Assessing the Potential of Diverse Forage Mixtures to Reduce Enteric Methane Emissions In Vitro
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. In Vitro Incubation
2.3. Sample Analysis
2.3.1. Analysis of the Chemical Composition
2.3.2. Analysis of Tannin Composition
2.4. Statistical Analysis
3. Results
3.1. Measured Parameters
3.1.1. Forage Quality Was High for All Species and across Both Years
3.1.2. Plant Secondary Metabolites Varied across Species with L. pedunculatus and S. minor Showing Highest Tannin Concentrations
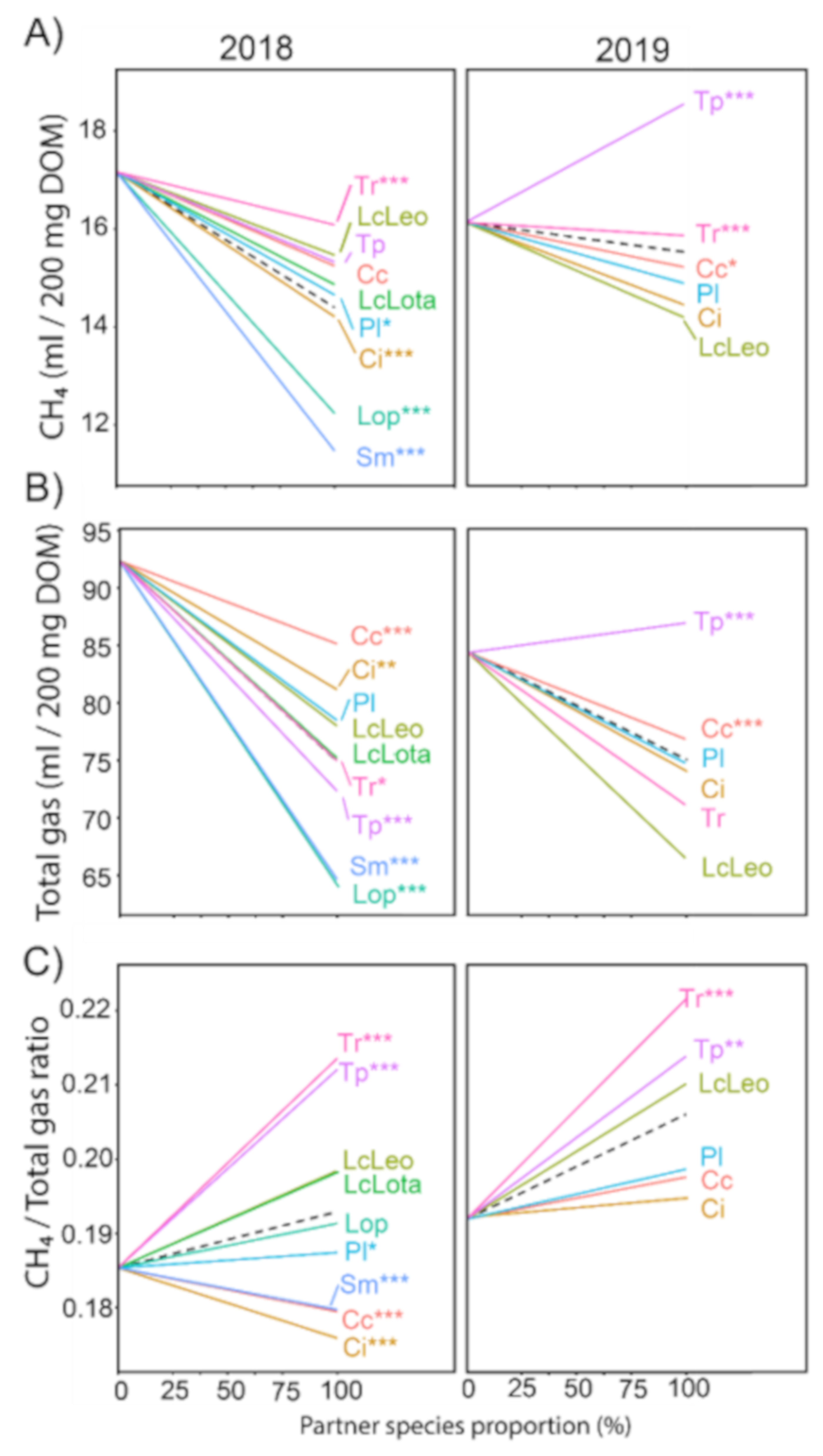
3.1.3. Total Gas Production Was Generally Linked to the Mixture Composition Linearly, While Changes in Methane Were Less Predictable
3.1.4. Covariates Did Not Improve the Model’s Ability to Predict Rumen Fermentation Parameters
3.2. Modeled Fermentation Parameters
3.2.1. Tannin-Containing Species Reduce Methane Formation Most, but Do Not Provide the Lowest Methane Concentrations
3.2.2. Despite Differences in Total Gas and Methane Formation, Trends in Methane Concentration Are Stable across Both Experimental Years
3.3. Performance of Model Compared to Measured Data
4. Discussion
4.1. The Experimental Setup Was Suitable to Answer the Research Question
4.2. Tannin-Rich Forages Reduced Methane Formation Most, but Chicory Was the Best Species for Reducing Methane Concentration
4.3. Unlike Digestibility, Methane Formation Cannot Be Estimated Accurately Based on the Share and Type of Partner Species, or from Forage Quality Parameters Alone
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Parameter | Plant Group | N | Mean | Range | SEC | R2 | SEP |
|---|---|---|---|---|---|---|---|
| ME (MJ/kg DM) | Whole sward | 251 | 10.810 | 8.38–12.62 | 0.179 | 0.959 | 0.195 |
| ME (MJ/kg DM) | Grasses | 248 | 10.806 | 8.38–12.62 | 0.173 | 0.956 | 0.194 |
| Legumes | 168 | 10.737 | 8.55–12.41 | 0.15 | 0.961 | 0.196 | |
| Herbs | 117 | 10.691 | 8.38–12.54 | 0.154 | 0.963 | 0.211 | |
| NEL (MJ/kg DM) | Whole sward | 249 | 6.567 | 4.79–7.81 | 0.138 | 0.955 | 0.173 |
| NEL (MJ/kg DM) | Grasses | 250 | 6.569 | 4.79–7.87 | 0.136 | 0.949 | 0.193 |
| Legumes | 167 | 6.512 | 4.92–7.77 | 0.106 | 0.965 | 0.152 | |
| Herbs | 115 | 6.514 | 4.80–7.784 | 0.112 | 0.964 | 0.163 | |
| DOM (g/kg DM) | Whole sward | 249 | 809.45 | 642.6–919.0 | 8.85 | 0.967 | 9.14 |
| DOM (g/kg DM) | Grasses | 248 | 821.04 | 632.1–918.8 | 10.83 | 0.956 | 10.97 |
| Legumes | 173 | 803.63 | 657.6–902.6 | 10.36 | 0.944 | 12.96 | |
| Herbs | 118 | 817.42 | 642.6–904.3 | 13.51 | 0.943 | 12.45 | |
| N (g/kg DM) | Whole sward | 268 | 26.69 | 8.7–54.2 | 0.951 | 0.991 | 1.071 |
| N (g/kg DM) | Grasses | 277 | 22.59 | 9–54.9 | 0.802 | 0.991 | 0.914 |
| Legumes | 178 | 35.07 | 14.7–57.1 | 1.116 | 0.981 | 1.131 | |
| Herbs | 86 | 26.39 | 10.3–39.9 | 0.749 | 0.995 | 1.317 |
References
- Lemaire, G.; Gastal, F.; Franzluebbers, A.; Chabbi, A. Grassland–cropping rotations: An avenue for agricultural diversification to reconcile high production with environmental quality. Environ. Manag. 2015, 56, 1065–1077. [Google Scholar] [CrossRef] [PubMed]
- Nyfeler, D.; Huguenin-Elie, O.; Suter, M.; Frossard, E.; Connolly, J.; Lüscher, A. Strong mixture effects among four species in fertilized agricultural grassland led to persistent and consistent transgressive overyielding. J. Appl. Ecol. 2009, 46, 683–691. [Google Scholar] [CrossRef]
- Finn, J.A.; Kirwan, L.; Connolly, J.; Sebastià, M.T.; Helgadottir, A.; Baadshaug, O.H.; Bélanger, G.; Black, A.; Brophy, C.; Collins, R.P.; et al. Ecosystem function enhanced by combining four functional types of plant species in intensively managed grassland mixtures: A 3-year continental-scale field experiment. J. Appl. Ecol. 2013, 50, 365–375. [Google Scholar] [CrossRef]
- Suter, M.; Connolly, J.; Finn, J.A.; Loges, R.; Kirwan, L.; Sebastià, M.-T.; Lüscher, A. Nitrogen yield advantage from grass-legume mixtures is robust over a wide range of legume proportions and environmental conditions. Glob. Chang. Biol. 2015, 21, 2424–2438. [Google Scholar] [CrossRef] [PubMed]
- Connolly, J.; Sebastià, M.-T.; Kirwan, L.; Finn, J.A.; Llurba, R.; Suter, M.; Collins, R.P.; Porqueddu, C.; Helgadóttir, Á.; Baadshaug, O.H.; et al. Weed suppression greatly increased by plant diversity in intensively managed grasslands: A continental-scale experiment. J. Appl. Ecol. 2018, 55, 852–862. [Google Scholar] [CrossRef] [PubMed]
- Lüscher, A.; Mueller-Harvey, I.; Soussana, J.F.; Rees, R.M.; Peyraud, J.L. Potential of legume-based grassland–livestock systems in Europe: A review. Grass Forage Sci. 2014, 69, 206–228. [Google Scholar] [CrossRef]
- Mueller-Harvey, I.; Bee, G.; Dohme-Meier, F.; Hoste, H.; Karonen, M.; Kölliker, R.; Lüscher, A.; Niderkorn, V.; Pellikaan, W.F.; Salminen, J.-P.; et al. Benefits of condensed tannins in forage legumes fed to ruminants: Importance of structure, concentration, and diet composition. Crop Sci. 2019, 59, 861–885. [Google Scholar] [CrossRef]
- Mueller-Harvey, I. Unravelling the conundrum of tannins in animal nutrition and health. J. Sci. Food Agric. 2006, 86, 2010–2037. [Google Scholar] [CrossRef]
- Min, B.R.; Barry, T.N.; Attwood, G.T.; McNabb, W.C. The effect of condensed tannins on the nutrition and health of ruminants fed fresh temperate forages: A review. Anim. Feed. Sci. Technol. 2003, 106, 3–19. [Google Scholar] [CrossRef]
- Niderkorn, V.; Baumont, R.; Le Morvan, A.; Macheboeuf, D. Occurrence of associative effects between grasses and legumes in binary mixtures on in vitro rumen fermentation characteristics. J. Anim. Sci. 2011, 89, 1138–1145. [Google Scholar] [CrossRef]
- Thorpe, A. Enteric fermentation and ruminant eructation: The role (and control?) of methane in the climate change debate. Clim. Chang. 2009, 93, 407–431. [Google Scholar] [CrossRef]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Mottet, A.; Opio, C.; Dijkman, J.; Falcucci, A.; Tempio, G. Tackling Climate Change through Livestock—A Global Assessment of Emissions and Mitigation Opportunities; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013; p. 139. Available online: http://www.fao.org/3/i3437e/i3437e.pdf (accessed on 12 March 2021).
- Verma, S.; Taube, F.; Malisch, C.S. Examining the variables leading to apparent incongruity between antimethanogenic potential of tannins and their observed effects in ruminants—A Review. Sustainability 2021, 13, 2743. [Google Scholar] [CrossRef]
- Aerts, R.J.; Barry, T.N.; McNabb, W.C. Polyphenols and agriculture: Beneficial effects of proanthocyanidins in forages. Agric. Ecosyst. Environ. 1999, 75, 1–12. [Google Scholar] [CrossRef]
- Waghorn, G. Beneficial and detrimental effects of dietary condensed tannins for sustainable sheep and goat production—Progress and challenges. Anim. Feed Sci. Technol. 2008, 147, 116–139. [Google Scholar] [CrossRef]
- Chung, Y.-H.; Mc Geough, E.J.; Acharya, S.; McAllister, T.A.; McGinn, S.M.; Harstad, O.M.; Beauchemin, K.A. Enteric methane emission, diet digestibility, and nitrogen excretion from beef heifers fed sainfoin or alfalfa1. J. Anim. Sci. 2013, 91, 4861–4874. [Google Scholar] [CrossRef]
- Huyen, N.T.; Fryganas, C.; Uittenbogaard, G.; Mueller-Harvey, I.; Verstegen, M.W.A.; Hendriks, W.H.; Pellikaan, W.F. Structural features of condensed tannins affect in vitro ruminal methane production and fermentation characteristics. J. Agric. Sci. 2016, 154, 1474–1487. [Google Scholar] [CrossRef]
- Jayanegara, A.; Marquardt, S.; Wina, E.; Kreuzer, M.; Leiber, F. In vitro indications for favourable non-additive effects on ruminal methane mitigation between high-phenolic and high-quality forages. Br. J. Nutr. 2013, 109, 615–622. [Google Scholar] [CrossRef]
- Jayanegara, A.; Marquardt, S.; Kreuzer, M.; Leiber, F. Nutrient and energy content, in vitro ruminal fermentation characteristics and methanogenic potential of alpine forage plant species during early summer: Ruminal fermentation traits of alpine forage plants. J. Sci. Food Agric. 2011, 91, 1863–1870. [Google Scholar] [CrossRef]
- Top, S.M.; Preston, C.M.; Dukes, J.S.; Tharayil, N. Climate Influences the Content and Chemical Composition of Foliar Tannins in Green and Senesced Tissues of Quercus rubra. Front. Plant Sci. 2017, 8, 423. [Google Scholar] [CrossRef]
- Malisch, C.S.; Salminen, J.-P.; Kölliker, R.; Engström, M.T.; Suter, D.; Studer, B.; Lüscher, A. Drought effects on proanthocyanidins in sainfoin ( Onobrychis viciifolia Scop.) are dependent on the plant’s ontogenetic stage. J. Agric. Food Chem. 2016, 64, 9307–9316. [Google Scholar] [CrossRef]
- Selmar, D.; Kleinwächter, M. Stress enhances the synthesis of secondary plant products: The impact of stress-related over-reduction on the accumulation of natural products. Plant Cell Physiol. 2013, 54, 817–826. [Google Scholar] [CrossRef]
- Moore, K.J.; Lenssen, A.W.; Fales, S.L. Factors Affecting Forage Quality. In Forages; Moore, K.J., Collins, M., Nelson, C.J., Redfearn, D.D., Eds.; Wiley: Hoboken, NJ, USA, 2020; pp. 701–717. ISBN 978-1-119-43657-7. [Google Scholar] [CrossRef]
- Minnée, E.M.K.; Leach, C.M.T.; Dalley, D.E. Substituting a pasture-based diet with plantain (Plantago lanceolata) reduces nitrogen excreted in urine from dairy cows in late lactation. Livest. Sci. 2020, 239, 104093. [Google Scholar] [CrossRef]
- Menke, K.; Steingass, H. Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen fluid. Anim. Res. Dev. 1988, 28, 7–55. [Google Scholar]
- De Boever, J.L.; Cottyn, B.G.; Andries, J.I.; Buysse, F.X.; Vanacker, J.M. The use of a cellulase technique to predict digestibility, metabolizable and net energy of forages. Anim. Feed Sci. Technol. 1988, 19, 247–260. [Google Scholar] [CrossRef]
- Malisch, C.S.; Lüscher, A.; Baert, N.; Engström, M.T.; Studer, B.; Fryganas, C.; Suter, D.; Mueller-Harvey, I.; Salminen, J.-P. Large variability of proanthocyanidin content and composition in Sainfoin (Onobrychis viciifolia). J. Agric. Food Chem. 2015, 63, 10234–10242. [Google Scholar] [CrossRef]
- Engström, M.T.; Pälijärvi, M.; Fryganas, C.; Grabber, J.H.; Mueller-Harvey, I.; Salminen, J.-P. Rapid qualitative and quantitative analyses of proanthocyanidin oligomers and polymers by UPLC-MS/MS. J. Agric. Food Chem. 2014, 62, 3390–3399. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: http://www.r-project.org (accessed on 12 March 2021).
- Pinheiro, J.C.; Bates, D.M.; DebRoy, S.; Sarkar, D.; R Core Team. nlme: Linear and Nonlinear Mixed Effects Models, 3.1-128. 2016. Available online: https://CRAN.R-project.org/package=nlme (accessed on 12 March 2021).
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous Inference in General Parametric Models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef]
- Nakagawa, S.; Schielzeth, H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol. Evol. 2013, 4, 133–142. [Google Scholar] [CrossRef]
- Akaike, H. A New Look at the Statistical Model Identification. In Selected Papers of Hirotugu Akaike; Parzen, E., Tanabe, K., Kitagawa, G., Eds.; Springer Series in Statistics; Springer: New York, NY, USA, 1974; pp. 215–222. ISBN 978-1-4612-7248-9. [Google Scholar] [CrossRef]
- Beauchemin, K.A.; Kreuzer, M.; O’Mara, F.; McAllister, T.A. Nutritional management for enteric methane abatement: A review. Aust. J. Exp. Agric. 2008, 48, 21–27. [Google Scholar] [CrossRef]
- Yáñez-Ruiz, D.R.; Bannink, A.; Dijkstra, J.; Kebreab, E.; Morgavi, D.P.; O’Kiely, P.; Reynolds, C.K.; Schwarm, A.; Shingfield, K.J.; Yu, Z.; et al. Design, implementation and interpretation of in vitro batch culture experiments to assess enteric methane mitigation in ruminants—A review. Anim. Feed Sci. Technol. 2016, 216, 1–18. [Google Scholar] [CrossRef]
- Loza, C.; Reinsch, T.; Loges, R.; Taube, F.; Kluß, C.; Hasler, M.; Malisch, C.S. Methane Emission and Milk Production from Jersey Cows Grazing Perennial Ryegrass-White Clover and Multispecies Forage Mixtures. Agriculture 2021, 11, 175. [Google Scholar] [CrossRef]
- Tiemann, T.T.; Avila, P.; Ramírez, G.; Lascano, C.E.; Kreuzer, M.; Hess, H.D. In vitro ruminal fermentation of tanniniferous tropical plants: Plant-specific tannin effects and counteracting efficiency of PEG. Anim. Feed Sci. Technol. 2008, 146, 222–241. [Google Scholar] [CrossRef]
- Robinson, P.H.; Getachew, G.; Cone, J.W. Evaluation of the extent of associative effects of two groups of four feeds using an in vitro gas production procedure. Anim. Feed Sci. Technol. 2009, 150, 9–17. [Google Scholar] [CrossRef]
- Goel, G.; Makkar, H.P.S. Methane mitigation from ruminants using tannins and saponins. Trop. Anim. Health Prod. 2012, 44, 729–739. [Google Scholar] [CrossRef]
- Lee, J.M.; Hemmingson, N.R.; Minnee, E.M.K.; Clark, C.E.F. Management strategies for chicory (Cichorium intybus) and plantain (Plantago lanceolata): Impact on dry matter yield, nutritive characteristics and plant density. Crop Pasture Sci. 2015, 66, 168. [Google Scholar] [CrossRef]
- Durmic, Z.; Moate, P.J.; Jacobs, J.L.; Vadhanabhuti, J.; Vercoe, P.E. In vitro fermentability and methane production of some alternative forages in Australia. Anim. Prod. Sci. 2016, 56, 641. [Google Scholar] [CrossRef]
- Williams, A.R.; Peña-Espinoza, M.A.; Boas, U.; Simonsen, H.T.; Enemark, H.L.; Thamsborg, S.M. Anthelmintic activity of chicory (Cichorium intybus): In vitro effects on swine nematodes and relationship to sesquiterpene lactone composition. Parasitology 2016, 143, 770–777. [Google Scholar] [CrossRef]
- Peña-Espinoza, M.; Valente, A.H.; Bornancin, L.; Simonsen, H.T.; Thamsborg, S.M.; Williams, A.R.; López-Muñoz, R. Anthelmintic and metabolomic analyses of chicory (Cichorium intybus) identify an industrial by-product with potent in vitro antinematodal activity. Vet. Parasitol. 2020, 280, 109088. [Google Scholar] [CrossRef] [PubMed]
- Foo, L.Y.; Lu, Y.; Molan, A.L.; Woodfield, D.R.; McNabb, W.C. The phenols and prodelphinidins of white clover flowers. Phytochemistry 2000, 54, 539–548. [Google Scholar] [CrossRef]
- Burggraaf, V.; Waghorn, G.; Woodward, S.; Thom, E. Effects of condensed tannins in white clover flowers on their digestion in vitro. Anim. Feed Sci. Technol. 2008, 142, 44–58. [Google Scholar] [CrossRef]
- Winters, A.L.; Minchin, F.R.; Michaelson-Yeates, T.P.T.; Lee, M.R.F.; Morris, P. Latent and active polyphenol oxidase (PPO) in red clover (Trifolium pratense) and use of a low PPO mutant to study the role of PPO in proteolysis reduction. J. Agric. Food Chem. 2008, 56, 2817–2824. [Google Scholar] [CrossRef]
- Copani, G.; Niderkorn, V.; Anglard, F.; Quereuil, A.; Ginane, C. Silages containing bioactive forage legumes: A promising protein-rich feed source for growing lambs. Grass Forage Sci. 2016, 71, 622–631. [Google Scholar] [CrossRef]
- Copani, G.; Ginane, C.; Le Morvan, A.; Niderkorn, V. Patterns of in vitro rumen fermentation of silage mixtures including sainfoin and red clover as bioactive legumes. Anim. Feed Sci. Technol. 2015, 208, 220–224. [Google Scholar] [CrossRef]
- Gidlund, H.; Hetta, M.; Huhtanen, P. Milk production and methane emissions from dairy cows fed a low or high proportion of red clover silage and an incremental level of rapeseed expeller. Livest. Sci. 2017, 197, 73–81. [Google Scholar] [CrossRef]

| Species/Cultivar † | Common Name | Abb * | Plant Family | Year | |
|---|---|---|---|---|---|
| Common | Lolium perenne | Perennial ryegrass | L. perenne | Poaceae | 2018, 2019 |
| Partner | Carum carvi | Caraway | C. carvi | Apiaceae | 2018, 2019 |
| Cichorium intybus | Chicory | C. intybus | Asteraceae | 2018, 2019 | |
| Lotus corniculatus “Leo” | Birdsfoot trefoil | L. corniculatus Leo | Fabaceae | 2018, 2019 | |
| Lotus corniculatus “Lotanava” | Birdsfoot trefoil | L. corniculatus Lotanava | Fabaceae | 2018 | |
| Lotus pedunculatus | Big trefoil | L. pedunculatus | Fabaceae | 2018 | |
| Trifolium pratense | Red clover | T. pratense | Fabaceae | 2018, 2019 | |
| Trifolium repens | White clover | T. repens | Fabaceae | 2018, 2019 | |
| Plantago lanceolata | Ribwort plantain | P. lanceolata | Plantaginaceae | 2018, 2019 | |
| Sanguisorba minor | Salad burnet | S. minor | Rosaceae | 2018 |
| Year | Species | MEgas | MENIRS | NEL | NDF | ADF | DOM | CP |
|---|---|---|---|---|---|---|---|---|
| (MJ/kg DM) | (g/kg DM) | |||||||
| 2018 | L. perenne | 11.6 | 10.5 | 6.5 | 508 | 271 | 850 | 108 |
| C. carvi | 11.0 | 11.0 | 6.7 | 303 | 233 | 930 | 67 | |
| C. intybus | 11.1 | 11.3 | 6.7 | 346 | 236 | 920 | 100 | |
| L. corniculatus Leo | 11.2 | 11.2 | 6.8 | 351 | 217 | 840 | 178 | |
| L. corniculatus Lotanava | 10.7 | 10.9 | 6.6 | 375 | 240 | 830 | 182 | |
| L. pedunculatus | 9.6 | 10.3 | 6.1 | 449 | 290 | 800 | 173 | |
| T. pratense | 10.9 | 10.8 | 6.6 | 412 | 221 | 860 | 214 | |
| T. repens | 11.0 | 10.6 | 6.6 | 418 | 257 | 860 | 209 | |
| P. lanceolata | 11.1 | 11.3 | 6.9 | 341 | 232 | 910 | 133 | |
| S. minor | 10.6 | 12.2 | 7.0 | 417 | 253 | 890 | 149 | |
| 2019 | L. perenne | 12.1 | 11.7 | 7.2 | 394 | 200 | 910 | 100 |
| C. carvi | 10.2 | 10.3 | 6.3 | 414 | 291 | 870 | 145 | |
| C. intybus | 10.4 | 10.4 | 6.4 | 513 | 276 | 870 | 162 | |
| L. corniculatus Leo | 9.3 | 9.9 | 5.9 | 425 | 310 | 810 | 186 | |
| T. pratense | NA | 11.3 | 6.9 | 359 | 204 | 850 | 172 | |
| T. repens | 11.4 | 11.4 | 7.1 | 338 | 189 | 890 | 214 | |
| P. lanceolata | 9.9 | 9.8 | 6.1 | 582 | 310 | 790 | 169 | |
| Year | Species | PP | F | T | HT | PA | PD Share |
|---|---|---|---|---|---|---|---|
| (mg/g DM) | (%) | ||||||
| 2018 | C. carvi | 8.8 | 4.1 | 0.0 | |||
| C. intybus | 9.3 | 2.4 | 0.0 | ||||
| L. corniculatus Leo | 3.1 | 1.4 | 1.7 | 0.2 | 1.5 | 25 | |
| L. corniculatus Lotanava | 5.6 | 1.5 | 4.1 | 0.3 | 3.9 | 36 | |
| L. pedunculatus | 20.8 | 2.4 | 18.4 | 0.2 | 18.2 | 79 | |
| T. pratense | 4.5 | 4.0 | 0.3 | 0.0 | 0.3 | 44 | |
| T. repens | 6.1 | 4.0 | 1.8 | 0.0 | 1.8 | 100 | |
| P. lanceolata | 1.2 | 0.1 | 0.0 | ||||
| S. minor | 47.6 | 5.6 | 41.0 | 41.0 | 0.0 | 0 | |
| 2019 | C. carvi | 5.9 | 2.9 | 0.0 | |||
| C. intybus | 4.2 | 1.4 | 0.0 | ||||
| L. corniculatus Leo | 3.6 | 1.3 | 2.0 | 0.1 | 1.9 | 30 | |
| T. pratense | 3.3 | 3.1 | 0.1 | 0.0 | 0.1 | 0 | |
| T. repens | 2.8 | 2.8 | 0.0 | ||||
| P. lanceolata | 1.1 | 0.3 | 0.0 | ||||
| Species | Partner Prop. | Total Gas (mL/200 mg DOM 1) | Methane (mL/200 mg DOM) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2018 | 2019 | 2018 | 2019 | ||||||||||
| L. perenne | 1 | 93.4 | (3.23) | 80.8 | (2.38) | 19.2 | (1.68) | 15.6 | (0.24) | ||||
| C. carvi | 0.25 | 94.2 | (0.68) | 83.9 | (0.68) | 16.1 | (0.07) | 16.4 | (0.32) | ||||
| 0.375 | 89.5 | (1.94) | 82.4 | (0.64) | 16.5 | (0.45) | 16.2 | (0.33) | |||||
| 0.5 | 90.0 | (0.72) | 74.9 | (0.41) | 15.3 | (0.34) | 14.5 | (0.16) | |||||
| 0.675 | 86.8 | (1.14) | 82.2 | (0.72) | 14.3 | (0.09) | 16.1 | (0.08) | |||||
| 0.75 | 87.7 | (1.22) | 77.8 | (0.64) | 16.0 | (0.43) | 15.4 | (0.45) | |||||
| 1 | 83.8 | (0.95) | 77.3 | (5.31) | 16.6 | (0.46) | 15.1 | (1.29) | |||||
| C. intybus | 0.25 | 94.3 | (0.28) | 81.2 | A | (1.10) | 16.0 | (0.06) | 15.9 | AB | (0.28) | ||
| 0.375 | 86.2 | (1.40) | 81.3 | A | (0.55) | 14.4 | (0.17) | 16.2 | A | (0.13) | |||
| 0.5 | 88.1 | (1.57) | 75.3 | AB | (0.74) | 15.4 | (0.22) | 14.4 | AB | (0.07) | |||
| 0.675 | 86.2 | (1.66) | 80.9 | A | (0.69) | 13.6 | (0.22) | 15.9 | AB | (0.09) | |||
| 0.75 | 84.8 | (1.10) | 75.5 | B | (1.49) | 15.3 | (0.38) | 14.7 | B | (0.55) | |||
| 1 | 79.5 | (0.69) | 72.7 | AB | (0.25) | 15.7 | (0.62) | 14.0 | AB | (0.15) | |||
| L. corniculatus Leo | 0.25 | 91.4 | AB | (2.25) | 78.8 | AB | (1.16) | 16.7 | AB | (0.24) | 15.3 | A | (0.19) |
| 0.375 | 88.0 | AC | (1.34) | 79.6 | A | (0.50) | 16.9 | A | (0.41) | 16.3 | AB | (0.08) | |
| 0.5 | 83.6 | CD | (1.28) | 76.7 | AB | (1.00) | 16.2 | AB | (0.18) | 14.9 | A | (0.18) | |
| 0.675 | 81.9 | ABCD | (2.03) | 75.7 | ABC | (1.46) | 14.7 | AB | (0.17) | 15.8 | AB | (0.22) | |
| 0.75 | 83.6 | jBD | (1.05) | 70.9 | kBC | (0.73) | 16.2 | B | (0.44) | 14.8 | AB | (0.72) | |
| 1 | 75.7 | BD | (0.69) | 63.8 | C | (2.23) | 16.5 | AB | (0.37) | 12.8 | B | (0.23) | |
| T. pratense | 0.25 | 87.1 | A | (1.84) | 86.5 | (0.87) | 16.0 | A | (0.34) | 17.1 | A | (0.28) | |
| 0.375 | 82.8 | ABC | (2.33) | 87.6 | (0.40) | 16.5 | A | (0.53) | 17.5 | AB | (0.37) | ||
| 0.5 | 82.9 | AB | (0.72) | 86.3 | (1.07) | 16.6 | AB | (0.27) | 17.5 | AB | (0.34) | ||
| 0.675 | 80.2 | ABC | (1.82) | 89.4 | (2.02) | 14.9 | AB | (0.15) | 18.3 | B | (0.35) | ||
| 0.75 | 80.1 | BC | (3.20) | 87.2 | (2.06) | 16.8 | B | (0.35) | 18.5 | B | (0.08) | ||
| 1 | 72.0 | C | (0.78) | 82.8 | (1.06) | 16.7 | AB | (0.62) | 17.3 | B | (0.20) | ||
| T. repens | 0.25 | 96.0 | A | (0.80) | 81.4 | AB | (0.67) | 17.1 | (0.03) | 16.4 | (0.37) | ||
| 0.375 | 82.9 | AB | (1.62) | 80.1 | AB | (0.23) | 16.5 | (0.44) | 16.4 | (0.41) | |||
| 0.5 | 89.1 | ABC | (0.55) | 75.6 | AC | (0.44) | 16.7 | (0.15) | 15.0 | (0.43) | |||
| 0.675 | 81.5 | ABC | (0.69) | 79.3 | AB | (1.04) | 15.3 | (0.08) | 16.6 | (0.22) | |||
| 0.75 | 77.9 | C | (1.16) | 73.5 | CD | (1.19) | 16.3 | (0.51) | 16.3 | (0.41) | |||
| 1 | 75.4 | BC | (0.90) | 69.8 | BD | (0.20) | 17.5 | (0.55) | 15.1 | (0.04) | |||
| P. lanceolata | 0.25 | 95.3 | A | (0.95) | 82.4 | (1.83) | 16.4 | ABCD | (0.11) | 16.2 | (0.57) | ||
| 0.375 | 87.0 | AB | (0.84) | 79.6 | (0.82) | 15.2 | AB | (0.10) | 15.8 | (0.20) | |||
| 0.5 | 87.5 | A | (0.82) | 82.6 | (0.53) | 16.5 | AC | (0.23) | 16.4 | (0.09) | |||
| 0.675 | 83.9 | AB | (1.54) | 79.5 | (1.14) | 14.3 | CD | (0.24) | 15.9 | (0.36) | |||
| 0.75 | 81.2 | AB | (1.40) | 77.5 | (2.36) | 15.4 | ABCD | (0.43) | 15.7 | (0.45) | |||
| 1 | 77.3 | B | (1.50) | 72.6 | (4.55) | 16.1 | BD | (0.37) | 13.7 | (0.75) | |||
| Species | Partner Prop. | Total Gas | Methane | ||||
|---|---|---|---|---|---|---|---|
| (mL/200 mg DOM 1) | |||||||
| L. corniculatus Lotanava | 0.25 | 90.7 | AB | (2.70) | 16.3 | AB | (0.42) |
| 0.375 | 87.8 | A | (1.59) | 16.7 | A | (0.42) | |
| 0.5 | 81.7 | BC | (1.18) | 15.6 | AB | (0.35) | |
| 0.675 | 82.7 | ABC | (1.51) | 14.6 | AB | (0.11) | |
| 0.75 | 80.7 | BC | (1.29) | 15.7 | B | (0.46) | |
| 1 | 71.9 | C | (2.46) | 15.8 | AB | (0.47) | |
| L. pedunculatus | 0.25 | 87.7 | AB | (3.70) | 15.5 | AB | (0.56) |
| 0.375 | 82.1 | AC | (0.96) | 14.1 | A | (0.06) | |
| 0.5 | 79.7 | ABCD | (0.29) | 14.9 | AB | (0.06) | |
| 0.675 | 73.4 | BD | (3.14) | 12.5 | B | (0.25) | |
| 0.75 | 71.7 | CD | (1.07) | 13.7 | AB | (0.38) | |
| 1 | 63.6 | D | (1.59) | 13.3 | AB | (0.52) | |
| S. minor | 0.25 | 89.3 | A | (1.17) | 15.4 | ABCD | (0.21) |
| 0.375 | 79.8 | A | (1.58) | 14.5 | AB | (0.38) | |
| 0.5 | 79.5 | AB | (1.96) | 15.0 | AC | (0.44) | |
| 0.675 | 72.8 | ABC | (2.32) | 11.9 | BD | (0.14) | |
| 0.75 | 71.8 | BC | (1.15) | 13.0 | CD | (0.34) | |
| 1 | 63.3 | C | (1.31) | 13.4 | BD | (0.68) | |
| Year | Species | Total Gas | Methane | Methane/Total Gas | |||
|---|---|---|---|---|---|---|---|
| Adjusted R2 | |||||||
| 2018 | C. carvi | 0.19 | ** | −0.02 | ns | 0.12 | * |
| C. intybus | 0.49 | *** | 0.00 | ns | 0.31 | *** | |
| L. corniculatus Leo | 0.52 | *** | 0.00 | ns | 0.26 | *** | |
| L. corniculatus Lotanava | 0.48 | *** | 0.03 | ns | 0.28 | *** | |
| L. pedunculatus | 0.72 | *** | 0.12 | * | 0.46 | *** | |
| T. pratense | 0.33 | *** | −0.02 | ns | 0.48 | *** | |
| T. repens | 0.46 | *** | −0.02 | ns | 0.47 | *** | |
| P. lanceolata | 0.59 | *** | 0.00 | ns | 0.39 | *** | |
| S. minor | 0.74 | *** | 0.22 | ** | 0.20 | ** | |
| 2019 | C. carvi | 0.23 | ** | 0.09 | ns | −0.03 | ns |
| C. intybus | 0.40 | *** | 0.34 | *** | 0.01 | ns | |
| L. corniculatus Leo | 0.74 | *** | 0.39 | *** | 0.00 | ns | |
| T. pratense | −0.01 | ns | 0.11 | ns | 0.23 | ** | |
| T. repens | 0.69 | *** | 0.02 | ns | 0.27 | ** | |
| P. lanceolata | 0.25 | ** | 0.17 | ** | −0.03 | ns | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loza, C.; Verma, S.; Wolffram, S.; Susenbeth, A.; Blank, R.; Taube, F.; Loges, R.; Hasler, M.; Kluß, C.; Malisch, C.S. Assessing the Potential of Diverse Forage Mixtures to Reduce Enteric Methane Emissions In Vitro. Animals 2021, 11, 1126. https://doi.org/10.3390/ani11041126
Loza C, Verma S, Wolffram S, Susenbeth A, Blank R, Taube F, Loges R, Hasler M, Kluß C, Malisch CS. Assessing the Potential of Diverse Forage Mixtures to Reduce Enteric Methane Emissions In Vitro. Animals. 2021; 11(4):1126. https://doi.org/10.3390/ani11041126
Chicago/Turabian StyleLoza, Cecilia, Supriya Verma, Siegfried Wolffram, Andreas Susenbeth, Ralf Blank, Friedhelm Taube, Ralf Loges, Mario Hasler, Christof Kluß, and Carsten Stefan Malisch. 2021. "Assessing the Potential of Diverse Forage Mixtures to Reduce Enteric Methane Emissions In Vitro" Animals 11, no. 4: 1126. https://doi.org/10.3390/ani11041126
APA StyleLoza, C., Verma, S., Wolffram, S., Susenbeth, A., Blank, R., Taube, F., Loges, R., Hasler, M., Kluß, C., & Malisch, C. S. (2021). Assessing the Potential of Diverse Forage Mixtures to Reduce Enteric Methane Emissions In Vitro. Animals, 11(4), 1126. https://doi.org/10.3390/ani11041126










