Neurobiology of Maternal Behavior in Nonhuman Mammals: Acceptance, Recognition, Motivation, and Rejection
Abstract
Simple Summary
Abstract
1. Introduction
2. Active and Passive Maternal Behaviors
3. Natural Stimuli That Facilitate the Expression of Maternal Behavior
3.1. Hormones
3.2. Vagino-Cervical Stimulation
3.3. Exposure to Pups
3.4. Mating
3.5. Maternal Experience
3.6. Environmental Enrichment
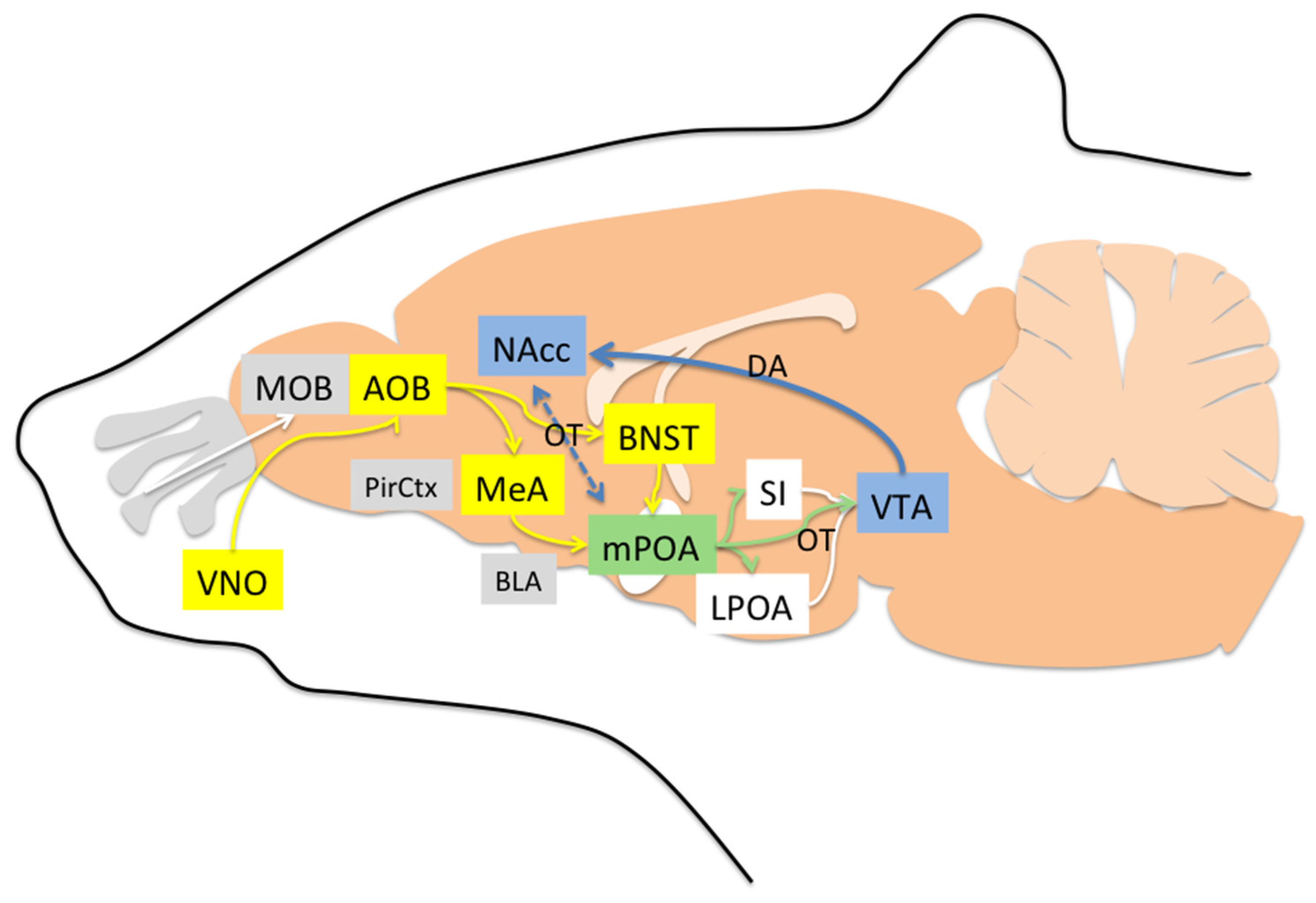
4. Neural Pathways Underlying Maternal Behavior
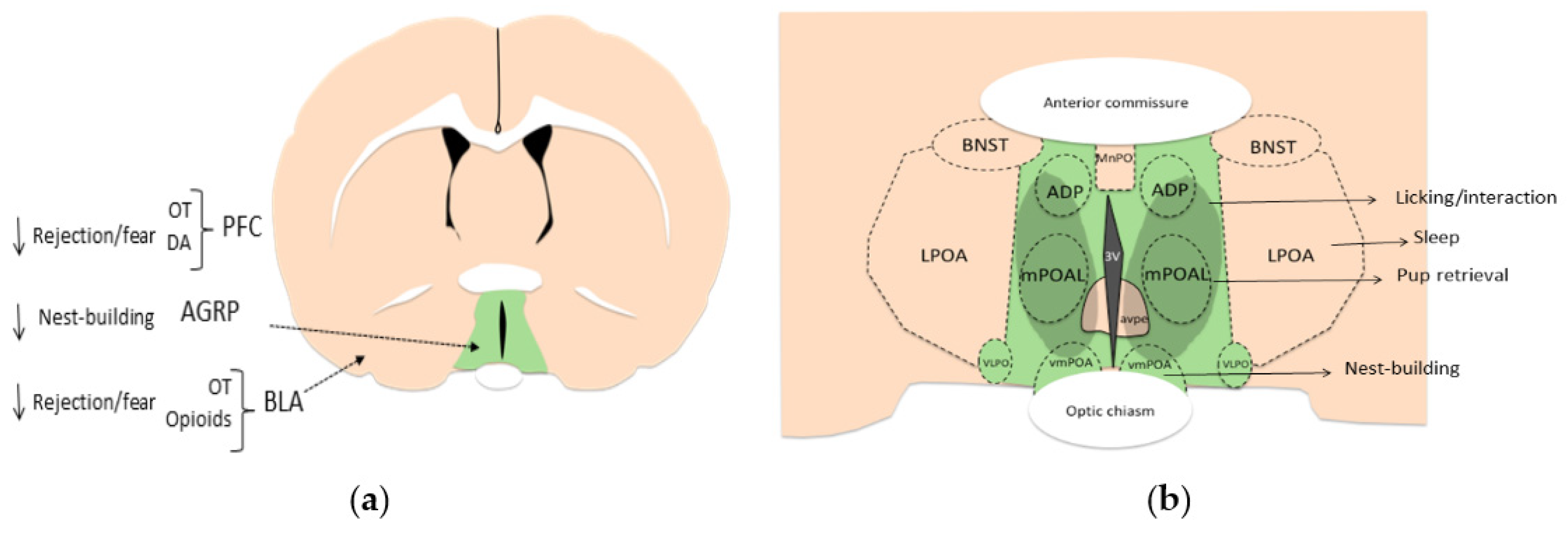
4.1. Areas Involved in Acceptance and Social Recognition
4.2. Areas Mainly Involved in Increasing Motivation
4.3. Areas Mainly Involved in Reducing Rejection/Fear
| Brain Area | Neurochemical | Effect | Representative References |
|---|---|---|---|
| MeA BNST CoA | OT | Acceptance, social recognition | [74,79,83] |
| mPOA | E2 PRL OT DA Galanin Vgat HCRT-1 AGRP | Pup retrieval Pup retrieval, nest-building Acceptance, licking/grooming Motivation, licking, grooming Motor coordination, motivation, recognition, grooming, inhibition of aggression Nest-building Licking/retrieval Inhibition of nest-building | [32,33,40] [36] [102,103] [103,104,130] [130] [106] [107] |
| VTA | OT | Approach, interaction | [74,79,86] |
| BLA | OT opioids | Inhibit rejection/fear | [85,108,121,124] |
| NAc | DA OT | Motivation, approach, interaction, pup retrieval | [99,100,101,112] |
| PFC | OT DA | Inhibit rejection/fear | [125,126] |
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kendrick, K.M.; Hinton, M.R.; Atkins, K.; Haupt, M.A.; Skinner, J.D. Mothers determine sexual preferences. Nature 1998, 395, 229–230. [Google Scholar] [CrossRef] [PubMed]
- Crowell-Davis, S.L.; Houpt, K.A. Maternal Behavior. Veter. Clin. N. Am. Equine Pract. 1986, 2, 557–571. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, M.; He, T.; Long, S.; Guo, Y.; Chen, Z. Effect of Different Cross-Fostering Strategies on Growth Performance, Stress Status and Immunoglobulin of Piglets. Animals 2021, 11, 499. [Google Scholar] [CrossRef]
- Grota, L.J. Effects of litter size, age of young, and parity on foster mother behaviour in Rattus norvegicus. Anim. Behav. 1973, 21, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Scharis, I.; Amundin, M. Cross-fostering in gray wolves (Canis lupus lupus). Zoo Biol. 2015, 34, 217–222. [Google Scholar] [CrossRef] [PubMed]
- von Keyserlingk, M.A.; Weary, D.M. Maternal behavior in cattle. Horm. Behav. 2007, 52, 106–113. [Google Scholar] [CrossRef]
- Dienske, H.; Van Vreeswijk, W. Regulation of nursing in chimpanzees. Dev. Psychobiol. 1987, 20, 71–83. [Google Scholar] [CrossRef]
- Davenport, R.K., Jr.; Menzel, E.W., Jr.; Rogers, C.M. Maternal care during infancy: Its effect on weight gain and mortality in the chimpanzee. Am. J. Orthopsychiatry 1961, 31, 803–809. [Google Scholar] [CrossRef]
- Schulte, I.; Hoy, S. Nursing and suckling behavior and mother-child contacts in domestic rabbits. Berl. Munch. Tierarztl. Wochenschr. 1997, 110, 134–138. [Google Scholar]
- Kleiman, D. Reproduction in the Canidae. Int. Zoo Yearb. 1968, 8, 3–8. [Google Scholar] [CrossRef]
- Denenberg, V.H.; Zarrow, M.; Taylor, R.E. Maternal Behavior in the Rat: An Investigation and Quantification of Nest Building. Behaviour 1969, 34, 1–16. [Google Scholar] [CrossRef]
- Benedek, I.; Altbäcker, V.; Zsolnai, A.; Molnár, T. Exploring the Genetic Background of the Differences in Nest-Building Behavior in European Rabbit. Animals 2020, 10, 1579. [Google Scholar] [CrossRef] [PubMed]
- Bleicher, N. Behavior of the bitch during parturition. J. Am. Veter. Med. Assoc. 1962, 140, 1076–1082. [Google Scholar]
- Ferrari, J.; Monteiro-Filho, E.L.A. Canid Familiaris-Comparative Analysis of pre and Postpartum Behavioral Patterns; Universidade Federal do Paraná: Curitiba, Brazil, 2004. [Google Scholar]
- Santos, N.R.; Beck, A.; Fontbonne, A. A review of maternal behaviour in dogs and potential areas for further research. J. Small Anim. Pract. 2019, 61, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Lidfors, L. Parental Behavior in Bovines. Adv. Neurobiol. 2022, 27, 177–212. [Google Scholar] [CrossRef] [PubMed]
- Rørvang, M.V.; Nielsen, B.L.; Herskin, M.S.; Jensen, M.B. Prepartum Maternal Behavior of Domesticated Cattle: A Comparison with Managed, Feral, and Wild Ungulates. Front. Veter. Sci. 2018, 5, 45. [Google Scholar] [CrossRef]
- Schweizer, C.M.; Meyers-Wallen, V.M. Medical managment of dystocia and indications of cesarean section in the bitch. In Current Veterinary Therapy XIII; Bonagura, W.B., Ed.; Saunders Co: Philadelphia, PA, USA, 2000; pp. 933–939. [Google Scholar]
- Rheingold, H.L. Maternal behavior in the dog. In Maternal Behaviors; Mammals, I., Reingold, H.L., Eds.; John Wiley and Sons: New York, NY, USA, 1963; pp. 169–202. [Google Scholar]
- Moore, C.L.; Morelli, G.A. Mother rats interact differently with male and female offspring. J. Comp. Physiol. Psychol. 1979, 93, 677–684. [Google Scholar] [CrossRef]
- Lévy, F.; Keller, M.; Poindron, P. Olfactory regulation of maternal behavior in mammals. Horm. Behav. 2004, 46, 284–302. [Google Scholar] [CrossRef]
- Moore, C. Maternal behavior of rats is affected by hormonal condition of pups. J. Comp. Physiol. Psychol. 1982, 96, 123–129. [Google Scholar] [CrossRef]
- Moore, C.L. Maternal contributions to the development of masculine sexual behavior in laboratory rats. Dev. Psychobiol. 1984, 17, 347–356. [Google Scholar] [CrossRef]
- Nagasawa, M.; Shibata, Y.; Yonezawa, A.; Takahashi, T.; Kanai, M.; Ohtsuka, H.; Suenaga, Y.; Yabana, Y.; Mogi, K.; Kikusui, T. Basal cortisol concentrations related to maternal behavior during puppy development predict post-growth resilience in dogs. Horm. Behav. 2021, 136, 105055. [Google Scholar] [CrossRef] [PubMed]
- Weaver, I.C.; Cervoni, N.; Champagne, F.A.; D’Alessio, A.C.; Sharma, S.; Seckl, J.R.; Dymov, S.; Szyf, M.; Meaney, M.J. Epi-genetic programming by maternal behavior. Nat. Neurosci. 2004, 7, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, M.; Shibata, Y.; Yonezawa, A.; Morita, T.; Kanai, M.; Mogi, K.; Kikusui, T. The behavioral and endocrinological development of stress response in dogs. Dev. Psychobiol. 2013, 56, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Santos, N.R.; Beck, A.; Maenhoudt, C.; Billy, C.; Fontbonne, A. Profile of Dogs’ Breeders and Their Considerations on Female Reproduction, Maternal Care and the Peripartum Stress—An International Survey. Animals 2021, 11, 2372. [Google Scholar] [CrossRef] [PubMed]
- Abitbol, M.L.; Inglis, S.R. Role of amniotic fluid in newborn acceptance and bonding in canines. J. Matern.-Fetal Med. 1997, 6, 49–52. [Google Scholar] [CrossRef]
- Rosenblatt, J.S.; Mayer, A.D.; Giordano, A.L. Hormonal basis during pregnancy for the onset of maternal behavior in the rat. Psychoneuroendocrinology 1988, 13, 29–46. [Google Scholar] [CrossRef] [PubMed]
- Nelson, R.J. An Introduction to Behavioral Endocrinology, 2nd ed.; Sinauer Associates: Sunderland, MA, USA, 2000. [Google Scholar]
- Bridges, R.S. The role of lactogenic hormones in maternal behavior in female rats. Acta Paediatr. Suppl. 1994, 83, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.C.; Musatov, S.; Shteyler, A.; Simanduyev, S.; Arrieta-Cruz, I.; Ogawa, S.; Pfaff, D.W. siRNA silencing of estrogen receptor-α expression specifically in medial preoptic area neurons abolishes maternal care in female mice. Proc. Natl. Acad. Sci. USA 2012, 109, 16324–16329. [Google Scholar] [CrossRef]
- Catanese, M.; Vandenberg, L.N. Bisphenol S (BPS) alters maternal behavior and brain in mice exposed during pregnancy/lactation and their daughters. Endocrinology 2016, 158, 516–530. [Google Scholar] [CrossRef]
- Fang, Y.-Y.; Yamaguchi, T.; Song, S.C.; Tritsch, N.X.; Lin, D. A Hypothalamic Midbrain Pathway Essential for Driving Maternal Behaviors. Neuron 2018, 98, 192–207.e10. [Google Scholar] [CrossRef]
- Wei, Y.-C.; Wang, S.-R.; Jiao, Z.-L.; Zhang, W.; Lin, J.-K.; Li, X.-Y.; Li, S.-S.; Zhang, X.; Xu, X.-H. Medial preoptic area in mice is capable of mediating sexually dimorphic behaviors regardless of gender. Nat. Commun. 2018, 9, 279. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.S.E.; Aoki, M.; Ladyman, S.R.; Phillipps, H.R.; Wyatt, A.; Boehm, U.; Grattan, D.R. Prolactin action in the medial preoptic area is necessary for postpartum maternal nursing behavior. Proc. Natl. Acad. Sci. USA 2017, 114, 10779–10784. [Google Scholar] [CrossRef] [PubMed]
- Lévy, F. Neuroendocrine control of maternal behavior in non-human and human mammals. Ann. d’Endocrinol. 2016, 77, 114–125. [Google Scholar] [CrossRef]
- Kendrick, K.; Lévy, F.; Keverne, E. Importance of vaginocervical stimulation for the formation of maternal bonding in primiparous and multiparous parturient ewes. Physiol. Behav. 1991, 50, 595–600. [Google Scholar] [CrossRef]
- Keverne, E.B.; Levy, F.; Poindron, P.; Lindsay, D.R. Vaginal Stimulation: An Important Determinant of Maternal Bonding in Sheep. Science 1983, 219, 81–83. [Google Scholar] [CrossRef] [PubMed]
- Ahdieh, H.B.; Mayer, A.D.; Rosenblatt, J.S. Effects of Brain Antiestrogen Implants on Maternal Behavior and on Postpartum Estrus in Pregnant Rats. Neuroendocrinology 1987, 46, 522–531. [Google Scholar] [CrossRef]
- Gandelman, R.; McDermott, N.J.; Kleinman, M.; DeJianne, D. Maternal nest building by pseudopregnant mice. Reproduction 1979, 56, 697–699. [Google Scholar] [CrossRef]
- Steuer, M.A.; Thompson, A.C.; Doerr, J.C.; Youakim, M.; Kristal, M.B. Induction of maternal behavior in rats: Effects of pseudopregnancy termination and placenta-smeared pups. Behav. Neurosci. 1987, 101, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Misner, T.L.; A Houpt, K. Animal behavior case of the month. Aggression that began 4 days after ovariohysterectomy. J. Am. Veter. Med. Assoc. 1998, 213, 1260–1262. [Google Scholar]
- Rosenblatt, J.S.; Siegel, H.I. Hysterectomy-induced maternal behavior during pregnancy in the rat. J. Comp. Physiol. Psychol. 1975, 89, 685–700. [Google Scholar] [CrossRef]
- Rosenblatt, J.S.; Hazelwood, S.; Poole, J. Maternal Behavior in Male Rats: Effects of Medial Preoptic Area Lesions and Presence of Maternal Aggression. Horm. Behav. 1996, 30, 201–215. [Google Scholar] [CrossRef] [PubMed]
- Perkinson, M.R.; Kim, J.S.; Iremonger, K.J.; Brown, C.H. Visualising oxytocin neurone activity in vivo: The key to unlocking central regulation of parturition and lactation. J. Neuroendocr. 2021, 33, e13012. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wang, S.C.; Liu, X.; Jia, S.; Wang, X.; Li, T.; Yu, J.; Parpura, V.; Wang, Y.-F. Neural Functions of Hypothalamic Oxytocin and its Regulation. ASN Neuro 2022, 14, 17590914221100706. [Google Scholar] [CrossRef] [PubMed]
- Terkel, J.; Rosenblatt, J.S. Humoral factors underlying maternal behavior at parturition: Corss transfusion between freely moving rats. J. Comp. Physiol. Psychol. 1972, 80, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Rosenblatt, J.S. Nonhormonal Basis of Maternal Behavior in the Rat. Science 1967, 156, 1512–1514. [Google Scholar] [CrossRef]
- Jakubowski, M.; Terkel, J. Transition from pup killing to parental behavior in male and virgin female albino rats. Physiol. Behav. 1985, 34, 683–686. [Google Scholar] [CrossRef]
- Harding, K.M.; Lonstein, J.S. Extensive juvenile “babysitting” facilitates later adult maternal responsiveness, decreases anxiety, and increases dorsal raphe tryptophan hydroxylase-2 expression in female laboratory rats. Dev. Psychobiol. 2016, 58, 492–508. [Google Scholar] [CrossRef]
- Carcea, I.; Caraballo, N.L.; Marlin, B.J.; Ooyama, R.; Riceberg, J.S.; Navarro, J.M.M.; Opendak, M.; Diaz, V.E.; Schuster, L.; Torres, M.I.A.; et al. Oxytocin neurons enable social transmission of maternal behaviour. Nature 2021, 596, 553–557. [Google Scholar] [CrossRef]
- Mennella, J.A.; Moltz, H. Infanticide in rats: Male strategy and female counter-strategy. Physiol. Behav. 1988, 42, 19–28. [Google Scholar] [CrossRef]
- vom Saal, F.S. Time-contingent change in infanticide and parental behavior induced by ejaculation in male mice. Physiol. Behav. 1985, 34, 7–15. [Google Scholar] [CrossRef]
- Dulac, C.; O’Connell, L.A.; Wu, Z. Neural control of maternal and paternal behaviors. Science 2014, 345, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Coolen, L.M.; Peters, H.J.; Veening, J.G. Fos immunoreactivity in the rat brain following consummatory elements of sexual behavior: A sex comparison. Brain Res. 1996, 738, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Malsbury, C.W. Facilitation of male rat copulatory behavior by electrical stimulation of the medial preoptic area. Physiol. Behav. 1971, 7, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Shimura, T.; Yamamoto, T.; Shimokochi, M. The medial preoptic area is involved in both sexual arousal and performance in male rats: Re-evaluation of neuron activity in freely moving animals. Brain Res. 1994, 640, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Arendash, G.W.; Gorski, R.A. Effects of discrete lesions of the sexually dimorphic nucleus of the preoptic area or other medial preoptic regions on the sexual behavior of male rats. Brain Res. Bull. 1983, 10, 147–154. [Google Scholar] [CrossRef]
- Guardini, G.; Bowen, J.; Raviglione, S.; Farina, R.; Gazzano, A. Maternal behaviour in domestic dogs: A comparison between primiparous and multiparous dogs. Dog Behav. 2015, 1, 23–33. [Google Scholar] [CrossRef]
- Vicentini, R.R.; El Faro, L.; Ujita, A.; Lima, M.L.P.; Oliveira, A.P.; Sant’Anna, A.C. Is maternal defensiveness of Gyr cows (Bos taurus indicus) related to parity and cows’ behaviors during the peripartum period? PLoS ONE 2022, 17, e0274392. [Google Scholar] [CrossRef]
- Jensen, M.B.; Webb, L.E.; Vaarst, M.; Bokkers, E. The effect of hides and parity on behavior of periparturient dairy cows at pasture. J. Dairy Sci. 2022, 105, 6196–6206. [Google Scholar] [CrossRef]
- Aguggia, J.P.; Suárez, M.M.; Rivarola, M.A. Multiparity Dampened the Neurobehavioral Consequences of Mother–Pup Separation Stress in Dams. Neuroscience 2019, 416, 207–220. [Google Scholar] [CrossRef]
- Lv, S.-J.; Yang, Y.; Li, F.-K. Parity and litter size effects on maternal behavior of Small Tail Han sheep in China. Anim. Sci. J. 2015, 87, 361–369. [Google Scholar] [CrossRef]
- Zhang, Y.-M.; Cheng, Y.-Z.; Wang, Y.-T.; Wei, R.-M.; Ge, Y.-J.; Kong, X.-Y.; Li, X.-Y. Environmental Enrichment Reverses Maternal Sleep Deprivation-Induced Anxiety-Like Behavior and Cognitive Impairment in CD-1 Mice. Front. Behav. Neurosci. 2022, 16, 943900. [Google Scholar] [CrossRef] [PubMed]
- Núñez-Murrieta, M.A.; Noguez, P.; Coria-Avila, G.A.; García-García, F.; Santiago-García, J.; Bolado-García, V.E.; Corona-Morales, A.A. Maternal behavior, novelty confrontation, and subcortical c-Fos expression during lactation period are shaped by gestational environment. Behav. Brain Res. 2021, 412, 113432. [Google Scholar] [CrossRef]
- Coria-Avila, G.A.; Manzo, J.; Garcia, L.I.; Carrillo, P.; Miquel, M.; Pfaus, J.G. Neurobiology of social attachments. Neurosci. Biobehav. Rev. 2014, 43, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Poindron, P.; Otal, J.; Ferreira, G.; Keller, M.; Guesdon, V.; Nowak, R.; Lévy, F. Amniotic fluid is important for the maintenance of maternal responsiveness and the establishment of maternal selectivity in sheep. Animal 2010, 4, 2057–2064. [Google Scholar] [CrossRef] [PubMed]
- Levy, F.; Poindron, P.; Le Neindre, P. Attraction and repulsion by amniotic fluids and their olfactory control in the ewe around parturition. Physiol. Behav. 1983, 31, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, G.; Terrazas, A.; Poindron, P.; Nowak, R.; Orgeur, P.; Lévy, F. Learning of olfactory cues is not necessary for early lamb recognition by the mother. Physiol. Behav. 2000, 69, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Rymer, T.L. The Role of Olfactory Genes in the Expression of Rodent Paternal Care Behavior. Genes 2020, 11, 292. [Google Scholar] [CrossRef]
- Fallon, J.H.; Moore, R.Y. Catecholamine innervation of the basal forebrain IV. Topography of the dopamine projection to the basal forebrain and neostriatum. J. Comp. Neurol. 1978, 180, 545–579. [Google Scholar] [CrossRef]
- Bielsky, I.F.; Hu, S.-B.; Szegda, K.L.; Westphal, H.; Young, L.J. Profound Impairment in Social Recognition and Reduction in Anxiety-Like Behavior in Vasopressin V1a Receptor Knockout Mice. Neuropsychopharmacology 2003, 29, 483–493. [Google Scholar] [CrossRef]
- Bielsky, I.F.; Young, L.J. Oxytocin, vasopressin, and social recognition in mammals. Peptides 2004, 25, 1565–1574. [Google Scholar] [CrossRef]
- Kavaliers, M.; Colwell, D.; Choleris, E.; Ågmo, A.; Muglia, L.J.; Ogawa, S.; Pfaff, D.W. Impaired discrimination of and aversion to parasitized male odors by female oxytocin knockout mice. Genes Brain Behav. 2003, 2, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, J.N.; Aldag, J.M.; Insel, T.R.; Young, L.J. Oxytocin in the Medial Amygdala is Essential for Social Recognition in the Mouse. J. Neurosci. 2001, 21, 8278–8285. [Google Scholar] [CrossRef] [PubMed]
- Dumais, K.M.; Bredewold, R.; Mayer, T.E.; Veenema, A.H. Sex differences in oxytocin receptor binding in forebrain regions: Correlations with social interest in brain region- and sex- specific ways. Horm. Behav. 2013, 64, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Caughey, S.D.; Klampfl, S.M.; Bishop, V.R.; Pfoertsch, J.; Neumann, I.D.; Bosch, O.J.; Meddle, S.L. Changes in the Intensity of Maternal Aggression and Central Oxytocin and Vasopressin V1a Receptors Across the Peripartum Period in the Rat. J. Neuroendocr. 2011, 23, 1113–1124. [Google Scholar] [CrossRef]
- Young, L.J.; Lim, M.M.; Gingrich, B.; Insel, T.R. Cellular Mechanisms of Social Attachment. Horm. Behav. 2001, 40, 133–138. [Google Scholar] [CrossRef]
- Fuchs, A.-R.; Fuchs, F.; Husslein, P.; Soloff, M.S.; Fernström, M.J. Oxytocin Receptors and Human Parturition: A Dual Role for Oxytocin in the Initiation of Labor. Science 1982, 215, 1396–1398. [Google Scholar] [CrossRef]
- Gimpl, G.; Fahrenholz, F. The Oxytocin Receptor System: Structure, Function, and Regulation. Physiol. Rev. 2001, 81, 629–683. [Google Scholar] [CrossRef]
- Insel, T.R.; Gingrich, B.S.; Young, L.J. Oxytocin: Who needs it? Prog. Brain Res. 2001, 133, 59–66. [Google Scholar]
- Grieb, Z.A.; Lonstein, J.S. Oxytocin receptor expression in the midbrain dorsal raphe is dynamic across female reproduction in rats. J. Neuroendocr. 2021, 33, e12926. [Google Scholar] [CrossRef]
- Quiñones-Jenab, V.; Jenab, S.; Ogawa, S.; Adan, R.A.; Burbach, P.H.; Pfaff, D.W. Effects of Estrogen on Oxytocin Receptor Messenger Ribonucleic Acid Expression in the Uterus, Pituitary, and Forebrain of the Female Rat. Neuroendocrinology 1997, 65, 9–17. [Google Scholar] [CrossRef]
- Keller, M.; Perrin, G.; Meurisse, M.; Ferreira, G.; Lévy, F. Cortical and medial amygdala are both involved in the formation of olfactory offspring memory in sheep. Eur. J. Neurosci. 2004, 20, 3433–3441. [Google Scholar] [CrossRef] [PubMed]
- Ogi, A.; Mariti, C.; Pirrone, F.; Baragli, P.; Gazzano, A. The Influence of Oxytocin on Maternal Care in Lactating Dogs. Animals 2021, 11, 1130. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Jia, S.-W.; Hou, D.; Liu, X.; Li, D.; Liu, Y.; Cui, D.; Wang, X.; Hou, C.; Brown, C.H.; et al. Intranasal Oxytocin Restores Maternal Behavior and Oxytocin Neuronal Activity in the Supraoptic Nucleus in Rat Dams with Cesarean Delivery. Neuroscience 2021, 468, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.H.; French, J.A. Oxytocin and vasopressin enhance responsiveness to infant stimuli in adult marmosets. Horm. Behav. 2015, 75, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Turcsán, B.; Román, V.; Lévay, G.; Lendvai, B.; Kedves, R.; Petró, E.; Topál, J. Intranasal Oxytocin Improves Social Behavior in Laboratory Beagle Dogs (Canis familiaris) Using a Custom-Made Social Test Battery. Front. Veter. Sci. 2022, 9, 785805. [Google Scholar] [CrossRef] [PubMed]
- Romero, T.; Nagasawa, M.; Mogi, K.; Hasegawa, T.; Kikusui, T. Intranasal administration of oxytocin promotes social play in domestic dogs. Commun. Integr. Biol. 2015, 8, e1017157. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.; Su, W.; Zhou, R.; Feng, J.; Lin, Y.; Zhang, Y.; Wang, X.; Chen, X.; Li, J. Intranasal oxytocin administration improves depression-like behaviors in adult rats that experienced neonatal maternal deprivation. Behav. Pharmacol. 2016, 27, 689–696. [Google Scholar] [CrossRef]
- Ramos, L.; Hicks, C.; Caminer, A.; Goodwin, J.; McGregor, I.S. Oxytocin and MDMA (‘Ecstasy’) enhance social reward in rats. Psychopharmacology 2015, 232, 2631–2641. [Google Scholar] [CrossRef]
- Ogi, A.; Naef, V.; Santorelli, F.M.; Mariti, C.; Gazzano, A. Oxytocin Receptor Gene Polymorphism in Lactating Dogs. Animals 2021, 11, 3099. [Google Scholar] [CrossRef]
- Ermisch, A.; Rühle, H.-J.; Landgraf, R.; Hess, J. Blood—Brain Barrier and Peptides. J. Cereb. Blood Flow Metab. 1985, 5, 350–357. [Google Scholar] [CrossRef]
- Kenkel, W.M.; Perkeybile, A.-M.; Yee, J.R.; Pournajafi-Nazarloo, H.; Lillard, T.S.; Ferguson, E.F.; Wroblewski, K.L.; Ferris, C.F.; Carter, C.S.; Connelly, J.J. Behavioral and epigenetic consequences of oxytocin treatment at birth. Sci. Adv. 2019, 5, eaav2244. [Google Scholar] [CrossRef] [PubMed]
- Walsh, C.J.; Fleming, A.S.; Lee, A.; Magnusson, J.E. The effects of olfactory and somatosensory desensitization on Fos-like immunoreactivity in the brains of pup-exposed postpartum rats. Behav. Neurosci. 1996, 110, 134–153. [Google Scholar] [CrossRef] [PubMed]
- Stack, E.C.; Balakrishnan, R.; Numan, M.J.; Numan, M. A functional neuroanatomical investigation of the role of the medial preoptic area in neural circuits regulating maternal behavior. Behav. Brain Res. 2002, 131, 17–36. [Google Scholar] [CrossRef] [PubMed]
- Tsuneoka, Y.; Maruyama, T.; Yoshida, S.; Nishimori, K.; Kato, T.; Numan, M.; Kuroda, K.O. Functional, anatomical, and neurochemical differentiation of medial preoptic area subregions in relation to maternal behavior in the mouse. J. Comp. Neurol. 2012, 521, 1633–1663. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Clancy, S.; Fleming, A.S. Mother rats bar-press for pups: Effects of lesions of the mpoa and limbic sites on maternal behavior and operant responding for pup-reinforcement. Behav. Brain Res. 1999, 100, 15–31. [Google Scholar] [CrossRef]
- Lee, A.; Li, M.; Watchus, J.; Fleming, A.S. Neuroanatomical basis of maternal memory in postpartum rats: Selective role for the nucleus accumbens. Behav. Neurosci. 1999, 113, 523–538. [Google Scholar] [CrossRef]
- Gaffori, O.; Le Moal, M. Disruption of maternal behavior and appearance of cannibalism after ventral mesencephalic tegmentum lesions. Physiol. Behav. 1979, 23, 317–323. [Google Scholar] [CrossRef]
- Shahrokh, D.K.; Zhang, T.-Y.; Diorio, J.; Gratton, A.; Meaney, M.J. Oxytocin-Dopamine Interactions Mediate Variations in Maternal Behavior in the Rat. Endocrinology 2010, 151, 2276–2286. [Google Scholar] [CrossRef]
- Tsuneoka, Y.; Funato, H. Cellular Composition of the Preoptic Area Regulating Sleep, Parental, and Sexual Behavior. Front. Neurosci. 2021, 15, 649159. [Google Scholar] [CrossRef]
- Kohl, J.; Babayan, B.M.; Rubinstein, N.D.; Autry, A.E.; Marin-Rodriguez, B.; Kapoor, V.; Miyamishi, K.; Zweifel, L.S.; Luo, L.; Uchida, N.; et al. Functional circuit architecture underlying parental behaviour. Nature 2018, 556, 326–331. [Google Scholar] [CrossRef]
- Tsuneoka, Y.; Yoshida, S.; Takase, K.; Oda, S.; Kuroda, M.; Funato, H. Neurotransmitters and neuropeptides in gonadal steroid receptor-expressing cells in medial preoptic area subregions of the male mouse. Sci. Rep. 2017, 7, 9809. [Google Scholar] [CrossRef] [PubMed]
- Rivas, M.; Torterolo, P.; Ferreira, A.; Benedetto, L. Hypocretinergic system in the medial preoptic area promotes maternal behavior in lactating rats. Peptides 2016, 81, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-Y.; Han, Y.; Zhang, W.; Wang, S.-R.; Wei, Y.-C.; Li, S.-S.; Lin, J.-K.; Yan, J.-J.; Chen, A.-X.; Zhang, X.; et al. AGRP Neurons Project to the Medial Preoptic Area and Modulate Maternal Nest-Building. J. Neurosci. 2018, 39, 456–471. [Google Scholar] [CrossRef]
- Insel, T.R.; E Shapiro, L. Oxytocin receptor distribution reflects social organization in monogamous and polygamous voles. Proc. Natl. Acad. Sci. USA 1992, 89, 5981–5985. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Z. Nucleus accumbens oxytocin and dopamine interact to regulate pair bond formation in female prairie voles. Neuroscience 2003, 121, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Aragona, B.J.; Liu, Y.; Curtis, J.T.; Stephan, F.K.; Wang, Z. A Critical Role for Nucleus Accumbens Dopamine in Partner-Preference Formation in Male Prairie Voles. J. Neurosci. 2003, 23, 3483–3490. [Google Scholar] [CrossRef] [PubMed]
- Gingrich, B.; Liu, Y.; Cascio, C.; Wang, Z.; Insel, T.R. Dopamine D2 receptors in the nucleus accumbens are important for social attachment in female prairie voles (Microtus ochrogaster). Behav. Neurosci. 2000, 114, 173–183. [Google Scholar] [CrossRef]
- Hansen, S.; Bergvall, A.H.; Nyiredi, S. Interaction with pups enhances dopamine release in the ventral striatum of maternal rats: A microdialysis study. Pharmacol. Biochem. Behav. 1993, 45, 673–676. [Google Scholar] [CrossRef]
- Champagne, F.A.; Chretien, P.; Stevenson, C.W.; Zhang, T.Y.; Gratton, A.; Meaney, M.J. Variations in Nucleus Accumbens Dopamine Associated with Individual Differences in Maternal Behavior in the Rat. J. Neurosci. 2004, 24, 4113–4123. [Google Scholar] [CrossRef]
- Keer, S.; Stern, J. Dopamine Receptor Blockade in the Nucleus Accumbens Inhibits Maternal Retrieval and Licking, but Enhances Nursing Behavior in Lactating Rats. Physiol. Behav. 1999, 67, 659–669. [Google Scholar] [CrossRef]
- Numan, M.; Numan, M.J.; Pliakou, N.; Stolzenberg, D.S.; Mullins, O.J.; Murphy, J.M.; Smith, C.D. The effects of D1 or D2 dopamine receptor antagonism in the medial preoptic area, ventral pallidum, or nucleus accumbens on the maternal retrieval response and other aspects of maternal behavior in rats. Behav. Neurosci. 2005, 119, 1588–1604. [Google Scholar] [CrossRef] [PubMed]
- Romero-Morales, L.; García-Saucedo, B.; Martínez-Torres, M.; Cárdenas, M.; Cárdenas-Vázquez, R.; Luis, J. Neural activation associated with maternal and aversive interactions with pups in the mongolian gerbil (Meriones unguiculatus). Behav. Brain Res. 2022, 437, 114153. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A.S.; Korsmit, M. Plasticity in the maternal circuit: Effects of maternal experience on Fos-Lir in hypothalamic, limbic, and cortical structures in the postpartum rat. Behav. Neurosci. 1996, 110, 567–582. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A.S.; Rosenblatt, J.S. Maternal behavior in the virgin and lactating rat. J. Comp. Physiol. Psychol. 1974, 86, 957–972. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A.S.; Rosenblatt, J.S. Olfactory regulation of maternal behavior in rats: I. Effects of olfactory bulb removal in experienced and inexperienced lactating and cycling females. J. Comp. Physiol. Psychol. 1974, 86, 221–232. [Google Scholar] [CrossRef]
- Fleming, A.S.; Rosenblatt, J.S. Olfactory regulation of maternal behavior in rats: II. Effects of peripherally induced anosmia and lesions of the lateral olfactory tract in pup-induced virgins. J. Comp. Physiol. Psychol. 1974, 86, 233–246. [Google Scholar] [CrossRef]
- Inoue, S.; Kamiyama, H.; Matsumoto, M.; Yanagawa, Y.; Hiraide, S.; Saito, Y.; Shimamura, K.-I.; Togashi, H. Synaptic Modulation via Basolateral Amygdala on the Rat Hippocampus–Medial Prefrontal Cortex Pathway in Fear Extinction. J. Pharmacol. Sci. 2013, 123, 267–278. [Google Scholar] [CrossRef]
- Lahoud, N.; Maroun, M. Oxytocinergic manipulations in corticolimbic circuit differentially affect fear acquisition and extinction. Psychoneuroendocrinology 2013, 38, 2184–2195. [Google Scholar] [CrossRef]
- Deji, C.; Yan, P.; Ji, Y.; Yan, X.; Feng, Y.; Liu, J.; Liu, Y.; Wei, S.; Zhu, Y.; Lai, J. The Basolateral Amygdala to Ventral Hippocampus Circuit Controls Anxiety-Like Behaviors Induced by Morphine Withdrawal. Front. Cell. Neurosci. 2022, 16, 894886. [Google Scholar] [CrossRef]
- Wardlaw, S.L.; Frantz, A.G. Brainβ-Endorphin during Pregnancy, Parturition, and the Postpartum Period. Endocrinology 1983, 113, 1664–1668. [Google Scholar] [CrossRef]
- Neumann, I.; Russell, J.; Landgraf, R. Chapter 8: Endogenous opioids regulate intracerebral oxytocin release during parturition in a region-specific manner. Prog. Brain Res. 1992, 91, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Kalivas, P.W.; Abhold, R. Enkephalin release into the ventral tegmental area in response to stress: Modulation of mesocorticolimbic dopamine. Brain Res. 1987, 414, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Beauregard, M.; Leroux, J.-M.; Bergman, S.; Arzoumanian, Y.; Beaudoin, G.; Bourgouin, P.; Stip, E. The functional neuroanatomy of major depression: An fMRI study using an emotional activation paradigm. Neuroreport 1998, 9, 3253–3258. [Google Scholar] [CrossRef] [PubMed]
- Broad, K.; Hinton, M.; Keverne, E.; Kendrick, K. Involvement of the medial prefrontal cortex in mediating behavioural responses to odour cues rather than olfactory recognition memory. Neuroscience 2002, 114, 715–729. [Google Scholar] [CrossRef]
- Stamatakis, A.; Kalpachidou, T.; Raftogianni, A.; Zografou, E.; Tzanou, A.; Pondiki, S.; Stylianopoulou, F. Rat dams exposed repeatedly to a daily brief separation from the pups exhibit increased maternal behavior, decreased anxiety and altered levels of receptors for estrogens (ERα, ERβ), oxytocin and serotonin (5-HT1A) in their brain. Psychoneuroendocrinology 2015, 52, 212–228. [Google Scholar] [CrossRef]
- Wu, Z.; Autry, A.E.; Bergan, J.F.; Watabe-Uchida, M.; Dulac, C.G. Galanin neurons in the medial preoptic area govern parental behaviour. Nature 2014, 509, 325–330. [Google Scholar] [CrossRef]
- Osterhout, J.A.; Kapoor, V.; Eichhorn, S.W.; Vaughn, E.; Moore, J.D.; Liu, D.; Lee, D.; DeNardo, L.A.; Luo, L.; Zhuang, X.; et al. A preoptic neuronal population controls fever and appetite during sickness. Nature 2022, 606, 937–944. [Google Scholar] [CrossRef]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates; Academic Press, Inc.: San Diego, CA, USA, 1998. [Google Scholar]
| Responses | Acceptance | Recognition | Motivation | Rejection/Fear |
|---|---|---|---|---|
| Passive | Allow proximity | Selective proximity | Staying in nest | Avoidance |
| Nursing | Selective nursing | Crouching over | ||
| Active | Directed calls | Licking | Aggression | |
| Directed attention | Retrieving | Infanticide | ||
| Placenta consumption | ||||
| Defense | ||||
| Nest-building |
| Stimuli | Effect | Behavioral Response | Representative References |
|---|---|---|---|
| Hormones E2 P4 PRL OT | ⇑ ⇑ ⇑ ⇑ | Pup retrieval Retrieving Nest-building Nursing | [34,35] [45] [36] [47] |
| Parturition VCS | ⇑ | Nursing Retrieving | [38,39] [40] |
| Mating | ⇓ | Infanticide | [54,55] |
| Exposure to pups | ⇑ | Nest-building, retrieving, licking, crouching posture | [48,49,50] |
| Experience Multiparous | ⇑ ⇑ ⇑ ⇑ ⇓ | Licking, nursing, contact Defense Suckling Following, grooming Rejection | [60] [61] [64] |
| Environmental enrichment | ⇓ ⇑ | Anxiety Licking, grooming, crouching posture, defense | [65] [66] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coria-Avila, G.A.; Herrera-Covarrubias, D.; García, L.I.; Toledo, R.; Hernández, M.E.; Paredes-Ramos, P.; Corona-Morales, A.A.; Manzo, J. Neurobiology of Maternal Behavior in Nonhuman Mammals: Acceptance, Recognition, Motivation, and Rejection. Animals 2022, 12, 3589. https://doi.org/10.3390/ani12243589
Coria-Avila GA, Herrera-Covarrubias D, García LI, Toledo R, Hernández ME, Paredes-Ramos P, Corona-Morales AA, Manzo J. Neurobiology of Maternal Behavior in Nonhuman Mammals: Acceptance, Recognition, Motivation, and Rejection. Animals. 2022; 12(24):3589. https://doi.org/10.3390/ani12243589
Chicago/Turabian StyleCoria-Avila, Genaro A., Deissy Herrera-Covarrubias, Luis I. García, Rebeca Toledo, María Elena Hernández, Pedro Paredes-Ramos, Aleph A. Corona-Morales, and Jorge Manzo. 2022. "Neurobiology of Maternal Behavior in Nonhuman Mammals: Acceptance, Recognition, Motivation, and Rejection" Animals 12, no. 24: 3589. https://doi.org/10.3390/ani12243589
APA StyleCoria-Avila, G. A., Herrera-Covarrubias, D., García, L. I., Toledo, R., Hernández, M. E., Paredes-Ramos, P., Corona-Morales, A. A., & Manzo, J. (2022). Neurobiology of Maternal Behavior in Nonhuman Mammals: Acceptance, Recognition, Motivation, and Rejection. Animals, 12(24), 3589. https://doi.org/10.3390/ani12243589











