Modified Black Soldier Fly Larva Fat in Broiler Diet: Effects on Performance, Carcass Traits, Blood Parameters, Histomorphological Features and Gut Microbiota
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Birds and Diets
- (i)
- MBSF1 was supplemented with 8–9% glycerol monobutyrin to obtain an equimolar content of monobutyrin and monolaurin;
- (ii)
- MBSF2 was supplemented with 21–22% glycerol monobutyrin such that the amount of FA was equimolar to the amount of added monobutyrin in the resulting product.
2.2. Chemical Composition and Fatty Acid Profile of the Experimental Diets
2.3. Growth Performance
2.4. Blood Parameters
2.5. Slaughter Procedures and Muscle Sampling
2.6. Chemical Composition of Meat
2.7. Histomorphological Investigations
2.8. DNA Extraction and 16S Metataxonomic Approach
2.9. Bioinformatics and Statistical Analysis
3. Results
3.1. Growth Performance
3.2. Blood Parameters
3.3. Slaughter Performance and Meat Quality Traits
3.4. Gut Morphology
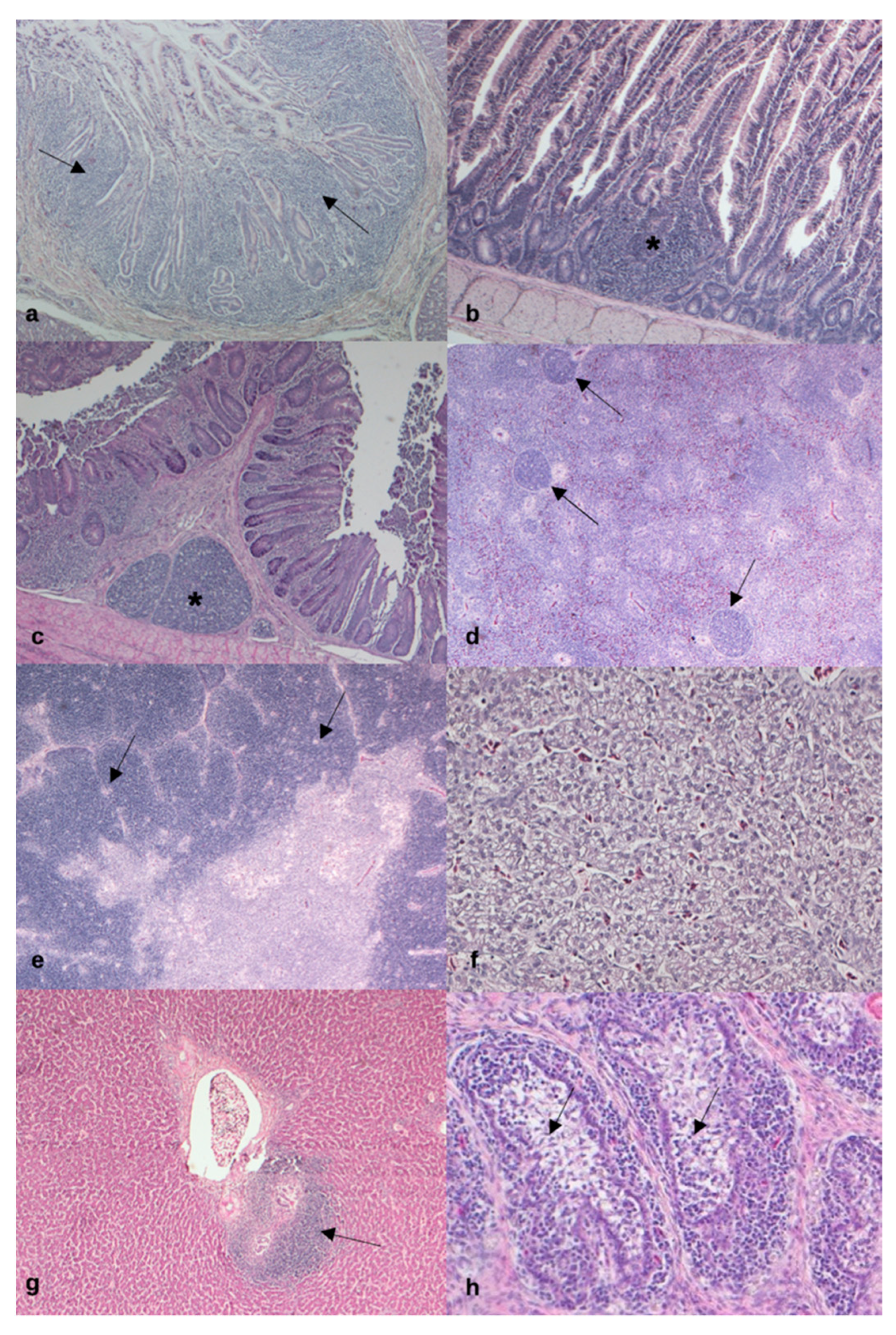
3.5. Histopathological Findings
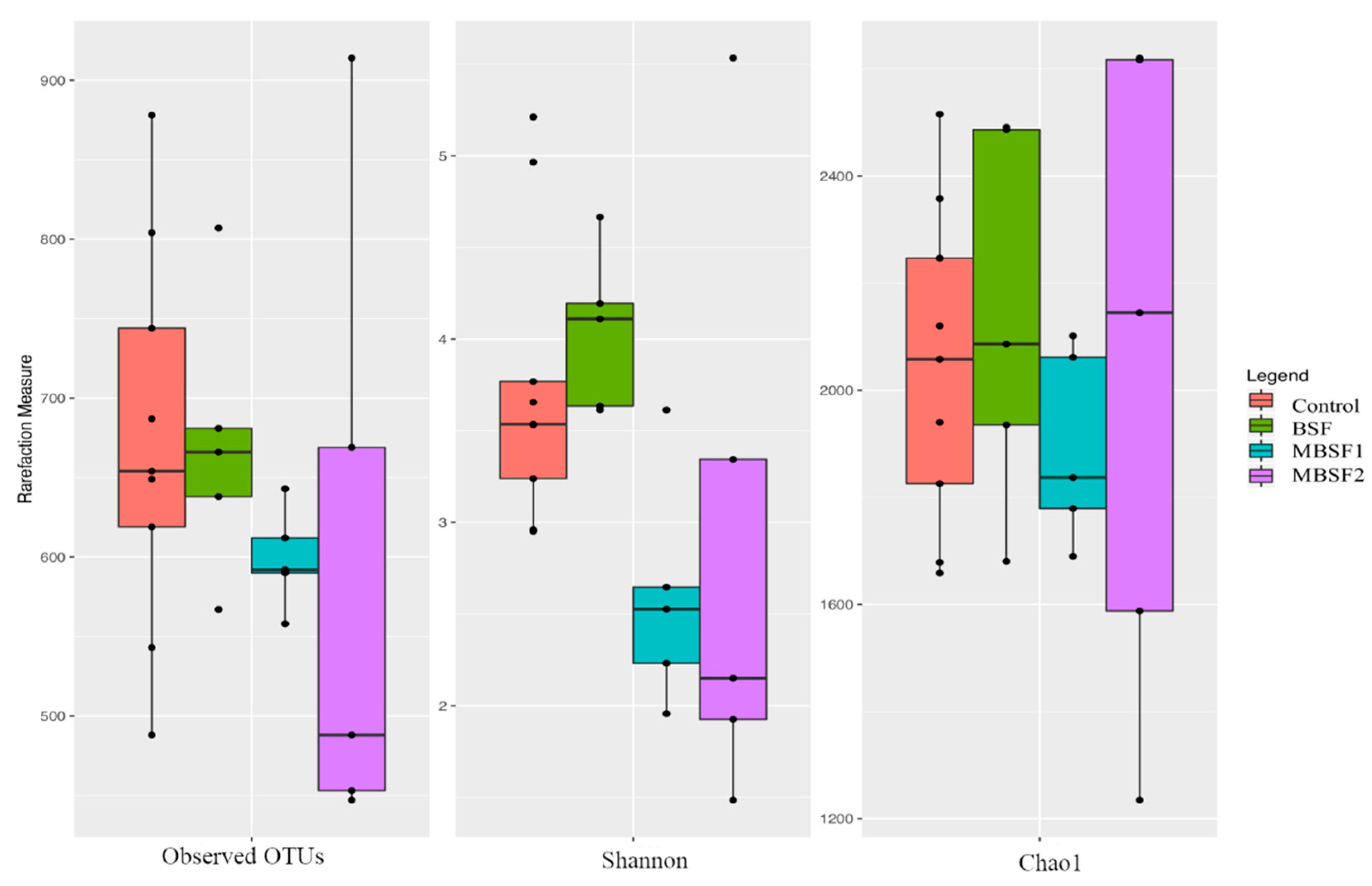
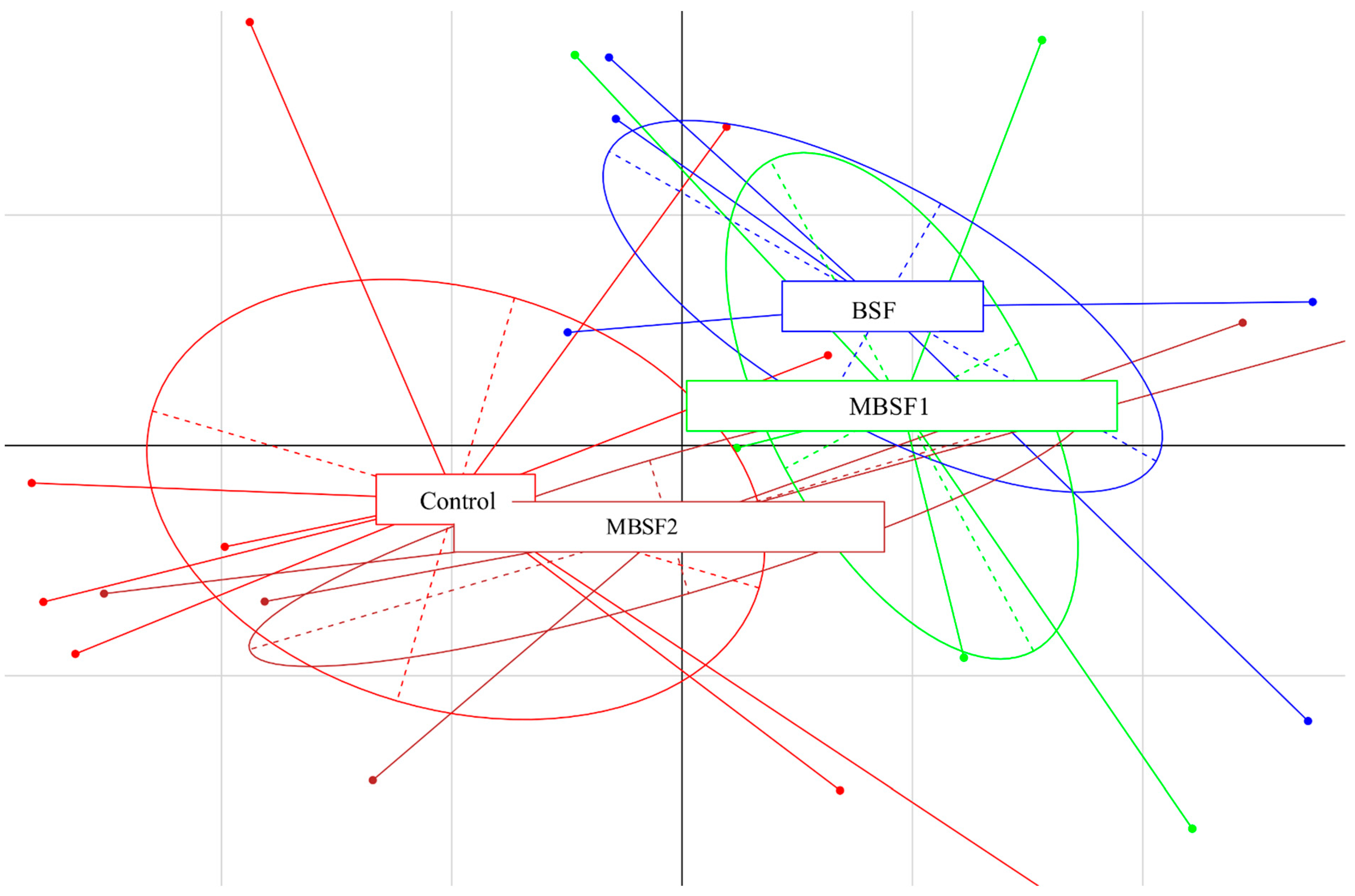
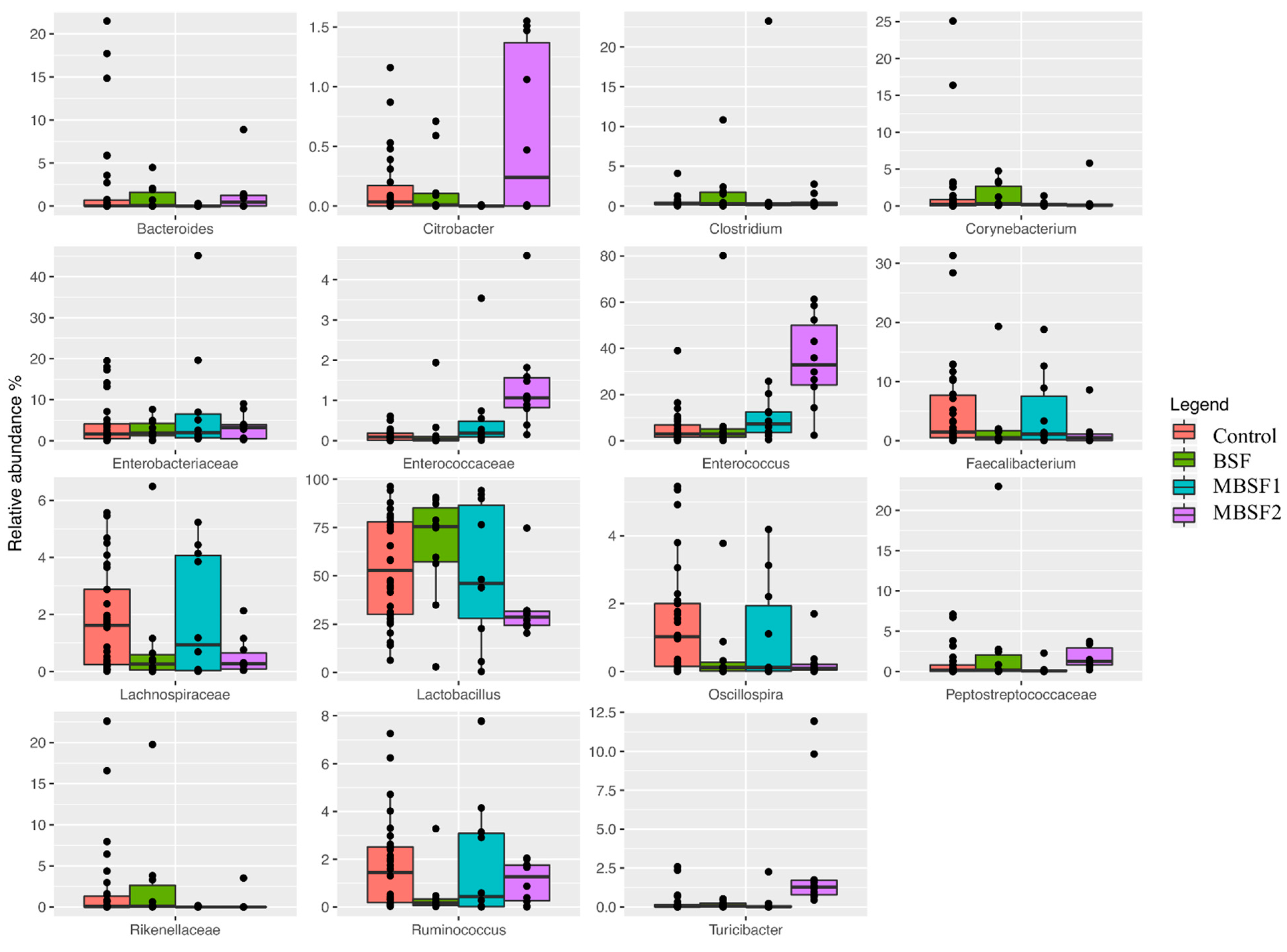
3.6. Microbiota Characterization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Muaz, K.; Riaz, M.; Akhtar, S.; Park, S.; Smail, I. Antibiotic residues in chicken meat: Global prevalence, threats, and decontamination strategies: A review. J. Food Prot. 2019, 81, 619–627. [Google Scholar] [CrossRef]
- Yadav, S.; Jha, R. Strategies to modulate the intestinal microbiota and their effects on nutrient utilization, performance, and health of poultry. J. Anim. Sci. Biotechnol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ji, H.; Zhang, B.; Tian, J.; Zhou, J.; Yu, H. Influence of black soldier fly (Hermetia illucens) larvae oil on growth performance, body composition, tissue fatty acid composition and lipid deposition in juvenile Jian carp (Cyprinus carpio var. Jian). Aquaculture 2016, 465, 43–52. [Google Scholar] [CrossRef]
- Schiavone, A.; Cullere, M.; De Marco, M.; Meneguz, M.; Biasato, I.; Bergagna, S.; Dezzutto, D.; Gai, F.; Dabbou, S.; Gasco, L.; et al. Partial or total replacement of soybean oil by black soldier larvae (Hermetia illucens L.) fat in broiler diets: Effect on growth performances, feed-choice, blood traits, carcass characteristics and meat quality. Ital. J. Anim. Sci. 2017, 16, 93–100. [Google Scholar] [CrossRef] [Green Version]
- Schiavone, A.; Dabbou, S.; De Marco, M.; Cullere, M.; Biasato, I.; Biasibetti, E.; Capucchio, M.; Bergagna, S.; Dezzutto, D.; Meneguz, M.; et al. Black soldier fly larva fat inclusion in finisher broiler chicken diet as an alternative fat source. Animal 2018, 12, 2032–2039. [Google Scholar] [CrossRef]
- Cullere, M.; Schiavone, A.; Dabbou, S.; Gasco, L.; Dalle Zotte, A. Meat quality and sensory traits of finisher broiler chickens fed with black soldier fly (Hermetia illucens L.) larvae fat as alternative fat source. Animals 2019, 9, 140. [Google Scholar] [CrossRef] [Green Version]
- Gasco, L.; Dabbou, S.; Trocino, A.; Xiccato, G.; Capucchio, M.T.; Biasato, I.; Dezzutto, D.; Meneguz, M.; Schiavone, A.; Gai, F. Effect of dietary supplementation with insect fats on growth performance, digestive efficiency and health of rabbits. J. Anim. Sci. Biotechnol. 2019, 10. [Google Scholar] [CrossRef]
- Gasco, L.; Dabbou, S.; Gai, F.; Brugiapaglia, A.; Schiavone, A.; Birolo, M.; Xiccato, G.; Trocino, A. Quality and consumer acceptance of meat from rabbits fed diets in which soybean oil is replaced with black soldier fly and yellow mealworm fats. Animals 2019, 9, 629. [Google Scholar] [CrossRef] [Green Version]
- Sypniewski, J.; Kierończyk, B.; Benzertiha, A.; Mikołajczak, Z.; Pruszyńska Oszmałek, E.; Kołodziejski, P.; Sassekc, M.; Rawskid, M.; Czekałae, W.; Józefiak, D. Replacement of soybean oil by Hermetia illucens fat in turkey nutrition: Effect on performance, digestibility, microbial community, immune and physiological status and final product quality. Br. Poult. Sci. 2020, 61, 294–302. [Google Scholar] [CrossRef]
- Kumar, V.; Femi, J.F.; Nicholas, R.; Md Sakhawat, H.; Shyam, N.L.; Ken, O.; Brian, C.S. Insect (black soldier fly, Hermetia illucens) meal supplementation prevents the soybean meal-induced intestinal enteritis in rainbow trout and health benefits of using insect oil. Fish Shellfish Immunol. 2021, 109, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Ewald, N.; Vidakovic, A.; Langeland, M.; Kiessling, A.; Sampels, S.; Lalander, C. Fatty acid composition of black soldier fly larvae (Hermetia illucens)—Possibilities and limitations for modification through diet. Waste Manag. 2020, 102, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Meneguz, M.; Schiavone, A.; Gai, F.; Dama, A.; Lussiana, C.; Renna, M.; Gasco, L. Effect of rearing substrate on growth performance, waste reduction efficiency and chemical composition of black soldier fly (Hermetia illucens) larva. J. Sci. Food Agric. 2018, 98, 5776–5784. [Google Scholar] [CrossRef]
- Spranghers, T.; Ottoboni, M.; Klootwijk, C.; Ovyn, A.; Deboosere, S.; De Meulenaer, B.; Michiels, J.; Eeckhout, M.; De Clercq, P.; De Smet, S. Nutritional composition of black soldier fly (Hermetia illucens) prepupae reared on different organic waste substrates. J. Sci. Food Agric. 2017, 97, 2594–2600. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, X.; Wang, H.; Yang, Q.Q.; Rehman, K.U.; Li, W.; Cai, M.M.; Li, Q.; Mazza, L.; Zhang, J.B.; et al. Dynamic changes of nutrient composition throughout the entire life cycle of black soldier fly. PLoS ONE 2017, 12, e0182601. [Google Scholar] [CrossRef] [Green Version]
- Lalander, C.; Diener, S.; Zurbrügg, C.; Vinnerås, B. Effects of feedstock on larval development and process efficiency in waste treatment with black soldier fly (Hermetia illucens). J. Clean. Prod. 2019, 208, 211–219. [Google Scholar] [CrossRef]
- Boyen, F.; Haesebrouck, F.; Vanparys, A.; Volf, J.; Mahu, M.; Van Immerseel, F.; Rychlik, I.; Dewulf, J.; Ducatelle, R.; Pasmans, F. Coated fatty acids alter virulence properties of Salmonella typhimurium and decrease intestinal colonization of pigs. Vet. Microbiol. 2008, 132, 319–327. [Google Scholar] [CrossRef]
- Baltic, B.M.; Janjic, J.; Brankovic Lazic, I.; Mrdovic, B.; Ciric, J.; Markovic, R.; Mitrovic, R. Relationships between broiler final weights and microbiota of certain segments of the intestine. IOP Conf. Ser. Earth Environ. Sci. 2019, 333, 012045. [Google Scholar] [CrossRef]
- Van Immerseel, F.; Russel, J.B.; Flythe, M.D.; Gantois, I.; Timbermont, L.; Pasmans, F.; Haesebrouck, F.; Ducatelle, R. The use of organic acids to combat Salmonella in poultry: A mechanistic explanation of the efficacy. Avian Pathol. 2006, 35, 182–188. [Google Scholar] [CrossRef] [Green Version]
- Van der Hoeven-Hangoor, E.; Van Der Vossen, J.M.B.M.; Schuren, F.H.J.; Verstegen, M.W.A.; De Oliveira, J.E.; Montijn, R.C.; Hendriks, W.H. Ileal microbiota composition of broilers fed various commercial diet compositions. Poult. Sci. 2013, 92, 2713–2723. [Google Scholar] [CrossRef]
- Hermans, D.; Martel, A.; Van Deun, K.; Verlinden, M.; Van Immerseel, F.; Garmyn, A.; Messens, W.; Heyndrickx, M.; Haesebrouck, F.; Pasmans, F. Intestinal mucus protects Campylobacter jejuni in the ceca of colonized broiler chickens against the bactericidal effects of medium-chain fatty acids. Poult. Sci. 2010, 89, 1144–1155. [Google Scholar] [CrossRef]
- Van Gerwe, T.; Bouma, A.; Klinkenberg, D.; Wagenaar, J.A.; Jacobs-Reitsma, W.F.; Stegeman, A. Medium chain fatty acid feed supplementation reduces the probability of Campylobacter jejuni colonization in broilers. Vet. Microbiol. 2010, 143, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Zeitz, J.; Fennhoff, O.J.; Kluge, H.; Stangl, G.I.; Eder, K. Effects of dietary fats rich in lauric and myristic acid on performance, intestinal morphology, gut microbes, and meat quality in broilers. Poult. Sci. 2015, 94, 2404–2413. [Google Scholar] [CrossRef]
- Antongiovanni, M.; Buccioni, A.; Minieri, S.; Galigani, I.; Rapaccini, S. Monobutyrine: A novel feed additive in the diet of broiler chickens. Ital. J. Anim. Sci. 2010, 9, e69. [Google Scholar] [CrossRef]
- Timbermont, L.; Lanckriet, A.; Dewulf, J.; Nollet, N.; Schwarzer, K.; Haesebrouck, F.; Ducatelle, R.; van Immerseel, F. Control of clostridium perfringens-induced necrotic enteritis in broilers by target-released butyric acid, fatty acids and essential oils. Avian Pathol. 2010, 39, 117–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeiger, K.; Popp, J.; Becker, A.; Hankel, J.; Visscher, C.; Klein, G.; Meemken, D. Lauric acid as feed additive—An approach to reducing Campylobacter spp. in broiler meat. PLoS ONE 2017, 12, e0175693. [Google Scholar] [CrossRef] [Green Version]
- Fortuoso, B.F.; Dos Reis, J.H.; Gebert, R.R.; Barreta, M.; Griss, L.G.; Casagrande, R.A.; de Cristo, T.G.; Santiani, F.; Campigotto, G.; Rampazzo, L.; et al. Glycerol monolaurate in the diet of broiler chickens replacing conventional antimicrobials: Impact on health, performance and meat quality. Microb Pathog. 2019, 129, 161–167. [Google Scholar] [CrossRef]
- Kabara, J.J.; Swieczkowski, D.M.; Conley, A.J.; Truant, J.P. Fatty acids and derivatives as antimicrobial agents. Antimicrob. Agents Chemother. 1972, 2, 23–28. [Google Scholar] [CrossRef] [Green Version]
- Martin, D.; Moran-Valero, M.I.; Vázquez, L.; Reglero, G.; Torres, C.F. Comparative in vitro intestinal digestion of 1,3-diglyceride and 1-monoglyceride rich oils and their mixtures. Food Res. Int. 2014, 64, 603–609. [Google Scholar] [CrossRef]
- Sek, L.; Porter, C.J.H.; Kaukonen, A.M.; Charman, W.N. Evaluation of the in vitro digestion profiles of long and medium chain glycerides and the phase behaviour of their lipolytic products. J. Pharm. Pharmacol. 2002, 54, 29–41. [Google Scholar] [CrossRef]
- Batovska, D.I.; Todorova, I.T.; Tsvetkova, I.V.; Najdenski, H.M. Antibacterial study of the medium chain fatty acids and their 1-monoglycerides: Individual effects and synergistic relationships. Pol. J. Microbiol. 2009, 58, 43–47. [Google Scholar]
- Amara, S.; Bourlieu, C.; Humbert, L.; Rainteau, D.; Carrière, F. Variations in gastrointestinal lipases, pH and bile acid levels with food intake, age and diseases: Possible impact on oral lipid-based drug delivery systems. Adv. Drug Deliv. Rev. 2019, 142, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Association of Official Analytical Chemists. Official Method of Analysis, 18th ed.; Association of Official Analytical Chemists (AOAC): Washington, DC, USA, 2005. [Google Scholar]
- Glass, R.L.; Christopherson, S.W. A method for the differential analysis of mixtures of esterified and free fatty acids. Chem. Phys. Lipids 1969, 3, 405–408. [Google Scholar] [CrossRef]
- Salamano, G.; Mellia, E.; Tarantola, M.; Gennero, M.S.; Doglione, L.; Schiavone, A. Acute phase proteins and heterophil-lymphocyte ratio in laying hens in different housing systems. Vet. Rec. 2010, 167, 749–751. [Google Scholar] [CrossRef] [PubMed]
- Commission Internationale de L’Éclairage (CIE). Recommendations on Uniform Colour Spaces-Colour Difference Equations, Psychometric Colour Terms; Supplement No 2 to CIE Publication No 15; Commission Internationale de L’Éclairage: Paris, France, 1976. [Google Scholar]
- Association of Official Analytical Chemists. Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists (AOAC): Arlington, VA, USA, 1990. [Google Scholar]
- Folch, J.; Lees, M.; Sloane Stanley, H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 479–509. [Google Scholar] [CrossRef]
- Laudadio, V.; Passantino, L.; Perillo, A.; Lopresti, G.; Passantino, A.; Khan, R.U.; Tufarelli, V. Productive performance and histological features of intestinal mucosa of broiler chickens fed different dietary protein levels. Poult. Sci. 2012, 91, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Qaisrani, S.N.; Moquet, P.C.; van Krimpen, M.M.; Kwakkel, R.P.; Verstegen, M.W.; Hendriks, W.H. Protein source and dietary structure influence growth performance, gut morphology, and hindgut fermentation characteristics in broilers. Poult. Sci. 2014, 93, 3053–3064. [Google Scholar] [CrossRef]
- Dabbou, S.; Ferrocino, I.; Kovitvadhi, A.; Dabbou, S.; Bergagna, S.; Dezzuto, D.; Schiavone, A.; Cocolin, L.; Gai, F.; Santoro, V.; et al. Bilberry pomace in rabbit nutrition: Effects on growth performance, apparent digestibility, caecal traits, bacterial community and antioxidant status. Animal 2019, 13, 53–63. [Google Scholar] [CrossRef] [Green Version]
- Biasato, I.; Ferrocino, I.; Biasibetti, E.; Grego, E.; Dabbou, S.; Sereno, A.; Gai, F.; Gasco, L.; Schiavone, A.; Cocolin, L.; et al. Modulation of intestinal microbiota, morphology and mucin composition by dietary insect meal inclusion in free-range chickens. BMC Vet. Res. 2018, 14, 383. [Google Scholar] [CrossRef] [Green Version]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dixon, A.L.; Liang, L.; Moffatt, M.F.; Chen, W.; Heath, S.; Wong, K.C.C.; Taylor, J.; Burnett, E.; Gut, I.; Farrall, M.; et al. A genome-wide association study of global gene expression. Nat. Genet. 2007, 39, 1202–1207. [Google Scholar] [CrossRef]
- Danieli, P.P.; Lussiana, C.; Gasco, L.; Amici, A.; Ronchi, B. The effects of diet formulation on the yield, proximate composition, and fatty acid profile of the black soldier fly (Hermetia illucens L.) prepupae intended for animal feed. Animals 2019, 9, 178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, Z.; Wang, W.; Lu, X.; Zhu, F.; Liu, W.; Wang, X.P.; Lei, C. Bioconversion performance and life table of black soldier fly (Hermetia illucens) on fermented maize straw. J. Clean. Prod. 2019, 230, 974–980. [Google Scholar] [CrossRef]
- Kawasaki, K.; Hashimoto, Y.; Hori, A.; Kawasaki, T.; Hirayasu, H.; Iwase, S.; Hashizume, A.; Ido, A.; Miura, C. Evaluation of black soldier fly (Hermetia illucens) larvae and pre-pupae raised on household organic waste, as potential ingredients for poultry feed. Animals 2019, 9, 98. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.B.; Kim, D.H.; Jeong, S.B.; Lee, J.W.; Kim, T.H.; Lee, H.G.; Lee, K.W. Black soldier fly larvae oil as an alternative fat source in broiler nutrition. Poult. Sci. 2020, 99, 3133–3143. [Google Scholar] [CrossRef] [PubMed]
- Kierónczyk, B.; Rawski, M.; Józefiak, A.; Mazurkiewicz, J.; Świątkiewicz, S.; Siwek, M.; Bednarczyk, M.; Szumacher-Strabel, M.; Cieślak, A.; Benzertiha, A. Effects of replacing soybean oil with selected insect fats on broilers. Anim. Feed Sci. Technol. 2018, 240, 170–183. [Google Scholar] [CrossRef]
- Benzertiha, A.; Kierónczyk, B.; Rawski, M.; Kołodziejski, P.; Bryszak, M.; Józefiak, D. Insect oil as an alternative to palm oil and poultry fat in broiler chicken nutrition. Animals 2019, 9, 116. [Google Scholar] [CrossRef] [Green Version]
- Lumej, J.T. Clinical Biochemistry of Domestic Animals, 6th ed.; Kaneko, J.J., Harwey, J.W., Bruss, M.L., Eds.; Elsevier Academic Press: Oxford, UK, 2008; pp. 839–872. [Google Scholar]
- Ricke, S.C. Perspectives on the use of organic acids and short chain fatty acids as antimicrobials. Poult. Sci. 2003, 82, 632–639. [Google Scholar] [CrossRef]
- Cox, N.A.; McHan, F.; Bailey, J.S. Effect of butyric or lactic acid on the in vivo colonization of Salmonella typhimurium. J. Appl. Poultry. Res. 1994, 3, 315–318. [Google Scholar] [CrossRef]
- Sampugna, J.; Quinn, J.G.; Pitas, R.E.; Carpenter, D.L.; Jensen, R.G. Digestion of butyrate glycerides by pancreatic lipase. Lipids 1967, 2, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Fernández Rubio, C.; Ordóñez, C.; Abad-González, J.; Garcia-Gallego, A.; Pilar Honrubia, M.; Jose Mallo, J.; Balaña-Fouce, R. Butyric acid-based feed additives help protect broiler chickens from Salmonella enteritidis infection. Poult. Sci. 2009, 88, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.H.; Jiang, Y.; Zhu, Q.F.; Gao, F.; Dai, S.F.; Chen, J.; Zhou, G.H. Sodium butyrate maintains growth performance by regulating the immune response in broiler chickens. Br. Poult. Sci. 2011, 52, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Leeson, S.; Namkung, H.; Antongiovanni, M.; Lee, E.H. Effect of butyric acid on the performance and carcass yield of broiler chickens. Poult. Sci. 2005, 84, 1418–1422. [Google Scholar] [CrossRef] [PubMed]
- Namkung, H.; Yu, H.; Gong, J.; Leeson, S. Antimicrobial activity of butyrate glycerides toward Salmonella typhimurium and Clostridium perfringens. Poult. Sci. 2011, 90, 2217–2222. [Google Scholar] [CrossRef]
- Wei, S.; Morrison, M.; Yu, Z. Bacterial census of poultry intestinal microbiome. Poult. Sci. 2013, 92, 671–683. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Si, W.; Forster, R.J.; Huang, R.; Yu, H.; Yin, Y.; Yang, C.; Han, Y. 16S rRNA gene-based analysis of mucosa-associated bacterial community and phylogeny in the chicken gastrointestinal tracts: From crops to ceca. FEMS Microbiol. Ecol. 2007, 59, 147–157. [Google Scholar] [CrossRef] [Green Version]
- Danzeisen, J.L.; Kim, H.B.; Isaacson, R.E.; Tu, Z.J.; Johnson, T.J. Modulations of the chicken cecal microbiome and metagenome in response to anticoccidial and growth promoter treatment. PLoS ONE 2011, 6, e27949. [Google Scholar] [CrossRef]
- Stanley, D.; Hughes, R.J.; Geier, M.S.; Moore, R.J. Bacteria within the gastrointestinal tract microbiota correlated with improved growth and feed conversion: Challenges presented for the identification of performance enhancing probiotic bacteria. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [Green Version]
- Oakley, B.B.; Kogut, M.H. Spatial and temporal changes in the broiler chicken cecal and fecal microbiomes and correlations of bacterial taxa with cytokine gene expression. Front. Vet. Sci. 2016, 3. [Google Scholar] [CrossRef] [Green Version]
- Glendinning, L.; Stewart, R.D.; Pallen, M.J.; Watson, K.A.; Watson, M. Assembly of hundreds of novel bacterial genomes from the chicken caecum. Genome Biol. 2020, 21. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Brisbin, J.; Yu, H.; Wang, Q.; Yin, F.; Zhang, Y.; Sabour, P.; Sharif, S.; Gong, J. Selected lactic acid-producing bacterial isolates with the capacity to reduce Salmonella translocation and virulence gene expression in chickens. PLoS ONE 2014, 9, e93022. [Google Scholar]
- Chen, F.; Gao, S.S.; Zhu, L.Q.; Qin, S.Y.; Qiu, H.L. Effects of dietary Lactobacillus rhamnosus CF supplementation on growth, meat quality, and microenvironment in specific pathogen-free chickens. Poult. Sci. 2018, 97, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Biddle, A.; Stewart, L.; Blanchard, J.; Leschine, S. Untan-Gling the genetic basis of fibrolytic specialization by Lachnospiraceae and Ruminococcaceae in diverse gut communities. Diversity 2013, 5, 627–640. [Google Scholar] [CrossRef]
- Hang, I.; Rinttila, T.; Zentek, J.; Kettunen, A.; Alaja, S.; Apajalahti, J.; Harmoinen, J.; de Vos, W.M.; Spillmann, T. Effect ofhigh contents of dietary animal-derived protein or carbohydrates on canine faecal microbiota. BMC Vet. Res. 2012, 8. [Google Scholar] [CrossRef] [Green Version]
- Warn, P.; Thommes, P.; Sattar, A.; Corbett, D.; Flattery, A.; Zhang, Z.; Black, T.; Hernandez, L.D.; Therien, A.G. Disease progression and resolution in rodent models of clostridium difficile infection and impact of antitoxin antibodies and vancomycin. Antimicrob. Agents Chemother. 2016, 60, 6471–6482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Cesare, A.; do Valle, I.F.; Sala, C.; Sirri, F.; Astolfi, A.; Castellani, G.; Manfreda, G. Effect of a low protein diet on chicken ceca microbiome and productive performances. Poult. Sci. 2019, 98, 3963–3976. [Google Scholar] [CrossRef]
- Borrelli, L.; Coretti, L.; Dipineto, L.; Bovera, F.; Menna, F.; Chiariotti, L.; Nizza, A.; Lembo, F.; Fioretti, A. Insect based diet, a promising nutritional source, modulates gut microbiota composition and SCFAs production in laying hens. Sci. Rep. 2017, 7, 16269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burkovski, A. Pathogenesis of Corynebacterium diphtheriae and Corynebacterium ulcerans. In Human Emerging and Re-Emerging Infections: Viral and Parasitic Infections; Wiley-Blackwell: Hoboken, NJ, USA, 2015; pp. 699–709. [Google Scholar] [CrossRef]
- Enurah, L.; Olubade, T.; Nwamo, A.; Sadiku, R. An outbreak of Corynebacterium diphtheriae infection in broiler chickens in Lagos, Nigeria. Glob. J. Med. Res. Vet. Sci. Vet. Med. 2016, 16, e14. [Google Scholar]
| Ingredients (g/kg) | Starter Period (1–11 d) | Grower-Finisher Period (11–33 d) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| C | BSF | MBSF1 | MBSF2 | C | BSF | MBSF1 | MBSF2 | ||
| Corn meal | 508.8 | 508.8 | 508.8 | 508.8 | 536.1 | 536.1 | 536.1 | 536.1 | |
| Soybean meal 48 | 345.3 | 345.3 | 345.3 | 345.3 | 332.0 | 332.0 | 332.0 | 332.0 | |
| Corn gluten feed | 54.0 | 54.0 | 54.0 | 54.0 | 35.0 | 35.0 | 35.0 | 35.0 | |
| Soybean oil | 46.3 | 43.4 | 38.3 | 42.8 | 59.3 | 57.7 | 55.3 | 57.5 | |
| BSF fat | - | 2.9 | - | - | - | 1.6 | - | - | |
| MBSF1 | - | - | 8.0 | - | - | - | 4.0 | - | |
| MBSF2 | - | - | - | 3.5 | - | - | - | 1.8 | |
| Calcium phosphate | 5.3 | 5.3 | 5.3 | 5.3 | 3.5 | 3.5 | 3.5 | 3.5 | |
| Calcium carbonate | 18.5 | 18.5 | 18.5 | 18.5 | 17.2 | 17.2 | 17.2 | 17.2 | |
| Sodium chloride | 2.3 | 2.3 | 2.3 | 2.3 | 2.3 | 2.3 | 2.3 | 2.3 | |
| Sodium bicarbonate | 1.3 | 1.3 | 1.3 | 1.3 | 1.3 | 1.3 | 1.3 | 1.3 | |
| L-lysine | 5.1 | 5.1 | 5.1 | 5.1 | 3.8 | 3.8 | 3.8 | 3.8 | |
| DL-methionine | 2.0 | 2.0 | 2.0 | 2.0 | 1.8 | 1.8 | 1.8 | 1.8 | |
| Threonine | 1.9 | 1.9 | 1.9 | 1.9 | 1.5 | 1.5 | 1.5 | 1.5 | |
| 3-phytase * | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |
| Vitamin and mineral premix a | 8.0 | 8.0 | 8.0 | 8.0 | 5.0 | 5.0 | 5.0 | 5.0 | |
| Choline chloride | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | |
| Total | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | |
| AME (kcal/kg) (calculated) | 3000 | 3000 | 3000 | 3000 | 3100 | 3100 | 3100 | 3100 | |
| Chemical Composition (Analyzed) | |||||||||
| Dry matter (DM), % | 89.6 | 89.7 | 90.0 | 89.7 | 90.1 | 90.0 | 89.8 | 90.2 | |
| Ash, % DM | 6.32 | 6.19 | 6.39 | 6.62 | 6.12 | 6.25 | 6.45 | 6.47 | |
| Crude protein, % DM | 23.0 | 23.0 | 23.0 | 23.0 | 21.5 | 21.5 | 21.5 | 21.5 | |
| Ether extract, %DM | 6.41 | 6.86 | 6.28 | 6.07 | 7.96 | 8.18 | 7.72 | 7.28 | |
| Lysine, % | 1.44 | 1.44 | 1.44 | 1.44 | 1.29 | 1.29 | 1.29 | 1.29 | |
| Methionine, % | 0.56 | 0.56 | 0.56 | 0.56 | 0.51 | 0.51 | 0.51 | 0.51 | |
| Threonine, % | 0.97 | 0.97 | 0.97 | 0.97 | 0.88 | 0.88 | 0.88 | 0.88 | |
| Calcium, % | 0.96 | 0.96 | 0.96 | 0.96 | 0.87 | 0.87 | 0.87 | 0.87 | |
| Phosphorus, % | 0.48 | 0.48 | 0.48 | 0.48 | 0.43 | 0.43 | 0.43 | 0.43 | |
| Fatty Acid | Dietary Fats | Experimental Diets | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Starter Diet | Grower-Finisher Diet | |||||||||||
| SO | BSF | MBSF1 | MBSF2 | C | BSF | MBSF1 | MBSF2 | C | BSF | MBSF1 | MBSF2 | |
| C12:0 | 0.00 | 46.86 | 45.46 | 47.35 | 0.00 | 1.49 | 1.28 | 0.58 | 0.00 | 0.65 | 0.62 | 0.30 |
| C14:0 | 0.08 | 9.87 | 9.80 | 10.06 | 0.10 | 0.40 | 0.33 | 0.22 | 0.00 | 0.22 | 0.19 | 0.13 |
| C16:0 | 10.92 | 14.38 | 15.17 | 14.56 | 12.30 | 12.65 | 12.00 | 12.17 | 11.39 | 11.35 | 11.64 | 12.31 |
| C16:1 | 0.09 | 2.78 | 2.92 | 2.77 | 0.16 | 0.17 | 0.00 | 0.00 | 0.00 | 0.00 | 0.15 | 0.16 |
| C18:0 | 3.43 | 1.79 | 1.98 | 1.80 | 3.28 | 3.20 | 3.30 | 3.30 | 3.43 | 3.38 | 3.34 | 3.31 |
| C18:1 n9 c | 25.23 | 7.73 | 8.26 | 7.57 | 25.78 | 24.92 | 25.22 | 25.58 | 25.90 | 25.57 | 25.43 | 25.05 |
| C18:2 n6 | 52.30 | 12.77 | 13.38 | 12.47 | 51.73 | 50.62 | 51.83 | 51.74 | 52.82 | 51.99 | 51.55 | 51.64 |
| C18:3 n3 | 6.22 | 0.98 | 1.03 | 0.95 | 4.90 | 4.83 | 4.95 | 4.97 | 5.35 | 5.29 | 5.27 | 5.28 |
| C20:0 | 0.31 | 0.07 | 0.00 | 0.00 | 0.35 | 0.33 | 0.36 | 0.36 | 0.38 | 0.37 | 0.34 | 0.33 |
| C20:1 | 0.26 | 0.77 | 0.74 | 0.81 | 0.30 | 0.30 | 0-36 | 0.28 | 0.31 | 0.32 | 0.30 | 0.24 |
| ΣSFA | 14.95 | 74.24 | 73.40 | 74.99 | 16.20 | 18.32 | 17.27 | 16.83 | 15.19 | 16.16 | 16.31 | 16.63 |
| ΣMUFA | 25.75 | 11.58 | 11.98 | 11.30 | 26.40 | 25.53 | 25.57 | 25.85 | 26.21 | 25.88 | 26.09 | 25.61 |
| ΣPUFA | 58.74 | 14.00 | 14.60 | 13.62 | 57.25 | 56.01 | 57.14 | 57.30 | 58.58 | 57.95 | 57.58 | 57.62 |
| Other FAs | 1.19 | 1.74 | 2.02 | 0.91 | 0.76 | 0.80 | 0.37 | 0.59 | 0.42 | 0.67 | 0.76 | 0.92 |
| Items | Experimental Diets | SEM | p-Value | |||
|---|---|---|---|---|---|---|
| C | BSF | MBSF1 | MBSF2 | |||
| LW at 1 d, g | 45.2 | 45.3 | 45.3 | 45.3 | 0.074 | 0.983 |
| LW at 11 d, g | 318.3 | 320.7 | 313.8 | 317.3 | 2.917 | 0.887 |
| LW at 33 d, g | 2002.5 | 1937.2 | 1920.5 | 2067.8 | 23.636 | 0.096 |
| ADG 1–11 d, g | 24.8 | 25.0 | 24.4 | 24.7 | 0.263 | 0.882 |
| ADG 11–33 d, g | 76.6 | 73.5 | 73.0 | 79.6 | 1.042 | 0.080 |
| ADG 1–33 d, g | 59.3 | 57.3 | 56.8 | 61.3 | 0.716 | 0.096 |
| ADFI 1–11 d, g | 27.4 | 28.6 | 27.0 | 28.0 | 0.343 | 0.390 |
| ADFI 11–33 d, g | 101.3 | 97.5 | 98.3 | 113.1 | 2.393 | 0.062 |
| ADFI 1–33 d, g | 77.5 | 74.7 | 75.2 | 86.0 | 1.775 | 0.074 |
| FCR 1–11 d | 1.11 | 1.13 | 1.10 | 1.06 | 0.010 | 0.172 |
| FCR 11–33 d | 1.26 | 1.31 | 1.29 | 1.34 | 0.015 | 0.325 |
| FCR 1–33 d | 1.25 | 1.28 | 1.27 | 1.32 | 0.011 | 0.245 |
| Items | Experimental Diets | SEM | p-Value | |||
|---|---|---|---|---|---|---|
| C | BSF | MBSF1 | MBSF2 | |||
| ALB (g/dL) | 1.35 | 1.38 | 1.39 | 1.45 | 0.020 | 0.441 |
| ALT (UI/L) | 2.80 | 2.57 | 2.67 | 2.90 | 0.063 | 0.264 |
| AST (UI/L) | 319.5 | 330.8 | 317.8 | 339.8 | 6.042 | 0.546 |
| ALP (UI/L) | 5492.6 | 5705.6 | 6519.0 | 5983.0 | 379.55 | 0.835 |
| GGT (UI/L) | 29.4 | 29.6 | 29.9 | 29.7 | 1.294 | 0.999 |
| Total cholesterol (mg/dL) | 113.5 | 116.6 | 115.1 | 119.7 | 2.096 | 0.765 |
| HDL-cholesterol (mg/dL) | 85.1 | 90.9 | 89.1 | 92.6 | 1.651 | 0.424 |
| LDL-cholesterol (mg/dL) | 29.1 | 26.7 | 26.0 | 27.8 | 1.112 | 0.786 |
| Triglycerides (mg/dL) | 41.8 | 40.6 | 40.7 | 37.0 | 1.435 | 0.669 |
| Uric acid (mg/dL) | 4.98 | 5.00 | 4.74 | 4.89 | 0.174 | 0.955 |
| Creatinine (mg/dL) | 0.09 | 0.10 | 0.08 | 0.10 | 0.005 | 0.692 |
| Total protein (g/dL) | 3.22 | 3.13 | 3.23 | 3.34 | 0.048 | 0.469 |
| Phosphorus (mg/dL) | 90.6 | 70.1 | 63.9 | 94.4 | 5.404 | 0.125 |
| Chlorine (mmol/L) | 118.6 | 117.9 | 116.6 | 125.2 | 1.529 | 0.171 |
| Potassium (mmol/L) | 6.81 | 7.04 | 6.64 | 7.66 | 0.156 | 0.088 |
| Magnesium (mEq/L) | 6.01 | 6.03 | 5.80 | 5.95 | 0.107 | 0.885 |
| Iron (µg/dL) | 99.7 | 88.3 | 96.4 | 90.8 | 2.618 | 0.401 |
| Sodium (mmol/L) | 154.2 | 165.1 | 163.6 | 174.9 | 3.428 | 0.194 |
| Calcium (mg/dL) | 48.7 | 48.7 | 53.2 | 49.6 | 0.749 | 0.121 |
| Items | Experimental Diets | SEM | p-Value | |||
|---|---|---|---|---|---|---|
| C | BSF | MBSF1 | MBSF2 | |||
| Slaughter weight (SW), g | 2032.2 | 2021.8 | 2017.6 | 2108.8 | 19.806 | 0.327 |
| Carcass weight (CCW), g | 1396.9 | 1382.5 | 1364.8 | 1442.4 | 15.336 | 0.328 |
| Slaughter yield, % SW | 68.8 | 68.3 | 67.6 | 68.4 | 0.249 | 0.439 |
| Breast, % CCW | 31.8 | 32.2 | 32.5 | 33.0 | 0.256 | 0.389 |
| Thigh, % CCW | 31.3 | 30.5 | 30.6 | 30.8 | 0.305 | 0.755 |
| Spleen, % SW | 0.11 | 0.11 | 0.11 | 0.11 | 0.004 | 0.985 |
| Liver, % SW | 2.04 | 2.02 | 2.02 | 2.06 | 0.020 | 0.886 |
| Bursa of Fabricius, % SW | 0.24 | 0.28 | 0.27 | 0.30 | 0.009 | 0.222 |
| Heart, % SW | 0.66 | 0.70 | 0.68 | 0.68 | 0.017 | 0.891 |
| Proventriculus (glandular stomach), % SW | 0.37 | 0.36 | 0.36 | 0.37 | 0.008 | 0.998 |
| Gizzard (muscular stomach), % SW | 1.38 | 1.38 | 1.54 | 1.32 | 0.047 | 0.400 |
| Intestine, % SW | 4.87 | 5.37 | 5.13 | 5.14 | 0.153 | 0.727 |
| Items | Experimental Diets | SEM | p-Value | |||
|---|---|---|---|---|---|---|
| C | BSF | MBSF1 | MBSF2 | |||
| Breast meat | ||||||
| pH | 5.99 | 5.99 | 5.97 | 6.04 | 0.013 | 0.288 |
| L | 52.9 | 52.6 | 52.2 | 53.2 | 0.366 | 0.814 |
| a* | 0.72 | 0.71 | 0.61 | 0.90 | 0.179 | 0.951 |
| b* | 12.5 | 13.0 | 11.9 | 12.3 | 0.275 | 0.552 |
| Chemical Composition (%) | ||||||
| Water | 24.3 | 24.6 | 24.6 | 24.1 | 0.095 | 0.231 |
| Ash | 1.24 | 1.21 | 1.25 | 1.19 | 0.017 | 0.576 |
| Crude protein | 22.7 | 22.5 | 22.4 | 22.5 | 0.138 | 0.940 |
| Total lipids | 1.45 a | 1.33 ab | 1.16 b | 1.44 a | 0.040 | 0.029 |
| Thigh meat | ||||||
| pH | 6.18 | 6.16 | 6.17 | 6.16 | 0.012 | 0.966 |
| Chemical Composition (%) | ||||||
| Water | 28.2 | 27.3 | 27.7 | 28.6 | 0.337 | 0.586 |
| Ash | 1.04 | 1.14 | 1.04 | 1.08 | 0.018 | 0.132 |
| Crude protein | 18.9 | 19.2 | 18.4 | 18.9 | 0.115 | 0.114 |
| Total lipids | 9.28 | 8.22 | 8.98 | 7.90 | 0.308 | 0.357 |
| Index | Fixed Effect | d.f. 3 | F | p-Value 4 |
|---|---|---|---|---|
| Vh (mm) | Diet 1 | 3 | 0.166 | 0.919 |
| Intestinal segment 2 | 2 | 155.045 | <0.001 | |
| Diet × Intestinal segment | 6 | 0.820 | 0.556 | |
| Cd (mm) | Diet | 3 | 0.888 | 0.448 |
| Intestinal segment | 2 | 0.133 | 0.875 | |
| Diet × Intestinal segment | 6 | 1.220 | 0.298 | |
| Vh/Cd (mm/mm) | Diet | 3 | 0.872 | 0.457 |
| Intestinal segment | 2 | 143.538 | <0.001 | |
| Diet × Intestinal segment | 6 | 0.547 | 0.772 |
| Index | Fixed Effect | Effect Levels | Least Square Mean 1 | SEM |
|---|---|---|---|---|
| Vh (mm) | Diet 2 | C | 2.28 | 0.08 |
| BSF | 2.28 | |||
| MBSF1 | 2.23 | |||
| MBSF2 | 2.30 | |||
| Intestinal segment 3 | DU | 3.24 a | 0.07 | |
| JE | 1.95 b | |||
| IL | 1.63 c | |||
| Cd (mm) | Diet 2 | C | 0.17 | 0.01 |
| BSF | 0.18 | |||
| MBSF1 | 0.18 | |||
| MBSF2 | 0.18 | |||
| Intestinal segment 3 | DU | 0.18 | 0.00 | |
| JE | 0.18 | |||
| IL | 0.18 | |||
| Vh/Cd (mm/mm) | Diet 2 | C | 13.13 | 0.46 |
| BSF | 12.33 | |||
| MBSF12 | 12.31 | |||
| MBSF | 12.99 | |||
| Intestinal segment 3 | DU | 18.12 a | 0.40 | |
| JE | 10.8 b | |||
| IL | 9.15 c |
| Items | Experimental Diets | SEM | p-Value | |||
|---|---|---|---|---|---|---|
| C | BSF | MBSF1 | MBSF2 | |||
| Spleen | 0.53 | 0.27 | 0.27 | 0.07 | 0.06 | 0.072 |
| Liver | 0.23 | 0.27 | 0.33 | 0.27 | 0.06 | 0.883 |
| Thymus | 0.06 | 0 | 0.06 | 0 | 0.03 | 0.591 |
| Bursa of Fabricius | 0.60 | 0.77 | 0.67 | 0.5 | 0.08 | 0.650 |
| Glandular stomach | 1.87 | 1.80 | 1.93 | 1.87 | 0.08 | 0.873 |
| Gut | 2.40 | 2.20 | 2.00 | 1.73 | 0.16 | 0.531 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dabbou, S.; Lauwaerts, A.; Ferrocino, I.; Biasato, I.; Sirri, F.; Zampiga, M.; Bergagna, S.; Pagliasso, G.; Gariglio, M.; Colombino, E.; et al. Modified Black Soldier Fly Larva Fat in Broiler Diet: Effects on Performance, Carcass Traits, Blood Parameters, Histomorphological Features and Gut Microbiota. Animals 2021, 11, 1837. https://doi.org/10.3390/ani11061837
Dabbou S, Lauwaerts A, Ferrocino I, Biasato I, Sirri F, Zampiga M, Bergagna S, Pagliasso G, Gariglio M, Colombino E, et al. Modified Black Soldier Fly Larva Fat in Broiler Diet: Effects on Performance, Carcass Traits, Blood Parameters, Histomorphological Features and Gut Microbiota. Animals. 2021; 11(6):1837. https://doi.org/10.3390/ani11061837
Chicago/Turabian StyleDabbou, Sihem, Angelo Lauwaerts, Ilario Ferrocino, Ilaria Biasato, Federico Sirri, Marco Zampiga, Stefania Bergagna, Giulia Pagliasso, Marta Gariglio, Elena Colombino, and et al. 2021. "Modified Black Soldier Fly Larva Fat in Broiler Diet: Effects on Performance, Carcass Traits, Blood Parameters, Histomorphological Features and Gut Microbiota" Animals 11, no. 6: 1837. https://doi.org/10.3390/ani11061837
APA StyleDabbou, S., Lauwaerts, A., Ferrocino, I., Biasato, I., Sirri, F., Zampiga, M., Bergagna, S., Pagliasso, G., Gariglio, M., Colombino, E., Narro, C. G., Gai, F., Capucchio, M. T., Gasco, L., Cocolin, L., & Schiavone, A. (2021). Modified Black Soldier Fly Larva Fat in Broiler Diet: Effects on Performance, Carcass Traits, Blood Parameters, Histomorphological Features and Gut Microbiota. Animals, 11(6), 1837. https://doi.org/10.3390/ani11061837











