Protective Effects of α-Lipoic Acid and Chlorogenic Acid on Cadmium-Induced Liver Injury in Three-Yellow Chickens
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Treatments and Chemicals
2.2. Measurement of Serum Biochemical Indexes
2.3. Histopathological Studies
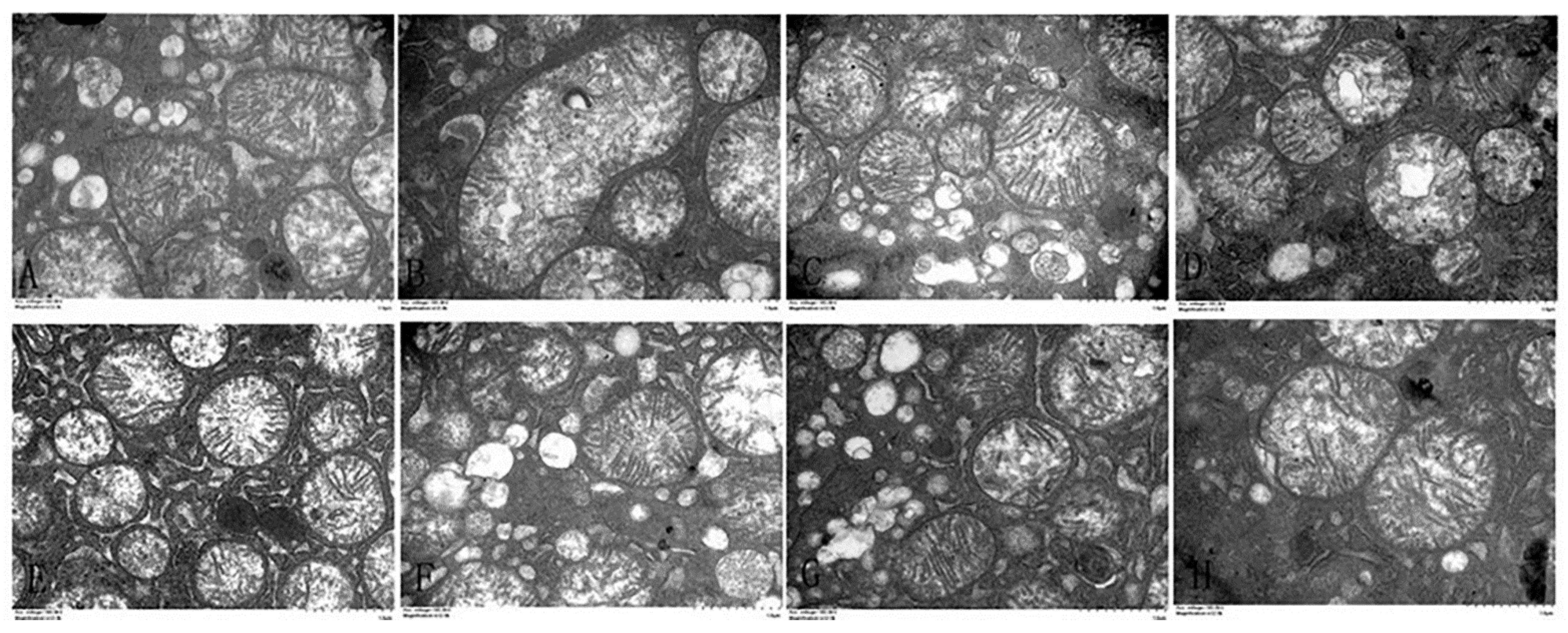
2.4. Observation of the Ultrastructure of Liver
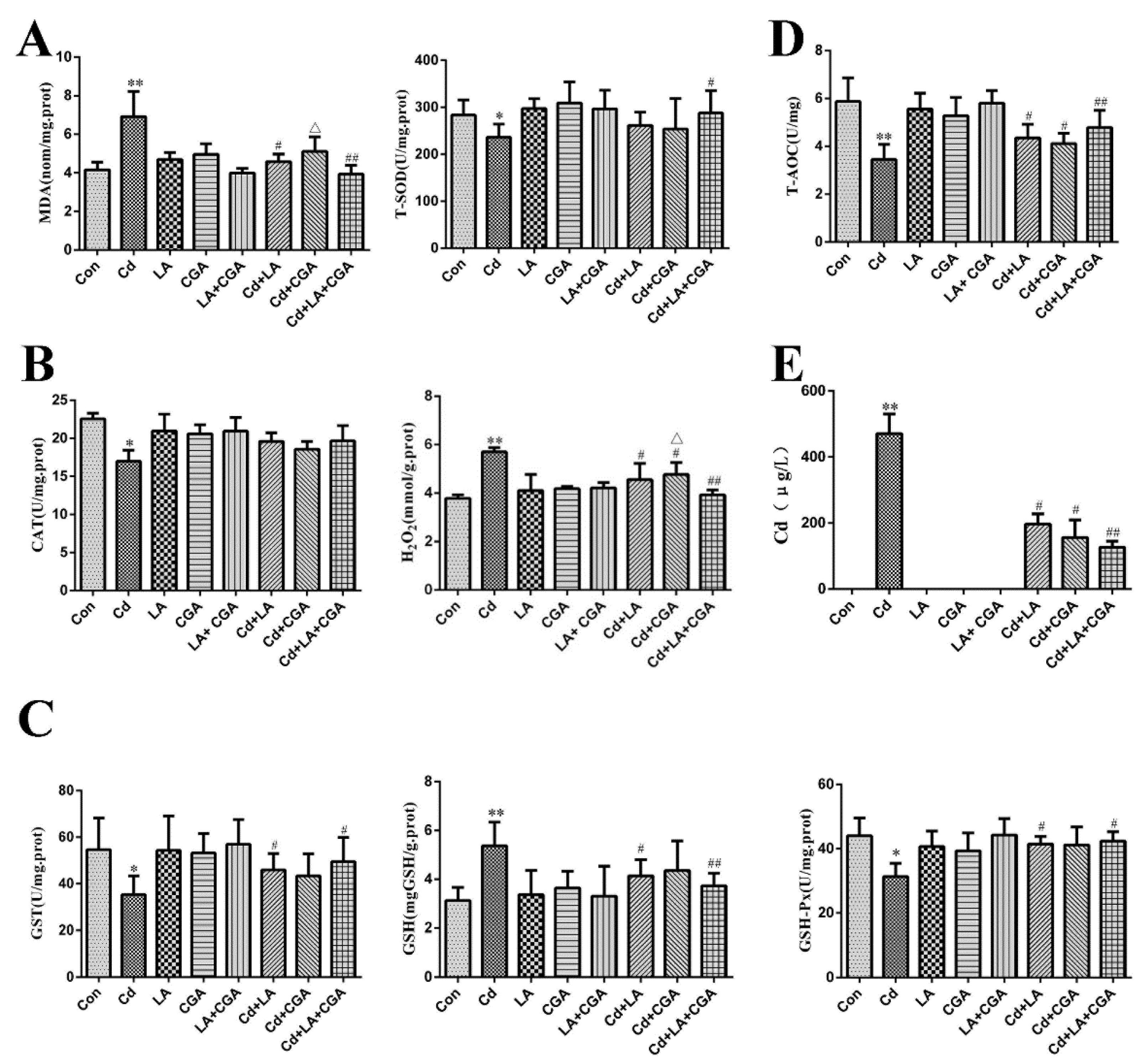
2.5. Measurement of Oxidants and Antioxidants
2.6. Determination of Trace Elements in Liver
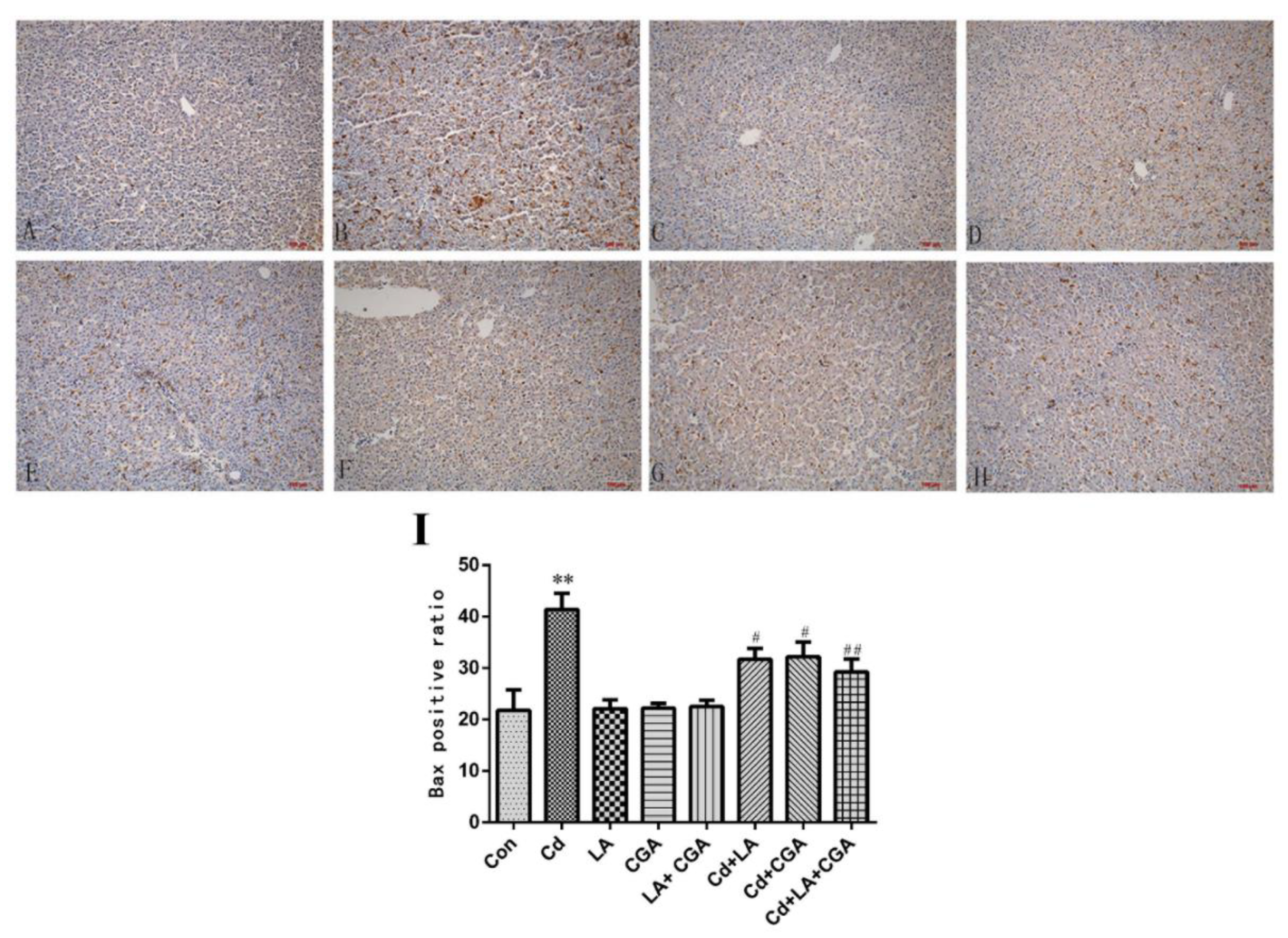
2.7. Immunohistochemical Assay of Bax Protein
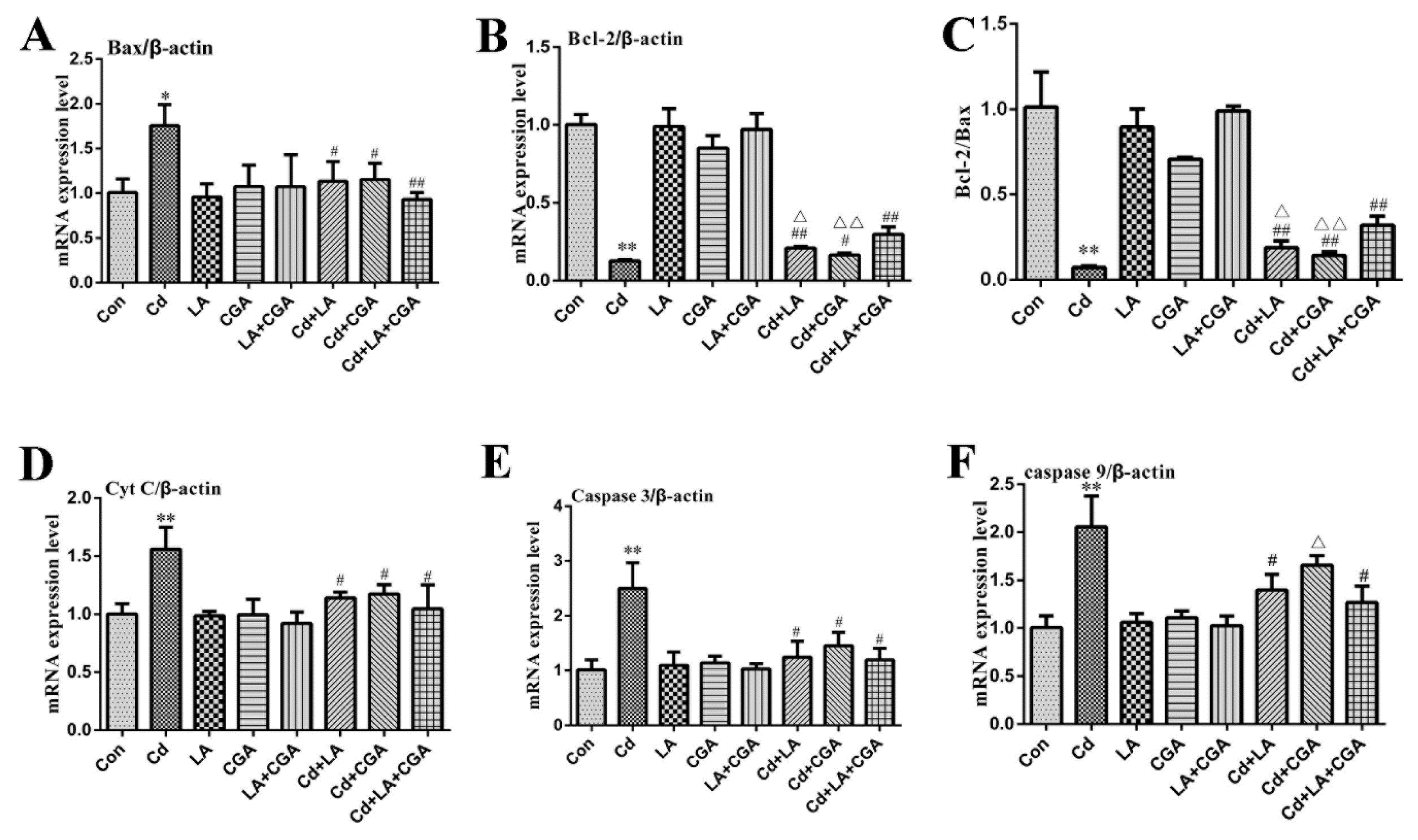
2.8. Total RNA Extraction and qRT-PCR
2.9. Statistical Analysis
3. Results
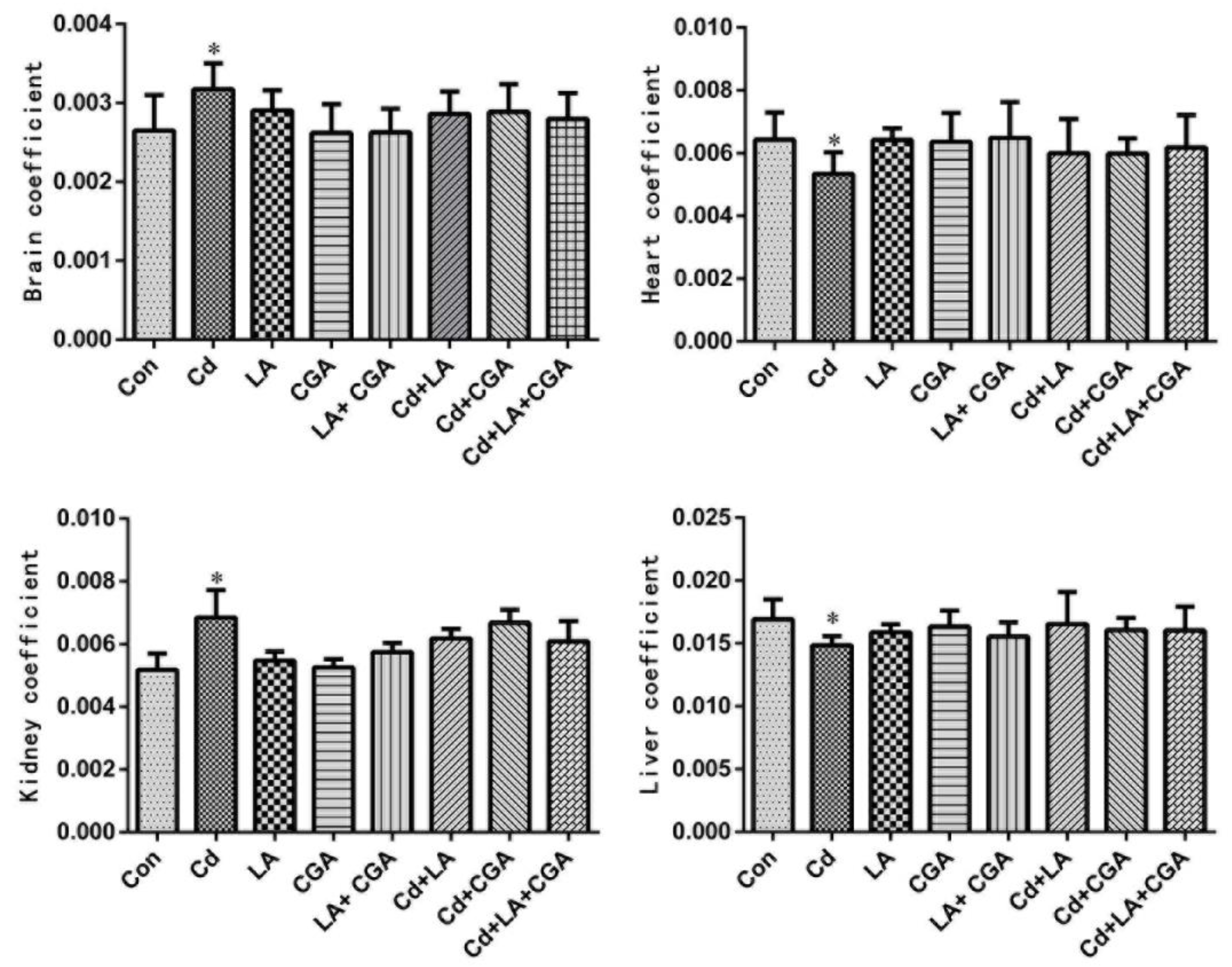
3.1. Effects of α-Lipoic Acid and Chlorogenic Acid on the Growth Performance of Cadmium-Poisoned Chickens
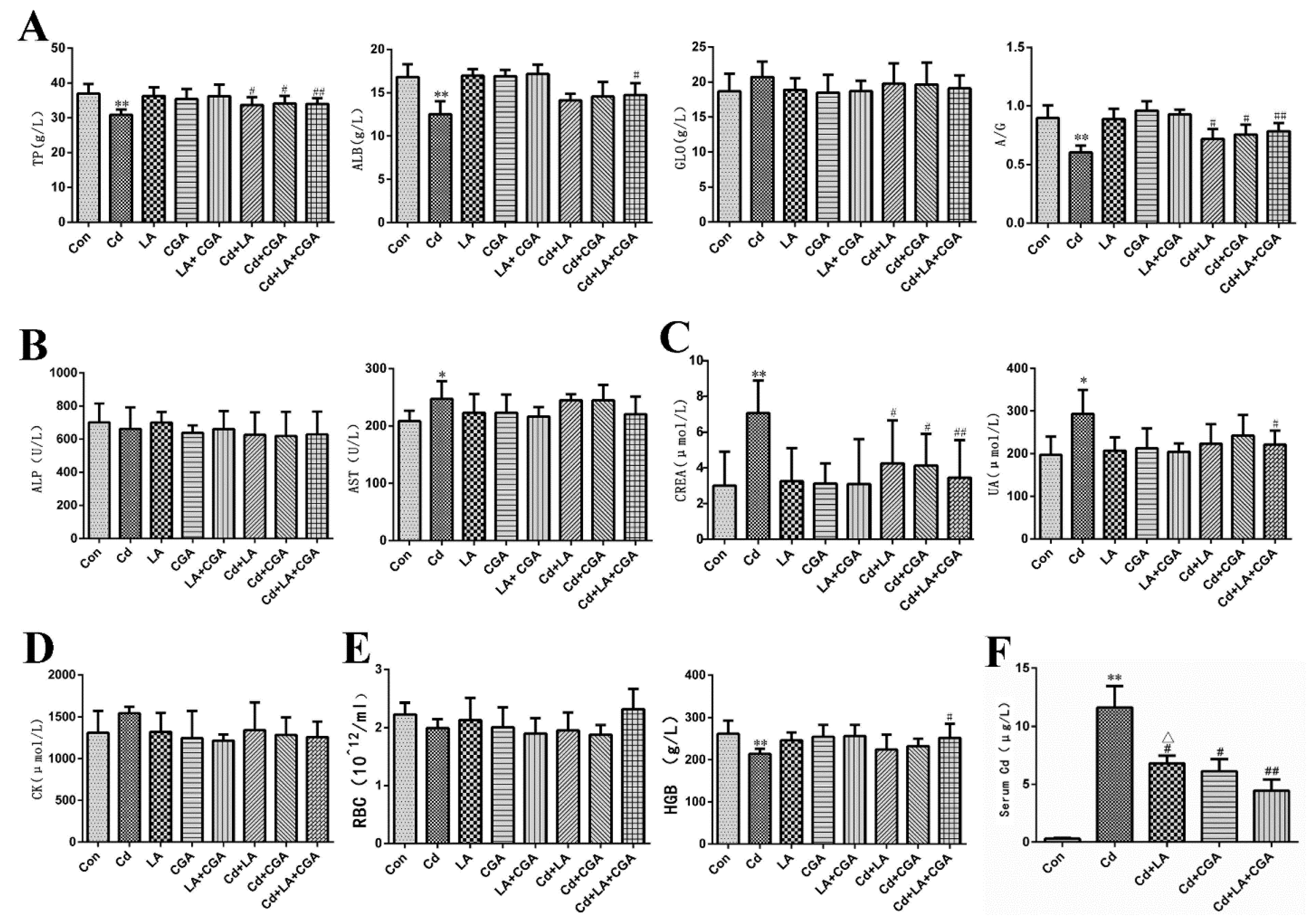
3.2. Effects of α-Lipoic Acid and Chlorogenic Acid on Serum Biochemical Markers of Cadmium-Poisoned Chickens
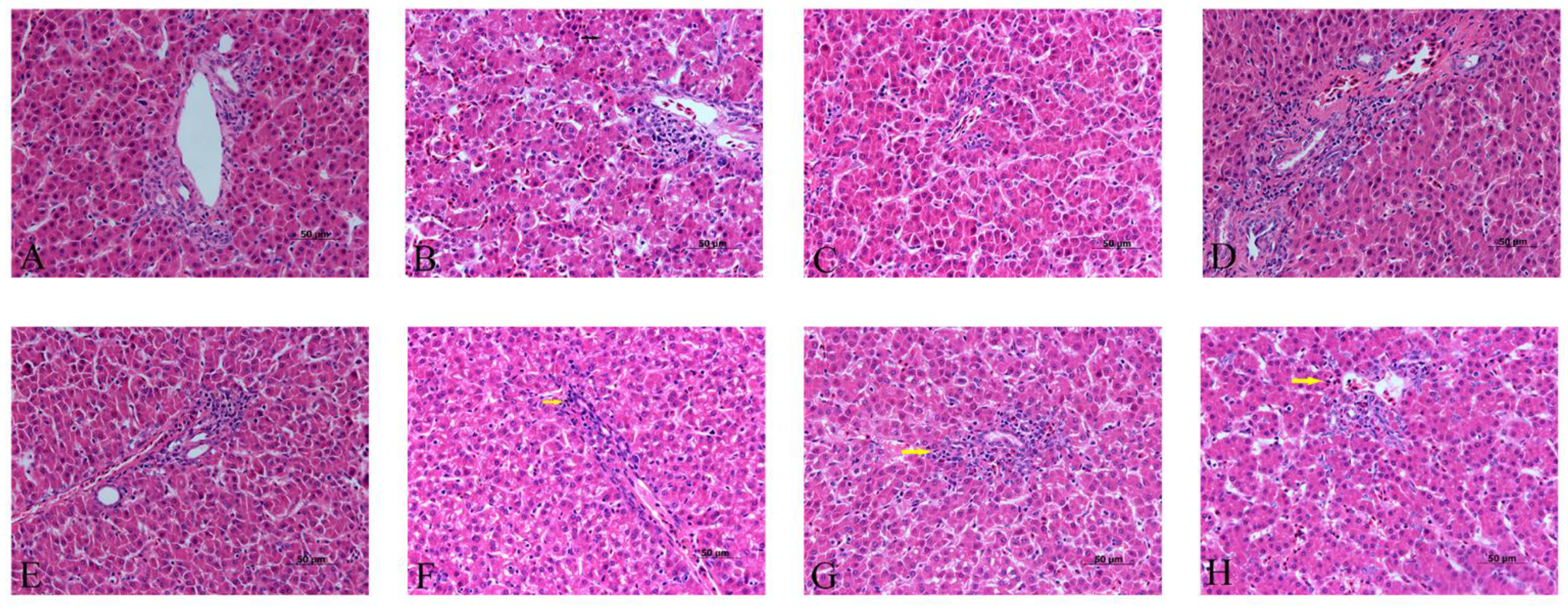
3.3. Effects of α-Lipoic Acid and Chlorogenic Acid on the Histopathology of Cadmium-Exposed Chicken Livers
3.4. Effects of α-Lipoic Acid and Chlorogenic Acid Antioxidative Properties on Cadmium-Exposed Chicken Liver
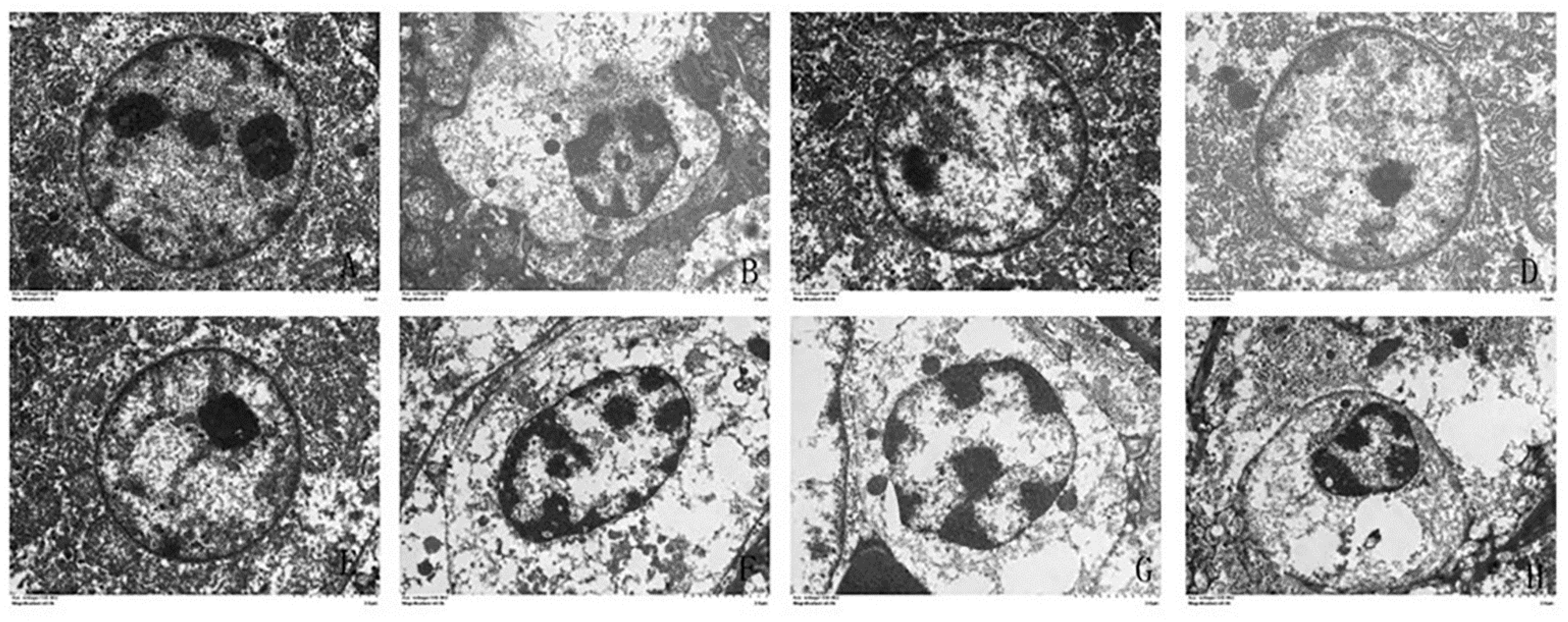
3.5. Effects of α-Lipoic Acid and Chlorogenic Acid on the Mitochondrial Apoptosis Pathway in Cadmium-Exposed Chicken Liver Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nawrot, T.S.; Staessen, J.A.; Roels, H.A.; Munters, E.; Cuypers, A.; Richart, T.; Ruttens, A.; Smeets, K.; Clijsters, H.; Vangronsveld, J. Cadmium exposure in the population: From health risks to strategies of prevention. BioMetals 2010, 23, 769–782. [Google Scholar] [CrossRef]
- Cai, J.; Zhang, Y.; Yang, J.; Liu, Q.; Zhao, R.; Hamid, S.; Wang, H.; Xu, S.; Zhang, Z. Antagonistic effects of selenium against necroptosis injury via adiponectin-necrotic pathway induced by cadmium in heart of chicken. RSC Adv. 2017, 7, 44438–44446. [Google Scholar] [CrossRef]
- Branca, J.J.V.; Fiorillo, C.; Carrino, D.; Paternostro, F.; Taddei, N.; Gulisano, M.; Pacini, A.; Becatti, M. Cadmium-Induced Oxidative Stress: Focus on the Central Nervous System. Antioxidants 2020, 9, 492. [Google Scholar] [CrossRef] [PubMed]
- Mężyńska, M.; Brzóska, M.M. Review of polyphenol-rich products as potential protective and therapeutic factors against cadmium hepatotoxicity. J. Appl. Toxicol. 2019, 39, 117–145. [Google Scholar] [CrossRef] [PubMed]
- Sanjeev, S.; Bidanchi, R.M.; Murthy, M.K.; Gurusubramanian, G.; Roy, V.K. Influence of ferulic acid consumption in ameliorating the cadmium-induced liver and renal oxidative damage in rats. Environ. Sci. Pollut. Res. 2019, 26, 20631–20653. [Google Scholar] [CrossRef]
- Martins, A.D.C., Jr.; Carneiro, M.F.H.; Grotto, D.; A Adeyemi, J.; Barbosa, F., Jr. Arsenic, cadmium, and mercury-induced hypertension: Mechanisms and epidemiological findings. J. Toxicol. Environ. Heal. Part B 2018, 21, 61–82. [Google Scholar] [CrossRef]
- Renu, K.; Chakraborty, R.; Myakala, H.; Koti, R.; Famurewa, A.C.; Madhyastha, H.; Vellingiri, B.; George, A.; Gopalakrishnan, A.V. Molecular mechanism of heavy metals (Lead, Chromium, Arsenic, Mercury, Nickel and Cadmium)-induced hepatotoxicity–A review. Chemosphere 2021, 271, 129735. [Google Scholar] [CrossRef]
- Johri, N.; Jacquillet, G.; Unwin, R. Heavy metal poisoning: The effects of cadmium on the kidney. BioMetals 2010, 23, 783–792. [Google Scholar] [CrossRef]
- Figueiredo-Pereira, M.E.; Yakushin, S.; Cohen, G. Disruption of the Intracellular Sulfhydryl Homeostasis by Cadmium-induced Oxidative Stress Leads to Protein Thiolation and Ubiquitination in Neuronal Cells. J. Biol. Chem. 1998, 273, 12703–12709. [Google Scholar] [CrossRef] [PubMed]
- Renugadevi, J.; Prabu, S.M. Cadmium-induced hepatotoxicity in rats and the protective effect of naringenin. Exp. Toxicol. Pathol. 2010, 62, 171–181. [Google Scholar] [CrossRef]
- Cheng, C.-H.; Ma, H.-L.; Deng, Y.-Q.; Feng, J.; Jie, Y.-K.; Guo, Z.-X. Oxidative stress, cell cycle arrest, DNA damage and apoptosis in the mud crab (Scylla paramamosain) induced by cadmium exposure. Chemosphere 2021, 263, 128277. [Google Scholar] [CrossRef] [PubMed]
- Dżugan, M.; Trybus, W.; Lis, M.; Wesołowska, M.; Trybus, E.; Kopacz-Bednarska, A.; Król, T. Cadmium-induced ultrastructural changes in primary target organs of developing chicken embryos (Gallus domesticus). J. Trace Elem. Med. Biol. 2018, 50, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Gobe, G.; Crane, D. Mitochondria, reactive oxygen species and cadmium toxicity in the kidney. Toxicol. Lett. 2010, 198, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.-M.; Wang, I.-C.; Yang, J.-L. Roles of JNK, p38 and ERK mitogen-activated protein kinases in the growth inhibition and apoptosis induced by cadmium. Carcinogenesis 2000, 21, 1423–1432. [Google Scholar] [CrossRef]
- Dżugan, M.; Lis, M.W.; Droba, M.; Niedziółka, J.W. Protective effect of zinc on cadmium embryotoxicity and antioxidant status of blood plasma in newly hatched chicks. J. Environ. Sci. Heal. Part A 2012, 47, 1288–1293. [Google Scholar] [CrossRef] [PubMed]
- Gong, P.; Chen, F.-X.; Wang, L.; Wang, J.; Jin, S.; Ma, Y.-M. Protective effects of blueberries (Vaccinium corymbosum L.) extract against cadmium-induced hepatotoxicity in mice. Environ. Toxicol. Pharmacol. 2014, 37, 1015–1027. [Google Scholar] [CrossRef]
- El-Senousey, H.; Chen, B.; Wang, J.; Atta, A.; Mohamed, F.; Nie, Q. Effects of dietary vitamin C, vitamin E, and alpha-lipoic acid supplementation on the antioxidant defense system and immune-related gene expression in broilers exposed to oxidative stress by dexamethasone. Poult. Sci. 2018, 97, 30–38. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, Q.; Ci, X.; Chen, S.; Xie, Z.; Li, H.; Zhang, H.; Chen, F.; Xie, Q. Evaluation of the efficacy of chlorogenic acid in reducing small intestine injury, oxidative stress, and inflammation in chickens challenged with Clostridium perfringens type A. Poult. Sci. 2020, 99, 6606–6618. [Google Scholar] [CrossRef]
- Târtea, G.C.; Florescu, D.R.; Mihailovici, A.R.; Donoiu, I.; Istrătoaie, O. Alpha-lipoic acid and vitamin B complex slow down the changes in mice diabetic cardiomyopathy. Rom. J. Morphol. Embryol. 2020, 61, 521–528. [Google Scholar] [CrossRef]
- Hiller, S.; DeKroon, R.; Hamlett, E.D.; Xu, L.; Osorio, C.; Robinette, J.; Winnik, W.; Simington, S.; Maeda, N.; Alzate, O.; et al. Alpha-lipoic acid supplementation protects enzymes from damage by nitrosative and oxidative stress. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2016, 1860, 36–45. [Google Scholar] [CrossRef]
- Markiewicz-Górka, I.; Pawlas, K.; Jaremków, A.; Januszewska, L.; Pawłowski, P.; Pawlas, N. Alleviating Effect of α-Lipoic Acid and Magnesium on Cadmium-Induced Inflammatory Processes, Oxidative Stress and Bone Metabolism Disorders in Wistar Rats. Int. J. Environ. Res. Public Heal. 2019, 16, 4483. [Google Scholar] [CrossRef]
- Bustamante, J. α-Lipoic Acid in Liver Metabolism and Disease. Free. Radic. Biol. Med. 1998, 24, 1023–1039. [Google Scholar] [CrossRef]
- Park, J.-S.; Han, K. The Spermatogenic Effect of Yacon Extract and Its Constituents and Their Inhibition Effect of Testosterone Metabolism. Biomol. Ther. 2013, 21, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Deng, W.; Liu, H. Effects of chlorogenic acid-enriched extract from Eucommia ulmoides leaf on performance, meat quality, oxidative stability, and fatty acid profile of meat in heat-stressed broilers. Poult. Sci. 2019, 98, 3040–3049. [Google Scholar] [CrossRef] [PubMed]
- A Abduljaleel, S.; Shuhaimi-Othman, M. Toxicity of Cadmium and Lead in Gallus gallus domesticus Assessment of Body Weight and Metal Content in Tissues after Metal Dietary Supplements. Pak. J. Biol. Sci. 2013, 16, 1551–1556. [Google Scholar] [CrossRef] [PubMed]
- El-Senousey, H.K.; Fouad, A.M.; Yao, J.H.; Zhang, Z.G.; Shen, Q.W. Dietary Alpha Lipoic Acid Improves Body Composition, Meat Quality and Decreases Collagen Content in Muscle of Broiler Chickens. Asian Australas. J. Anim. Sci. 2013, 26, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Huang, S.; Tong, X.; Zhang, L.; Jiang, X.; Zhang, H.; Mehmood, K.; Li, J. Chlorogenic Acid Alleviates Thiram-Induced Tibial Dyschondroplasia by Modulating Caspases, BECN1 Expression and ECM Degradation. Int. J. Mol. Sci. 2019, 20, 3160. [Google Scholar] [CrossRef]
- Pinheiro, F.C.; Babos, D.V.; Barros, A.I.; Pereira-Filho, E.R.; Nóbrega, J.A. Microwave-assisted digestion using dilute nitric acid solution and investigation of calibration strategies for determination of As, Cd, Hg and Pb in dietary supplements using ICP-MS. J. Pharm. Biomed. Anal. 2019, 174, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Branca, J.J.V.; Pacini, A.; Gulisano, M.; Taddei, N.; Fiorillo, C.; Becatti, M. Cadmium-Induced Cytotoxicity: Effects on Mitochondrial Electron Transport Chain. Front. Cell Dev. Biol. 2020, 8, 604377. [Google Scholar] [CrossRef]
- Zhang, C.; Huang, Y.; Talukder, M.; Ge, J.; Lv, M.-W.; Bi, S.-S.; Li, J.-L. Selenium sources differ in their potential to alleviate the cadmium-induced testicular dysfunction. Environ. Pollut. 2020, 267, 115610. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Mohajeri, M.; Rezaee, M.; Sahebkar, A. Cadmium-induced toxicity is rescued by curcumin: A review. BioFactors 2017, 43, 645–661. [Google Scholar] [CrossRef] [PubMed]
- Abou-Kassem, D.E.; Mahrose, K.M.; Alagawany, M. The role of vitamin E or clay in growing Japanese quail fed diets polluted by cadmium at various levels. Animal 2016, 10, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Dirksen, K.; Burgener, I.; Rothuizen, J.; Ingh, T.V.D.; Penning, L.; Spee, B.; Fieten, H. Sensitivity and Specificity of Plasma ALT, ALP, and Bile Acids for Hepatitis in Labrador Retrievers. J. Veter. Intern. Med. 2017, 31, 1017–1027. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Fang, Z.; Hang, D.; Wang, F.; Polychronidis, G.; Wang, L.; Lo, C.; Wang, K.; Zhong, R.; Knudsen, M.D.; et al. Circulating liver function markers and colorectal cancer risk: A prospective cohort study in the UK Biobank. Int. J. Cancer 2021, 148, 1867–1878. [Google Scholar] [CrossRef]
- Oladipo, O.O.; Ayo, J.O.; Ambali, S.F.; Mohammed, B. Evaluation of hepatorenal impairments in Wistar rats coexposed to low-dose lead, cadmium and manganese: Insights into oxidative stress mechanism. Toxicol. Mech. Methods 2016, 26, 674–684. [Google Scholar] [CrossRef]
- Wang, X.; Han, C.; Cui, Y.; Li, S.; Jin, G.; Shi, W.; Bao, Y. Florfenicol causes excessive lipid peroxidation and apoptosis induced renal injury in broilers. Ecotoxicol. Environ. Saf. 2021, 207, 111282. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Awan, Z.; Mumtaz, S.; Shakir, H.A.; Ahmad, F.; Ulhaq, M.; Tahir, H.M.; Awan, M.S.; Sharif, S.; Irfan, M.; et al. Cardiac toxicity of heavy metals (cadmium and mercury) and pharmacological intervention by vitamin C in rabbits. Environ. Sci. Pollut. Res. 2020, 27, 29266–29279. [Google Scholar] [CrossRef]
- Rogival, D.; Scheirs, J.; De Coen, W.; Verhagen, R.; Blust, R. Metal Blood Levels and Hematological Characteristics in Wood Mice (Apodemus sylvaticus L.) along a Metal Pollution Gradient. Environ. Toxicol. Chem. 2006, 25, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Horiguchi, H.; Oguma, E.; Kayama, F. Cadmium Induces Anemia through Interdependent Progress of Hemolysis, Body Iron Accumulation, and Insufficient Erythropoietin Production in Rats. Toxicol. Sci. 2011, 122, 198–210. [Google Scholar] [CrossRef]
- Zhu, M.; Li, H.; Bai, L.; Wang, L.; Zou, X. Histological changes, lipid metabolism, and oxidative and endoplasmic reticulum stress in the liver of laying hens exposed to cadmium concentrations. Poult. Sci. 2020, 99, 3215–3228. [Google Scholar] [CrossRef]
- Bjørklund, G.; Crisponi, G.; Nurchi, V.M.; Cappai, R.; Djordjevic, A.B.; Aaseth, J. A Review on Coordination Properties of Thiol-Containing Chelating Agents Towards Mercury, Cadmium, and Lead. Molecules 2019, 24, 3247. [Google Scholar] [CrossRef]
- De Moura, C.F.G.; Ribeiro, F.A.P.; Handan, B.A.; Aguiar, O.; Oshima, C.T.F.; Ribeiro, D.A. Grape Juice Concentrate Protects Rat Liver Against Cadmium Intoxication: Histopathology, Cytochrome C and Metalloproteinases Expression. Drug Res. 2016, 66, 339–344. [Google Scholar] [CrossRef]
- Teng, X.; Zhang, W.; Song, Y.; Wang, H.; Ge, M.; Zhang, R. Protective effects of Ganoderma lucidum triterpenoids on oxidative stress and apoptosis in the spleen of chickens induced by cadmium. Environ. Sci. Pollut. Res. 2019, 26, 23967–23980. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Yin, H.; Yang, Z.; Tan, M.; Wang, F.; Chen, K.; Zuo, Z.; Shu, G.; Cui, H.; Ouyang, P.; et al. Vitamin E protects against cadmium-induced sub-chronic liver injury associated with the inhibition of oxidative stress and activation of Nrf2 pathway. Ecotoxicol. Environ. Saf. 2021, 208, 111610. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, H.; Chang, W.; Chen, R.; Xu, S.; Tao, D. Protective effects of selenium yeast against cadmium-induced necroptosis via inhibition of oxidative stress and MAPK pathway in chicken liver. Ecotoxicol. Environ. Saf. 2020, 206, 111329. [Google Scholar] [CrossRef]
- Petrović, T.; Kijanović, A.; Tomašević, N.K.; Gavrić, J.; Despotović, S.; Gavrilović, B.; Radovanović, T.; Vukov, T.; Faggio, C.; Prokić, M. Effects of Desiccation on Metamorphic Climax in Bombina variegata: Changes in Levels and Patterns of Oxidative Stress Parameters. Animal 2021, 11, 953. [Google Scholar] [CrossRef]
- El-Sokkary, G.H.; Nafady, A.A.; Shabash, E.H. Melatonin administration ameliorates cadmium-induced oxidative stress and morphological changes in the liver of rat. Ecotoxicol. Environ. Saf. 2010, 73, 456–463. [Google Scholar] [CrossRef]
- Souid, G.; Souayed, N.; Yaktiti, F.; Maaroufi, K. Effect of acute cadmium exposure on metal accumulation and oxidative stress biomarkers of Sparus aurata. Ecotoxicol. Environ. Saf. 2013, 89, 1–7. [Google Scholar] [CrossRef]
- Islam, O.; Bacchetti, T.; Ferretti, G. Alterations of Antioxidant Enzymes and Biomarkers of Nitro-oxidative Stress in Tissues of Bladder Cancer. Oxidative Med. Cell. Longev. 2019, 2019, 2730896. [Google Scholar] [CrossRef] [PubMed]
- Anetor, J.I. Rising Environmental Cadmium Levels in Developing Countries: Threat to Genome Stability and Health. J. Environ. Anal. Toxicol. 2012, 27, 103–115. [Google Scholar] [CrossRef]
- Ou, L.; Wang, H.; Wu, Z.; Wang, P.; Yang, L.; Li, X.; Sun, K.; Zhu, X.; Zhang, R. Effects of cadmium on osteoblast cell line: Exportin 1 accumulation, p-JNK activation, DNA damage and cell apoptosis. Ecotoxicol. Environ. Saf. 2021, 208, 111668. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Nie, G.; Zhuang, Y.; Hu, R.; Wu, H.; Xing, C.; Li, G.; Hu, G.; Yang, F.; Zhang, C. Inhibition of autophagy enhances cadmium-induced apoptosis in duck renal tubular epithelial cells. Ecotoxicol. Environ. Saf. 2020, 205, 111188. [Google Scholar] [CrossRef] [PubMed]
- Fan, R.-F.; Li, Z.-F.; Zhang, D.; Wang, Z.-Y. Involvement of Nrf2 and mitochondrial apoptotic signaling in trehalose protection against cadmium-induced kidney injury. Metallomics 2020, 12, 2098–2107. [Google Scholar] [CrossRef] [PubMed]
- Jiaxin, S.; Shengchen, W.; Yirong, C.; Shuting, W.; Shu, L. Cadmium exposure induces apoptosis, inflammation and immunosuppression through CYPs activation and antioxidant dysfunction in common carp neutrophils. Fish Shellfish Immunol. 2020, 99, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Ben, P.; Zhang, Z.; Xuan, C.; Sun, S.; Shen, L.; Gao, Y.; Cao, X.; Zhou, Y.; Lan, L.; Yin, Z.; et al. Protective Effect of l-Theanine on Cadmium-Induced Apoptosis in PC12 Cells by Inhibiting the Mitochondria-Mediated Pathway. Neurochem. Res. 2015, 40, 1661–1670. [Google Scholar] [CrossRef] [PubMed]
| Gene Name | Primer Sequence |
|---|---|
| β-actin | F: CCGCTCTA TGAAGGCTACGC |
| R: CTCTCGGCTGTGGTGGTGAA | |
| Bax | F: TCCATTCAGGTTCTCTTGACC |
| R: GCCAAACATCCAAACACAGA | |
| Bcl-2 | F: GAGTTCGGCGGCGTGATGTG |
| R: TTCAGGTACTCGGTCATCCAGGTG | |
| Cyt C | F: CTTCTTCCTCCTGGTGAACG |
| R: GCACTCCGAAAGTCTCCTGA | |
| Caspase 3 | F: CTGAAGGCTCCTGGTTTA |
| R: TGCCACT CTGCGATTTAC | |
| Caspase 9 | F: ATTCCTTTCCAGGCTCCATC |
| R: CACTCACCTTGTCCCTCCAG |
| Group | Initial Weight (g) | 30 d Weight (g) | 60 d Weight (g) | 90 d Weight (g) |
|---|---|---|---|---|
| Control | 76.6 ± 6.6 | 386.2 ± 59.1 | 761.5 ± 138.9 | 1067.0 ± 215.7 |
| Cd | 79.2 ± 8.3 | 327.1 ± 32.9 * | 644.5 ± 66.9 * | 892.1 ± 66.5 * |
| α-LA | 81.3 ± 6.0 | 373.6 ± 43.1 | 784.2 ± 62.7 | 1107.7 ± 108.3 |
| CGA | 78.2 ± 6.2 | 364.5 ± 37.7 | 764.1 ± 99.0 | 1108.0 ± 102.0 |
| α-LA + CGA | 80.8 ± 6.4 | 376.1 ± 40.4 | 757.0 ± 83.4 | 1061.2 ± 76.9 |
| Cd + α-LA | 76.1 ± 3.1 | 350.4 ± 30.1 | 692.0 ± 78.8 | 994.6 ± 123.2 |
| Cd + CGA | 79.5 ± 5.6 | 360.6 ± 54.8 | 764.2 ± 86.3 # | 995.7 ± 158.1 |
| Cd + α-LA + CGA | 77.9 ± 5.2 | 360.3 ± 31.8 | 726.2 ± 96.0 | 993.9 ± 107.7 |
| Group | Mn (μg/g) | Fe (μg/g) | Cu (μg/g) | Zn (μg/g) | Se (μg/g) |
|---|---|---|---|---|---|
| Control | 16.69 ± 1.78 | 565.22 ± 17.09 | 20.61 ± 0.76 | 175.08 ± 28.23 | 4.13 ± 0.20 |
| Cd | 7.23 ± 0.54 ** | 354.72 ± 37.35 ** | 11.62 ± 0.89 ** | 103.61 ± 3.58 ** | 2.75 ± 0.12 ** |
| α-LA | 17.8 ± 1.37 | 668.32 ± 138.6 | 21.76 ± 2.92 | 181.62 ± 7.18 | 4.32 ± 0.75 |
| CGA | 17.71 ± 0.68 | 606.53 ± 119.80 | 23.50 ± 1.00 | 184.24 ± 7.84 | 4.57 ± 0.15 |
| α-LA + CGA | 16.96 ± 1.57 | 582.98 ± 43.73 | 22.85 ± 2.04 | 173.39 ± 5.95 | 4.34 ± 0.18 |
| Cd + α-LA | 11.59 ± 0.99 # | 468.42 ± 35.83 | 15.48 ± 0.54 # | 130.46 ± 6.76 # | 3.39 ± 0.61 |
| Cd + CGA | 9.74 ± 0.57 # | 418.42 ± 35.83 | 14.03 ± 1.48 | 115.22 ± 6.61 Δ | 2.98 ± 0.32 |
| Cd + α-LA + CGA | 12.43 ± 0.79 ## | 477.46 ± 17.78 # | 17.24 ± 1.17 # | 154.32 ± 5.77 # | 3.82 ± 0.25 # |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, J.; Chang, X.; Zou, H.; Gu, J.; Yuan, Y.; Liu, X.; Liu, Z.; Bian, J. Protective Effects of α-Lipoic Acid and Chlorogenic Acid on Cadmium-Induced Liver Injury in Three-Yellow Chickens. Animals 2021, 11, 1606. https://doi.org/10.3390/ani11061606
Shi J, Chang X, Zou H, Gu J, Yuan Y, Liu X, Liu Z, Bian J. Protective Effects of α-Lipoic Acid and Chlorogenic Acid on Cadmium-Induced Liver Injury in Three-Yellow Chickens. Animals. 2021; 11(6):1606. https://doi.org/10.3390/ani11061606
Chicago/Turabian StyleShi, Jiabin, Xiaocui Chang, Hui Zou, Jianhong Gu, Yan Yuan, Xuezhong Liu, Zongping Liu, and Jianchun Bian. 2021. "Protective Effects of α-Lipoic Acid and Chlorogenic Acid on Cadmium-Induced Liver Injury in Three-Yellow Chickens" Animals 11, no. 6: 1606. https://doi.org/10.3390/ani11061606
APA StyleShi, J., Chang, X., Zou, H., Gu, J., Yuan, Y., Liu, X., Liu, Z., & Bian, J. (2021). Protective Effects of α-Lipoic Acid and Chlorogenic Acid on Cadmium-Induced Liver Injury in Three-Yellow Chickens. Animals, 11(6), 1606. https://doi.org/10.3390/ani11061606







