Systematic Analysis of Breed, Methodological, and Geographical Impact on Equine Sperm Progressive Motility
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
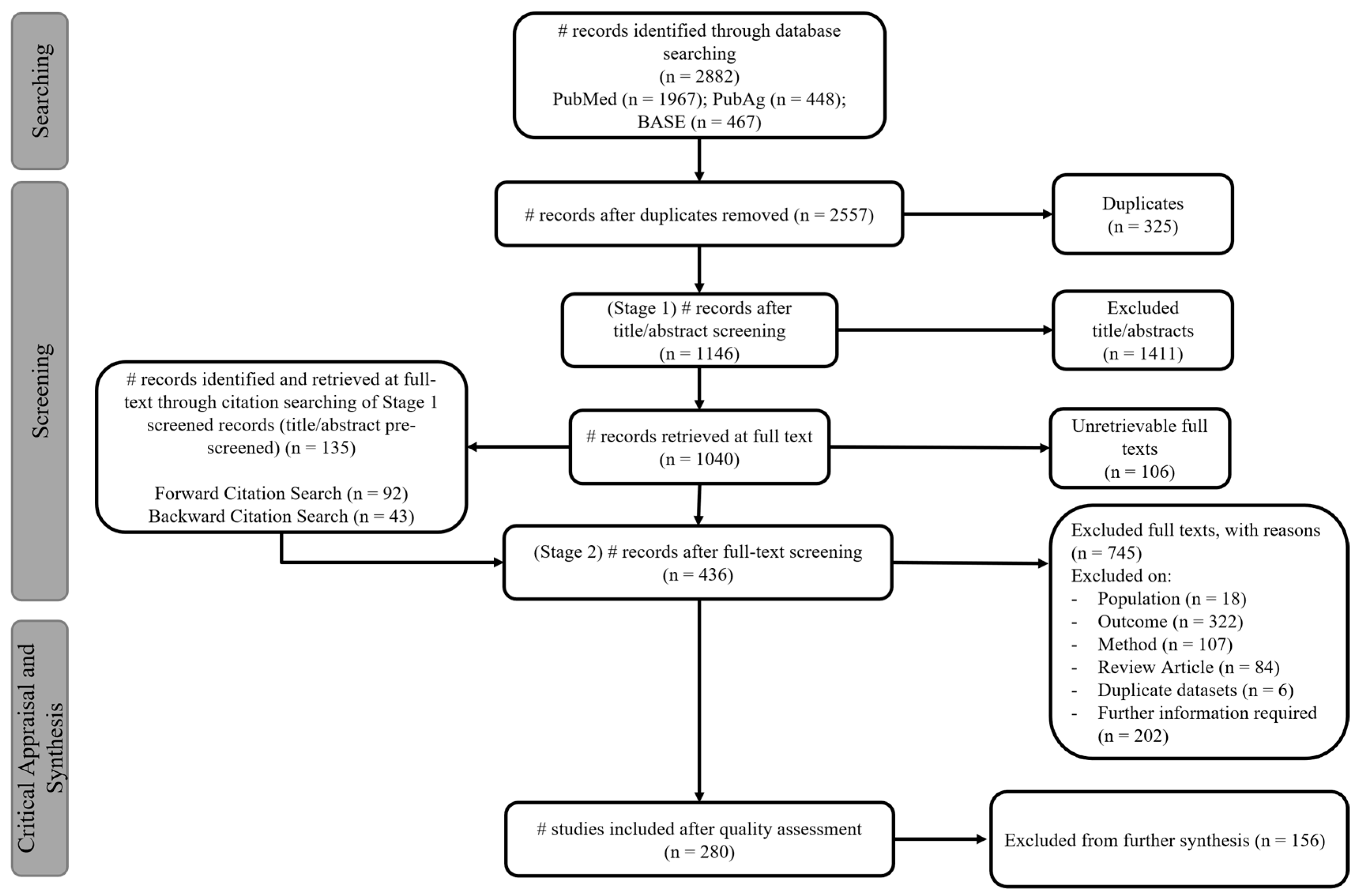
2.2. Systematic Review
2.2.1. Eligibility Criteria
2.2.2. Article Screening
2.3. Quality Assessment
2.4. Data Extraction
2.5. Statistical Analysis
3. Results
3.1. Year of Semen Collection
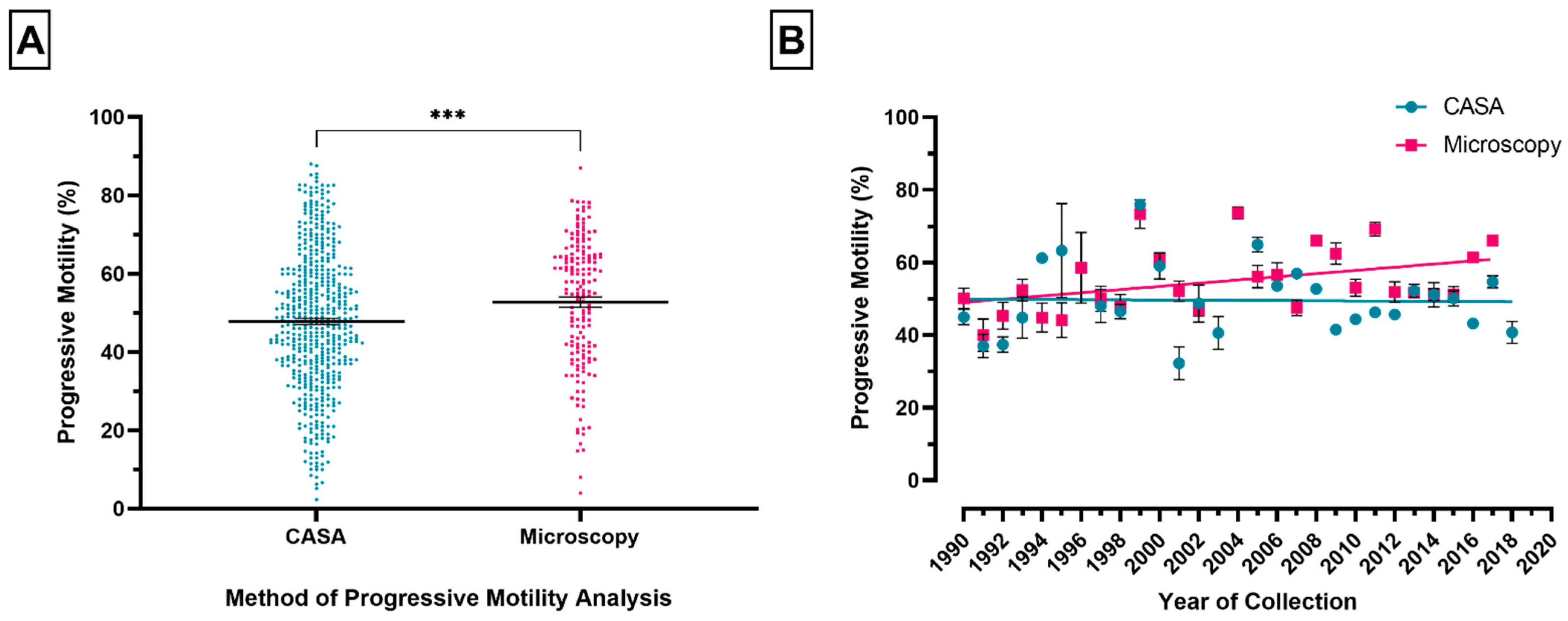
3.2. Method of Sperm Motility Assessment
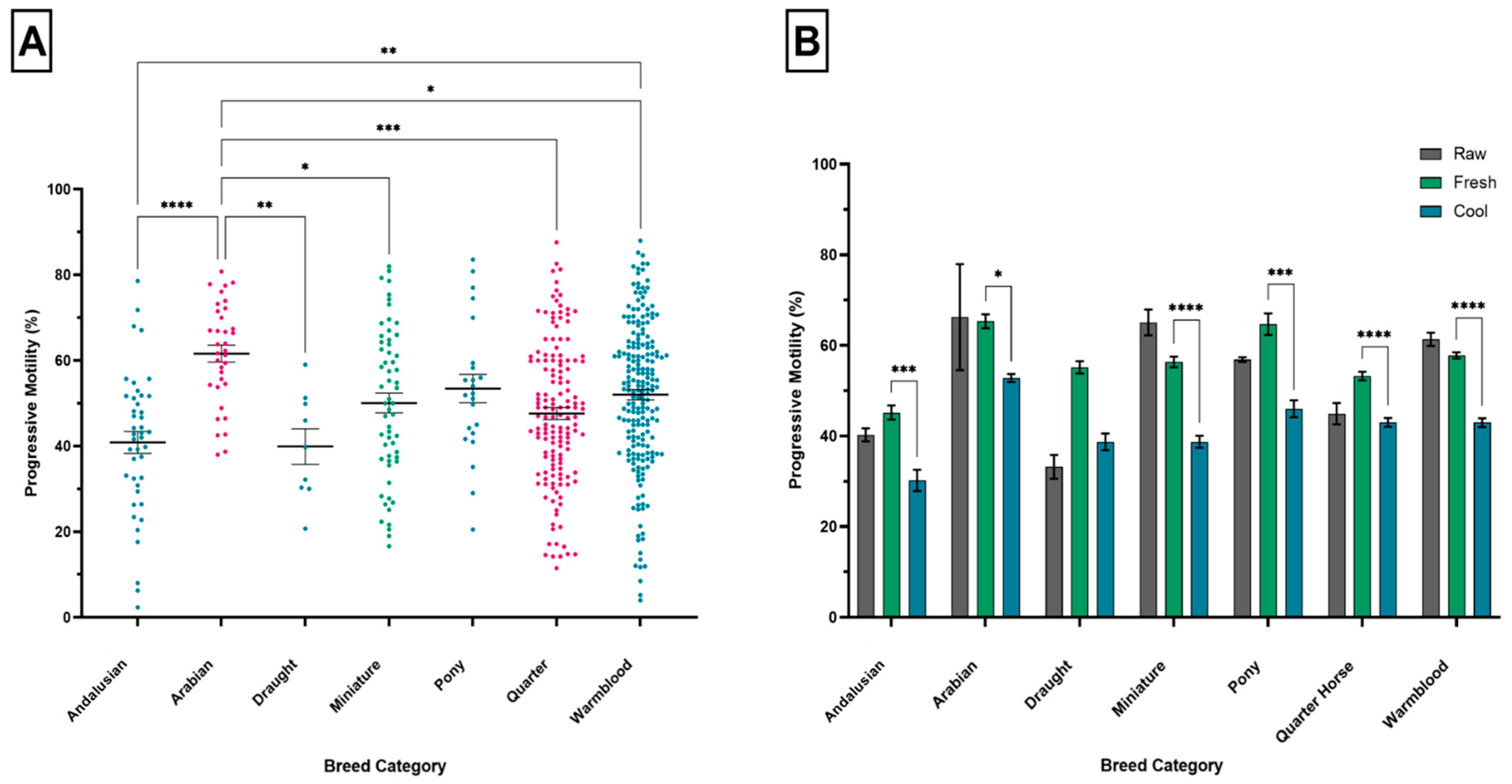
3.3. Breed Category
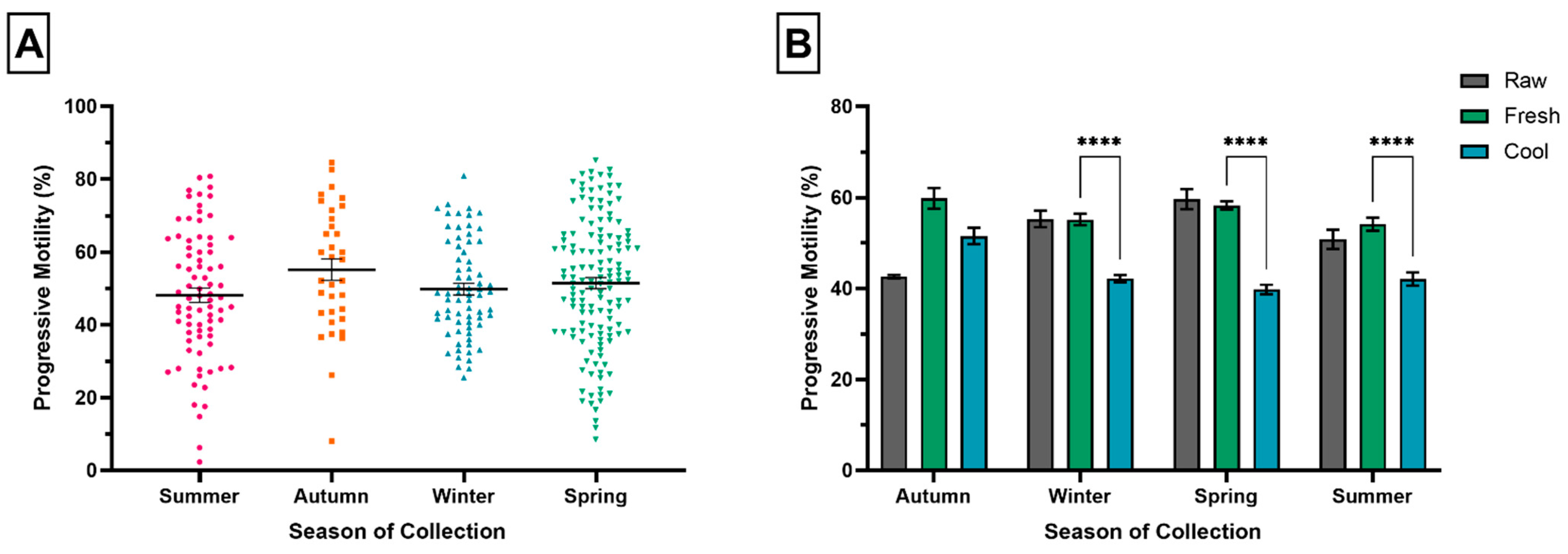
3.4. Season of Semen Collection
3.5. Geographical Location
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organisation. WHO Laboratory Manual for the Examination and Processing of Human Semen, 5th ed.; World Health Organisation: Geneva, Switzerland, 2010; ISBN 978-92-4-154778-9. [Google Scholar]
- Wang, C.; Swerdloff, R.S. Limitations of semen analysis as a test of male fertility and anticipated needs from newer tests. Fertil. Steril. 2014, 102, 1502–1507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carlsen, E.; Giwercman, A.; Keiding, N.; Skakkebaek, N.E. Evidence for decreasing quality of semen during past 50 years. Br. Med. J. 1992, 48, 200–202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wahl, R.L.; Reif, J.S. Temporal trends in bull semen quality: A comparative model for human health? Environ. Res. 2009, 109, 273–280. [Google Scholar] [CrossRef]
- Lea, R.G.; Byers, A.S.; Sumner, R.N.; Rhind, S.M.; Zhang, Z.; Freeman, S.L.; Moxon, R.; Richardson, H.M.; Green, M.; Craigon, J.; et al. Environmental chemicals impact dog semen quality in vitro and may be associated with a temporal decline in sperm motility and increased cryptorchidism. Sci. Rep. 2016, 6, 31281. [Google Scholar] [CrossRef] [PubMed]
- Levine, H.; Jørgensen, N.; Martino-Andrade, A.; Mendiola, J.; Weksler-Derri, D.; Mindlis, I.; Pinotti, R.; Swan, S.H. Temporal trends in sperm count: A systematic review and meta-regression analysis. Hum. Reprod. Update 2017, 23, 646–659. [Google Scholar] [CrossRef] [PubMed]
- Serrano, T.; Chevrier, C.; Multigner, L.; Cordier, S.; Jégou, B. International geographic correlation study of the prevalence of disorders of male reproductive health. Hum. Reprod. 2013, 28, 1974–1986. [Google Scholar] [CrossRef] [Green Version]
- Olsen, G.W.; Bodner, K.M.; Ramlow, J.M.; Ross, C.E.; Lipshultz, L.I. Have sperm counts been reduced 50 percent in 50 years? A statistical model revisited. Fertil. Steril. 1995, 63, 887–893. [Google Scholar] [CrossRef]
- Cooper, T.G.; Handelsman, D.J. Falling sperm counts and global oestrogenic pollution: Postscript. Asian J. Androl. 2013, 15, 208–211. [Google Scholar] [CrossRef] [Green Version]
- Pacey, A. Are sperm counts declining? or did we just change our spectacles? Asian J. Androl. 2013, 15, 187–190. [Google Scholar] [CrossRef] [Green Version]
- Swan, S.H.; Elkin, E.F.; Fenster, L. Have sperm densities declined? A reanalysis of global trend data. Environ. Health Perspect. 1997, 105, 1228–1232. [Google Scholar] [CrossRef]
- Swan, S.H.; Elkin, E.P.; Fenster, L. The question of declining sperm density revisited: An analysis of 101 studies published 1934-1996. Environ. Health Perspect. 2000, 108, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Mendiola, J.; Jørgensen, N.; Mínguez-Alarcón, L.; Sarabia-Cos, L.; López-Espín, J.J.; Vivero-Salmerón, G.; Ruiz-Ruiz, K.J.; Fernández, M.F.; Olea, N.; Swan, S.H.; et al. Sperm counts may have declined in young university students in Southern Spain. Andrology 2013, 1, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Le Moal, J.; Rolland, M.; Goria, S.; Wagner, V.; De Crouy-, P.; Santé, C.; France, P. Semen quality trends in French regions are consistent with a global change in environmental exposure. Reproduction 2014, 147, 567–574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jørgensen, N.; Vierula, M.; Jacobsen, R.; Pukkala, E.; Perheentupa, A.; Virtanen, H.E.; Skakkebæk, N.E.; Toppari, J. Recent adverse trends in semen quality and testis cancer incidence among Finnish men. Int. J. Androl. 2011, 34, e37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Multigner, L.; Magistrini, M.; Ducot, B.; Spira, A. Secular sperm trends in stallions between 1981 and 1996. J. Androl. 1999, 20, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Multigner, L.; Magistrini, M.; Ducot, B.; Spira, A. Environnement et declin de la qualite du sperme. Evolution chez l’etalon au cours des deux dernieres decennies. Rev. Epidemiol. Sante Publique 2000, 48 (Suppl. 2), 2S72–2S78. [Google Scholar]
- Lemazurier, E.; Moslemi, S.; Sourdaine, P.; Desjardins, I.; Plainfosse, B.; Seralini, G.-E. Free and conjugated estrogens and androgens in stallion semen. Gen. Comp. Endocrinol. 2002, 125, 272–282. [Google Scholar] [CrossRef]
- Colenbrander, B.; Gadella, B.; Stout, T. The predictive value of semen analysis in the evaluation of stallion fertility. Reprod. Domest. Anim. 2003, 38, 305–311. [Google Scholar] [CrossRef]
- Hinrichs, K. Assisted reproductive techniques in mares. Reprod. Domest. Anim. 2018, 53, 4–13. [Google Scholar] [CrossRef]
- Claes, A.; Cuervo-Arango, J.; van den Broek, J.; Galli, C.; Colleoni, S.; Lazzari, G.; Deelen, C.; Beitsma, M.; Stout, T.A. Factors affecting the likelihood of pregnancy and embryonic loss after transfer of cryopreserved in vitro produced equine embryos. Equine Vet. J. 2019, 51, 446–450. [Google Scholar] [CrossRef]
- Squires, E. Current Reproductive Technologies Impacting Equine Embryo Production. J. Equine Vet. Sci. 2020, 89, 102981. [Google Scholar] [CrossRef] [PubMed]
- Griffin, R.A.; Swegen, A.; Baker, M.; Aitken, R.J.; Skerrett-Byrne, D.A.; Rodriguez, A.S.; Martín-Cano, F.E.; Nixon, B.; Peña, F.J.; Delehedde, M.; et al. Mass spectrometry reveals distinct proteomic profiles in high- And low-quality stallion spermatozoa. Reproduction 2020, 160, 695–707. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S. Meta-analysis of Observational Studies in Epidemiology: A Proposal for Reporting. J. Am. Med. Assoc. 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.; Welch, V. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions, Version 6.2, Cochrane, Online. 2021. Available online: www.training.cochrane.org/handbook (accessed on 22 October 2021).
- Vandenbroucke, J.P.; von Elm, E.; Altman, D.G.; Gøtzsche, P.C.; Mulrow, C.D.; Pocock, S.J.; Poole, C.; Schlesselman, J.J.; Egger, M.; Blettner, M.; et al. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. Int. J. Surg. 2014, 12, 1500–1524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Houghton, C.; Murphy, K.; Meehan, B.; Thomas, J.; Brooker, D.; Casey, D. From screening to synthesis: Using NVivo to enhance transparency in qualitative evidence synthesis. J. Clin. Nurs. 2017, 26, 873–881. [Google Scholar] [CrossRef]
- Gottschalk, M.; Sieme, H.; Martinsson, G.; Distl, O. Analysis of breed effects on semen traits in light horse, warmblood, and draught horse breeds. Theriogenology 2016, 85, 1375–1381. [Google Scholar] [CrossRef]
- Ghasemi, A.; Zahediasl, S. Normality tests for statistical analysis: A guide for non-statisticians. Int. J. Endocrinol. Metab. 2012, 10, 486–489. [Google Scholar] [CrossRef] [Green Version]
- Jasko, D.J.; Little, T.V.; Lein, D.H.; Foote, R.H. Comparison of spermatozoal movement and semen characteristics with fertility in stallions: 64 cases (1987–1988). J. Am. Vet. Med. Assoc. 1992, 200, 979–985. [Google Scholar]
- Love, C.C.; Noble, J.K.; Standridge, S.A.; Bearden, C.T.; Blanchard, T.L.; Varner, D.D.; Cavinder, C.A. The relationship between sperm quality in cool-shipped semen and embryo recovery rate in horses. Theriogenology 2015, 84, 1587–1593.e4. [Google Scholar] [CrossRef]
- Broekhuijse, M.L.W.J.; Šostarić, E.; Feitsma, H.; Gadella, B.M. Additional value of computer assisted semen analysis (CASA) compared to conventional motility assessments in pig artificial insemination. Theriogenology 2011, 76, 1473–1486.e1. [Google Scholar] [CrossRef]
- Proctor, J.G.; William, M.H.A.; Boone, R.; Lee, H.; Iii, H.; Boone, W.R. Comparison of the Manual, IVOS, and SCA Methods for Semen Analysis Reporting. J. Clin. Embryol. 2009, 12, 5–7. [Google Scholar]
- Iguer-ouada, M.; Verstegen, J.P. Evaluation of the ‘Hamilton thorn computer-based automated system’ for dog semen analysis. Theriogenology 2001, 55, 733–749. [Google Scholar] [CrossRef]
- Jasko, D.; Moran, D.; Farlin, M.; Squires, E. Effect of seminal plasma dilution or removal on spermatozoal motion characteristics of cooled stallion semen. Theriogenology 1991, 35, 1059–1068. [Google Scholar] [CrossRef]
- Brinsko, S.P.; Rowan, K.R.; Varner, D.D.; Blanchard, T.L. Effects of transport container and ambient storage temperature on motion characteristics of equine spermatozoa. Theriogenology 2000, 53, 1641–1655. [Google Scholar] [CrossRef]
- Gottschalk, M.; Metzger, J.; Martinsson, G.; Sieme, H.; Distl, O. Genome-wide association study for semen quality traits in German Warmblood stallions. Anim. Reprod. Sci. 2016, 171, 81–86. [Google Scholar] [CrossRef]
- Gottschalk, M.; Sieme, H.; Martinsson, G.; Distl, O. Heritability of semen traits in German Warmblood stallions. Anim. Reprod. Sci. 2016, 170, 10–14. [Google Scholar] [CrossRef]
- Dowsett, K.F.; Knott, L.M. The influence of age and breed on stallion semen. Theriogenology 1996, 46, 397–412. [Google Scholar] [CrossRef]
- Krakowski, L.; Obara, J.; Wąchocka, A.; Piech, T.; Bartoszek, P.; Kostro, K.; Tatara, M.R. Assessment of extent of apoptosis and DNA defragmentation in chilled semen of stallions during the breeding season. Reprod. Domest. Anim. 2013, 48, 826–832. [Google Scholar] [CrossRef]
- Gurgul, A.; Jasielczuk, I.; Semik-Gurgul, E.; Pawlina-Tyszko, K.; Stefaniuk-Szmukier, M.; Szmatoła, T.; Polak, G.; Tomczyk-Wrona, I.; Bugno-Poniewierska, M. A genome-wide scan for diversifying selection signatures in selected horse breeds. PLoS ONE 2019, 14, e0210751. [Google Scholar] [CrossRef] [Green Version]
- Gómez, M.D.; Sánchez, M.J.; Bartolomé, E.; Cervantes, I.; Poyato-Bonilla, J.; Demyda-Peyrás, S.; Valera, M. Phenotypic and genetic analysis of reproductive traits in horse populations with different breeding purposes. Animal 2020, 14, 1351–1361. [Google Scholar] [CrossRef]
- Splingart, C.; Frapsauce, C.; Veau, S.; Barthélémy, C.; Royère, D.; Guérif, F. Semen variation in a population of fertile donors: Evaluation in a French centre over a 34-year period. Int. J. Androl. 2012, 35, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Nazem, T.G.; Gounko, D.; Lee, J.; Bar-Chama, N.; Shamonki, J.M.; Antonelli, C.; Copperman, A.B. Eleven year longitudinal study of U.S. sperm donors demonstrates declining sperm count and motility. Fertil. Steril. 2018, 110, e54–e55. [Google Scholar] [CrossRef]
- Swan, S.H.; Brazil, C.; Drobnis, E.Z.; Liu, F.; Kruse, R.L.; Hatch, M.; Redmon, J.B.; Wang, C.; Overstreet, J.W. Geographic Differences in Semen Quality of Fertile U.S. Males. Environ. Health Perspect. 2003, 111, 414–420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joffe, M. Semen quality analysis and the idea of normal fertility. Asian J. Androl. 2010, 12, 79–82. [Google Scholar] [CrossRef] [Green Version]
- Sumner, R.N.; Harris, I.T.; Van der Mescht, M.; Byers, A.; England, G.C.W.; Lea, R.G. The dog as a sentinel species for environmental effects on human fertility. Reproduction 2020, 159, R265–R276. [Google Scholar] [CrossRef] [PubMed]
- Cimenci, O.; Vandevijvere, S.; Goscinny, S.; Van Den Bergh, M.A.; Hanot, V.; Vinkx, C.; Bolle, F.; Van Loco, J. Dietary exposure of the Belgian adult population to non-dioxin-like PCBs. Food Chem. Toxicol. 2013, 59, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Monnolo, A.; Clausi, M.T.; Mercogliano, R.; Fusco, G.; Fiorentino, M.L.; Buono, F.; Lama, A.; Ferrante, M.C. Levels of polychlorinated biphenyls and organochlorine pesticides in donkey milk: Correlation with the infection level by intestinal strongyles. Chemosphere 2020, 258, 127287. [Google Scholar] [CrossRef]
- Mullen, K.R.; Rivera, B.N.; Tidwell, L.G.; Ivanek, R.; Anderson, K.A.; Ainsworth, D.M. Environmental surveillance and adverse neonatal health outcomes in foals born near unconventional natural gas development activity. Sci. Total Environ. 2020, 731, 138497. [Google Scholar] [CrossRef]
- Whitesell, K.; Stefanovski, D.; McDonnell, S.; Turner, R. Evaluation of the effect of laboratory methods on semen analysis and breeding soundness examination (BSE) classification in stallions. Theriogenology 2020, 142, 67–76. [Google Scholar] [CrossRef]
- Cooper, T.G.; Noonan, E.; Von Eckardstein, S.; Auger, J.; Gordon Baker, H.W.; Behre, H.M.; Haugen, T.B.; Kruger, T.; Wang, C.; Mbizvo, M.T.; et al. World Health Organization reference values for human semen characteristics. Hum. Reprod. 2010, 16, 231–245. [Google Scholar] [CrossRef]
- Dillon, P.; Snijders, S.; Buckley, F.; Harris, B.; O’Connor, P.; Mee, J.F. A comparison of different dairy cow breeds on a seasonal grass-based system of milk production: 2. Reproduction and survival. Livest. Prod. Sci. 2003, 83, 35–42. [Google Scholar] [CrossRef]


| Inclusion | Exclusion |
|---|---|
|
|
|
|
| Category for Analysis | Data Extracted from Individual Studies | |
|---|---|---|
| Geographic Location | Countries Included | |
| Africa | Egypt, South Africa | |
| Asia | India, Iran, Saudi Arabia, Thailand | |
| North America | United States, Canada, Mexico | |
| South America | Argentina, Brazil, Chile, Colombia | |
| Europe | Austria, Belgium, Czech Republic, Finland, France, Germany, Italy, Netherlands, Poland, Portugal, Spain, Sweden, Switzerland | |
| UK | United Kingdom | |
| Other | Australia, New Zealand, Russia, Turkey | |
| Breed category 1 | Horse breeds included | |
| Andalusian | Lusitano, Peruvian Paso, Lipizzaner, Mangalarga Marchador, Brazilian Jumping, Spanish Purebred, Sorraia, Garrano | |
| Arabian | Arabian, Anglo-Arab | |
| Draught | Draught, Polish Coldblood | |
| Miniature | Miniature, Shetland, Miniature Caspian Pony | |
| Pony | Brazilian Pony, Pony, Connemara, Welsh Pony | |
| Quarter Horse | American Quarter Horse; Azteca Horse, Thoroughbred; Trakehner, Manipuri, Standardbred, American Paint Horse, Friesians, Thai Native X, Pantaneiro, Trotter, Maremmano, Finnhorse | |
| Warmblood | Hanoverian, Holsteiner, Appaloosa, Dutch Warmblood, German Warmblood, Oldenburg Warmblood, Rhinelander, Chilean Purebred, Marwari, Kathiawari, Zanskari, French Saddlebred, Westphalian, Swedish Warmblood, Old Kladruber, Criollo Colombiano, Brandenburg, Belgium Draft, Franches-Montagnes, French Warmblood, Polish Warmblood, Haflinger | |
| Month of Collection | Northern Hemisphere | Southern Hemisphere |
| November-February | Winter | Summer |
| March-June | Spring | Autumn |
| July-August | Summer | Winter |
| September-October | Autumn | Spring |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perrett, J.; Harris, I.T.; Maddock, C.; Farnworth, M.; Pyatt, A.Z.; Sumner, R.N. Systematic Analysis of Breed, Methodological, and Geographical Impact on Equine Sperm Progressive Motility. Animals 2021, 11, 3088. https://doi.org/10.3390/ani11113088
Perrett J, Harris IT, Maddock C, Farnworth M, Pyatt AZ, Sumner RN. Systematic Analysis of Breed, Methodological, and Geographical Impact on Equine Sperm Progressive Motility. Animals. 2021; 11(11):3088. https://doi.org/10.3390/ani11113088
Chicago/Turabian StylePerrett, Jodie, Imogen Thea Harris, Christy Maddock, Mark Farnworth, Alison Z. Pyatt, and Rebecca Nicole Sumner. 2021. "Systematic Analysis of Breed, Methodological, and Geographical Impact on Equine Sperm Progressive Motility" Animals 11, no. 11: 3088. https://doi.org/10.3390/ani11113088
APA StylePerrett, J., Harris, I. T., Maddock, C., Farnworth, M., Pyatt, A. Z., & Sumner, R. N. (2021). Systematic Analysis of Breed, Methodological, and Geographical Impact on Equine Sperm Progressive Motility. Animals, 11(11), 3088. https://doi.org/10.3390/ani11113088








