Transovarial Transmission of Borrelia hermsii by Its Tick Vector and Reservoir Host Ornithodoros hermsi
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strain
2.2. Tick Feeding and Colony Maintenance
2.3. Spirochete Infection in Mice
2.4. Spirochete Infection in Eggs and Larvae
2.5. Examination of Unfed Larvae by Dark-Field Microscopy
2.6. Experimental Infection and Transmission of a Genomic Group I B. hermsii
2.7. Statistical Analyses
2.8. Ethics Statement
3. Results
3.1. Fecundity and Transovarial Transmission by the Founding Female
3.2. Prevalence of Spirochete Infection in F-1 Adults and Transovarial Transmission
3.3. Prevalence of Larval Cohorts Infected by Transovarial Transmission as the Colony Aged
3.4. Prevalence of Infection in Nymphal and Adult O. hermsi
3.5. Isolation of Spirochetes from Infected Nymphs
3.6. Microscopic Examination of Single Eggs Versus Same-Clutch Larval Feeding
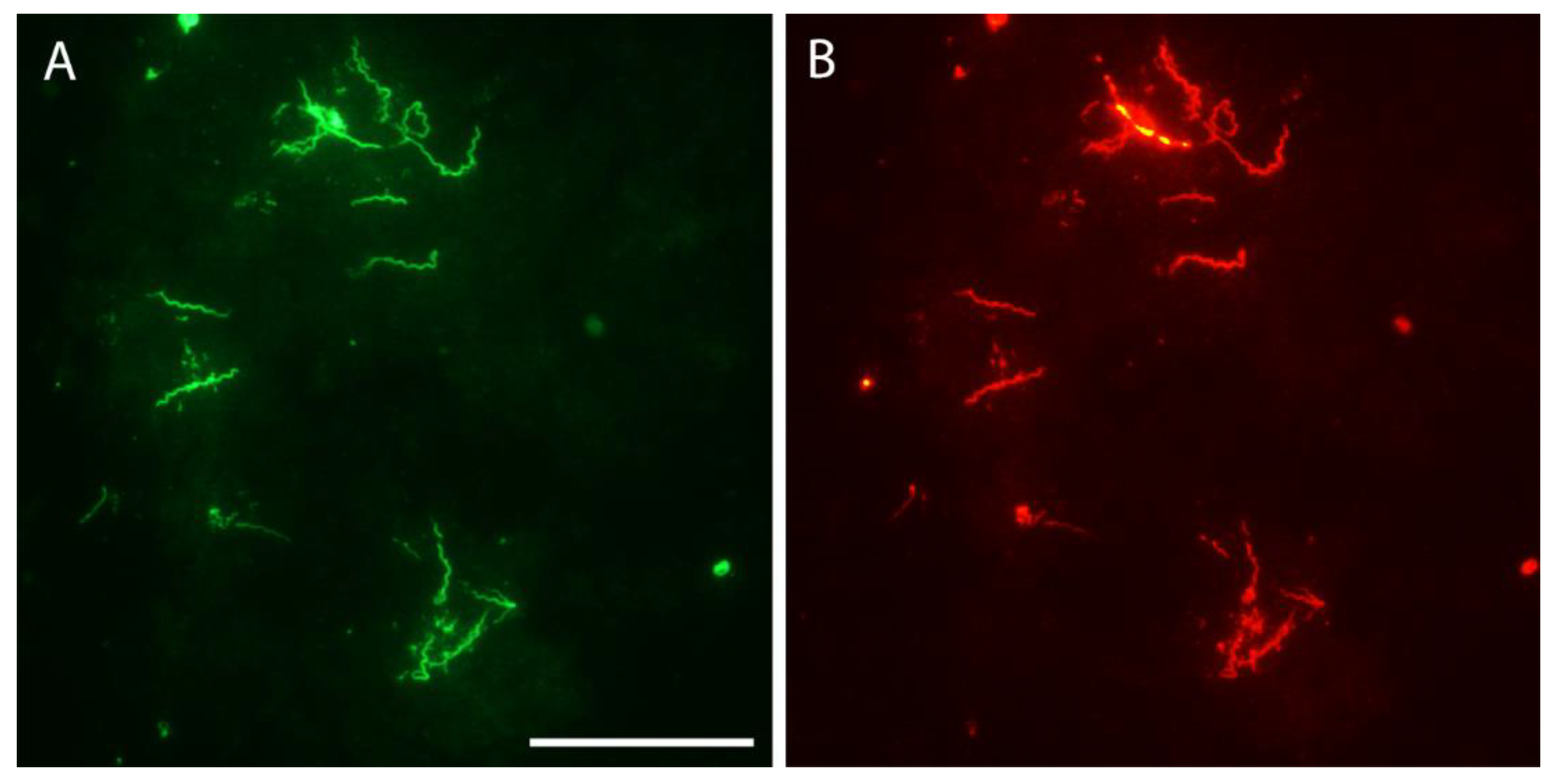
3.7. Microscopic Examination of Unfed Larvae
3.8. Tick Infection and Transovarial Passage of a Genomic Group I B. hermsii
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Smith, T.; Kilborne, F.L. Investigations into the Nature, Causation, and Prevention of Texas or Southern Cattle Fever; U.S. Department of Agriculture, Bureau of Animal Industry: Washington, DC, USA, 1893; Bull. 1; pp. 1–301. [Google Scholar]
- Dutton, J.E.; Todd, J.L. The Nature of Human Tick-Fever in the Eastern Part of the Congo Free State with Notes on the Distribution and Bionomics of the Tick; Memoirs of the Liverpool School of Tropical Medicine; Williams & Norgate: London, UK, 1905; pp. 1–18. [Google Scholar]
- Koch, R. Vorläufige Mitteilungen über die Ergebnisse einer Forchungsreise nach Ostafrika. Dtsch. Med. Wochenschr. 1905, 31, 1865–1869. [Google Scholar] [CrossRef] [Green Version]
- Koch, R. Ueber afrikanischen Recurrens. Berl. Klin. Wochenschr. 1906, 43, 185–194. [Google Scholar]
- Burgdorfer, W. Analyse des infektionsverlaufes bei Ornithodorus moubata (Murray) und der naturlichen uebertragung von Spirochaeta duttoni. Acta Trop. 1951, 8, 193–262. [Google Scholar] [PubMed]
- Felsenfeld, O. Borrelia. Strains, Vectors, Human and Animal Borreliosis; Warren H. Green, Inc.: St. Louis, MO, USA, 1971. [Google Scholar]
- Hoogstraal, H. Ticks and spirochetes. Acta Trop. 1979, 36, 133–136. [Google Scholar]
- Barbour, A.G.; Schwan, T.G. Borrelia, Swellengrebel 1907, 582AL. In Bergey’s Manual of Systematics of Archaea and Bacteria; Whitman, W.B., Rainey, R., Kämpfe, P.P., Trujillo, M., Chun, J., DeVos, P., Hedlund, B., Dedysh, S., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018; pp. 1–22. [Google Scholar]
- Dworkin, M.S.; Schwan, T.G.; Anderson, D.E.; Borchardt, S.M. Tick-borne relapsing fever. Infect. Dis. Clin. N. Am. 2008, 22, 449–468. [Google Scholar] [CrossRef] [Green Version]
- Davis, G.E. Species unity or plurality of the relapsing fever spirochetes. Am. Assoc. Adv. Sci. Monogr. 1942, 18, 41–47. [Google Scholar]
- Johnson, T.L.; Fischer, R.J.; Raffel, S.J.; Schwan, T.G. Host associations and genomic diversity of Borrelia hermsii in an endemic focus of tick-borne relapsing fever in western North America. Parasites Vectors 2016, 9, 575. [Google Scholar] [CrossRef] [Green Version]
- Herms, W.B.; Wheeler, C.M. Ornithodoros hermsi Wheeler as a vector of relapsing fever in California. J. Parasitol. 1936, 22, 276–282. [Google Scholar] [CrossRef]
- Wheeler, C.M. Progress of spirochaete infection in the developmental stages of the host tick, Ornithodoros hermsi, Wheeler. Am. J. Trop. Med. 1938, 18, 413–419. [Google Scholar] [CrossRef]
- Longanecker, D.S. Laboratory and field studies on the biology of the relapsing fever tick vector (Ornithodoros hermsi Wheeler) in the high mountains of California. Am. J. Trop. Med. 1951, 31, 373–380. [Google Scholar] [CrossRef]
- Schwan, T.G.; Raffel, S.J.; Schrumpf, M.E.; Webster, L.S.; Marques, A.R.; Spano, R.; Rood, M.; Burns, J.; Hu, R. Tick-borne relapsing fever and Borrelia hermsii, Los Angeles County, California, USA. Emerg. Infect. Dis. 2009, 15, 1026–1031. [Google Scholar] [CrossRef]
- Endris, R.G.; Haslett, T.M.; Monahan, M.J.; Hess, W.R.; Rutledge, L.C. Techniques for mass rearing soft ticks (Acari: Argasidae). J. Med. Entomol. 1986, 23, 225–229. [Google Scholar] [CrossRef]
- McCoy, B.N.; Raffel, S.J.; Lopez, J.E.; Schwan, T.G. Bloodmeal size and spirochete acquisition of Ornithodoros hermsi (Acari: Argasidae) during feeding. J. Med. Entomol. 2010, 47, 1164–1172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwan, T.G.; Hinnebusch, B.J. Bloodstream- versus tick-associated variants of a relapsing fever bacterium. Science 1998, 280, 1938–1940. [Google Scholar] [CrossRef] [Green Version]
- Porcella, S.F.; Raffel, S.J.; Anderson Jr, D.E.; Gilk, S.D.; Bono, J.L.; Schrumpf, M.E.; Schwan, T.G. Variable tick protein in two genomic groups of the relapsing fever spirochete Borrelia hermsii in western North America. Infect. Immun. 2005, 73, 6647–6658. [Google Scholar] [CrossRef] [Green Version]
- Schwan, T.G.; Raffel, S.J.; Schrumpf, M.E.; Porcella, S.F. Diversity and distribution of Borrelia hermsii. Emerg. Infect. Dis. 2007, 13, 436–442. [Google Scholar] [CrossRef]
- Möllers, B. Experimentelle Studien über die Uebertragung des Rückfallfiebers durch Zecken. Z. Hyg. Infektionskr. 1907, 58, 277–286. [Google Scholar] [CrossRef]
- Carter, R.M. The presence of Spirochaeta duttoni in the ova of Ornithodoros moubata. Ann. Trop. Med. Parasit. 1908, 1, 157–160. [Google Scholar]
- Geigy, R.; Wagner, O.; Aeschlimann, A. Transmission génitale de Borrelia duttoni chez Ornithodorus moubata. Acta Trop. 1954, 11, 81–82. [Google Scholar] [PubMed]
- Aeschlimann, A. Developpement embryonnaire d’Ornithodorus moubata (Murray) et transmission transovarienne de Borrelia duttoni. Acta Trop. 1958, 15, 15–64. [Google Scholar] [PubMed]
- Burgdorfer, W.; Varma, M.G.R. Trans-stadial and transovarial development of disease agents in arthropods. Ann. Rev. Entomol. 1967, 12, 347–376. [Google Scholar] [CrossRef]
- Telford, S.R.; Goethert, H.K. Perpetuation of Borreliae. Curr. Issues Mol. Biol. 2021, 42, 267–306. [Google Scholar]
- Pavlovsky, Y.N. Tick-borne relapsing fever. In Human Diseases with Natural Foci; Pavlovsky, Y.N., Ed.; Foreign Languages Publishing House: Moscow, Russia, 1963; pp. 138–184. [Google Scholar]
- Balashov, Y.S. Bloodsucking ticks (Ixodoidea)—Vectors of diseases of man and animals. Misc. Publ. Entomol. Soc. Am. 1972, 8, 161–376. [Google Scholar]
- Gaber, M.S.; Khalil, G.M.; Hoogstraal, H.; Aboul-Nasr, A.E. Borrelia crocidurae localization and transmission in Ornithodoros erraticus and O. savignyi. Parasitology 1984, 88, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Zaher, M.A.; Soliman, Z.R.; Diab, F.M. An experimental study of Borrelia anserina in four species of Argas ticks. 2. Transtadial survival and transovarial transmission. Z. Parasitenkd. 1977, 53, 213–223. [Google Scholar] [CrossRef]
- Brumpt, E. Étude du Spirochaeta turicatae, n. sp., agent de la fièvre récurrente sporadique des Etats-Unis transmise par Ornithodorus turicata. C. R. Soc. Biol. 1933, 113, 1369–1372. [Google Scholar]
- Brumpt, E. Fièvre récurrente sporadique des Etats-Unis due a Spirochoeta turicatae n. sp. et transmise dans la nature par Ornithodorus turicata. In Proceedings of the Octava Reunión de la Sociedad Argentina de Patologia Regional del Norte, Santiago, Chile, 2–3 October 1933; pp. 566–571. [Google Scholar]
- Francis, E. Rat-bite fever and relapsing fever in the United States. Trans. Assoc. Am. Physicians 1932, 47, 143–149. [Google Scholar]
- Francis, E. Longevity of the tick Ornithodoros turicata and of Spirochaeta recurrentis within this tick. Public Health Rep. 1938, 53, 2220–2241. [Google Scholar] [CrossRef]
- Davis, G.E. The identification of spirochetes from human cases of relapsing fever by xenodiagnoses with comments of local specificity of tick vectors. Exp. Parasitol. 1956, 5, 271–275. [Google Scholar] [CrossRef]
- Davis, G.E. Relapsing fever: The tick Ornithodoros turicata as a spirochetal reservoir. Public Health Rep. 1943, 58, 839–842. [Google Scholar] [CrossRef]
- Davis, G.E. Biology as an aid to the identification of two closely related species of ticks of the genus Ornithodoros. J. Parasitol. 1952, 38, 477–480. [Google Scholar] [CrossRef]
- Cooley, R.A.; Kohls, G.M. The Argasidae of North America, Central America and Cuba; The American Midland Naturalist. Monograph no. 1; Notre Dame University Press: Notre Dame, IN, USA, 1944; pp. 1–152. [Google Scholar]
- Mans, B.J.; Featherston, J.; Kvas, M.; Pillay, K.-A.; de Klerk, D.G.; Pienaar, R.; de Castro, M.H.; Schwan, T.G.; Lopez, J.E.; Teel, P.; et al. Argasid and ixodid systematics: Implications for soft tick evolution and systematics, with a new argasid species list. Ticks Tick-Borne Dis. 2019, 10, 219–240. [Google Scholar] [CrossRef]
- Schwan, T.G.; Raffel, S.J.; Schrumpf, M.E.; Policastro, P.F.; Rawlings, J.A.; Lane, R.S.; Breitschwerdt, E.B.; Porcella, S.F. Phylogenetic analysis of the spirochetes Borrelia parkeri and Borrelia turicatae and the potential for tick-borne relapsing fever in Florida. J. Clin. Microbiol. 2005, 43, 3851–3859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lane, R.S.; Burgdorfer, W.; Hayes, S.F.; Barbour, A.G. Isolation of a spirochete from the soft tick, Ornithodoros coriaceus: A possible agent of epizootic bovine abortion. Science 1985, 230, 85–87. [Google Scholar] [CrossRef]
- Lane, R.S.; Manweiler, S.A. Borrelia coriaceae in its tick vector, Ornithodoros coriaceus (Acari: Argasidae), with emphasis on transstadial and transovarial infection. J. Med. Entomol. 1988, 25, 172–177. [Google Scholar] [CrossRef]
- Davis, G.E. Relapsing fever: Ornithodoros hermsi a vector in Colorado. Public Health Rep. 1939, 54, 2178–2180. [Google Scholar] [CrossRef]
- Diehl, P.A.; Aeschlimann, A.; Obenchain, F.D. Tick reproduction: Oogenesis and oviposition. In Physiology of Ticks; Obenchain, F.D., Galun, R., Eds.; Pergamon Press Ltd.: Oxford, UK, 1982; pp. 277–350. [Google Scholar]
- Ogihara, M.H.; Taylor, D. Female reproduction system: Anatomy, physiology, and molecular biology. In Biology of Ticks, 2nd ed.; Sonenshine, D.E., Roe, R.M., Eds.; Oxford University Press: Oxford, UK, 2013; Volume 1, pp. 449–483. [Google Scholar]
- Wheeler, C.M. A new species of tick which is a vector of relapsing fever in California. Am. J. Trop. Med. 1935, 15, 435–438. [Google Scholar] [CrossRef]
- Raffel, S.J.; Battisti, J.M.; Fischer, R.J.; Schwan, T.G. Inactivation of genes for antigenic variation in the relapsing fever spirochete Borrelia hermsii reduces infectivity in mice and transmission by ticks. PLoS Pathog. 2014, 10, e1004056. [Google Scholar] [CrossRef] [Green Version]
- Rich, S.M.; Sawyer, S.A.; Barbour, A.G. Antigen polymorphism in Borrelia hermsii, a clonal pathogenic bacterium. Proc. Natl. Acad. Sci. USA 2001, 98, 15038–15043. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barbour, A.G.; Dai, Q.; Restrepo, B.I.; Stoenner, H.G.; Frank, S.A. Pathogen escape from host immunity by a genome program for antigenic variation. Proc. Natl. Acad. Sci. USA 2006, 103, 18290–18295. [Google Scholar] [CrossRef] [Green Version]
- Lopez, J.E.; McCoy, B.N.; Krajacich, B.J.; Schwan, T.G. Acquisition and subsequent transmission of Borrelia hermsii by the soft tick Ornithodoros hermsi. J. Med. Entomol. 2011, 48, 891–895. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krajacich, B.J.; Lopez, J.E.; Raffel, S.J.; Schwan, T.G. Vaccination with the variable tick protein of the relapsing fever spirochete Borrelia hermsii protects mice from infection by tick-bite. Parasites Vectors 2015, 8, 546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marcsisin, R.A.; Lewis, E.R.G.; Barbour, A.G. Expression of the tick-associated Vtp protein of Borrelia hermsii in a murine model of relapsing fever. PLoS ONE 2016, 11, e0153906. [Google Scholar]
- Schwan, T.G.; Raffel, S.J.; Battisti, J.M. Transgenic functional complementation with a transmission-associated protein restores spirochete infectivity by tick bite. Ticks Tick-Borne Dis. 2020, 11, 101377. [Google Scholar] [CrossRef]
- Christensen, J.; Fischer, R.J.; McCoy, B.N.; Raffel, S.J.; Schwan, T.G. Tickborne relapsing fever, Bitterroot Valley, Montana, USA. Emerg. Infect. Dis. 2015, 21, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Fukunaga, M.; Takahashi, Y.; Tsuruta, Y.; Matsushita, O.; Ralph, D.; McClelland, M.; Nakao, M. Genetic and phenotypic analysis of Borrelia miyamotoi sp. nov., isolated from the ixodid tick Ixodes persulcatus, the vector for Lyme disease in Japan. Int. J. Syst. Bacteriol. 1995, 45, 804–810. [Google Scholar] [CrossRef] [Green Version]
- Fukunaga, M.; Koreki, Y. The flagellin gene of Borrelia miyamotoi sp. nov. and its phylogenetic relationship among Borrelia species. FEMS Microbiol. Lett. 1995, 134, 255–258. [Google Scholar] [CrossRef]
- Laveran, A. Sur la spirillose des bovidés. C. R. Acad. Sci. 1903, 136, 939–941. [Google Scholar]
- Theiler, A. Spirillosis of cattle. J. Comp. Pathol. Ther. 1904, 17, 47–55. [Google Scholar] [CrossRef]
- Theiler, A. Transmission and Inoculability of Spirillum Theileri (Laveran). Proc. R. Soc. Lond. Ser. B Contain. Pap. Biol. Character 1905, 76, 504–506. [Google Scholar]
- Theiler, A. Transmission des spirilles et des piroplasmes par différentes espèces de tiques. Bull. Soc. Pathol. Exot. 1909, 2, 293–294. [Google Scholar]
- Smith, R.D.; Brener, J.; Osorno, M.; Ristic, M. Pathobiology of Borrelia theileri in the tropical cattle tick, Boophilus microplus. J. Invert. Pathol. 1978, 32, 182–190. [Google Scholar] [CrossRef]
- Smith, R.D.; Miranpuri, G.S.; Adams, J.H.; Ahrens, E.H. Borrelia theileri: Isolation from ticks (Boophilus microplus) and tick-borne transmission between splenectomized calves. Am. J. Vet. Res. 1985, 46, 1396–1398. [Google Scholar] [PubMed]
- Cutler, S.; Vayssier-Taussat, M.; Estrada-Peña, A.; Potkonjak, A.; Mihalca, A.D.; Zeller, H. A new Borrelia on the block: Borrelia miyamotoi—A human health risk? Eurosurveillance 2019, 24, 1800170. [Google Scholar] [CrossRef] [Green Version]
- Wagemakers, A.; Staarink, P.J.; Sprong, H.; Hovius, J.W.R. Borrelia miyamotoi: A widespread tick-borne relapsing fever spirochete. Trends Parasitol. 2015, 31, 260–269. [Google Scholar] [CrossRef]
- Richter, D.; Debski, A.; Hubalek, Z.; Matuschka, F.-R. Absence of Lyme disease spirochetes in larval Ixodes ricinus ticks. Vector Borne Zoonotic Dis. 2012, 12, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Rollend, L.; Fish, D.; Childs, J.E. Transovarial transmission of Borrelia spirochetes by Ixodes scapularis: A summary of the literature and recent observations. Ticks Tick-Borne Dis. 2013, 4, 46–51. [Google Scholar] [CrossRef]
- Scoles, G.A.; Papero, M.; Beati, L.; Fish, D. A relapsing fever group spirochete transmitted by Ixodes scapularis ticks. Vector Borne Zoonotic Dis. 2001, 1, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Lubelczyk, C.; Hickling, G.J.; Belperron, A.A.; Bockenstedt, L.K.; Tsao, J.I. Vertical transmission rates of Borrelia miyamotoi in Ixodes scapularis collected from white-tailed deer. Ticks Tick-Borne Dis. 2019, 10, 682–689. [Google Scholar] [CrossRef]
- Lynn, G.E.; Breuner, N.E.; Eisen, L.; Hojgaard, A.; Replogle, A.J.; Eisen, R.J. An immunocompromised mouse model to infect Ixodes scapularis ticks with the relapsing fever spirochete, Borrelia miyamotoi. Ticks Tick-Borne Dis. 2019, 10, 352–359. [Google Scholar] [CrossRef]
- Keesing, F.; McHenry, D.J.; Hersh, M.H.; Ostfeld, R.S. Spatial and temporal patterns of the emerging tick-borne pathogen Borrelia miyamotoi in blacklegged ticks (Ixodes scapularis) in New York. Parasites Vectors 2021, 14, 51. [Google Scholar] [CrossRef]
- Van Duijvendijk, G.; Coipan, C.; Wagemakers, A.; Fonville, M.; Ersöz, J.; Oei, A.; Földvári, G.; Hovius, J.; Takken, W.; Sprong, H. Larvae of Ixodes ricinus transmit Borrelia afzelii and B. miyamotoi to vertebrate hosts. Parasites Vectors 2016, 9, 97. [Google Scholar] [CrossRef] [Green Version]
- Stromdahl, E.Y.; Williamson, P.C.; Kollars, T.M., Jr.; Evans, S.R.; Barry, R.K.; Vince, M.A.; Dobbs, N.A. Evidence of Borrelia lonestari DNA in Amblyomma americanum (Acari: Ixodidae) removed from humans. J. Clin. Microbiol. 2003, 41, 5557–5562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Platonov, A.E.; Karan, L.S.; Kolyasnikova, N.M.; Makhneva, N.A.; Toporkova, M.G.; Maleev, V.V.; Fish, D.; Krause, P.J. Humans infected with relapsing fever spirochete Borrelia miyamotoi, Russia. Emerg. Infect. Dis. 2011, 17, 1816–1823. [Google Scholar] [CrossRef] [PubMed]
- Gugliotta, J.L.; Goethert, H.K.; Berardi, V.P.; Telford, S.R., III. Meningoencephalitis from Borrelia miyamotoi in an immunocompromised patient. N. Engl. J. Med. 2013, 368, 240–245. [Google Scholar] [CrossRef] [Green Version]
- Krause, P.J.; Narasimhan, S.; Wormser, G.P.; Rollend, L.; Fikrig, E.; Lepore, T.; Barbour, A.; Fish, D. Human Borrelia miyamotoi infection in the United States. N. Engl. J. Med. 2013, 368, 291–293. [Google Scholar] [CrossRef] [Green Version]
- Hovius, J.W.R.; de Wever, B.; Sohne, M.; Brouwer, M.C.; Coumou, J.; Wagemakers, A.; Oei, A.; Knol, H.; Narasimhan, S.; Hodiamont, C.J.; et al. A case of meningoencephalitis by the relapsing fever spirochaete Borrelia miyamotoi in Europe. Lancet 2013, 382, 658. [Google Scholar] [CrossRef] [Green Version]
- Breuner, N.E.; Hojgaard, A.; Replogle, A.J.; Boegler, K.A.; Eisen, L. Transmission of the relapsing fever spirochete, Borrelia miyamotoi, by single transovarially-infected larval Ixodes scapularis ticks. Ticks Tick-Borne Dis. 2018, 9, 1464–1467. [Google Scholar] [CrossRef] [PubMed]
- Piesman, J.; Schwan, T.G. Ecology of borreliae and their arthropod vectors. In Borrelia: Molecular Biology, Host Interaction and Pathogenesis; Samuels, D.S., Radolf, J.D., Eds.; Caister Academic Press: Norfolk, UK, 2010; pp. 251–278. [Google Scholar]
- Replogle, A.J.; Sexton, C.; Young, J.; Kingry, L.C.; Schriefer, M.E.; Dolan, M.; Johnson, T.L.; Connally, N.P.; Padgett, K.A.; Petersen, J.M. Isolation of Borrelia miyamotoi and other borreliae using a modified BSK medium. Sci. Rep. 2021, 11, 1926. [Google Scholar] [CrossRef]
- Mount, G.A.; Haile, D.G.; Daniels, E. Simulation of blacklegged tick (Acari: Ixodidae) population dynamics and transmission of Borrelia burgdorferi. J. Med. Entomol. 1997, 34, 461–484. [Google Scholar] [CrossRef]
- Randolph, S.E. The relative contributions of transovarial and transstadial transmission to the maintenance of tick-borne diseases. In Lyme Borreliosis; Axford, J.S., Rees, D.H.E., Eds.; Plenum Press: New York, NY, USA, 1994; pp. 131–134. [Google Scholar]
- Johnson, T.L.; Landguth, E.L.; Stone, E.F. Modeling relapsing disease dynamics in a host-vector community. PLoS Negl. Trop. Dis. 2016, 10, e0004428. [Google Scholar] [CrossRef] [PubMed]




| Founding Female History | Larval Assays | Nymphal Assays | |||||
|---|---|---|---|---|---|---|---|
| Feeding | Date Fed | Transmission to Mouse | DF a | Number Fed | Transmission to Mouse | Stage Fed b | Transmission to Mouse |
| 1st | 4 March 2009 | ND c | 2/2 | 33 | ND | N2 | + d |
| 2nd | 16 December 2009 | ND | 25 | + | N2, N3 | +,+ | |
| 3rd | 11 April 2010 | + | 35 | ND | N1 | + | |
| 4th | 27 August 2010 | ND | 18 | ND | N2, N3 | +,+ | |
| 5th | 27 June 2011 | ND | 1/1 | 45 | + | N1, N2, N3 | +,+,+ |
| 6th | 18 December 2011 | + | 1/1 | 27 | + | N1, N2, N3 | +,+,+ |
| 7th | 27 April 2012 | + | 2/2 | 6 | + | N1 | + |
| 8th | 8 November 2012 | - e | 0 f | NA g | 0 | NA | |
| 9th | 16 April 2013 h | - | 0 | NA | 0 | NA | |
| Female ID | Clutch Origin a | Date Female First Fed | Infection by Feeding | Larval Cohorts Infected b,c |
|---|---|---|---|---|
| F-1A | First | 25 January 2011 | + | 2/2 |
| F-1B | First | “ | + | 4/5 |
| F-1C | First | “ | + | 3/3 |
| F-1D | First | 6 August 2011 | + | 3/3 |
| F-1E | First | “ | + | 3/3 |
| F-1F | First | “ | + | 1/1 |
| F-1G | First | “ | - | 2/2 |
| F-1H | First | 1 September 2011 | + | None Produced |
| F-II | First | “ | + | 4/4 |
| F-1J | Second | 10 November 2012 | + | 2/2 |
| F-1K | Second | “ | + | 1/2 |
| F-1L | Second | “ | + | 0/2 |
| F-1M | Second | “ | + | 2/2 |
| F-1N | Second | “ | + | 1/1 |
| F-1O | Second | “ | + | 0/2 |
| F-1P | Second | 16 April 2013 | + | 1/2 |
| F-1Q | Second | “ | + | 1/1 |
| F-1R | Second | 11 December 2012 | + | 2/2 |
| F-1S | Second | 1 March 2013 | + | 1/1 |
| Totals | 18/19 | 33/40 (82.5%) |
| Dates Larvae Fed | Source Females a | Larval Cohorts Fed | Larvae Per Cohort | Larval Cohorts Infected | Percentage Cohorts Infected |
|---|---|---|---|---|---|
| April 2010–December 2011 | 4 | 5 | 25–60 | 5 | 100% |
| April–November 2012 | 7 | 9 | 6–70 | 9 | 100% |
| July–September 2013 | 18 | 18 | 5–70 | 15 | 83.3% |
| October 2014 | 13 | 13 | 14–57 | 9 | 69.2% |
| Total | 20 | 45 b | 38 | 84.4% |
| Single Ticks Fed | Pooled Ticks Fed | |||||
|---|---|---|---|---|---|---|
| Dates Ticks Fed | Number Ticks Infected | Number Ticks Fed | % Infected | Number Pools Infected | Number Pools Fed | % Infected |
| June 2007–December 2011 | 43 | 47 | 91.5% | 14 | 15 | 93.3% |
| April–December 2012 | 24 | 26 | 92.3% | 28 | 30 | 93.3% |
| March–December 2013 | 18 | 26 | 69.2% | 55 | 71 | 77.5% |
| April–December 2014 | 3 | 4 | 75.0% | 29 | 37 | 78.4% |
| Total | 88 | 103 a | 85.4% | 126 | 153 b | 82.4% |
| Female ID | Eggs Infected/Eggs Examined | Average Number Spirochetes Per Egg (Range) | Number of Larvae Fed in Cohort | Larval Cohort Transmission a |
|---|---|---|---|---|
| F-1C | 15/38 | 25.5 (2–128) | 23 | + |
| F-1E | 13/24 | 43.0 (1–110) | 5 | + |
| F-1F | 11/12 | 97.0 b (15–TNC) | 29 | + |
| F-1S | 3/24 | 3.7 (1–9) | 8 | + |
| F-1G | 0/12 | 0 | 17 | + |
| F-1M | 0/12 | 0 | 41 | + |
| F-1P | 0/12 | 0 | 15 | + |
| F-1Q | 0/12 | 0 | 34 | + |
| F-1R | 0/12 | 0 | 35 | + |
| F-2A | 0/5 | 0 | 27 | + |
| F-1B | 0/6 | 0 | 9 | - |
| F-1L | 0/12 | 0 | 22 | - |
| F-1O | 0/15 | 0 | 18 | - |
| Totals | 42/196 (21.4%) |
| Female a | Number Larvae Infected/ Number Larvae Examined b | Percentage Larvae Infected |
|---|---|---|
| F-1F | 8/12 | 66.7% |
| F-1I | 9/12 | 75% |
| F-1J | 2/12 | 16.7% |
| F-1Q | 10/12 | 83.3% |
| F-1T | 4/12 | 33.3% |
| Total | 33/60 | 55% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwan, T.G.; Raffel, S.J. Transovarial Transmission of Borrelia hermsii by Its Tick Vector and Reservoir Host Ornithodoros hermsi. Microorganisms 2021, 9, 1978. https://doi.org/10.3390/microorganisms9091978
Schwan TG, Raffel SJ. Transovarial Transmission of Borrelia hermsii by Its Tick Vector and Reservoir Host Ornithodoros hermsi. Microorganisms. 2021; 9(9):1978. https://doi.org/10.3390/microorganisms9091978
Chicago/Turabian StyleSchwan, Tom G., and Sandra J. Raffel. 2021. "Transovarial Transmission of Borrelia hermsii by Its Tick Vector and Reservoir Host Ornithodoros hermsi" Microorganisms 9, no. 9: 1978. https://doi.org/10.3390/microorganisms9091978





