disease and Acute Kidney Injury. Vet. Clin. N. Am. Small Anim. Pract. 2016, 46, 961–993. [Google Scholar] [CrossRef]
- Hall, J.A.; Yerramilli, M.; Obare, E.; Yerramilli, M.; Jewell, D.E. Comparison of Serum Concentrations of Symmetric Dimethylarginine and Creatinine as Kidney Function Biomarkers in Cats with Chronic Kidney Disease. J. Vet. Intern. Med. 2014, 28, 1676–1683. [Google Scholar] [CrossRef]
- Doogue, M.P.; Polasek, T.M. Drug Dosing in Renal Disease. Clin. Biochem. Rev. 2011, 32, 69–73. [Google Scholar] [PubMed]
- Roberts, D.M.; Sevastos, J.; Carland, J.E.; Stocker, S.L.; Lea-Henry, T.N. Clinical Pharmacokinetics in Kidney Disease: Application to Rational Design of Dosing Regimens. CJASN 2018, 13, 1254–1263. [Google Scholar] [CrossRef]
- Papich, M. Drugs for Treating Gastrointestinal Diseases. In Veterinary Pharmacology and Therapeutics; Wiley-Blackwell: Hoboken, NJ, USA, 2018; pp. 1245–1277. ISBN 978-1-118-85588-1. [Google Scholar]
- Littman, M.P. Protein-Losing Nephropathy in Small Animals. Vet. Clin. N. Am. Small Anim. Pr. 2011, 41, 31–62. [Google Scholar] [CrossRef] [PubMed]
- Jansen, J.; Jankowski, J.; Gajjala, P.R.; Wetzels, J.F.M.; Masereeuw, R. Disposition and Clinical Implications of Protein-Bound Uremic Toxins. Clin. Sci. 2017, 131, 1631–1647. [Google Scholar] [CrossRef] [PubMed]
- Riviere, J.E. Pharmacokinetics. In Veterinary Pharmacology and Therapeutics; Wiley-Blackwell: Hoboken, NJ, USA, 2017; ISBN 978-1-118-85588-1. [Google Scholar]
- Santana Machado, T.; Poitevin, S.; Paul, P.; McKay, N.; Jourde-Chiche, N.; Legris, T.; Mouly-Bandini, A.; Dignat-George, F.; Brunet, P.; Masereeuw, R.; et al. Indoxyl Sulfate Upregulates Liver P-Glycoprotein Expression and Activity through Aryl Hydrocarbon Receptor Signaling. J. Am. Soc. Nephrol. 2018, 29, 906–918. [Google Scholar] [CrossRef]
- Bricker, N.S.; Morrin, P.A.F.; Kime, S.W. The Pathologic Physiology of Chronic Bright’s Disease. Am. J. Med. 1960, 28, 77–98. [Google Scholar] [CrossRef]
- Pradhan, S.; Duffull, S.B.; Walker, R.J.; Wright, D.F.B. The Intact Nephron Hypothesis as a Model for Renal Drug Handling. Eur. J. Clin. Pharm. 2019, 75, 147–156. [Google Scholar] [CrossRef]
- Bonate, P.L.; Reith, K.; Weir, S. Drug Interactions at the Renal Level: Implications for Drug Development. Clin. Pharmacokinet. 1998, 34, 375–404. [Google Scholar] [CrossRef]
- Sun, H.; Huang, Y.; Frassetto, L.; Benet, L.Z. Effects of Uremic Toxins on Hepatic Uptake and Metabolism of Erythromycin. Drug Metab. Dispos. 2004, 32, 1239–1246. [Google Scholar] [CrossRef]
- Michaud, J.; Dubé, P.; Naud, J.; Leblond, F.A.; Desbiens, K.; Bonnardeaux, A.; Pichette, V. Effects of Serum from Patients with Chronic Renal Failure on Rat Hepatic Cytochrome P450: Human Uremic Serum Downregulates Cytochrome P450. Br. J. Pharmacol. 2005, 144, 1067–1077. [Google Scholar] [CrossRef]
- Lefebvre, H.P. Dosage Regimen Adjustment in Renal Failure: Why, When and How. Ph.D. Thesis, National Veterinary School of Toulouse, Toulouse, France, 2002. [Google Scholar]
- Gabardi, S.; Abramson, S. Drug Dosing in Chronic Kidney Disease. Med. Clin. N. Am. 2005, 89, 649–687. [Google Scholar] [CrossRef] [PubMed]
- Swan, S.K.; Bennett, W.M. Drug Dosing Guidelines in Patients with Renal Failure. West. J. Med. 1992, 156, 633–638. [Google Scholar] [PubMed]
- Wiebe, V.J. Drug Therapy for Infectious Diseases of the Dog and Cat: Wiebe/Drug Therapy for Infectious Diseases of the Dog and Cat; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; ISBN 978-1-118-91116-7. [Google Scholar]
- Drug Dose Adjustments for Disease (Proceedings). Available online: https://www.dvm360.com/view/drug-dose-adjustments-disease-proceedings (accessed on 10 October 2021).
- Lefebvre, H.P.; Braun, J.P.; Toutain, P.L. Drug Prescription in Renal-Impaired Dogs. Rev. Méd. Vét. 1996, 147, 757–782. [Google Scholar]
- Hornum, M.; Feldt-Rasmussen, B. Drug Dosing and Estimated Renal Function—Any Step Forward from Effersoe? Nephron 2017, 136, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Aloy, B.; Launay-Vacher, V.; Bleibtreu, A.; Bortolotti, P.; Faure, E.; Filali, A.; Gauzit, R.; Gilbert, M.; Lesprit, P.; Mahieu, R.; et al. Antibiotics and Chronic Kidney Disease: Dose Adjustment Update for Infectious Disease Clinical Practice. Méd. Mal. Infect. 2020, 50, 323–331. [Google Scholar] [CrossRef]
- Boothe, D.M. Therapeutic Drug Monitoring. In Small Animal Clinical Diagnosis by Laboratory Methods; Elsevier: St. Louis, MO, USA, 2012; ISBN 978-1-4377-0657-4. [Google Scholar]
- Groman, R.P. Aminoglycosides. In Small Animal Critical Care Medicine; Elsevier Health Sciences: Amsterdam, The Netherlands, 2014; ISBN 978-1-4557-0306-7. [Google Scholar]
- Plumb, D.C. Plumb’s Veterinary Drug Handbook, 8th ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2015; Volume 93, ISBN 978-1-118-91192-1. [Google Scholar]
- Monaghan, K.N.; Labato, M.A.; Papich, M.G. Ampicillin Pharmacokinetics in Azotemic and Healthy Dogs. J. Vet. Intern. Med. 2021, 35, 987–992. [Google Scholar] [CrossRef]
- Benson, K.K.; Quimby, J.M.; Dowers, K.L.; Sieberg, L.G.; Daniels, J.B.; Langston, C.E.; Lunghofer, P.J.; Gustafson, D.L. Pilot Study of Side Effects and Serum and Urine Concentrations of Amoxicillin–Clavulanic Acid in Azotemic and Non-Azotemic Cats. J. Feline Med. Surg. 2020, 22, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Matzke, G.R.; Aronoff, G.R.; Atkinson, A.J.; Bennett, W.M.; Decker, B.S.; Eckardt, K.-U.; Golper, T.; Grabe, D.W.; Kasiske, B.; Keller, F.; et al. Drug Dosing Consideration in Patients with Acute and Chronic Kidney Disease—A Clinical Update from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 2011, 80, 1122–1137. [Google Scholar] [CrossRef]
- Trepanier, L.A. Applying Pharmacokinetics to Veterinary Clinical Practice. Vet. Clin. N. Am. Small Anim. Pract. 2013, 43, 1013–1026. [Google Scholar] [CrossRef]
- Papich, M.G. Sulfonamides and Potentiated Sulfonamides. In Veterinary Pharmacology and Therapeutics; Wiley-Blackwell: Hoboken, NJ, USA, 2017; ISBN 978-1-118-85588-1. [Google Scholar]
- Trouchon, T.; Lefebvre, S. A Review of Enrofloxacin for Veterinary Use. OJVM 2016, 06, 40–58. [Google Scholar] [CrossRef]
- McLean, M.K.; Khan, S.A. Toxicology of Frequently Encountered Nonsteroidal Anti-Inflammatory Drugs in Dogs and Cats. Vet. Clin. N. Am. Small Anim. Pract. 2018, 48, 969–984. [Google Scholar] [CrossRef]
- Monteiro-Steagall, B.P.; Steagall, P.V.M.; Lascelles, B.D.X. Systematic Review of Nonsteroidal Anti-Inflammatory Drug-Induced Adverse Effects in Dogs. J. Vet. Intern. Med. 2013, 27, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Papich, M.G. An Update on Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) in Small Animals. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 1243–1266. [Google Scholar] [CrossRef] [PubMed]
- Lomas, A.L.; Grauer, G.F. The Renal Effects of NSAIDs in Dogs. J. Am. Anim. Hosp. Assoc. 2015, 51, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Waters, C.B.; Adams, L.G.; Scott-Moncrieff, J.C.; DeNicola, D.B.; Snyder, P.W.; White, M.R.; Gasparini, M. Effects of Glucocorticoid Therapy on Urine Protein-to-Creatinine Ratios and Renal Morphology in Dogs. J. Vet. Intern. Med. 1997, 11, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Vaden, S.L.; Elliott, J. Management of Proteinuria in Dogs and Cats with Chronic Kidney Disease. Vet. Clin. N. Am. Small Anim. Pract. 2016, 46, 1115–1130. [Google Scholar] [CrossRef]
- Acierno, M.J.; Brown, S.; E. Coleman, A.; E. Jepson, R.; Papich, M.; L. Stepien, R.; M. Syme, H. ACVIM Consensus Statement: Guidelines for the Identification, Evaluation, and Management of Systemic Hypertension in Dogs and Cats. J. Jpn. Assoc. Vet. Nephrol. Urol. 2020, 12, 30–49. [Google Scholar] [CrossRef]
- Coleman, A.E.; Brown, S.A.; Traas, A.M.; Bryson, L.; Zimmering, T.; Zimmerman, A. Safety and Efficacy of Orally Administered Telmisartan for the Treatment of Systemic Hypertension in Cats: Results of a Double-blind, Placebo-controlled, Randomized Clinical Trial. J. Vet. Intern. Med. 2019, 33, 478–488. [Google Scholar] [CrossRef]
- Lourenço, B.N.; Coleman, A.E.; Brown, S.A.; Schmiedt, C.W.; Parkanzky, M.C.; Creevy, K.E. Efficacy of Telmisartan for the Treatment of Persistent Renal Proteinuria in Dogs: A Double-masked, Randomized Clinical Trial. J. Vet. Intern. Med. 2020, 34, 2478–2496. [Google Scholar] [CrossRef]
- Miyagawa, Y.; Akabane, R.; Sakatani, A.; Ogawa, M.; Nagakawa, M.; Miyakawa, H.; Takemura, N. Effects of Telmisartan on Proteinuria and Systolic Blood Pressure in Dogs with Chronic Kidney Disease. Res. Vet. Sci. 2020, 133, 150–156. [Google Scholar] [CrossRef]
- Taylor, S.S.; Sparkes, A.H.; Briscoe, K.; Carter, J.; Sala, S.C.; Jepson, R.E.; Reynolds, B.S.; Scansen, B.A. ISFM Consensus Guidelines on the Diagnosis and Management of Hypertension in Cats. J. Feline Med. Surg. 2017, 19, 288–303. [Google Scholar] [CrossRef]
- Orvalho, J.S.; Cowgill, L.D. Cardiorenal Syndrome. Vet. Clin. N. Am. Small Anim. Pract. 2017, 47, 1083–1102. [Google Scholar] [CrossRef]
- Papich, M.G. Anticoagulant, Antiplatelet, and Hemostatic Drugs. In Veterinary Pharmacology and Therapeutics; Wiley-Blackwell: Hoboken, NJ, USA, 2018; ISBN 978-1-118-85588-1. [Google Scholar]
- Capodanno, D.; Angiolillo, D.J. Antithrombotic Therapy in Patients With Chronic Kidney Disease. Circulation 2012, 125, 2649–2661. [Google Scholar] [CrossRef]
- Segal, R.; Lubart, E.; Leibovitz, A.; Iaina, A.; Caspi, D. Renal Effects of Low Dose Aspirin in Elderly Patients. Isr. Med. Assoc. J. 2006, 8, 679–682. [Google Scholar] [PubMed]
- Kim, A.J.; Lim, H.J.; Ro, H.; Ko, K.-P.; Han, S.Y.; Chang, J.H.; Lee, H.H.; Chung, W.; Jung, J.Y. Low-Dose Aspirin for Prevention of Cardiovascular Disease in Patients with Chronic Kidney Disease. PLoS ONE 2014, 9, e104179. [Google Scholar] [CrossRef]
- Tsai, M.-H.; Liou, H.-H.; Huang, Y.-C.; Lee, T.-S.; Chen, M.; Fang, Y.-W. Hazardous Effect of Low-Dose Aspirin in Patients with Predialysis Advanced Chronic Kidney Disease Assessed by Machine Learning Method Feature Selection. Healthcare 2021, 9, 1484. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.J.; Kim, A.J.; Ro, H.; Chang, J.H.; Lee, H.H.; Chung, W.; Hyun, Y.Y.; Lee, J.; Kim, Y.H.; Han, S.H.; et al. Low-Dose Aspirin Was Associated with an Increased Risk of Cardiovascular Events in Patients with Chronic Kidney Disease and Low Bodyweight: Results from KNOW-CKD Study. Sci. Rep. 2021, 11, 6691. [Google Scholar] [CrossRef] [PubMed]
- Papich, M.G. Saunders Handbook of Veterinary Drugs: Small and Large Animal, 4th ed.; Elsevier: St. Louis, MI, USA, 2016; ISBN 978-0-323-24485-5. [Google Scholar]
- Sparkes, A.H.; Heiene, R.; Lascelles, B.D.X.; Malik, R.; Real, L.; Robertson, S.; Scherk, M.; Taylor, P. ISFM and AAFP Consensus Guidelines: Long-Term Use of NSAIDs in Cats. J. Feline Med. Surg. 2010, 12, 521–538. [Google Scholar] [CrossRef] [PubMed]
- Deray, G.; Bagnis, C.; Brouard, R.; Necciari, J.; Leenhardt, A.F.; Raymond, F.; Baumelou, A. Clopidogrel Activities in Patients with Renal Function Impairment. Clin. Drug Investig. 1998, 16, 319–328. [Google Scholar] [CrossRef]
- Tanios, B.Y.; Itani, H.S.; Zimmerman, D.L. Clopidogrel Use in End-Stage Kidney Disease. Semin. Dial. 2015, 28, 276–281. [Google Scholar] [CrossRef]
- Jiang, X.-L.; Samant, S.; Lesko, L.J.; Schmidt, S. Clinical Pharmacokinetics and Pharmacodynamics of Clopidogrel. Clin. Pharm. 2015, 54, 147–166. [Google Scholar] [CrossRef]
- Nishi, T.; Ariyoshi, N.; Nakayama, T.; Fujimoto, Y.; Sugimoto, K.; Wakabayashi, S.; Hanaoka, H.; Kobayashi, Y. Impact of Chronic Kidney Disease on Platelet Inhibition of Clopidogrel and Prasugrel in Japanese Patients. J. Cardiol. 2017, 69, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Shropshire, S.; Johnson, T.; Olver, C. Platelet Aggregometry Testing during Aspirin or Clopidogrel Treatment and Measurement of Clopidogrel Metabolite Concentrations in Dogs with Protein-losing Nephropathy. J. Vet. Intern. Med. 2020, 34, 710–718. [Google Scholar] [CrossRef] [PubMed]
- Segev, G.; Naylor, S.; Cowgill, L.D. Hematological and Neurological Side Effects Associated with the Use of Aluminum Based Phosphate Binders in Dogs with Chronic Kidney Disease. Isr. J. Vet. Med. 2016, 71, 31–38. [Google Scholar]
- Manlucu, J.; Tonelli, M.; Ray, J.G.; Papaioannou, A.; Youssef, G.; Thiessen-Philbrook, H.R.; Holbrook, A.; Garg, A.X. Dose-Reducing H2 Receptor Antagonists in the Presence of Low Glomerular Filtration Rate: A Systematic Review of the Evidence. Nephrol Dial. Transpl. 2005, 20, 2376–2384. [Google Scholar] [CrossRef] [PubMed]
- Marks, S.L.; Kook, P.H.; Papich, M.G.; Tolbert, M.K.; Willard, M.D. ACVIM Consensus Statement: Support for Rational Administration of Gastrointestinal Protectants to Dogs and Cats. J. Vet. Intern. Med. 2018, 32, 1823–1840. [Google Scholar] [CrossRef] [PubMed]
- Sierra, F.; Suarez, M.; Rey, M.; Vela, M.F. Systematic Review: Proton Pump Inhibitor-Associated Acute Interstitial Nephritis: Systematic Review: PPI-Induced Interstitial Nephritis. Aliment. Pharmacol. Ther. 2007, 26, 545–553. [Google Scholar] [CrossRef]
- Lazarus, B.; Chen, Y.; Wilson, F.P.; Sang, Y.; Chang, A.R.; Coresh, J.; Grams, M.E. Proton Pump Inhibitor Use and the Risk of Chronic Kidney Disease. JAMA Intern. Med. 2016, 176, 238. [Google Scholar] [CrossRef]
- de la Puente-Redondo, V.A.; Siedek, E.M.; Benchaoui, H.A.; Tilt, N.; Rowan, T.G.; Clemence, R.G. The Anti-Emetic Efficacy of Maropitant (Cerenia?) In the Treatment of Ongoing Emesis Caused by a Wide Range of Underlying Clinical Aetiologies in Canine Patients in Europe. J. Small Anim. Pract. 2007, 48, 93–98. [Google Scholar] [CrossRef]
- Ramsey, D.S.; Kincaid, K.; Watkins, J.A.; Boucher, J.F.; Conder, G.A.; Eagleson, J.S.; Clemence, R.G. Safety and Efficacy of Injectable and Oral Maropitant, a Selective Neurokinin 1 Receptor Antagonist, in a Randomized Clinical Trial for Treatment of Vomiting in Dogs. J. Vet. Pharmacol. Ther. 2008, 31, 538–543. [Google Scholar] [CrossRef]
- Quimby, J.M.; Brock, W.T.; Moses, K.; Bolotin, D.; Patricelli, K. Chronic Use of Maropitant for the Management of Vomiting and Inappetence in Cats with Chronic Kidney Disease: A Blinded, Placebo-Controlled Clinical Trial. J. Feline Med. Surg. 2015, 17, 692–697. [Google Scholar] [CrossRef]
- Fitzpatrick, R.L.; Wittenburg, L.A.; Hansen, R.J.; Gustafson, D.L.; Quimby, J.M. Limited Sampling Pharmacokinetics of Subcutaneous Ondansetron in Healthy Geriatric Cats, Cats with Chronic Kidney Disease, and Cats with Liver Disease. J. Vet. Pharmacol. Therap. 2016, 39, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Quimby, J.M.; Gustafson, D.L.; Lunn, K.F. The Pharmacokinetics of Mirtazapine in Cats with Chronic Kidney Disease and In Age-Matched Control Cats. J. Vet. Intern. Med. 2011, 25, 985–989. [Google Scholar] [CrossRef]
- Ferguson, L.E.; McLean, M.K.; Bates, J.A.; Quimby, J.M. Mirtazapine Toxicity in Cats: Retrospective Study of 84 Cases (2006–2011). J. Feline Med. Surg. 2016, 18, 868–874. [Google Scholar] [CrossRef]
- Benson, K.K.; Zajic, L.B.; Morgan, P.K.; Brown, S.R.; Hansen, R.J.; Lunghofer, P.J.; Wittenburg, L.A.; Gustafson, D.L.; Quimby, J.M. Drug Exposure and Clinical Effect of Transdermal Mirtazapine in Healthy Young Cats: A Pilot Study. J. Feline Med. Surg. 2017, 19, 998–1006. [Google Scholar] [CrossRef]
- Giorgi, M.; Yun, H. Pharmacokinetics of Mirtazapine and Its Main Metabolites in Beagle Dogs: A Pilot Study. Vet. J. 2012, 192, 239–241. [Google Scholar] [CrossRef] [PubMed]
- Bianciardi, P.; Brovida, C.; Valente, M.; Aresu, L.; Cavicchioli, L.; Vischer, C.; Giroud, L.; Castagnaro, M. Administration of Miltefosine and Meglumine Antimoniate in Healthy Dogs: Clinicopathological Evaluation of the Impact on the Kidneys. Toxicol. Pathol. 2009, 37, 770–775. [Google Scholar] [CrossRef] [PubMed]
- Daza González, M.A.; Miró, G.; Fermín Rodríguez, M.; Rupérez Noguer, C.; Fragío Arnold, C. Short Term Impacts of Meglumine Antimoniate Treatment on Kidney Function in Dogs with Clinical Leishmaniosis. Res. Vet. Sci. 2019, 126, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Kasabalis, D.; Chatzis, M.K.; Apostolidis, K.; Petanides, T.; Athanasiou, L.V.; Xenoulis, P.G.; Mataragka, A.; Ikonomopoulos, J.; Leontides, L.S.; Saridomichelakis, M.N. A Randomized, Blinded, Controlled Clinical Trial Comparing the Efficacy of Aminosidine (Paromomycin)-Allopurinol Combination with the Efficacy of Meglumine Antimoniate-Allopurinol Combination for the Treatment of Canine Leishmaniosis Due to Leishmania Infantum. Exp. Parasitol. 2020, 214, 107903. [Google Scholar] [CrossRef]
- Woerly, V.; Maynard, L.; Sanquer, A.; Eun, H.-M. Clinical Efficacy and Tolerance of Miltefosine in the Treatment of Canine Leishmaniosis. Parasitol. Res. 2009, 105, 463. [Google Scholar] [CrossRef]
- Manna, L.; Vitale, F.; Reale, S.; Picillo, E.; Neglia, G.; Vescio, F.; Gravino, A.E. Study of Efficacy of Miltefosine and Allopurinol in Dogs with Leishmaniosis. Vet. J. 2009, 182, 441–445. [Google Scholar] [CrossRef]
- Mateo, M.; Maynard, L.; Vischer, C.; Bianciardi, P.; Miró, G. Comparative Study on the Short Term Efficacy and Adverse Effects of Miltefosine and Meglumine Antimoniate in Dogs with Natural Leishmaniosis. Parasitol. Res. 2009, 105, 155. [Google Scholar] [CrossRef]
- Andrade, H.M.; Toledo, V.P.C.P.; Pinheiro, M.B.; Guimarães, T.M.P.D.; Oliveira, N.C.; Castro, J.A.; Silva, R.N.; Amorim, A.C.; Brandão, R.M.S.S.; Yoko, M.; et al. Evaluation of Miltefosine for the Treatment of Dogs Naturally Infected with L. infantum (=L. chagasi) in Brazil. Vet. Parasitol. 2011, 181, 83–90. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Iarussi, F.; Paradies, P.; Foglia Manzillo, V.; Gizzarelli, M.; Caratozzolo, M.F.; Navarro, C.; Greco, B.; Rubino, G.T.R.; Oliva, G.; Sasanelli, M. Comparison of Two Dosing Regimens of Miltefosine, Both in Combination With Allopurinol, on Clinical and Parasitological Findings of Dogs With Leishmaniosis: A Pilot Study. Front. Vet. Sci. 2020, 7, 577395. [Google Scholar] [CrossRef] [PubMed]
- Roura, X.; Cortadellas, O.; Day, M.J.; Benali, S.L.; Zatelli, A.; Canine Leishmaniosis Working Group; D’Anna, N.; Fondati, A.; Gradoni, L.; Lubas, G.; et al. Canine Leishmaniosis and Kidney Disease: Q&A for an Overall Management in Clinical Practice. J. Small Anim. Pr. 2021, 62, 3. [Google Scholar] [CrossRef]
- Pennisi, M.G.; Persichetti, M.F. Feline Leishmaniosis: Is the Cat a Small Dog? Vet. Parasitol. 2018, 251, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Dorsch, R.; Teichmann-Knorrn, S.; Sjetne Lund, H. Urinary Tract Infection and Subclinical Bacteriuria in Cats: A Clinical Update. J. Feline Med. Surg. 2019, 21, 1023–1038. [Google Scholar] [CrossRef]
- Hindar, C.; Chang, Y.; Syme, H.M.; Jepson, R.E. The Association of Bacteriuria with Survival and Disease Progression in Cats with Azotemic Chronic Kidney Disease. J. Vet. Intern. Med. 2020, 34, 2516–2524. [Google Scholar] [CrossRef]
- Lamoureux, A.; Da Riz, F.; Cappelle, J.; Boulouis, H.; Benchekroun, G.; Cadoré, J.; Krafft, E.; Maurey, C. Frequency of Bacteriuria in Dogs with Chronic Kidney Disease: A Retrospective Study of 201 Cases. J. Vet. Intern. Med. 2019, 33, 640–647. [Google Scholar] [CrossRef] [PubMed]
- St Peter, W.L.; Redic-Kill, K.A.; Halstenson, C.E. Clinical Pharmacokinetics of Antibiotics in Patients with Impaired Renal Function. Clin. Pharmacokinet. 1992, 22, 169–210. [Google Scholar] [CrossRef]
- Swan, S.K. Aminoglycoside Nephrotoxicity. Semin. Nephrol. 1997, 17, 27–33. [Google Scholar]
- Greene, C.E.; Boothe, D.M. Antimicrobial Chemotherapy. In Infectious Diseases of the Dog and Cat; Elsevier/Saunders: St. Louis, Mo, USA, 2012; ISBN 978-1-4160-6130-4. [Google Scholar]
- Yeary, R.A. Systemic Toxic Effects of Chemotherapeutic Agents in Domestic Animals. Vet. Clin. N. Am. 1975, 5, 51–69. [Google Scholar] [CrossRef]
- Beaucaire, G. Does Once-Daily Dosing Prevent Nephrotoxicity in All Aminoglycosides Equally? Clin. Microbiol. Infect. 2000, 6, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, L.S.; Spencer, J.P. Aminoglycosides: A Practical Review. Am. Fam. Physician 1998, 58, 1811–1820. [Google Scholar]
- Albarellos, G.; Montoya, L.; Ambros, L.; Kreil, V.; Hallu, R.; Rebuelto, M. Multiple Once-Daily Dose Pharmacokinetics and Renal Safety of Gentamicin in Dogs. J. Vet. Pharm. 2004, 27, 21–25. [Google Scholar] [CrossRef]
- Avent, M.L.; Rogers, B.A.; Cheng, A.C.; Paterson, D.L. Current Use of Aminoglycosides: Indications, Pharmacokinetics and Monitoring for Toxicity: Aminoglycosides: Review and Monitoring. Intern. Med. J. 2011, 41, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Pagkalis, S.; Mantadakis, E.; Mavros, M.N.; Ammari, C.; Falagas, M.E. Pharmacological Considerations for the Proper Clinical Use of Aminoglycosides. Drugs 2011, 71, 2277–2294. [Google Scholar] [CrossRef] [PubMed]
- Riviere, J.E.; Carver, M.P.; Coppoc, G.L.; Carlton, W.W.; Lantz, G.C.; Shy-Modjeska, J. Pharmacokinetics and Comparative Nephrotoxicity of Fixed-Dose versus Fixed-Interval Reduction of Gentamicin Dosage in Subtotal Nephrectomized Dogs. Toxicol. Appl. Pharmacol. 1984, 75, 496–509. [Google Scholar] [CrossRef]
- IRIS Kidney—Guidelines—Preventing Aminoglycoside-Induced AKI. Available online: http://www.iris-kidney.com/education/prevention.html (accessed on 30 September 2021).
- Papich, M.G. Table of Common Drugs: Approximate Dosages. In Kirk’s Current Veterinary Therapy XIV; Saunders/Elsevier: Philadelphia, PA, USA, 2008; ISBN 978-1-4377-1152-3. [Google Scholar]
- Horber, F.F.; Frey, F.J.; Descoeudres, C.; Murray, A.T.; Reubi, F.C. Differential Effect of Impaired Renal Function on the Kinetics of Clavulanic Acid and Amoxicillin. Antimicrob. Agents Chemother. 1986, 29, 614–619. [Google Scholar] [CrossRef]
- Caprile, K.A. The Cephalosporin Antimicrobial Agents: A Comprehensive Review. J. Vet. Pharmacol. Ther. 1988, 11, 1–32. [Google Scholar] [CrossRef]
- Klepser, M.E.; Marangos, M.N.; Patel, K.B.; Nicolau, D.P.; Quintiliani, R.; Nightingale, C.H. Clinical Pharmacokinetics of Newer Cephalosporins. Clin. Pharmacokinet. 1995, 28, 361–384. [Google Scholar] [CrossRef]
- Trepanier, L.A. Delayed Hypersensitivity Reactions to Sulphonamides: Syndromes, Pathogenesis and Management. Vet. Derm. 1999, 10, 241–248. [Google Scholar] [CrossRef]
- Trepanier, L.A.; Danhof, R.; Toll, J.; Watrous, D. Clinical Findings in 40 Dogs with Hypersensitivity Associated with Administration of Potentiated Sulfonamides. J. Vet. Intern. Med. 2003, 17, 647–652. [Google Scholar] [CrossRef]
- Giger, U.; Werner, L.L.; Millichamp, N.J.; Gorman, N.T. Sulfadiazine-Induced Allergy in Six Doberman Pinschers. J. Am. Vet. Med. Assoc. 1985, 186, 479–484. [Google Scholar]
- Cribb, A.E. Idiosyncratic Reactions to Sulfonamides in Dogs. J. Am. Vet. Med. Assoc. 1989, 195, 1612–1614. [Google Scholar] [PubMed]
- Cribb, A.E.; Spielberg, S.P. An in Vitro Investigation of Predisposition to Sulphonamide Idiosyncratic Toxicity in Dogs. Vet. Res. Commun. 1990, 14, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Hadrick, M.K.; Vaden, S.L.; Geoly, F.J.; Cullen, J.M.; Douglass, J.P. Acute Tubulointerstitial Nephritis with Eosinophiluria in a Dog. J. Vet. Intern. Med. 1996, 10, 45–47. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, H.P.; Schneider, M.; Dupouy, V.; Laroute, V.; Costes, G.; Delesalle, L.; Toutain, P.L. Effect of Experimental Renal Impairment on Disposition of Marbofloxacin and Its Metabolites in the Dog. J. Vet. Pharmacol. Ther. 1998, 21, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Mathews, K.A. Nonsteroidal Anti-Inflammatory Analgesics. Vet. Clin. N. Am. Small Anim. Pract. 2000, 30, 783–804. [Google Scholar] [CrossRef]
- Mathews, K.; Kronen, P.W.; Lascelles, D.; Nolan, A.; Robertson, S.; Steagall, P.V.; Wright, B.; Yamashita, K. Guidelines for Recognition, Assessment and Treatment of Pain: WSAVA Global Pain Council Members and Co-Authors of This Document. J. Small Anim. Pr. 2014, 55, E10–E68. [Google Scholar] [CrossRef]
- Hampshire, V.A.; Doddy, F.M.; Post, L.O.; Koogler, T.L.; Burgess, T.M.; Batten, P.O.; Hudson, R.; McAdams, D.R.; Brown, M.A. Adverse Drug Event Reports at the United States Food and Drug Administration Center for Veterinary Medicine. J. Am. Vet. Med. Assoc. 2004, 225, 533–536. [Google Scholar] [CrossRef]
- Lascelles, B.D.X.; McFarland, J.M.; Swann, H. Guidelines for Safe and Effective Use of NSAIDs in Dogs. Vet. Ther. 2005, 6, 237–251. [Google Scholar]
- Lascelles, B.D.X.; Court, M.H.; Hardie, E.M.; Robertson, S.A. Nonsteroidal Anti-Inflammatory Drugs in Cats: A Review. Vet. Anaesth. Analg. 2007, 34, 228–250. [Google Scholar] [CrossRef]
- Launay-Vacher, V.; Karie, S.; Fau, J.-B.; Izzedine, H.; Deray, G. Treatment of Pain in Patients with Renal Insufficiency: The World Health Organization Three-Step Ladder Adapted. J. Pain 2005, 6, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Inker, L.A.; Astor, B.C.; Fox, C.H.; Isakova, T.; Lash, J.P.; Peralta, C.A.; Kurella Tamura, M.; Feldman, H.I. KDOQI US Commentary on the 2012 KDIGO Clinical Practice Guideline for the Evaluation and Management of CKD. Am. J. Kidney Dis. 2014, 63, 713–735. [Google Scholar] [CrossRef] [PubMed]
- Baker, M.; Perazella, M.A. NSAIDs in CKD: Are They Safe? Am. J. Kidney Dis. 2020, 76, 546–557. [Google Scholar] [CrossRef] [PubMed]
- Kongara, K.; Cave, N.; Weidgraaf, K.; Rao Dukkipati, V.S. Effect of Non-Steroidal Anti-Inflammatory Drugs on Glomerular Filtration Rate and Urinary N-Acetyl-β-D-Glucosaminidase Activity in Cats after Dental Surgery. Vet. Anaesth. Analg. 2020, 47, 631–636. [Google Scholar] [CrossRef]
- Khan, K.N.M.; Venturini, C.M.; Bunch, R.T.; Brassard, J.A.; Koki, A.T.; Morris, D.L.; Trump, B.E.; Maziasz, T.J.; Alden, C.L. Interspecies Differences in Renal Localization of Cyclooxygenase Isoforms: Implications in Nonsteroidal Antiinflammatory Drug-Related Nephrotoxicity. Toxicol. Pathol. 1998, 26, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Papich, M.G. Pharmacologic Considerations for Opiate Analgesic and Nonsteroidal Anti-Inflammatory Drugs. Vet. Clin. N. Am. Small Anim. Pract. 2000, 30, 815–837. [Google Scholar] [CrossRef]
- Horii, Y.; Ikenaga, M.; Shimoda, M.; Kokue, E. Pharmacokinetics of Flunixin in the Cat: Enterohepatic Circulation and Active Transport Mechanism in the Liver. J. Vet. Pharm. 2004, 27, 65–69. [Google Scholar] [CrossRef]
- Castro, E.; Soraci, A.; Fogel, F.; Tapia, O. Chiral Inversion of R(-) Fenoprofen and Ketoprofen Enantiomers in Cats. J. Vet. Pharm. 2000, 23, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, B.; Steagall, P.V.M.; Lascelles, B.D.X.; Robertson, S.; Murrell, J.C.; Kronen, P.W.; Wright, B.; Yamashita, K. Long-Term Use of Non-Steroidal Anti-Inflammatory Drugs in Cats with Chronic Kidney Disease: From Controversy to Optimism. J. Small Anim. Pract. 2019, 60, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Pages, J.-P. Néphropathies Dues Aux Anti-Inflammatoires Non Stéroïdiens [AINS] Chez Le Chat: 21 Observations (1993–2001). Prat. Méd. Chir. L’animal Cie 2005, 40, 177–181. [Google Scholar]
- Gunew, M.N.; Menrath, V.H.; Marshall, R.D. Long-Term Safety, Efficacy and Palatability of Oral Meloxicam at 0.01–0.03 Mg/Kg for Treatment of Osteoarthritic Pain in Cats. J. Feline Med. Surg. 2008, 10, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Gowan, R.A.; Lingard, A.E.; Johnston, L.; Stansen, W.; Brown, S.A.; Malik, R. Retrospective Case—Control Study of the Effects of Long-Term Dosing with Meloxicam on Renal Function in Aged Cats with Degenerative Joint Disease. J. Feline Med. Surg. 2011, 13, 752–761. [Google Scholar] [CrossRef]
- Gowan, R.A.; Baral, R.M.; Lingard, A.E.; Catt, M.J.; Stansen, W.; Johnston, L.; Malik, R. A Retrospective Analysis of the Effects of Meloxicam on the Longevity of Aged Cats with and without Overt Chronic Kidney Disease. J. Feline Med. Surg. 2012, 14, 876–881. [Google Scholar] [CrossRef]
- King, J.N.; King, S.; Budsberg, S.C.; Lascelles, B.D.X.; Bienhoff, S.E.; Roycroft, L.M.; Roberts, E.S. Clinical Safety of Robenacoxib in Feline Osteoarthritis: Results of a Randomized, Blinded, Placebo-Controlled Clinical Trial. J. Feline Med. Surg. 2016, 18, 632–642. [Google Scholar] [CrossRef]
- Surdyk, K.K.; Brown, C.A.; Brown, S.A. Evaluation of Glomerular Filtration Rate in Cats with Reduced Renal Mass and Administered Meloxicam and Acetylsalicylic Acid. Am. J. Vet. Res. 2013, 74, 648–651. [Google Scholar] [CrossRef]
- KuKanich, K.; George, C.; Roush, J.K.; Sharp, S.; Farace, G.; Yerramilli, M.; Peterson, S.; Grauer, G.F. Effects of Low-Dose Meloxicam in Cats with Chronic Kidney Disease. J. Feline Med. Surg. 2021, 23, 138–148. [Google Scholar] [CrossRef]
- Surdyk, K.K.; Sloan, D.L.; Brown, S.A. Evaluation of the Renal Effects of Ibuprofen and Carprofen in Euvolemic and Volume-Depleted Dogs. Int. J. Appl. Res. Vet. Med. 2011, 9, 130. [Google Scholar]
- Surdyk, K.K.; Sloan, D.L.; Brown, S.A. Renal Effects of Carprofen and Etodolac in Euvolemic and Volume-Depleted Dogs. Am. J. Vet. Res. 2012, 73, 1485–1490. [Google Scholar] [CrossRef]
- KuKanich, B.; Bidgood, T.; Knesl, O. Clinical Pharmacology of Nonsteroidal Anti-Inflammatory Drugs in Dogs. Vet. Anaesth. Analg. 2012, 39, 69–90. [Google Scholar] [CrossRef]
- Nine Ways to Minimize the Risks of Nonsteroidal Anti-Inflammatory Drugs (NSAIDs). Available online: https://www.aaha.org/aaha-guidelines/pain-management-config/implementation-toolkit/nine-ways-to-minimize-the-risks-of-nonsteroidal-anti-inflammatory-drugs-nsaids/ (accessed on 30 September 2021).
- Smets, P.M.Y.; Lefebvre, H.P.; Aresu, L.; Croubels, S.; Haers, H.; Piron, K.; Meyer, E.; Daminet, S. Renal Function and Morphology in Aged Beagle Dogs Before and after Hydrocortisone Administration. PLoS ONE 2012, 7, e31702. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, J.; Soblechero, P.; Duque, F.J.; Macías-García, B.; Ruiz, P.; Zaragoza, C.; Barrera, R. Effects of Oral Prednisone Administration on Serum Cystatin C in Dogs. J. Vet. Intern. Med. 2017, 31, 1765–1770. [Google Scholar] [CrossRef]
- IRIS Canine GN Study Group Established Pathology Subgroup; Segev, G.; Cowgill, L.D.; Heiene, R.; Labato, M.A.; Polzin, D.J. Consensus Recommendations for Immunosuppressive Treatment of Dogs with Glomerular Disease Based on Established Pathology. J. Vet. Intern. Med. 2013, 27, S44–S54. [Google Scholar] [CrossRef]
- Elliott, J.; Barber, P.J.; Syme, H.M.; Rawlings, J.M.; Markwell, P.J. Feline Hypertension: Clinical Findings and Response to Antihypertensive Treatment in 30 Cases. J. Small Anim. Pr. 2001, 42, 122–129. [Google Scholar] [CrossRef]
- Jepson, R.E.; Brodbelt, D.; Vallance, C.; Syme, H.M.; Elliott, J. Evaluation of Predictors of the Development of Azotemia in Cats. J. Vet. Intern. Med. 2009, 23, 806–813. [Google Scholar] [CrossRef] [PubMed]
- Schmieder, R. Mechanisms for the Clinical Benefits of Angiotensin II Receptor Blockers. Am. J. Hypertens. 2005, 18, 720–730. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, H.; Brown, S.; Chetboul, V.; King, J.; Pouchelon, J.-L.; Toutain, P. Angiotensin-Converting Enzyme Inhibitors in Veterinary Medicine. CPD 2007, 13, 1347–1361. [Google Scholar] [CrossRef] [PubMed]
- Grauer, G.F.; Greco, D.S.; Getzy, D.M.; Cowgill, L.D.; Vaden, S.L.; Chew, D.J.; Polzin, D.J.; Barsanti, J.A. Effects of Enalapril versus Placebo as a Treatment for Canine Idiopathic Glomerulonephritis. J. Vet. Intern. Med. 2000, 14, 526–533. [Google Scholar] [CrossRef] [PubMed]
- King, J.N.; Gunn-Moore, D.A.; Tasker, S.; Gleadhill, A.; Strehlau, G. Tolerability and Efficacy of Benazepril in Cats with Chronic Kidney Disease. J. Vet. Intern. Med. 2006, 20, 1054–1064. [Google Scholar] [CrossRef] [PubMed]
- Mizutani, H.; Koyama, H.; Watanabe, T.; Kitagawa, H.; Nakano, M.; Kajiwara, K.; King, J.N. Evaluation of the Clinical Efficacy of Benazepril in the Treatment of Chronic Renal Insufficiency in Cats. J. Vet. Intern. Med. 2006, 20, 1074–1079. [Google Scholar] [CrossRef] [PubMed]
- Tenhündfeld, J.; Wefstaedt, P.; Nolte, I.J.A. A Randomized Controlled Clinical Trial of the Use of Benazepril and Heparin for the Treatment of Chronic Kidney Disease in Dogs. J. Am. Vet. Med. Assoc. 2009, 234, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Harley, L.; Langston, C. Proteinuria in Dogs and Cats. Can. Vet. J. 2012, 53, 631–638. [Google Scholar]
- Toutain, P.L.; Lefebvre, H.P. Pharmacokinetics and Pharmacokinetic/Pharmacodynamic Relationships for Angiotensin-Converting Enzyme Inhibitors. J. Vet. Pharm. 2004, 27, 515–525. [Google Scholar] [CrossRef] [PubMed]
- King, J.N.; Strehlau, G.; Wernsing, J.; Brown, S.A. Effect of Renal Insufficiency on the Pharmacokinetics and Pharmacodynamics of Benazepril in Cats. J. Vet. Pharm. 2002, 25, 371–378. [Google Scholar] [CrossRef]
- Lefebvre, H.P.; Laroute, V.; Concordet, D.; Toutain, P.L. Effects of Renal Impairment on the Disposition of Orally Administered Enalapril, Benazepril, and Their Active Metabolites. J. Vet. Intern. Med. 1999, 13, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.L.; Coleman, A.E.; Schmiedt, C.W.; Brown, S.A. Attenuation of the Pressor Response to Exogenous Angiotensin by Angiotensin Receptor Blockers and Benazepril Hydrochloride in Clinically Normal Cats. Am. J. Vet. Res. 2015, 76, 807–813. [Google Scholar] [CrossRef]
- Tissier, R.; Perrot, S.; Enriquez, B. Amlodipine: One of the Main Anti-Hypertensive Drugs in Veterinary Therapeutics. J. Vet. Cardiol. 2005, 7, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Pouchelon, J.L.; Atkins, C.E.; Bussadori, C.; Oyama, M.A.; Vaden, S.L.; Bonagura, J.D.; Chetboul, V.; Cowgill, L.D.; Elliot, J.; Francey, T.; et al. Cardiovascular–Renal Axis Disorders in the Domestic Dog and Cat: A Veterinary Consensus Statement. J. Small Anim. Pr. 2015, 56, 537–552. [Google Scholar] [CrossRef]
- IRIS Canine GN Study Group Standard Therapy Subgroup; Brown, S.; Elliott, J.; Francey, T.; Polzin, D.; Vaden, S. Consensus Recommendations for Standard Therapy of Glomerular Disease in Dogs. J. Vet. Intern. Med. 2013, 27, S27–S43. [Google Scholar] [CrossRef] [PubMed]
- Lennon, E.M.; Hanel, R.M.; Walker, J.M.; Vaden, S.L. Hypercoagulability in Dogs with Protein-Losing Nephropathy as Assessed by Thromboelastography. J. Vet. Intern. Med. 2013, 27, 462–468. [Google Scholar] [CrossRef] [PubMed]
- de Laforcade, A.; Bacek, L.; Blais, M.-C.; Goggs, R.; Lynch, A.; Rozanski, E. Consensus on the Rational Use of Antithrombotics in Veterinary Critical Care (CURATIVE): Domain 1-Defining Populations at Risk. J. Vet. Emerg. Crit. Care 2019, 29, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Geesaman, B.; Bach, J.; Monaghan, K. Heparin-Induced Hyperkalemia in a Dog Receiving Continuous Renal Replacement Treatment. J. Vet. Intern. Med. 2015, 29, 1629–1632. [Google Scholar] [CrossRef][Green Version]
- Rayhel, L.H.; Quimby, J.M.; Cianciolo, R.E.; Cléroux, A.; McLeland, S.M.; Franken, T. Clinicopathologic and Pathologic Characteristics of Feline Proteinuric Kidney Disease. J. Feline Med. Surg. 2020, 22, 1219–1229. [Google Scholar] [CrossRef]
- Hogan, D.F.; Fox, P.R.; Jacob, K.; Keene, B.; Laste, N.J.; Rosenthal, S.; Sederquist, K.; Weng, H.-Y. Secondary Prevention of Cardiogenic Arterial Thromboembolism in the Cat: The Double-Blind, Randomized, Positive-Controlled Feline Arterial Thromboembolism; Clopidogrel vs. Aspirin Trial (FAT CAT). J. Vet. Cardiol. 2015, 17, S306–S317. [Google Scholar] [CrossRef]
- Giustino, G.; Baber, U.; Sartori, S.; Mehran, R.; Mastoris, I.; Kini, A.S.; Sharma, S.K.; Pocock, S.J.; Dangas, G.D. Duration of Dual Antiplatelet Therapy After Drug-Eluting Stent Implantation. J. Am. Coll. Cardiol. 2015, 65, 1298–1310. [Google Scholar] [CrossRef]
- Maton, P.N.; Burton, M.E. Antacids Revisited: A Review of Their Clinical Pharmacology and Recommended Therapeutic Use. Drugs 1999, 57, 855–870. [Google Scholar] [CrossRef]
- Segev, G.; Bandt, C.; Francey, T.; Cowgill, L.D. Aluminum Toxicity Following Administration of Aluminum-Based Phosphate Binders in 2 Dogs with Renal Failure. J. Vet. Intern. Med. 2008, 22, 1432–1435. [Google Scholar] [CrossRef]
- Xie, Y.; Bowe, B.; Li, T.; Xian, H.; Balasubramanian, S.; Al-Aly, Z. Proton Pump Inhibitors and Risk of Incident CKD and Progression to ESRD. JASN 2016, 27, 3153–3163. [Google Scholar] [CrossRef]
- Schubert, M.L. Adverse Effects of Proton Pump Inhibitors: Fact or Fake News? Curr. Opin. Gastroenterol. 2018, 34, 451–457. [Google Scholar] [CrossRef]
- Nagashima, R. Mechanisms of Action of Sucralfate. J. Clin. Gastroenterol. 1981, 3, 117–127. [Google Scholar] [PubMed]
- Leung, A.C.; Henderson, I.S.; Halls, D.J.; Dobbie, J.W. Aluminium Hydroxide versus Sucralfate as a Phosphate Binder in Uraemia. BMJ 1983, 286, 1379–1381. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.A. Sucralfate, Intestinal Aluminum Absorption, and Aluminum Toxicity in a Patient on Dialysis. Ann. Intern. Med. 1989, 111, 179. [Google Scholar] [CrossRef]
- Conder, G.A.; Sedlacek, H.S.; Boucher, J.F.; Clemence, R.G. Efficacy and Safety of Maropitant, a Selective Neurokinin 1 Receptor Antagonist, in Two Randomized Clinical Trials for Prevention of Vomiting Due to Motion Sickness in Dogs. J. Vet. Pharmacol. Ther. 2008, 31, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Benchaoui, H.A.; Cox, S.R.; Schneider, R.P.; Boucher, J.F.; Clemence, R.G. The Pharmacokinetics of Maropitant, a Novel Neurokinin Type-1 Receptor Antagonist, in Dogs. J. Vet. Pharm. 2007, 30, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Hickman, M.A.; Cox, S.R.; Mahabir, S.; Miskell, C.; Lin, J.; Bunger, A.; McCALL, R.B. Safety, Pharmacokinetics and Use of the Novel NK-1 Receptor Antagonist Maropitant (CereniaTM ) for the Prevention of Emesis and Motion Sickness in Cats. J. Vet. Pharm. 2008, 31, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Quimby, J.M.; Lake, R.C.; Hansen, R.J.; Lunghofer, P.J.; Gustafson, D.L. Oral, Subcutaneous, and Intravenous Pharmacokinetics of Ondansetron in Healthy Cats. J. Vet. Pharmacol. Therap. 2014, 37, 348–353. [Google Scholar] [CrossRef]
- Quimby, J.M.; Lunn, K.F. Mirtazapine as an Appetite Stimulant and Anti-Emetic in Cats with Chronic Kidney Disease: A Masked Placebo-Controlled Crossover Clinical Trial. Vet. J. 2013, 197, 651–655. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Deng, C.; Huang, X.-F. The Role of Hypothalamic H1 Receptor Antagonism in Antipsychotic-Induced Weight Gain. CNS Drugs 2013, 27, 423–434. [Google Scholar] [CrossRef]
- Schellekens, H.; De Francesco, P.N.; Kandil, D.; Theeuwes, W.F.; McCarthy, T.; van Oeffelen, W.E.P.A.; Perelló, M.; Giblin, L.; Dinan, T.G.; Cryan, J.F. Ghrelin’s Orexigenic Effect Is Modulated via a Serotonin 2C Receptor Interaction. ACS Chem. Neurosci. 2015, 6, 1186–1197. [Google Scholar] [CrossRef]
- Quimby, J.M.; Gustafson, D.L.; Samber, B.J.; Lunn, K.F. Studies on the Pharmacokinetics and Pharmacodynamics of Mirtazapine in Healthy Young Cats: Mirtazapine Pharmacokinetics and Pharmacodynamics in Cats. J. Vet. Pharmacol. Ther. 2011, 34, 388–396. [Google Scholar] [CrossRef]
- Fantinati, M.; Trnka, J.; Signor, A.; Dumond, S.; Jourdan, G.; Verwaerde, P.; Priymenko, N. Appetite-Stimulating Effect of Gabapentin vs Mirtazapine in Healthy Cats Post-Ovariectomy. J. Feline Med. Surg. 2020, 22, 1176–1183. [Google Scholar] [CrossRef]
- Quimby, J.M.; Benson, K.K.; Summers, S.C.; Saffire, A.; Herndon, A.K.; Bai, S.; Gustafson, D.L. Assessment of Compounded Transdermal Mirtazapine as an Appetite Stimulant in Cats with Chronic Kidney Disease. J. Feline Med. Surg. 2020, 22, 376–383. [Google Scholar] [CrossRef]
- Fitzpatrick, R.L.; Quimby, J.M.; Benson, K.K.; Ramirez, D.; Sieberg, L.G.; Wittenburg, L.A.; Gustafson, D.L. In Vivo and in Vitro Assessment of Mirtazapine Pharmacokinetics in Cats with Liver Disease. J. Vet. Intern. Med. 2018, 32, 1951–1957. [Google Scholar] [CrossRef] [PubMed]
- Buhles, W.; Quimby, J.M.; Labelle, D.; Williams, V.S. Single and Multiple Dose Pharmacokinetics of a Novel Mirtazapine Transdermal Ointment in Cats. J. Vet. Pharm. Ther. 2018, 41, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Costa, F.A.L.; Goto, H.; Saldanha, L.C.B.; Silva, S.M.M.S.; Sinhorini, I.L.; Silva, T.C.; Guerra, J.L. Histopathologic Patterns of Nephropathy in Naturally Acquired Canine Visceral Leishmaniasis. Vet. Pathol. 2003, 40, 677–684. [Google Scholar] [CrossRef]
- Zatelli, A.; Borgarelli, M.; Santilli, R.; Bonfanti, U.; Nigrisoli, E.; Zanatta, R.; Tarducci, A.; Guarraci, A. Glomerular Lesions in Dogs Infected with Leishmania Organisms. Am. J. Vet. Res. 2003, 64, 558–561. [Google Scholar] [CrossRef] [PubMed]
- Aresu, L.; Benali, S.; Ferro, S.; Vittone, V.; Gallo, E.; Brovida, C.; Castagnaro, M. Light and Electron Microscopic Analysis of Consecutive Renal Biopsy Specimens From Leishmania-Seropositive Dogs. Vet. Pathol. 2013, 50, 753–760. [Google Scholar] [CrossRef]
- Koutinas, A.F.; Koutinas, C.K. Pathologic Mechanisms Underlying the Clinical Findings in Canine Leishmaniosis Due to Leishmania Infantum/Chagasi. Vet. Pathol. 2014, 51, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Vaden, S.L. Glomerular Disease. In Nephrology and Urology of Small Animals; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2011; ISBN 978-0-8138-1717-0. [Google Scholar]
- Oliva, G.; Dvm, X.R.; Crotti, A.; Maroli, M.; Castagnaro, M.; Gradoni, L.; Lubas, G.; Paltrinieri, S.; Zatelli, A.; Zini, E. Guidelines for Treatment of Leishmaniasis in Dogs. J. Am. Vet. Med. Assoc. 2010, 236, 1192–1198. [Google Scholar] [CrossRef] [PubMed]
- Solano-Gallego, L.; Miró, G.; Koutinas, A.; Cardoso, L.; Pennisi, M.G.; Ferrer, L.; Bourdeau, P.; Oliva, G.; Baneth, G. LeishVet Guidelines for the Practical Management of Canine Leishmaniosis. Parasites Vectors 2011, 4, 86. [Google Scholar] [CrossRef]
- Plevraki, K.; Koutinas, A.F.; Kaldrymidou, H.; Roumpies, N.; Papazoglou, L.G.; Saridomichelakis, M.N.; Savvas, I.; Leondides, L. Effects of Allopurinol Treatment on the Progression of Chronic Nephritis in Canine Leishmaniosis (Leishmania infantum). J. Vet. Intern. Med. 2006, 20, 228–233. [Google Scholar] [CrossRef]
- Pierantozzi, M.; Roura, X.; Paltrinieri, S.; Poggi, M.; Zatelli, A. Variation of Proteinuria in Dogs with Leishmaniasis Treated with Meglumine Antimoniate and Allopurinol: A Retrospective Study. J. Am. Anim. Hosp. Assoc. 2013, 49, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Proverbio, D.; Spada, E.; de Giorgi, G.B.; Perego, R. Proteinuria Reduction after Treatment with Miltefosine and Allopurinol in Dogs Naturally Infected with Leishmaniasis. Vet. World 2016, 9, 904–908. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.; Pastor, J.; Roura, X.; Tabar, M.D.; Espada, Y.; Font, A.; Balasch, J.; Planellas, M. Adverse Urinary Effects of Allopurinol in Dogs with Leishmaniasis: Urinary Adverse Effects of Allopurinol. J. Small Anim. Pr. 2016, 57, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Paltrinieri, S.; Solano-Gallego, L.; Fondati, A.; Lubas, G.; Gradoni, L.; Castagnaro, M.; Crotti, A.; Maroli, M.; Oliva, G.; Roura, X.; et al. Guidelines for Diagnosis and Clinical Classification of Leishmaniasis in Dogs. J. Am. Vet. Med. Assoc. 2010, 236, 1184–1191. [Google Scholar] [CrossRef]
- Pennisi, M.G.; Hartmann, K.; Lloret, A.; Addie, D.; Belák, S.; Boucraut-Baralon, C.; Egberink, H.; Frymus, T.; Gruffydd-Jones, T.; Hosie, M.J.; et al. Leishmaniosis in Cats: ABCD Guidelines on Prevention and Management. J. Feline Med. Surg. 2013, 15, 638–642. [Google Scholar] [CrossRef]
- Fernandez-Gallego, A.; Feo Bernabe, L.; Dalmau, A.; Esteban-Saltiveri, D.; Font, A.; Leiva, M.; Ortuñez-Navarro, A.; Peña, M.-T.; Tabar, M.-D.; Real-Sampietro, L.; et al. Feline Leishmaniosis: Diagnosis, Treatment and Outcome in 16 Cats. J. Feline Med. Surg. 2020, 22, 993–1007. [Google Scholar] [CrossRef]
- Pennisi, M.-G.; Cardoso, L.; Baneth, G.; Bourdeau, P.; Koutinas, A.; Miró, G.; Oliva, G.; Solano-Gallego, L. LeishVet Update and Recommendations on Feline Leishmaniosis. Parasites Vectors 2015, 8, 302. [Google Scholar] [CrossRef]
| Class | Drug | Comments |
|---|---|---|
| Antimicrobial agents | Aminoglycosides | Potentially nephrotoxic. Their use is generally discouraged in CKD patients [55]. When clinically indicated, the patients should be hemodynamically stable, and the concomitant use of other nephrotoxic drugs should be avoided. SDD regimen is recommended in association with TDM. The duration of treatment should be minimized (<7 days) [59]. |
| Penicillins | High therapeutic index [60]. Due to the possible accumulation, dose adjustment could be considered for ampicillin in dogs with severe azotemia [61]. Although penicillins have been classified as “probably safe” in cats with CKD [7,13], the need for dose adjustment in azotemic feline patients remains to be determined [62]. | |
| Cephalosporins | Dosage regimen adjustment based on an extension of the administration interval is recommended in humans with a moderate to severe reduction in GFR [63]; interval extension should be considered in dogs and cats with moderate or severe CKD [64]. | |
| Sulfonamides | Potential nephrotoxicity due to crystalluria and hypersensitivity reactions. Dehydration should be avoided [65]. In patients with reduced renal function dose adjustment could be considered (i.e., halving the dose or doubling the interval of administration) [64]. | |
| Tetracyclines | Increased risk of nephrotoxicity for water-soluble medications (e.g., oxytetracycline). In patients with altered renal function, doxycycline or minocycline should be preferred [60]. | |
| Fluoroquinolones | Undergo renal and non-renal elimination pathways. Dose adjustment may not be required in dogs [66]. Due to the high risk of retinopathy, an interval extension is recommended in CKD feline patients with moderate disease [7]. | |
| Anti-inflammatory drugs | NSAIDs | Potentially nephrotoxic [67,68]. When clinically indicated, the patients should be hemodynamically stable and the concomitant use of other nephrotoxic drugs should be avoided; close monitoring of renal function and administration of the lowest effective dose are recommended [7,69,70]. A washout period should be envisaged between different NSAIDs molecules and corticosteroids [69]. |
| Corticosteroids | Poor evidence of nephrotoxicity in dogs and cats. Possible worsening of azotemia and development of proteinuria reported in dogs [71]. | |
| Antiproteinuric and antihypertensive drugs | ACEis | Potentially nephrotoxic. Patients should be hemodynamically stabilized before administration and adoption of a lower starting dose and monitoring of renal function, SBP and serum electrolytes are recommended [72]. Caution should be used in an advanced stage of CKD and if administered in association with ARBs [73]. |
| ARBs | Potentially nephrotoxic. Telmisartan is safe and effective in reducing UPC and SBP in dogs and cats with CKD [74,75,76]. Caution should be used in advanced CKD stages and if administered in association with an ACEi [73]. | |
| CCBs | Amlodipine is a safe antihypertensive agent in cats with CKD [7,73,77]. Since CCBs preferentially dilate the renal afferent arteriole, exposing the glomerulus to increased hydrostatic pressure, their use as the sole antihypertensive drug in dogs is discouraged [73]. | |
| Diuretics | Furosemide | Potentially nephrotoxic due to excessive volume depletion. Contraindicated in patients with unstable renal function or volume depletion conditions; close monitoring of renal function is strongly recommended and association with other nephrotoxic drugs is discouraged [55,60]. Its use may be required in CRS [78]. |
| Antithrombotic agents | LMWHs | Risk of accumulation should be considered under conditions of reduced renal function [79]. Interval extension is recommended in humans with stage 4 CKD [80]. |
| Acetylsalicylic acid | Potentially dangerous in human CKD patients [81,82,83,84].No controlled clinical studies are available in animals [85]. Caution should be used under renal hypoperfusion conditions [86]. The concomitant administration of other NSAIDs should be avoided and renal function should be monitored. Prolonged time of excretion in cats [85]. | |
| Clopidogrel | Dose adjustments seem to not be required in humans with CKD [87,88]; however, the marked variability in pharmacokinetics of clopidogrel justifies its cautious use in patients with advanced CKD [80,88,89]; a lower antiplatelet effect has been observed in CKD patients [90]. Drug metabolism may not be altered in dogs with PLN and early CKD [91]. | |
| Gastroprotectants | Antacids | Although they generally lack systemic effects, the possible accumulation of aluminum and hypermagnesemia should be considered for aluminum/magnesium-containing salts in dogs with advanced renal failure [92]. No specific indications for dose adjustment are available in dogs and cats with CKD. |
| H2RAs | Dose adjustments recommended in humans [93]. Although no specific indications are available in dogs and cats [94], either decreasing the dose or extending the dosing interval can be used in IRIS Stages 3 and 4. | |
| PPIs | Nephrotoxicity (interstitial nephritis) reported in humans [95,96]. Poor evidence of nephrotoxicity in dogs and cats [94]. | |
| Sucralfate | Relatively safe compound. Caution should be used with long-term treatment in patients with renal insufficiency to avoid aluminum intoxication [94]. | |
| Antiemetics | Metoclopramide | Administration at standard constant rate infusion dosages (1–2 mg/kg/day) may cause tremors and ataxia in azotemic patients. The dose could be reduced by up to the 25–50% of the standard dose and titrate to dosage, which elicit a therapeutic effect without tremor [64]. |
| Maropitant | Safe in dogs and cats with renal impairment [97,98,99]. | |
| Ondansentron | Safe in cats with renal impairment [100]. No pharmacokinetic data are available for dogs. | |
| Appetite stimulants | Mirtazapine | 1.88 mg/cat q 24–48 h may be suitable as a starting dose in cats with CKD [101,102,103]. Limited data are available for dogs [104]. |
| Antileishmanial drugs | Meglumine antimoniate | Despite the limited evidence of nephrotoxicity, dose reduction and a monitoring of renal function are recommended [105,106,107]. Use in dogs with advanced renal failure is discouraged. Limited data on use in cats. |
| Miltefosine | Low impact on renal function in dogs [105,108,109,110,111,112]. Limited data on use in cats. | |
| Allopurinol | In cases of xantinuria, mineralization or uroliths, the interval of administration should be prolonged (i.e., 10 mg/kg q24 h) [113]. Limited data are available for cats; development of AKI reported [114]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Santis, F.; Boari, A.; Dondi, F.; Crisi, P.E. Drug-Dosing Adjustment in Dogs and Cats with Chronic Kidney Disease. Animals 2022, 12, 262. https://doi.org/10.3390/ani12030262
De Santis F, Boari A, Dondi F, Crisi PE. Drug-Dosing Adjustment in Dogs and Cats with Chronic Kidney Disease. Animals. 2022; 12(3):262. https://doi.org/10.3390/ani12030262
Chicago/Turabian StyleDe Santis, Francesca, Andrea Boari, Francesco Dondi, and Paolo Emidio Crisi. 2022. "Drug-Dosing Adjustment in Dogs and Cats with Chronic Kidney Disease" Animals 12, no. 3: 262. https://doi.org/10.3390/ani12030262
APA StyleDe Santis, F., Boari, A., Dondi, F., & Crisi, P. E. (2022). Drug-Dosing Adjustment in Dogs and Cats with Chronic Kidney Disease. Animals, 12(3), 262. https://doi.org/10.3390/ani12030262






