Effect of a Single and Triple Dose of Levamisole on Hematological Parameters in Controlled Inflammation Model
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Animals
2.2. Experimental Pleuritis Model
- IP—Control inflammation—18 females. Rats received 0.15 mL of 1% of carrageenan solution intrapleurally. Blood samples were collected at 24, 48, and 72 h from baseline.
- LMS I 24—6 females. Induction of pleuritis with subsequent single administration (IP) of levamisole at a dose of 2.5 mg/kg bw, 2 h after inflammation induction. Blood samples were collected at 24 h from baseline.
- LMS I 48—6 females. Induction of pleuritis with subsequent single administration (IP) of levamisole at a dose of 2.5 mg/kg bw 2 h after inflammation induction. Blood samples were collected at 48 h from baseline.
- LMS I 72—6 females. Induction of pleuritis with subsequent single administration (IP) of levamisole at a dose of 2.5 mg/kg bw 2 h after inflammation induction. Blood samples were collected at 72 h from baseline.
- LMS II 48—6 females. Induction of pleuritis with subsequent double administration (IP) of levamisole at a dose of 2.5 mg/kg bw 2 and 24 h after inflammation induction. Blood samples were collected at 48 h from baseline.
- LMS III 72—6 females. Induction of pleuritis with subsequent triple administration (IP) of levamisole at a dose of 2.5 mg/kg bw 2, 24 and 48 h after inflammation induction. Blood samples were collected at 72 h from baseline.
2.3. Blood Sample Collection and Hematological Analysis
2.4. Histological Analysis
2.5. Statistical Analysis
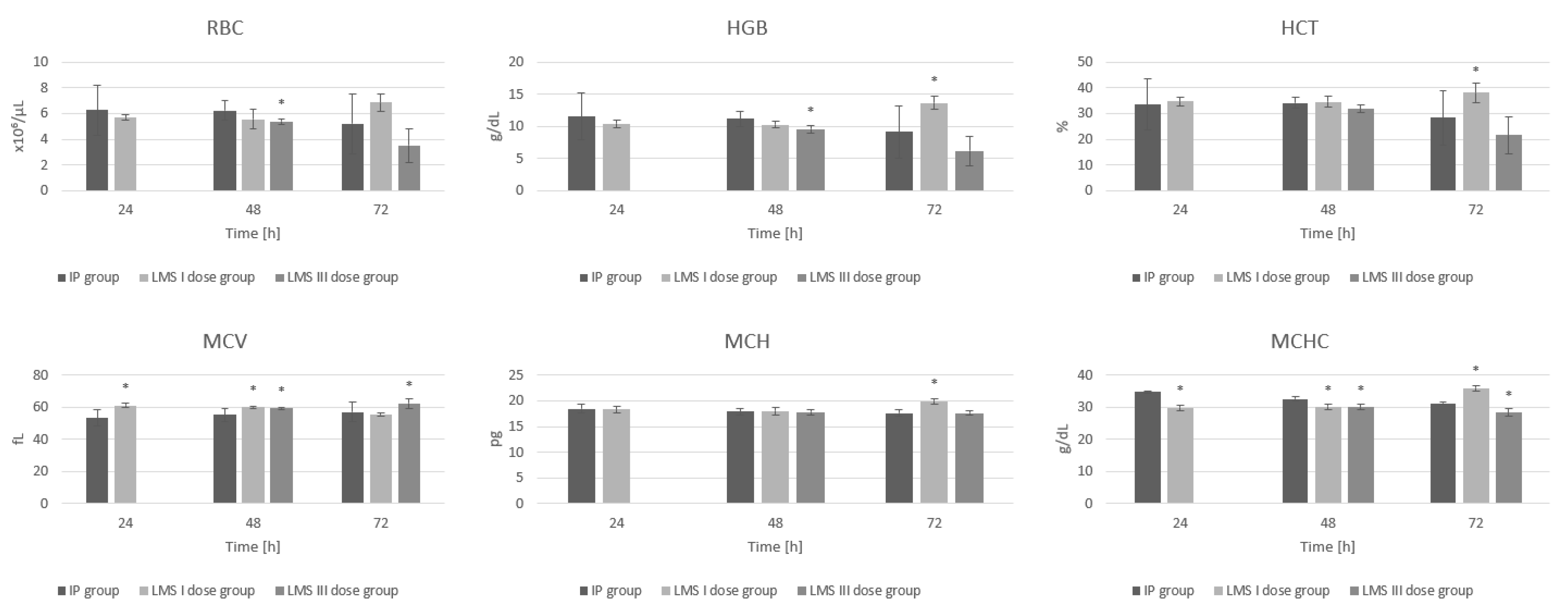
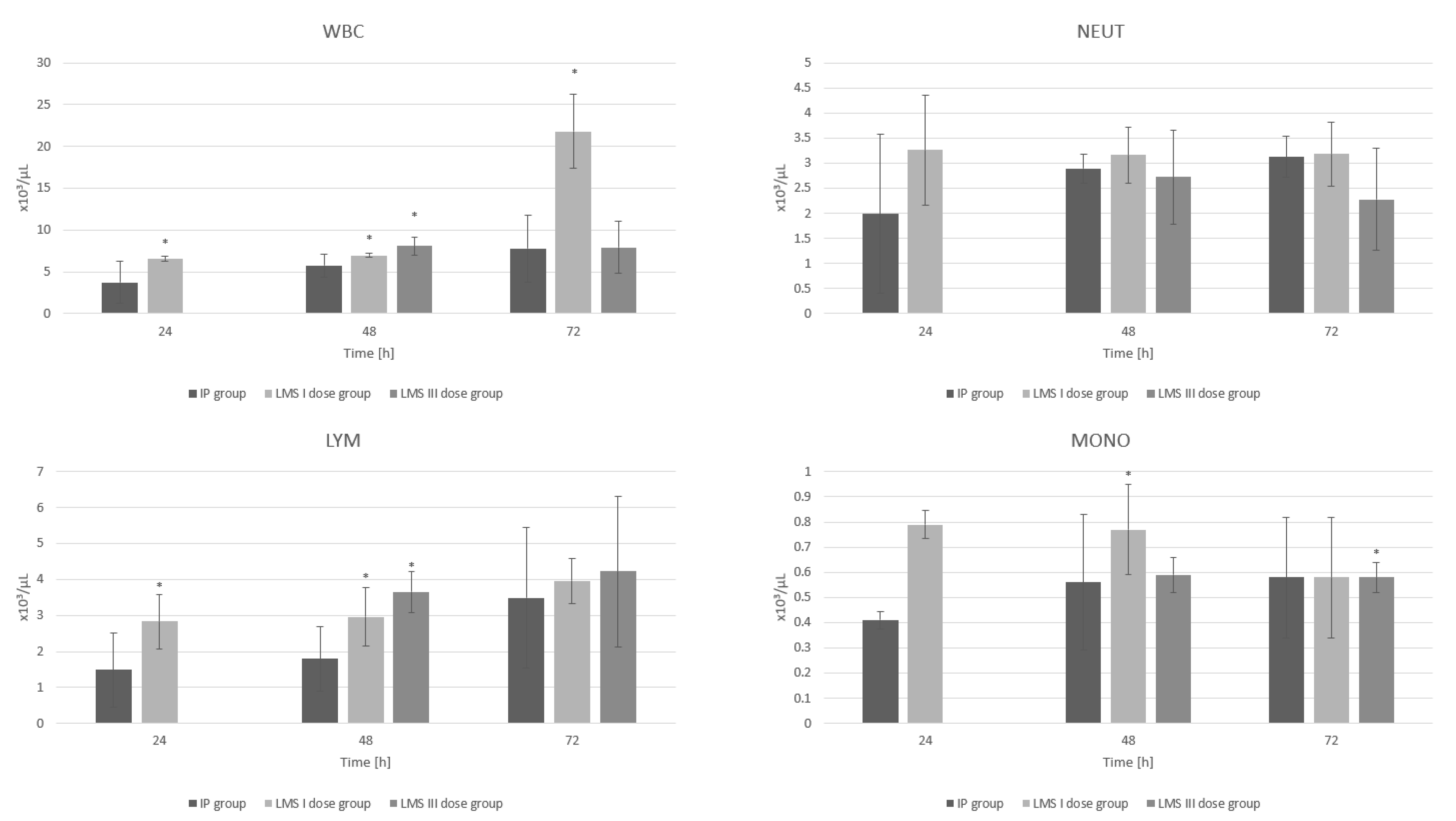
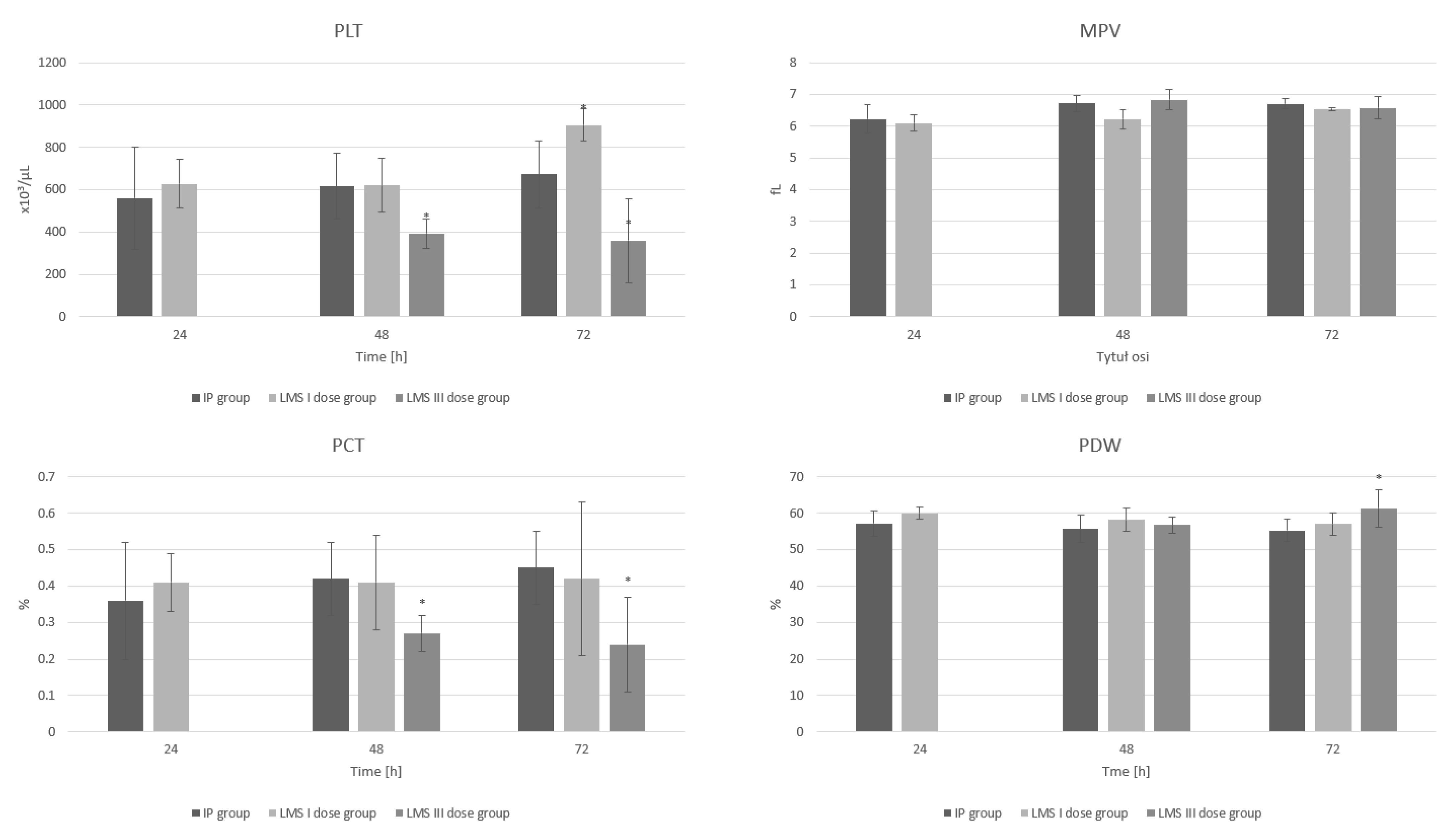
3. Results
3.1. Red Blood Cell System
3.2. White Blood Cell System
3.3. Platelets
3.4. Histological Evaluation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gross, R.L.; Brucker, J.; Bahce-Altuntas, A.; Abadi, M.A.; Lipoff, J.; Kotlyar, D.; Barland, P.; Putterman, C. A novel cutaneous vasculitis syndrome induced by levamisole-contaminated cocaine. Clin. Rheumatol. 2011, 30, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Dy, I.; Pokuri, V.; Olichney, J.; Wiernik, P. Levamisole-adulterated in cocaine causing agranulocytosis, vasculopathy, and acquired protein S deficiency. Ann. Hematol. 2012, 91, 477–478. [Google Scholar] [CrossRef] [PubMed]
- Scheinfeld, N.; Rosenberg, J.D.; Weinberg, J.M. Levamisole in dermatology: A review. Am. J. Clin. Dermatol. 2004, 5, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, J.A.; Oyer, R.J.; Patel, N.R.; Jacquet, G.A.; Bornikova, L.; Thienelt, C.; Shriver, D.A.; Shockley, L.W.; Wilson, M.L.; Hurlbut, K.M.; et al. A Confirmed Case of Agranulocytosis after Use of Cocaine Contaminated with Levamisole. J. Med Toxicol. 2010, 6, 160–164. [Google Scholar] [CrossRef]
- Ince, S.; Kozan, E.; Kucukkurt, I.; Bacak, E. The effect of levamisole and levamisole+vitamin C on oxidative damage in rats naturally infected with Syphacia muris. Exp. Parasitol. 2010, 124, 448–452. [Google Scholar] [CrossRef]
- Martin, R.J.; Bai, G.; Clark, C.L.; Robertson, A.P. Methyridine (2-[2-methoxyethyl]-pyridine]) and levamisole activate different ACh receptor subtypes in nematode parasites: A new lead for levamisole-resistance. J. Cereb. Blood Flow Metab. 2003, 140, 1068–1076. [Google Scholar] [CrossRef]
- Rehni, A.K.; Singh, T.G. Levamisole-induced reduction in seizure threshold: A possible role of nicotinic acetylcholine receptor-mediated pathway. Naunyn-Schmiedebergs Arch. fur Exp. Pathol. und Pharmakol. 2010, 382, 279–285. [Google Scholar] [CrossRef]
- Walker, J.; Hoekstra, R.; Roos, M.H.; Wiley, L.J.; Weiss, A.S.; Sangster, N.C.; Tait, A. Cloning and structural analysis of partial acetylcholine receptor subunit genes from the parasitic nematode Teladorsagia circumcincta. Veter.- Parasitol. 2001, 97, 329–335. [Google Scholar] [CrossRef]
- Marshall, J.L.; Haller, D.G.; De Gramont, A.; Hochster, H.S.; Lenz, H.-J.; Ajani, J.A.; Goldberg, R.M. Adjuvant Therapy for Stage II and III Colon Cancer: Consensus Report of the International Society of Gastrointestinal Oncology. Gastrointest. Cancer Res. GCR 2007, 1, 146–154. [Google Scholar]
- Kayatas, M. Levamisole Treatment Enhances Protective Antibody Response to Hepatitis B Vaccination in Hemodialysis Patients. Artif. Organs 2002, 26, 492–496. [Google Scholar] [CrossRef]
- Hibi, D.; Imazawa, T.; Kijima, A.; Suzuki, Y.; Ishii, Y.; Jin, M.; Umemura, T.; Nishikawa, A. Investigation of carcinogenicity for levamisole administered in the diet to F344 rats. Food Chem. Toxicol. 2010, 48, 3321–3326. [Google Scholar] [CrossRef] [PubMed]
- Won, T.H.; Park, S.Y.; Kim, B.S.; Seo, P.S.; Park, S.D. Levamisole Monotherapy for Oral Lichen Planus. Ann. Dermatol. 2009, 21, 250–254. [Google Scholar] [CrossRef]
- Chen, L.-Y.; Lin, Y.-L.; Chiang, B.-L. Levamisole enhances immune response by affecting the activation and maturation of human monocyte-derived dendritic cells. Clin. Exp. Immunol. 2008, 151, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Janjatović, A.K.; Lacković, G.; Božić, F.; Popović, M.; Valpotić, I. Levamisole synergizes proliferation of intestinal IgA+ cells in weaned pigs immunized with vaccine candidate F4ac+ nonenterotoxigenic Escherichia coli strain. J. Vet. Pharmacol. Ther. 2008, 31, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Figer, A.; Nissan, A.; Shani, A.; Borovick, R.; Stiener, M.; Baras, M.; Freund, H.R.; Sulkes, A.; Stojadinovic, A.; Peretz, T. Mature Results of a Prospective Randomized Trial Comparing 5-Flourouracil with Leucovorin to 5-Flourouracil with Levamisole as Adjuvant Therapy of Stage II and III Colorectal Cancer- The Israel Cooperative Oncology Group (ICOG) Study. J. Cancer 2011, 2, 177–185. [Google Scholar] [CrossRef]
- Tsai, W.-S.; Hsieh, P.-S.; Yeh, C.-Y.; Chiang, J.-M.; Tang, R.; Chen, J.-S.; Changchien, C.R.; Wang, J.Y. Long-term survival benefits of adjuvant chemotherapy by decreasing incidence of tumor recurrence without delaying relapse in stage III colorectal cancer. Int. J. Color. Dis. 2011, 26, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Dahl, O.; Fluge, Ø.; Carlsen, E.; Wiig, J.N.; Myrvold, H.E.; Vonen, B.; Podhorny, N.; Bjerkeset, O.; Eide, T.J.; Halvorsen, T.B.; et al. Final results of a randomised phase III study on adjuvant chemotherapy with 5 FU and levamisol in colon and rectum cancer stage II and III by the Norwegian Gastrointestinal Cancer Group. Acta Oncol. 2009, 48, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Ramanadham, M.; Nageshwari, B. Anti-proliferative effect of levamisole on human myeloma cell lines in vitro. J. Immunotoxicol. 2010, 7, 327–332. [Google Scholar] [CrossRef]
- Artwohl, M.; Hölzenbein, T.; Wagner, L.; Freudenthaler, A.; Waldhäusl, W.; Baumgartner-Parzer, S.M. Levamisole induced apoptosis in cultured vascular endothelial cells. Br. J. Pharmacol. 2000, 131, 1577–1583. [Google Scholar] [CrossRef]
- Mota, A.; Silva, P.; Neves, D.; Lemos, C.; Calhau, C.; Torres, D.; Martel, F.; Fraga, H.; Ribeiro, L.; Alçada, M.N.M.P.; et al. Characterization of rat heart alkaline phosphatase isoenzymes and modulation of activity. Braz. J. Med. Biol. Res. 2008, 41, 600–609. [Google Scholar] [CrossRef]
- de Medina, F.S.; Martínez-Augustin, O.; González, R.; Ballester, I.; Nieto, A.; Gálvez, J.; Zarzuelo, A. Induction of alkaline phosphatase in the inflamed intestine: A novel pharmacological target for inflammatory bowel disease. Biochem. Pharmacol. 2004, 68, 2317–2326. [Google Scholar] [CrossRef] [PubMed]
- Saif, M.W.; Alexander, D.; Wicox, C.M. Serum Alkaline Phosphatase Level as a Prognostic Tool in Colorectal Cancer: A Study of 105 patients. J. Appl. Res. 2005, 5, 88–95. [Google Scholar] [PubMed]
- Purzyc, L.; Całkosiński, I. Ecto-ATPase from rat lymphocytes--in vivo studies on the influence of levamisole. Pol. J. Pharmacol. 1998, 50, 239–251. [Google Scholar] [PubMed]
- Purzyc, L.; Całkosiński, I. Influence of Levamisole, Nitrogranulogen and Dietyldithiocarbamate on the activity of ecto-ATPase from rats lymphocytes. Acta Pol. Pharm. 1998, 55, 41–47. [Google Scholar]
- Obmińska-Domoradzka, B.; Całkosiński, I. Modulation of humoral response in rats by levamisole, mechlorethamine and sodium diethyldithiocarbamate. Acta Pol. Pharm.-Drug Res. 1994, 51, 95–99. [Google Scholar]
- Gostomska-Pampuch, K.; Ostrowska, A.; Kuropka, P.; Dobrzyński, M.; Ziółkowski, P.; Kowalczyk, A.; Łukaszewicz, E.; Gamian, A.; Całkosiński, I. Protective effects of levamisole, acetylsalicylic acid, and α-tocopherol against dioxin toxicity measured as the expression of AhR and COX-2 in a chicken embryo model. Histochem. Cell Biol. 2017, 147, 523–536. [Google Scholar] [CrossRef]
- Całkosiński, I.; Majda, J.; Terlecki, G.; Gostomska-Pampuch, K.; Małolepsza-Jarmołowska, K.; Sobolewska, S.; Całkosińska, A.; Kumala, A.; Gamian, A. Dynamic Analysis of Changes of Protein Levels and Selected Biochemical Indices in Rat Serum in the Course of Experimental Pleurisy. Inflammation 2016, 39, 1076–1089. [Google Scholar] [CrossRef][Green Version]
- Całkosiński, I.; Rosińczuk-Tonderys, J.; Bazan, J.; Dzierzba, K.; Całkosińska, M.; Majda, J.; Dobrzyński, M.; Bronowicka-Szydełko, A. The Influence of 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) on Hematological Parameters During Experimentally Induced Pleuritis in Rats. Inflammation 2013, 36, 387–404. [Google Scholar] [CrossRef][Green Version]
- Całkosiński, I.; Dobrzyński, M.; Całkosińska, M.; Seweryn, E.; Bronowicka-Szydełko, A.; Dzierzba, K.; Ceremuga, I.; Gamian, A. Characterization of an inflammatory response. Postepy Hig. I Med. Dosw. 2009, 63, 395–408. [Google Scholar]
- Calkosinski, I.; Rosinczuk-Tonderys, J.; Dobrzynski, M.; Palka, L.; Bazan, J. Occurrence of Disseminated Intravascular Coagulation in 2,3,7,8-Tetrachlorodibenzo-p-Dioxin-Induced Pneumonia in the Rat. Atherosclerosis 2013, 788, 283–292. [Google Scholar] [CrossRef]


| t [h] | RBC | HGB | HCT | WBC | NEUT | LYM | PLT |
|---|---|---|---|---|---|---|---|
| IP | r = −0.256 | r = −0.311 | r = −0.259 | r = 0.533 | r = −0.137 | r = 0.338 | r = 0.256 |
| p = 0.305 | p = 0.209 | p = 0.300 | p = 0.023 | p = 0.589 | p = 0.170 | p = 0.305 | |
| LMS I dose | r = 0.599 | r = 0.776 | r = 0.471 | r = 0.833 | r = −0.044 | r = 0.648 | r = 0.687 |
| p = 0.009 | p < 0.001 | p = 0.049 | p < 0.001 | p = 0.861 | p = 0.004 | p = 0.002 | |
| LMS III doses | r = −0.740 | r = −0.771 | r = −0.777 | r = 0.301 | r = −0.669 | r = 0.734 | r = −0.629 |
| p < 0.001 | p < 0.001 | p < 0.001 | p = 0.225 | p = 0.002 | p = 0.001 | p = 0.005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuropka, P.; Leśków, A.; Małolepsza-Jarmołowska, K.; Dobrzyński, M.; Tarnowska, M.; Majda, J.; Janeczek, M.; Żybura-Wszoła, K.; Gamian, A. Effect of a Single and Triple Dose of Levamisole on Hematological Parameters in Controlled Inflammation Model. Animals 2022, 12, 2110. https://doi.org/10.3390/ani12162110
Kuropka P, Leśków A, Małolepsza-Jarmołowska K, Dobrzyński M, Tarnowska M, Majda J, Janeczek M, Żybura-Wszoła K, Gamian A. Effect of a Single and Triple Dose of Levamisole on Hematological Parameters in Controlled Inflammation Model. Animals. 2022; 12(16):2110. https://doi.org/10.3390/ani12162110
Chicago/Turabian StyleKuropka, Piotr, Anna Leśków, Katarzyna Małolepsza-Jarmołowska, Maciej Dobrzyński, Małgorzata Tarnowska, Jacek Majda, Maciej Janeczek, Katarzyna Żybura-Wszoła, and Andrzej Gamian. 2022. "Effect of a Single and Triple Dose of Levamisole on Hematological Parameters in Controlled Inflammation Model" Animals 12, no. 16: 2110. https://doi.org/10.3390/ani12162110
APA StyleKuropka, P., Leśków, A., Małolepsza-Jarmołowska, K., Dobrzyński, M., Tarnowska, M., Majda, J., Janeczek, M., Żybura-Wszoła, K., & Gamian, A. (2022). Effect of a Single and Triple Dose of Levamisole on Hematological Parameters in Controlled Inflammation Model. Animals, 12(16), 2110. https://doi.org/10.3390/ani12162110








