Metataxonomic Analysis of Bacteria Entrapped in a Stalactite’s Core and Their Possible Environmental Origins
Abstract
1. Introduction
2. Material and Methods
2.1. Study Site
2.2. Sample Collection
2.3. DNA Extraction and DNA Sequence Analysis
2.4. Bioinformatics and Statistical Analysis
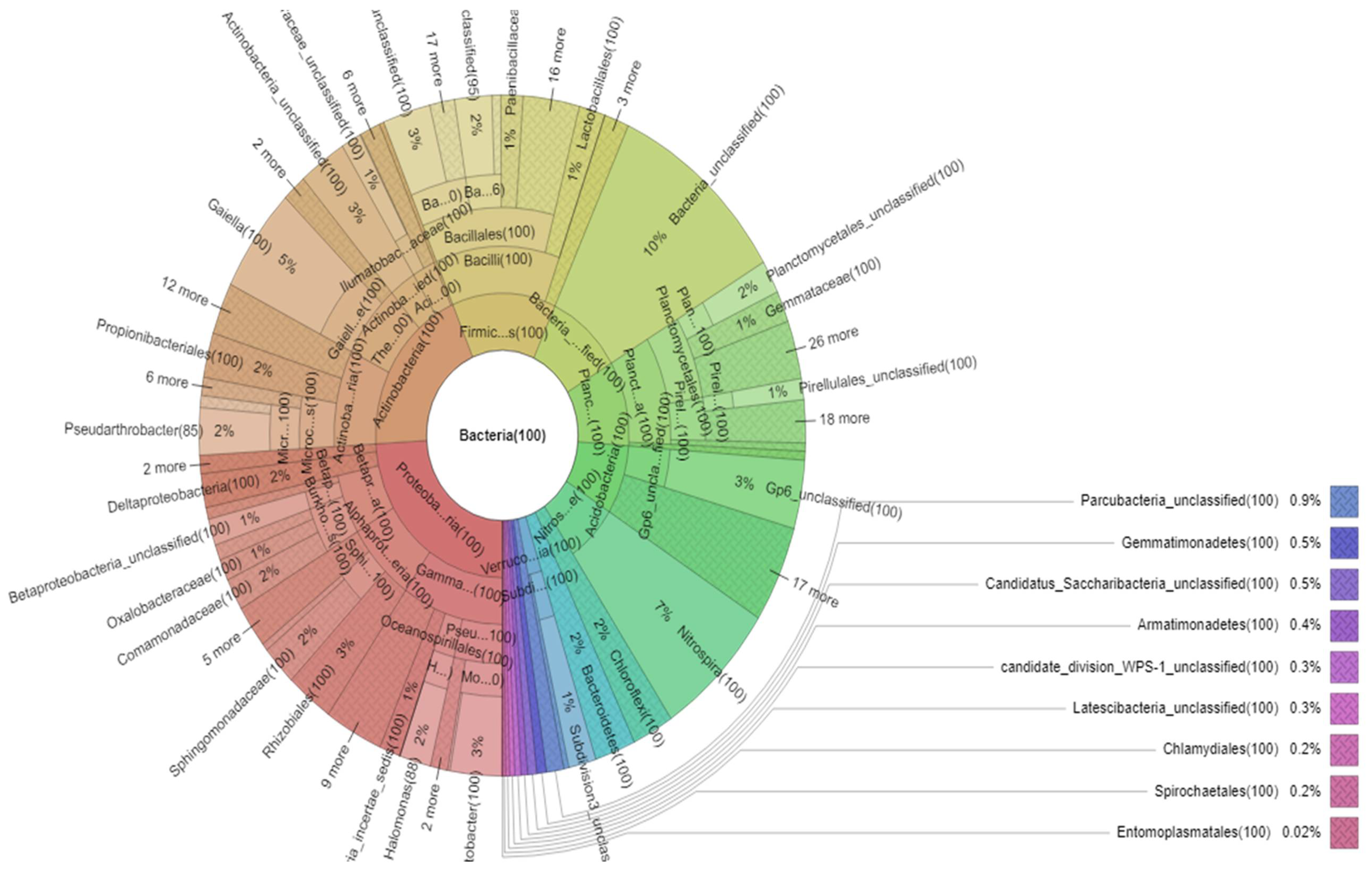
3. Results
3.1. Bacterial Communities Encapsulated during the Formation of Stalactite
3.2. Proteobacteria
3.3. α-Proteobacteria
3.4. β-Proteobacteria
3.5. γ-Proteobacteria
3.6. Actinobacteria
3.7. Firmicutes
3.8. Acidobacteria
3.9. Bacteroidetes
3.10. Verrucomicrobia
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vanderwolf, K.J.; Malloch, D.; McAlpine, D.F.; Forbes, G.J. A world review of fungi, yeasts, and slime molds in caves. Int. J. Speleol. 2013, 42, 77–96. [Google Scholar] [CrossRef]
- Mason-Williams, A.; Benson-Evans, K. Summary of results obtained during a preliminary investigation into the bacterial and botanical flora of caves in South Wales. Int. J. Speleol. 1967, 2, 397–402. [Google Scholar] [CrossRef][Green Version]
- Boere, A.C.; Abbas, B.; Rijpstra, W.I.C.; Versteegh, G.J.M.; Volkman, J.K.; Sinninghe Damsté, J.S.; Coolen, M.J.L. Late-Holocene succession of dinoflagellates in an Antarctic fjord using a multi-proxy approach: Paleoenvironmental genomics, lipid biomarkers and palynomorphs. Geobiology 2009, 7, 265–281. [Google Scholar] [CrossRef] [PubMed]
- Coolen, M.J.L. 7000 years of Emiliania huxleyi viruses in the Black Sea. Science 2011, 22, 451–452. [Google Scholar] [CrossRef] [PubMed]
- Coolen, M.J.; Overmann, J. 217 000-year-old DNA sequences of green sulfur bacteria in Mediterranean sapropels and their implications for the reconstruction of the paleoenvironment. Environ. Microbiol. 2007, 9, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Yun, Y.; Wang, H.; Man, B.; Xiang, X.; Zhou, J.; Qiu, X.; Duan, Y.; Engel, A.S. The relationship between pH and bacterial communities in a single karst ecosystem and its implication for soil acidification. Front. Microbiol. 2016, 7, 1955. [Google Scholar] [CrossRef] [PubMed]
- Affolter, S.; Hauselmann, A.; Fleitmann, D.; Edwards, R.L.; Cheng, H.; Leuenberger, M. Central Europe temperature constrained by speleothem fluid inclusion water isotopes over the past 14,000 years. Sci. Adv. 2019, 5, aav3809. [Google Scholar] [CrossRef]
- Sankaranarayanan, K.; Timofeeff, M.N.; Spathis, R.; Lowenstein, T.K.; Lum, J.K. Ancient microbes from halite fluid inclusions: Optimized surface sterilization and DNA extraction. PLoS ONE 2011, 6, e20683. [Google Scholar] [CrossRef]
- Jones, B.; Renaut, R.W.; Rosen, M.R. Microbial construction of siliceous stalactites at geysers and hot springs: Examples from the Whakarewarewa Geothermal Area, North Island, New Zealand. Palaios 2001, 16, 73–94. [Google Scholar] [CrossRef]
- Jones, B. The preferential association of dolomite with microbes in stalactites from Cayman Brac, British West Indies. Sediment. Geol. 2010, 226, 94–109. [Google Scholar] [CrossRef]
- Pacton, M.; Breitenbach, S.F.; Lechleitner, F.A.; Vaks, A.; Rollion-Bard, C.; Gutareva, O.S.; Osintcev, A.V.; Vasconcelos, C. The role of microorganisms in the formation of a stalactite in Botovskaya Cave, Siberia–paleoenvironmental implications. Biogeosciences 2013, 10, 6115–6130. [Google Scholar] [CrossRef]
- Borgonie, G.; Linage-Alvarez, B.; Ojo, A.; Shivambu, S.; Kuloyo, O.; Cason, E.D.; Maphanga, S.; Vesmeulen, J.-G.; Litthauer, D.; Ralston, S.D.; et al. Deep subsurface mine stalactites trap endemic fissure fluid Archaea, Bacteria, and Nematoda possibly originating from ancient seas. Front. Microbiol. 2015, 6, 833. [Google Scholar] [CrossRef] [PubMed]
- Dhami, N.K.; Mukherjee, A.; Watkin, E.L. Microbial diversity and mineralogical-mechanical properties of calcitic cave speleothems in natural and in vitro biomineralization conditions. Front. Microbiol. 2018, 9, 40. [Google Scholar] [CrossRef]
- Candiroglu, B.; Gungor, N.D. Cave ecosystems: Microbiological view. Eur. J. Biol. 2017, 76, 36–42. [Google Scholar] [CrossRef]
- Gabriel, C.R.; Northup, D.E. Microbial ecology: Caves as an extreme habitat. In Cave Microbiomes: A Novelresource for Drug Discovery; Springer: New York, NY, USA, 2013; pp. 85–108. [Google Scholar] [CrossRef]
- Zhu, H.Z.; Zhang, Z.F.; Zhou, N.; Jiang, C.Y.; Wang, B.J.; Cai, L.; Liu, S.J. Diversity, distribution and co-occurrence patterns of bacterial communities in a karst cave system. Front. Microbiol. 2019, 10, 1726. [Google Scholar] [CrossRef]
- De Mandal, S.; Chatterjee, R.; Kumar, N.S. Dominant bacterial phyla in caves and their predicted functional roles in C and N cycle. BMC Microbiol. 2017, 17, 90. [Google Scholar] [CrossRef]
- Thompson, T.P.; Kelly, S.A.; Skvortsov, T.; Plunkett, G.; Ruffell, A.; Hallsworth, J.E.; Hopps, J.; Gilmore, B.F. Microbiology of a NaCl stalactite ‘salticle’ in Triassic halite. Environ. Microbiol. 2021, 23, 3881–3895. [Google Scholar] [CrossRef]
- Adler, C.J.; Dobney, K.; Weyrich, L.S.; Kaidonis, J.; Walker, A.W.; Haak, W. Sequencing ancient calcified dental plaque shows changes in oral microbiota with dietary shifts of the neolithic and industrial revolutions. Nat. Genet. 2013, 45, 450e–455e. [Google Scholar] [CrossRef]
- Warinner, C.; Rodrigues, J.F.M.; Vyas, R.; Trachsel, C.; Shved, N.; Grossmann, J. Pathogens and host immunity in the ancient human oral cavity. Nat. Genet. 2014, 46, 336–344. [Google Scholar] [CrossRef]
- Harkins, K.M.; Stone, A.C. Ancient pathogen genomics: Insights into timing and adaptation. J. Hum. Evol. 2015, 79, 137–149. [Google Scholar] [CrossRef]
- Arriola, L.A.; Cooper, A.; Weyrich, L.S. Palaeomicrobiology: Application of ancient DNA sequencing to better understand bacterial genome evolution and adaptation. Front. Ecol. Evol. 2020, 16, 40. [Google Scholar] [CrossRef]
- Durazzi, F.; Sala, C.; Castellani, G.; Manfreda, G.; Remondini, D.; De Cesare, A. Comparison between 16S rRNA and shotgun sequencing data for the taxonomic characterization of the gut microbiota. Sci. Rep. 2021, 11, 3030. [Google Scholar] [CrossRef] [PubMed]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef]
- Kozich, J.J.; Westcott, S.L.; Baxter, N.T.; Highlander, S.K.; Schloss, P.D. Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl. Environ. Microbiol. 2013, 79, 5112–5120. [Google Scholar] [CrossRef]
- Afgan, E.; Baker, D.; Batut, B.; van den Beek, M.; Bouvier, D.; Čech, M.; Chilton, J.; Clements, D.; Coraor, N.; Grüning, B.; et al. The Galaxy platform for accessible, reproducible and collabo-rative biomedical analyses: 2018 update. Nucleic Acids Res. 2018, 46, W537–W544. [Google Scholar] [CrossRef] [PubMed]
- Balkwill, D.L.; Fredrickson, J.K.; Romine, M.F. Sphingomonas and Related Genera (No. PNNL-SA-39356); Pacific Northwest National Lab. (PNNL): Richland, WA, USA, 2003. [Google Scholar]
- de Souza, J.A.M.; Alves, L.M.C.; de Mello Varani, A.; de Macedo Lemos, E.G. The family bradyrhizobiaceae. In The Prokaryotes; Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., Thompson, F., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 135–154. [Google Scholar]
- Alves, L.M.C.; de Souza, J.A.M.; de Mello Varani, A.; de Macedo Lemos, E.G. The family rhizobiaceae. In The Prokaryotes; Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., Thompson, F., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 419–437. [Google Scholar]
- La Scola, B.; Birtles, R.J.; Mallet, M.N.; Raoult, D. Massilia timonae gen. nov., sp. nov., isolated from blood of an immunocompromised patient with cerebellar lesions. J. Clin. Microbiol. 1998, 36, 2847–2852. [Google Scholar] [CrossRef]
- Irgens, R.L.; Gosink, J.J.; Staley, J.T. Polaromonas vacuolata gen. nov., sp. nov., a psychrophilic, marine, gas vacuolate bacterium from Antarctica. Int. J. Syst. Bacteriol. 1996, 46, 822–826. [Google Scholar] [CrossRef][Green Version]
- Gawor, J.; Grzesiak, J.; Sasin-Kurowska, J.; Borsuk, P.; Gromadka, R.; Górniak, D.; Świątecki, A.; Aleksandrzak-Piekarczyk, T.; Zdanowski, M.K. Evidence of adaptation, niche separation and microevolution within the genus Polaromonas on Arctic and Antarctic glacial surfaces. Extremophiles 2016, 20, 403–413. [Google Scholar] [CrossRef]
- Yabuuchi, E.; Kosako, Y.; Yano, I.; Hotta, H.; Nishiuchi, Y. Transfer of two Burkholderia and an Alcaligenes species to Ralstonia gen. nov.: Proposal of Ralstonia pickettii (Ralston, Palleroni and Doudoroff 1973) comb. nov., Ralstonia solanacearum (Smith 1896) comb. nov. and Ralstonia eutropha (Davis 1969) comb. nov. Microbiol. Immunol. 1995, 39, 897–904. [Google Scholar] [CrossRef]
- Towner, K.J. The genus acinetobacter. In The Prokaryotes; Springer: New York, NY, USA, 1992; pp. 3137–3143. [Google Scholar]
- Madigan, M.; Martinko, J. Brock Biology of Microorganisms, 11th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2005; ISBN 0-13-144329-1. [Google Scholar]
- Vreeland, R.H.; Litchfield, C.D.; Martin, E.L.; Elliot, E. Halomonas elongata, a new genus and species of extremely salt-tolerant bacteria. Int. J. Syst. Evol. Microbiol. 1980, 30, 485–495. [Google Scholar] [CrossRef]
- Reichenbach, H. The genus lysobacter. In The Prokaryotes; Springer: New York, NY, USA, 1992; pp. 3256–3275. [Google Scholar]
- Welch, R.A. The genus escherichia. In The Prokaryotes; Springer: New York, NY, USA, 2006; pp. 60–71. [Google Scholar]
- Grimont, P.A.; Grimont, F. The genus serratia. Annu. Rev. Microbiol. 1978, 32, 221–248. [Google Scholar] [CrossRef]
- Robinson, R.K. Encyclopedia of Food Microbiology; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Im, W.T.; Liu, Q.M.; Yang, J.E.; Kim, M.S.; Kim, S.Y.; Lee, S.T.; Yi, T.H. Panacagrimonas perspica gen. nov., sp. nov., a novel member of Gammaproteobacteria isolated from soil of a ginseng field. J. Microbiol. 2010, 48, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Takai, K.; Miyazaki, M.; Hirayama, H.; Nakagawa, S.; Querellou, J.; Godfroy, A. Isolation and physiological characterization of two novel, piezophilic, thermophilic chemolithoautotrophs from a deep-sea hydrothermal vent chimney. Environ. Microbiol. 2009, 11, 1983–1997. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, N.; Guo, X.; Zhang, Y.; Ye, B. Comparative analysis of bacterial community structure in the rhizo-sphere of maize by high-throughput pyrosequencing. PLoS ONE 2017, 12, e0178425. [Google Scholar] [CrossRef]
- Johnston-Monje, D.; Lundberg, D.S.; Lazarovits, G.; Reis, V.M.; Raizada, M.N. Bacterial populations in juvenile maize rhizospheres originate from both seed and soil. Plant Soil 2016, 405, 337–355. [Google Scholar] [CrossRef]
- Smil, V. Nitrogen in crop production: An account of global flows. Glob. Biogeochem. Cycles 1999, 13, 647–662. [Google Scholar] [CrossRef]
- Anandan, R.; Dharumadurai, D.; Manogaran, G.P. Anandan, R.; Dharumadurai, D.; Manogaran, G.P. An introduction to actinobacteria. In Actinobacteria-Basics and Biotechnological Applications; IntechOpen: London, UK, 2016; Available online: https://www.intechopen.com/chapters/49873 (accessed on 18 June 2021).
- Banks, E.D.; Taylor, N.M.; Gulley, J.; Lubbers, B.R.; Giarrizzo, J.G.; Bullen, H.A.; Hoehler, T.M.; Barton, H.A. Bacterial calcium carbonate precipitation in cave environments: A function of calcium homeostasis. Geomicrobiol. J. 2010, 27, 444–454. [Google Scholar] [CrossRef]
- Pašić, L.; Kovce, B.; Sket, B.; Herzog-Velikonja, B. Diversity of microbial communities colonizing the walls of a Karstic cave in Slovenia. FEMS Microbiol. Ecol. 2010, 71, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Marques, E.L.; Silva, G.S.; Dias, J.C.; Gross, E.; Costa, M.S.; Rezende, R.P. Cave drip water-related samples as a natural environment for aromatic hydrocarbon-degrading bacteria. Microorganisms 2019, 7, 33. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Herraiz, M.; Jurado, V.; Cuezva, S.; Laiz, L.; Pallecchi, P.; Tiano, P.; Sanchez-Moral, S.; Saiz-Jimenez, C. Deterioration of an Etruscan tomb by bacteria from the order Rhizobiales. Sci. Rep. 2014, 4, 3610. [Google Scholar] [CrossRef] [PubMed]
- Novello, G.; Gamalero, E.; Bona, E.; Boatti, L.; Mignone, F.; Massa, N.; Cesaro, P.; Lingua, G.; Berta, G. The rhizosphere bacterial microbiota of Vitis vinifera cv. Pinot Noir in an integrated pest management vineyard. Front. Microbiol. 2017, 8, 1528. [Google Scholar] [CrossRef] [PubMed]
- Floc’h, J.B.; Hamel, C.; Lupwayi, N.; Harker, K.N.; Hijri, M.; St-Arnaud, M. Bacterial communities of the canola rhizosphere: Network analysis reveals a core bacterium shaping microbial interactions. Front. Microbiol. 2020, 11, 1587. [Google Scholar] [CrossRef] [PubMed]
- Nordstedt, N.P.; Jones, M.L. Isolation of rhizosphere bacteria that improve quality and water stress tolerance in greenhouse ornamentals. Front. Plant Sci. 2020, 11, 826. [Google Scholar] [CrossRef]
- Lazcano, C.; Boyd, E.; Holmes, G.; Hewavitharana, S.; Pasulka, A.; Ivors, K. The rhizosphere microbiome plays a role in the resistance to soil-borne pathogens and nutrient uptake of strawberry cultivars under field conditions. Sci. Rep. 2021, 11, 3188. [Google Scholar] [CrossRef]
- Zhao, Y.; Fu, W.; Hu, C.; Chen, G.; Xiao, Z.; Chen, Y.; Wang, Z.; Cheng, H. Variation of rhizosphere microbial community in continuous mono-maize seed production. Sci. Rep. 2021, 11, 1544. [Google Scholar] [CrossRef]
- White, W.B. Paleoclimate records from speleothems in limestone caves. In Studies of Cave Sediments; Springer: Dordrecht, The Netherlands, 2007; pp. 135–175. [Google Scholar] [CrossRef]
- Willems, A. The family comamonadaceae. In The Prokaryotes: Alphaproteobacteria and Betaproteobacteria; Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., Thompson, F., Eds.; Springer: Berlin, Germany, 2014; pp. 777–843. [Google Scholar]
- Smarž, J.; Kováč, L.; Mikeš, J.; Lukešová, A. Microwhip scorpions (palpigradi) feed on heterotrophic cyanobacteria in Slovak caves—A curiosity among arachnida. PLoS ONE 2013, 8, e75989. [Google Scholar] [CrossRef]
- Barton, H.A.; Jurado, V. What’s up down there? Microbial diversity in caves. Microbe 2007, 2, 132–138. [Google Scholar]
- Czerwik-Marcinkowska, J.; Massalski, A. Diversity of Cyanobacteria on Limestone Caves, Cyanobacteria; Tiwari, A., Ed.; IntechOpen: London, UK, 2018; Available online: https://www.intechopen.com/chapters/62780 (accessed on 18 June 2021).
- Czerwik-Marcinkowska, J.; Mrozińska, T. Algae and cyanobacteria in caves of the Polish Jura. Pol. Bot. J. 2011, 56, 203–243. [Google Scholar]
- Lamprinou, V.; Tryfinopoulou, K.; Velonakis, E.N.; Vatopoulos, A.; Antonopoulou, S.; Fragopoulou, E.; Pantazidou, A.; Economou-Amilli, A. Cave cyanobacteria showing activity. Int. J. Speleol. 2015, 44, 231–238. [Google Scholar] [CrossRef]
- Henrici, A.T.; Johnson, D.E. Studies of freshwater bacteria. II: Stalked bacteria, a new order of Schizomycetes. J. Bacteriol. 1935, 30, 61–93. [Google Scholar] [CrossRef]
- Liu, Y.; Yao, T.; Jiao, N.; Kang, S.; Zeng, Y.; Huang, S. Microbial community structure in moraine lakes and glacial meltwaters, Mount Everest. FEMS Microbiol. Lett. 2006, 265, 98–105. [Google Scholar] [CrossRef]
- Schlesner, H.; Jenkins, C.; Staley, J.T. The phylum verrucomicrobia: A phylogenetically heterogeneous bacterial group. Prokaryotes 2006, 7, 881–896. [Google Scholar] [CrossRef]
- Tierney, J.E.; Poulsen, C.J.; Montañez, I.P.; Bhattacharya, T.; Feng, R.; Ford, H.L.; Hönisch, B.; Inglis, G.N.; Petersen, S.V.; Sagoo, N.; et al. Past climates inform our future. Science 2020, 370, eaay3701. [Google Scholar] [CrossRef] [PubMed]
- Dawson, W.; Hör, J.; Egert, M.; van Kleunen, M.; Pester, M. A small number of low-abundance bacteria dominate plant species-specific responses during rhizosphere colonization. Front. Microbiol. 2017, 8, 975. [Google Scholar] [CrossRef] [PubMed]
- Troussellier, M.; Escalas, A.; Bouvier, T.; Mouillot, D. Sustaining rare marine microorganisms: Macroorganisms as repositories and dispersal agents of microbial diversity. Front. Microbiol. 2017, 8, 947. [Google Scholar] [CrossRef]
- Fernández-Gómez, B.; Díez, B.; Polz, M.F.; Arroyo, J.I.; Alfaro, F.D.; Marchandon, G.; Sanhueza, C.; Farias, L.; Trefault, N.; Marquet, P.A.; et al. Bacterial community structure in a sympagic habitat expanding with global warming: Brackish ice brine at 85–90 °N. ISME J. 2019, 13, 316–333. [Google Scholar] [CrossRef]
- Liang, Y.; Xian, X.; Nuccio, E.E.; Yuan, M.; Zhang, N.; Xue, K.; Cohan, F.M.; Zhou, J.; Sun., B. Differentiation strategies of soil rare and abundant microbial taxa in response to changing climatic regimes. Environ. Microbiol. 2020, 22, 1327–1340. [Google Scholar] [CrossRef] [PubMed]
- Pascoal, F.; Magalhães, C.; Costa, R. The link between the ecology of the prokaryotic rare biosphere and its biotechnological potential. Front. Microbiol. 2020, 11, 231. [Google Scholar] [CrossRef]


| Phylum | Class | Order | Family (F) | Genus | Ecology | References |
|---|---|---|---|---|---|---|
| Proteobacteria (24% of Bacteria) | α-Proteobacteria (28% of Proteobacteria), 23,934 reads | Sphingomonadales (33% of α-Proteobacteria) | Sphingomonadaceae (85% of Sphingomonadales) | Sphingomonas (70% of F) | The sphingomonads are widely distributed in nature, having been isolated from many different aqueous and terrestrial habitats, as well as from plant root systems, clinical specimens, and other sources. | [28] |
| Rhizobiales (37% α-Proteobacteria) | Bradyrhizobiaceae (31% of Rhizobiales) | unclassified | Bradyrhizobiaceae is a family presenting a broad taxonomic affiliation with organisms from different environments, such as soil, plant, or animal hosts. | |||
| Hyphomicrobiaceae | Pedomicrobium (11% of F) | Pedomicrobium is a ubiquitous bacterium dominant in biofilms of man-made aquatic environments such as water distribution systems. | [29] | |||
| Rhizobiaceae (40% of Rhizobiales) | unclassified | Rhizobiaceae is a family of the Rhizobiales order in the Alphaproteobacteria class that presents genera associated with soil and planta hosts. | [30] | |||
| β-Proteobacteria (21% of Proteobacteria) 17,931 reads | Burkholderiales (60% of β-Proteobacteria) | Oxalobacteraceae (31% of Burkholderiales) | Massilia (6% of F) | Massilia timonae gen. nov., sp. nov., isolated from blood of an immunocompromised patient with cerebellar lesions. | [31] | |
| Comamonadaceae (47% of Burkholderiales) | Polaromonas (9% of F) | Polaromonas vacuolata gen. nov., sp. nov., a psychrophilic, marine, gas vacuolate bacterium from Antarctica. Polaromonas is one of the most abundant genera found on glacier surfaces, yet its ecology remains poorly described. | [32,33] | |||
| Burkholderiaceae (13% of Burkholderiales) | Ralstonia (8% of F) | Most common pathogens for causing nosocomial infections. It colonises the xylem, causing bacterial wilt in a very wide range of potential host plants. | [34] | |||
| γ-Proteobacteria (40% of Proteobacteria), 34,298 reads | Pseudomonadales (39% of γ-Proteobacteria) | Moraxellaceae (79% of Pseudomonadales) | Acinetobacter (100% of F) | Acinetobacters are common, free-living saprophytes found in soil, water, sewage and foods. | [35] | |
| Pseudomonadaceae (24% of Pseudomonadales) | Pseudomonas (94% of F) | The members of the genus demonstrate a great deal of metabolic diversity and consequently are able to colonize a wide range of niches. | [36] | |||
| Oceanospirillales (17% of γ-Proteobacteria) | Halomonadaceae (100% of Oceanospirillales) | Halomonas (99% of F) | Halomonas elongata, a new genus and species of extremely salt-tolerant bacteria. | [37] | ||
| Xanthomonadales (11% of γ-Proteobacteria) | Xanthomonadaceae (92% of Xanthomonadales) | Lysobacter | The Lysobacter species live in soil, decaying organic matter, and fresh water. | [38] | ||
| Rhodanobacteraceae (2% of Xanthomonadales) | unclassified | |||||
| Enterobacterales (7% of γ-Proteobacteria) | Enterobacteriaceae (36% of Enterobacterales) | Escherichia/Shigella (94% of F) | A Common inhabitant of the gastrointestinal tract of humans and animals. | [39] | ||
| Yersiniaceae (34% of Enterobacterales) | Serratia (100% of F) | Found in water, soil, plants, and animals. Some members of this genus produce a characteristic red pigment, Prodigiosin. | [40] | |||
| Erwiniaceae (29% of Enterobacterales) | Pantoea (100% of F) | Pantoea species have been isolated from feculent material, in soil, water, plant (as epiphytes or endophytes), seeds, fruits (e.g., pineapple, mandarin oranges), and the human and animal gastrointestinal tracts, in dairy products, in blood and in urine. | [41] | |||
| Nevskiales (4% of γ-Proteobacteria) | Steroidobacteraceae (49% of Nevskiales) | unclassified | ||||
| Nevskiaceae (49% of Nevskiales) | Panacagrimonas (62% of F) | Soil bacteria. | [42] | |||
| Chromatiales (2% of γ-Proteobacteria) | Thioprofundaceae (12% of Chromatiales) | Thioprofundum (100% of F) | Thermophilic chemolithoautotrophs from the deep sea. | [43] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michail, G.; Karapetsi, L.; Madesis, P.; Reizopoulou, A.; Vagelas, I. Metataxonomic Analysis of Bacteria Entrapped in a Stalactite’s Core and Their Possible Environmental Origins. Microorganisms 2021, 9, 2411. https://doi.org/10.3390/microorganisms9122411
Michail G, Karapetsi L, Madesis P, Reizopoulou A, Vagelas I. Metataxonomic Analysis of Bacteria Entrapped in a Stalactite’s Core and Their Possible Environmental Origins. Microorganisms. 2021; 9(12):2411. https://doi.org/10.3390/microorganisms9122411
Chicago/Turabian StyleMichail, George, Lefkothea Karapetsi, Panagiotis Madesis, Angeliki Reizopoulou, and Ioannis Vagelas. 2021. "Metataxonomic Analysis of Bacteria Entrapped in a Stalactite’s Core and Their Possible Environmental Origins" Microorganisms 9, no. 12: 2411. https://doi.org/10.3390/microorganisms9122411
APA StyleMichail, G., Karapetsi, L., Madesis, P., Reizopoulou, A., & Vagelas, I. (2021). Metataxonomic Analysis of Bacteria Entrapped in a Stalactite’s Core and Their Possible Environmental Origins. Microorganisms, 9(12), 2411. https://doi.org/10.3390/microorganisms9122411









