Trichomonosis in Austrian Songbirds—Geographic Distribution, Pathological Lesions and Genetic Characterization over Nine Years
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection and Laboratory Investigations
2.2. DNA Extraction for Sequence Analysis
2.3. Trichomonad Partial Genome Sequencing
2.4. Bird Monitoring Counts
3. Results
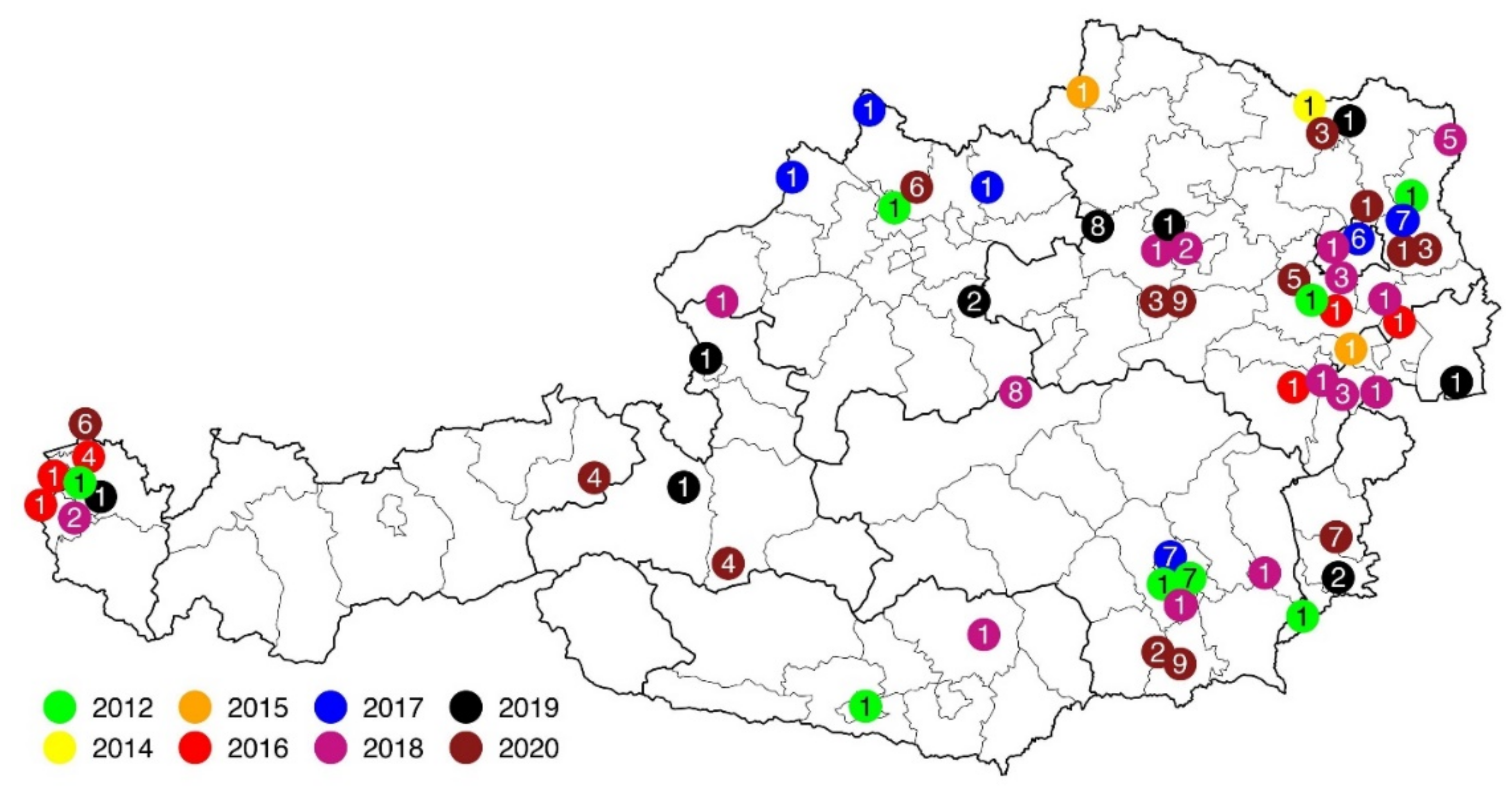
3.1. Dead Bird Reports
3.2. Macroscopic Findings
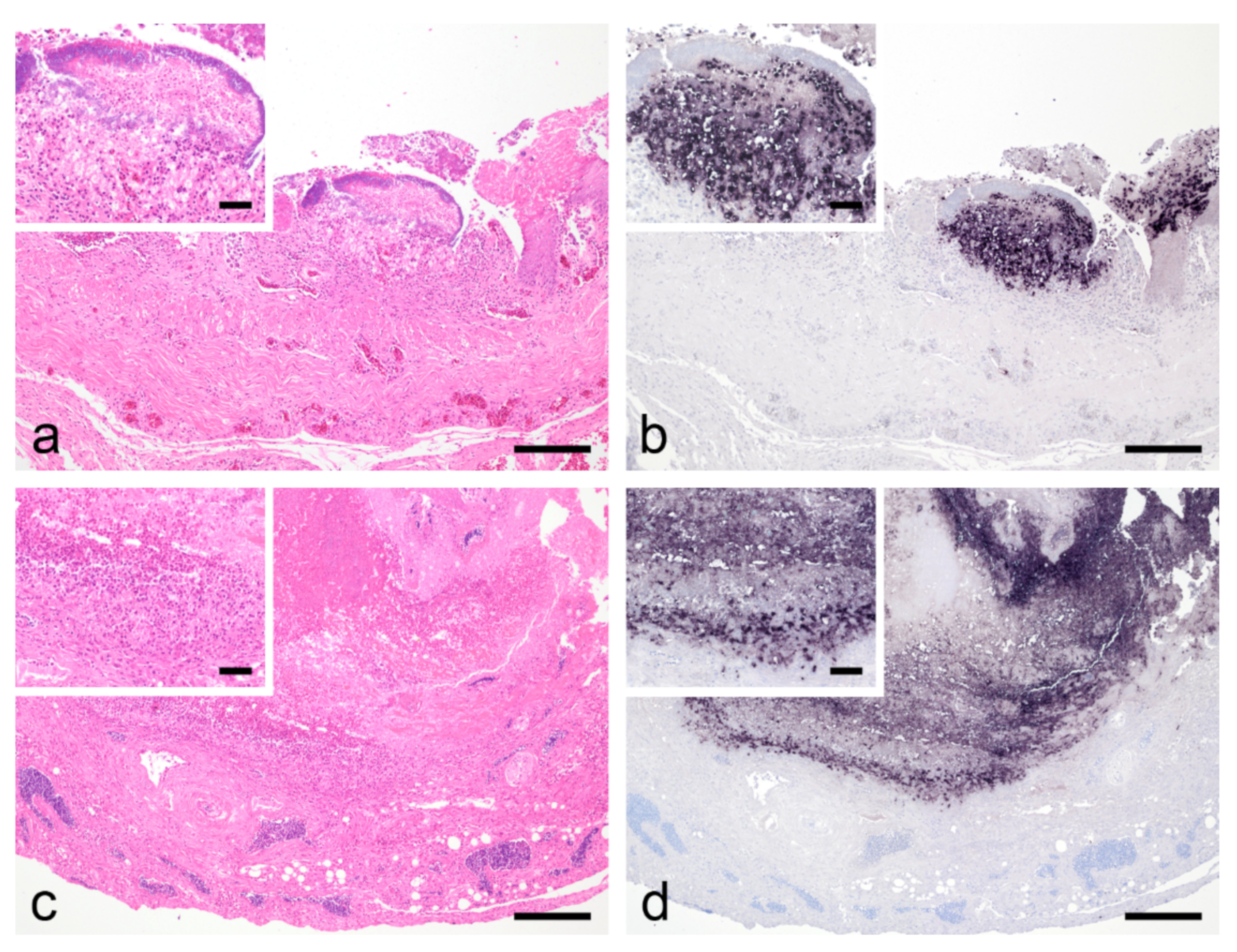
3.3. Histological Findings, PCR, In Situ Hybridization and Bacteriological Examination
3.4. Sequence Analysis
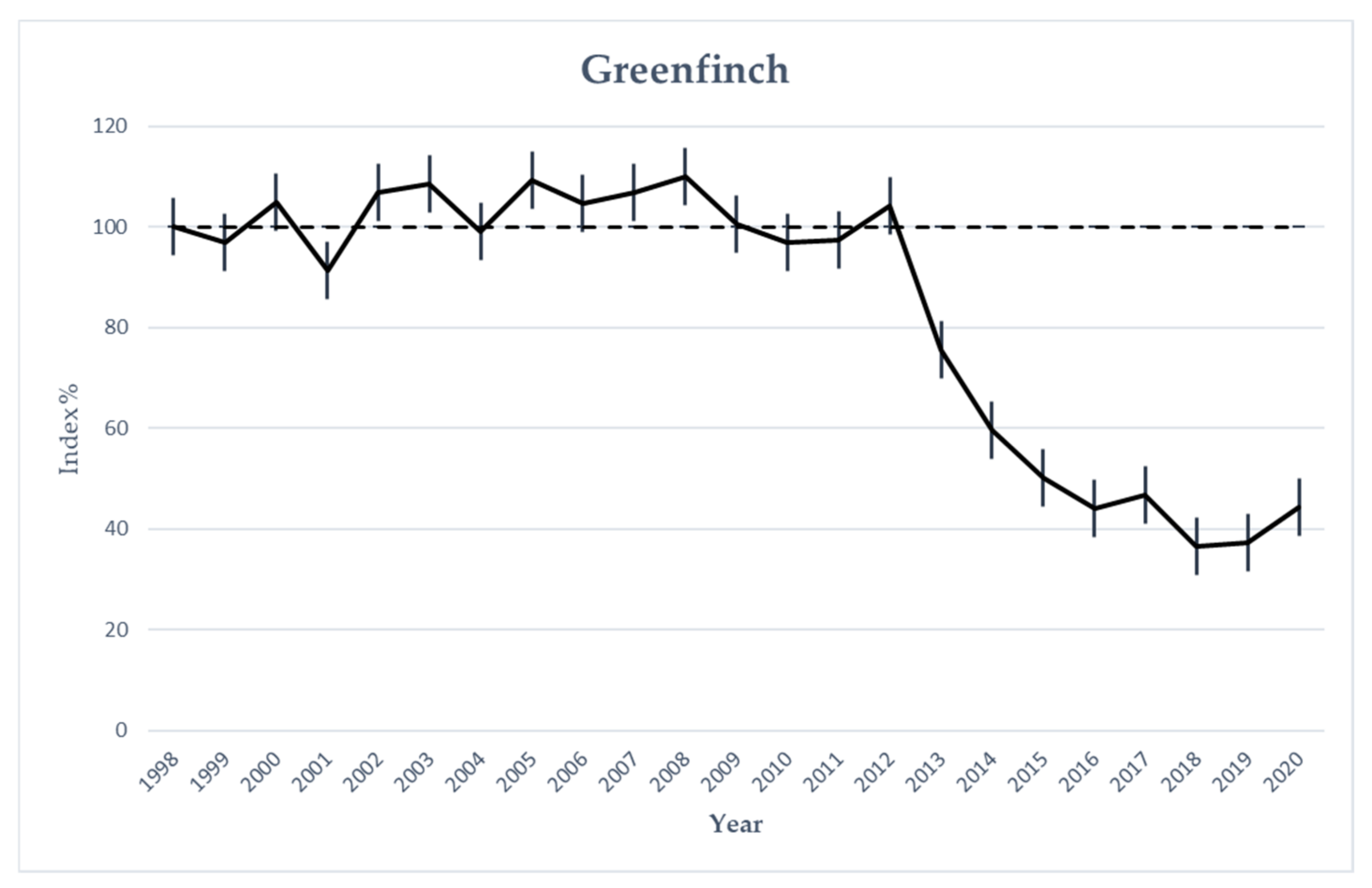
3.5. Greenfinch Monitoring Data
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ausarbeitung des Österreichischen Berichts Gemäß Artikel 12 der Vogelschutzrichtlinie, 2009/147/EG Berichtszeitraum 2008 bis 2012. Available online: https://www.noe.gv.at/noe/Naturschutz/Endbericht_VS_Art12_2014_hp.pdf (accessed on 24 March 2022).
- Massey, J.G. Diseases and medical management of wild passeriformes. Semin. Avian Exot. Pet Med. 2003, 12, 29–36. [Google Scholar] [CrossRef]
- Lehikoinen, A.; Lehikoinen, E.; Valkama, J.; Väisänen, R.A.; Isomursu, M. Impacts of trichomonosis epidemics on Greenfinch Chloris chloris and Chaffinch Fringilla coelebs populations in Finland. Ibis 2013, 155, 357–366. [Google Scholar]
- Lawson, B.; Robinson, R.A.; Colvile, K.M.; Peck, K.M.; Chantrey, J.; Pennycott, T.W.; Simpson, V.R.; Toms, M.P.; Cunningham, A.A. The emergence and spread of finch trichomonosis in the British Isles. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 2852–2863. [Google Scholar] [CrossRef]
- Peters, M.; Kilwinski, J.; Reckling, D.; Henning, K. Gehäufte Todesfälle von wild lebenden Grünfinken an Futterstellen infolge Trichomonas gallinae-Infektionen—Ein aktuelles Problem in Norddeutschland. Kleintierprax 2009, 54, 433–438. [Google Scholar]
- Robinson, R.A.; Lawson, B.; Toms, M.P.; Peck, K.M.; Kirkwood, J.K.; Chantrey, J.; Clatworthy, I.R.; Evans, A.D.; Hughes, L.A.; Hutchinson, O.C.; et al. Emerging Infectious Disease Leads to Rapid Population Declines of Common British Birds. PLoS ONE 2010, 5, e12215. [Google Scholar] [CrossRef]
- Quillfeldt, P.; Schumm, Y.R.; Marek, C.; Mader, V.; Fischer, D.; Marx, M. Prevalence and genotyping of Trichomonas infections in wild birds in central Germany. PLoS ONE 2018, 13, e0200798. [Google Scholar] [CrossRef]
- Cousquer, G. Ingluvitis and oesophagitis in wild finches. Vet. Rec. 2005, 157, 455. [Google Scholar] [CrossRef]
- Holmes, P.; Duff, P. Ingluvitis and oesophagitis in wild finches. Vet. Rec. 2005, 157, 455. [Google Scholar] [CrossRef]
- Pennycott, T.; Lawson, B.; Cunningham, A.; Simpson, V.; Chantrey, J. Necrotic ingluvitis in wild finches. Vet. Rec. 2005, 157, 360. [Google Scholar] [CrossRef]
- Lawson, B.; Cunningham, A.; Chantrey, J.; Hughes, L.; Kirkwood, J.; Pennycott, T.; Simpson, V. Epidemic finch mortality. Vet. Rec. 2006, 159, 367. [Google Scholar] [CrossRef]
- Simpson, V.; Molenaar, F. Increase in trichomonosis in finches. Vet. Rec. 2006, 159, 606. [Google Scholar] [CrossRef]
- Forzán, M.J.; Vanderstichel, R.; Melekhovets, Y.F.; McBurney, S. Trichomoniasis in finches from the Canadian Maritime provinces—An emerging disease. Can. Vet. J. Rev. Vet. Can. 2010, 51, 391–396. [Google Scholar]
- Neimanis, A.S.; Handeland, K.; Isomursu, M.; Ågren, E.; Mattsson, R.; Hamnes, I.S.; Bergsjø, B.; Hirvelä-Koski, V. First Report of Epizootic Trichomoniasis in Wild Finches (Family Fringillidae) in Southern Fennoscandia. Avian Dis. 2010, 54, 136–141. [Google Scholar] [CrossRef]
- Rijks, J.M.; Laumen, A.A.G.; Slaterus, R.; Stahl, J.; Gröne, A.; Kik, M.L. Trichomonosis in Greenfinches (Chloris chloris) in the Netherlands 2009–2017: A Concealed Threat. Front. Vet. Sci. 2019, 6, 425. [Google Scholar] [CrossRef]
- Chavatte, J.-M.; Giraud, P.; Esperet, D.; Place, G.; Cavalier, F.; Landau, I. An outbreak of trichomonosis in European green-finches Chloris chloris and European goldfinches Carduelis carduelis wintering in Northern France. Parasite 2019, 26, 21. [Google Scholar]
- Zadravec, M.; Marhold, C.; Slavec, B.; Rojs, O.Z.; Răcnik, J. Trichomonosis in finches in Slovenia. Vet. Rec. 2012, 171, 253–254. [Google Scholar] [CrossRef]
- Gourlay, P.; Decors, A.; Jouet, D.; Treilles, D.; Lemberger, K.; Faure, E.; Moinet, M.; Chi, J.; Tyler, K.; Cunningham, A.; et al. Finch trichomonosis spreads to France. Eur. Sect. Wildl. Dis. Assoc. Bull. 2011, 2, 9–10. [Google Scholar]
- Anderson, N.L.; Johnson, C.K.; Fender, S.; Heckly, S.; Metzler, M.; Nave, P.; Yim, J. Clinical Signs and Histopathologic Findings Associated with a Newly Recognized Protozoal Disease (Trichomonas gallinae) in Free-Ranging House Finches (Carpodacus mexicanus). J. Zoo Wildl. Med. 2010, 41, 249–254. [Google Scholar] [CrossRef]
- Mostegl, M.M.; Richter, B.; Nedorost, N.; Lang, C.; Maderner, A.; Dinhopl, N.; Weissenböck, H. First evidence of previously undescribed trichomonad species in the intestine of pigs? Vet. Parasitol. 2012, 185, 86–90. [Google Scholar]
- Mostegl, M.M.; Wetscher, A.; Richter, B.; Nedorost, N.; Dinhopl, N.; Weissenböck, H. Detection of Tritrichomonas foetus and Pentatrichomonas hominis in intestinal tissue specimens of cats by chromogenic in situ hybridization. Vet. Parasitol. 2012, 183, 209–214. [Google Scholar] [CrossRef]
- Felleisen, R.S. Comparative sequence analysis of 5.8S rRNA genes and internal transcribed spacer (ITS) regions of tricho-monadid protozoa. Parasitology 1997, 115 Pt 2, 111–119. [Google Scholar]
- Lawson, B.; Cunningham, A.A.; Chantrey, J.; Hughes, L.A.; John, S.K.; Bunbury, N.; Bell, D.J.; Tyler, K.M. A clonal strain of Trichomonas gallinae is the aetiologic agent of an emerging avian epidemic disease. Infect. Genet. Evol. 2011, 11, 1638–1645. [Google Scholar] [CrossRef]
- Hall, T. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Teufelbauer, N.; Seaman, B.S.; Dvorak, M. Population changes of common Austrian breeding birds in the period 1998-2016—Results of the breeding bird monitoring scheme. Egretta 2017, 55, 43–76. [Google Scholar]
- Van Strien, A.; Pannekoek, J.; Hagemeijer, W.; Verstrael, T. A loglinear Poisson regression method to analyse bird monitoring data. Bird Census News 2004, 13, 33–39. [Google Scholar]
- Grzimek, B.; Meise, W.; Niethammer, G.; Steinbacher, J. Die Finkenfamilie. In Grzimeks Tierleben Vögel 3, 1st ed.; Grzimek, B., Ed.; Deutscher Taschenbuch Verlag GmbH & Co.KG: München, Germany, 1993; p. 396. [Google Scholar]
- Gordo, F.P.; Herrera, S.; Castro, A.; Durán, B.G.; Díaz, R.M. Parasites from farmed ostriches (Struthio camelus) and rheas (Rhea americana) in Europe. Vet. Parasitol. 2002, 107, 137–160. [Google Scholar] [CrossRef]
- Mirzaei, M.; Ghashghaei, O.; Khedri, J. First report of an outbreak trichomoniasis in turkey in Sistan, Iran. J. Parasit. Dis. 2014, 40, 61–64. [Google Scholar] [CrossRef][Green Version]
- Pennycott, T.W. Carriage of trichomonads, Hexamita species and Blastocystis species by adult pheasants. Vet. Rec. 1998, 143, 142–143. [Google Scholar] [CrossRef]
- Forrester, D.J.; Foster, G.W. Trichomonosis. In Parasitic Diseases of Wild Birds; Atkinson, C.T., Thomas, N.J., Hunter, D.B., Thomas, N.J., Eds.; Wiley-Blackwell: Ames, IA, USA, 2008; pp. 120–153. [Google Scholar]
- Chalmers, A.; Alberta, G. Trichomoniasis in finches. Can. Vet. J. Rev. Vet. Can. 1992, 33, 616–617. [Google Scholar]
- Ecco, R.; Preis, I.S.; Vilela, D.A.; Luppi, M.M.; Malta, M.C.; Beckstead, R.B.; Stimmelmayer, R.; Gerhold, R. Molecular confirmation of Trichomonas gallinae and other parabasalids from Brazil using the 5.8S and ITS-1 rRNA regions. Vet. Parasitol. 2012, 190, 36–42. [Google Scholar] [CrossRef]
- Baker, J.R. Trichomoniasis, a major cause of vomiting in budgerigars. Vet. Rec. 1986, 118, 447–449. [Google Scholar] [CrossRef]
- McKeon, T.; Dunsmore, J.; Raidal, S. Trichomonas gallinae in budgerigars and columbid birds in Perth, Western Australia. Aust. Vet. J. 1997, 75, 652–655. [Google Scholar] [CrossRef]
- Deplazes, P.; Joachim, A.; Mathis, A.; Strube, C.; Taubert, A.; von Samson-Himmelstjerna, G.; Zahner, H. Parasitologie für die Tiermedizin, 4th ed.; Georg Thieme Verlag: Stuttgart, Germany; New York, NY, USA, 2021; pp. 57–58. [Google Scholar]
- Haugen, A.O. Trichomoniasis in Alabama Mourning Doves. J. Wildl. Manag. 1952, 16, 164. [Google Scholar] [CrossRef]
- Gerhold, R.W.; Tate, C.M.; Gibbs, S.E.; Mead, D.G.; Allison, A.B.; Fischer, J.R. Necropsy Findings and Arbovirus Surveillance in Mourning Doves from the Southeastern United States. J. Wildl. Dis. 2007, 43, 129–135. [Google Scholar] [CrossRef][Green Version]
- Rogers, K.H.; Girard, Y.A.; Koenig, W.D.; Johnson, C.K. Ecologic Drivers and Population Impacts of Avian Trichomonosis Mortality Events in Band-Tailed Pigeons (Patagioenas Fasciata) in California, USA. J. Wildl. Dis. 2016, 52, 484–494. [Google Scholar] [CrossRef][Green Version]
- Höfle, U.; Gortázar, C.; Ortiz, J.A.; Knispel, B.; Kaleta, E.F. Outbreak of trichomoniasis in a woodpigeon (Columba palumbus) wintering roost. Eur. J. Wildl. Res. 2004, 50, 73–77. [Google Scholar] [CrossRef]
- Reisen, W.K.; Chiles, R.E.; Green, E.N.; Fang, Y.; Mahmood, F.; Martinez, V.M.; Laver, T. Effects of immunosuppression on encephalitis virus infection in the house finch, Carpodacus mexicanus. J. Med Èntomol. 2003, 40, 206–214. [Google Scholar] [CrossRef]
- Ganas, P.; Jaskulska, B.; Lawson, B.; Zadravec, M.; Hess, M.; Bilic, I. Multi-locus sequence typing confirms the clonality of Trichomonas gallinae isolates circulating in European finches. Parasitology 2014, 141, 652–661. [Google Scholar]
- Lawson, B.; Robinson, R.A.; Neimanis, A.; Handeland, K.; Isomursu, M.; Ågren, E.O.; Hamnes, I.S.; Tyler, K.M.; Chantrey, J.; Hughes, L.A.; et al. Evidence of Spread of the Emerging Infectious Disease, Finch Trichomonosis, by Migrating birds. EcoHealth 2011, 8, 143–153. [Google Scholar] [CrossRef]
- Normann, D. Chaffinch Fringilla coelebs. In The Migration Atlas: Movements of the Birds of Britain and Ireland; Wenham, C.V., Toms, M.P., Marchant, J.H., Clark, J.A., Siriwardena, G.M., Baille, S.R., Eds.; T. & A.D. Poyser: London, UK, 2002; pp. 637–640. [Google Scholar]
- Svensson, L.; Grant, P.J.; Mullarney, K.; Zetterström, D. Sperlingsvögel. In Der Kosmos Vogelführer: Alle Arten Europas, Nordafrikas und Vorderasiens, 2nd ed.; Svensson, L., Grant, P.J., Mullarney, K., Zetterström, D., Eds.; Franckh-Kosmos Ver-lags-GmbH & Co: Stuttgart, Germany, 2018; p. 350. [Google Scholar]
- Bakken, V.; Runde, O.; Tjørve, E. [Norsk ringmerkingsatlas] The Norwegian Bird Ringing Atlas. Stavanger Museum: Stavanger, Norway, 2006; Volume 2, p. 446. [Google Scholar]
- Kocan, R.M.; Herman, C.M. Trichomoniasis. In Infectious and Parasitic Diseases of Wild Birds, 1st ed.; Davis, J.W., Anderson, R.C., Karstad, L., Trainer, D.O., Eds.; Iowa State University Press: Ames, IA, USA, 1971; pp. 282–290. [Google Scholar]
- Dorrestein, G.M.; Kummerfeld, N. Singvögel. In Krankheiten der Heimtiere, 8th ed.; Fehr, M., Sassenburg, L., Zwart, P., Eds.; Schlütersche Verlagsgesellschaft mbH & Co. KG: Hannover, Germany, 2014; pp. 425–492. [Google Scholar]
- Hudson, C.B.; Tudor, D.C. Salmonella typhimurium infection in feral birds. Cornell Vet. 1957, 47, 394–395. [Google Scholar]
- Wilson, J.; Macdonald, J. Salmonella Infection in Wild Birds. Br. Vet. J. 1967, 123, 212–219. [Google Scholar] [CrossRef]
- Schaal, E.; Ernst, H. Enzootic occurrence of salmonella infections in free-living, domestic bird species. Berl. Munch. Tierarztl. Wochenschr. 1967, 80, 13–16. [Google Scholar]
- Locke, L.N.; Shillinger, R.B.; Jareed, T. Salmonellosis in Passerine Birds in Maryland and West Virginia. J. Wildl. Dis. 1973, 9, 144–145. [Google Scholar] [CrossRef]
- Hurvell, B.; Jevring, J. Infections with Salmonella typhimurium in passerine birds in Sweden (author’s transl). Nord. Vet. Med. 1974, 26, 392–399. [Google Scholar]
- Kapperud, G.; Stenwig, H.; Lassen, J. Epidemiology of Salmonella typhimurium O: 4-12 Infection in Norway: Evidence of Transmission from an Avian Wildlife Reservoir. Am. J. Epidemiol. 1998, 147, 774–782. [Google Scholar] [CrossRef]
- Daoust, P.Y.; Busby, D.G.; Ferns, L.; Goltz, J.; McBurney, S.; Poppe, C.; Whitney, H. Salmonellosis in songbirds in the Canadian Atlantic provinces during winter-summer 1997–1998. Can. Vet. J. Rev. Vet. Can. 2000, 41, 54–59. [Google Scholar]
- Alley, M.R.; Connolly, J.; Fenwick, S.; Mackereth, G.; Leyland, M.; Rogers, L.; Haycock, M.; Nicol, C.; Reed, C. An epidemic of salmonellosis caused by Salmonella Typhimurium DT160 in wild birds and humans in New Zealand. N. Z. Vet. J. 2002, 50, 170–176. [Google Scholar] [CrossRef]
- Refsum, T.; Vikøren, T.; Handeland, K.; Kapperud, G.; Holstad, G. Epidemiologic and Pathologic Aspects of Salmonella Typhimurium Infection in Passerine Birds in Norway. J. Wildl. Dis. 2003, 39, 64–72. [Google Scholar] [CrossRef]
- Hall, A.J.; Saito, E.K. Avian Wildlife Mortality Events Due to Salmonellosis in the United States, 1985–2004. J. Wildl. Dis. 2008, 44, 585–593. [Google Scholar] [CrossRef]
- Une, Y.; Sanbe, A.; Suzuki, S.; Niwa, T.; Kawakami, K.; Kurosawa, R.; Izumiya, H.; Watanabe, H.; Kato, Y. Salmonella enterica serotype Typhimurium infection causing mortality in eurasian tree sparrows (Passer montanus) in Hokkaido. Jpn. J. Infect. Dis. 2008, 61, 166–167. [Google Scholar]
- Lawson, B.; Howard, T.; Kirkwood, J.K.; Macgregor, S.K.; Perkins, M.; Robinson, R.A.; Ward, L.R.; Cunningham, A.A. Epi-demiology of salmonellosis in garden birds in England and Wales, 1993 to 2003. Ecohealth 2010, 7, 294–306. [Google Scholar]
- Pennycott, T.W.; Mather, H.A.; Bennett, G.; Foster, G. Salmonellosis in garden birds in Scotland, 1995 to 2008: Geographic region, Salmonella enterica phage type and bird species. Vet. Rec. 2010, 166, 419–421. [Google Scholar] [CrossRef]
- Hernandez, S.M.; Keel, K.; Sanchez, S.; Trees, E.; Gerner-Smidt, P.; Adams, J.K.; Cheng, Y.; Ray, A.; Martin, G.; Presotto, A.; et al. Epidemiology of a Salmonella enterica subsp. enterica Serovar Typhimurium Strain Associated with a Songbird Outbreak. Appl. Environ. Microbiol. 2012, 78, 7290–7298. [Google Scholar] [CrossRef]
- Krawiec, M.; Pietkiewicz, M.; Wieliczko, A. Salmonella spp. as a cause of mortality and clinical symptoms in free-living garden bird species in Poland. Pol. J. Vet. Sci. 2014, 17, 729–731. [Google Scholar] [CrossRef]
- Brunthaler, R.; Spergser, J.; Weissenböck, H. Multiple Epidemics in Austrian Fringillidae Caused by A Single Variant of Salmonella Typhimurium. J. Wildl. Dis. 2021, 57, 891–899. [Google Scholar] [CrossRef]
- Knispel, B.H.M. Morphologische und molekularbiologische Untersuchungen (PCR und REA der 5,8S rRNA-Region) an Trichomonas gallinae und Tetratrichomonas gallinarum verschiedener Vogelarten. Ph.D. Thesis, Justus-Liebig-Universität, Gießen, Germany, 2005. [Google Scholar]
- Anderson, N.L.; Grahn, R.A.; Van Hoosear, K.; BonDurant, R.H. Studies of trichomonad protozoa in free ranging songbirds: Prevalence of Trichomonas gallinae in house finches (Carpodacus mexicanus) and corvids and a novel trichomonad in mockingbirds (Mimus polyglottos). Vet. Parasitol. 2009, 161, 178–186. [Google Scholar] [CrossRef]
- Greiner, E.C.; Ritchie, B.W. Parasites. In Avian Medicine: Principles and Application; Ritchie, B.W., Harrison, G.J., Harrison, L.R., Eds.; Wingers Publishing: Lake Worth, FL, USA, 1994; pp. 1007–1029. [Google Scholar]
- Mehlhorn, H.; Al-Quraishy, S.; Aziza, A.; Hess, M. Fine structure of the bird parasites Trichomonas gallinae and Tetra-trichomonas gallinarum from cultures. Parasitol. Res. 2009, 105, 751–756. [Google Scholar]
- Mostegl, M.M.; Richter, B.; Nedorost, N.; Maderner, A.; Dinhopl, N.; Kulda, J.; Liebhart, D.; Hess, M.; Weissenböck, H. Design and validation of an oligonucleotide probe for the detection of protozoa from the order Trichomonadida using chromogenic in situ hybridization. Vet. Parasitol. 2010, 171, 1–6. [Google Scholar] [CrossRef][Green Version]
- Mcburney, S.; Kelly-Clark, W.K.; Forzán, M.J.; Lawson, B.; Tyler, K.M.; Greenwood, S.J. Molecular characterization of Trichomonas gallinae isolates recovered from the Canadian Maritime provinces’ wild avifauna reveals the presence of the genotype responsible for the European finch trichomonosis epidemic and additional strains. Parasitology 2015, 142, 1053–1062. [Google Scholar] [CrossRef]
- Boal, C.W.; Mannan, R.W. Cooper’s Hawks in Urban and Exurban Areas: A Reply. J. Wildl. Manag. 2000, 64, 601–604. [Google Scholar]
- Gerhold, R.W.; Maestas, L.P.; Harnage, P.M. Persistence of Two Trichomonas gallinae Isolates in Chlorinated and Distilled Water with or without Organic Material. Avian Dis. 2013, 57, 681–683. [Google Scholar] [CrossRef]
- Kaleta, E.F.; Tschimmel, S.; Kraus, S.; Rullof, R. Beobachtungen bei der spontanen (natürlichen) Infektion von Brieftauben mit Trichomonas sp. Proc. VII Tag. Vogelkrankheiten DVG Hann. 1990, 171–176. [Google Scholar]
- Einer der wärmsten Sommer der Messgeschichte. Available online: https://www.zamg.ac.at/cms/de/klima/news/einer-der-waermsten-sommer-der-messgeschichte (accessed on 22 February 2022).
- Stabler, R.M. Trichomonas gallinae: A review. Exp. Parasitol. 1954, 3, 368–402. [Google Scholar] [CrossRef]
- Cole, R.A. Trichomoniasis. In Field Manual of Wildlife Diseases: General Field Procedures and Diseases of Birds; Friend, M., Franson, J.C., Eds.; U.S. Dept. of the Interior U.S. Geological Survey: Washington, DC, USA, 1999; pp. 201–206. [Google Scholar]
- Amin, A.; Bilic, I.; Liebhart, D.; Hess, M. Trichomonads in birds—A review. Parasitology 2014, 141, 733–747. [Google Scholar]
- Sansano-Maestre, J.; Garijo-Toledo, M.M.; Gómez-Muñoz, M.T. Prevalence and genotyping of Trichomonas gallinae in pigeons and birds of prey. Avian Pathol. 2009, 38, 201–207. [Google Scholar]
- Marx, M.; Reiner, G.; Willems, H.; Rocha, G.; Hillerich, K.; Masello, J.F.; Mayr, S.L.; Moussa, S.; Dunn, J.C.; Thomas, R.C.; et al. High prevalence of Trichomonas gallinae in wild columbids across western and southern Europe. Parasites Vectors 2017, 10, 242. [Google Scholar] [CrossRef]
- Jiang, X.; Sun, J.; Wang, F.; Li, H.; Zhao, X. Prevalence of Trichomonas spp. in domestic pigeons in Shandong Province, China, and genotyping by restriction fragment length polymorphism. Vet. J. 2016, 211, 88–93. [Google Scholar] [CrossRef]
- Lennon, R.J.; Dunn, J.C.; Stockdale, J.E.; Goodman, S.J.; Morris, A.J.; Hamer, K.C. Trichomonad parasite infection in four species of Columbidae in the UK. Parasitology 2013, 140, 1368–1376. [Google Scholar] [CrossRef]
- Zadravec, M.; Slavec, B.; Krapež, U.; Gombač, M.; Švara, T.; Poljšak-Prijatelj, M.; Gruntar, I.; Račnik, J. Trichomonosis outbreak in a flock of canaries (Serinus canaria f. domestica) caused by a finch epidemic strain of Trichomonas gallinae. Vet. Parasitol. 2017, 239, 90–93. [Google Scholar] [CrossRef]
- Cooper, J.E.; Petty, S.J. Trichomoniasis in Free-Living Goshawks (Accipiter Gentilis Gentilis) from Great Britain. J. Wildl. Dis. 1988, 24, 80–87. [Google Scholar] [CrossRef]
- Keymer, I. Diseases of birds of prey. Vet. Rec. 1972, 90, 579–594. [Google Scholar] [CrossRef]
- Krone, O.; Altenkamp, R.; Kenntner, N. Prevalence of Trichomonas Gallinae in Northern Goshawks from the Berlin Area of Northeastern Germany. J. Wildl. Dis. 2005, 41, 304–309. [Google Scholar] [CrossRef]
- Real, J.; Mañosa, S.; Muñoz, E. Trichomoniasis in a Bonelli’s eagle population in Spain. J. Wildl. Dis. 2000, 36, 64–70. [Google Scholar]
- Samour, J.H.; Bailey, T.A.; Cooper, J.E. Trichomoniasis in birds of prey (order Falconiformes) in Bahrain. Vet. Rec. 1995, 136, 358–362. [Google Scholar] [CrossRef]
- Kunca, T.; Šmejkalová, P.; Cepicka, I.; Èepièka, I. Trichomonosis in Eurasian sparrowhawks in the Czech Republic. Folia Parasitol. 2015, 62, 035. [Google Scholar] [CrossRef][Green Version]
- Himmel, T.; Harl, J.; Matt, J.; Weissenböck, H. A citizen science-based survey of avian mortality focusing on haemosporidian infections in wild passerine birds. Malar. J. 2021, 20, 417. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brunthaler, R.; Teufelbauer, N.; Seaman, B.; Nedorost, N.; Bittermann, K.; Matt, J.; Weissenbacher-Lang, C.; Weissenböck, H. Trichomonosis in Austrian Songbirds—Geographic Distribution, Pathological Lesions and Genetic Characterization over Nine Years. Animals 2022, 12, 1306. https://doi.org/10.3390/ani12101306
Brunthaler R, Teufelbauer N, Seaman B, Nedorost N, Bittermann K, Matt J, Weissenbacher-Lang C, Weissenböck H. Trichomonosis in Austrian Songbirds—Geographic Distribution, Pathological Lesions and Genetic Characterization over Nine Years. Animals. 2022; 12(10):1306. https://doi.org/10.3390/ani12101306
Chicago/Turabian StyleBrunthaler, René, Norbert Teufelbauer, Benjamin Seaman, Nora Nedorost, Karin Bittermann, Julia Matt, Christiane Weissenbacher-Lang, and Herbert Weissenböck. 2022. "Trichomonosis in Austrian Songbirds—Geographic Distribution, Pathological Lesions and Genetic Characterization over Nine Years" Animals 12, no. 10: 1306. https://doi.org/10.3390/ani12101306
APA StyleBrunthaler, R., Teufelbauer, N., Seaman, B., Nedorost, N., Bittermann, K., Matt, J., Weissenbacher-Lang, C., & Weissenböck, H. (2022). Trichomonosis in Austrian Songbirds—Geographic Distribution, Pathological Lesions and Genetic Characterization over Nine Years. Animals, 12(10), 1306. https://doi.org/10.3390/ani12101306







