Measures Matter—Determining the True Nutri-Physiological Value of Feed Ingredients for Swine
Simple Summary
Abstract
1. Introduction
2. The “Disconnect” between Traditional Chemical Measurements and Physiological Responses
2.1. The Role of Water
2.2. Energy Systems
2.2.1. Carbohydrates
2.2.2. Lipids
2.3. Protein and Amino Acids
2.4. Minerals
3. Benefits and Limitations Using In Vitro and Ex Vivo Determinations of the Nutri-Physiological Value of Feed Ingredients
3.1. In Vitro Methods
3.1.1. Near-Infrared Reflectance Spectroscopy (NIRS)
- (1)
- Rapid scanning of the samples (less than 1 min).
- (2)
- Only a small amount of sample is needed for analysis.
- (3)
- Low cost because no chemical reagents are needed and a single operator can analyze a large number of samples in a short period of time.
- (4)
- Results are highly reproducible.
- (5)
- Multiple analytes can be determined in one operation.
- (6)
- Minimal (drying and grinding) or no sample preparation is needed.
- (7)
- Equipment can easily be used in different environments (e.g., ingredient processing, grain harvest, laboratory, feed mill).
- (8)
- High accessibility for online data capture and storage.
- (9)
- Some optical probes allow analyzing samples in situ.
- (10)
- Equipment is portable.
- (1)
- It is a secondary method that requires the use of data derived from chemical analysis or in vivo studies as reference values.
- (2)
- A large number of samples with variable composition and data with large variation is required for accurate and robust calibrations.
- (3)
- Highly trained personnel are required for calibration and validation of the results.
- (4)
- Continuous maintenance and updating of the calibration database is required.
- (5)
- Changes in chemical structure of nutrients that occur during the digestion process cannot be predicted using NIRS technology.
- (6)
- High initial cost for purchasing NIRS instruments.
3.1.2. Closed In Vitro Filtration Methods
3.2. Ex Vivo Methods
3.2.1. Ussing Chambers
| References | Age | Intestinal Segment | Probes (Markers) 1 | Findings | |||||
|---|---|---|---|---|---|---|---|---|---|
| HRP | Man | GlySar | Na-Flu | Na+-Gluc | Na+-Glut | ||||
| [280] | 26 | Jejunum | X | X | X | X | Postweaning feed intake level did not change gut permeability in nursery pigs | ||
| [281] | 28 | Jejunum | X | X | Intestinal molecular permeability was not affected by the age of weaning and creep feeding | ||||
| [282] | 28 | Jejunum | X | Dietary modification from milk- to grain-based sources did not affect HRP fluxes in nursery pigs | |||||
| [283] | 25 | Jejunum | X | X | Intestinal macromolecular permeability was not affected by supplemental dietary tryptophan | ||||
| [284] | 26 | Jejunum | X | X | Intestinal permeability was not different between piglets fed a high-lactulose and low-protein (HL/LP) diet compared with piglets fed control (milk-based) diet | ||||
| [285] | 26 | Jejunum | X | X | Feeding dry pellets elevated transcellular permeability compared with wet feeding | ||||
| Jejunum | X | X | |||||||
| [286,287] | 28 | jejunum | X | X | X | Paracellular permeability was not affected by supplementation of various probiotics (E. farcium and B. cereus var. toyoi) | |||
| [288] | 24 | Ileum, Colon | X | X | Feeding diets containing 2.5% and 5% spray-dried porcine plasma reduced ileal permeability of pigs on day 7 postweaning | ||||
| [289] | 28 | Ileum | X | X | Increasing dietary Zn level from 100 to 2500 ppm at weaning increased intestinal permeability and reduced diarrhea | ||||
| [290] | 28 | Jejunum | X | X | Dietary copper disturbed intestinal-barrier function by increasing transepithelial conductance | ||||
| [291] | 7 | Ileum | X | X | Intestinal permeability increased by 89% in the ileum of piglets deficient in dietary threonine (6.5 g/kg) compared with piglets fed the control diet containing 9.3 g/kg threonine | ||||
| [292] | 14–17 | Jejunum | X | Long-chain (n-3) PUFA supplementation of maternal diets had no effect on total or passive ion transport of their progeny | |||||
| [293] | 15–19 | Jejunum | X | Feeding maternal diets containing long-chain (n−3) PUFA resulted in upregulated glucose flux in piglet jejunum | |||||
3.2.2. Enteroids
4. Nutrient Digestion Kinetics of Feed Ingredients
5. Functional Ingredients and Nutrients
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Saddoris-Clemons, K.; Schneider, J.; Feoli, C.; Cook, D.; Newton, B. Cost-effective feeding strategies for grow-finish pigs. Adv. Pork Prod. 2011, 22, 187–194. [Google Scholar]
- Coffey, D.; Dawson, K.; Ferket, P.; Connolly, A. Review of the feed industry from a historical perspective and implications for its future. J. Appl. Anim. Nutr. 2016, 4, 1–11. [Google Scholar]
- FAO. Livestock’s Long Shadow—Environmental Issues and Options; Food and Agriculture Organization of the United Nations: Rome, Italy, 2006. [Google Scholar]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Mottet, A.; Opio, C.; Dijkman, J.; Falcucci, A.; Tempio, G. Tackling Climate Change through Livestock—A Global Assessment of Emissions and Mitigation Opportunities; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013. [Google Scholar]
- LEAP. Environmental Performance of Animal Feed Supply Chains: Guidelines for Assessment; Livestock Environmental Assessestment and Performance Partnership; Food and Agriculture Organization: Rome, Italy, 2015. [Google Scholar]
- Uwizeye, A.; Gerber, P.J.; Schulte, R.P.O.; de Boer, I.J.M. A comprehensive framework to assess the sustainability of nutrient use in global livestock supply chains. J. Clean. Prod. 2016, 129, 647–658. [Google Scholar] [CrossRef]
- Eriksson, I.S.; Elquist, H.; Stern, S.; Nybrandt, T. Environmental systems analysis of pig production—The impact of feed choice. Int. J. LCA 2005, 10, 143–154. [Google Scholar] [CrossRef]
- Wilfart, A.; Espagnol, S.; Dauguet, S.; Tailleur, A.; Gac, A.; Garcia-Launay, F. ECOALIM: A dataset of environmental impacts of feed ingredients used in French animal production. PLoS ONE 2016, 11, e0167343. [Google Scholar] [CrossRef] [PubMed]
- De Quelen, F.; Brossard, L.; Wilfart, A.; Dourmad, J.-Y.; Garcia-Launay, F. Eco-friendly feed formulation reduces the environmental impacts of pig production without consequences on animal performance. In Proceedings of the 12th International Conference on Life Cycle Assessment of Food 2020 (LCA Food 2020)—Towards Sustainable Agri-Food Systems, Berlin, Germany, 13–16 October 2020. Abstract 321. [Google Scholar]
- Beltran-Alcrudo, D.; Falco, J.R.; Raizman, E.; Dietze, K. Transboundary spread of pig diseases: The role of international trade and travel. BMC Vet. Res. 2019, 15, 54. [Google Scholar] [CrossRef]
- Jones, C.K.; Woodworth, J.; Dritz, S.S.; Paulk, C.B. Reviewing the risk of feed as a vehicle for swine pathogen transmission. Vet. Med. Sci. 2019, 6, 527–534. [Google Scholar] [CrossRef]
- Trudeau, M.P.; Verma, H.; Sampedro, F.; Urriola, P.E.; Shurson, G.C.; Goyal, S.M. Environmental persistence of porcine coronaviruses in feed and feed ingredients. PLoS ONE 2017, 12, e0178094. [Google Scholar] [CrossRef] [PubMed]
- Dee, S.A.; Bauermann, F.V.; Niederwerder, M.C.; Singrey, A.; Clement, T.; de Lima, M.; Long, C.; Patterson, G.; Sheahan, M.A.; Stolan, A.M.M.; et al. Survival of viral pathogens in animal feed ingredients under transboundary shipping models. PLoS ONE 2018, 13, e0194509. [Google Scholar] [CrossRef] [PubMed]
- Patterson, G.; Niederwerder, M.C.; Dee, S.A. Risks to animal health associated with imported feed ingredients. J. Am. Vet. Med. Assoc. 2019, 254, 790–791. [Google Scholar] [CrossRef] [PubMed]
- Shurson, G. Biosecurity of Feed Supply Chains in an Era of Global Animal Disease Epidemics; Technical Bulletin QC2020-03; U.S. Soybean Export Council, Southeast Asia: Chesterfield, MO, USA, 2020; 21p.
- Cochrane, R.A.; Dritz, S.S.; Woodworth, J.C.; Stark, C.R.; Huss, A.R.; Cano, J.P.; Thompson, R.W.; Fahrenholz, A.C.; Jones, C.K. Feed mill biosecurity plans: A systematic approach to prevent biological pathogens in swine feed. J. Swine Health Prod. 2016, 24, 154–164. [Google Scholar]
- Barton, M. Antibiotic use in animal feed and its impact on human health. Nutr. Res. Rev. 2000, 13, 279–299. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Guidelines on Use of Medically Important Antimicrobials in Food-Producing Animals; WHO: Geneva, Switzerland, 2017; 88p, ISBN 9789241550130. [Google Scholar]
- Lillehoj, H.; Liu, Y.; Calsamiglia, S.; Fernandez-Miyakawa, M.E.; Chi, F.; Cravens, R.L.; Oh, S.; Gay, C.G. Phytochemicals as antibiotic alternatives to promote growth and enhance host health. Vet. Res. 2018, 49, 76. [Google Scholar] [CrossRef]
- Liu, Y.; Espinosa, C.D.; Abelilla, J.J.; Casas, G.A.; Lagos, L.V.; Lee, S.A.; Kwon, W.B.; Mathai, J.K.; Navarro, D.M.D.L.; Jaworski, N.W.; et al. Non-antibiotic feed additives in diets for pigs: A review. Anim. Nutr. 2018, 4, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Berckmans, D.; Guarino, M. Precision livestock farming for the global livestock sector. Anim. Front. 2017, 7, 4–5. [Google Scholar] [CrossRef]
- Benhazi, T.M.; Babinszky, L.; Halas, V.; Tscharke, M. Precision livestock farming: Precision feeding technologies and sustainable livestock production. Int. J. Agric. Biol. Eng. 2012, 5, 54–61. [Google Scholar]
- Pomar, C.; van Milgen, J.; Remus, A. Precision livestock feeding, principle and practice. In Poultry and Pig Nutrition; Wageningen Academic Publishers: Wageningen, The Netherlands, 2019; pp. 397–418. ISBN 9789086863334. [Google Scholar] [CrossRef]
- Van Kempen, T.A.T.G.; Simmins, P.H. Near-infrared spectroscopy in precision feed formulation. J. Appl. Poult. Res. 1997, 6, 471–477. [Google Scholar] [CrossRef]
- Pomar, C.; Hauschild, L.; Zhang, G.H.; Pomar, J.; Lovatto, P.A. Applying precision feeding techniques in growing-finishing pig operations. R. Bras. Zootec. 2009, 38, 226–237. [Google Scholar] [CrossRef]
- Henneberg, W.; Stohmann, F. Begründung Einer Rationellen Fütterung der Wiederkäuer; Schwetschtke und Sohn: Braunschweig, Germany, 1860; Volume I. [Google Scholar]
- Henneberg, W.; Stohmann, F. Begründung Einer Rationellen Fütterung der Wiederkäuer; Schwetschtke und Sohn: Braunschweig, Germany, 1864; Volume II. [Google Scholar]
- Johnson, D.E. Contributions of animal nutrition research to nutrition principles: Energetics. J. Nutr. 2007, 137, 698–701. [Google Scholar] [CrossRef][Green Version]
- Noblet, J.; Bontems, V.; Tran, G. Estimation of the energy value of feed for pork. INRA Prod. Anim. 2003, 16, 197–210. [Google Scholar] [CrossRef]
- Stein, H.; Sève, B.M.F.; Fuller, M.F.; Moughan, P.J.; de Lange, C.F.M. Invited review: AA bioavailability and digestibility in pig feed ingredients: Terminology and application. J. Anim. Sci. 2007, 85, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Jondreville, C.; Dourmad, J.-Y. Phosphorus in pig nutrition. INRA Prod. Anim. 2005, 18, 183–192. [Google Scholar] [CrossRef]
- Stein, H.H. Procedures for determining digestibility of amino acids, lipids, starch, fibre, phosphorus, and calcium in feed ingredients fed to pigs. Anim. Prod. Sci. 2017, 57, 2317–2324. [Google Scholar] [CrossRef]
- Zhang, F.; Adeola, O. Techniques for evaluating digestibility of energy, amino acids, phosphorus, and calcium in feed ingredients for pigs. Anim. Nutr. 2017, 3, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Stein, H.H.; Lagos, L.V.; Casas, G.A. Nutritional value of feed ingredients of plant origin fed to pigs. Anim. Feed Sci. Technol. 2016, 218, 33–69. [Google Scholar] [CrossRef]
- Noblet, J.; van Milgen, J. Energy value of pig feeds: Effect of pig body weight and energy evaluation system. J. Anim. Sci. 2004, 82, E229–E238. [Google Scholar]
- Jones, D.P.; Park, Y.; Ziegler, T.R. Nutritional metabolomics: Progress in addressing complexity in diet and health. Annu. Rev. Nutr. 2012, 32, 183–202. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chen, C. Emerging applications of metabolomics in studying chemopreventative phytochemicals. AAPS J. 2013, 15, 941–950. [Google Scholar] [CrossRef]
- Chen, C.; Perez de Nanclares, M.; Kurtz, J.F.; Trudeau, M.; Wang, L.; Yao, D.; Saqui-Salces, M.; Urriola, P.E.; Mydland, L.T.; Shurson, G.C.; et al. Identification of redox imbalance as a prominent metabolic response elicited by rapeseed feeding in the swine metabolome. J. Anim. Sci. 2018, 96, 1757–1768. [Google Scholar] [CrossRef] [PubMed]
- Loor, J.J.; Vailati-Riboni, M.; McCann, J.C.; Zhou, Z.; Bionaz, M. Triennial Lactation Symposium: Nutrigenomics in livestock: Systems biology meets nutrition. J. Anim. Sci. 2015, 93, 5554–5574. [Google Scholar] [CrossRef]
- Jansman, A.J.M.; van Laar, H.; Veldkamp, T.; Gerrits, W.J.J. Masterplan for the Development of Nutrient Based Dynamic Mechanistic Response Models for Pigs and Poultry; Livestock Research Report 765; Wageningen UR Livestock Research: Wageningen, The Netherlands, 2014; pp. 1–43. [Google Scholar]
- Cromwell, G.L.; Calvert, C.C.; Cline, T.R.; Crenshaw, J.D.; Crenshaw, T.D.; Easter, R.A.; Ewan, R.C.; Hamilton, C.R.; Hill, G.M.; Lewis, A.J.; et al. Variability among sources and laboratories in nutrient analyses of corn and soybean meal. NCR-42 Committee on Swine Nutrition. J. Anim. Sci. 1999, 77, 3262–3273. [Google Scholar] [CrossRef]
- Zambrano, M.V.; Dutta, B.; Mercer, D.G.; MacLean, H.L.; Touchie, M.F. Assessment of moisture content measurement methods of dried food products in small-scale operation in developing countries: A review. Trends Food Sci. Technol. 2019, 88, 484–496. [Google Scholar] [CrossRef]
- Beuchat, L.R. Microbial stability as affected by water activity. Cereal Foods World 1981, 26, 345–349. [Google Scholar]
- Fontana, A.J. Understanding the importance of water activity in food. Cereal Foods World 2000, 45, 7–10. [Google Scholar]
- Lowe, J.A.; Kershaw, S.J. Water activity—Moisture content relationship as a predictive indicator for control of spoilage in commercial pet diet components. Anim. Feed Sci. Technol. 1995, 56, 187–194. [Google Scholar] [CrossRef]
- Ghnimi, S.; Budilarto, E.; Kamal-Eldin, A. The new paradigm for lipid oxidation and insights to microencapsulation of omega-3 fatty acids. Comprehen. Rev. Food Sci. Food Saf. 2017, 16, 1206–1218. [Google Scholar] [CrossRef] [PubMed]
- Eichner, K.; Karel, M. Influence of water content and water activity on the sugar-amino browning reaction in model systems under various conditions. J. Agric. Food Chem. 1972, 20, 218–223. [Google Scholar] [CrossRef]
- Hemmler, D.; Roullier-Gall, C.; Marshall, J.W.; Rychlik, M.; Taylor, A.J.; Schmitt-Kopplin, P. Insights into the chemistry of non-enzymatic browning reactions in different ribose-AA model systems. Sci. Rep. 2018, 8, 16879. [Google Scholar] [CrossRef]
- Monsan, P.; Combes, D. Effect of water activity on enzyme action and stability. Enz. Eng. 1984, 434, 48–60. [Google Scholar] [CrossRef]
- Anthonsen, T.; Sjursnes, B.J. Importance of water activity for enzymatic catalysts in non-aqueous organic systems. In Methods and Tools in Biosciences and Medicine; Methods in Non-Aqueous Enzymology; Gupta, M.N., Ed.; Birkhäuser Verlag: Basel, Switzerland, 2000; pp. 14–35. [Google Scholar]
- Arya, S.S.; Thakur, B.R. Effect of water activity on vitamin A degradation in wheat flour (atta). J. Food Proc. Preserv. 1990, 14, 123–134. [Google Scholar] [CrossRef]
- Laing, B.M.; Schlueter, D.L.; Labuza, T.P. Degradation kinetics of ascorbic acid at high temperature and water activity. J. Food Sci. 2006, 43, 1440–1443. [Google Scholar] [CrossRef]
- Beuchat, L.R. Influence of water activity on growth, metabolic activities and survival of yeasts and molds. J. Food Protect. 1983, 46, 135–141. [Google Scholar] [CrossRef]
- Hemmingsen, A.K.T.; Stevik, A.M.; Claussen, I.C.; Lundblad, K.K.; Prestløkken, E.; Sørensen, M.; Eikevik, T.M. Water adsorption in feed ingredients for animal pellets at different temperatures, particle size, and ingredient combinations. Dry. Technol. 2008, 26, 738–748. [Google Scholar] [CrossRef]
- FAO; IFIF. Good Practices for the Feed Sector—Implementing the Codex Alimentarius Code of Practice on Good Animal Feeding; FAO Animal Production and Health Manual No. 24; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Velayudhan, D.E.; Kim, I.H.; Nyachoti, C.M. Invited Review: Characterization of dietary energy in swine feed and feed ingredients: A review of recent research results. Asian Australas. J. Anim. Sci. 2015, 28, 1–13. [Google Scholar] [CrossRef]
- Wenk, C.; Colombani, P.C.; van Milgen, J.; Lemme, A. Glossary: Terminology in animal and human energy metabolism. In Energy Metabolism in Animals; No. 103; Chwalibog, A., Jakobsen, K., Eds.; EAAP Publication: Snekkersten, Denmark; Wageningen Press: Wageningen, The Netherlands, 2000; pp. 409–421. [Google Scholar]
- Le Goff, G.; Noblet, J. Comparative total tract digestibility of dietary energy and nutrients in growing pigs and adult sows. J. Anim. Sci. 2001, 79, 2418–2427. [Google Scholar] [CrossRef]
- Birkett, S.; de Lange, K. A computational frame for a nutrient flow representation of energy utilization by growing monogastric animals. Br. J. Nutr. 2001, 86, 661–674. [Google Scholar] [CrossRef]
- Velayudhan, D.E.; Nyachoti, C.M. Validation of net energy system of feed formulation in growing pigs fed barley based diets with alternative feed ingredients. J. Anim. Sci. 2014, 92 (Suppl. S2), 645. [Google Scholar]
- Noblet, J.; Perez, J.M. Prediction of digestibility of nutrients and energy values of pig diets from chemical analysis. J. Anim. Sci. 1993, 71, 3389–3398. [Google Scholar] [CrossRef]
- Noblet, J.; Fortune, H.; Shi, X.S.; Dubois, S. Prediction of net energy value of feeds for growing pigs. J. Anim. Sci. 1994, 72, 344–354. [Google Scholar] [CrossRef] [PubMed]
- NRC. Nutrient Requirements of Swine, 11th ed.; National Academy Press: Washington, DC, USA, 2012.
- Adeola, O. Digestion and balance techniques in pigs. In Swine Nutrition, 2nd ed.; Lewis, A.J., Southern, L.L., Eds.; CRC Press: Washington, DC, USA, 2001; pp. 903–916. [Google Scholar]
- Van Milgen, J.; Noblet, J. Partitioning of energy intake to heat, protein, and fat in growing pigs. J. Anim. Sci. 2003, 81 (Suppl. S2), E86–E93. [Google Scholar]
- Kil, D.Y.; Kim, B.G.; Stein, H.H. Invited review: Feed energy evaluation for growing pigs. Asian Australas. J. Anim. Sci. 2013, 26, 1205–1217. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, H.; Li, Y.; Lv, Z.; Liu, L.; Lai, C.; Wang, J.; Wang, F.; Li, D.; Zhang, S. Methodologies on estimating the energy requirements for maintenance and determining the net energy content of feed ingredients in swine: A review of recent work. J. Anim. Sci. Biotechnol. 2018, 9, 39. [Google Scholar] [CrossRef]
- Rijnen, M.M.J.A.; Doorenbos, J.; Mallo, J.; den Hartog, L.A. The application of the net energy systems for swine. In Proceedings of the 25th Western Nutrition Conference, Saskatoon, SK, Canada, 28–30 September 2004. [Google Scholar]
- Boisen, S.; Verstegen, M.W.A. Evaluation of feedstuffs and pig diets. Energy or nutrient-based evaluation systems. Acta Agric. Scand. Sect. A Anim. Sci. 1998, 48, 86–94. [Google Scholar]
- Noblet, J. Digestive and metabolic utilization of feed energy in swine: Application to energy evaluation systems. J. Appl. Anim. Res. 2000, 17, 113–132. [Google Scholar] [CrossRef]
- Van Milgen, J.; Noblet, J.; Dubois, S. Energetic efficiency of starch, protein and lipid utilization in growing pigs. J. Nutr. 2001, 131, 1309–1318. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, Z.; Zhang, S.; Page, G.; Jaworski, N.W. The role of lactose in weanling pig nutrition: A literature and meta-analysis review. J. Anim. Sci. Biotechnol. 2021, 12, 10. [Google Scholar]
- O’Doherty, J.V.; Dillon, S.; Figat, S.; Callan, J.J.; Sweeney, T. The effects of lactose inclusion and seaweed extract derived from Laminaria spp. on performance, digestibility of diet components and microbial populations in newly weaned pigs. Anim. Feed Sci. Technol. 2010, 157, 173–180. [Google Scholar] [CrossRef]
- Mahan, D.C. Efficacy of dried whey and its lactalbumin and lactose components at two dietary lysine levels on post weaning pig performance and nitrogen balance. J. Anim. Sci. 1992, 70, 2182–2187. [Google Scholar] [CrossRef] [PubMed]
- Charalampopoulos, D.; Wang, R.; Pandiella, S.S.; Webb, C. Application of cereals and cereal components in functional foods: A review. Int. J. Food Microbiol. 2002, 79, 131–141. [Google Scholar] [CrossRef]
- Bach Knudsen, K.E. Lactose in diet influences the degradation of mixed linked β (1–3; 1–4)-D-glucan in the small intestine of pigs. J. Anim. Sci. 2012, 90, 125–127. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sánchez-Muros, M.-J.; Barroso, F.G.; Manzano-Agugliaro, F. Insect meal as renewable source of food for animal feeding: A review. J. Clean. Prod. 2014, 65, 16–27. [Google Scholar] [CrossRef]
- Mohan, K.; Ganesan, A.R.; Muralisankar, T.; Jayakumar, R.; Sathishkumar, P.; Uthayakumar, V.; Chandirasekar, R.; Revathi, N. Recent insights into the extraction, characterization, and bioactivities of chitin and chitosan from insects. Trends Food Sci. Technol. 2020, 105, 17–42. [Google Scholar] [CrossRef] [PubMed]
- Bach Knudsen, K.E.; Lærke, H.N.; Jorgensen, H. Carbohydrates and carbohydrate utilization in swine. In Sustainable Swine Nutrition; Chiba, L.I., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; pp. 109–137. [Google Scholar]
- Navarro, D.M.D.L.; Abelilla, J.J.; Stein, H.H. Structures and characteristics of carbohydrates in diets fed to pigs: A review. J. Anim. Sci. Biotechnol. 2019, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Woyengo, T.A.; Beltranena, E.; Zijlstra, R.T. Nonruminant Nutrition Symposium: Controlling feed cost by including alternative ingredients into pig diets: A review. J. Anim. Sci. 2014, 92, 1293–1305. [Google Scholar] [CrossRef] [PubMed]
- Jha, R.; Berrocoso, J.D. Review: Dietary fiber utilization and its effects on physiological functions and gut health of swine. Animal 2015, 9, 1441–1452. [Google Scholar] [CrossRef] [PubMed]
- Agyekum, A.K.; Nyachoti, C.M. Nutritional and metabolic consequences of feeding high-fiber diets to swine: A review. Engineering 2017, 3, 716–725. [Google Scholar] [CrossRef]
- Swiatkiewicz, S.; Swiatkiewicz, M.; Arczewska-Wlosek, A.; Jozefiak, D. Efficacy of feed enzymes in pig and poultry diets containing distillers dried grains with solubles: A review. J. Anim. Physiol. Anim. Nutr. 2015, 100, 15–26. [Google Scholar] [CrossRef]
- Torres-Pitarch, A.; Manzanilla, E.G.; Gardiner, G.E.; O’Doherty, J.V. Systematic review and meta-analysis of the effect of feed enzymes on growth and nutrient digestibility in grow-finisher pigs: Effect of enzyme type and cereal source. Anim. Feed Sci. Technol. 2019, 251, 153–165. [Google Scholar] [CrossRef]
- Zhao, J.; Bai, Y.; Zhang, G.; Liu, L.; Lai, C. Relationship between dietary fiber fermentation and volatile fatty acids’ concentration in growing pigs. Animals 2020, 10, 263. [Google Scholar] [CrossRef]
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Le Sciellour, M.; Labussière, E.; Zemb, O.; Renaudeau, D. Effect of dietary fiber content on nutrient digestibility and fecal microbiota composition in growing-finishing pigs. PLoS ONE 2018, 13, e0206159. [Google Scholar]
- Tran, T.H.T.; Everaert, N.; Bindlle, J. Review on the effects of potential prebiotics on controlling intestinal enteropathogens Salmonella and Escherichia coli in pig production. J. Anim. Physiol. Anim. Nutr. 2018, 102, 17–32. [Google Scholar] [CrossRef]
- Jha, R.; Fouhse, J.M.; Tiwari, U.P.; Li, L.; Willing, B. Dietary fiber and intestinal health of monogastric animals. Front. Vet. Sci. 2019, 6, 48. [Google Scholar] [CrossRef]
- Van Soest, P.J.; Wine, R.H. Use of detergents in the analysis of fibrous feeds. IV. Determination of plant cell wall consituents. J. Assoc. Off. Anal. Chem. 1967, 50, 50–55. [Google Scholar]
- Mertens, D.R. Challenges in measuring insoluble dietary fiber. J. Anim. Sci. 2003, 81, 3233–3249. [Google Scholar] [CrossRef]
- Bach Knudsen, K.E. The nutritional significance of dietary fibre analysis. Anim. Feed Sci. Technol. 2001, 90, 3–20. [Google Scholar] [CrossRef]
- McRorie, J.W.; Fahey, G.C. Fiber supplements and clinically meaningful health benefits: Identifying the physiochemical characteristics of fiber that drive specific physiologic effects. In Dietary Supplements in Health Promotion; Wallace, T.C., Ed.; CRC Press: Boca Raton, FL, USA, 2015; pp. 161–206. [Google Scholar]
- Bach Knudsen, K.E. Triennial Growth Symposium: Effects of polymeric carbohydrates on growth and development in pigs. J. Anim. Sci. 2011, 89, 1965–1980. [Google Scholar] [CrossRef]
- Hung, Y.-T.; Zhu, J.; Shurson, G.C.; Saqui-Salces, M.; Urriola, P.E. Influence of viscosity and fiber on nutrient digestibility and intestinal physiology in growing pigs. J. Anim. Sci. 2020, 98 (Suppl. S3), 67–68. [Google Scholar] [CrossRef]
- Kaczmarek, J.L.; Thompson, S.V.; Holscher, H.D. Complex interactions of circadian rhythms, eating behaviors, and the gastrointestinal microbiota and their potential on health. Nutr. Rev. 2017, 75, 673–682. [Google Scholar] [CrossRef] [PubMed]
- Sinturel, F.; Gerber, A.; Mauvoisin, D.; Wang, J.; Gatfield, D.; Stubblefield, J.J.; Green, C.B.; Gachon, F.; Schibler, U. Diurnal oscillations in liver mass and cell size accompany assembly cycles. Cell 2017, 169, 651–663.e14. [Google Scholar] [CrossRef] [PubMed]
- Parkar, S.G.; Kalsbeek, A.; Cheeseman, J.F. Host circadian rhythms and metabolic health. Microorganisms 2019, 7, 41. [Google Scholar] [CrossRef]
- Uhr, G.T.; Dohnalová, L.; Thaiss, C.A. The dimension of time in host-microbiome interactions. mSystems 2019, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hopwood, D.E.; Pluske, J.R.; Hampson, D.J. Dietary manipulation of infectious bowel disease, Chapter 12. Biol. Grow. Anim. 2006, 4, 365–385. [Google Scholar]
- Hopwood, D.E.; Pethick, D.W.; Pluske, J.R.; Hampson, D.J. Addition of pearl barley to a rice-based diet for newly weaned piglets increases the viscosity of the intestinal contents, reduces starch digestibility and exacerbates post-weaning colibacillosis. Br. J. Nutr. 2004, 92, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Molist, F.; van Oostrum, M.; Pérez, J.F.; Mateos, G.G.; Nyachoti, C.M.; van der Aar, P.J. Relevance of functional properties of dietary fibre in diets for weanling pigs. Anim. Feed Sci. Technol. 2014, 189, 1–10. [Google Scholar] [CrossRef]
- Kim, B.G.; Kil, D.Y.; Zhang, Y.; Stein, H.H. Concentrations of analyzed or reactive lysine, but not crude protein, may predict the concentration of digestible lysine in distillers dried grains with soluble fed to pigs. J. Anim. Sci. 2012, 90, 3798–3808. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.M.; Opapeju, F.O.; Pluske, J.R.; Kim, J.C.; Hampson, D.J.; Nyachoti, C.M. Gastrointestinal health and function in weaned pigs: A review of feeding strategies to control post-weaning diarrhoea without using in-feed antimicrobial compounds. J. Anim. Physiol. Anim. Nutr. 2013, 97, 2017–2237. [Google Scholar] [CrossRef]
- Saqui-Salces, M.; Huang, Z.; Ferrandis Vila, M.; Li, J.; Mielke, J.A.; Urriola, P.E.; Shurson, G.C. Modulation of intestinal cell differentiation in growing pigs is dependent on the fiber source in the diet. J. Anim. Sci. 2017, 95, 1179–1190. [Google Scholar] [CrossRef]
- Williams, B.A.; Verstegen, M.W.A.; Tamminga, S. Fermentation in the large intestine of single-stomached animals and its relationship to animal health. Nutr. Res. Rev. 2001, 14, 207–227. [Google Scholar] [CrossRef]
- Jarrett, S.; Ashworth, C.J. The role of dietary fibre in pig production, with a particular emphasis on reproduction. J. Anim. Sci. Biotechnol. 2018, 9, 59. [Google Scholar] [CrossRef]
- Souza da Silva, C.; Bolhuis, J.E.; Gerrits, W.J.J.; Kemp, B.; van den Borne, J.J.G.C. Effects of dietary fibers with different fermentation characteristics on feeding motivation in adult female pigs. Physiol. Behav. 2013, 110-111, 148–157. [Google Scholar] [CrossRef]
- Ferguson, E.M.; Slevin, J.; Hunter, M.G.; Edwards, S.A.; Ashworth, C.J. Beneficial effects of a high fibre diet on oocyte maturity and embryo survival in gilts. Reproduction 2007, 133, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Weaver, A.C.; Kelly, J.M.; Kind, K.L.; Gatford, K.L.; Kennaway, D.J.; Herde, P.J.; van Wettre, W.H.E.J. Oocyte maturation and embryo survival in nulliparous female pigs (gilts) is improved by feeding a lupin-based high fibre diet. Reprod. Fertil. Dev. 2013, 25, 1216–1223. [Google Scholar] [CrossRef]
- Van der Peet-Schwering, C.M.C.; Kemp, B.; Binnendijk, G.P.; den Hartog, L.A.; Spoolder, H.A.M.; Verstegen, M.W.A. Performance of sows fed high levels of nonstarch polysaccharides during gestation and lactation over three parities. J. Anim. Sci. 2003, 81, 2247–2258. [Google Scholar] [CrossRef] [PubMed]
- Veum, T.L.; Crenshaw, J.D.; Crenshaw, T.D.; Cromwell, G.L.; Easter, R.A.; Ewan, R.C.; Nelssen, J.L.; Miller, E.R.; Pettigrew, J.E.; Ellersieck, M.R.; et al. The addition of ground wheat straw as a fiber source in the gestation diet of sows and the effect on sow and litter performance for three successive parities. J. Anim. Sci. 2009, 87, 1003–1012. [Google Scholar] [CrossRef]
- Peltoniemi, O.A.T.; Tast, A.; Heinonen, M.; Oravainen, J.; Munsterhjelm, C.; Hälli, O.; Oliviero, C.; Hämeenoja, P.; Virolainen, J.V. Fertility of sows fed ad libitum with a high fibre diet during pregnancy. Reprod. Domest. Anim. 2010, 45, 1008–1014. [Google Scholar] [CrossRef] [PubMed]
- Fairbairn, S.L.; Patience, J.F.; Classen, H.L.; Zijlstra, R.T. The energy content of barley fed to growing pigs: Characterizing the nature of its variability and developing prediction equations for its estimation. J. Anim. Sci. 1999, 77, 1502–1512. [Google Scholar] [CrossRef]
- Li, Q.; Zang, J.; Liu, D.; Piao, X.; Lai, C.; Li, D. Predicting corn digestible and metabolizable energy content from its chemical composition in growing pigs. J. Anim. Sci. Biotechnol. 2014, 5, 11. [Google Scholar] [CrossRef]
- Kerr, B.J.; Dozier, W.A., III; Shurson, G.C. Effects of reduced-oil corn distillers dried grains with solubles composition on digestible and metabolizable energy value and prediction in growing pigs. J. Anim. Sci. 2013, 91, 3231–3243. [Google Scholar] [CrossRef] [PubMed]
- Son, A.R.; Park, C.S.; Kim, B.G. Determination and prediction of digestible and metabolizable energy concentrations in byproduct feed ingredients fed to growing pigs. Asian Australas. J. Anim. Sci. 2017, 30, 546–553. [Google Scholar] [CrossRef]
- Palmquist, D.L.; Jenkins, T.C. Challenges with fats and fatty acid methods. J. Anim. Sci. 2003, 81, 3250–3254. [Google Scholar] [CrossRef] [PubMed]
- Kerr, B.J.; Kellner, T.A.; Shurson, G.C. Characteristics of lipids and their feeding value in swine diets. J. Anim. Sci. Biotechnol. 2015, 6, 30. [Google Scholar] [CrossRef] [PubMed]
- Luthria, D.L. (Ed.) Oil Extraction and Analysis: Critical Issues and Comparative Studies; AOCS Press: Champaign, IL, USA, 2004. [Google Scholar]
- Moller, J. Cereals, cereal-based products and animal feeding stuffs—Determination of crude fat and total fat content by the Randall extraction method: A collaborative study. In Quality Assurance and Safety Crops and Foods; Wiley: Hoboken, NJ, USA, 2010; pp. 1–6. [Google Scholar]
- Kim, B.G.; Kil, D.Y.; Stein, H.H. In growing pigs, the true ileal and total tract digestibility of acid hydrolyzed ether extract in extracted corn oil is greater than in intact sources of corn oil or soybean oil. J. Anim. Sci. 2013, 91, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.J.; Chwalibog, A.; Tauson, A.H.; Sawosz, E. Influence of different fibre sources on digestibility and nitrogen retention and energy balances in growing pigs. Arch. Anim. Nutr. 2006, 60, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Kil, D.Y.; Sauber, T.E.; Jones, D.B.; Stein, H.H. Effect of the form of dietary fat and the concentration of dietary neutral detergent fiber on ileal and total tract endogenous losses and apparent and true digestibility of fat by growing pigs. J. Anim. Sci. 2010, 88, 2959–2967. [Google Scholar] [CrossRef]
- Cera, K.R.; Mahan, D.C.; Reinhart, G.A. Evaluation of various extracted vegetable oils, roasted soybeans, medium-chain triglyceride and an animal-vegetable fat blend for postweaning swine. J. Anim. Sci. 1990, 68, 2756–2765. [Google Scholar] [CrossRef] [PubMed]
- Cera, K.R.; Mahan, D.C.; Reinhart, G.A. Apparent fat digestibilities and performance responses of postweaning swine fed diets supplemented with coconut oil, corn oil or tallow. J. Anim. Sci. 1989, 67, 2040–2047. [Google Scholar] [CrossRef]
- Cera, K.R.; Mahan, D.C.; Reinhart, G.A. Weekly digestibilities of diets supplemented with corn oil, lard or tallow by weanling swine. J. Anim. Sci. 1988, 66, 1430–1437. [Google Scholar] [CrossRef]
- Powles, J.; Wiseman, J.; Cole, D.J.A.; Jagger, S. Prediction of the apparent digestible energy value of fats given to pigs. Anim. Sci. 1995, 61, 149–154. [Google Scholar] [CrossRef]
- Powles, J.; Wiseman, J.; Cole, D.J.A.; Hardy, B. Effect of chemical structure of fats upon their apparent digestible energy value when given to young pigs. Anim. Prod. 1994, 58, 411–417. [Google Scholar] [CrossRef]
- Jorgensen, H.; Fernandez, J.A. Chemical composition and energy value of different fat sources for growing pigs. Acta Agric. Scand. Sect. A Anim. Sci. 2000, 50, 129–136. [Google Scholar] [CrossRef]
- Mendoza, S.M.; van Heugten, E. Effects of dietary lipid sources on performance and apparent total tract digestibility of lipids and energy when fed to nursery pigs. J. Anim. Sci. 2014, 92, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Rosero, D.S.; Odle, J.; Arellano, C.; Boyd, R.D.; van Heugten, E. Development of prediction equations to estimate the apparent digestibility energy content of lipids when fed to lactating sows. J. Anim. Sci. 2015, 93, 1165–1176. [Google Scholar] [CrossRef] [PubMed]
- Renner, R.; Hill, F.W. Factors affecting the absorbability of saturated fatty acids in the chick. J. Nutr. 1961, 74, 254–258. [Google Scholar] [CrossRef]
- Brockerhoff, H. Stereospecific analysis of triglycerides. Lipids 1971, 4, 942–956. [Google Scholar] [CrossRef]
- Kil, D.Y.; Ji, F.; Stewart, L.L.; Hinson, R.B.; Beaulieu, A.D.; Allee, G.L.; Patience, J.F.; Pettigrew, J.E.; Stein, H.H. Net energy of soybean oil and choice white grease in diets fed to growing and finishing pigs. J. Anim. Sci. 2011, 89, 448–459. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Chen, D.; Zhang, K.; Yu, B. Effects of oxidative stress on growth performance, nutrient digestibilities and activities of antioxidant enzymes of weanling pigs. Asian Australas. J. Anim. Sci. 2007, 20, 1600–1605. [Google Scholar] [CrossRef]
- Liu, P.; Chen, C.; Kerr, B.J.; Weber, T.E.; Johnston, L.J.; Shurson, G.C. Influence of thermally-oxidized vegetable oils and animal fats on energy and nutrient digestibility in young pigs. J. Anim. Sci. 2014, 92, 2971–2979. [Google Scholar] [CrossRef]
- David, R.O.; Sánchez-Muniz, F.J.; Bastida, S.; Benedi, J.; González-Muñoz, M.J. Gastric emptying and short-term digestibility of thermally oxidized sunflower oil used for frying in fasted and nonfasted rats. J. Agric. Food Chem. 2010, 58, 9242–9248. [Google Scholar] [CrossRef] [PubMed]
- Van Heugten, E.; Odle, J.; Boyd, R.D. Feeding value of fat for swine—Concepts and practice. In Proceedings of the Milk Specialties Global Conference, Pre-Conference to the Minnesota Nutrition Conference, Prior Lake, MN, USA, 16–17 September 2015. [Google Scholar]
- Kellner, T.A.; Patience, J.F. The digestible energy, metabolizable energy, and net energy content of dietary fat sources in thirteen- and fifty-kilogram pigs. J. Anim. Sci. 2017, 95, 3984–3995. [Google Scholar] [CrossRef]
- Su, Y.; She, Y.; Huang, Q.; Shi, C.; Li, Z.; Huang, C.; Piao, X.; Li, D. The effect of inclusion level of soybean oil and palm oil on their digestible and metabolizable energy content determined with the difference and regression method when fed to growing pigs. Asian Australas. J. Anim. Sci. 2015, 28, 1751–1759. [Google Scholar] [CrossRef][Green Version]
- Su, Y.; Bi, X.; Huang, Q.; Liu, L.; Piao, X.; Li, D. The effect of inclusion level and basal diet on the determination of the digestible and metabolisable energy content of soybean oil and its digestibility when fed to growing pigs. Anim. Prod. Sci. 2016, 56, 1167–1173. [Google Scholar] [CrossRef]
- Kerr, B.J.; Curry, S.M.; Lindblom, S.C. Digestibility of energy and caloric value in nursery pigs fed commercially available lipids. Appl. Anim. Sci. 2018, 35, 291–297. [Google Scholar] [CrossRef]
- Kerr, B.J.; Weber, T.E.; Dozier, W.A., III; Kidd, M.T. Digestible and metabolizable energy content of crude glycerin originating from different sources in nursery pigs. J. Anim. Sci. 2009, 87, 4042–4049. [Google Scholar] [CrossRef]
- Kerr, B.J.; Shurson, G.C. Determination of ether extract digestibility and energy content of specialty lipids with different fatty acid and free fatty acid content and the effect of lecithin, for nursery pigs. Prof. Anim. Sci. 2016, 33, 127–134. [Google Scholar] [CrossRef]
- Sauvant, D.; Perez, J.-M.; Tran, G. Tables of Composition and Nutritional Value of Feed Materials, 2nd ed.; Wageningen Academy Publishers: Wageningen, The Netherlands, 2004. [Google Scholar]
- Rostagno, H.S. (Ed.) Brazilian Tables for Poultry and Swine—Composition of Feedstuffs and Nutritional Requirements, 4th ed.; Universidade Federal de Viçosa: Viçosa, Brasil, 2017. [Google Scholar]
- Li, E.; Lv, Z.; Liu, H.; Liu, L.; Li, Y.; Li, Z.; Wang, F.; Li, D.; Zhang, S. Determination of net energy content of soybean oil fed to growing pigs using indirect calorimetry. Anim. Sci. J. 2018, 89, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Kerr, B.J.; Dozier, W.A., III; Shurson, G.C. Lipid digestibility and energy content of distillers’ corn oil in swine and poultry. J. Anim. Sci. 2016, 94, 2900–2908. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, J.; Powles, J.; Salvador, F. Comparison between pigs and poultry in the prediction of the dietary energy value of fats. Anim. Feed Sci. Technol. 1998, 71, 1–9. [Google Scholar] [CrossRef]
- Lindblom, S.C.; Gabler, N.K.; Kerr, B.J. Influence of feeding thermally peroxidized soybean oil on growth performance, digestibility, and gut integrity in growing pigs. J. Anim. Sci. 2018, 96, 558–569. [Google Scholar] [CrossRef]
- Song, R.; Shurson, G.C. Evaluation of lipid oxidation level in corn dried distillers grains with solubles. J. Anim. Sci. 2013, 91, 4383–4388. [Google Scholar] [CrossRef] [PubMed]
- Shurson, G.C.; Kerr, B.J.; Hanson, A.R. Evaluating the quality of feed fats and oils and their effects on pig growth performance. J. Anim. Sci. Biotechnol. 2015, 6, 10. [Google Scholar] [CrossRef]
- Spiteller, P.; Kern, W.; Reiner, J.; Spiteller, G. Aldehydic lipid peroxidation products derived from linoleic acid. Biochim. Biophys. Acta 2001, 1531, 188–208. [Google Scholar] [CrossRef]
- Seppanen, C.M.; Csallany, A.S. Formation of 4-hydroxynonenal, a toxic aldehyde, in soybean oil at frying temperature. J. Am. Oil Chem. Soc. 2002, 79, 1033–1038. [Google Scholar] [CrossRef]
- Belitz, H.D.; Grosch, W.; Schieberle, P. (Eds.) Lipids. In Food Chemistry; Springer: Berlin, Germany, 2009; pp. 158–247. [Google Scholar]
- Rocha, C.; Maiorka, A.; Valle, F.L.P.; Schramm, V.G.; Angeli, A.L.; Silva, A.V.F. The effect of soybean oil quality and vitamin E supplementation on turkey diet nutrition. J. Appl. Poult. Res. 2012, 21, 318–324. [Google Scholar] [CrossRef]
- Hung, Y.T.; Hanson, A.R.; Shurson, G.C.; Urriola, P.E. Peroxidized lipids reduce growth performance of poultry and swine: A meta-analysis. Anim. Feed Sci. Technol. 2017, 231, 47–58. [Google Scholar] [CrossRef]
- Boler, D.D.; Fernandez-Duenas, D.M.; Kutzler, L.W.; Zhao, J.; Harrell, R.L.; Campion, D.R.; Killefer, J.; McKeith, F.K.; Dilger, A.C. Effects of oxidaized corn oil and a synthetic antioxidant blend on performance, oxidative status of tissues, and fresh meat quality in finishing barrows. J. Anim. Sci. 2012, 90, 5159–5169. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Kerr, B.J.; Weber, T.E.; Chen, C.; Johnston, L.J.; Shurson, G.C. Effect of lipid source and oxidation level on metabolic oxidation status of young pigs. J. Anim. Sci. 2012, 90 (Suppl. S2), 75. [Google Scholar]
- Tahashaki, K.; Akiba, Y. Effect of oxidized fat on performance and some physiological responses in broiler chickens. Jpn. Poult. Sci. 1999, 36, 304–310. [Google Scholar] [CrossRef]
- Anjum, M.I.; Mirza, I.H.; Khan, A.G.; Azim, A. Effect of fresh versus oxidized soybean oil on growth performance, organ weights, and meat quality of broiler chicks. Pak. Vet. J. 2004, 24, 173–178. [Google Scholar]
- Van Heugten, E.; Rosero, D.S.; Chang, P.L.Y.C.; Zier-Rush, C.; Boyd, R.D. Peroxidized lipids in nursery pig diets—Why and when should we be concerned? In Proceedings of the Midwest Swine Nutrition Conference, Indianapolis, IN, USA, 9 September 2016; pp. 31–39. [Google Scholar]
- Dibner, J.J.; Atwell, C.A.; Kitchell, M.L.; Shermer, W.D.; Ivey, F.J. Feeding oxidized fats to broilers and swine: Effects on enterocyte turnover, hepatocyte proliferation and the gut associated lymphoid tissue. Anim. Feed Sci. Technol. 1996, 62, 1–13. [Google Scholar] [CrossRef]
- Liang, F.; Jiang, S.; Mo, Y.; Zhou, G.; Yang, L. Consumption of oxidized soybean oil increased intestinal oxidative stress and affected intestinal immune variables in yellow-feathered broilers. Asian Australas. J. Anim. Sci. 2015, 28, 1194–1201. [Google Scholar] [CrossRef]
- Evonik. Amino Dat 4.0® The AA Composition of Feedstuffs, 5th ed.; Degussa AG, Feed Additives: Hanau, Germany, 2010. [Google Scholar]
- Olukosi, O.A.; Adebiyi, A.O. Chemical composition and prediction of AA content of maize- and wheat-distillers’ dried grains with solubles. Anim. Feed Sci. Technol. 2013, 185, 182–189. [Google Scholar] [CrossRef]
- Cromwell, G.L.; Cline, T.R.; Crenshaw, J.D.; Crenshaw, T.D.; Easter, R.A.; Ewan, R.C.; Hamilton, C.R.; Hill, G.M.; Lewis, A.J.; Mahan, D.C.; et al. Variability among sources and laboratories in analyses of wheat middlings. NCR-42 Committee on Swine Nutrition. J. Anim. Sci. 2000, 78, 2652–2658. [Google Scholar] [CrossRef]
- Messad, F.; Létourneau-Montminy, M.P.; Charbonneau, E.; Sauvant, D.; Guay, F. Prediction of standardized ileal digestibility and essential AA content of ingredients in swine: A meta-analysis. Anim. Feed Sci. Technol. 2015, 207, 204–221. [Google Scholar] [CrossRef]
- Wu, G. Dietary requirements of synthesizable Amino acids by animals: A paradigm shift in protein nutrition. J. Anim. Sci. Biotechnol. 2014, 5, 34. [Google Scholar] [CrossRef]
- Kong, C.; Adeola, O. Invited review—Evaluation of AA and energy utilization in feedstuff for swine and poultry diets. Asian Australas. J. Anim. Sci. 2014, 27, 917–925. [Google Scholar] [CrossRef]
- Stein, H.H.; Kim, S.W.; Nielsen, T.T.; Easter, R.A. Standardized ileal protein and AA digestibility by growing pigs and sows. J. Anim. Sci. 2001, 79, 2113–2122. [Google Scholar] [CrossRef] [PubMed]
- Stein, H.H.; Pedersen, C.; Wirt, A.R.; Bohke, R.A. Additivity of values for apparent and standardized ileal digestibility of amino acids in mixed diets fed to growing pigs. J. Anim. Sci. 2005, 83, 2387–2395. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.A.; Jo, H.; Kong, C.; Kim, B.G. Use of digestible rather than total AA in diet formulation increases nitrogen retention and reduces nitrogen excretion from pigs. Livest. Sci. 2017, 197, 8–11. [Google Scholar] [CrossRef]
- Maillard, L.C. Synthéses des matiéres humiques par action des acides aminés sur les sucres réducteurs. Ann. Chim. 1916, 6, 258–317. [Google Scholar]
- Maillard, L.C. Action des acides aminés sur les sucres: Formation des mélanoidines per voie méthodologique. Comptes R. Acad. Sci. 1912, 154, 66–68. [Google Scholar]
- Maga, J.A. Measurement of available lysine using the guanidination reaction. J. Food Sci. 1981, 46, 132–134. [Google Scholar] [CrossRef]
- Hurrell, R.F.; Carpenter, K.J. The estimation of available lysine in foodstuffs after Maillard reactions. Prog. Food Sci. 1981, 5, 159–176. [Google Scholar]
- Finot, P.A. The absorption and metabolism of modified Amino acids in processed foods. J. AOAC Int. 2005, 88, 894–903. [Google Scholar] [CrossRef]
- Fontaine, J.; Zimmer, U.; Moughan, P.J.; Rutherford, S.M. Effect of heat damage in an autoclave on the reactive lysine contents of soy products and corn distillers dried grains with solubles. Use of the results to check lysine damage in common qualities of these ingredients. J. Agric. Food Chem. 2007, 55, 10737–10743. [Google Scholar] [CrossRef] [PubMed]
- Pahm, A.A.; Pedersen, C.; Stein, H.H. Application of the reactive lysine procedure to estimate lysine digestibility in distillers dried grains with soluble fed to growing pigs. J. Agric. Food Chem. 2008, 56, 9441–9446. [Google Scholar] [CrossRef] [PubMed]
- Rutherfurd, S.M. Use of the guanidination reaction for determining reactive lysine, bioavailable lysine and gut endogenous lysine. Amino Acids 2015, 47, 1805–1815. [Google Scholar] [CrossRef]
- Almeida, F.N.; Htoo, J.K.; Thomson, J.; Stein, H.H. AA digestibility of heat damaged distillers dried grains with solubles fed to pigs. J. Anim. Sci. Biotechnol. 2013, 4, 44. [Google Scholar] [CrossRef]
- Cromwell, G.L.; Herkelman, K.L.; Stahly, T.S. Physical, chemical, and nutritional characteristics of distillers dried grains with solubles for chicks and pigs. J. Anim. Sci. 1993, 71, 679–686. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fastinger, N.D.; Mahan, D.C. Determination of the ileal AA and energy digestibilities of corn distillers dried grains with solubles using grower-finisher pigs. J. Anim. Sci. 2006, 84, 1722–1728. [Google Scholar] [CrossRef]
- Urriola, P.E.; Johnston, L.J.; Stein, H.H.; Shurson, G.C. Prediction of the concentration of standardized ileal digestible Amino acids in distillers dried grains with solubles. J. Anim. Sci. 2013, 91, 4389–4396. [Google Scholar] [CrossRef][Green Version]
- American Oil Chemists Society. Official Methods and Recommended Practices, 5th ed.; AOCS: Urbana, IL, USA, 2000. [Google Scholar]
- Batal, A.B.; Douglas, M.W.; Engram, A.E.; Parsons, C.M. Protein dispersibility index as an indicator of adequately processed soybean meal. Poult. Sci. 2000, 79, 1592–1596. [Google Scholar] [CrossRef] [PubMed]
- Araba, M.; Dale, N.M. Evaluation of protein solubility as an indicator of overprocessing of soybean meal. Poult. Sci. 1990, 69, 76–83. [Google Scholar] [CrossRef]
- Araba, M.; Dale, N.M. Evaluation of protein solubility as an indicator of underprocessing of soybean meal. Poult. Sci. 1990, 69, 1749–1752. [Google Scholar] [CrossRef]
- Anderson-Hafermann, J.C.; Zhang, Y.; Parsons, C.M. Effect of heating on nutritional quality of conventional and Kunitz trypsin inhibitor-free soybeans. Poult. Sci. 1992, 71, 1700–1709. [Google Scholar] [CrossRef] [PubMed]
- Hsu, J.T.; Satter, L.D. Procedures for measuring the quality of heat-treated soybeans. J. Dairy Sci. 1995, 78, 1353–1361. [Google Scholar] [CrossRef]
- De Coca-Sinova, A.; Valencia, D.G.; Jiménez-Moreno, E.; Lázaro, R.; Mateos, G.G. Apparent ileal digestibility of energy, nitrogen, and Amino acids of soybean meals of different origin in broilers. Poult. Sci. 2008, 87, 2613–2623. [Google Scholar] [CrossRef] [PubMed]
- van Barneveld, R.J.; Graham, H.; Diffey, S. Predicting the nutritional quality of feed ingredients for pigs using near-infrared spectroscopy (NIRS) and chemical analysis. Anim. Prod. Sci. 2018, 58, 709–718. [Google Scholar] [CrossRef]
- Zeng, Z.K.; Shurson, G.C.; Urriola, P.E. Prediction of the concentration of standardized ileal digestible Amino acids and safety margins among sources of distillers dried grains with solubles for growing pigs: A meta-analysis approach. Anim. Feed Sci. Technol. 2017, 231, 150–159. [Google Scholar] [CrossRef]
- Święch, E. Alternative prediction methods of protein and energy evaluation of pig feeds. J. Anim. Sci. Biotech. 2017, 8, 39. [Google Scholar] [CrossRef]
- Kar, S.K.; Jansman, A.J.M.; Boeren, S.; Kruijt, L.; Smits, M.A. Protein, peptide, AA composition, and potential functional properties of existing and novel dietary protein sources for monogastrics. J. Anim. Sci. 2016, 94, 30–39. [Google Scholar] [CrossRef]
- Celi, P.; Gabai, G. Oxidant/antioxidant balance in animal nutrition and health: The role of protein oxidation. Front. Vet. Sci. 2015, 2, 48. [Google Scholar] [CrossRef]
- Gebicki, J.M. Protein hydroperoxides as new reactive oxygen species. Redox. Rep. 1997, 3, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Korhonen, H.; Pihlanto-Leppälä, A.; Rantamäki, P.; Tupasela, T. Impact of processing on bioactive proteins and peptides. Trends Food Sci. Technol. 1998, 9, 307–319. [Google Scholar] [CrossRef]
- Hua, Y.; Cui, S.W.; Wang, Q.; Mine, Y.; Poysa, V. Heat induced gelling properties of soy protein isolates prepared from different defatted soybean flours. Food Res. Int. 2005, 38, 377–385. [Google Scholar] [CrossRef]
- Tang, X.; Wu, Q.; Le, G.; Shi, Y. Effects of heat treatment on structural modification and in vivo antioxidant capacity of soy protein. Nutrition 2012, 28, 1180–1185. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.W.; Chen, X.; Yang, X.; Leng, Z.X.; Yan, P.S.; Zhou, Y.M. Effects of heat treatment of soy protein isolate on the growth performance and immune function of broiler chickens. Poult. Sci. 2014, 93, 326–334. [Google Scholar] [CrossRef]
- Zhang, X.; Lu, P.; Xue, W.; Wu, D.; Wen, C.; Zhou, Y. An evaluation of heat on protein oxidation of soy protein isolate or soy protein isolate mixed with soybean oil in vitro and its consequences on redox status of broilers at early age. Asian Australas. J. Anim. Sci. 2017, 30, 1135–1142. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Xue, W.Y.; Zhang, X.L.; Wu, D.W.; Ding, L.R.; Wen, C.; Zhou, Y.M. Heat-induced protein oxidation of soybean meal impairs growth performance and antioxidant status of broilers. Poult. Sci. 2019, 98, 276–286. [Google Scholar] [CrossRef]
- Frame, C.A.; Huff-Lonergan, E.J.; Serao, M.C.R. Impact of storage conditions on protein oxidation of rendered by-product meals. Transl. Anim. Sci. 2020, 4, 1–9. [Google Scholar] [CrossRef]
- Frame, C.A.; Johnson, E.; Kilburn, L.; Huff-Lonergan, E.; Kerr, B.J.; Serao, M.R. Impact of dietary oxidized protein on oxidative status and performance in growing pigs. J. Anim. Sci. 2020, 98, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Crenshaw, T.D. Calcium, phosphorus, vitamin D, and vitamin K. In Swine Nutrition, 2nd ed.; Lewis, A.J., Southern, L.L., Eds.; CRC Press: Washington, DC, USA, 2001; pp. 187–212. [Google Scholar]
- Cromwell, G.L. The biological availability of phosphorus in feedstuffs for pigs. Pig News Inf. 1992, 13, 75N–78N. [Google Scholar]
- NRC. Nutrient Requirements of Swine, 10th ed.; National Academies Press: Washington, DC, USA, 1998.
- Baker, D.H.; Stein, H.H. Bioavailability of minerals and vitamins in feedstuffs. In Sustainable Swine Nutrition; Chiba, L.I., Ed.; Wiley-Blackwell: Oxford, UK, 2013; pp. 341–364. [Google Scholar]
- Shen, Y.; Fan, M.Z.; Ajakiye, A.; Archibold, T. Use of the regression analysis technique to determine the true digestible phosphorus digestibility and the endogenous phosphorus output associated with corn in growing pigs. J. Nutr. 2002, 132, 1199–1206. [Google Scholar] [CrossRef]
- She, Y.; Wang, Q.; Stein, H.H.; Liu, L.; Li, D.; Zhang, S. Additivity of values for phosphorus digestibility in corn, soybean meal, and canola meal in diets fed to growing pigs. Asian Australas. J. Anim. Sci. 2018, 31, 1301–1307. [Google Scholar] [CrossRef]
- Gonçalves, M.A.D.; Dritz, S.S.; Tokach, M.D.; DeRouchey, J.M.; Woodworth, J.C.; Goodband, R.D. Fact sheet—Ingredient database management for swine: Phosphorus. J. Swine Health Prod. 2017, 25, 76–78. [Google Scholar]
- Adeola, O.; Cowieson, A.J. Board-invited review: Opportunities and challenges in using exogenous enzymes to improve nonruminant animal production. J. Anim. Sci. 2011, 89, 3189–3218. [Google Scholar] [CrossRef]
- Dersjant-Li, Y.; Awati, A.; Schulze, H.; Partridge, G. Phytase in non-ruminant animal nutrition: A critical review on phytase activities in the gastrointestinal tract and influencing factors. J. Sci. Food Agric. 2014, 95, 878–896. [Google Scholar] [CrossRef]
- Humer, E.; Schwarz, C.; Schedle, K. Phytate in pig and poultry nutrition. J. Anim. Physiol. Anim. Nutr. 2015, 99, 605–625. [Google Scholar] [CrossRef] [PubMed]
- Cowieson, A.J.; Ruckebusch, J.P.; Knap, I.; Guggenbuhl, P.; Fru-Nji, F. Phytate-free nutrition: A new paradigm in monogastric animal production. Anim. Feed Sci. Technol. 2016, 222, 180–189. [Google Scholar] [CrossRef]
- Rosenfelder-Kuon, P.; Siegert, W.; Rodehutscord, M. Effect of microbial phytase supplementation on P digestibility in pigs: A meta-analysis. Arch. Anim. Nutr. 2020, 74, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Moughan, P.J. In vitro techniques for the assessment of the nutritive value of feed grains for pigs: A review. Aust. J. Agric. Res. 1999, 50, 871–880. [Google Scholar] [CrossRef]
- Rollin, B.E. Toxicology and new social ethics for animals. Toxicol. Pathol. 2003, 31, 128–131. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.W.; Vavak, D.L.; Satterlee, L.; Miller, G.A. A multienzyme technique for estimating protein digestibility. J. Food Sci. 1977, 42, 1269–1273. [Google Scholar]
- Pedersen, B.; Eggum, B.O. Prediction of protein digestibility by an in vitro enzymatic pH-stat procedure. Z. Tierphysiol. Tierernahr. Futtermittelkd. 1983, 49, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Wolzak, A.; Bressani, R.; Gomez, B.R. A comparison of in vivo and in vitro estimates of protein digestibility of native and thermally processed vegetable proteins. Plant Foods Hum. Nutr. 1981, 31, 31–43. [Google Scholar] [CrossRef]
- Fontaine, J.; Hörr, J.; Schirmer, B. Near-infrared reflectance spectroscopy enables the fast and accurate prediction of the essential AA contents in soy, rapeseed meal, sunflower meal, peas, fishmeal, meat meal products, and poultry meal. J. Agric. Food Chem. 2001, 49, 57–66. [Google Scholar] [CrossRef]
- Fontaine, J.; Hörr, J.; Schirmer, B. AA contents in raw materials can be precisely analyzed in a global network of near-infrared spectrometers: Collaborative trials prove the positive effects of instrument standardization and repeatability files. J. Agric. Food Chem. 2004, 52, 701–708. [Google Scholar] [CrossRef]
- Jiang, H.Y.; Zhu, Y.J.; Wei, L.M.; Dai, J.R.; Song, T.M.; Yan, Y.L.; Chen, S.J. Analysis of protein, starch and oil content of single intact kernels by near infrared reflectance spectroscopy (NIRS) in maize (Zea mays L.). Plant Breed. 2007, 126, 492–497. [Google Scholar] [CrossRef]
- Yang, Z.; Han, L.; Fernández Pierna, J.A.; Dardenne, P.; Baeten, V. Review—The potential of near infrared microscopy to detect, identify; quantify processed animal by-products. J. NIRS 2011, 19, 211–231. [Google Scholar]
- Tena, N.; Boix, A.; von Holst, C. Identification of botanical and geographical origin of distillers dried grains with solubles by near infrared microscopy. Food Control 2015, 54, 103–110. [Google Scholar] [CrossRef]
- Fontaine, J.; Schirmer, B.; Hörr, J. Near-infrared reflectance spectroscopy (NIRS) enables the fast and accurate prediction of essential AA contents. 2. Results for wheat, barley, corn, triticale, wheat bran/middlings, rice bran, and sorghum. J. Agric. Food Chem. 2002, 50, 3902–3911. [Google Scholar] [CrossRef]
- Zhou, L.J.; Zhang, L.Y.; Zhang, E.X.; Li, J.T.; Yang, W.J.; Wang, Z.Y. Rapid determination of swine available energy and Amino acids in corn distillers dried grains with solubles by near-infrared reflectance spectroscopy. Anim. Feed Sci. Technol. 2012, 175, 198–202. [Google Scholar] [CrossRef]
- Van Barneveld, R.J. Understanding the nutritional chemistry of lupin (Lupinus spp.) seed to improve livestock production efficiency. Nutr. Res. Rev. 1999, 12, 203–230. [Google Scholar] [CrossRef]
- McCann, M.E.E.; McCracken, K.J.; Agnew, R.E. The use of near infrared reflectance spectroscopy (NIRS) for prediction of the nutritive value of barley for growing pigs. Irish J. Agric. Food Res. 2006, 45, 187–195. [Google Scholar]
- Zijlstra, R.T.; Swift, M.L.; Wang, L.F.; Scott, T.A.; Edney, M.J. Near infrared reflectance spectroscopy accurately predicts the digestible energy content of barley for pigs. Can. J. Anim. Sci. 2011, 91, 301–304. [Google Scholar] [CrossRef]
- Li, J.; Li, Q.; Li, D.; Chen, Y.; Wang, X.; Yang, W.; Zhang, L. Use of near-infrared reflectance spectroscopy for the rapid determination of the digestible energy and metabolizable energy content of corn fed to growing pigs. J. Anim. Sci. Biotechnol. 2016, 7, 45. [Google Scholar]
- Cozannet, P.; Primot, Y.; Gady, C.; Métayer, J.P.; Lessire, M.; Skiba, F. Energy value of wheat distillers grains with solubles for growing pigs and adult sows. J. Anim. Sci. 2010, 88, 2382–2392. [Google Scholar] [CrossRef] [PubMed]
- Jha, R.; Tiwari, U.P. Rapid technique for feed evaluation: Scope and limitations. In Proceedings of the NZ Poultry Industry Conference, Wellington, New Zealand, 4–5 October 2016; Volume 13, pp. 84–102. [Google Scholar]
- Sheffner, A.L.; Eckfeldt, G.A.; Spector, H. The pepsin-digest-residue (PDR) AA index of net protein utilization. J. Nutr. 1956, 60, 105–120. [Google Scholar] [CrossRef]
- Maga, J.A.; Lorenz, K.; Onayemi, O. Digestive acceptability of proteins as measured by the intestinal rate of in vitro proteolysis. J. Food Sci. 1973, 38, 173–174. [Google Scholar] [CrossRef]
- Buchanan, R.A. In vivo and in vitro methods of measuring nutritive value of leaf-protein preparations. Br. J. Nutr. 1969, 23, 533–545. [Google Scholar] [CrossRef][Green Version]
- Saunders, R.M.; Kohler, G.O. In vitro determination of protein digestibility in wheat mill feeds for monogastric animals. Cereal Chem. 1972, 49, 98–103. [Google Scholar]
- Akeson, W.R.; Stahmann, M.A. A pepsin pancreatin digest index of protein quality evaluation. J. Nutr. 1964, 83, 257–261. [Google Scholar] [CrossRef]
- Büchmann, N.B. In vitro digestibility of protein from barley and other cereals. J. Sci. Food Agric. 1979, 30, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Dierick, N.; Vervaeke, I.; Decuypere, J.; Henderickx, H. Protein digestion in pigs measured in vivo and in vitro. In Digestive Physiology in the Pigs; Just, A., Jørgensen, H., Fernández, J.A., Eds.; National Institute of Animal Science: Copenhagen, Denmark, 1985; pp. 329–332. [Google Scholar]
- Saunders, R.M.; Connor, M.A.; Booth, A.N.; Bickoff, E.M.; Kohler, G.O. Measurement of digestibility of alfalfa protein concentrates by in vivo and in vitro methods. J. Nutr. 1973, 103, 530–535. [Google Scholar] [CrossRef]
- Furuya, S.; Sakamoto, K.; Takahashi, S. A new in vitro method for the estimation of digestibility using the intestinal fluid of the pig. Br. J. Nutr. 1979, 41, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Sauer, W.C.; He, J.; Hwangbo, J.; Wang, X. The nutritive value of hulled and hulless barley for growing pigs. 1. Determination of energy and protein digestibility with the in vivo and in vitro method. J. Anim. Feed Sci. 2003, 12, 759–769. [Google Scholar] [CrossRef]
- Huang, G.; Sauer, W.C.; He, J.; Ramirez, M. The nutritive value of hulled and hulless barley for growing pigs. 2. Determination of in vivo and in vitro energy and in vivo ileal AA digestibility. J. Anim. Feed Sci. 2003, 12, 771–784. [Google Scholar] [CrossRef]
- Regmi, P.R.; Sauer, W.C.; Zijstra, R.T. Prediction of in vivo apparent total tract energy digestibility of barley in grower pigs using an in vitro digestibility technique. J. Anim. Sci. 2008, 86, 2619–2626. [Google Scholar] [CrossRef] [PubMed]
- Boisen, S.; Fernández, J.A. Prediction of the total tract digestibility of energy in feedstuffs and pig diets by in vitro analyses. Anim. Feed Sci. Technol. 1997, 68, 277–286. [Google Scholar] [CrossRef]
- Regmi, P.R.; Ferguson, N.S.; Zijstra, R.T. In vitro digestibility techniques to predict apparent total tract energy digestibility of wheat in grower pigs. J. Anim. Sci. 2009, 87, 3620–3629. [Google Scholar] [CrossRef]
- Vervaeke, I.J.; Dierick, N.A.; Demeyer, D.I.; Decuypere, J.A. Approach to the energetic importance of fibre digestion in pigs. II. An experimental approach to hindgut digestion. Anim. Feed Sci. Technol. 1989, 23, 169–194. [Google Scholar] [CrossRef]
- Boisen, S.; Fernández, J.A. Prediction of the apparent ileal digestibility of protein and Amino acids in feedstuffs and feed mixtures for pigs by in vitro analyses. Anim. Feed Sci. Technol. 1995, 51, 29–43. [Google Scholar] [CrossRef]
- Slominski, B.A.; Simbaya, J.; Campbell, L.D.; Rakow, G.; Guenter, W. Nutritive value for broilers of meals derived from newly developed varieties of yellow-seeded canola. Anim. Feed Sci. Technol. 1999, 78, 249–262. [Google Scholar] [CrossRef]
- Maenz, D.D.; Engele-Schaan, C.M.; Newkirk, R.W.; Classen, H.L. The effect of minerals and mineral chelators on the formation of phytase-resistant and phytase-susceptible forms of phytic acid in solution and in a slurry of canola meal. Anim. Feed Sci. Technol. 1999, 81, 177–192. [Google Scholar] [CrossRef]
- Zyla, K.; Ledoux, D.R.; Garcia, A.; Veum, T.L. An in vitro procedure for studying enzymic dephosphorylation of phytate in maize-soybean feeds for turkey poults. Br. J. Nutr. 1995, 74, 3–17. [Google Scholar] [CrossRef]
- Zyla, K.; Koreleski, J. In-vitro and in-vivo dephosphorylation of rapeseed meal by means of phytate-degrading enzymes derived from Aspergillus niger. J. Sci. Food Agric. 1993, 61, 1–6. [Google Scholar] [CrossRef]
- Zhu, X.S.; Seib, P.A.; Allee, G.L.; Liang, Y.T. Preparation of a low-phytate feed mixture and bioavailability of its phosphorus to chicks. Anim. Feed Sci. Technol. 1990, 27, 341–351. [Google Scholar] [CrossRef]
- Ajakaiye, A.; Fan, M.Z.; Archbold, T.; Hacker, R.R.; Forsberg, C.W.; Phillips, J.P. Determination of true digestive utilization of phosphorus and the endogenous phosphorus outputs associated with soybean meal for growing pigs. J. Anim. Sci. 2003, 81, 2766–2775. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Kerr, B.J.; Shurson, G.C.; Urriola, P.E. In vitro prediction of standardized total tract digestibility of phosphorus among sources of animal protein meals. J. Anim. Sci. 2018, 96 (Suppl. S2), 117–118.–118. [Google Scholar] [CrossRef]
- Bauer, E.; Williams, B.A.; Bosch, M.W.; Voigt, C.; Mosenthin, R.; Verstegen, M.W.A. Differences in microbial activity of digesta from three sections of the porcine large intestine according to in vitro fermentation of carbohydrate-rich substrates. J. Sci. Food Agric. 2004, 84, 2097–2104. [Google Scholar] [CrossRef]
- Bindelle, J.; Buldgen, A.; Boudry, C.; Leterme, P. Effect of inoculum and pepsin-pancreatin hydrolysis on fibre fermentation measured by the gas production technique in pigs. Anim. Feed Sci. Technol. 2007, 132, 111–122. [Google Scholar] [CrossRef]
- Williams, B.A.; Bosch, M.W.; Boer, H.; Verstegen, M.W.A.; Tamminga, S. An in vitro batch culture method to assess potential fermentability of feed ingredients for monogastric diets. Anim. Feed Sci. Technol. 2005, 123, 445–462. [Google Scholar] [CrossRef]
- Christensen, D.N.; Bach Knudsen, K.E.; Wolstrup, J.; Jensen, B.B. Integration of ileum cannulated pigs and in vitro fermentation to quantify the effect of diet composition on the amount of short-chain fatty acids available from fermentation in the large intestine. J. Sci. Food Agric. 1999, 79, 755–762. [Google Scholar] [CrossRef]
- Bauer, E.; Williams, B.A.; Bosch, M.W.; Voigt, C.; Mosenthin, R.; Verstegen, M.W.A. Microbial activities of faeces from unweaned and adult pigs, in relation to selected fermentable carbohydrates. Anim. Sci. 2001, 73, 313–322. [Google Scholar] [CrossRef]
- Pastorelli, G.; Faustini, M.; Attard, E. In vitro fermentation of feed ingredients by fresh or frozen pig fecal inocula. Anim. Sci. J. 2014, 85, 690–697. [Google Scholar] [CrossRef]
- Jang, J.-C.; Zeng, Z.; Shurson, G.C.; Urriola, P.E. Effects of gas production recording system and pig fecal inoculum volume on kinetics and variation of in vitro fermentation using corn distiller’s dried grains with solubles and soybean hulls. Animals 2019, 9, 773. [Google Scholar] [CrossRef]
- Tao, S.; Bai, Y.; Zhou, X.; Zhao, J.; Yang, H.; Zhang, S.; Wang, J. In vitro fermentation characteristics for different ratios of soluble to insoluble dietary fiber by fresh fecal microbiota from growing pigs. ACS Omega 2019, 4, 15158–15167. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Urriola, P.E.; Shurson, G.C. Prediction of digestible and metabolizable energy of corn distillers dried grains with solubles for growing pigs using in vitro digestible nutrients. J. Anim. Sci. 2018, 96, 1818–1824. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Urriola, P.E.; Shurson, G.C. Use of in vitro dry matter digestibility and gas production to predict apparent total tract digestibility and total dietary fiber for growing pigs. J. Anim. Sci. 2017, 95, 5474–5484. [Google Scholar] [CrossRef]
- Huang, Z.; Urriola, P.E.; Salfer, I.J.; Stern, M.D.; Shurson, G.C. Differences in in vitro hydrolysis and fermentation among and within high-fiber ingredients using a modified three-step procedure for growing pigs. J. Anim. Sci. 2017, 95, 5497–5506. [Google Scholar] [CrossRef]
- Ussing, H.H.; Zerahn, K. Active transport of sodium as the source of electric current in the short-circuited isolated frog skin. Acta. Physiol. Scand. 1951, 23, 110–127. [Google Scholar] [CrossRef]
- Smith, P.L. Methods for Evaluating Intestinal Permeability and Metabolism in Vitro. In Models for Assessing Drug Absorption and Metabolism; Pharmaceutical Biotechnology Series; Borchardt, R.T., Smith, P.L., Wilson, G., Eds.; Springer: Boston, MA, USA, 1996; Volume 8, pp. 13–34. [Google Scholar]
- Holtug, K.; Hansen, M.B.; Skadhauge, E. Experimental studies of intestinal ion and water transport. Scand. J. Gastroenterol. Suppl. 1996, 216, 95–110. [Google Scholar] [CrossRef]
- Wijtten, P.J.; van der Meulen, J.; Verstegen, M.W. Intestinal barrier function and absorption in pigs after weaning: A review. Br. J. Nutr. 2011, 105, 967–981. [Google Scholar] [CrossRef] [PubMed]
- Boudry, G. The Ussing chamber technique to evaluate alternatives to in-feed antibiotics for young pigs. Anim. Res. 2005, 54, 219–230. [Google Scholar] [CrossRef]
- Haslam, I.S.; O’Reilly, D.A.; Sherlock, D.J.; Kauser, A.; Womack, C.; Coleman, T. Pancreatoduodenectomy as a source of human small intestine for Ussing chamber investigations and comparative studies with rat tissue. Biopharm. Drug Dispos. 2011, 32, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Rozehnal, V.; Nakai, D.; Hoepner, U.; Fischer, T.; Kamiyama, E.; Takahasi, M.; Yasuda, S.; Mueller, J. Human small intestinal and colonic tissue mounted in the Ussing chamber as a tool for characterizing the intestinal absorption of drugs. Eur. J. Pharm. Sci. 2012, 46, 367–373. [Google Scholar] [CrossRef]
- Verdonk, J.M.A.J.; Bruininx, E.M.A.M.; van der Meulen, J.; Verstegen, M.W.A. Post-weaning feed intake level modulates gut morphology but not gut permeability in weaned piglets. Livest. Sci. 2007, 108, 146–149. [Google Scholar] [CrossRef]
- Van der Meulen, J.; Koopmans, S.J.; Dekker, R.A.; Hoogendoorn, A. Increasing weaning age of piglets from 4 to 7 weeks reduces stress, increases post-weaning feed intake but does not improve intestinal functionality. Animal 2010, 4, 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Boudry, G.; Lalle’s, J.P.; Malbert, C.H.; Bobillier, E.; Sève, B. Diet-related adaptation of the small intestine at weaning in pigs is functional rather than structural. J. Pediatr. Gastroenterol. Nutr. 2002, 34, 180–187. [Google Scholar] [CrossRef]
- Koopmans, S.J.; Guzik, A.C.; Van Der Meulen, J.; Dekker, R.; Kogut, J.; Kerr, B.J.; Southern, L.L. Effects of supplemental L-tryptophan on serotonin, cortisol, intestinal integrity, and behavior in weanling piglets. J. Anim. Sci. 2006, 84, 963–971. [Google Scholar] [CrossRef]
- Spreeuwenberg, M.A.M.; Verdonk, J.M.A.J.; Gaskins, H.R.; Verstegen, M.W.A. Small intestine epithelial barrier function is compromised in pigs with low feed intake at weaning. J. Nutr. 2001, 131, 1520–1527. [Google Scholar] [CrossRef] [PubMed]
- Verdonk, J.M.A.J. Nutritional Strategy Affects Gut Wall Integrity in Weaned Piglets. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2006. [Google Scholar]
- Lodemann, U.; Hübener, K.; Jansen, N.; Martens, H. Effects of Enterococcus faecium NCIMB 10415 as probiotic supplement on intestinal transport and barrier function of piglets. Arch. Anim. Nutr. 2006, 60, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Lodemann, U.; Lorenz, B.M.; Weyrauch, K.D.; Martens, H. Effects of Bacillus cereus var. toyoi as probiotic feed supplement on intestinal transport and barrier function in piglets. Arch. Anim. Nutr. 2008, 62, 87–106. [Google Scholar] [CrossRef]
- Peace, R.M.; Campbell, J.; Polo, J.; Crenshaw, J.; Russell, L.; Moeser, A. Spray-dried porcine plasma influences intestinal barrier function, inflammation, and diarrhea in weaned pigs. J. Nutr. 2011, 141, 1312–1317. [Google Scholar] [CrossRef]
- Carlson, D.; Poulsen, H.D.; Sehested, J. Influence of weaning and effect of post weaning dietary zinc and copper on electrophysiological response to glucose, theophylline and 5-HT in piglet small intestinal mucosa. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2004, 137, 757–765. [Google Scholar] [CrossRef]
- Carlson, D.; Sehested, J.; Feng, Z.; Poulsen, H.D. Serosal zinc attenuate serotonin and vasoactive intestinal peptide induced secretion in piglet small intestinal epithelium in vitro. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2008, 149, 51–58. [Google Scholar] [CrossRef]
- Hamard, A.; Mazurais, D.; Boudry, G.; Le Huërou-Luron, I.; Sève, B.; Le Floc’h, N. A moderate threonine deficiency affects gene expression profile, para- cellular permeability and glucose absorption capacity in the ileum of piglets. J. Nutr. Biochem. 2010, 21, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Gabler, N.K.; Radcliffe, J.S.; Spencer, J.D.; Webel, D.M.; Spurlock, M.E. Feeding long-chain n-3 polyunsaturated fatty acids during gestation increases intestinal glucose absorption potentially via the acute activation of AMPK. J. Nutr. Biochem. 2009, 20, 17–25. [Google Scholar] [CrossRef]
- Gabler, N.K.; Spencer, J.D.; Webel, D.M.; Spurlock, M.E. In utero and postnatal exposure to long chain (n-3) PUFA enhances intestinal glucose absorption and energy stores in weanling pigs. J. Nutr. 2007, 137, 2351–2358. [Google Scholar] [CrossRef] [PubMed]
- Barrenetxe, J.; Sánchez, O.; Barber, A.; Gascón, S.; Rodríguez-Yoldi, M.J.; Lostao, M.P. TNFα regulates sugar transporters in the human intestinal epithelial cell line Caco-2. Cytokine 2013, 64, 181–187. [Google Scholar] [CrossRef]
- Tan, B.L.; Norhaizan, M.E.; Yeap, S.K.; Roselina, K. Water extract of brewers’ rice induces antiproliferation of human colorectal cancer (HT-29) cell lines via the induction of apoptosis. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 1022–1029. [Google Scholar]
- Yu, Z.T.; Nanthakumar, N.N.; Newburg, D.S. The Human Milk Oligosaccharide 2′-Fucosyllactose Quenches Campylobacter jejuni-Induced Inflammation in Human Epithelial Cells HEp-2 and HT-29 and in Mouse Intestinal Mucosa. J. Nutr. 2016, 146, 1980–1990. [Google Scholar] [CrossRef] [PubMed]
- Kwak, Y.; Ju, J. Inhibitory activities of Perilla frutescens britton leaf extract against the growth, migration, and adhesion of human cancer cells. Nutr. Res. Pract. 2015, 9, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.H.; Kim, W.K.; Ha, A.W.; Kim, M.H.; Chang, M.J. Anti-inflammatory effect of lycopene in SW480 human colorectal cancer cells. Nutr. Res. Pract. 2017, 11, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Bein, A.; Shin, W.; Jalili-Firoozinezhad, S.; Park, M.H.; Sontheimer-Phelps, A.; Tovaglieri, A.; Chalkiadaki, A.; Kim, H.J.; Ingber, D.E. Microfluidic Organ-on-a-Chip Models of Human Intestine. Cell. Mol. Gastroenterol. Hepatol. 2018, 5, 659–668. [Google Scholar] [CrossRef]
- Yin, Y.; Metselaar, H.J.; Sprengers, D.; Peppelenbosch, M.P.; Pan, Q. Rotavirus in organ transplantation: Drug-virus-host interactions. Am. J. Transplant. 2015, 15, 585–593. [Google Scholar] [CrossRef]
- Yin, Y.-B.; de Jonge, H.R.; Wu, X.; Yin, Y.-L. Enteroids for nutritional studies. Mol. Nutr. Food Res. 2019, 63, 1801143. [Google Scholar] [CrossRef] [PubMed]
- Seeger, B. Farm animal-derived models of the epithelium: Recent advances and future applications of intestinal organoids. Alt. Lab. Anim. 2020. [Google Scholar] [CrossRef]
- Resende, T.P.; Medida, R.L.; Vanuci, F.A.; Saqui-Salces, M.; Gebhart, C. Evaluation of swine enteroids as in vitro models for Lawsonia intracellularis infection. J. Anim. Sci. 2020, 98, 1–5. [Google Scholar] [CrossRef]
- Luo, H.; Zheng, J.; Chen, Y.; Wang, T.; Zhang, Z.; Shan, Y.; Xu, J.; Yue, M.; Fang, W.; Li, X. Utility evaluation of porcine enteroids as PDCoV infection model in vitro. Front. Microbiol. 2020, 11, 821. [Google Scholar] [CrossRef]
- Sato, T.; Clevers, H. Growing self-organizing mini-guts from a single intestinal stem cell: Mechanism and applications. Science 2013, 340, 1190–1194. [Google Scholar] [CrossRef]
- Just, A.; Jørgensen, H.; Fernández, J. Correlations of protein deposited in growing female pigs to ileal and faecal digestible crude protein and Amino acids. Livest. Prod. Sci. 1985, 12, 145–159. [Google Scholar] [CrossRef]
- Millet, S.; Aluwé, M.; Van Den Broeke, A.; Leen, F.; De Boever, J.; De Campeneere, S. Review: Pork production with maximal nitrogen efficiency. Animal 2018, 12, 1060–1067. [Google Scholar] [CrossRef]
- Geiger, E. The role of the time factor in protein synthesis. Science 1950, 111, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Chen, H. Protein Digestion Kinetics in Pigs and Poultry. Ph.D. Dissertation, Wageningen University, Wageningen, The Netherlands, 2017. [Google Scholar]
- Gerrits, W.J.J.; Schop, M.T.A.; de Vries, S.; Dijkstra, J. ASAS-NANP symposium: Digestion kinetics in pigs: The next step in feed evaluation and a ready-to-use modeling exercise. J. Anim. Sci. 2021, 99, 1–8. [Google Scholar] [CrossRef]
- Englyst, H.N.; Kingman, S.M.; Cummings, J.H. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 46 (Suppl. S2), S33–S50. [Google Scholar] [PubMed]
- Van Kempen, T.A.T.G.; Regmi, P.R.; Matte, J.J.; Zijlstra, R.T. In vitro starch digestion kinetics, corrected for estimated gastric emptying, predict portal glucose appearance in pigs. J. Nutr. 2010, 140, 1227–1233. [Google Scholar] [CrossRef] [PubMed]
- Gerrits, W.J.J.; Bosch, M.W.; van den Borne, J.J.G.C. Quantifying resistant starch using novel, in vivo methodology and the energetic utilization of fermented starch in pigs. J. Nutr. 2012, 142, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Patience, J.F.; Bedford, M.R.; Classen, H.L.; Inborr, J. The effect of dietary enzyme supplementation of rye-and barley-based diets on digestion and subsequent performance in weanling pigs. Can. J. Anim. Sci. 1992, 72, 97–105. [Google Scholar] [CrossRef]
- Dekker, R.A.; Smits, B.; Cone, J.W. Het Verloop van de Enzymatische Vertering en de Fermentatieve Afbraak van Rantsoenen Met Een Hoog Aandeel Aan Ontsloten Mais- of Natief Erwtenzetmeel in Het Maagdarm-Kanaal van Vleesvarkens; ID-DLO: Intern Raport; Institute for Animal Science and Health: Lelystad, The Netherlands, 1995. [Google Scholar]
- Blok, M.C.; Brandsma, G.; Bosch, G.; Gerrits, W.J.J.; Jansman, A.J.M.; Fledderus, J.; Everts, H. A New Dutch Net Energy Formula for Feed and Feedstuffs for Growing and Fattening Pigs; CVB-Documentation Report 56; WUR Livestock Research, Department of Animal Nutrition: Wageningen, The Netherlands, 2015; pp. 1–39. Available online: https://edepot.wur.nl/375605 (accessed on 19 February 2021).
- Urriola, P.E.; Shurson, G.C.; Stein, H.H. Digestibility of dietary fiber in distillers coproducts fed to growing pigs. J. Anim. Sci. 2010, 88, 2373–2381. [Google Scholar] [CrossRef]
- Salmas, G.; DeVries, J.W.; Plank, D. Challenges for dietary fiber: Benefits and costs of new U.S. regulations. Cereal Foods World 2017, 62, 88–94. [Google Scholar] [CrossRef]
- Urriola, P.E.; Stein, H.H. Comparative digestibility of energy and nutrients in fibrous feed ingredients fed to Meishan and Yorkshire pigs. J. Anim. Sci. 2012, 90, 802–812. [Google Scholar] [CrossRef]
- Shrestha, D.R. Effects of Supplemented NSP-Degrading Enzymes on Nutrient Digestibility of Diets Containing Co-Products Fed to Grower Pigs. Master’s Thesis, University of Alberta, Edmonton, AB, Canada, 2012. [Google Scholar]
- Ndou, S.P.; Kiarie, E.; Agyekum, A.K.; Heo, J.M.; Romero, L.F.; Arent, S.; Lorentsen, R.; Nyachoti, C.M. Comparative efficacy of xylanases on growth performance and digestibility in growing pigs fed wheat and wheat bran- or corn and corn DDGS-based diets supplemented with phytase. Anim. Feed Sci. Technol. 2015, 209, 230–239. [Google Scholar] [CrossRef]
- Urriola, P.E.; Zeng, Z.K.; Shurson, G.C. Strategies to Modify Fiber Structure and Increase Digestible Energy in Corn Distillers Dried Grains with Solubles—NPB #17-036; Report to the National Pork Board; National Pork Board: Des Moines, IA, USA, 2017.
- Abelilla, J.J.; Stein, H.H. Degradation of dietary fiber in the stomach, small intestine, and large intestine of growing pigs fed corn- or wheat-based diets without or with microbial xylanase. J. Anim. Sci. 2019, 97, 338–352. [Google Scholar] [CrossRef] [PubMed]
- Ferrandis Vila, M.; Trudeau, M.P.; Hung, Y.-T.; Zeng, Z.; Urriola, P.E.; Shurson, G.C.; Saqui-Salces, M. Dietary fiber sources and non-starch polysaccharide-degrading enzymes modify mucin expression and the immune profile of the swine ileum. PLoS ONE 2018, 13, e0207196. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.; Fledderus, J.; Busink, R.; Smits, C.; Ramaekers, P.J.L.; Jaworski, N.W. Interaction between protein sources (wheat gluten and protein concentrate from soy and potato) and starch sources (pre-gelatinized and native pea starch) on weanling pig growth performance and diarrhea incidence. J. Anim. Sci. 2018, 96, 4967285. [Google Scholar] [CrossRef]
- Jaworski, N.W.; Simard, F.; Leduc, M.; Ramaekers, P.; Fledderus, J.; Ferguson, N. Utilizing in vitro protein digestion kinetics and resistant fiber to steer ingredient composition of nursery pig diets for reduced risk of post-weaning diarrhea. In Proceedings of the Danish Seges: Zero Zinc Summit 2019, Copenhagen, Denmark, 17–18 June 2019; pp. 1–5. [Google Scholar]
- Ratanpaul, V.; Williams, B.A.; Black, J.L.; Gidley, M.J. Review: Effects of fibre, grain starch digestion rate and the ileal brake on voluntary feed intake in pigs. Animal 2019, 13, 2745–2754. [Google Scholar] [CrossRef] [PubMed]
- Ratanpaul, V.; Williams, B.A.; Black, J.L.; Gidley, M.J. Apparent amylase diffusion rates in milled cereal grains determined in vitro: Potential relevance to digestion in the small intestine of pigs. J. Cereal Sci. 2018, 82, 42–48. [Google Scholar] [CrossRef]
- Sanders, M.E. Overview of functional foods: Emphasis on probiotic bacteria. Int. Dairy J. 1998, 8, 341–347. [Google Scholar] [CrossRef]
- Calder, P.C. Feeding the immune system. Conference Proceedings on Transforming the Nutrition Landscape in Africa, Plenary Session 1. Proc. Nutr. Soc. 2013, 72, 299–309. [Google Scholar] [CrossRef]
- Venter, C.; Eyerich, S.; Sarin, T.; Klatt, K.C. Nutrition and the immune system: A complicated tango. Nutrients 2020, 12, 818. [Google Scholar] [CrossRef]
- Humphrey, B.; Zhao, J.; Faris, R. Review: Link between intestinal immunity and practical approaches to swine nutrition. Animal 2019, 11, 2736–2744. [Google Scholar] [CrossRef] [PubMed]
- Wu, G. Functional Amino acids in growth, reproduction, and health. Adv. Nutr. 2010, 1, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Liao, S. Physiological effects of dietary Amino acids on gut health and functions of swine. Front. Vet. Sci. 2019, 6, 169. [Google Scholar] [CrossRef] [PubMed]
- Van Dijk, A.J.; Everts, H.; Nabuurs, N.J.A.; Margry, R.J.C.F.; Beynen, A.C. Growth performance of waned pigs fed spray-dried animal plasma: A review. Livest. Prod. Sci. 2001, 68, 263–274. [Google Scholar] [CrossRef]
- Lallès, J.P.; Boudry, G.; Favier, C.; Le Floc’h, N.; Luron, I.; Montagne, L.; Oswald, I.P.; Pié, S.; Piel, C.; Sève, B. Gut function and dysfunction in young pigs: Physiology. Anim. Res. 2004, 53, 301–316. [Google Scholar] [CrossRef]
- O’Connell, J.M.; Callan, J.J.; O’Doherty, J.V. The interaction between cereal type and lactose level on piglet performance and diet digestibility post weaning. Anim. Sci. 2005, 81, 265–269. [Google Scholar] [CrossRef]
- Stein, H.H.; Kil, D.Y. Reduced use of antibiotic growth promoters in diets fed to weanling pigs: Dietary tools, part 2. Anim. Biotechnol. 2006, 17, 217–231. [Google Scholar] [CrossRef] [PubMed]
- De Lange, C.F.M.; Pluske, J.; Gong, J.; Nyachoti, C.M. Strategic use of feed ingredients and feed additives to stimulate gut health and development in young pigs. Livest. Sci. 2010, 134, 124–134. [Google Scholar] [CrossRef]
- Lombardi, P.; Musco, N.; Calabrò, S.; Tudisco, R.; Mastellone, V.; Vastolo, A.; Infascelli, F.; Cutrignelli, M.I. Different carbohydrate sources affect swine performance and post-prandial glycaemic response. Ital. J. Anim. Sci. 2020, 19, 421–430. [Google Scholar] [CrossRef]
- Poole, K. Review: At the nexus of antibiotics and metals: The impact of Cu and Zn on antibiotic activity and resistance. Trends Microbiol. 2017, 25, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Shannon, M.C.; Hill, G.M. Trace mineral supplementation for the intestinal health of young monogastric animals. Front. Vet. Sci. 2019, 6, 73. [Google Scholar] [CrossRef] [PubMed]
- Rosero, D.S.; Boyd, R.D.; Odle, J.; van Heugten, E. Optimizing dietary lipid use to improve essential fatty acid status and reproductive performance of the modern lactating sows: A review. J. Anim. Sci. Biotechnol. 2016, 7, 34. [Google Scholar] [CrossRef] [PubMed]
- Rosero, D.S.; Boyd, R.D.; McCulley, M.; Odle, J.; van Heugten, E. Essential fatty acid supplementation during lactation is required to maximize the subsequent reproductive performance of the modern sow. Anim. Reprod. Sci. 2016, 168, 151–163. [Google Scholar] [CrossRef]
- Lauridsen, C. Board invited review: Effects of dietary fatty acids on gut health and function of pigs pre- and post-weaning. J. Anim. Sci. 2020, 98, 1–12. [Google Scholar] [CrossRef]
- Missotten, J.A.M.; Michiels, J.; Degroote, J.; De Smet, S. Fermented liquid feed for pigs: An ancient technique for the future. J. Anim. Sci. Biotechnol. 2015, 6, 4. [Google Scholar] [CrossRef]
- Mukherjee, R.; Chakraborty, R.; Dutta, A. Role of fermentation in improving nutritional quality of soybean meal—A review. Asian Australas. J. Anim. Sci. 2016, 29, 1523–1529. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Chang, J.; Yin, Q.; Liu, M.; Di, Y.; Wang, P.; Wang, Z.; Wang, E.; Lu, F. Fermented soybean meal improves the growth performance, nutrient digestibility, and microbial flora in piglets. Anim. Nutr. 2017, 3, 19–24. [Google Scholar] [CrossRef]
- Viveros, A.; Chamorro, S.; Pizarro, M.; Arija, I.; Centeno, C.; Brenes, A. Effects of dietary polyphenol-rich grape products on intestinal microflora and gut morphology in broiler chicks. Poult. Sci. 2011, 90, 566–578. [Google Scholar] [CrossRef]
- Fiesel, A.; Gessner, D.K.; Erika Most, E.; Eder, K. Effects of dietary polyphenol-rich plant product from grape or hop on pro-inflammatory gene expression in the intestine, nutrient digestibility, and faecal microbiota of weaned pigs. BMC Vet. Res. 2014, 10, 196. [Google Scholar] [CrossRef] [PubMed]
- Verhelst, R.; Schroyen, M.; Buys, N.; Niewold, T. Dietary polyphenols reduce diarrhea in enterotoxigenic Escherichia coli (ETEC) infected post-weaning piglets. Livest. Sci. 2014, 160, 138–140. [Google Scholar] [CrossRef]
- Gerasopoulos, K.; Stagos, D.; Petrotos, K.; Kokkas, S.; Kantas, D.; Goulas, P.; Kouretas, D. Feed supplemented with polyphenolic by-product from olive mill wastewater processing improves redox status in blood and tissues of piglets. Food Chem. Toxicol. 2015, 86, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, J.E. Fiber effects in nutrition and gut health in pigs. J. Anim. Sci. Biotechnol. 2014, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. Review: The impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef]
- Beukema, M.; Faas, M.M.; de Vos, P. The effects of different dietary fiber pectin structures on the gastrointestinal immune barrier: Impact via gut microbiota and direct effects on immune cells. Exp. Mol. Med. 2020, 52, 1364–1376. [Google Scholar] [CrossRef]
- Zentek, J.; Buchheit-Renko, S.; Ferrara, F.; Vahjen, W.; van Kessel, A.G.; Pieper, R. Nutritional and physiological role of medium-chain triglycerides and medium-chain fatty acids in piglets. Anim. Health Res. Rev. 2011, 12, 83–93. [Google Scholar] [CrossRef]
- Cochrane, R.A.; Dritz, S.S.; Woodworth, J.C.; Stark, C.R.; Saensukjaroenphon, M.; Gebhardt, J.T.; Bai, J.; Hesse, R.A.; Poulsen, E.G.; Chen, Q.; et al. Assessing the effects of medium-chain fatty acids and fat sources on PEDV infectivity. Transl. Anim. Sci. 2020, 4, 1051–1059. [Google Scholar] [CrossRef]
- Cochrane, R.A.; Huss, A.R.; Aldrich, G.C.; Stark, C.R.; Jones, C.K. Evaluating chemical mitigation of Salmonella typhimurium ATCC 14028 in animal feed ingredients. J. Food Prot. 2016, 79, 672–676. [Google Scholar] [CrossRef] [PubMed]
- Hanczakowska, E. The use of medium-chain fatty acids in piglet feeding—A review. Ann. Anim. Sci. 2017, 17, 967–977. [Google Scholar] [CrossRef]
- Jackman, J.A.; Boyd, R.D.; Elrod, C.C. Medium-chain fatty acids and monoglycerides as feed additives for pig production: Towards gut health improvement and feed pathogen mitigation. J. Anim. Sci. Biotechnol. 2020, 11, 44. [Google Scholar] [CrossRef]
- Gebhardt, J.T.; Thomson, K.A.; Woodworth, J.C.; Dritz, S.S.; Tokach, M.D.; DeRouchey, J.M.; Goodband, R.D.; Jones, C.K.; Cochrane, R.A.; Niederwerder, M.C.; et al. Effect of dietary medium-chain fatty acids on nursery pig growth performance, fecal microbial composition, and mitigation properties against porcine epidemic diarrhea virus following storage. J. Anim. Sci. 2020, 98, 1–11. [Google Scholar] [CrossRef]
- Dahmer, P.L.; Leubcke, G.E.; Lerner, A.B.; Jones, C.K. Effects of medium-chain fatty acids as alternatives to ZnO or antibiotics in nursery pig diets. Transl. Anim. Sci. 2020, 4, 1–8. [Google Scholar] [CrossRef]
- Morais, T.; Inácio, A.; Coutinho, T.; Ministro, M.; Cotas, J.; Pereira, L.; Bahcevandziev, K. Seaweed potential in animal feed: A review. J. Mar. Sci. Eng. 2020, 8, 559. [Google Scholar] [CrossRef]
- Madeira, M.S.; Cardoso, C.; Lopes, P.A.; Coelho, D.; Afonso, C.; Bandarra, N.M.; Prates, J.A.M. Microalgae as feed ingredients for livestock production and meat quality: A review. Livest. Sci. 2017, 205, 111–121. [Google Scholar] [CrossRef]
- Smith, B.N.; Dilger, R.N. Immunomodulatory potential of dietary soybean-derived isoflavones and saponins in pigs. J. Anim. Sci. 2018, 96, 1288–1304. [Google Scholar] [CrossRef]
- Schweer, W.P.; Patience, J.F.; Burrough, E.R.; Kerr, B.J.; Gabler, N.K. Impact of PRRSV infection and dietary soybean meal on ileal amino acids digestibility and endogenous amino acidss losses in growing pigs. J. Anim. Sci. 2018, 96, 1846–1859. [Google Scholar] [CrossRef]
- Sinbad, O.O.; Folorunsho, A.A.; Olabisi, O.L.; Ayoola, O.A.; Temitope, E.J. Vitamins as antioxidants. J. Food Sci. Nutr. Res. 2019, 2, 214–235. [Google Scholar]
- Lauridsen, C.; Matte, L.L.; Lessard, M.; Celi, P.; Litta, G. Role of vitamins for gastro-intestinal functionality and health in pigs. Anim. Feed Sci. Technol. 2021, 114823. [Google Scholar] [CrossRef]
- World Health Organization. Diet, Nutrition, and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation; WHO Technical Report Series 916; WHO: Geneva, Switzerland, 2002. [Google Scholar]
- Saura-Calixto, F. Dietary fiber as a carrier of dietary antioxidants: An essential physiological function. J. Agric. Food Chem. 2011, 59, 43–49. [Google Scholar] [CrossRef]
- Lipiński, K.; Antoszkiewicz, Z.; Mazur-Kuśnirek, M.; Korniewicz, D.; Kotlarczyk, S. The effect of polyphenols on the performance and antioxidant status of sows and piglets. Ital. J. Anim. Sci. 2019, 18, 174–181. [Google Scholar] [CrossRef]
- Vitaglione, P.; Napolitano, A.; Fogliano, V. Cereal dietary fibre: A natural functional ingredient to deliver phenolic compounds to the gut. Trends Food Sci. Technol. 2008, 19, 451–463. [Google Scholar] [CrossRef]
- Quirós-Sauceda, A.E.; Palafox-Carlos, H.; Sáyago-Ayerdi, S.G.; Ayala-Zavala, J.F.; Bello-Perez, L.A.; Álvarez-Parrilla, E.; de la Rosa, L.A.; González-Córdova, A.F.; González-Aguilar, G.A. Dietary fiber and phenolic compounds as functional ingredients: Interaction and possible effect after ingestion. Food Funct. 2014, 5, 1063–1072. [Google Scholar] [CrossRef]
- Palafox-Carlos, H.; Ayala-Zavala, J.F.; González-Aguilar, G.A. The role of dietary fiber in the bioaccessibility and bioavailability of fruit and vegetable antioxidants. J. Food Sci. 2011, 76, R6–R15. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.Y.; Han, S.N. The role of vitamin E in immunity. Nutrients 2018, 10, 1614. [Google Scholar] [CrossRef]
- Smith, A.D.; Panickar, K.S.; Urban, J.F., Jr.; Dawson, H.D. Impact of micronutrients on the immune response of animals. Annu. Rev. Anim. Biosci. 2018, 6, 227–254. [Google Scholar] [CrossRef]
- De Santis, S.; Cavalcanti, E.; Mastronardi, M.; Jirillo, E.; Chieppa, M. Nutritional keys for intestinal barrier modulation. Front. Immunol. 2015, 6, 612. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Kunisawa, J. Vitamin-mediated immune regulation in the development of inflammatory diseases. Endocr. Metab. Immune Disord. Drug Targets 2015, 15, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Albers, R.; Antoine, J.M.; Blum, S.; Bourdet-Sicard, R.; Ferns, G.A.; Folkerts, G.; Friedmann, P.S.; Frost, G.S.; Guarner, F.; et al. Inflammatory disease processes and interactions with nutrition. Br. J. Nutr. 2009, 101, 1–45. [Google Scholar] [CrossRef] [PubMed]
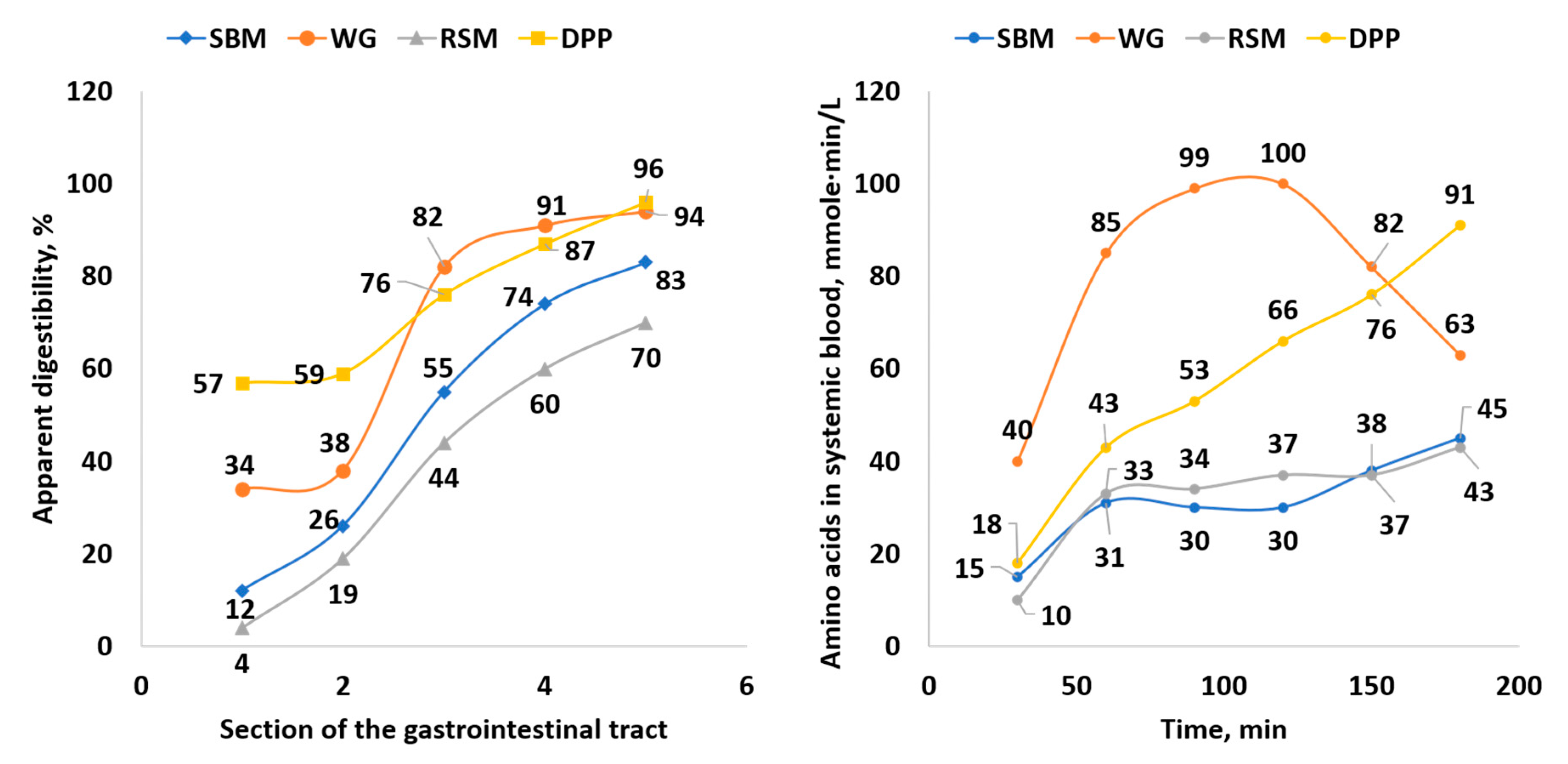
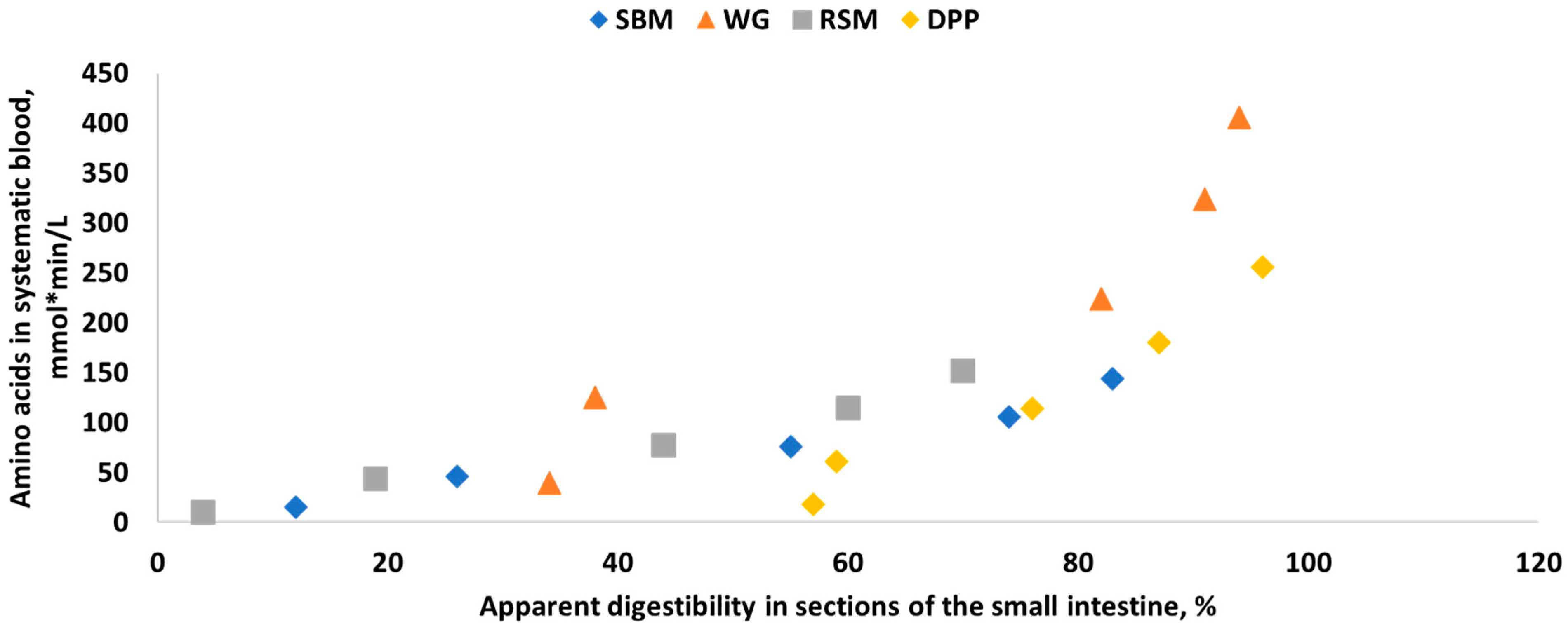
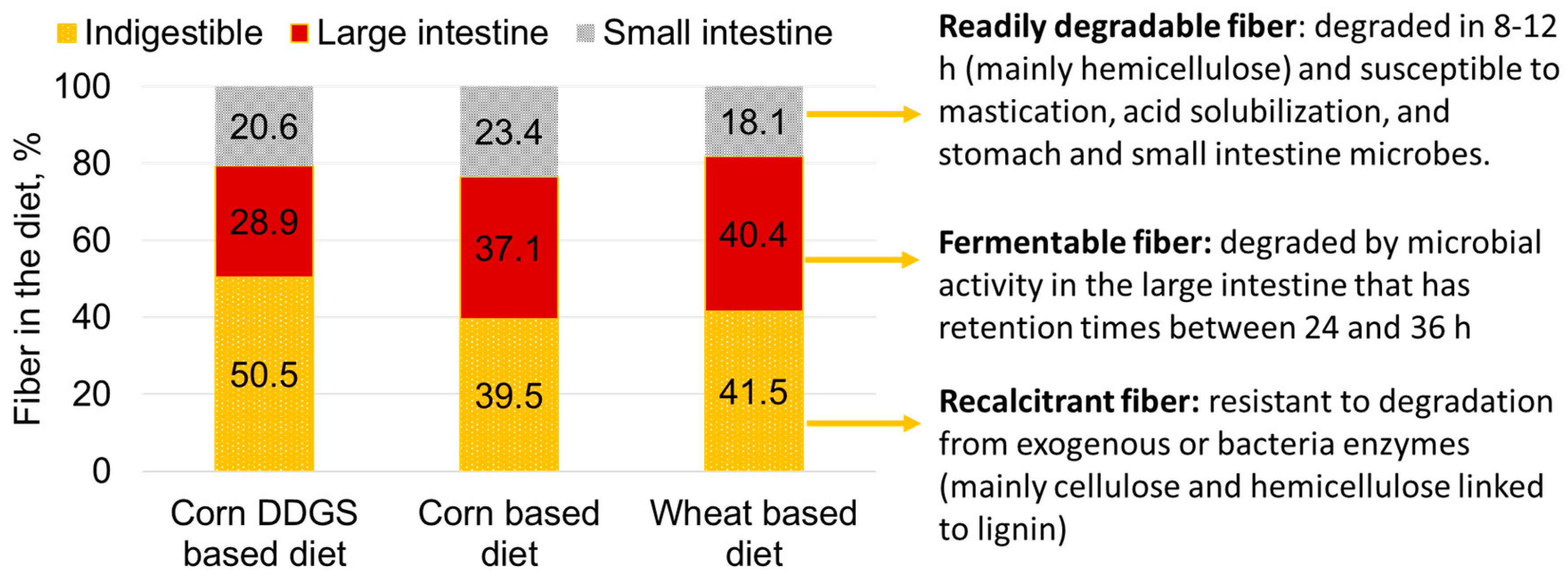
| Grain | Moisture Content (%) at Various aw | |||
|---|---|---|---|---|
| Water activity, aw | 0.60 | 0.65 | 0.70 | 0.75 |
| Rice | 13.2 | 13.8 | 14.2 | 15.0 |
| Oats | 11.2 | 12.2 | 13.0 | 14.0 |
| Rye | 12.2 | 12.8 | 13.6 | 14.6 |
| Barley | 12.2 | 13.0 | 14.0 | 15.0 |
| Corn | 12.8 | 13.4 | 14.2 | 15.2 |
| Sorghum | 12.0 | 13.0 | 13.8 | 14.8 |
| Wheat | 13.0 | 13.6 | 14.6 | 15.8 |
| Ingredient | Mean Particle Size (μm) | Water Activity, aw | Water Content, % | |||
|---|---|---|---|---|---|---|
| Coarse | Fine | Coarse | Fine | Coarse | Fine | |
| Soybean meal | 1430 | 342 | 0.657 | 0.596 | 11.8 | 11.4 |
| Barley | 2423 | 314 | 0.638 | 0.496 | 13.0 | 11.0 |
| Rapeseed cake | 708 | 310 | 0.502 | 0.481 | 8.7 | 8.2 |
| Corn 1 | - | - | 0.605 | 0.564 | 12.4 | 11.9 |
| Characteristic | Nonstarch Polysaccharides | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cellulose | Hemicelluloses | β-glucans | Pectins | Gums and Mucilages | Fructans | |||||
| Fractions | - | Arabinogalactans Glucans Arabinoxylans Glucuronoxylans Xyloglucans Galactomannans Pectin substances | Oat β-glucan, Barley β-glucan | - | Galactomannans Guar (PHGG) Locust bean gum (carob) Carob galactomannan Tara galactomannan | Gum (acacia) Gum (karaya) Gum (tragacanth) | Alginates Agar Carrageenan | Xanthan Gellan | Psyllium | Inulin Oligofructose |
| Ingredient sources | Brans Legumes Nuts Peas Cereals Functional fibers | Brans Cereal grains Legumes Nuts Vegetables Fruit Functional fibers | Oats Barley Rye | Fruits Vegetables Legumes Potato Sugar beets | Legumes Seed extracts (endosperm) | Tree extracts | Seaweed extracts (algal polysaccharides) | Microbial gums | Outer layer of seeds of plantain family | Chicory root Onion Artichoke Agave Wheat |
| Physicochemical properties a,b,c,d | Insoluble, Poorly to moderately fermentable | Insoluble, Poorly to moderately fermentable | Soluble, Viscous, Readily fermentable | Soluble, Readily fermentable | Soluble, Viscous (some), Readily fermentable | Soluble, Viscous (some gums), Readily fermentable | Soluble, Viscous (some), Readily fermentable | Soluble, Viscous (some), Readily fermentable | Soluble, Highly viscous, Not fermentable | Soluble, Nonviscous, Fermentable |
| Characteristic | Nondigestible Oligosaccharides (Short-Chain Oligosaccharides) | Other Fibers Found in Plants | Fibers Found in Animals (Fungi, Yeast, and Invertebrates) | ||||
|---|---|---|---|---|---|---|---|
| Lignin | Cutin | Suberin | Waxes | Chitin | Chitosan (Commercially Produced from Chitin) | ||
| Fractions | Fructooligosaccharides (FOS)/Neosugar Galactooligosaccharides Xylooligosaccharides Arabinoxylanoligosaccharides (AXOS) Soybean oligosaccharides | - | - | - | - | - | - |
| Ingredient sources | - | Woody plants or outer layer of cereal grains | - | - | - | - | - |
| Physicochemical properties a,b,c,d | Soluble, Nonviscous, Readily/rapidly fermentable | Insoluble, Poorly fermentable | Insoluble, Poorly fermentable | Insoluble, Poorly fermentable | Insoluble, Poorly fermentable | Insoluble, Poorly fermentable | Insoluble, Poorly fermentable |
| Characteristic | Resistant Starch | Chemically Synthesized Carbohydrate Compounds | ||||||
|---|---|---|---|---|---|---|---|---|
| Physically Inaccessible Starch | Native Starch Granules | Retrograded Starch | Chemically Modified Resistant Starch | Dextrin | Polydextrose (e.g., Litesse™) | Cellulose-Derived | PolyGlycopleX (PGX) | |
| Fractions | - | High amylose starch High amylose maize resistant starch type 2 High-maize 260 | - | - | Resistant maltodextrin 1 Fibersol Pine fiber Wheat dextrin Nutriose™ Corn fiber | - | Methylcellulose Hydroxypropylmethylcellulose Hydroxyethylmethylcellulose Ethylmethylcellulose/cellulose gum | - |
| Ingredient sources | Legumes Seeds Whole grains | Unripe banana Raw potato (ungelatinized starch granules) | Heated and cooled starch | Modified food starch | Hydrolyzed food starch | - | - | - |
| Physicochemical properties a,b,c,d | Poorly soluble, Variable fermentability | Poorly soluble, Variable fermentability | Poorly soluble, Variable fermentability | Soluble, Nonviscous, Readily fermentable | Viscous, Variable fermentability | Nonviscous, Readily fermentable | Viscous, Not fermentable Not gel-forming | - |
| Pig Body Weight | Diet Inclusion Rate | DE, kcal/kg | ME, kcal/kg | Reference |
|---|---|---|---|---|
| 13 kg | 5% | 8993–9038 | 8813–8856 | [141] |
| 50 kg | 5% | 8181–9049 | 8017–8868 | [141] |
| 38 kg | 4, 6, 8, 10% | 4% = 8243 | 4% = 7966 | [142] |
| 6% = 8419 | 6% = 8190 | |||
| 10% = 8911 | 8% = 8422 | |||
| 8% = 8775 | 10% = 8797 | |||
| 34 kg | 5 and 10% using two different basal diets | Corn–soybean meal | Corn–soybean meal | [143] |
| 5% = 8357 | 5% = 8099 | |||
| 10% = 8410 | 10% = 8854 | |||
| Corn starch casein | Corn starch casein | |||
| 5% = 8054 | 5% = 7896 | |||
| 10% = 8410 | 10% = 8319 | |||
| 19 kg | 7.13% | 9979 | - | [144] |
| 10 kg | 6.7% | 8567 | 8469 | [145] |
| 15 kg | 10% | 8315 | 8368 | [146] |
| - | - | 8749 | 8574 | [63] |
| - | - | 7977 | 7906 | [147] |
| - | - | 8600 | 8300 | [148] |
| Pig Body Weight | Diet Inclusion Rate | NE, kcal/kg | Reference |
|---|---|---|---|
| 22 kg | 5 or 10% | 5% = 4561 10% = 4781 | [136] |
| 84 kg | 5 or 10% | 5% = 5585 10% = 4578 | [136] |
| 31 kg | 5 or 10% | 5% = 7989 10% = 8132 | [149] |
| 13 kg | 5% | 7756–7795 | [141] |
| 50 kg | 5% | 7055–7804 | [141] |
| - | - | 7545 | [63] |
| - | - | 7117 | [147] |
| - | - | 7364 | [148] |
| Pig Body Weight | Equation 1 | R 2 | Reference |
|---|---|---|---|
| 19 kg | DE (kcal/kg) = 10,267 − (110.3 × FFA, %) − (41.8 × C16:0, %) − (39.7 × C18:0, %) − (98.0 × U:S) + (6.4 × iodine value) | 0.97 | [144] 2 |
| 13 kg | DE (Mcal/kg) = 9.363 − (0.097 × FFA, %) − (0.016 × n-6:n-3) − (1.24 × C20:0, %) − (5.054 × insoluble impurities, %) + (0.014 × C16:0, %) | 0.81 | [141] 3 |
| ME (Mcal/kg) = 9.176 − (0.095 × FFA, %) − (0.016 × n-6:n-3) − (1.215 × C20:0, %) − (4.953 × insoluble impurities, %) + (0.014 × C16:0, %) | 0.81 | [141] | |
| NE (Mcal/kg) = 8.075 − (0.093 × FFA, %) − (0.014 × n-6:n-3) − (1.07 × C20:0, %) − (4.359 × insoluble impurities, %) + (0.013 × C16:0, %) | 0.81 | [141] | |
| DE (kcal/kg) = 37.89 – (0.0051 × FFA, g/kg) – 8.20(−0.515 × U:S)/0.004184 | - | [129] | |
| 50 kg | DE (Mcal/kg) = 8.357 + (0.189 × U:S) − (0.195 × FFA, %) − (6.768 × C22:0, %) + (0.024 × PUFA, %) | 0.81 | [141] |
| ME (Mcal/kg) = 8.19 + (0.185 × U:S) − (0.191 × FFA, %) − (6.633 × C22:0, %) + (0.023 × PUFA, %) | 0.81 | [141] | |
| NE (Mcal/kg) = 7.207 + (0.163 × U:S) − (0.168 × FFA, %) − (5.836 × C22:0, %) + (0.021 × PUFA, %) | 0.81 | [141] | |
| DE (kcal/kg) = 36.898 − (0.0046 × FFA, g/kg) − 7.33(−0.906 × U:S)/0.004184 | [129] | ||
| Lactating sows | DE (kcal/kg) = 8381 − (80.6 × FFA, %) + (0.4 × FFA 2, %) + (248.8 × U:S) − (28.1 × U:S 2) + (12.8 × FFA, % × U:S) | 0.74 | [133] 4 |
| Criterion | Corn Oil Composition | ||||
|---|---|---|---|---|---|
| Free fatty acids % | 0.04 | 4.9 | 12.8 | 13.9 | 93.8 |
| UFA:SFA 1 | 6.13 | 5.00 | 5.61 | 5.00 | 4.81 |
| DE actual, kcal/kg | 8814 | 8828 | 8036 | 8465 | 8921 |
| DE predicted, kcal/kg | 8972 | 8848 | 8794 | 8741 | 7775 |
| Lipid Source | Actual DE, Mcal/kg | Powles et al. [129] predicted DE 1, Mcal/kg | Kellner and Patience [141] Predicted DE 2,3, Mcal/kg |
|---|---|---|---|
| 13 kg BW | |||
| Animal–vegetable blend | 8.81 | 8.40 | 8.34 |
| Canola oil | 8.59 | 8.82 | 8.56 |
| Choice white grease source A | 8.32 | 8.45 | 8.69 |
| Choice white grease source B | 8.67 | 8.46 | 8.79 |
| Coconut oil | 7.65 | 7.08 | 7.64 |
| Corn oil source A | 6.90 | 8.66 | 7.14 |
| Corn oil source B | 8.52 | 8.80 | 8.28 |
| Fish oil | 8.69 | 8.37 | 8.78 |
| Flax oil | 8.06 | 8.66 | 8.03 |
| Palm oil | 8.81 | 8.10 | 8.62 |
| Poultry fat | 8.67 | 8.57 | 8.38 |
| Soybean oil source A | 9.04 | 8.81 | 8.95 |
| Soybean oil source B | 8.99 | 8.81 | 8.97 |
| Tallow | 8.33 | 8.06 | 8.76 |
| 50 kg | |||
| Animal–vegetable blend | 7.51 | 8.40 | 7.69 |
| Canola oil | 9.53 | 8.82 | 9.52 |
| Choice white grease source A | 9.31 | 8.45 | 8.75 |
| Choice white grease source B | 8.72 | 8.46 | 8.77 |
| Coconut oil | 7.97 | 7.08 | 8.34 |
| Corn oil source A | 7.43 | 8.66 | 7.54 |
| Corn oil source B | 8.55 | 8.80 | 8.50 |
| Fish oil | 7.77 | 8.37 | 7.85 |
| Flax oil | 9.43 | 8.66 | 9.54 |
| Palm oil | 8.50 | 8.10 | 8.76 |
| Poultry fat | 8.14 | 8.57 | 7.93 |
| Soybean oil source A | 9.05 | 8.81 | 8.66 |
| Soybean oil source B | 8.18 | 8.81 | 8.71 |
| Tallow | 8.22 | 8.06 | 7.92 |
| Lipid Source | DE Actual, kcal/kg | DE Predicted 1, kcal/kg | DE Predicted 2, kcal/kg |
|---|---|---|---|
| Coconut oil | 9380 | 7104 | 8518 |
| Butter | 8911 | 7496 | 8471 |
| Tallow | 8071 | 7828 | 8464 |
| Palm oil | 8304 | 7861 | 7595 |
| Lard | 8648 | 7968 | 8118 |
| Fish oil | 9464 | 8059 | 8524 |
| Soybean oil | 9979 | 8944 | 8769 |
| Olive oil | 9606 | 8947 | 8639 |
| Flaxseed oil | 8584 | 8873 | 7764 |
| Canola oil | 9474 | 9053 | 8589 |
| Method | Enzymes Used | References |
|---|---|---|
| 1-step | Pepsin | [239] |
| Trypsin | [240] | |
| Papain | [241] | |
| Pronase | [242] | |
| 2-step | Pepsin–Pancreatin | [243,244,245] |
| Pepsin–Trypsin | [246] | |
| Pepsin–Pronase | [245] | |
| Pepsin–Jejunal fluid | [247] | |
| 3-step | Pepsin–Pancreatin–Cellulase | [248,249,250] |
| Pepsin–Pancreatin–Viscozyme | [251,252] | |
| Pepsin–Pancreatin–Rumen fluid | [253] |
| Ingredient | Bioactive Compounds | Functions | References |
|---|---|---|---|
| AAs | Glutamate, glutamine, glycine, proline, arginine | Signaling pathways regulating gene expression, intracellular turnover, nutrient metabolism, oxidative defense, and reducing intestinal damage | [171,333,334] |
| Animal plasma | Immunoglobulins | Improves immune response and gut-barrier function | [288,335,336] |
| Barley | Β-glucans, resistant starch, soluble and insoluble NSPs | Prebiotic to increase lactic acid production; improve gut health | [82,337,338,339,340] |
| Copper | Copper sulfate Tribasic copper chloride | Antibacterial alters gut microbiome | [341,342] |
| Essential fatty acids | Linoleic acid and linolenic acid | Improve reproductive performance in sows, affect inflammatory reactions and immune-response bacterial challenges and epithelial barrier function | [343,344,345] |
| Fermented liquid feed | Naturally occurring lactic acid bacteria and yeast | Production of lactic acid, acetic acid, and ethanol; reduces pH; prevents proliferation of pathogens | [338,346] |
| Fermented soybean meal | - | Decreased antinutritional factors, increased peptides, improves nutrient digestibility and gut microbiome | [347,348] |
| Functional fibers | Various types of NSPs | Alter gut microbiome, prebiotic, production of short-chain fatty acids, improve innate and adaptive immune responses, antioxidant and bactericidal properties | [82,83,89,90,94,349,350,351,352,353,354,355] |
| Lactose | Glucose and galactose | Improves nutrient digestibility, prebiotic, fermentation to lactic acid and volatile fatty acids in young pigs | [73,74,75,76] |
| Medium-chain fatty acids and monoglycerides | Caproic acid, caprylic acid, capric acid, and lauric acid; Glycerol monocaproate, glycerol monocaprylate, glycerol monocaprate, glycerol monolaurate | Antibacterial, antiviral, immune modulation activity, and improved gut health in pigs; feed-pathogen mitigation | [356,357,358,359,360,361,362] |
| Macroalgae–Seaweed | Functional fiber | Improve immune response and prebiotic; component of clay-based antimycotoxin agents | [363] |
| Microalgae | Omega-3 fatty acids | Improve immune response and reproduction; component of clay-based antimycotoxin agents | [364] |
| Oats | Β-glucans, resistant starch, soluble and insoluble NSPs | Prebiotic to increase lactic acid production; improve gut health | [82,337,338,339] |
| Soybean meal, soy-protein concentrate, and soy-protein isolate | Isoflavones | Anti-inflammatory, antioxidant, antiviral, decrease intestinal epithelial permeability, enhance growth and immune responses from PRRSV infection | [365,366] |
| Soybean meal, soy-protein concentrate, and soy-protein isolate | Saponins | Antioxidants, potential enhancers of passive immunity, vaccine adjuvants to increase immune response | [365] |
| Vitamins | A (carotenoids), C, D, E, K, niacin, pyridoxine, riboflavin | Antioxidants; gastrointestinal function and health | [367,368] |
| Zinc | Zinc oxide | Antibacterial, alters gut microbiome | [341,342] |
| Item | Barley | Corn | Oats | Rye | Wheat | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Grain | Bran | Grain | Bran | Grain | Bran | Grain | Bran | Grain | Bran | |
| Total dietary fiber, % | 14.6–27.1 | - | 13.1–19.6 | 86.7 | 11.5–37.7 | 18.1–25.2 | 15.2–20.9 | 35.8 | 11.6–17.0 | 36.5–52.4 |
| Insoluble dietary fiber, % | 12.0–22.1 | - | 11.6–16.0 | 86.5 | 8.6–33.9 | 14.5–20.2 | 11.1–16.0 | 30.5 | 10.2–14.7 | 35.0–48.4 |
| Soluble dietary fiber, % | 2.6–5.0 | - | 1.5–3.6 | 0.2 | 2.9–3.8 | 3.6–5.0 | 3.7–4.5 | 5.3 | 1.4–2.3 | 1.5–4.0 |
| Ferulic acid, mg/kg | 168–723 | 2002–2017 | 380–1759 | 26,100–33,000 | 359 | - | 6–860 | 25–2780 | 4.5–1270 | 1942–5400 |
| p-coumeric acid, mg/kg | 4–374 | 2565–3367 | 31 | 3000–4000 | - | - | 41 | 100–190 | 0.2–37.2 | 100–457 |
| Vanillic acid, mg/kg | 29.2–33.4 | 82–117 | 4.6 | - | 17 | - | 3–22 | 10 | 0.6–35 | 100–164 |
| Sinapic acid, mg/kg | - | - | 57 | - | 55 | - | 2–120 | 53–100 | 1.3–63 | 300 |
| Total phenolic content, mg gallic acid equivalent/kg | - | - | 2194–3010 | - | 1223 | 1950 | - | 5840 | 350–1505 | 2800–5643 |
| Vitamin | Mechanism | Impact |
|---|---|---|
| B-vitamins, E | Inhibition of inflammation via reduction of PGE2 | Reduced inflammation provides a less favorable environment for ETEC |
| Carotenoids, C, D, E, K, niacin, pyridoxine, and riboflavin | Control oxidative stress | Minimize production of reactive oxygen species during an induced inflammatory response and prevention of enteric infection |
| A,B-complex, C, D, and E | Improved immune cell activity, response, and homeostasis | Formation of immune cells and signals and modulation of immune cell responses |
| A and D | Improved intestinal barrier function | Regulation of tight junction molecules and prevention of barrier damage |
| A and D | Regulation of innate and adaptive immunity and resolution of inflammation | Immune cell differentiation and cytokine suppression in response to injury and infection and resolution of inflammation |
| A and D | Production of antimicrobial peptides | Enhance innate immunity and composition of commensal microbiota |
| A, B6, B12, thiamin, riboflavin, C, D, E, and K | Affect microbiome composition | Regenerate commensal microbiota |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shurson, G.C.; Hung, Y.-T.; Jang, J.C.; Urriola, P.E. Measures Matter—Determining the True Nutri-Physiological Value of Feed Ingredients for Swine. Animals 2021, 11, 1259. https://doi.org/10.3390/ani11051259
Shurson GC, Hung Y-T, Jang JC, Urriola PE. Measures Matter—Determining the True Nutri-Physiological Value of Feed Ingredients for Swine. Animals. 2021; 11(5):1259. https://doi.org/10.3390/ani11051259
Chicago/Turabian StyleShurson, Gerald C., Yuan-Tai Hung, Jae Cheol Jang, and Pedro E. Urriola. 2021. "Measures Matter—Determining the True Nutri-Physiological Value of Feed Ingredients for Swine" Animals 11, no. 5: 1259. https://doi.org/10.3390/ani11051259
APA StyleShurson, G. C., Hung, Y.-T., Jang, J. C., & Urriola, P. E. (2021). Measures Matter—Determining the True Nutri-Physiological Value of Feed Ingredients for Swine. Animals, 11(5), 1259. https://doi.org/10.3390/ani11051259








