1. Introduction
Cognition is a system involved in knowledge acquisition, and people who own dogs tend to claim that these animals have great cognitive skills [
1]. Dogs are widely used in cognition studies, mainly due to their cognitive capacities, which, depending on the task, may be greater than those of other species, such as wolves and chimpanzees [
2,
3]. Throughout domestication, dogs went through a process of selection of skills for interacting and communicating with humans, including relying on human gestures and making eye contact [
4,
5], which may have contributed to the development of their cognitive resources. For instance, dogs follow human pointing gestures, an ability that has been related to the absolute brain size of the species [
6,
7]. In tasks which consisted of understanding social cues given by humans to find hidden food, although dogs tended to act strongly based on olfactory cues, their performance was evident [
8,
9].
Dogs evolved while coexisting with humans, and friendly interactions between the two species are known to promote positive effects on canine welfare [
10,
11]. The selection for canine characteristics to facilitate the interactions of these animals with humans, as well as the intense coexistence between the two species may have been responsible for stimulating the development of attachment bonds in dogs. Dogs tend to play and explore an unfamiliar environment more when their owners are present [
12]; when dogs feel threatened by a stranger, both their heart rate and its variation increase if the owner is not present, whereas his/her presence reduces these increases [
13]. Taking effects like these into consideration, researchers state that attachment bonds between dogs and humans are similar to those developed between human babies and their mothers [
14]. From this perspective, the separation of dogs from their attachment figures—their owners—may favor the development of separation-related problems, which in turn, increases the chance of developing a pessimistic cognitive bias, an effect common in pet dogs, and also reported in dogs from reallocation centers [
15].
The life history of dogs was shown to affect relevant aspects of their social competence, such as dog’s persistence on gazing behavior, or at being close to humans [
16]. For instance, shelter and companion dogs looked more at human faces, and spent more time close to humans, compared to dogs that lived on the streets [
16]. Shelter dogs performed well in tests that involved vocal cues but had difficulty learning visual cues [
17]. In another study, using an A-not-B task, in which dogs had to learn that a reward was relocated and reach it [
18]—shelter dogs did not differ from companion dogs in the number of correct responses in the training stage, but learned more slowly and had a greater number of incorrect choices during the test stage. Such differences might be related to the fewer opportunities shelter dogs have during their life to observe people’s behaviors, or to be rewarded for following their directions. Companion dogs live with humans; therefore, they are constantly being stimulated by humans, and some of their responses in the presence of certain stimuli increase because these have led to a high probability of reinforcement in the past [
19]. On the other hand, shelter dogs do not underperform companion dogs in all abilities. No differences were found between these dogs in discriminating generous human attitudes from selfish ones [
20], an important skill for social interaction. Shelter dogs have also shown contagion with human yawning, although this phenomenon depends on context and the dog’s excitement [
21].
Friendly human–animal interactions, understood as mutual interactions between human beings and animals that have a relaxing effect on animals, have been recommended in order to improve the welfare of animals [
22]. A type of interspecific interaction that is common between people and dogs is training [
23]. Training is usually performed with the use of operant conditioning techniques, which consists in an individual associating his/her actions (e.g., giving a paw) with a consequence (e.g., receiving a reward), and this association usually has effects on the probability of this individual repeating that action [
24].
A previous study by our team [
10] found out that positive reinforcement training, a type of operant conditioning, had relaxing effects on dogs and wolves, an effect which varied with trainer. Studies have pointed to human influences on dogs’ learning processes. Although some studies have found no effect of training method on the behavior or welfare of animals [
25], some have raised evidence that the use of punitive methods in training may, for instance, affect a dog’s ability to learn a new task [
26], or increase their stress levels (e.g., [
27,
28]). A dog’s ability to attend to a cue may also be influenced by the vocal information the trainer includes before the cue (e.g., saying the dog’s name or an unknown word [
29]); even if the dog is familiar with the cue, this vocal information may reduce the dog’s performance. Studies have been conducted on the motivation and effects of people’s tones of voice when communicating with dogs (e.g., [
30,
31,
32]) but as far as we know, no studies have investigated associations between trainer vocal behavior and the behavioral responses of dogs during training. A study of vocal cue learning, considering the effect of dog origin and possible associations between trainers’ behaviors and dogs’ responses, has the potential to improve our understanding of dog learning processes and abilities.
The purpose of this study was to investigate how: (a) the origin of dogs (in terms of their interactions with humans), and (b) trainer behavior correlate with dogs’ abilities to learn vocal cues. We hypothesized that shelter dogs would perform differently from companion dogs in the tasks. We also expected some behaviors of trainers to be correlated with animals’ responses. More specifically, our predictions for this study were: (a) shelter dogs would need a greater number of sessions to learn the cues, more repetitions of cues, and would show greater latencies in responding to them; (b) both shelter and companion dogs would perform better in sessions when the trainers exhibit friendly behaviors for longer. Shelter dogs responded to more cues per session, with shorter latencies and fewer cue repetitions. Furthermore, these dogs showed more excitement during the sessions, maybe due to their history of social deprivation. However, the data supported our second prediction—the use of a neutral tone of voice and laughter was associated with dogs’ performance and excitement. Our outcomes suggest that friendly interactions with humans may contribute to dogs’ performance and—possibly—welfare.
4. Discussion
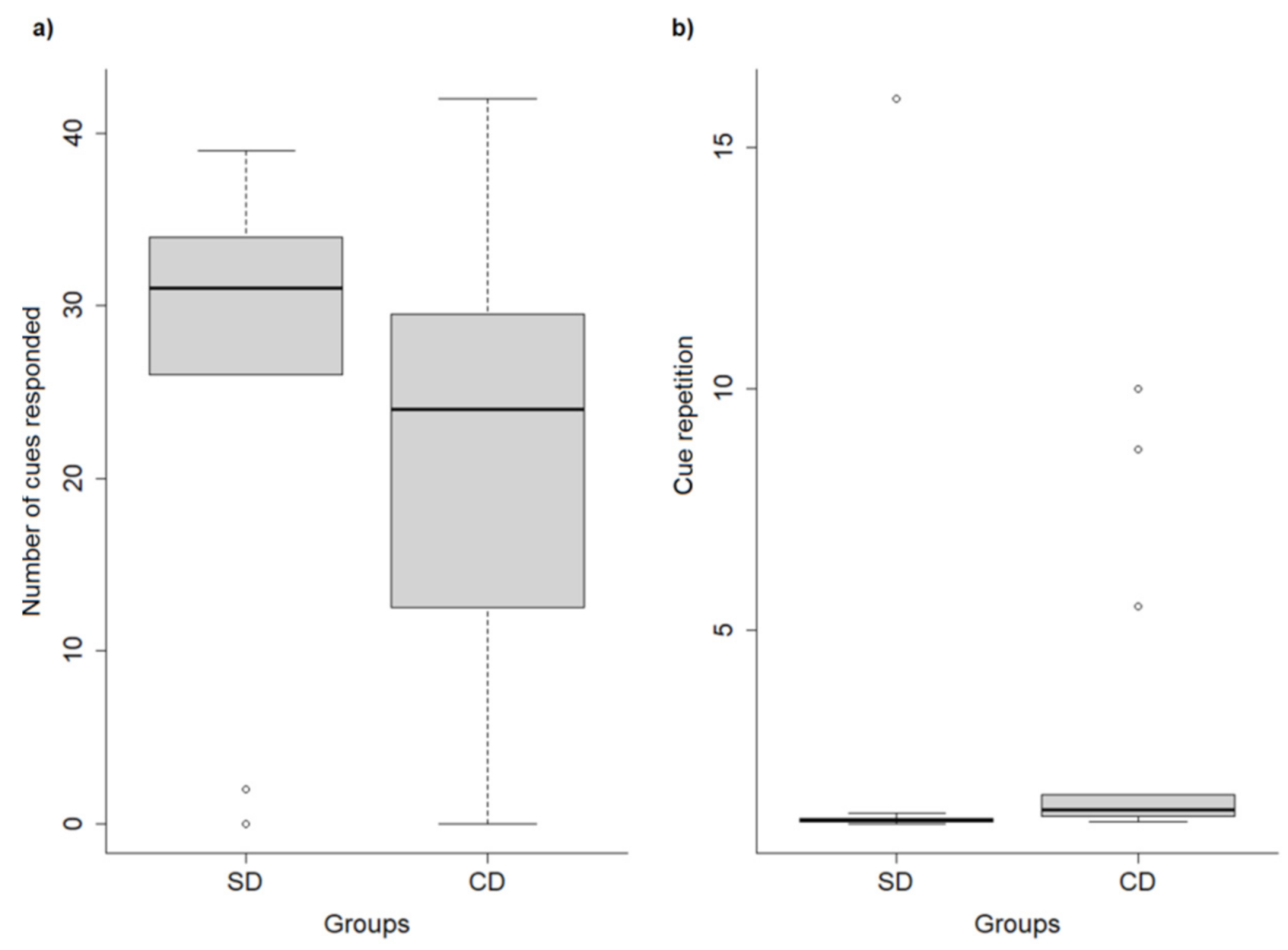
In this study, we aimed to assess the responses of shelter and companion dogs during operant conditioning training to learn two vocal cues, considering the associations between trainer behavior and dogs’ responses. We found dog origin (shelter vs. companion), dog sex, and trainer (Trainer 1 vs. Trainer 2) to be associated with differences in dog performance. Shelter dogs responded to more cues, with shorter latencies, and fewer repetitions of cues required for them to respond than CDs. Male dogs took more time, needed more cue repetitions to respond, and responded to fewer cues than females. We also found correlations between the behavior of the trainers and dogs’ performance and general behaviors. Although the use of a reproachful speech was positively associated with training performance (lower latencies, fewer cue repetitions, and more cues responded to), it was also linked to shorter durations of dogs within one meter of the trainer and wagging their tails, and to longer durations of NTB. On the other hand, the use of neutral and gentle speech was associated with a greater number of cues responded to, and more time spent tail-wagging. Moreover, the orientation of trainers to dogs was positively associated with the orientation of the dogs to the trainers.
Some studies have found SDs to perform similarly to CDs (e.g., [
18,
46]). The SDs in our study, however, performed better than CDs in three out of five of the parameters measured: the number of cues responded to, latency in responding to cues, and the number of cues repeated. This indicates that, despite their routine with a low rate of interaction with humans, the ability of shelter dogs to learn basic vocal tasks was not impaired. One possible explanation has to do with ontogenic homeostasis. Ontogenic homeostasis is a process in which, despite living in a poor environment or having defective genes, an individual somehow develops normally [
47]. Such phenomenon applies usually to systems or abilities that are essential for survival, such as in social cognition. An example of such an effect is the classical study by the Harlows involving the deprivation of contact with the mother in infant rhesus monkeys, who, although exhibiting abnormal social and sexual behaviors, presented normal physical development [
48]. Another study that reported this effect—this one on human cognitive development—found that, despite the nutritional deprivation of mothers during their pregnancy throughout the Dutch famine of 1944–1945, their children did not show differences in mental performance in relation to children whose mothers did not experience deprivation, even nineteen years later [
49]. This buffering effect on cognition development/maintenance might be responsible for the good performance of SDs in our study. However, as ontogenic homeostasis processes refer to the deprivation animals suffer while developing, and we did not have information about the age at which the study shelter dogs arrived at the shelters, this explanation may not apply to all of them. Nevertheless, the deprivation of human contact in the experience of SDs may have predisposed them to interact with unfamiliar people (i.e., the trainers) more than CDs did [
11,
12,
50,
51]. Another factor that might have contributed to the difference in performance between SDs and CDs in our study is the separation of CDs from their owners during training. Although the protocol was standardized for both dog groups, CDs might have been affected more by the procedure due to a strong attachment to their owners [
12,
14]. However, we think this is improbable, since their reactions when the owners left consisted mostly of looking at the door for a few seconds; none of the CDs remained by the door or refused to take part in the training sessions.
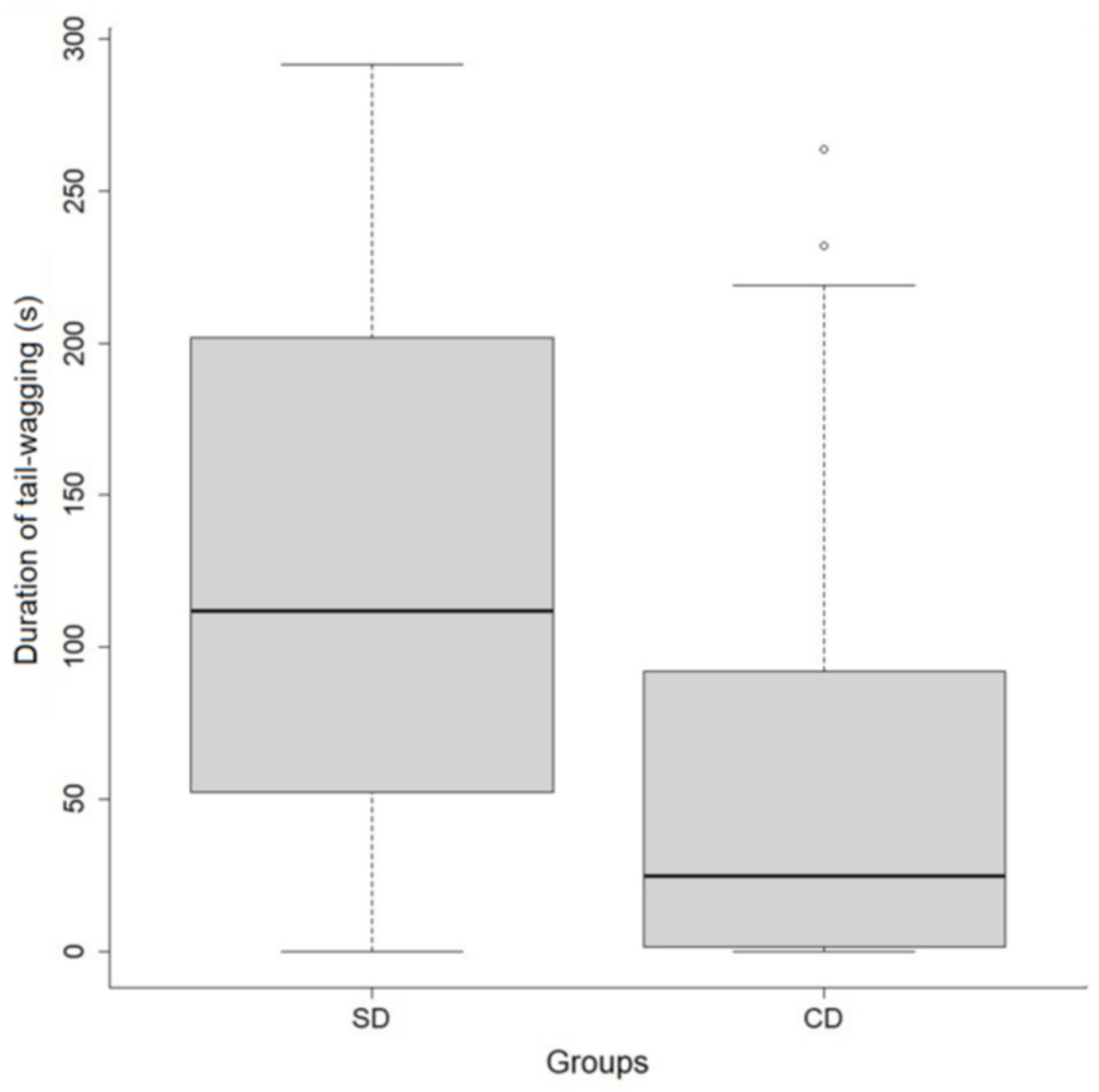
In addition to the differences in performance discussed above, the duration of tail wagging—usually reported as an expression of a positive emotional disposition [
52,
53,
54]—was greater for shelter dogs and had no relation to the identity of the trainer. The novelty—and possibly the value—of training interactions with humans was possibly greater for SDs than for CDs, regardless of with whom the interaction took place. This outcome is in line with a study [
20], in which SDs, during the extinction phase of previously-taught behaviors, spent more time next to the experimenter than CDs did, possibly because they were more willing to interact with humans, or because the human presence somehow buffered the frustrating effect of not being rewarded for a behavior [
51,
55]. In another study, when approached by an unfamiliar experimenter, although SDs displayed more fear-related behaviors towards the experimenter, they stayed close to her for longer periods compared to CDs [
56]. Such results suggest a greater need among SDs for contact with humans [
57].
We found differences in some parameters related to performance between sexes: although females responded to more cues, at a faster speed, and with fewer repetitions, males wagged their tails for longer during the sessions. Males and females have been reported to excel in different tasks—males have shown greater performance in maintaining eye contact with experimenters and in short-term memory tests [
58], whereas females were better at leaving a maze [
59]. The inferior performance of males in our study may be related to their possibly lower trainability, as already reported [
60]. One could also suggest that, as males present more separation-related problems [
61], their separation from the owners during the sessions might have affected them more than it affected females. However, their longer duration of tail wagging suggests that this was not the case. An alternative explanation for this difference could be that males responded to a smaller number of cues because they were less attentive or focused than females, possibly because they were more excited.
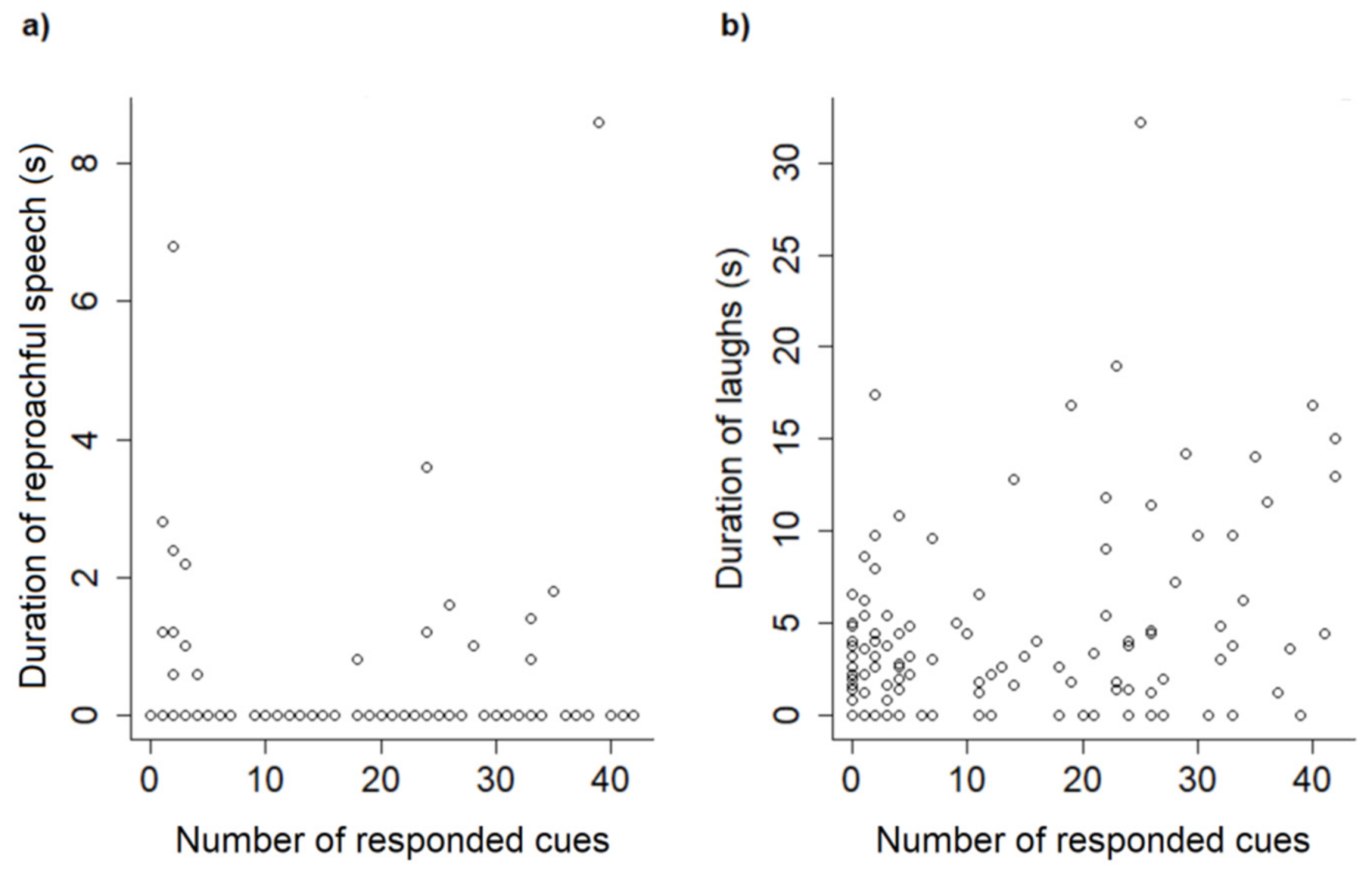
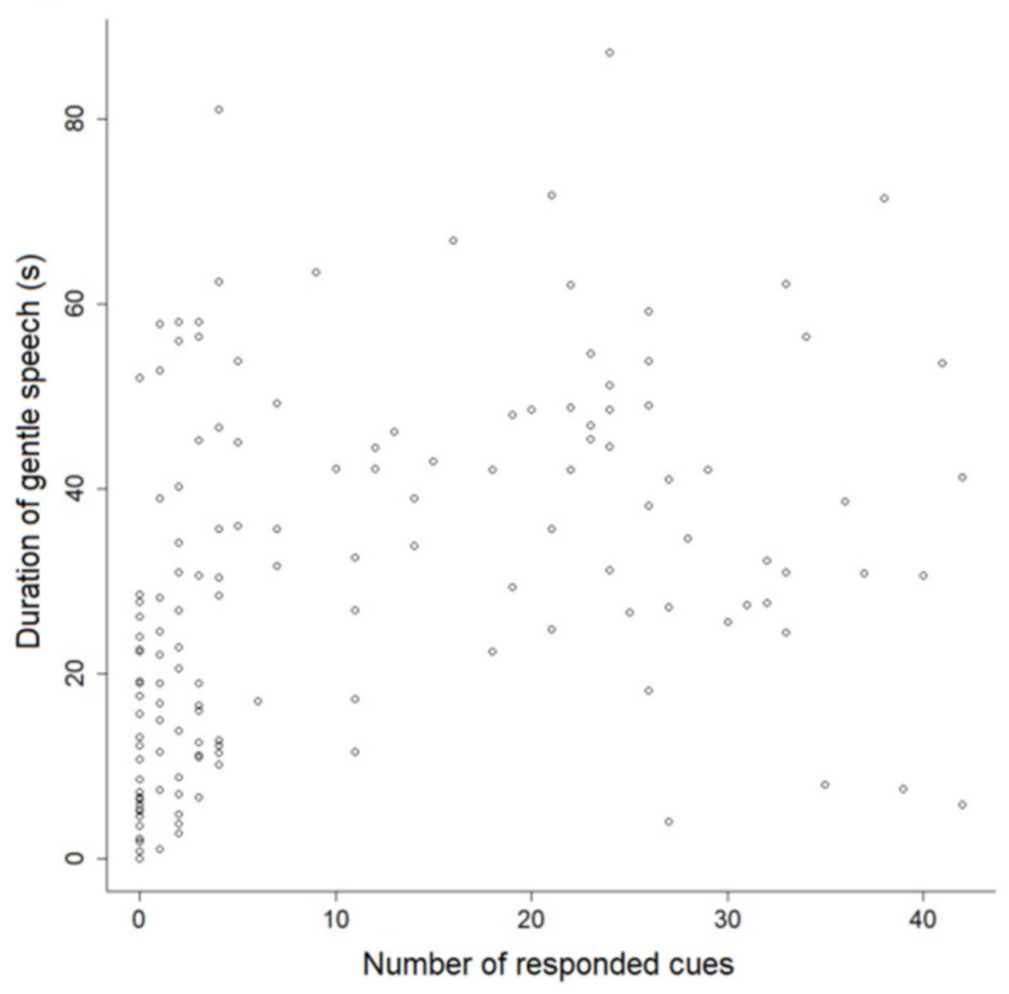
Dog performance was associated with the type of speech used by the trainers during the sessions, as well as their laughing. The use of reproachful speech was connected to performance—shorter latencies and fewer cue repetitions, and a greater number of cues responded to. However, this type of discourse was also correlated with dogs’ general behaviors, especially those indicative of internal states. In sessions when this type of speech was used for longer, dogs wagged their tails less, stayed within 1 m of the trainer for less time, and exhibited more NTB (behaviors that suggested the animals were not focused, or not interested in the interactions). Although most training procedures were based on positive reinforcement (i.e., when the consequence of an individuals’ actions is being rewarded, which has proven to be beneficial for reducing stress [
10]), it is possible that dogs perceived reproachful speech as a punishment [
23] and, although responding to the cues, were emotionally affected by the interaction, as some studies have shown (for reviews, see [
25,
62]). On the other hand, the use of gentle speech was related to a greater number of cues attended to; neutral speech was positively associated with tail wagging, and laughter—although linked to increased latency—was also associated with fewer cue repetitions and more cues attended to. It is possible that these behaviors created a good, less stressful atmosphere for the dogs; therefore, they possibly learnt in a more relaxed manner. An alternative interpretation for these associations was that the trainers behaved more positively (i.e., using neutral or gentle tones of voice) in sessions when the dogs performed better. However, considering the fact that we also recorded reproachful speech used in tandem with good dog performance, and considering studies which have reported the effects of human behavior on their responses (e.g., [
26,
27,
28,
30,
31,
32]), we think this interpretation is improbable. Different emotional responses of dogs to interactions with humans have been demonstrated to be dependent on human behavior/attitude. For example, Horváth and colleagues studied the effects of a 3-min playing session on military dogs with their handlers [
63]. Although the researchers instructed all handlers to play with the dogs, some handlers mostly disciplined their animals during the sessions. These animals showed increases in cortisol levels, suggesting a stress response—the opposite outcome to that observed in dogs with handlers who genuinely played with them.
Our results point to a possible effect that vocal trainer behavior may have on animals during training. Some studies have investigated responses of dogs to some types of speech (e.g., [
30,
32,
64]). Gentle speech, and maybe also laughter, fit within a specific type of speech used with dogs, characterized by a high-pitched voice (high frequency) and affectionate content, known as “dog-directed speech”. Dogs prefer when humans use this type of speech to talk to them, demonstrating this by getting closer and looking at them for longer, and both the patterns of rhythm and sound (prosody) and content of the speech matter [
31,
65]. In our study, when using gentle speech, the trainers praised the dogs, and praise may also function as a type of positive reinforcement [
23]. Here, for the first time—as far as we know—we report an association of this type of speech with dog performance during training.
The duration of the trainers’ neutral speech was surprisingly negatively correlated with the duration of the dogs’ orientation to them. One could interpret this result as an aversive response from dogs to neutral speech. This interpretation is possible, but perhaps not likely, given that the same type of speech was associated with a greater duration of tail wagging. Therefore, an alternative—and possibly more sensible—interpretation would be that this result reflects a response not from dogs, but from trainers, who may have used neutral speech for longer when dogs were not directing their attention to them [
32,
66]. The same rationale can be used to understand the negative correlation between the duration of trainers’ orientation to dogs and the number of cues responded to, and the positive correlation between the time trainers spent oriented to dogs and their latency to respond to them.
We found that the orientation of trainers to dogs was correlated with the dogs’ orientation to them. These results suggest the more focused trainers were on animals, the greater the animals’ attention to them. An interpretation in the opposite direction is also possible (i.e., dog orientation affecting trainer orientation), but the first explanation is in line with studies that found that the trainer’s involvement with the dog during training has an influence on the animal’s attention [
51,
67]. Dogs seem to perceive the human state of attention, responding faster/more often to cues given by people looking at them [
67], and ignoring when the instructor requests a response while looking at someone else instead of the dog. Dogs also prefer to ask for food from people who make eye contact with them than from people who look away [
67], which shows that they evaluate the visual attention they receive, being sensitive to human orientation.
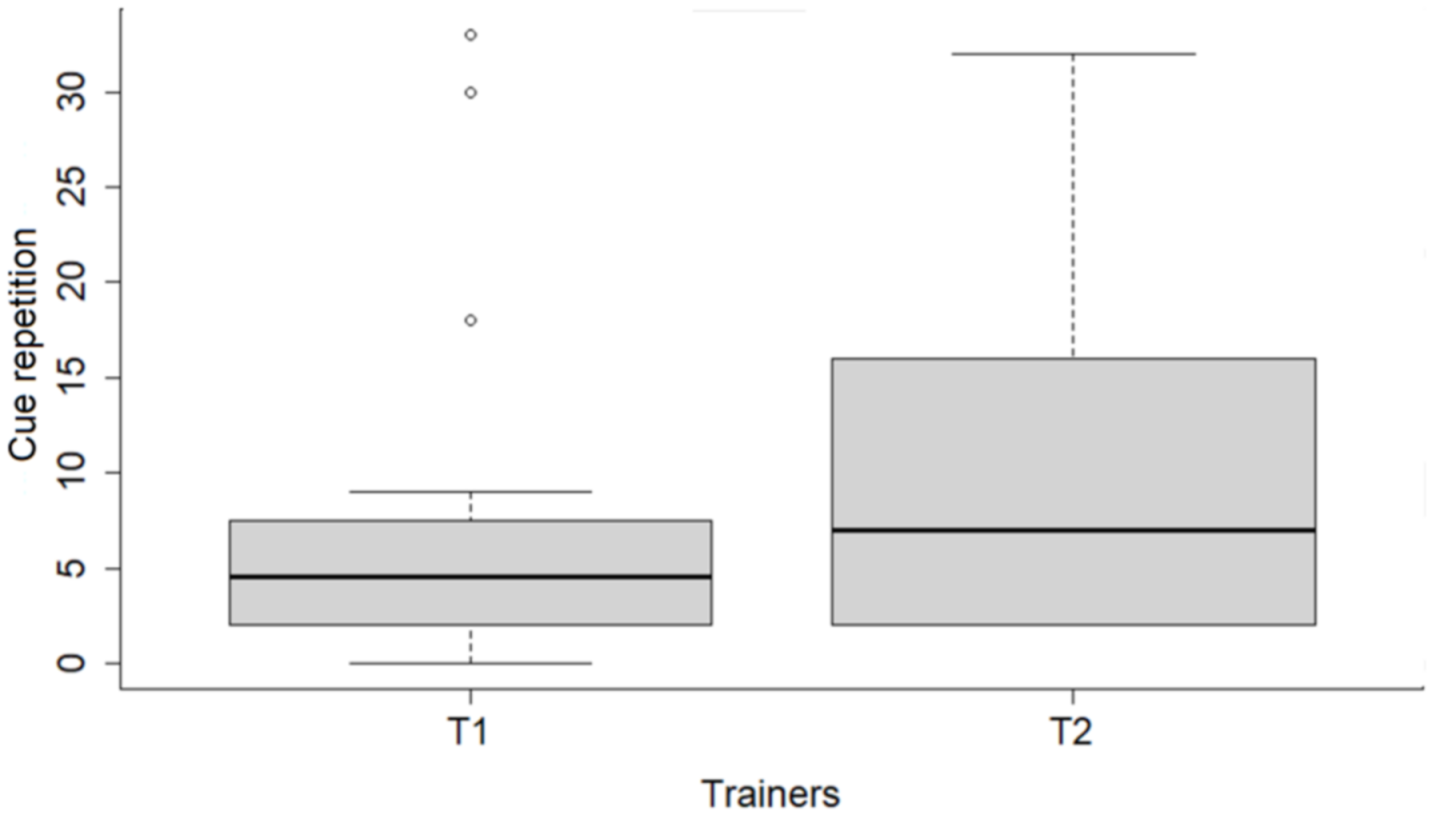
Differences in the use of types of speech, as well as the use of laughter, were detected between trainers. Although the number of sessions each trainer conducted differed, this difference in trainer behavior may have contributed to the greater demand for cue repetitions on the part of dogs in sessions with Trainer 2. One possible explanation for the greater use of reproachful instead of gentle speech by Trainer 2 is a lower level of patience. The level of patience, in tandem with the amount of rewards provided, and trainer involvement in the interactions, has been shown to influence animal learning [
26]. Trainer 1, who used gentle speech and laughter for longer, obtained responses from the dogs with longer latencies, but these did not prevent dogs from responding to more cues per session than when trained by Trainer 2. Considering the potential effects of a pleasant atmosphere for promoting a positive affective state in dogs [
63], our outcomes recommend the use of a soft tone of voice and a relaxed atmosphere in training sessions, with the potential also for improving dogs’ performance.
Our results, although original and relevant, must be interpreted carefully due to some study limitations. Dog breed and age were not balanced in our sample; therefore, we could not investigate the possible effects of these variables on our animals’ responses. Our sample size was limited by the number of shelters that allowed our study to be run in their facilities and in controlled conditions. Nevertheless, small sample sizes are common in dog cognition/learning studies (a review has shown that 57.91% of studies were composed of up to 25 individuals [
68]). Although none of the dogs in this study refused to take part in the training interactions, the absence of the CDs’ owners during the sessions may have had an effect on their learning ability or interest in the sessions [
69]. Finally, as our training sessions were naturalistic, we cannot draw definitive conclusions about the direction of the associations found between dog and trainer behavior. Future studies to investigate these associations further should test dogs’ responses in sessions with standardized trainer behaviors.
Our study highlighted the possible effects of dog origin on their responses during interactions with humans. Dogs were selected during the domestication process to interact and communicate with humans [
4], but a possible side-effect of this selection is a greater ecological and psychological dependence of these animals on human beings. For example, dogs that live inside the house, as family members, show greater dependence on their owners for solving tasks, compared to dogs that live outside [
70]. Shelter dogs traditionally experience a life deprived of human contact, and studies like ours suggest that good-quality interactions with humans may be beneficial for them, improving their quality of life. We suggest that shelters should create training programs for their dogs. Such programs, in addition to having the potential to benefit dog welfare [
10,
71], could increase their chances of being adopted [
72]. An increase in the frequency of interactions with humans has been shown to improve shelter dogs’ chances of being adopted [
73] by up to 1.4 times [
32]. This effect may be attributed to the greater behavioral repertoire developed by these dogs, which is attractive for potential owners. Training programs for shelter dogs may be developed with shelter employees, or even volunteers, at a low or negligible cost.