Seroprevalence of Leptospira in Racehorses and Broodmares in New Zealand
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sample Collection and Serological Testing
2.3. Questionnaire
2.4. Statistical Analysis
3. Results
3.1. Population Description
3.2. Serovar Results
3.3. Questionnaire Results
3.4. Logistic Regression Analysis
3.4.1. Any Serovar
3.4.2. Serovar Ballum
3.4.3. Serovar Copenhageni
3.4.4. Serovar Hardjo (Bovis)
3.4.5. Serovar Pomona
3.4.6. Serovar Tarassovi
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sanhueza, J.M.; Baker, M.G.; Benschop, J.; Collins-Emerson, J.M.; Wilson, P.R.; Heuer, C. Estimation of the burden of leptospirosis in New Zealand. Zoonoses Public Health 2020, 67, 167–176. [Google Scholar] [CrossRef]
- Institute of Environmental Science and Research Limited (ESR). Surveillance Report Notifiable and Other Diseases in New Zealand, Annual Report. 2014–2018. Available online: https://surv.esr.cri.nz/PDF_surveillance/AnnualRpt/AnnualSurv/ (accessed on 2 September 2020).
- El-Tras, W.F.; Bruce, M.; Holt, H.R.; Eltholth, M.M.; Merien, F. Update on the status of leptospirosis in New Zealand. Acta Trop. 2018, 188, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Yupiana, Y.; Vallee, E.; Wilson, P.; Collins-Emerson, J.; Weston, J.; Benschop, J.; Heuer, C. Emerging Leptospira strain poses public health risk for dairy farmers in New Zealand. Prev. Vet. Med. 2019, 170, 104727. [Google Scholar] [CrossRef] [PubMed]
- Dreyfus, A.; Wilson, P.; Benschop, J.; Collins-Emerson, J.; Verdugo, C.; Heuer, C. Seroprevalence and herd-level risk factors for seroprevalence of Leptospira spp. in sheep, beef cattle and deer in New Zealand. N. Z. Vet. J. 2018, 66, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Stevenson, B.; Adler, B. Leptospirosis in horses. Vet. Microbiol. 2013, 167, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Boggs, J.; Donecker, J. Equine Leptospirosis: Disease Overview and the Risks and Economic Ramifications of Leptospira-Associated Recurrent Uveitis and Leptospiral Abortion; Technical Bulletin; LEI-00003; Zoetis: Morris County, NJ, USA, 2015. [Google Scholar]
- Gilger, B.C. Association of acute leptospirosis with systemic disease and uveitis in horses. Equine Vet. Educ. 2018, 30, 137–138. [Google Scholar] [CrossRef]
- Frellstedt, L. Equine recurrent uveitis: A clinical manifestation of leptospirosis. Equine Vet. Educ. 2009, 21, 546–552. [Google Scholar] [CrossRef]
- Hamond, C.; Pestana, C.P.; Rocha-de-Souza, C.M.; Cunha, L.E.R.; Brandão, F.Z.; Medeiros, M.A.; Lilenbaum, W. Presence of leptospires on genital tract of mares with reproductive problems. Vet. Microbiol. 2015, 179, 264–269. [Google Scholar] [CrossRef]
- Witkowski, L.; Cywinska, A.; Paschalis-Trela, K.; Crisman, M.; Kita, J. Multiple etiologies of equine recurrent uveitis—A natural model for human autoimmune uveitis: A brief review. Comp. Immunol. Microbiol. Infect. Dis. 2016, 44, 14–20. [Google Scholar] [CrossRef]
- Hilbrink, F.; Penrose, M. Serological reactions against Leptospira interrogans serovars in New Zealand horses. N. Z. Vet. J. 1990, 38, 124–125. [Google Scholar] [CrossRef]
- Julian, A. Equine abortions in New Zealand between 1974 and 1990. Surveillance 1992, 19, 24–25. [Google Scholar]
- Julian, A. An outbreak of equine leptospiral abortions in Thoroughbred mares. Vetscript 1998, 11, 24–25. [Google Scholar]
- Anonymous. Quarterly review of diagnostic cases: April-June 2017. Surveillance 2017, 44, 39–47. [Google Scholar]
- Anonymous. Quarterly review of diagnostic cases. Surveillance 2020, 47, 13. [Google Scholar]
- Gee, E.; Rogers, C.; Bolwell, C. Commercial equine production in New Zealand 1: Reproduction and breeding. Anim. Prod. Sci. 2017. [Google Scholar] [CrossRef]
- Bolwell, C.F.; Rogers, C.W.; Gee, E.K.; Rosanowski, S.M. Descriptive statistics and the pattern of horse racing in New Zealand: Part 1 Thoroughbred Racing. Anim. Prod. Sci. 2014, 56, 77–81. [Google Scholar] [CrossRef]
- Rogers, C.W.; Gee, E.K.; Firth, E.C. A cross-sectional survey of Thoroughbred stud farm management in the North Island of New Zealand. N. Z. Vet. J. 2007, 55, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Bengtsson, J.; Rogers, C.; Back, P.; Emanuelson, U.; Roca, J.; Gee, E.; Bolwell, C. Characteristics of the grazing and farm management of broodmares on commercial Thoroughbred stud farms during spring. N. Z. J. Anim. Sci. Prod. 2018, 78, 88–91. [Google Scholar]
- Wilson, P.; Mannewald, A.; Collins-Emerson, J.; Dreyfus, A.; Sanhueza, J.M.; Benschop, J.; Verdugo, C.; Emanuelson, U.; Boqvist, S.; Heuer, C. Serological study of Leptospira interrogans serovar Copenhageni and L. borgpetersenii serovars Tarassovi and Ballum in beef cattle, sheep and deer in New Zealand. N. Z. Vet. J. 2020. accepted. [Google Scholar]
- Marshall, R.B.; Manktelow, B.W. Fifty years of leptospirosis research in New Zealand: A perspective. N. Z. Vet. J. 2002, 50, 61–63. [Google Scholar] [CrossRef] [PubMed]
- Benschop, J. Communicating Leptospirosis outbreaks—It takes a village. In Proceedings of the 10th International Leptospirosis Society Meeting, Palmerston North, New Zealand, 27 November–1 December 2017. [Google Scholar]
- Bolwell, C.F.; Rogers, C.; Gee, E.; Rosanowski, S. Commercial equine production in New Zealand 3: The Racing and Sport industries. Anim. Prod. Sci. 2017. [Google Scholar] [CrossRef]
- Faine, S. Guidelines for the Control of Leptospirosis; World Health Organisation: Geneva, Switzerland, 1982. [Google Scholar]
- Blackmore, D.K.; Bahaman, A.R.; Marshall, R.B. The epidemiological interpretation of serological responses to leptospiral serovars in sheep. N. Z. Vet. J. 1982, 30, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Dohoo, I.; Martin, W.; Stryhn, H. Veterinary Epidemiologic Research; AVC Inc.: Charlottetown, PE, Canada, 2003; p. 309. [Google Scholar]
- Jung, B.Y.; Lee, K.W.; Ha, T.Y. Seroprevalence of Leptospira spp. in Clinically Healthy Racing Horses in Korea. J. Vet. Med. Sci. 2010, 72, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Dickeson, D.; Love, D.N. A serological survey of dogs, cats and horses in south-eastern Australia for leptospiral antibodies. Aust. Vet. J. 1993, 70, 389–390. [Google Scholar] [CrossRef]
- Farias, D.K.; Dick, G.; Bunn, S.; Thaler Neto, A.; Rech, C.; Menin, Á.; Saito, M.E. Risk factors related to seropositivity for Leptospira spp. in horses in the Serra Catarinense region. Cienc. Rural 2020, 50. [Google Scholar] [CrossRef]
- Vera, E.; Taddei, S.; Cavirani, S.; Schiavi, J.; Angelone, M.; Cabassi, C.S.; Schiano, E.; Quintavalla, F. Leptospira Seroprevalence in Bardigiano Horses in Northern Italy. Animals 2020, 10, 23. [Google Scholar] [CrossRef]
- Trimble, A.; Blevins, C.; Beard, L.; Deforno, A.; Davis, E. Seroprevalence, frequency of leptospiuria, and associated risk factors in horses in Kansas, Missouri, and Nebraska from 2016-2017. PLoS ONE 2018, 13, e0206639. [Google Scholar] [CrossRef]
- Blatti, S.; Overesch, G.; Gerber, V.; Frey, J.; Hussy, D. Seroprevalence of Leptospira spp. in clinically healthy horses in Switzerland. Schweiz Arch Tierheilkd 2011, 153, 449–456. [Google Scholar] [CrossRef]
- Fagre, A.C.; Mayo, C.E.; Pabilonia, K.L.; Landolt, G.A. Seroprevalence of Leptospira spp. in Colorado equids and association with clinical disease. J. Vet. Diagn. Investig. 2020. [Google Scholar] [CrossRef]
- Divers, T.J.; Chang, Y.-F.; Irby, N.L.; Smith, J.L.; Carter, C.N. Leptospirosis: An important infectious disease in North American horses. Equine Vet. J. 2019, 51, 287–292. [Google Scholar] [CrossRef]
- Siqueira, C.C.; Fraga, D.B.; Chagas-Junior, A.D.; Athanazio, D.A.; Silva, M.M.; Cerqueira, R.B.; McBride, F.W.; Pinna, M.H.; Ayres, M.C. Seroprevalence and risk factors associated with equineleptospirosis in the metropolitan region of Salvador and Recôncavo Baiano region, Bahia state (NE Brazil). Trop. Anim. Health Prod. 2019. [Google Scholar] [CrossRef] [PubMed]
- Tsegay, K.; Potts, A.D.; Aklilu, N.; Lötter, C.; Gummow, B. Circulating serovars of Leptospira in cart horses of central and southern Ethiopia and associated risk factors. Prev. Vet. Med. 2016, 125, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Gee, E.K.; Firth, E.C.; Morel, P.C.H.; Fennessy, P.F.; Grace, N.D.; Mogg, T.D. Enlargements of the distal third metacarpus and metatarsus in thoroughbred foals at pasture from birth to 160 days of age. N. Z. Vet. J. 2005, 53, 438–447. [Google Scholar] [CrossRef] [PubMed]
- Rogers, C.W.; Gee, E.K.; Back, P.J.; van Zon, S.; Hirst, R.L.; Pomroy, W.E. Pasture use and management in commercial equine production systems. N. Z. J. Anim. Sci. Prod. 2018, 78, 40–44. [Google Scholar]
- Wood, I.J.; Lancaster, B.E.; Rogers, C.W. The feeding and management of Thoroughbred and Standardbred Racehorses displaying clinical signs of recurrent exertional rhabdomyolysis. N. Z. J. Anim. Sci. Prod. 2019, 79, 26–31. [Google Scholar]
- Fang, F.; Collins-Emerson, J.M.; Cullum, A.; Heuer, C.; Wilson, P.R.; Benschop, J. Shedding and Seroprevalence of Pathogenic Leptospira spp. in Sheep and Cattle at a New Zealand Abattoir. Zoonoses Public Health 2015, 62, 258–268. [Google Scholar] [CrossRef]
- Simbizi, V.; Saulez, M.N.; Potts, A.; Lötter, C.; Gummow, B. A study of leptospirosis in South African horses and associated risk factors. Prev. Vet. Med. 2016, 134, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Rosanowski, S.M.; Cogger, N.; Rogers, C.W. An investigation of the movement patterns and biosecurity practices on Thoroughbred and Standardbred stud farms in New Zealand. Prev. Vet. Med. 2013, 108, 178–187. [Google Scholar] [CrossRef]
- Hamond, C.; Martins, G.; Lawson-Ferreira, R.; Medeiros, M.A.; Lilenbaum, W. The role of horses in the transmission of leptospirosis in an urban tropical area. Epidemiol. Infect. 2012, 141, 33–35. [Google Scholar] [CrossRef]
- Bott, R.C.; Greene, E.A.; Koch, K.; Martinson, K.L.; Siciliano, P.D.; Williams, C.; Trottier, N.L.; Burk, A.; Swinker, A. Production and Environmental Implications of Equine Grazing. J. Equine Vet. Sci. 2013, 33, 1031–1043. [Google Scholar] [CrossRef]
- Gellin, G.; Carter, C.; Caldwell, G.; Timoney, J.F.; Steinman, M.; Fardo, D.; Sanderson, W. Serological evidence of potential occupational exposure to leptospirosis in Kentucky horse farm workers and veterinarians. In Proceedings of the American Public Health Association 140th Annual Meeting & Expo, San Francisco, CA, USA, 27–31 October 2012. [Google Scholar]
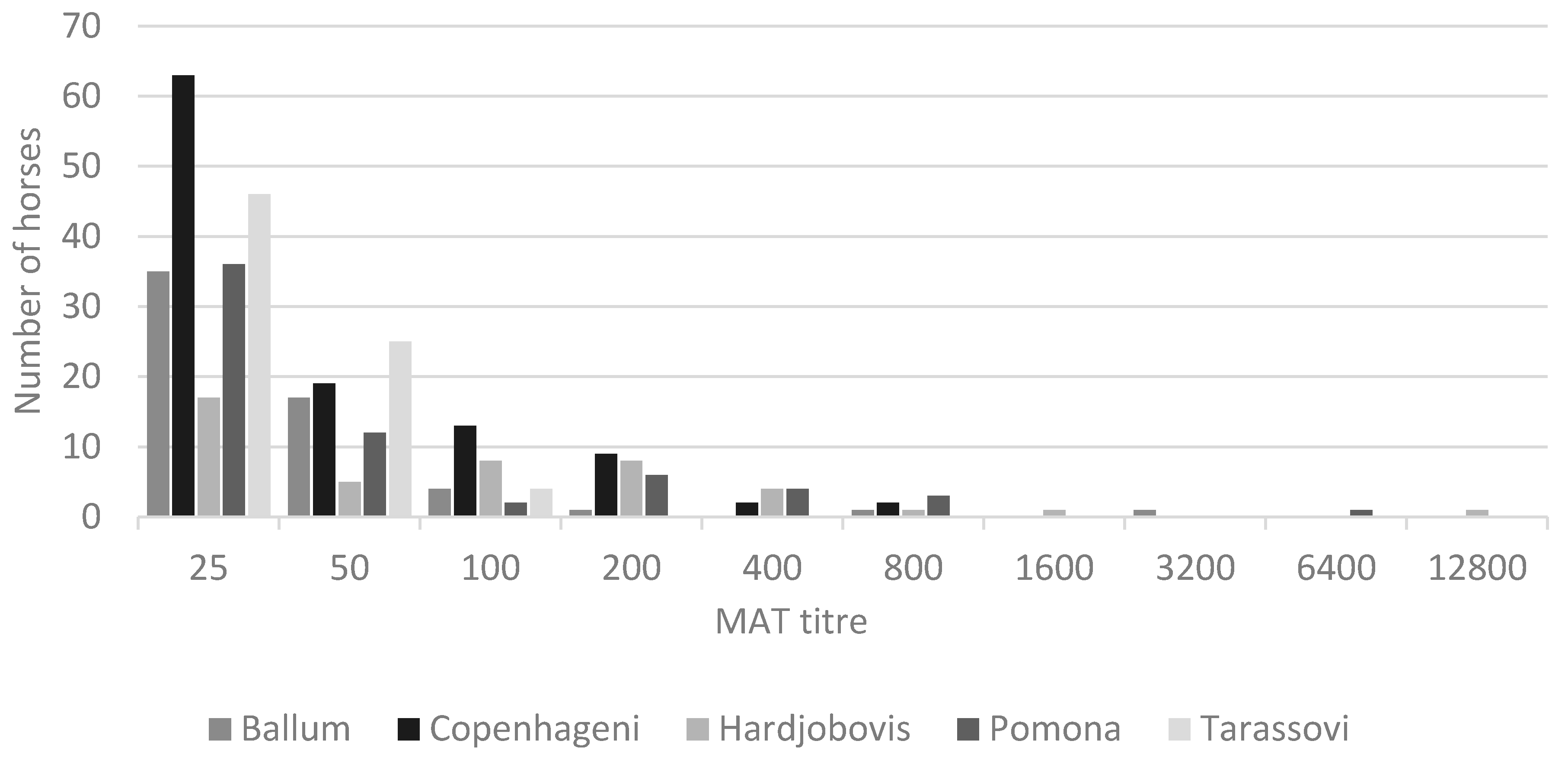
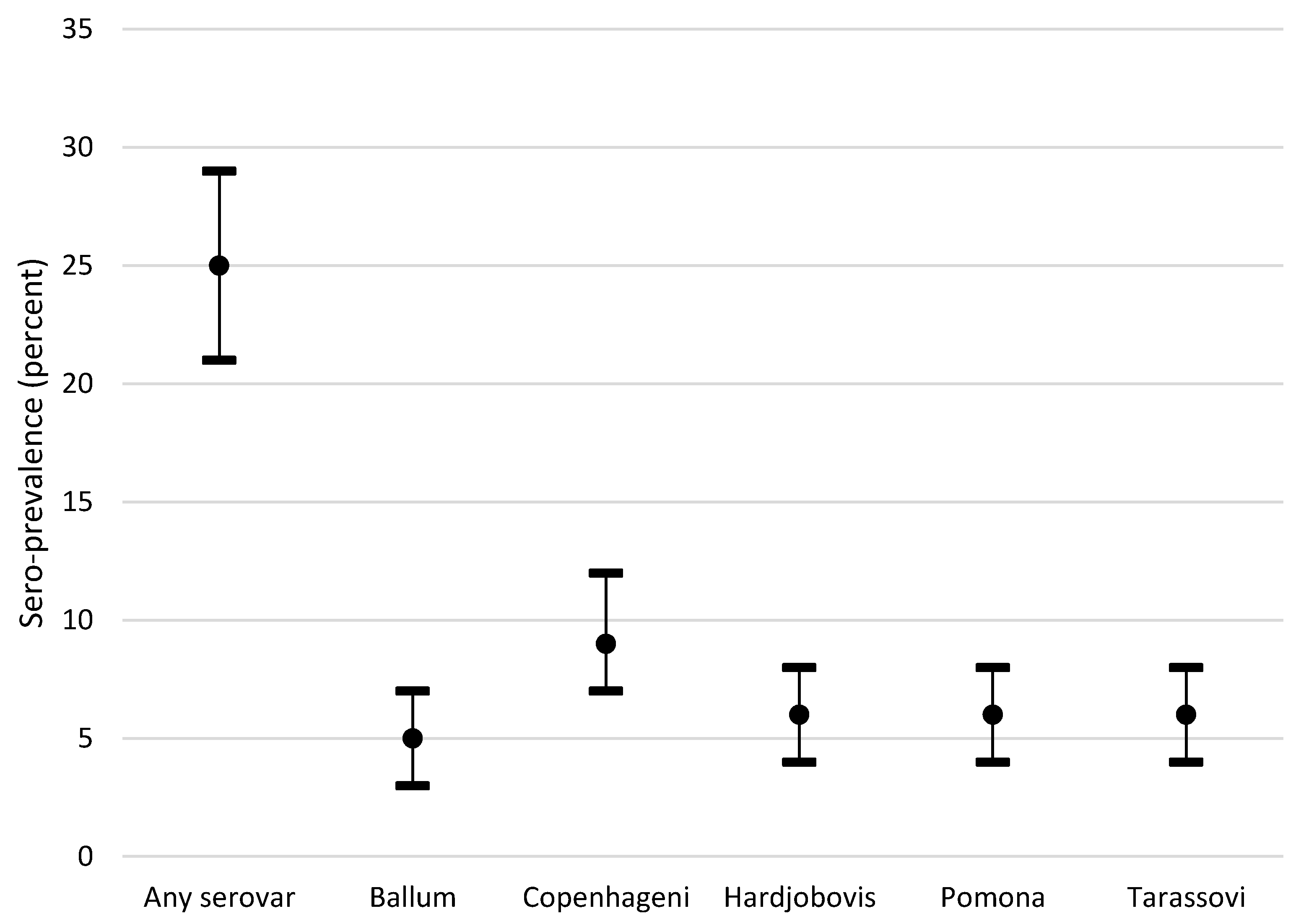
| Variable | Level | Any Serovar | Ballum | Copenhageni | Hardjo (Bovis) | Pomona | Tarassovi | |
|---|---|---|---|---|---|---|---|---|
| Total Horses | Number of Horses Positive (%) | Number of Horses Positive (%) | Number of Horses Positive (%) | Number of Horses Positive (%) | Number of Horses Positive (%) | Number of Horses Positive (%) | ||
| Horse-level | ||||||||
| Sex | Female | 400 | 111 (27.8) | 22 (5.5) | 41 (10.3) | 22 (5.5) | 27 (6.8) | 28 (7.0) |
| Male | 99 | 13 (13.1) | 2 (2.0) | 4 (4.0) | 6 (6.1) | 1 (1.0) | 1 (1.0) | |
| Role | Broodmare | 335 | 103 (30.8) | 21 (6.3) | 38 (11.3) | 20 (6.0) | 26 (7.8) | 27 (8.1) |
| Racehorse | 164 | 21 (12.8) | 3 (1.8) | 7 (4.3) | 8 (4.9) | 2 (1.2) | 2 (1.2) | |
| Vaccinated against other pathogens * | No/unsure | 105 | 14 (13.3) | 1 (0.9) | 6 (5.7) | 6 (5.7) | 0 | 2 (1.9) |
| Yes | 394 | 110 (27.9) | 23 (5.8) | 39 (9.9) | 22 (5.5) | 28 (7) | 27 (6.8) | |
| Property-level | ||||||||
| Region | Manawatu | 315 | 72 (22.8) | 14 (4.4) | 25 (5.9) | 19 (6.0) | 17 (5.4) | 16 (5.1) |
| Waikato | 136 | 40 (29.4) | 7 (5.2) | 16 (11.8) | 7 (5.2) | 8 (5.9) | 10 (7.4) | |
| Auckland | 48 | 12 (25) | 3 (6.3) | 4 (8.3) | 2 (4.2) | 3 (6.3) | 3 (6.3) | |
| Natural water source on property | No | 69 | 17 (24.6) | 1 (1.5) | 6 (8.7) | 5 (7.3) | 5 (7.3) | 4 (5.8) |
| Yes | 430 | 107 (24.9) | 23 (5.4) | 39 (9.1) | 23 (5.4) | 23 (5.4) | 25 (5.8) | |
| Natural water source type * | Bore | 2 | 2 (100) | 1 (50.0) | 1 (50.0) | 0 (0.0) | 2 (100) | 1 (50.0) |
| River | 211 | 55 (26.1) | 11 (5.2) | 18 (8.5) | 12 (5.7) | 12 (5.7) | 11 (5.2) | |
| Stream | 16 | 4 (25) | 1 (6.3) | 3 (18.8) | 0 (0.0) | 0 (0.0) | 1 (6.3) | |
| Creek | 193 | 42 (21.8) | 9 (4.7) | 15 (7.8) | 11 (5.7) | 9 (4.7) | 10 (5.2) | |
| Swamp | 8 | 4 (50) | 1 (12.5) | 2 (25.0) | 0 (0.0) | 0 (0.0) | 2 (25.0) | |
| Flooding on property in the last 12 months | No | 259 | 73 (28.2) | 12 (4.6) | 27 (10.4) | 19 (7.3) | 18 (7.0) | 16 (6.2) |
| Yes | 240 | 51 (21.2) | 12 (5.0) | 18 (7.5) | 9 (3.8) | 10 (4.2) | 13 (5.4) | |
| Evidence of wildlife on the property | ||||||||
| Rats | No | 86 | 16 (18.6) | 3 (3.5) | 5 (5.8) | 5 (5.8) | 5 (5.8) | 4 (4.7) |
| Yes | 413 | 108 (26.2) | 21 (5.1) | 40 (9.7) | 23 (5.6) | 23 (5.6) | 25 (6.1) | |
| Mice | No | 9 | 9 (11.1) | 0 (0.0) | 1 (11.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Yes | 490 | 124 (24.9) | 24 (4.9) | 44 (9.0) | 28 (5.7) | 28 (5.7) | 29 (5.9) | |
| Possums | No | 114 | 24 (21.1) | 6 (5.3) | 9 (7.9) | 4 (3.5) | 6 (5.3) | 5 (4.4) |
| Yes | 385 | 100 (26.0) | 18 (4.7) | 36 (9.4) | 24 (6.2) | 22 (5.7) | 24 (6.2) | |
| Hedgehogs | No | 246 | 53 (21.5) | 13 (5.3) | 21 (8.5) | 12 (4.9) | 12 (4.9) | 11 (4.5) |
| Yes | 253 | 71 (28.1) | 11 (4.4) | 24 (9.5) | 16 (6.3) | 16(6.3) | 18 (7.1) | |
| Rabbits | No | 19 | 6 (31.6) | 1 (5.3) | 0 (0.0) | 2 (10.5) | 0 (0.0) | 3 (15.8) |
| Yes | 480 | 118 (24.6) | 23 (4.8) | 45 (9.4) | 26 (5.4) | 28 (5.8) | 26 (5.4) | |
| Mustelids | No | 364 | 92 (25.3) | 21 (5.8) | 38 (10.4) | 18 (5.0) | 20 (5.5) | 21 (5.8) |
| Yes | 135 | 32 (23.7) | 3 (2.2) | 7 (5.2) | 10 (7.4) | 8 (5.9) | 8 (5.9) | |
| Traps for rodents on property | No | 19 | 2 (10.5) | 0 (0.0) | 0 (0.0) | 2 (10.5) | 0 (0.0) | 0 (0.0) |
| Yes | 480 | 122 (25.4) | 24 (5.0) | 45 (9.4) | 26 (5.4) | 28 (5.8) | 29 (6.0) | |
| Feed storage prior to opening | Feed shed | 333 | 85 (25.5) | 17 (5.1) | 33 (9.9) | 15 (4.5) | 21 (6.3) | 20 (6.0) |
| Other shed | 50 | 11 (22.0) | 2 (4.0) | 3 (6.0) | 3 (6.0) | 4 (8.0) | 3 (6.0) | |
| Shed and silo | 44 | 8 (18.2) | 1 (2.3) | 2 (4.6) | 6 (13.6) | 0 (0.0) | 1 (2.3) | |
| Other (silo/kegs) | 72 | 20 (27.8) | 4 (5.6) | 7 (9.7) | 4 (5.6) | 3 (4.2) | 5 (6.9) | |
| Feed storage once opened | Open feed bags | 72 | 17(23.6) | 7 (9.7) | 9 (12.5) | 0 (0.0) | 5 (6.9) | 3 (4.2) |
| Unsealed feed bins | 73 | 15 (20.6) | 2 (2.7) | 5 (6.9) | 6 (8.2) | 2 (2.7) | 3 (4.1) | |
| Sealed feed bins | 230 | 53 (23.0) | 10 (4.4) | 17 (7.4) | 12 (5.2) | 11 (4.8) | 14 (6.1) | |
| Silo | 85 | 26 (30.6) | 3 (3.5) | 7 (8.2) | 8 (9.4) | 8 (9.4) | 6 (7.1) | |
| Other | 39 | 13 (33.3) | 2 (5.1) | 7 (18.0) | 2 (5.1) | 2 (5.1) | 3 (7.7) | |
| Animals on property | ||||||||
| Cats | No | 64 | 13 (20.3) | 3 (4.7) | 4 (6.3) | 2 (3.1) | 5 (7.8) | 3 (4.7) |
| Yes | 435 | 111 (25.5) | 21 (4.8) | 41 (9.4) | 26 (6.0) | 23 (5.3) | 26 (6.0) | |
| Dogs | No | 22 | 5 (22.7) | 1 (4.6) | 3 (13.6) | 0 (0.0) | 0 (0.0) | 2 (9.1) |
| Yes | 477 | 119 (25.0) | 23 (4.8) | 42 (8.8) | 28 (5.9) | 28 (5.9) | 27 (5.7) | |
| Pigs | No | 499 | 124 (24.9) | 24 (4.8) | 45 (9.0) | 28 (5.6) | 28 (5.6) | 29 (5.8) |
| Yes | 0 | - | - | - | - | - | - | |
| Goats | No | 465 | 112 (24.1) | 24 (5.2) | 42 (9.0) | 24 (5.2) | 23 (5.0) | 27 (5.8) |
| Yes | 34 | 12 (35.3) | 0 (0.0) | 3 (8.8) | 4 (11.8) | 5 (14.7) | 2 (5.9) | |
| Dairy cattle | No | 383 | 95 (24.8) | 19 (5.0) | 36 (9.4) | 19 (5.0) | 21 (5.5) | 21 (5.5) |
| Yes | 116 | 29 (25.0) | 5 (4.3) | 9 (7.8) | 9 (7.8) | 7 (6.1) | 8 (6.9) | |
| Beef cattle | No | 101 | 14 (13.9) | 3 (3.0) | 7 (6.9) | 4 (4.0) | 1 (1.0) | 2 (2.0) |
| Yes | 398 | 110 (27.6) | 21 (5.2) | 38 (9.5) | 24 (6.0) | 27 (6.8) | 27 (6.8) | |
| Sheep | No | 217 | 46 (21.2) | 5 (2.3) | 19 (8.8) | 9 (4.2) | 10 (4.6) | 11 (5.1) |
| Yes | 282 | 78 (27.6) | 19 (6.7) | 26 (9.2) | 19 (6.7) | 18 (6.4) | 18 (6.4) | |
| Deer | No | 451 | 110 (24.4) | 22 (4.9) | 37 (8.2) | 26 (5.8) | 26 (5.8) | 26 (5.8) |
| Yes | 48 | 14 (29.2) | 2 (4.2) | 8 (16.7) | 2 (4.2) | 2 (4.2) | 3 (6.3) | |
| Animals on property vaccinated for lepto ^ | No | 305 | 73 (23.9) | 14 (4.6) | 31 (10.1) | 12 (3.9) | 16 (5.3) | 19 (6.2) |
| Yes | 130 | 31 (23.9) | 9 (6.9) | 12 (9.2) | 7 (5.4) | 7 (5.4) | 5 (3.9) | |
| Do not know | 64 | 20 (31.3) | 1 (1.6) | 2 (3.1) | 9 (14.1) | 5 (7.8) | 5 (7.8) | |
| Contact with other animals | ||||||||
| Contact with cattle | No | 77 | 9 (11.7) | 2 (2.6) | 5 (6.5) | 1 (1.3) | 1 (1.3) | 1 (1.3) |
| Yes | 422 | 115 (27.3) | 22 (5.2) | 40 (9.5) | 27 (6.4) | 27 (6.4) | 28 (6.6) | |
| Graze horses same time as cattle | No | 321 | 76 (23.7) | 14 (4.4) | 28 (8.8) | 14 (4.4) | 14 (4.4) | 19 (5.9) |
| Yes | 178 | 48 (27.0) | 10 (5.6) | 17 (9.6) | 14 (7.9) | 14 (7.9) | 10 (5.6) | |
| Graze horses alternately with cattle | No | 151 | 26 (17.2) | 8 (5.3) | 16 (10.6) | 1 (0.7) | 5 (3.3) | 4 (2.7) |
| Yes | 348 | 98 (28.2) | 16 (4.6) | 29 (8.3) | 27 (7.8) | 23 (6.6) | 25 (7.2) | |
| Horses share water source with cattle | No | 277 | 63 (22.7) | 16 (5.8) | 25 (9.0) | 10 (3.6) | 14 (5.1) | 13 (4.7) |
| Yes | 222 | 61 (27.5) | 8 (3.6) | 20 (9.0) | 18 (8.1) | 14 (6.3) | 16 (7.2) | |
| Contact with cattle over fence | No | 300 | 69 (23.0) | 17 (5.7) | 27 (9.0) | 12 (4.8) | 17 (5.7) | 14 (4.7) |
| Yes | 199 | 55 (27.6) | 7 (3.5) | 18 (9.1) | 16 (8.0) | 11 (5.5) | 15 (7.5) | |
| Contact with sheep | No | 68 | 6 (8.8) | 0 (0.0) | 4 (5.9) | 1 (1.5) | 0 (0.0) | 1 (1.5) |
| Yes | 431 | 118 (27.4) | 24 (5.6) | 41 (9.5) | 27 (6.3) | 28 (6.5) | 28 (6.5) | |
| Graze horses same time as sheep | No | 340 | 86 (25.3) | 13 (3.8) | 30 (8.8) | 18 (5.3) | 18 (5.3) | 22 (6.5) |
| Yes | 159 | 38 (23.9) | 11 (6.9) | 15 (9.4) | 10 (6.3) | 10(6.3) | 7 (4.4) | |
| Graze horses alternately with sheep | No | 271 | 61 (22.5) | 11 (4.1) | 28 (10.3) | 9 (3.3) | 14 (5.2) | 14 (5.2) |
| Yes | 228 | 63 (27.6) | 13 (5.7) | 17 (7.5) | 19 (8.3) | 14 (6.1) | 15 (6.6) | |
| Horses share water source with sheep | No | 365 | 90 (24.7) | 17 (4.7) | 36 (9.9) | 16 (4.4) | 20 (5.5) | 21 (5.8) |
| Yes | 134 | 34 (25.4) | 7 (5.2) | 9 (6.7) | 12 (9.0) | 8 (6.0) | 8 (6.0) | |
| Contact with sheep over fence | No | 371 | 94 (25.3) | 18 (4.9) | 38 (10.2) | 18 (4.9) | 23 (6.2) | 20 (5.4) |
| Yes | 128 | 30 (23.4) | 6 (4.7) | 7 (5.5) | 10 (7.8) | 5 (3.9) | 9 (7.0) | |
| Contact with deer | No | 269 | 58 (21.6) | 14 (5.2) | 22 (8.2) | 13 (4.8) | 12 (4.5) | 14 (5.2) |
| Yes | 230 | 66 (28.7) | 10 (4.3) | 23 (10.0) | 15 (6.5) | 16 (7.0) | 15 (6.5) | |
| Graze horses same time as deer | No | 499 | 124 (24.9) | 24 (4.8) | 45 (9.0) | 28 (5.6) | 28 (5.6) | 29 (5.8) |
| Yes | 0 | - | - | - | - | - | - | |
| Graze horses alternately with deer | No | 490 | 123 (25.1) | 24 (4.9) | 44 (9.0) | 28 (5.7) | 28 (5.7) | 29 (5.9) |
| Yes | 9 | 1 (11.1) | 0 (0.0) | 1 (11.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Horses share water source with deer | No | 490 | 123 (25.1) | 24 (4.9) | 44 (9.0) | 28 (5.7) | 28 (5.7) | 29 (5.9) |
| Yes | 9 | 1 (11.1) | 0 (0.0) | 1 (11.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Contact with deer over fence | No | 451 | 110 (24.4) | 22 (4.9) | 37 (8.2) | 26 (5.8) | 26 (5.8) | 26 (5.8) |
| Yes | 48 | 14 (29.2) | 2 (4.2) | 8 (16.7) | 2 (4.2) | 2 (4.2) | 3 (6.3) | |
| Property | 1 | 16 | 4 (25.0) | 1 (6.3) | 3 (18.8) | 0 (0.0) | 0 (0.0) | 1 (6.3) |
| 2 | 12 | 2 (16.7) | 0 (0.0) | 0 (0.0) | 2 (16.7) | 0 (0.0) | 0 (0.0) | |
| 3 | 13 | 1 (7.7) | 0 (0.0) | 0 (0.0) | 1 (7.7) | 0 (0.0) | 0 (0.0) | |
| 4 | 7 | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| 5 | 2 | 2 (100) | 1 (50.0) | 1 (50.0) | 0 (0.0) | 2 (100) | 1 (50.0) | |
| 6 | 21 | 7 (33.3) | 1 (4.8) | 1 (4.8) | 2 (9.5) | 2 (9.5) | 2 (9.5) | |
| 7 | 8 | 4 (50) | 1 (12.5) | 2 (25.0) | 0 (0.0) | 0 (0.0) | 2 (25.0) | |
| 8 | 14 | 1 (7.1) | 0 (0.0) | 1 (7.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| 9 | 9 | 1 (11.1) | 0 (0.0) | 1 (11.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| 10 | 18 | 2 (11.1) | 1 (5.6) | 0 (0.0) | 0 (0.0) | 1 (5.6) | 0 (0.0) | |
| 11 | 46 | 11 (23.9) | 5 (10.9) | 7 (15.2) | 0 (0.0) | 4 (8.7) | 1 (2.2) | |
| 12 | 12 | 3 (25) | 1 (8.3) | 2 (16.7) | 1 (8.3) | 0 (0.0) | 1 (8.3) | |
| 13 | 19 | 6 (31.6) | 1 (5.3) | 0 (0.0) | 2 (10.5) | 0 (0.0) | 3 (15.8) | |
| 14 | 11 | 4 (36.4) | 0 (0.0) | 2 (18.2) | 2 (18.2) | 2 (18.2) | 1 (9.1) | |
| 15 | 20 | 3 (15) | 0 (0.0) | 0 (0.0) | 3 (15.0) | 0 (0.0) | 0 (0.0) | |
| 16 | 12 | 1 (8.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (8.3) | |
| 17 | 9 | 1 (11.1) | 0 (0.0) | 1 (11.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| 18 | 21 | 6 (28.6) | 2 (9.5) | 0 (0.0) | 2 (9.5) | 1 (4.8) | 1 (4.8) | |
| 19 | 15 | 1 (6.7) | 0 (0.0) | 1 (6.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| 20 | 5 | 1 (20) | 0 (0.0) | 1 (20.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| 21 | 25 | 11 (44) | 0 (0.0) | 2 (8.0) | 4 (16.0) | 5 (20.0) | 2 (8.0) | |
| 22 | 67 | 23 (34.3) | 5 (7.5) | 7 (10.5) | 5 (7.5) | 6 (9.0) | 5 (7.5) | |
| 23 | 30 | 4 (13.3) | 0 (0.0) | 2 (6.7) | 0 (0.0) | 0 (0.0) | 2 (6.7) | |
| 24 | 39 | 13 (33.3) | 2 (5.1) | 7 (18.0) | 2 (5.1) | 2 (5.1) | 3 (7.7) | |
| 25 | 48 | 12 (25.0) | 3 (6.3) | 4 (8.3) | 2 (4.2) | 3 (6.3) | 3 (6.3) | |
| Risk Factor | Level | Odds Ratio | Robust SE | 95% Confidence Interval | p-Value | Likelihood Ratio Test p-Value |
|---|---|---|---|---|---|---|
| Horse age | 1.15 | 0.03 | 1.09–1.20 | <0.001 | <0.001 | |
| Role | Racehorse | Ref | - | - | - | <0.001 |
| Broodmare | 3.02 | 0.79 | 1.80–5.05 | <0.001 | ||
| Vaccinated for other pathogens | No/unsure | Ref | - | - | - | 0.001 |
| Yes | 2.52 | 0.77 | 1.37–4.61 | 0.003 | ||
| Region | Manawatu | Ref | - | - | - | 0.34 |
| Waikato | 1.41 | 0.33 | 0.89–2.21 | 0.14 | ||
| Auckland | 1.13 | 0.40 | 0.56–2.28 | 0.74 | ||
| Natural water source on property | No | Ref | - | - | - | 0.97 |
| Yes | 1.01 | 0.30 | 0.56–1.83 | 0.97 | ||
| Flooding on property in the last 12 months | No | Ref | - | - | - | 0.07 |
| Yes | 0.69 | 0.14 | 0.46–1.04 | 0.07 | ||
| Evidence of wildlife on the property | ||||||
| Rats | No | Ref | - | - | - | 0.13 |
| Yes | 1.55 | 0.46 | 0.86–2.78 | 0.14 | ||
| Mice | No | Ref | - | - | - | 0.30 |
| Yes | 2.68 | 2.86 | 0.33–21.65 | 0.36 | ||
| Possums | No | Ref | - | - | - | 0.27 |
| Yes | 1.32 | 0.34 | 0.79–2.18 | 0.29 | ||
| Hedgehogs | No | Ref | - | - | - | 0.09 |
| Yes | 1.42 | 0.30 | 0.94–2.14 | 0.09 | ||
| Rabbits | No | Ref | - | - | - | 0.50 |
| Yes | 0.71 | 0.36 | 0.26–1.90 | 0.49 | ||
| Mustelids | No | Ref | - | - | - | 0.72 |
| Yes | 0.92 | 0.22 | 0.58–1.46 | 0.72 | ||
| Traps for rodents on property | No | Ref | - | - | - | 0.11 |
| Yes | 2.90 | 2.19 | 0.66–12.72 | 0.16 | ||
| Feed storage prior to opening | Feed shed | Ref | - | - | - | 0.63 |
| Shed and silo | 0.65 | 0.27 | 0.29–1.45 | 0.29 | ||
| Other shed | 0.82 | 0.30 | 0.40–1.68 | 0.59 | ||
| Other | 1.12 | 0.33 | 0.63–1.99 | 0.69 | ||
| Feed storage once opened | Sealed feed bins | Ref | - | - | - | 0.40 |
| Open feed bags | 1.03 | 0.33 | 0.55–1.93 | 0.92 | ||
| Unsealed feed bins | 0.86 | 0.28 | 0.45–1.65 | 0.66 | ||
| Silo | 1.47 | 0.42 | 0.85–2.56 | 0.17 | ||
| Other | 1.67 | 0.62 | 0.80–3.48 | 0.17 | ||
| Animals on property | ||||||
| Cats | No | Ref | - | - | - | 0.36 |
| Yes | 1.34 | 0.44 | 0.70–2.56 | 0.37 | ||
| Dogs | No | Ref | - | - | - | 0.81 |
| Yes | 1.13 | 0.59 | 0.41–3.13 | 0.81 | ||
| Goats | No | Ref | - | - | - | 0.16 |
| Yes | 1.72 | 0.64 | 0.82–3.58 | 0.15 | ||
| Contact with other animals | ||||||
| Graze horses same time as cattle | No | Ref | - | - | - | 0.42 |
| Yes | 1.19 | 0.25 | 0.78–1.81 | 0.42 | ||
| Graze horses alternately with cattle | No | Ref | - | - | - | 0.008 |
| Yes | 1.88 | 0.46 | 1.16–3.05 | 0.01 | ||
| Graze horses same time as sheep | No | Ref | - | - | - | 0.74 |
| Yes | 0.93 | 0.21 | 0.60–1.44 | 0.74 | ||
| Graze horses alternately with sheep | No | Ref | - | - | - | 0.19 |
| Yes | 1.31 | 0.27 | 0.88–1.97 | 0.19 | ||
| Yes | 0.90 | 0.22 | 0.56–1.44 | 0.67 | ||
| Contact with deer over fence | No | Ref | - | - | - | 0.47 |
| Yes | 1.28 | 0.43 | 0.66–2.47 | 0.47 | ||
| Risk Factor | Level | Odds Ratio | Robust SE | 95% Confidence Interval | p-Value |
|---|---|---|---|---|---|
| Any serovar | |||||
| Horse age | 1.13 | 0.03 | 1.08–1.18 | <0.001 | |
| Role | Racehorse | Ref | - | - | - |
| Broodmare | 1.73 | 0.46 | 1.03–2.93 | 0.04 | |
| Flooding on property last 12 months | No | Ref | - | - | - |
| Yes | 0.59 | 0.09 | 0.43–0.80 | 0.001 | |
| Graze horses alternately with sheep | No | Ref | - | - | - |
| Yes | 1.4 | 0.19 | 1.07–1.84 | 0.01 | |
| Evidence of hedgehogs on the property | No | Ref | - | - | - |
| Yes | 0.77 | 0.12 | 0.55–1.05 | 0.1 | |
| Ballum | |||||
| Role | Racehorse | Ref | - | - | - |
| Broodmare | 4.49 | 2.36 | 1.29–15.6 | 0.02 | |
| Grazing same time as sheep | No | Ref | - | - | - |
| Yes | 2.45 | 1.06 | 1.05–5.72 | 0.03 | |
| Copenhageni | |||||
| Horse age | 1.14 | 0.04 | 1.07–1.22 | <0.001 | |
| Ferrets on the property | No | - | - | - | - |
| Yes | 0.39 | 0.17 | 0.16–0.91 | 0.03 | |
| Hardjo (bovis) | |||||
| Horses graze alternately with cattle | No | - | - | - | - |
| Yes | 10.2 | 10.5 | 1.3–77.6 | 0.02 | |
| Horses graze alternately with sheep | No | - | - | - | - |
| Yes | 1.86 | 0.78 | 0.81–4.24 | 0.14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bolwell, C.F.; Rogers, C.W.; Benschop, J.; Collins-Emerson, J.M.; Adams, B.; Scarfe, K.R.; Gee, E.K. Seroprevalence of Leptospira in Racehorses and Broodmares in New Zealand. Animals 2020, 10, 1952. https://doi.org/10.3390/ani10111952
Bolwell CF, Rogers CW, Benschop J, Collins-Emerson JM, Adams B, Scarfe KR, Gee EK. Seroprevalence of Leptospira in Racehorses and Broodmares in New Zealand. Animals. 2020; 10(11):1952. https://doi.org/10.3390/ani10111952
Chicago/Turabian StyleBolwell, Charlotte F., Chris W. Rogers, Jackie Benschop, Julie M. Collins-Emerson, Brooke Adams, Katherine R. Scarfe, and Erica K. Gee. 2020. "Seroprevalence of Leptospira in Racehorses and Broodmares in New Zealand" Animals 10, no. 11: 1952. https://doi.org/10.3390/ani10111952
APA StyleBolwell, C. F., Rogers, C. W., Benschop, J., Collins-Emerson, J. M., Adams, B., Scarfe, K. R., & Gee, E. K. (2020). Seroprevalence of Leptospira in Racehorses and Broodmares in New Zealand. Animals, 10(11), 1952. https://doi.org/10.3390/ani10111952





