1. Introduction
Pasteurella multocida (Pm) is a highly versatile opportunistic pathogen that causes infections in a wide range of domestic and wild animals and humans [
1]. It is found mainly in the oral cavity, nasopharyngeal and upper respiratory tract of animals [
2]. Goats are distributed worldwide and provide meat and milk for human consumption and fiber [
3]. Pasteurellosis causes two significant diseases in goats: pneumonic pasteurellosis and systemic pasteurellosis [
4]. The high incidence and rapid transmission of the diseases cause substantial economic losses to the goat industry.
The symptoms of Pm infection in goats usually begin with fever, lethargy, anorexia and edema with copious salivation, lacrimation, nasal discharge, coughing and dullness. These symptoms are rapidly followed by respiratory distress, septic shock with widespread hemorrhaging and death [
4,
5]. In addition, hematological, biochemical and pathological changes were observed [
4,
6]. Pm-infected goats showed a significant decrease in hemoglobin count and significant increase in white blood cells, neutrophil and monocyte counts, respectively. Infected goats revealed significantly lower concentrations of Ca and Mg but a considerably higher concentration of P in serum than healthy goats [
4]. Microscopical examination of the lungs of Pm-infected goats indicated that goats with acute pneumonia showed edema and dense infiltration of neutrophils in the alveolar spaces as well as severely congested inter-alveolar capillaries [
5]. Neutrophils, alveolar macrophages, fibrin and protein-rich oedematous fluid are present in the alveolar spaces. In more advanced cases, macrophages and fibrin were observed among the neutrophils [
5]. Despite extensive research on the clinical, pathological, hematological and biochemical parameters of Pm-infected goats, very little is known about the molecular mechanisms of the Pm infection of goats.
High-throughput sequencing (HTS) is widely used as a screening method for differentially expressed genes (DEGs), mainly in transcriptomics, genomics, epigenomics and immunomics. Research on the application of HTS in Pm-infected goats is limited. In contrast, the disease has been widely studied in mice, pigs, poultry and other animals. Wu et al. established a mouse model for
Pasteurella pneumonia and detected 4236 DEGs by transcriptome sequencing analysis [
7]. Newborn pig tracheal epithelial (NPTr) cells were used as an in vitro model and the results found that Pm infection disrupted the barrier functions of the NPTr cells. RNA sequencing showed 30 DEGs between the Pm-infected pig tracheal epithelial cells and the control group [
8]. The transcriptomes data from chicken lungs infected with two different lethality isolates Pm were used for the cell-type enrichment analysis, the results showed that immune cells were enriched in both infection groups [
9]. A number of studies evaluated the effects of Pm on transcripts and host immune reaction in Pm-infected animals, but none have used HTS to analyse the miRNA and mRNA expression and function mechanism in goat.
The objective of this study, therefore, was to characterize the histopathology, miRNA and mRNA expression profiles in goat lungs from uninfected (the control group) and Pm-infected groups (the Pm group) at 1, 2, 5 and 7 days. In addition, we also evaluated the potential relationship between the miRNAs and mRNA expression and explored their possible functions in the Pm-caused inflammation and host immune response.
4. Discussion
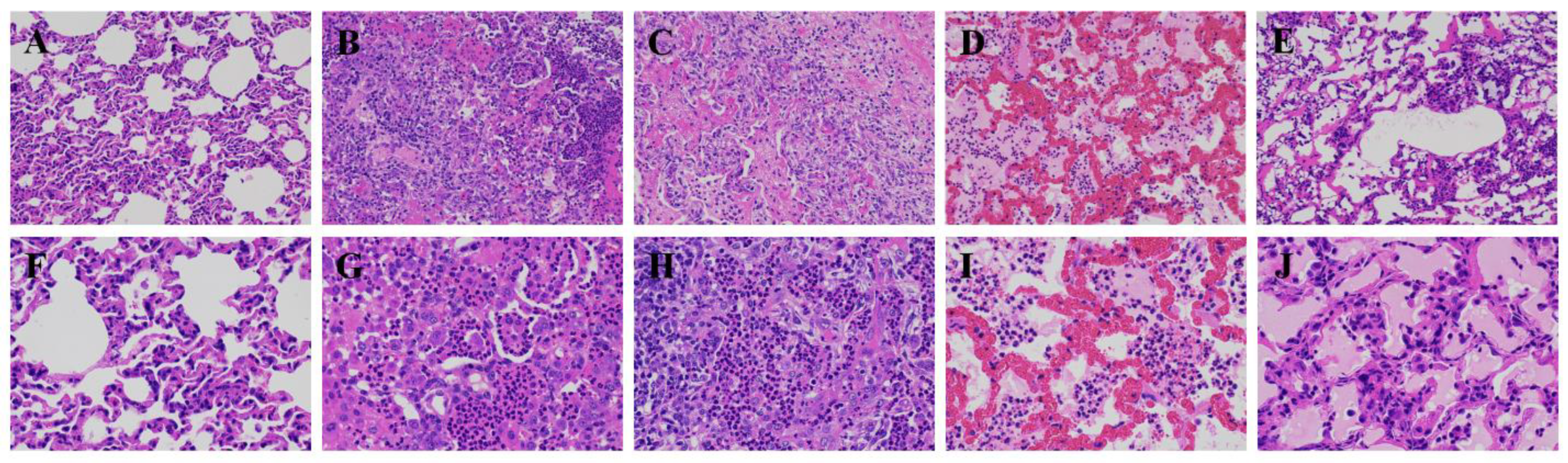
Respiratory disease is a severe ailment in goat breeding in tropical and subtropical areas. The disease, which causes enormous economic losses in the goat breeding industry, is caused by Pm. This study aimed to explore the pathogenic mechanisms of serotype D Pm–infected Hainan black goats. A previous study reported that Pm–challenged goats showed changes at the transcriptome level in the goat lung [
10]. Therefore, this study explored the effects of different infection periods on the miRNA and mRNA levels of goats. In addition, the changes of the clinical symptoms, body temperature and lung histopathology were monitored. Three goats died due to acute and fulminating conditions, while Pm triggered an extreme inflammatory response in the other goats. The lung lesions and HE staining results confirmed the inflammatory response (
Figure 2). This typical symptom was also observed in another study [
5]. The body temperature returned to normal 6 days after the Pm challenge (
Table 1). One possible reason is that the lung tissue lesions were more severe and acute in the first 5 days after the Pm infection. Correspondingly, the morphology of the lung tissue lesions gradually decreased in the samples collected 5 to 7 days after Pm infection (
Figure 2). Additionally, fewer DEGs were noticed in the 5 to 7 period after Pm infection than after 1 and 2 days (
Figure 4B).
The HE stained figures of the lung lesion showed several macrophages, monocytes and a marked neutrophil response at the site of the Pm infection lesion. Marker genes for these three cell types were also among the differentially expressed genes after Pm infection. CD163 is a macrophage-specific protein and the upregulated expression of this receptor is one of the significant changes in the macrophage switch to alternatively activated phenotypes in inflammation [
18]. Compared with the CK group, a high CD163 expression was observed after Pm infection after 1, 2, 5 and 7 days in this study. This was a characteristic of the lung tissue responding to pathogen-induced inflammation. The pulmonary macrophages are responsible primarily for recognizing pathogens and protecting the host by initiating inflammation and clearing organisms from the bronchioles and alveoli. It is practical to observe the pulmonary macrophages that phagocytose or kill bacteria, as a part of pathogenesis [
19]. A marked neutrophil response was observed in Pm-infected adult cattle [
20]. Consistent with this study, a large number of neutrophil infiltration was also observed in the lung lesion of Pm-infected goats in the current study. Therefore, the neutrophils play a role in response to pathogen infection in goats. However, inflammatory cell responses have been reported to be regulated by cytokines [
21]. The differentially expressed cytokines such as IL6, IL10, IL11, IL15, TNFSF10, TNFRSF6B and TNFAIP6 were detected in the lung of goats after Pm infection in this study. The expression of some of these was further validated by qPCR (
Figure 7).
Enrichment analysis of the DEGs in the different groups indicated that many immune GO terms and KEGG pathways were enriched. The GO enrichment results showed that several cellular components and leukocytes migration terms were enriched. The results indicated that Pm infection affected the cellular component. ‘Leukocytes migration’ GO terms may have facilitated leukocytes migrating to the lesions in the lung to eliminate the inflammatory trigger and contribute to tissue repair [
22]. This process is initiated mainly by pathogen-associated molecular patterns released by the invading pathogens and damage-associated molecular patterns derived from damaged and/or dead cells or in response to tissue and/or cellular injury [
23]. In addition, cytokine production and activity were also enriched. The cytokines may be released by effector T cells and macrophages to trigger leukocytes’ recruitment. Once in the interstitial tissue, leukocytes can exhibit multiple forms and numerous cellular and molecular regulatory mechanisms [
24,
25]. Leukocyte migration through activated venular walls is a fundamental immune response. The primary step in leukocyte migration is the establishment of adhesive interactions between leukocytes and endothelial cells of postcapillary venular walls close to inflamed tissues [
26]. This critical association is mediated by an array of cell-surface adhesion molecules. In our study, the DEGs significantly enriched cell adhesion molecules (CAMs) as a KEGG pathway after the Pm infection. This may indicated that leukocytes are leaving vessels and entering the inflammary tissue to destroy pathogens. In addition, the activation of endothelial cells is a decisive step in this process and can be induced by histamines, platelet-activating factors and cytokines [
27]. The enriched GO term ‘cytokines production’ strongly suggests that cytokines contributed to inducing endothelial cell activation in this study. Dysregulation of inflammatory cytokines is likely a common factor in developing Pm-caused pneumonia.
In the enriched KEGG pathways, multiple immune signaling pathways were significantly enriched. The cytokine–cytokine receptor interaction pathway, as one of the most enriched KEGG pathways (
Figure 5), was also influenced by avian pathogenic
Escherichia coli (APEC) infection in the chicken trachea [
28] and was involved in the rehabilitation process after respiratory syncytial virus infection [
29]. In addition, the DEGs were enriched in the activated complement and coagulation cascades KEGG pathway, suggesting that Pm infection can activate the complement and coagulation cascades. Rapid activation of the complement and coagulation cascades protects against invading pathogens and limits further bleeding. When these cascades are over-amplified by severe injury, the imbalanced response rapidly leads to the destruction rather than the repair of the injured tissue. This exaggerated and disordered response can result in multi-organ dysfunction syndrome (MODS), which is frequently fatal [
30]. These mechanisms are the likely reasons that caused the goat lung lesion and acute death 12 h after Pm infection. As an enriched KEGG pathway, the phagosome destroys and digests pathogens. Tight junctions form and regulate the paracellular barrier between epithelial and endothelial cell sheets. These sheets were also affected by the Pm infection since the lung epithelial barrier damage facilitated the Pm invasion. Praveena et al. reported that Pm infection caused ultrastructural changes in alveolar epithelia of the lungs [
31]. Moreover, disruption of the lung epithelial barrier structure by Pm was also reflected by HE staining in our study.
Differentially expressed miRNAs increased gradually from 1, 2, 5 and 7 days after infection (
Figure 4A). MiRNAs are mainly transcriptional and translational level regulators and the miRNAs change process is slow. Due to the innate immune response in the primary period, there was greater regulation of the inflammation reaction by other factors, such as various transcription factors and signaling pathways, and they were less dependent on miRNAs regulation. Compared with the CK group, five miRNAs were commonly differentially expressed in the Pm-infected animals after 1, 2, 5 and 7 days. Chi-miR-335-3p was the only significantly down-regulated miRNAs in the Pm-infected group. In the lung adenocarcinoma cells, miR-335-3p was targeted and down-regulated by the Coatomer Protein Complex Subunit beta 2 (COPB2) Gene [
32]. MiR-335-5p was down-regulated in the Parenchymal Lung Fibroblasts of smokers because cigarette smoking caused lung inflammation and tissue damage [
33]. In addition, miR-335-3p has lower expression in immune diseases such as T-cell acute lymphoblastic leukemia [
34] and childhood acute lymphoblastic leukemia [
35]. Therefore, miR-335-3p may regulate multiple target genes and other regulatory factors, such as lncRNA [
36], which are commonly involved in the host inflammation response reaction.
MiR-155-5p is a multifunctional miRNA enriched in the cells of the immune system and involved in the immune response. Chi-miR-155-5p was up-regulated in the Pm-infected groups in this study. Aziza’s analysis indicated that miR-155 could regulate chemokine production and pro-inflammatory chemokine receptor expression, contributing to Rheumatoid Arthritis pathogenesis and promoting inflammatory cell recruitment and retention in the RA synovium [
37]. Therefore, we speculated that the higher expression of chi-miR-155-5p was due to the Pm infection. Pm infection induced the release of cytokines, which promoted many inflammatory cells such as monocytes and macrophages. MiR-155-5p is highly expressed in activated B- and T-cells and monocytes/macrophages [
38]. This may be the reason for the up-regulation of miR-155-5p in the lung of the Pm-infected goats.
Chi-miR-21-3p and Chi-miR-21-5p are derived from opposite arms of the same pre-miRNA. Although both were up-regulated in the Pm infected groups, the expression of chi-miR-21-5p was more than 35,000 times that of chi-miR-21-3p. This study’s result agrees with the increased expression of miR-21-5p observed after
Mycobacterium tuberculosis infection of RAW264.7 and THP-1 cells [
39]. MiR-21-5p was also showed elevated expression in a murine macrophage cell line by activation of NF-κB -mediated inflammation with LPS stimulation [
40]. Therefore, we speculated that the high expression of chi-miR-21 regulated the Pm survival and inflammatory responses through multiple target genes and involved immune-related signaling pathways in the proliferated immune cells of the lung.
MiR-130b-3p has been reported to be associated with the immune system in the lung by its target genes [
41]. MiR-130b-3p was up-regulated after the
Mycoplasma gallisepticum-infected chicken by activating the PTEN/PI3K/AKT/NF-κB pathway to defend against pathogen invasion [
42]. Consistent with this study, miR-130b-3p was also up-regulated after Pm infection, maybe through the same pathway. However, this conjecture needs further validation.
5. Conclusions
In conclusion, we characterized the histopathlogy, miRNA and mRNA expression dynamics in lung of goats during infected with Pm for 1,2, 5 and 7 days and the clinical symptoms of animals. The infected lung tissue showed characteristics of suppurative pneumonia with inflammatory cells infiltration and lung structure destruction. Meanwhile, compared with the CK group, there were 3080, 3508, 2716 and 2675 differentially expressed genes, respectively, in the Pm_d1, Pm_d2, Pm_d5 and Pm_d7 groups, and 42, 69, 91 and 108 miRNAs were significantly expressed. The differentially expressed miRNAs and genes commonly involved and regulated the host immune reaction to the Pm infection. Finally, the potential mechanism of the host lesions in the lung caused by Pm was discussed in the context of the hosts’ genes and miRNAs. Although there were DEGs mapped to the marker genes of the immune cells, further details are still required through single-cell sequencing in the future.