Feed Safety and the Development of Poultry Intestinal Microbiota
Simple Summary
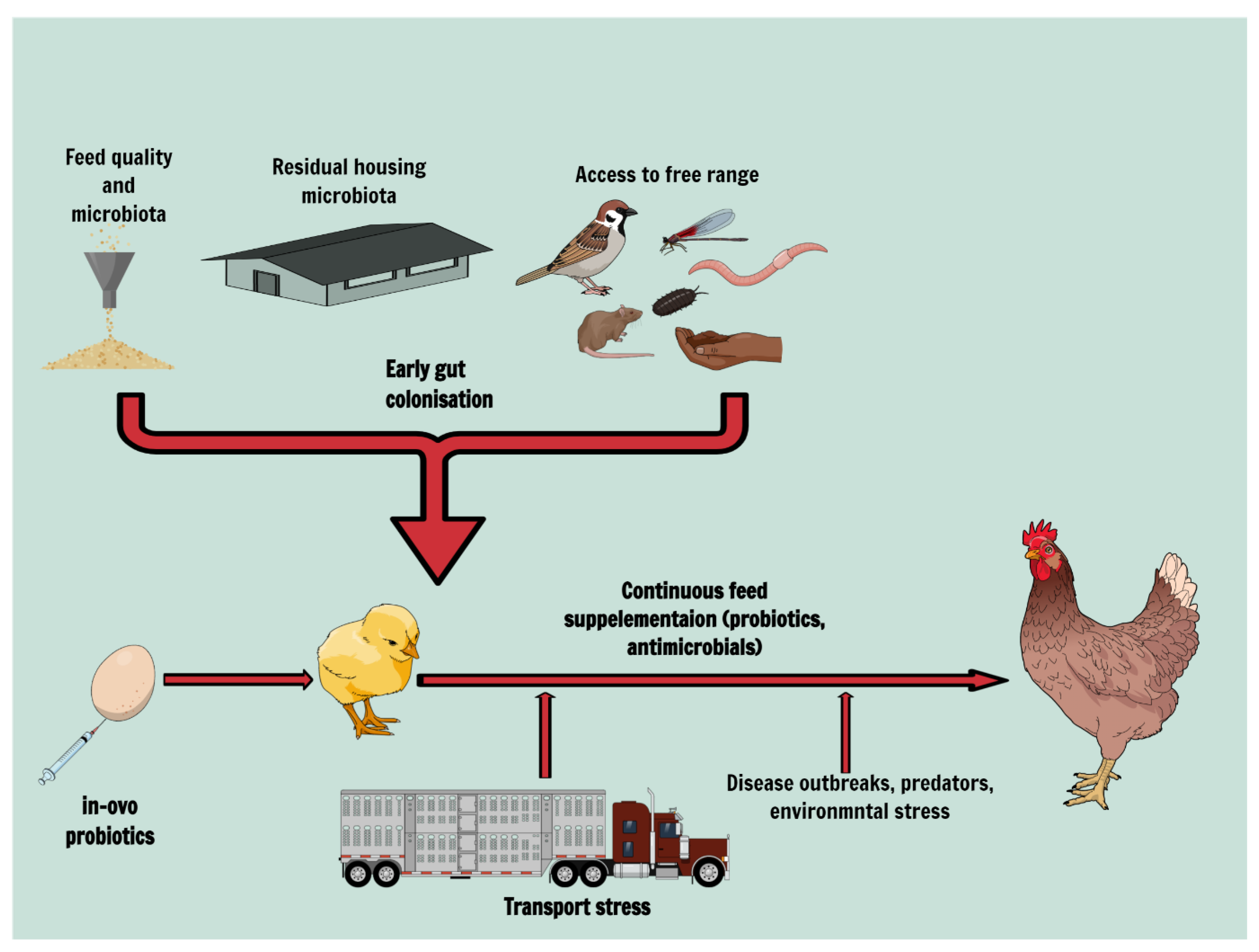
Abstract
1. Background
2. Gut Colonisation
3. Biological Contaminants in Feed
3.1. Microbial Contamination of Feed
3.2. Salmonella
3.3. Campylobacter
3.4. Clostridium Perfringens
3.5. Escherichia Coli
3.6. Bacteriophages
3.7. Feed Microbiota
4. Chemical Contaminants in Feed
4.1. Mycotoxins
4.2. Heavy Metals
4.3. Pesticides and Herbicides
4.4. Other Feed Contaminants
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Edwards, C.A. Determinants and Duration of Impact of Early Gut Bacterial Colonization. Ann. Nutr. Metab. 2017, 70, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Stecher, B.; Hardt, W.D. Mechanisms Controlling Pathogen Colonization of the Gut. Curr. Opin. Microbiol. 2011, 14, 82–91. [Google Scholar] [CrossRef]
- Apajalahti, J.; Kettunen, A.; Graham, H. Characteristics of the Gastrointestinal Microbial Communities, with Special Reference to the Chicken. Worlds Poult. Sci. J. 2004, 60, 223–232. [Google Scholar] [CrossRef]
- Wallace, J.G.; Gohir, W.; Sloboda, D.M. The Impact of Early Life Gut Colonization on Metabolic and Obesogenic Outcomes: What Have Animal Models Shown Us? J. Dev. Orig. Health Dis. 2016, 7, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.L.; Walker, W.A. Early Gut Colonization and Subsequent Obesity Risk. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Trinh, P.; Zaneveld, J.R.; Safranek, S.; Rabinowitz, P.M. One Health Relationships Between Human, Animal, and Environmental Microbiomes: A Mini-Review. Front. Public Health 2018, 6, 235. [Google Scholar] [CrossRef]
- Stanley, D.; Geier, M.S.; Hughes, R.J.; Denman, S.E.; Moore, R.J. Highly Variable Microbiota Development in the Chicken Gastrointestinal Tract. PLoS ONE 2013, 8, e84290. [Google Scholar] [CrossRef]
- Baldwin, S.; Hughes, R.J.; Hao Van, T.T.; Moore, R.J.; Stanley, D. At-Hatch Administration of Probiotic to Chickens Can Introduce Beneficial Changes in Gut Microbiota. PLoS ONE 2018, 13, e0194825. [Google Scholar] [CrossRef]
- Thofner, I.; Sandvang, D.; Aagaard, K.; Poulsen, L.L. Recovery of Probiotic Bacteria from the Intestinal Tract of Broilers after Noninvasive Pre-Hatch Application. Benef. Microbes 2021, 12, 467–477. [Google Scholar] [CrossRef]
- Rehan, I.F.; Rehan, A.F.; Abouelnaga, A.F.; Hussein, M.A.; El-Ghareeb, W.R.; Eleiwa, N.Z.; Elnagar, A.; Batiha, G.E.; Abdelgawad, M.A.; Ghoneim, M.M.; et al. Impact of Dietary Egg Yolk IgY Powder on Behavior, Meat Quality, Physiology, and Intestinal Escherichia coli Colonization of Broiler Chicks. Front. Vet. Sci. 2022, 9, 783094. [Google Scholar] [CrossRef]
- Shealy, N.G.; Yoo, W.; Byndloss, M.X. Colonization Resistance: Metabolic Warfare as a Strategy Against Pathogenic Enterobacteriaceae. Curr. Opin. Microbiol. 2021, 64, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Ducarmon, Q.R.; Zwittink, R.D.; Hornung, B.V.H.; van Schaik, W.; Young, V.B.; Kuijper, E.J. Gut Microbiota and Colonization Resistance against Bacterial Enteric Infection. Microbiol. Mol. Biol. Rev. 2019, 83. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.S.; Chatterjee, R.N.; Raju, M.; Prakash, B.; Rama Rao, S.V.; Yadav, S.P.; Kannan, A. Gut Microbial Composition Differs Extensively among Indian Native Chicken Breeds Originated in Different Geographical Locations and a Commercial Broiler Line, but Breed-Specific, as Well as Across-Breed Core Microbiomes, Are Found. Microorganisms 2021, 9, 391. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.C.; Kil, D.Y.; Sul, W.J. Cecal Microbiome Divergence of Broiler Chickens by Sex and Body Weight. J. Microbiol. 2017, 55, 939–945. [Google Scholar] [CrossRef]
- Sztandarski, P.; Marchewka, J.; Konieczka, P.; Zdanowska-Sasiadek, Z.; Damaziak, K.; Riber, A.B.; Gunnarsson, S.; Horbanczuk, J.O. Gut Microbiota Activity in Chickens from two Genetic Lines and with Outdoor-Preferring, Moderate-Preferring, and Indoor-Preferring Ranging Profiles. Poult. Sci. 2022, 101, 102039. [Google Scholar] [CrossRef]
- Chi, C.; Fan, Y.; Li, C.; Li, Y.; Guo, S.; Li, T.; Buys, N.; Clifton, V.L.; Colditz, P.B.; Yin, C.; et al. Early Gut Microbiota Colonisation of Premature Infants Fed with Breastmilk or Formula with or without Probiotics: A Cohort Study. Nutrients 2021, 13, 4068. [Google Scholar] [CrossRef]
- Nagpal, R.; Tsuji, H.; Takahashi, T.; Nomoto, K.; Kawashima, K.; Nagata, S.; Yamashiro, Y. Gut Dysbiosis Following C-Section Instigates Higher Colonisation of Toxigenic Clostridium Perfringens in Infants. Benef. Microbes 2017, 8, 353–365. [Google Scholar] [CrossRef]
- Wilkinson, N.; Hughes, R.J.; Bajagai, Y.S.; Aspden, W.J.; Hao Van, T.T.; Moore, R.J.; Stanley, D. Reduced Environmental Bacterial Load During Early Development and Gut Colonisation Has Detrimental Health Consequences in Japanese Quail. Heliyon 2020, 6, e03213. [Google Scholar] [CrossRef]
- Donaldson, E.E.; Stanley, D.; Hughes, R.J.; Moore, R.J. The Time-Course of Broiler Intestinal Microbiota Development after Administration of Cecal Contents to Incubating Eggs. PeerJ 2017, 5, e3587. [Google Scholar] [CrossRef]
- Volf, J.; Crhanova, M.; Karasova, D.; Faldynova, M.; Kubasova, T.; Seidlerova, Z.; Sebkova, A.; Zeman, M.; Juricova, H.; Matiasovicova, J.; et al. Eggshell and Feed Microbiota Do Not Represent Major Sources of Gut Anaerobes for Chickens in Commercial Production. Microorganisms 2021, 9, 1480. [Google Scholar] [CrossRef]
- Joat, N.; Van, T.T.H.; Stanley, D.; Moore, R.J.; Chousalkar, K. Temporal Dynamics of Gut Microbiota in Caged Laying Hens: A Field Observation from Hatching to End of Lay. Appl. Microbiol. Biotechnol. 2021, 105, 4719–4730. [Google Scholar] [CrossRef] [PubMed]
- Haberecht, S.; Bajagai, Y.S.; Moore, R.J.; Van, T.T.H.; Stanley, D. Poultry Feeds Carry Diverse Microbial Communities that Influence Chicken Intestinal Microbiota Colonisation and Maturation. AMB Express 2020, 10, 143. [Google Scholar] [CrossRef]
- Lu, T.; Marmion, M.; Ferone, M.; Wall, P.; Scannell, A.G.M. On Farm Interventions to Minimise Campylobacter spp. Contamination in Chicken. Br. Poult. Sci. 2021, 62, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Alves, M.B.; Fonseca, B.B.; Melo, R.T.; Mendonca, E.P.; Nalevaiko, P.C.; Girao, L.C.; Monteiro, G.P.; Silva, P.L.; Rossi, D.A. Feed can be a source of Campylobacter jejuni infection in broilers. Br. Poult. Sci. 2017, 58, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Withenshaw, S.M.; Cawthraw, S.; Gosling, B.; Newton, K.; Oastler, C.E.; Smith, R.P.; Davies, R.H. Risk factor analysis for Salmonella contamination of broiler chicken (Gallus gallus) hatcheries in Great Britain. Prev. Vet. Med. 2021, 196, 105492. [Google Scholar] [CrossRef]
- Ahaduzzaman, M.; Groves, P.J.; Walkden-Brown, S.W.; Gerber, P.F. A molecular based method for rapid detection of Salmonella spp. in poultry dust samples. MethodsX 2021, 8, 101356. [Google Scholar] [CrossRef]
- Hering, J.; Fromke, C.; von Munchhausen, C.; Hartmann, M.; Schneider, B.; Friese, A.; Rosler, U.; Kreienbrock, L.; Hille, K. Cefotaxime-resistant Escherichia coli in broiler farms-A cross-sectional investigation in Germany. Prev. Vet. Med. 2016, 125, 154–157. [Google Scholar] [CrossRef]
- Khan, I.; Bai, Y.; Zha, L.; Ullah, N.; Ullah, H.; Shah, S.R.H.; Sun, H.; Zhang, C. Mechanism of the Gut Microbiota Colonization Resistance and Enteric Pathogen Infection. Front. Cell Infect. Microbiol. 2021, 11, 716299. [Google Scholar] [CrossRef]
- Schumacher, L.L.; Cochrane, R.A.; Huss, A.R.; Gebhardt, J.T.; Woodworth, J.C.; Stark, C.R.; Jones, C.K.; Bai, J.; Main, R.G.; Chen, Q.; et al. Feed batch sequencing to decrease the risk of porcine epidemic diarrhea virus (PEDV) cross-contamination during feed manufacturing. J. Anim. Sci. 2018, 96, 4562–4570. [Google Scholar] [CrossRef]
- Namulawa, V.T.; Mutiga, S.; Musimbi, F.; Akello, S.; Nganga, F.; Kago, L.; Kyallo, M.; Harvey, J.; Ghimire, S. Assessment of Fungal Contamination in Fish Feed from the Lake Victoria Basin, Uganda. Toxins 2020, 12, 233. [Google Scholar] [CrossRef]
- Pereyra, C.M.; Cavaglieri, L.R.; Chiacchiera, S.M.; Dalcero, A.M. Mycobiota and mycotoxins contamination in raw materials and finished feed intended for fattening pigs production in eastern Argentina. Vet. Res. Commun. 2011, 35, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Abdollahia, M.R.; Ravindrana, V.; Svihus, B. Pelleting of broiler diets: An overview with emphasis on pellet quality and nutritional value. Anim. Feed. Sci. Technol. 2013, 179, 1–23. [Google Scholar] [CrossRef]
- Shirota, K.; Katoh, H.; Ito, T.; Otsuki, K. Salmonella contamination in commercial layer feed in Japan. J. Vet. Med. Sci. 2000, 62, 789–791. [Google Scholar] [CrossRef] [PubMed]
- Henderson, W.; Ostendorf, J.; Morehouse, G.L. The relative pathogenicity of some Salmonella serotypes for chicks. Avian Dis. 1960, 4, 103–109. [Google Scholar] [CrossRef]
- Sauli, I.; Danuser, J.; Geeraerd, A.H.; Van Impe, J.F.; Rufenacht, J.; Bissig-Choisat, B.; Wenk, C.; Stark, K.D. Estimating the probability and level of contamination with Salmonella of feed for finishing pigs produced in Switzerland—the impact of the production pathway. Int. J. Food. Microbiol. 2005, 100, 289–310. [Google Scholar] [CrossRef]
- Yang, S.; Wu, Z.; Lin, W.; Xu, L.; Cheng, L.; Zhou, L. Investigations into Salmonella contamination in feed production chain in Karst rural areas of China. Env. Sci Pollut. Res. Int. 2017, 24, 1372–1379. [Google Scholar] [CrossRef]
- Wierup, M.; Kristoffersen, T. Prevention of Salmonella contamination of finished soybean meal used for animal feed by a Norwegian production plant despite frequent Salmonella contamination of raw soy beans, 1994–2012. Acta. Vet. Scand. 2014, 56, 41. [Google Scholar] [CrossRef]
- Gosling, R.J.; Mawhinney, I.; Richardson, K.; Wales, A.; Davies, R. Control of Salmonella and Pathogenic E. coli Contamination of Animal Feed Using Alternatives to Formaldehyde-Based Treatments. Microorganisms 2021, 9, 263. [Google Scholar] [CrossRef]
- Ricke, S.C.; Richardson, K.; Dittoe, D.K. Formaldehydes in feed and their potential interaction with the poultry gastrointestinal tract microbial community-a review. Front. Vet. Sci. 2019, 6, 188. [Google Scholar] [CrossRef]
- Gebhardt, J.T.; Cochrane, R.A.; Woodworth, J.C.; Jones, C.K.; Niederwerder, M.C.; Muckey, M.B.; Stark, C.R.; Tokach, M.D.; DeRouchey, J.M.; Goodband, R.D.; et al. Evaluation of the effects of flushing feed manufacturing equipment with chemically treated rice hulls on porcine epidemic diarrhea virus cross-contamination during feed manufacturing. J. Anim. Sci. 2018, 96, 4149–4158. [Google Scholar] [CrossRef]
- Muckey, M.; Huss, A.R.; Yoder, A.; Jones, C. Research Note: Evaluating the roles of surface sanitation and feed sequencing on mitigating Salmonella Enteritidis contamination on animal food manufacturing equipment. Poult. Sci. 2020, 99, 3841–3845. [Google Scholar] [CrossRef] [PubMed]
- Hakeem, M.J.; Lu, X. Survival and Control of Campylobacter in Poultry Production Environment. Front. Cell Infect. Microbiol. 2020, 10, 615049. [Google Scholar] [CrossRef] [PubMed]
- Corcionivoschi, N.; Gundogdu, O. Foodborne Pathogen Campylobacter. Microorganisms 2021, 9, 1241. [Google Scholar] [CrossRef] [PubMed]
- Richardson, K.E.; Cox, N.A.; Berrang, M.E.; Powell, A.; Weller, C. Recovery of Campylobacter from feed using different enrichment media. J. Environ. Sci. Health B 2021, 56, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Iovine, N.M.; Blaser, M.J. Antibiotics in animal feed and spread of resistant Campylobacter from poultry to humans. Emerg. Infect. Dis. 2004, 10, 1158–1159. [Google Scholar] [CrossRef] [PubMed]
- Heres, L.; Engel, B.; Van Knapen, F.; Wagenaar, J.A.; Urlings, B.A. Effect of fermented feed on the susceptibility for Campylobacter jejuni colonisation in broiler chickens with and without concurrent inoculation of Salmonella enteritidis. Int. J. Food Microbiol. 2003, 87, 75–86. [Google Scholar] [CrossRef]
- Craven, S.E.; Stern, N.J.; Bailey, J.S.; Cox, N.A. Incidence of Clostridium perfringens in broiler chickens and their environment during production and processing. Avian Dis. 2001, 45, 887–896. [Google Scholar] [CrossRef]
- Tessari, E.N.C.; Cardoso, A.L.S.P.; Kanashiro, A.M.I.; Stoppa, G.F.Z.; Luciano, R.L.; de Castro, A.G.M. Analysis of the presence of Clostridium perfringens in feed and raw material used in poultry production. Food Nutr. Sci. 2014, 5, 614–617. [Google Scholar]
- Udhayavel, S.; Thippichettypalayam Ramasamy, G.; Gowthaman, V.; Malmarugan, S.; Senthilvel, K. Occurrence of Clostridium perfringens contamination in poultry feed ingredients: Isolation, identification and its antibiotic sensitivity pattern. Anim. Nutr. 2017, 3, 309–312. [Google Scholar] [CrossRef]
- Ge, B.; LaFon, P.C.; Carter, P.J.; McDermott, S.D.; Abbott, J.; Glenn, A.; Ayers, S.L.; Friedman, S.L.; Paige, J.C.; Wagner, D.D.; et al. Retrospective analysis of Salmonella, Campylobacter, Escherichia coli, and Enterococcus in animal feed ingredients. Foodborne Pathog. Dis. 2013, 10, 684–691. [Google Scholar] [CrossRef]
- Ngai, D.G.; Nyamache, A.K.; Ombori, O. Prevalence and antimicrobial resistance profiles of Salmonella species and Escherichia coli isolates from poultry feeds in Ruiru Sub-County, Kenya. BMC Res. Notes 2021, 14, 41. [Google Scholar] [CrossRef] [PubMed]
- Ezemba, C.C.; Obi, C.P.; Eleanya, L.C.; Udoye, I.; Ezemba, A.S.; Osuala, O.J.; Archibong, E.J. The incidence of Salmonella and E. coli in poultry feeds. J. Curr. Biomed. Res. 2021, 1, 45–56. [Google Scholar]
- da Costa, P.M.; Oliveira, M.; Bica, A.; Vaz-Pires, P.; Bernardo, F. Antimicrobial resistance in Enterococcus spp. and Escherichia coli isolated from poultry feed and feed ingredients. Vet. Microbiol. 2007, 120, 122–131. [Google Scholar] [CrossRef]
- Toribio-Avedillo, D.; Blanch, A.R.; Muniesa, M.; Rodriguez-Rubio, L. Bacteriophages as Fecal Pollution Indicators. Viruses 2021, 13, 1089. [Google Scholar] [CrossRef]
- McMinn, B.R.; Ashbolt, N.J.; Korajkic, A. Bacteriophages as indicators of faecal pollution and enteric virus removal. Lett. Appl. Microbiol. 2017, 65, 11–26. [Google Scholar] [CrossRef]
- Jofre, J.; Blanch, A.R.; Lucena, F.; Muniesa, M. Bacteriophages infecting Bacteroides as a marker for microbial source tracking. Water Res. 2014, 55, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ji, M.; Liu, Z.; Sun, K.; Li, Z.; Fan, X.; Li, Q. Bacteriophages in water pollution control: Advantages and limitations. Front. Environ. Sci. Eng. 2021, 15, 84. [Google Scholar] [CrossRef]
- Maciorowski, K.G.; Pillai, S.D.; Ricke, S.C. Presence of bacteriophages in animal feed as indicators of fecal contamination. J. Environ. Sci. Health B 2001, 36, 699–708. [Google Scholar] [CrossRef]
- Wu, H.; Rui, X.; Li, W.; Xiao, Y.; Zhou, J.; Dong, M. Whole-grain oats (Avena sativa L.) as a carrier of lactic acid bacteria and a supplement rich in angiotensin I-converting enzyme inhibitory peptides through solid-state fermentation. Food Funct. 2018, 9, 2270–2281. [Google Scholar] [CrossRef]
- Carrizo, S.L.; Montes de Oca, C.E.; Laino, J.E.; Suarez, N.E.; Vignolo, G.; LeBlanc, J.G.; Rollan, G. Ancestral Andean grain quinoa as source of lactic acid bacteria capable to degrade phytate and produce B-group vitamins. Food Res. Int. 2016, 89, 488–494. [Google Scholar] [CrossRef]
- Xia, Y.; Kong, Y.; Seviour, R.; Yang, H.E.; Forster, R.; Vasanthan, T.; McAllister, T. In situ identification and quantification of starch-hydrolyzing bacteria attached to barley and corn grain in the rumen of cows fed barley-based diets. FEMS Microbiol. Ecol. 2015, 91, fiv077. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pan, D.; Mionetto, A.; Tiscornia, S.; Bettucci, L. Endophytic bacteria from wheat grain as biocontrol agents of Fusarium graminearum and deoxynivalenol production in wheat. Mycotoxin Res. 2015, 31, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Ahlawat, O.P.; Yadav, D.; Kashyap, P.L.; Khippal, A.; Singh, G. Wheat endophytes and their potential role in managing abiotic stress under changing climate. J. Appl. Microbiol. 2021, 132, 2501–2520. [Google Scholar] [CrossRef]
- Abdallah, M.F.; De Boevre, M.; Landschoot, S.; De Saeger, S.; Haesaert, G.; Audenaert, K. Fungal Endophytes Control Fusarium graminearum and Reduce Trichothecenes and Zearalenone in Maize. Toxins 2018, 10, 493. [Google Scholar] [CrossRef]
- Makar, O.; Kuzniar, A.; Patsula, O.; Kavulych, Y.; Kozlovskyy, V.; Wolinska, A.; Skorzynska-Polit, E.; Vatamaniuk, O.; Terek, O.; Romanyuk, N. Bacterial Endophytes of Spring Wheat Grains and the Potential to Acquire Fe, Cu, and Zn under Their Low Soil Bioavailability. Biology 2021, 10, 409. [Google Scholar] [CrossRef] [PubMed]
- Ruiz Mostacero, N.; Castelli, M.V.; Barolo, M.I.; Amigot, S.L.; Fulgueira, C.L.; Lopez, S.N. Fungal endophytes in Peperomia obtusifolia and their potential as inhibitors of chickpea fungal pathogens. World J. Microbiol. Biotechnol. 2021, 37, 14. [Google Scholar] [CrossRef] [PubMed]
- Abrunhosa, L.; Morales, H.; Soares, C.; Calado, T.; Vila-Cha, A.S.; Pereira, M.; Venancio, A. A Review of Mycotoxins in Food and Feed Products in Portugal and Estimation of Probable Daily Intakes. Crit. Rev. Food Sci. Nutr. 2016, 56, 249–265. [Google Scholar] [CrossRef]
- Pleadin, J.; Frece, J.; Markov, K. Mycotoxins in food and feed. Adv. Food Nutr. Res. 2019, 89, 297–345. [Google Scholar]
- Guerre, P. Worldwide Mycotoxins Exposure in Pig and Poultry Feed Formulations. Toxins 2016, 8, 350. [Google Scholar] [CrossRef]
- Afsah-Hejri, L.; Jinap, S.; Hajeb, P.; Radu, S.; Shakibazadeh, S. A Review on Mycotoxins in Food and Feed: Malaysia Case Study. Compr. Rev. Food Sci. Food Saf. 2013, 12, 629–651. [Google Scholar] [CrossRef]
- Liew, W.P.; Mohd-Redzwan, S. Mycotoxin: Its Impact on Gut Health and Microbiota. Front. Cell Infect. Microbiol. 2018, 8, 60. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Meng, L.; Liu, H.; Wang, J.; Zheng, N. The Compromised Intestinal Barrier Induced by Mycotoxins. Toxins 2020, 12, 619. [Google Scholar] [CrossRef] [PubMed]
- Robert, H.; Payros, D.; Pinton, P.; Theodorou, V.; Mercier-Bonin, M.; Oswald, I.P. Impact of mycotoxins on the intestine: Are mucus and microbiota new targets? J. Toxicol. Environ. Health B Crit. Rev. 2017, 20, 249–275. [Google Scholar] [CrossRef] [PubMed]
- Piglowski, M. Heavy Metals in Notifications of Rapid Alert System for Food and Feed. Int. J. Environ. Res. Public Health 2018, 15, 365. [Google Scholar] [CrossRef]
- Guntinas, M.B.; Semeraro, A.; Wysocka, I.; Cordeiro, F.; Quetel, C.; Emteborg, H.; Charoud-Got, J.; Linsinger, T.P. Proficiency test for the determination of heavy metals in mineral feed. The importance of correctly selecting the certified reference materials during method validation. Food Addit. Contam. Part A 2011, 28, 1534–1546. [Google Scholar] [CrossRef][Green Version]
- Eskandari, M.H.; Pakfetrat, S. Aflatoxins and heavy metals in animal feed in Iran. Food Addit. Contam. Part B Surveill. 2014, 7, 202–207. [Google Scholar] [CrossRef]
- Kabeer, M.S.; Hameed, I.; Kashif, S.U.; Khan, M.; Tahir, A.; Anum, F.; Khan, S.; Raza, S. Contamination of heavy metals in poultry eggs: A study presenting relation between heavy metals in feed intake and eggs. Arch. Environ. Occup. Health 2021, 76, 220–232. [Google Scholar] [CrossRef]
- Yasotha, A.; Dabade, D.S.; Singh, V.P.; Sivakumar, T. Risk assessment of heavy metals in milk from cows reared around industrial areas in India. Environ. Geochem. Health 2021, 43, 1799–1815. [Google Scholar] [CrossRef]
- Yang, N.; Wang, H.; Wang, H.; Wang, Z.; Ran, J.; Guo, S.; Peng, Y. Screening maize (Zea mays L.) varieties with low accumulation of cadmium, arsenic, and lead in edible parts but high accumulation in other parts: A field plot experiment. Environ. Sci. Pollut. Res. Int. 2021, 28, 33583–33598. [Google Scholar] [CrossRef]
- Jia, H.; Ren, H.; Maita, M.; Satoh, S.; Endo, H.; Hayashi, T. Development of functional fish feed with natural ingredients to control heavy metals. Toxicol. Mech. Methods 2006, 16, 411–417. [Google Scholar] [CrossRef]
- Utomo, S.W.; Rahmadina, F.; Wispriyono, B.; Kusnoputranto, H.; Asyary, A. Metal Contents of Lake Fish in Area Close to Disposal of Industrial Waste. J. Environ. Public Health 2021, 2021, 6675374. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Wang, X.; Xu, X.; Cai, R.; Xie, S. Effects of heavy metals on the bioaccumulation, excretion and gut microbiome of black soldier fly larvae (Hermetia illucens). Ecotoxicol. Environ. Saf. 2020, 192, 110323. [Google Scholar] [CrossRef] [PubMed]
- Holzel, C.S.; Muller, C.; Harms, K.S.; Mikolajewski, S.; Schafer, S.; Schwaiger, K.; Bauer, J. Heavy metals in liquid pig manure in light of bacterial antimicrobial resistance. Environ. Res. 2012, 113, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Rilstone, V.; Vignale, L.; Craddock, J.; Cushing, A.; Filion, Y.; Champagne, P. The role of antibiotics and heavy metals on the development, promotion, and dissemination of antimicrobial resistance in drinking water biofilms. Chemosphere 2021, 282, 131048. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Gunn, L.; Wall, P.; Fanning, S. Antimicrobial resistance and its association with tolerance to heavy metals in agriculture production. Food Microbiol. 2017, 64, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.B.; Dancy, B.C.R.; Horton, C.L.; Lee, Y.S.; Madejczyk, M.S.; Xu, Z.Z.; Ackermann, G.; Humphrey, G.; Palacios, G.; Knight, R.; et al. Exposure to toxic metals triggers unique responses from the rat gut microbiota. Sci. Rep. 2018, 8, 6578. [Google Scholar] [CrossRef] [PubMed]
- Giller, K.E.; Witter, E.; Mcgrath, S.P. Toxicity of heavy metals to microorganisms and microbial processes in agricultural soils: A review. Soil Biol. Biochem. 1988, 30, 1389–1414. [Google Scholar] [CrossRef]
- Islam, E.; Yang, X.E.; He, Z.L.; Mahmood, Q. Assessing potential dietary toxicity of heavy metals in selected vegetables and food crops. J. Zhejiang Univ. Sci. B 2007, 8, 1–13. [Google Scholar] [CrossRef]
- Mondal, N.K. Prevalence of Arsenic in chicken feed and its contamination pattern in different parts of chicken flesh: A market basket study. Environ. Monit. Assess. 2020, 192, 590. [Google Scholar] [CrossRef]
- van der Lee, M.K.; van der Weg, G.; Traag, W.A.; Mol, H.G. Qualitative screening and quantitative determination of pesticides and contaminants in animal feed using comprehensive two-dimensional gas chromatography with time-of-flight mass spectrometry. J. Chromatogr. A. 2008, 1186, 325–339. [Google Scholar] [CrossRef]
- Walorczyk, S.; Drozdzynski, D. Improvement and extension to new analytes of a multi-residue method for the determination of pesticides in cereals and dry animal feed using gas chromatography-tandem quadrupole mass spectrometry revisited. J. Chromatogr. A. 2012, 1251, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Lim, W.; Song, G. Mediation of oxidative stress toxicity induced by pyrethroid pesticides in fish. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2020, 234, 108758. [Google Scholar] [CrossRef] [PubMed]
- Moebus, S.; Boedeker, W. Case Fatality as an Indicator for the Human Toxicity of Pesticides-A Systematic Scoping Review on the Availability and Variability of Severity Indicators of Pesticide Poisoning. Int. J. Environ. Res. Public Health 2021, 18, 8307. [Google Scholar] [CrossRef] [PubMed]
- Utembe, W.; Kamng′ona, A.W. Gut microbiota-mediated pesticide toxicity in humans: Methodological issues and challenges in the risk assessment of pesticides. Chemosphere 2021, 271, 129817. [Google Scholar] [CrossRef] [PubMed]
- De Boer, A. Labelling and denominating lung toxicity effects of pesticides. Curr. Opin. Pulm. Med. 2021, 27, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Rueda-Ruzafa, L.; Cruz, F.; Roman, P.; Cardona, D. Gut microbiota and neurological effects of glyphosate. Neurotoxicology 2019, 75, 1–8. [Google Scholar] [CrossRef]
- Sorensen, M.T.; Poulsen, H.D.; Katholm, C.L.; Hojberg, O. Review: Feed residues of glyphosate—potential consequences for livestock health and productivity. Animal 2021, 15, 100026. [Google Scholar] [CrossRef]
- Vicini, J.L.; Reeves, W.R.; Swarthout, J.T.; Karberg, K.A. Glyphosate in livestock: Feed residues and animal health. J. Anim. Sci. 2019, 97, 4509–4518. [Google Scholar] [CrossRef]
- Peeters, L.E.; Daeseleire, E.; Devreese, M.; Rasschaert, G.; Smet, A.; Dewulf, J.; Heyndrickx, M.; Imberechts, H.; Haesebrouck, F.; Butaye, P.; et al. Residues of chlortetracycline, doxycycline and sulfadiazine-trimethoprim in intestinal content and feces of pigs due to cross-contamination of feed. BMC Vet. Res. 2016, 12, 209. [Google Scholar] [CrossRef]
- Przenioslo-Siwczynska, M.; Patyra, E.; Grelik, A.; Chylek-Purchala, M.; Kozak, B.; Kwiatek, K. Contamination of Animal Feed with Undeclared Tetracyclines-Confirmatory Analysis by Liquid Chromatography-Mass Spectrometry after Microbiological Plate Test. Molecules 2020, 25, 2162. [Google Scholar] [CrossRef]
- Iammarino, M.; dell’Oro, D.; Bortone, N.; Chiaravalle, A.E. Beta Emitter Radionuclides ((90)Sr) Contamination in Animal Feed: Validation and Application of a Radiochemical Method by Ultra Low Level Liquid Scintillation Counting. Ital. J. Food Saf. 2015, 4, 4531. [Google Scholar] [PubMed]
- Torres, J.P.; Leite, C.; Krauss, T.; Weber, R. Landfill mining from a deposit of the chlorine/organochlorine industry as source of dioxin contamination of animal feed and assessment of the responsible processes. Environ. Sci. Pollut. Res. Int. 2013, 20, 1958–1965. [Google Scholar] [CrossRef] [PubMed]
- Pinto, B.; Garritano, S.L.; Cristofani, R.; Ortaggi, G.; Giuliano, A.; Amodio-Cocchieri, R.; Cirillo, T.; De Giusti, M.; Boccia, A.; Reali, D. Monitoring of polychlorinated biphenyl contamination and estrogenic activity in water, commercial feed and farmed seafood. Environ. Monit. Assess. 2008, 144, 445–453. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stanley, D.; Bajagai, Y.S. Feed Safety and the Development of Poultry Intestinal Microbiota. Animals 2022, 12, 2890. https://doi.org/10.3390/ani12202890
Stanley D, Bajagai YS. Feed Safety and the Development of Poultry Intestinal Microbiota. Animals. 2022; 12(20):2890. https://doi.org/10.3390/ani12202890
Chicago/Turabian StyleStanley, Dragana, and Yadav Sharma Bajagai. 2022. "Feed Safety and the Development of Poultry Intestinal Microbiota" Animals 12, no. 20: 2890. https://doi.org/10.3390/ani12202890
APA StyleStanley, D., & Bajagai, Y. S. (2022). Feed Safety and the Development of Poultry Intestinal Microbiota. Animals, 12(20), 2890. https://doi.org/10.3390/ani12202890






