Analysis of Morphokinetic Parameters of Feline Embryos Using a Time-Lapse System
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Source of Ovaries and Oocyte Collection
2.2. In Vitro Maturation (IVM) of Oocytes
2.3. In Vitro Fertilization (IVF)
2.4. Embryo Culture in the Time-Lapse System (Primo Vision)
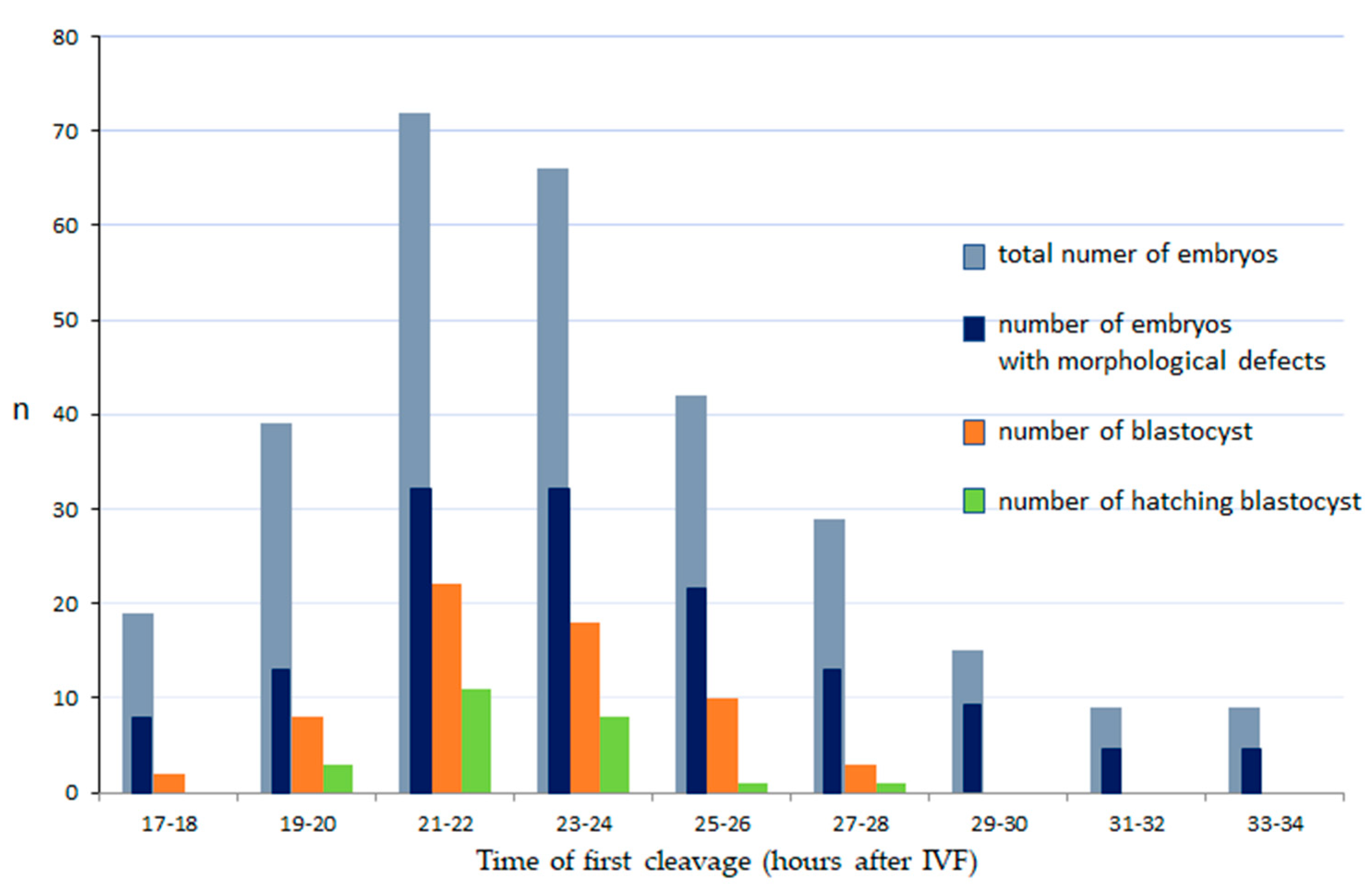
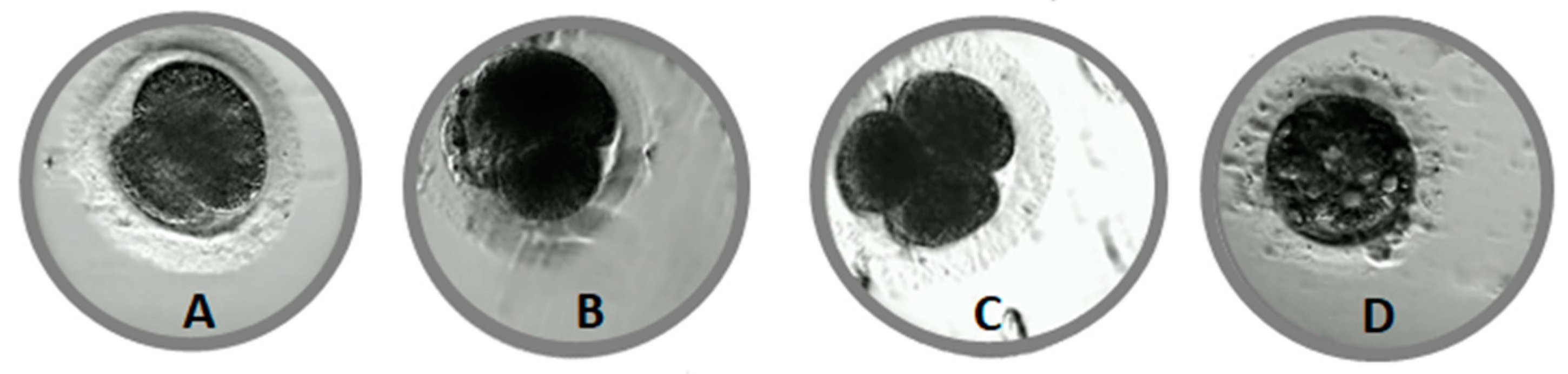
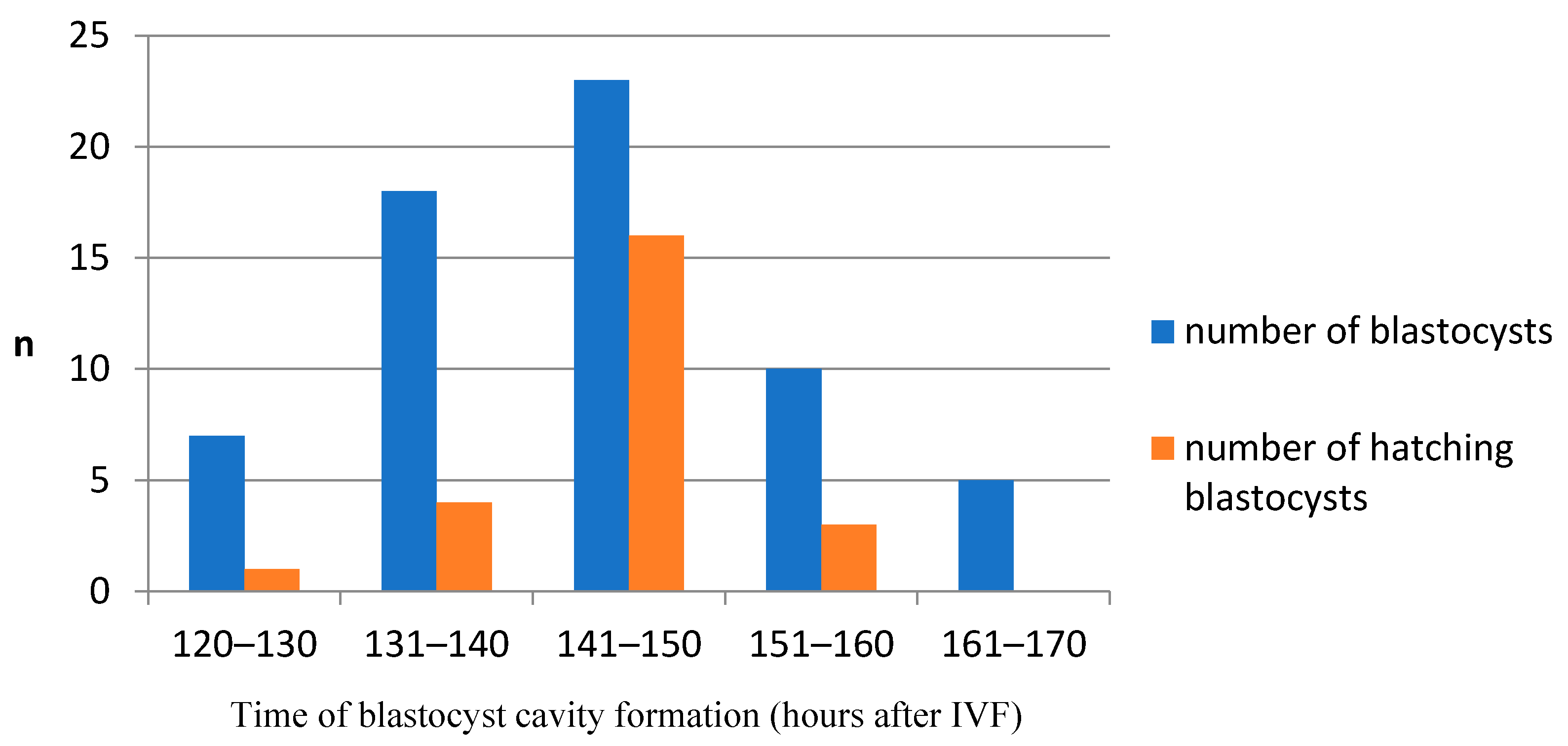
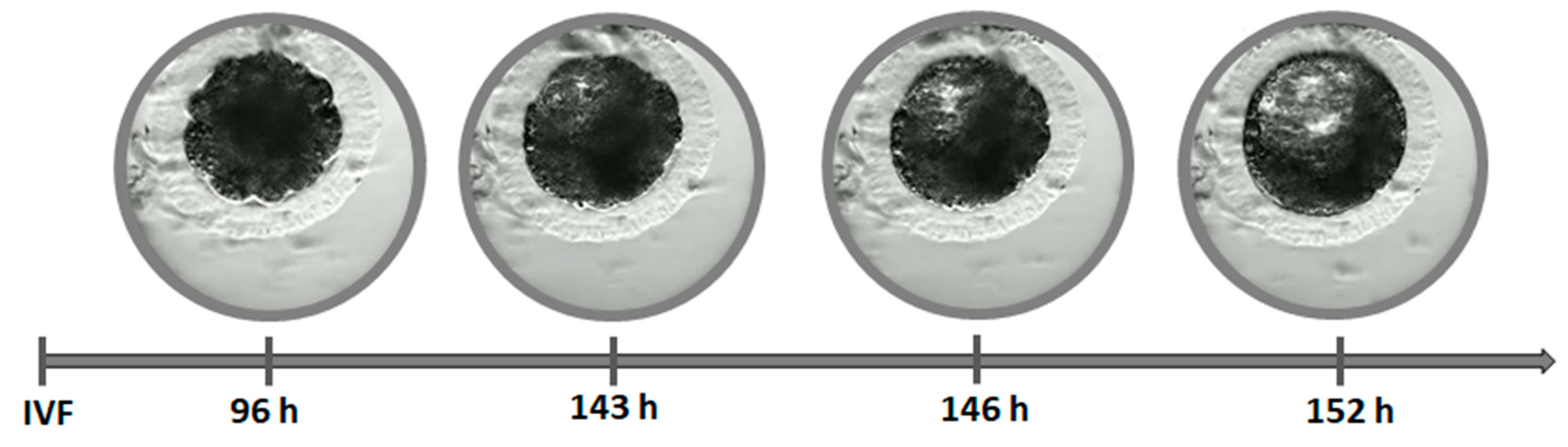
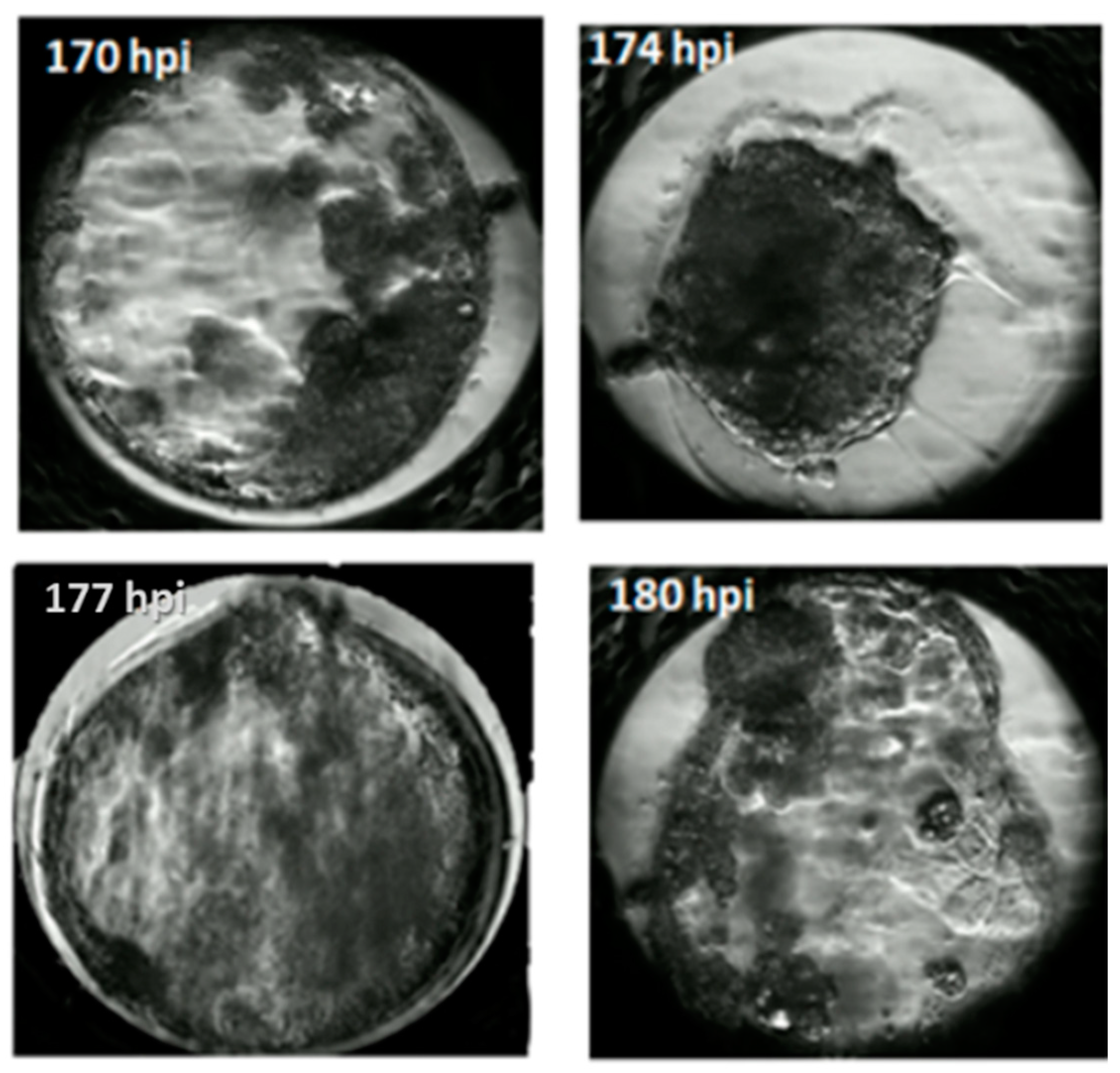
2.5. Embryo Evaluation
- t2, t3, t4—The timing of division to the 2, 3 and 4 blastomere stage
- BC—formation of the blastocyst cavity
- BL—expanded blastocyst
- BH—hatching blastocyst
- N—normal
- AS—asymmetry of blastomeres
- FR—cellular fragmentation
- DC—direct cleavage from one to three or more blastomeres
- RC—reverse cleavage (blastomere fusion after division)
- VAC—vacuoles in blastomeres
- MUL—presence of two or more morphological defects.
- Proportions of late embryo stages (blastocyst, hatching blastocysts) were calculated as a function of the total number of cleaved embryos.
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Pope, C.E. Aspects of in vivo oocyte production, blastocyst development and embryo transfer in the cat. Theriogenology 2014, 81, 126–137. [Google Scholar] [CrossRef]
- Kochan, J.; Nowak, A.; Młodawska, W.; Prochowska, S.; Partyka, A.; Skotnicki, J.; Niżański, W. Comparison of the morphology and developmental potential of oocytes obtained from prepubertal and adult domestic and wild cats. Animals 2021, 11, 1–11. [Google Scholar]
- Fernandez-Gonzalez, L.; Hribal, R.; Stagegaard, J.; Zahmel, J.; Jewgenow, K. Production of lion (Panthera leo) blastocysts after in vitro maturation of oocytes and intracytoplasmic sperm injection. Theriogenology 2015, 83, 995–999. [Google Scholar] [CrossRef]
- Goodrowe, K.L.; Wall, R.J.; O’Brien, S.J.; Schmidt, P.M.; Wildt, D.E. Developmental competence of domestic cat follicular oocytes after fertilization in vitro. Biol. Reprod. 1988, 39, 355–372. [Google Scholar] [CrossRef]
- Freistedt, P.; Stojkovic, M.; Wolf, E.; March, J.; Ii, A.J. Efficient in vitro production of cat embryos in modified synthetic oviduct fluid medium: Effects of season and ovarian status effect of sperm treatment of cat embryos. Biol. Reprod. 2001, 65, 9–13. [Google Scholar] [CrossRef]
- Gómez, M.C.; Pope, E.; Harris, R.; Mikota, S.; Dresser, B.L. Development of in vitro matured, in vitro fertilized domestic cat embryos following cryopreservation, culture and transfer. Theriogenology 2003, 60, 239–251. [Google Scholar] [CrossRef]
- Thongkittidilok, C.; Tharasanit, T.; Songsasen, N.; Sananmuang, T.; Buarpung, S.; Techakumphu, M. Epidermal growth factor improves developmental competence and embryonic quality of singly cultured domestic cat embryos. J. Reprod. Dev. 2015, 61, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Sananmuang, T.; Tharasanit, T.; Nguyen, C.; Phutikanit, N.; Techakumphu, M. Culture medium and embryo density influence on developmental competence and gene expression of cat embryos. Theriogenology 2011, 75, 1708–1719. [Google Scholar] [CrossRef] [PubMed]
- Hribal, R.; Braun, B.C.; Ringleb, J.; Jewgenow, K. Capabilities and challenges of examination of gene expression for quality assessment of domestic cat embryos. Reprod. Domest. Anim. 2012, 47, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Moro, L.N.; Sestelo, A.J.; Salamone, D.F. Evaluation of cheetah and leopard spermatozoa developmental capability after interspecific ICSI with domestic cat oocytes. Reprod. Domest. Anim. 2014, 49, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Ochota, M.; Wojtasik, B.; Niżański, W. Total cell number and its allocation to trophectoderm and inner cell mass in vitro obtained cats’ blastocysts. Reprod. Domest. Anim. 2016, 51, 339–345. [Google Scholar] [CrossRef]
- Ochota, M.; Niżański, W. Time of early cleavage affects the developmental potential of feline preimplantation embryos in vitro. Theriogenology 2017, 89, 26–31. [Google Scholar] [CrossRef]
- Ziebe, S.; Petersen, K.; Lindenberg, S.; Andersen, A.G.; Gabrielsen, A.; Andersen, A.N. Embryo morphology or cleavage stage: How to select the best embryos for transfer after in-vitro fertilization. Hum. Reprod. 1997, 12, 1545–1549. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.C.; Loewke, K.E.; Bossert, N.L.; Behr, B.; De Jonge, C.J.; Baer, T.M.; Reijo Pera, R.A. Non-invasive imaging of human embryos before embryonic genome activation predicts development to the blastocyst stage. Nat. Biotechnol. 2010, 28, 1115–1121. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, D.S.; Boatman, D.E.; Bavister, B.D. Kinematics of trophectoderm projections and locomotion in the peri-implantation hamster blastocyst. Dev. Dyn. 1996, 205, 435–444. [Google Scholar] [CrossRef]
- Sugimura, S.; Akai, T.; Imai, K. Selection of viable in vitro-fertilized bovine embryos using time-lapse monitoring in microwell culture dishes. J. Reprod. Dev. 2017, 63, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Sciorio, R.; Thong, J.K.; Pickering, S.J. Comparison of the development of human embryos cultured in either an EmbryoScope or benchtop inkubator. J. Assist. Reprod. Genet. 2017, 15, 1100–1106. [Google Scholar]
- Truong, T.; Gardner, D.K. Antioxidants improve IVF outcome and subsequent embryo development in the mouse. Hum. Reprod. 2017, 32, 2404–2413. [Google Scholar] [CrossRef]
- Fenwick, J.; Platteau, P.; Murdoch, A.P.; Herbert, M. Time from insemination to first cleavage predicts developmental competence of human preimplantation embryos in vitro. Hum. Reprod. 2002, 17, 407–412. [Google Scholar] [CrossRef]
- Leoni, G.G.; Succu, S.; Berlinguer, F.; Rosati, I.; Bebbere, D.; Bogliolo, L.; Ledda, S.; Naitana, S. Delay on the in vitro kineticdevelopment of prepubertal ovine embryos. Anim. Reprod. Sci. 2006, 92, 373–383. [Google Scholar] [CrossRef]
- Klincumhom, N.; Thongphakdee, A.; Techakumphu, M.; Chatdarong, K. Time of the first embryonic cleavage indicates cat blastocyst quality. Thai J. Vet. Med. 2012, 42, 67–72. [Google Scholar]
- Kij, B.; Kochan, J.; Nowak, A.; Niżański, W.; Prochowska, S.; Fry, K.; Bugno-Poniewierska, M. Using time lapse monitoring for determination of morphological defect frequency in feline embryos after in vitro fertilization (IVF). Animals 2020, 10, 3. [Google Scholar] [CrossRef]
- Kochan, J.; Nowak, A.; Niżański, W.; Prochowska, S.; Migdał, A.; Młodawska, W.; Partyka, A.; Witkowski, M. Developmental competence of cat (Felis domesticus) oocytes and embryos after parthenogenetic stimulation using different methods. Zygote 2018, 22, 1–8. [Google Scholar] [CrossRef]
- Nizanski, W.; Dejneka, G.J.; Klimowicz, M.; Dubiel, A. Evaluating some properties of domestic cat epididymal spermatozoa and their cryopreservation. Med. Weter. 2005, 61, 173–178. [Google Scholar]
- Dinnyés, A.; Lonergan, P.; Fair, T.; Boland, M.P.; Yang, X. Timing of the first cleavage posrfertilization affects cryosurvival of in vitro produced bovine blastocysts. Mol. Reprod. Dev. 1999, 53, 318–324. [Google Scholar] [CrossRef]
- Lundin, K.; Bergh, C.; Hardarson, T. Earlyembryo cleavage is a strong indicator of embryo quality in human IVF. Hum. Reprod. 2001, 16, 2652–2657. [Google Scholar] [CrossRef] [PubMed]
- Fryc, K.; Nowak, A.; Kij, B.; Kochan, J.; Bartlewski, P.M.; Murawski, M. Timing of cleavage divisions determined with time-lapse imaging is linked to blastocyst formation rates and quality of in vitro-produced ovine embryos. Theriogenology 2021, 159, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chapple, V.; Roberts, P.; Matson, P. Prevalence, consequence, and significance of reverse cleavage by human embryos viewed with the use of the Embryoscope time-lapse video system. Fertil. Steril. 2014, 102, 1295–1300. [Google Scholar] [CrossRef] [PubMed]
- Shiao-Hsuan, Y.; Cheng-Hsuan, W.; Yu-Ching, C.; Chueh-Ko, Y.; Tsui-Huang, W.; Pei-Chi, C.; Horng-Der, T. Effect of morphokinetics and morphological dynamics of cleavagestage on embryo developmental potential: A time-lapse study. Taiwan. J. Obstet. Gynecol. 2018, 57, 76–82. [Google Scholar]
- Spindler, R.; Crichton, E.; Agca, Y.; Loskutoff, N.; Critser, J.; Gardner, D.; Wildt, D. Improved felid embryo development by group culture is maintained with heterospecific companions. Theriogenology 2006, 66, 82–92. [Google Scholar] [CrossRef]
- Mizobe, Y.; Tokunaga, M.; Oya, N.; Iwakiri, R.; Yoshida, N.; Sato, Y.; Onoue, N.; Ezono, Y. Synchrony of the first division as an index of the blastocyst formation rate during embryonic development. Reprod. Med. Biol. 2017, 17, 64–70. [Google Scholar] [CrossRef]
- Cummins, J.M.; Breen, T.M.; Harrison, K.L.; Shaw, J.M.; Wilson, L.M.; Hennessey, J.F. A formula for scoring human embryo growth rates in in vitro fertilization: Its value in predicting pregnancy and in comparison with visual estimates of embryo quality. J. Vitro. Fert. Embryo Transf. 1986, 3, 284–295. [Google Scholar] [CrossRef] [PubMed]
- Rubio, I.; Kuhlmann, R.; Agerholm, I.; Kirk, J.; Herrero, J.; Escriba, M.J. Limited implantation success of direct-cleaved human zygotes: A time-lapse study. Fertil. Steril. 2012, 98, 1458–1463. [Google Scholar] [CrossRef]
- Stigliani, S.; Anserini, P.; Venturini, P.L.; Scaruffi, P. Mitochondrial DNA content in embryo culture medium is significantly associated with human embryo fragmentation. Hum. Reprod. 2013, 28, 2652–2660. [Google Scholar] [CrossRef] [PubMed]
- Jurisicova, A.; Varmuza, S.; Casper, R.F. Programmed cell death and human embryofragmentation. Mol. Hum. Reprod. 1996, 2, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Chatzimeletiou, K.; Morrison, E.E.; Prapas, N.; Prapas, Y.; Handyside, A.H. Spindle abnormalities in normally developing and arrested human preimplantation embryos in vitro identified by confocal laser scanning microscopy. Hum. Reprod. 2005, 20, 672–682. [Google Scholar] [CrossRef]
- Somfai, T.; Inaba, Y.; Aikawa, Y.; Ohtake, M.; Kobayashi, S.; Konishi, K. Relationship between the length of cell cycles, cleavage pattern and development al competence in bovine embryos generated by in vitro fertilization or parthenogenesis. J. Reprod. Dev. 2010, 56, 200–207. [Google Scholar] [CrossRef]
- Hickman, C.F.L.; Campbell, A.; Duffy, S.; Fishel, S. Reverse cleavage: Its significance with regards to human embryo morphokinetics ploidy and stimulation protocol. Hum. Reprod. 2012, 27 (Suppl. 2), 103–105. [Google Scholar]
- Ebner, T.; Moser, M.; Tews, G. Is oocyte morphology prognostic of embryo developmental potential after ICSI? Reprod. Biomed. Online 2006, 12, 507–512. [Google Scholar] [CrossRef]
- Rienzi, L.; Ubaldi, F.M.; Lacobelli, M.; Minasi, M.G.; Romano, S.; Ferrero, S.; Sapienza, F.; Baroni, E.; Litwicka, K.; Greco, E. Significance of metaphase II human oocyte morphology on ICSI outcome. Fertil. Steril. 2008, 90, 1692–1700. [Google Scholar] [CrossRef]
- Fancsovits, P.; Murber, A.; Gilan, Z.; Rigo, J.; Urbancsek, J. Human oocytes containing large cytoplasmic vacuoles can result in pregnancy and viable offspring. Reprod. Biomed. Online 2011, 23, 513–516. [Google Scholar] [CrossRef] [PubMed]
| Morphology of Embryos | N (%) |
|---|---|
| Total number of embryos | 300 (100) |
| Normal | 156 (52) |
| Abnormal | 144 (48) |
| MUL—multiple defects | 81 (27) |
| FGR—cellular fragmentation | 24 (8) |
| AS—asymmetry of blastomeres | 18 (6) |
| DC—direct cleavage | 9 (3) |
| RC—reverse cleavage | 6 (2) |
| VAC—vacuoles in blastomeres | 6 (2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kochan, J.; Nowak, A.; Kij, B.; Prochowska, S.; Niżański, W. Analysis of Morphokinetic Parameters of Feline Embryos Using a Time-Lapse System. Animals 2021, 11, 748. https://doi.org/10.3390/ani11030748
Kochan J, Nowak A, Kij B, Prochowska S, Niżański W. Analysis of Morphokinetic Parameters of Feline Embryos Using a Time-Lapse System. Animals. 2021; 11(3):748. https://doi.org/10.3390/ani11030748
Chicago/Turabian StyleKochan, Joanna, Agnieszka Nowak, Barbara Kij, Sylwia Prochowska, and Wojciech Niżański. 2021. "Analysis of Morphokinetic Parameters of Feline Embryos Using a Time-Lapse System" Animals 11, no. 3: 748. https://doi.org/10.3390/ani11030748
APA StyleKochan, J., Nowak, A., Kij, B., Prochowska, S., & Niżański, W. (2021). Analysis of Morphokinetic Parameters of Feline Embryos Using a Time-Lapse System. Animals, 11(3), 748. https://doi.org/10.3390/ani11030748








