Alterations of Gut Bacteria in Hirschsprung Disease and Hirschsprung-Associated Enterocolitis
Abstract
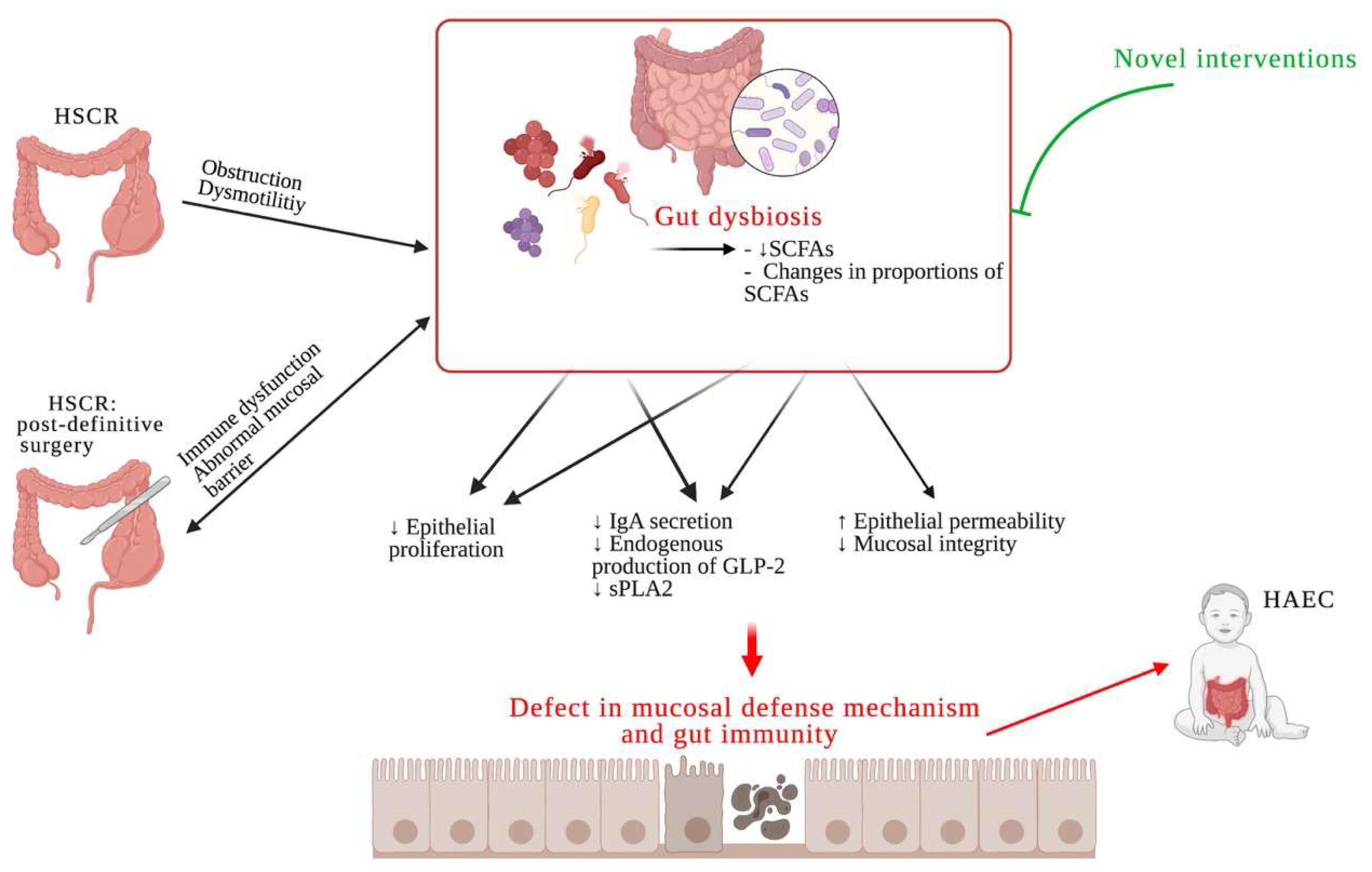
1. Introduction
2. The Association of Gut Microbiota with Human Gut Maturation, Immunity, and Mucosal Defense Mechanism
3. The Comparison of the Diversity of Gut Microbiota between Subjects with Hirschsprung Disease and Healthy Subjects: Evidence from Animal to Clinical Studies
4. The Alterations of Taxa in Gut Microbiota between Subjects with Hirschsprung Disease and Healthy Controls: Evidence from Animal to Clinical Studies
5. The Diversity of Gut Microbiota in Hirschsprung Disease with and without Enterocolitis: Evidence from Animal to Clinical Studies
6. The Alterations in Gut Microbiota Taxa in Hirschsprung Disease with and without Enterocolitis: Evidence from Animal to Clinical Studies
7. The Alterations in Gut Microbiota following Interventions of Enterocolitis in HSCR: Evidence from Animal to Clinical Studies
8. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
Appendix A
References
- Lager, J.C. Hirschsprung disease. In Holcomb and Ashcraft’s Pediatric Surgery, 7th ed.; Holcomb, G.W., Ed.; Elsevier: Philadelphia, PA, USA, 2020; Volume 7, pp. 557–558. [Google Scholar]
- Gosain, A.; Frykman, P.K.; Cowles, R.A.; Horton, J.; Levitt, M.; Rothstein, D.H.; Langer, J.C.; Goldstein, A.M. Guidelines for the diagnosis and management of Hirschsprung-associated enterocolitis. Pediatr. Surg. Int. 2017, 33, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Heuckeroth, R.O. Hirschsprung disease—Integrating basic science and clinical medicine to improve outcomes. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 152–167. [Google Scholar] [CrossRef] [PubMed]
- Gosain, A. Established and emerging concepts in Hirschsprung’s-associated enterocolitis. Pediatr. Surg. Int. 2016, 32, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Frykman, P.K.; Nordenskjold, A.; Kawaguchi, A.; Hui, T.T.; Granstrom, A.L.; Cheng, Z.; Tang, J.; Underhill, D.M.; Iliev, I.; Funari, V.A.; et al. Characterization of Bacterial and Fungal Microbiome in Children with Hirschsprung Disease with and without a History of Enterocolitis: A Multicenter Study. PLoS ONE 2015, 10, e0124172. [Google Scholar] [CrossRef]
- Cheng, Z.; Zhao, L.; Dhall, D.; Ruegger, P.M.; Borneman, J.; Frykman, P.K. Bacterial Microbiome Dynamics in Post Pull-Through Hirschsprung-Associated Enterocolitis (HAEC): An Experimental Study Employing the Endothelin Receptor B-Null Mouse Model. Front. Surg. 2018, 5, 30. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Su, Y.; Yuan, C.; Zhang, Y.; Zhou, L.; Peng, L.; Wang, P.; Chen, G.; Li, Y.; Li, H.; et al. Prospective study reveals a microbiome signature that predicts the occurrence of post-operative enterocolitis in Hirschsprung disease (HSCR) patients. Gut Microbes 2020, 11, 842–854. [Google Scholar] [CrossRef] [PubMed]
- Till, H.; Castellani, C.; Moissl-Eichinger, C.; Gorkiewicz, G.; Singer, G. Disruptions of the intestinal microbiome in necrotizing enterocolitis, short bowel syndrome, and Hirschsprung’s associated enterocolitis. Front. Microbiol. 2015, 6, 1154. [Google Scholar] [CrossRef]
- Pierre, J.F.; Barlow-Anacker, A.J.; Erickson, C.S.; Heneghan, A.F.; Leverson, G.E.; Dowd, S.E.; Epstein, M.L.; Kudsk, K.A.; Gosain, A. Intestinal dysbiosis and bacterial enteroinvasion in a murine model of Hirschsprung’s disease. J. Pediatr. Surg. 2014, 49, 1242–1251. [Google Scholar] [CrossRef]
- Ihekweazu, F.D.; Versalovic, J. Development of the Pediatric Gut Microbiome: Impact on Health and Disease. Am. J. Med. Sci. 2018, 356, 413–423. [Google Scholar] [CrossRef]
- Shi, N.; Li, N.; Duan, X.; Niu, H. Interaction between the gut microbiome and mucosal immune system. Mil. Med. Res. 2017, 4, 14. [Google Scholar] [CrossRef]
- McDermott, A.J.; Huffnagle, G.B. The microbiome and regulation of mucosal immunity. Immunology 2014, 142, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.Y.; Groer, M.; Dutra, S.V.O.; Sarkar, A.; McSkimming, D.I. Gut Microbiota and Immune System Interactions. Microorganisms 2020, 8, 1587. [Google Scholar] [CrossRef] [PubMed]
- Ward, N.L.; Pieretti, A.; Dowd, S.E.; Cox, S.B.; Goldstein, A.M. Intestinal aganglionosis is associated with early and sustained disruption of the colonic microbiome. Neurogastroenterol. Motil. 2012, 24, 874-e400. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, A.P.; Hascoet, J.; Berneau, P.; LeGouevec, F.; Georges, J.; Randuineau, G.; Formal, M.; Henno, S.; Boudry, G. A piglet model of iatrogenic rectosigmoid hypoganglionosis reveals the impact of the enteric nervous system on gut barrier function and microbiota postnatal development. J. Pediatr. Surg. 2021, 56, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Hegde, S.; Lin, Y.M.; Golovko, G.; Khanipov, K.; Cong, Y.; Savidge, T.; Fofanov, Y.; Shi, X.Z. Microbiota dysbiosis and its pathophysiological significance in bowel obstruction. Sci. Rep. 2018, 8, 13044. [Google Scholar] [CrossRef] [PubMed]
- Vandeputte, D.; Falony, G.; Vieira-Silva, S.; Tito, R.Y.; Joossens, M.; Raes, J. Stool consistency is strongly associated with gut microbiota richness and composition, enterotypes and bacterial growth rates. Gut 2016, 65, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Neuvonen, M.I.; Korpela, K.; Kyrklund, K.; Salonen, A.; de Vos, W.; Rintala, R.J.; Pakarinen, M.P. Intestinal Microbiota in Hirschsprung Disease. J. Pediatr. Gastroenterol. Nutr. 2018, 67, 594–600. [Google Scholar] [CrossRef]
- Wagner, N.R.F.; Zaparolli, M.R.; Cruz, M.R.R.; Schieferdecker, M.E.M.; Campos, A.C.L. Postoperative Changes in Intestinal Microbiota and Use of Probiotics in Roux-En-Y Gastric Bypass and Sleeve Vertical Gastrectomy: An Integrative Review. Arq. Bras. Cir. Dig. 2018, 31, e1400. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.G.; Bai, R.X.; Yan, W.M.; Yan, M.; Dong, L.Y.; Song, M.M. Differential composition of gut microbiota among healthy volunteers, morbidly obese patients and post-bariatric surgery patients. Exp. Ther. Med. 2019, 17, 2268–2278. [Google Scholar] [CrossRef] [PubMed]
- Steinert, R.E.; Rehman, A.; Souto Lima, E.J.; Agamennone, V.; Schuren, F.H.J.; Gero, D.; Schreiner, P.; Vonlanthen, R.; Ismaeil, A.; Tzafos, S.; et al. Roux-en-Y gastric bypass surgery changes fungal and bacterial microbiota in morbidly obese patients-A pilot study. PLoS ONE 2020, 15, e0236936. [Google Scholar] [CrossRef]
- Cong, J.; Zhu, H.; Liu, D.; Li, T.; Zhang, C.; Zhu, J.; Lv, H.; Liu, K.; Hao, C.; Tian, Z.; et al. A Pilot Study: Changes of Gut Microbiota in Post-surgery Colorectal Cancer Patients. Front. Microbiol. 2018, 9, 2777. [Google Scholar] [CrossRef]
- Pini Prato, A.; Bartow-McKenney, C.; Hudspeth, K.; Mosconi, M.; Rossi, V.; Avanzini, S.; Faticato, M.G.; Ceccherini, I.; Lantieri, F.; Mattioli, G.; et al. A Metagenomics Study on Hirschsprung’s Disease Associated Enterocolitis: Biodiversity and Gut Microbial Homeostasis Depend on Resection Length and Patient’s Clinical History. Front. Pediatr. 2019, 7, 326. [Google Scholar] [CrossRef]
- Toure, A.M.; Landry, M.; Souchkova, O.; Kembel, S.W.; Pilon, N. Gut microbiota-mediated Gene-Environment interaction in the TashT mouse model of Hirschsprung disease. Sci. Rep. 2019, 9, 492. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.H.; Shi, C.R.; Chen, J.J.; Yu, S.Y.; Wu, Y.; Yan, W.B. Detection of intestinal bifidobacteria and Lactobacilli in patients with Hirschsprung’s disease associated enterocolitis. World J. Pediatr. 2009, 5, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Shin, N.R.; Whon, T.W.; Bae, J.W. Proteobacteria: Microbial signature of dysbiosis in gut microbiota. Trends Biotechnol. 2015, 33, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Poroyko, V.; Yan, Z.; Pan, L.; Feng, Y.; Zhao, P.; Xie, Z.; Hong, L. Characterization of Intestinal Microbiomes of Hirschsprung’s Disease Patients with or without Enterocolitis Using Illumina-MiSeq High-Throughput Sequencing. PLoS ONE 2016, 11, e0162079. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Poroyko, V.; Gu, S.; Zhang, Z.; Pan, L.; Wang, J.; Bao, N.; Hong, L. Characterization of the intestinal microbiome of Hirschsprung’s disease with and without enterocolitis. Biochem. Biophys. Res. Commun. 2014, 445, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.Q.; Huang, Y.; Sun, M.; Ruan, Q.; Qi, Y.; He, R.; Huang, Y.J.; Ma, Y.P.; Ji, Y.H.; Sun, Z.R.; et al. Genetic polymorphism of UL144 open reading frame of human cytomegalovirus DNA detected in colon samples from infants with Hirschsprung’s disease. World J. Gastroenterol. 2007, 13, 4350–4354. [Google Scholar] [CrossRef] [PubMed]
- Tam, P.K.; Quint, W.G.; van Velzen, D. Hirschsprung’s disease: A viral etiology? Pediatr. Pathol. 1992, 12, 807–810. [Google Scholar] [CrossRef] [PubMed]
- Arbizu, R.A.; Collins, D.; Wilson, R.C.; Alekseyenko, A.V. Evidence for Differentiation of Colon Tissue Microbiota in Patients with and without Postoperative Hirschsprung’s Associated Enterocolitis: A Pilot Study. Pediatr. Gastroenterol. Hepatol. Nutr. 2021, 24, 30–37. [Google Scholar] [CrossRef]
- Singer, G.; Kashofer, K.; Castellani, C.; Till, H. Hirschsprung’s Associated Enterocolitis (HAEC) Personalized Treatment with Probiotics Based on Gene Sequencing Analysis of the Fecal Microbiome. Case Rep. Pediatr. 2018, 2018, 3292309. [Google Scholar] [CrossRef]
- Derrien, M.; Belzer, C.; de Vos, W.M. Akkermansia muciniphila and its role in regulating host functions. Microb. Pathog. 2017, 106, 171–181. [Google Scholar] [CrossRef]
- Macchione, I.G.; Lopetuso, L.R.; Ianiro, G.; Napoli, M.; Gibiino, G.; Rizzatti, G.; Petito, V.; Gasbarrini, A.; Scaldaferri, F. Akkermansia muciniphila: Key player in metabolic and gastrointestinal disorders. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 8075–8083. [Google Scholar] [CrossRef]
- Håkansson, Å.; Tormo-Badia, N.; Baridi, A.; Xu, J.; Molin, G.; Hagslätt, M.L.; Karlsson, C.; Jeppsson, B.; Cilio, C.M.; Ahrné, S. Immunological alteration and changes of gut microbiota after dextran sulfate sodium (DSS) administration in mice. Clin. Exp. Med. 2015, 15, 107–120. [Google Scholar] [CrossRef]
- Carvalho, F.A.; Koren, O.; Goodrich, J.K.; Johansson, M.E.; Nalbantoglu, I.; Aitken, J.D.; Su, Y.; Chassaing, B.; Walters, W.A.; González, A.; et al. Transient inability to manage Proteobacteria promotes chronic gut inflammation in TLR5-deficient mice. Cell Host Microbe 2012, 12, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Knights, D.; Silverberg, M.S.; Weersma, R.K.; Gevers, D.; Dijkstra, G.; Huang, H.; Tyler, A.D.; van Sommeren, S.; Imhann, F.; Stempak, J.M.; et al. Complex host genetics influence the microbiome in inflammatory bowel disease. Genome Med. 2014, 6, 107. [Google Scholar] [CrossRef] [PubMed]
- Morgan, X.C.; Tickle, T.L.; Sokol, H.; Gevers, D.; Devaney, K.L.; Ward, D.V.; Reyes, J.A.; Shah, S.A.; LeLeiko, N.; Snapper, S.B.; et al. Dysfunction of the intestinal microbiome in inflammatory bowel disease and treatment. Genome Biol. 2012, 13, R79. [Google Scholar] [CrossRef] [PubMed]
- Gong, D.; Gong, X.; Wang, L.; Yu, X.; Dong, Q. Involvement of Reduced Microbial Diversity in Inflammatory Bowel Disease. Gastroenterol. Res. Pract. 2016, 2016, 6951091. [Google Scholar] [CrossRef]
- Lavelle, A.; Lennon, G.; O’Sullivan, O.; Docherty, N.; Balfe, A.; Maguire, A.; Mulcahy, H.E.; Doherty, G.; O’Donoghue, D.; Hyland, J.; et al. Spatial variation of the colonic microbiota in patients with ulcerative colitis and control volunteers. Gut 2015, 64, 1553–1561. [Google Scholar] [CrossRef] [PubMed]
- Santoru, M.L.; Piras, C.; Murgia, A.; Palmas, V.; Camboni, T.; Liggi, S.; Ibba, I.; Lai, M.A.; Orrù, S.; Blois, S.; et al. Cross sectional evaluation of the gut-microbiome metabolome axis in an Italian cohort of IBD patients. Sci. Rep. 2017, 7, 9523. [Google Scholar] [CrossRef] [PubMed]
- Demehri, F.R.; Frykman, P.K.; Cheng, Z.; Ruan, C.; Wester, T.; Nordenskjold, A.; Kawaguchi, A.; Hui, T.T.; Granstrom, A.L.; Funari, V.; et al. Altered fecal short chain fatty acid composition in children with a history of Hirschsprung-associated enterocolitis. J. Pediatr. Surg. 2016, 51, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.; Linde, H.J.; Lehn, N.; Zimmermann, K.; Grossmann, J.; Falk, W.; Schölmerich, J. Immunomodulatory consequences of oral administration of Lactobacillus rhamnosus strain GG in healthy volunteers. J. Dairy Res. 2003, 70, 165–173. [Google Scholar] [CrossRef]
- Abu-Sbeih, H.; Wang, Y. Gut Microbiome and Immune Checkpoint Inhibitor-Induced Enterocolitis. Dig. Dis. Sci. 2020, 65, 797–799. [Google Scholar] [CrossRef] [PubMed]
- Rintala, R.J.; Lindahl, H. Sodium cromoglycate in the management of chronic or recurrent enterocolitis in patients with Hirschsprung’s disease. J. Pediatr. Surg. 2001, 36, 1032–1035. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Soh, H.J.; Nataraja, R.M.; Pacilli, M. Prevention and management of recurrent postoperative Hirschsprung’s disease obstructive symptoms and enterocolitis: Systematic review and meta-analysis. J. Pediatr. Surg. 2018, 53, 2423–2429. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Lim, T.; Puri, P. Probiotics for the prevention of Hirschsprung-associated enterocolitis: A systematic review and meta-analysis. Pediatr. Surg. Int. 2018, 34, 189–193. [Google Scholar] [CrossRef]
- El-Sawaf, M.; Siddiqui, S.; Mahmoud, M.; Drongowski, R.; Teitelbaum, D.H. Probiotic prophylaxis after pullthrough for Hirschsprung disease to reduce incidence of enterocolitis: A prospective, randomized, double-blind, placebo-controlled, multicenter trial. J. Pediatr. Surg. 2013, 48, 111–117. [Google Scholar] [CrossRef]
- Wang, X.; Li, Z.; Xu, Z.; Wang, Z.; Feng, J. Probiotics prevent Hirschsprung’s disease-associated enterocolitis: A prospective multicenter randomized controlled trial. Int. J. Colorectal Dis. 2015, 30, 105–110. [Google Scholar] [CrossRef]
- Cani, P.D.; Possemiers, S.; Van de Wiele, T.; Guiot, Y.; Everard, A.; Rottier, O.; Geurts, L.; Naslain, D.; Neyrinck, A.; Lambert, D.M.; et al. Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut 2009, 58, 1091–1103. [Google Scholar] [CrossRef]
| HSCR Model/Age (N) | Control/Age (N) | Specimens/Time of Collection | Methods | Diversity | Interpretation | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| Alpha | Type of Analysis | Beta | ||||||
| EdnrB-null mice/Early age [P16–18] (n = 6) Late age (P21–24) (n = 4) | EdnrB-het mice/Early age [P16–18] (n = 8) Late age [P21–24] (n = 11) | Cecal content/early and late ages | 16S rRNA sequencing | Early: ↑ Late: ↑ | Shannon | Significantly differed between the late control group and others. | Alpha diversity in Hirschsprung mice increased continuously over time, while the diversity was decreased in normal controls. | [9] |
| EdnrB null mice/P7, 20, 24 (n = 13) | WT/P7, 20, 24 (n = 13) | Fecal and colonic mucosa/P7,20,24 days | 16S rRNA sequencing | Colon ↑ P7 ↔ P20 and P24 ↑ with age Fecal: ↑ P7 ↑ P20 ↔ P24 | OTUs Shannon | Differences seen between both groups. Differences seen between all postnatal ages in both fecal and colonic specimens. | The alpha diversity in early age mutant mice was increased with age and greater than WT, while both fecal and colonic specimens had different beta diversity between both mutant-WT and all postnatal age groups. | [14] |
| 0.5% BAC serosal rectosigmoid soaked piglets/P26 (n = 7) | saline soaked piglets/P26 (n = 5) | Rectosigmoid content/P26 days | 16S rRNA sequencing (V3–4) | ↔ | Chao1 Shannon | Significant difference between BAC and control piglets. | There was no significant difference in the alpha diversity but a difference in the beta diversity between BAC and control piglets. | [15] |
| Ednrb-/- mice/PO 0, 14, and 28 days (n = 10) | WT mice/PO 0, 14, and 28 days (n = 8) | Stool/PO 0,14, and 28 days | 16S rRNA sequencing (V4–5) | ↓ | Shannon | Difference between mutant and WT. Difference between PO 0, 14 and 28 in both mutant and WT groups. | After surgery, the alpha diversity was significantly reduced in mutant mice, while the beta diversity differed between mutant-WT and in all postoperative periods. Surgery itself might alter microbiota. | [6] |
| HSCR patients/3–25 yrs (n = 34) | non-HSCR/2–7 yrs (n = 141) | Stool/post definitive surgery. | 16S rDNA sequencing (V3–4) | ↓ richness | N/A | The richness of gut microbiota in HSCR patients was significantly reduced after surgery. | [18] | |
| HSCR Model/Age (N) | Control/Age (N) | Specimens/Time of Collection | Methods | Taxonomy | Interpretation | Ref. | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phylum | Genus, Family, Species | |||||||||
| Proteobacteria | Firmicutes | Bacteroides | Others | |||||||
| EdnrB-null mice/Early age [P16–18] (n = 6) Late age [P21–24] (n = 4) | EdnrB-het mice/Early age [P16–18] (n = 8) Late age [P21–24] (n = 11) | Cecal content/early and late ages | 16S rRNA sequencing | Early: N/A Late: ↓Actinobacteria ↓TM7 | Late: ↑Escherichia | Early: ↔Lactobacillus Late: ↑Clostridium ↓Lactobacillus (↓overtime) | Late: ↑Bacteroides | The mutant mice showed a decrease in Lactobacillus over time with an increase in a possible pathogen (Escherichia coli), leading to EC. | [9] | |
| EdnrB-null mice/Early age [P7] Late age [P20,24] (n = 13) | WT mice/Early age [P7] Late age [P20,24] (n = 13) | Fecal and colonic mucosal microbiota/Early age [P7] Late age [P20,24] | 16S rRNA sequencing | Colon (Early): N/A Colon (Late): ↑Proteobacteria ↑↑Bacteroidetes ↓↓Firmicutes Fecal (Late): ↑Proteobacteria ↑↑Bacteroidetes ↓↓Firmicute | Colon (Early): ↑Staphylococcus (S. xylosus) ↑Lactobacillus ↑Coprobacillus ↓Clostridium Colon (Late): ↑Clostridium ↑↑Coprobacillus Absent Staphylococcus Absent Lactobacillus Fecal (Late): ↓Lactobacillus ↓Staphylococcus (S. Xylosus) | Colon (Early): ↑Bacteroides Colon (Late): ↑↑Bacteroides Fecal (Late): ↑Tannerella | The mutant fecal and colonic specimens increased in Bacteroidetes and Proteobacteria but decreased in Firmicutes esp. Lactobacillus and Staphylococcus over time and changed with age. | [14] | ||
| 0.5% BAC serosal rectosigmoid soaked piglets/P5 (n = 7) | saline soaked piglets/P5 (n = 5) | Rectosigmoid content/P26 days | 16S rRNA sequencing (V3–4) | Tendency ↑Proteobacteria | ↑Bilophilia | ↑Mogibacterium | ↑Fusobacterium (Fusobacteria) | BAC piglet stools showed a tendency to increase in Proteobacteria at the phylum level and increase in Fusobacterium, Mogibacterium, Bilophilia (proinflammatory bacteria). | [15] | |
| TashT Tg/Tg, mice/P21–22 (n = 9) and Holstein Tg/Tg mice/P21–22 (n = 3) | WT mice/P21–22 (n = 6) | fecal sample from the colon/weaning age | 16S rRNA sequencing (V5–6) | ↑↑Proteobacteria ↑Deferribacteres ↓↓Firmicutes | The mutant mice feces significantly increased in Proteobacteria and decreased in Firmicutes at the phylum level. | [24] | ||||
| HSCR patients/3–25 yrs. (n = 34) | Non-HSCR patients/3–25 yrs. (n = 141) | Stool/post definite surgery | 16S rDNA sequencing (V3–4) | ↑Proteobacteria | ↑Escherichia ↑Pseudomonas | ↑Dialister ↑Bacilli ↓Ruminococcaceae ↓Lachnospiraceae ↓Lactobacilli | ↑Prevotella ↓Bacteroidales | ↑Actinomyces (Actinobacteria) | After surgery, HSCR stools showed an expansion of Proteobacteria at the phylum level and Enterobacteria and Bacilli at order level. While Lactobacilli decreased significantly. | [18] |
| HSCR patients/2 wks–2 yrs. (n = 20) | Non-HSCR patients/2 wks–2 yrs. (n = 15) | Stool | 16S rRNA real-time PCR | ↓Lactobacilli | Lactobacilli significantly decreased in HSCR specimens. | [25] | ||||
| HSCR with EC Model/Age (N) | HSCR without EC Model/Age (N) | Specimens/Time of Collection | Methods | Diversity | Other Findings | Interpretation | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|
| Alpha | Type of Analysis | Beta | |||||||
| HAEC patients/2, 6 mo. (n = 2) | HSCR patients/7, 12 mo. (n = 2) | Intestinal content from different sections/during surgery | 16S rDNA sequencing (V1–3) | ↑ | OTUs | Difference | HSCR had greater diversity in distal than proximal samples. HAEC had greater diversity in proximal than distal samples. | HAEC samples increased the alpha diversity, while both HSCR-HAEC had a difference of microbiome between proximal (ganglionic) and distal (aganglionic) parts of the intestine. | [28] |
| HAEC/HAEC-R patients/10 d.–2 yrs. (n = 5/3) | HSCR patients/10 d.–2 yrs. (n = 5) | Intestinal contents from difference sites/during surgery | 16S rRNA sequencing (V4) | N/A | Difference (HSCR-HAEC) Similarity (HAEC-HAEC-R) | HSCR showed distinct microbiomes between the proximal-distal intestine. Both HAEC and HAEC-R specimens showed no different microbiota in sites. | HAEC specimens found a loss of a site-specific microbiome of HSCR and HAEC-R had persistent disturbance similar to HAEC even when symptoms of EC were resolved. | [27] | |
| HAEC patients/5 mo.–8 yrs. (n = 9) | HSCR patients/5 mo.–8 yrs. (n = 9) | Stools/after definitive surgery | 16S rRNA sequencing (V1–4) | ↑ ↓ (mycobiome) | Shannon OTUs | N/A | The stools of HSCR patients had an increased alpha diversity in the microbiome but a decreased alpha diversity in the mycobiome. | [5] | |
| HAEC patients/3–25 yrs. (n = 26) | HSCR patients/3–25 yrs. (n = 8) | Stools/post definitive surgery | 16S rDNA sequencing (V3–4) | ↓ richness | N/A | The loss of richness in the microbiota led to an increase in vulnerability to colonizing pathogens in HAEC. | [18] | ||
| HAEC episodes/3 yrs. (n = 3) | Non-HAEC episodes/3 yrs. (n = 3) | Self-comparisons of stools | 16S rRNA sequencing | ↔ | Chao1 | Difference | There was a difference in Beta diversity between HAEC and non-HAEC periods. | [32] | |
| post-op HAEC patients/mostly <3 mo. (n = 25) | HSCR patients/mostly <3 mo. (n = 50) | Mucosa at edge of dilated segment close to normal/at time of surgery | 16S rRNA sequencing (V4) | ↓ | OTUs Chao1 Shannon Simpson PD | Difference | In postoperative HAEC patients, a decrease in the alpha diversity of microbiota in a mucosal specimen at the time of surgery was found. | [7] | |
| post-op HAEC patients/mostly <1 mo. (n = 4) | HSCR patients/mostly <1 mo. (n = 4) | Aganglionic colonic tissue formalin and paraffin fixation/at time of surgery | 16S rDNA sequencing (V3,4) | ↑ | observed alpha diversity | No significant difference | In postoperative HAEC patients or HAEC potential patients, an increase in the alpha diversity of microbiota in aganglionic colon specimens at the time of surgery was found without significant difference in the beta diversity between HSCR and potential HAEC groups. | [31] | |
| Ednrb-/- mice with HAEC/3–6 wks. (n = 6) | WT mice/3–6 wks. (n = 4) | Stool/PO d0,14,28 | 16S rRNA sequencing (V4–5) | ↓ | OTUs Chao1 Shannon Simpson PD | N/A | In a potential HAEC model, a decrease in the alpha diversity of microbiota in stools at the time of surgery was found. | [6] | |
| HSCR with EC Model/Age (N) | HSCR without EC Model/Age (N) | Specimens/Time of Collection | Methods | Taxonomy | Interpretation | Ref. | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phylum | Genus, Family, Species | |||||||||
| Proteobacteria | Firmicutes | Bacteroides | Others | |||||||
| HAEC patients/2, 6 mo. (n = 2) | HSCR patients/7, 12 mo. (n = 2) | Intestinal content from different sections/during surgery | 16S rDNA sequencing (V1–3) | ↑Proteobacteria ↓Firmicutes | ↑Enterobacteriaceae ↓Acinetobacter | ↑Enterococcus | ↑Bacteroides | ↓Fusobacterium (Fusobacteria) ↓Eukaryota | HAEC specimens had an increase in Proteobacteria but a decrease in Firmicutes. | [28] |
| HAEC/HAEC-R patients/10 d.–2 yrs. (n = 5/3) | HSCR patients/10 d.–2 yrs. (n = 5) | Intestinal contents from different sites/during Surgery | 16S rRNA sequencing (V4) | ↑↑Proteobacteria ↓↓ Bacteroidetes ↓Firmicutes | ↑↑Escherichia ↓Acinetobacter | ↓Veillonella | ↓Bacteroidete | An increase in Proteobacteria and a deficiency in Bacteroides-Firmicutes might be associated with HAEC. | [27] | |
| HAEC patients/5 mo.–8 yrs. (n = 9) | HSCR patients/5 mo.–8 yrs. (n = 9) | Stool/after complete definitive surgery | 16S rRNA sequencing (V1–4) | ↑Proteobacteria ↑Bacteroidetes ↓Firmicutes ↓Verrucomicrobia | No significant difference | ↑C. albican (mycobiome) ↓C. tropicalis (mycobiome) ↓Malassezia (mycobiome) ↓Saccharomyces sp. (mycobiome) | An increase in Proteobacteria, a decrease in Firmicutes, and an increase in the pathologic mycobiome were detected in the HAEC stools. | [5] | ||
| History of HAEC patients/3 mo–8 yrs. (n = 9) | HSCR patients/3 mo–8 yrs. (n = 9) | Stool/after definitive surgery | 16S rRNA sequencing (V1–4) | Dominated by non-SCFA-producing bacteria | The microbiota changed to be dominated by non-SCFA-producing bacteria in HAEC. | [42] | ||||
| HAEC patients/3–25 yrs. (n = 26) | HSCR patients/3–25 yrs. (n = 8) | Stool/post definitive surgery | 16S rDNA sequencing (V3–4) | ↑↑Escherichia ↑↑Shigella ↑Proteobacteria | ↑Lactococcus ↑Lactobacillus ↓↓Clostridia ↓Oscillospira ↓Holdemania | ↓↓Prevotella | HAEC increased vulnerability to colonization by pathogens. | [18] | ||
| HAEC patients/2 wks–2 yrs. (n = 10) | Non-HAEC patients/2 wks–2 yrs. (n = 20) | Stool | 16S rRNA real time PCR | ↓Bifidobacterium (Actinobacteria) | The scarcity of Bifidobacterium might be a predisposing cause of EC. | [25] | ||||
| HAEC episodes/3 yrs. (n = 3) | Non-HAEC episodes/3 yrs. (n = 3) | Self-comparison of stool | 16S rRNA sequencing | ↑Proteobacteria ↑Bacteroidetes ↑Cyanobacteria ↓Actinobacteria | ↓Bifidobacterium (Actinobacteria) | HAEC stools showed increased Proteobacteria, Bacteroidetes, Cyanobacteria, and significantly decreased Actinobacteria esp. Bifidobacterium. | [32] | |||
| post-op HAEC patients/mostly <3 mo. (n = 25) | HSCR patients/mostly <3 mo. (n = 50) | Mucosa at dilated segment close to normal/at time of surgery | 16S rRNA sequencing (V4) | ↑Enterobacteriaceae | ↑Koribactereceae (Acidobacteria) | An increase in Enterobacteriaceae was detected in HAEC potential patients. | [7] | |||
| post-op HAEC patients/mostly <1 mo. (n = 4) | HSCR patients/mostly <1 mo. (n = 4) | Aganglionic colonic tissue formalin and paraffin fixation/at time of surgery | 16S rDNA sequencing (V3,4) | ↑↑Firmicutes ↑Bacteroidetes ↑Cyanobacteria | ↑Escherichia | ↑Dolosigranulum ↑Streptococcus ↑Roseburia ↑Enterococcus | ↑Propionibacterium (Actinobacteria) | An increase in bacteria in phylum Firmicutes, especially Dolosigranulum, Streptococcus, and Roseburia was detected in postoperative HAEC patients. | [31] | |
| Ednrb-/- mice with HAEC/3–6 wks. (n = 6) | Ednrb-/- mice without HAEC/3–6 wks. (n = 4) | Stool/PO d0,14,28 | 16S rRNA sequencing (V4–5) | ↑↑Verrucomicrobia ↓Bacteroidetes ↔Firmicutes | ↓Clostridium XIVa | ↓Bacteroidetes ↓Dysgonomas | ↑↑Akkermansia (Verrucomicrobia) | Markedly increased Akkermansia and decreased Bacteroidetes might be associated with EC. | [6] | |
| Model/Age (N) | Control/Age (N) | Specimens/Time of Collection | Intervention | Methods | Diversity | Taxonomy | Interpretation | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Alpha | Beta | Phylum | Genus, Family, Species | |||||||
| TashT Tg/Tg mice a/P21–22 (n = 3) WT/P21–22 (n = 3) + ABO | TashT Tg/Tg mice a/P21–22 (n = 3) WT/P21–22 (n = 3) − ABO | fecal sample in colon /P21–22 | Oral broad spectrum ABO b prenatal 10 day—P36 | 16S rRNA sequencing (V5–6) | ↓ | N/A | ↑↑Tenericutes ↓Firmicutes ↓Bacteroidetes | ↑↑↑Mycoplasma (Tenericutes) ↑Enterobacter ales (Proteobacteria) ↓↓Clostridiales (Firmicutes) ↓↓Bacteroidetes (Bacteroidetes) ↓↓Campylobacterales (Proteobacteria) ↓↓Burkholderiales (Proteobacteria) | Antibiotics decreased fecal microbiota similarly in both mutant and WT, causing dysbiosis. | [24] |
| HAEC episode/3 yrs. (n = 6) Non-HAEC episode/3 yrs. (n = 8) + Probiotics | HAEC episode/3 yrs. (n = 6) Non-HAEC episode/3 yrs. (n = 8) − Probiotics | self-comparisons of stool | Probiotic c period 3 mo. | 16S rRNA sequencing | ↑ | Significant difference | ↑Bacteroidetes | ↑Bifidobacterium (Actinobacteria) ↑Streptococcus (Firmicutes) ↓Rikenellaceae (Bacteroides) ↓Pseudobutyrivibrio (Firmicutes) ↓Blautia (Firmicutes) ↓Lachnospiraceae (Firmicutes) | Probiotic significantly improved gut-dysbiosis in HAEC patients. | [32] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chantakhow, S.; Khorana, J.; Tepmalai, K.; Boonchooduang, N.; Chattipakorn, N.; Chattipakorn, S.C. Alterations of Gut Bacteria in Hirschsprung Disease and Hirschsprung-Associated Enterocolitis. Microorganisms 2021, 9, 2241. https://doi.org/10.3390/microorganisms9112241
Chantakhow S, Khorana J, Tepmalai K, Boonchooduang N, Chattipakorn N, Chattipakorn SC. Alterations of Gut Bacteria in Hirschsprung Disease and Hirschsprung-Associated Enterocolitis. Microorganisms. 2021; 9(11):2241. https://doi.org/10.3390/microorganisms9112241
Chicago/Turabian StyleChantakhow, Sireekarn, Jiraporn Khorana, Kanokkan Tepmalai, Nonglak Boonchooduang, Nipon Chattipakorn, and Siriporn C. Chattipakorn. 2021. "Alterations of Gut Bacteria in Hirschsprung Disease and Hirschsprung-Associated Enterocolitis" Microorganisms 9, no. 11: 2241. https://doi.org/10.3390/microorganisms9112241
APA StyleChantakhow, S., Khorana, J., Tepmalai, K., Boonchooduang, N., Chattipakorn, N., & Chattipakorn, S. C. (2021). Alterations of Gut Bacteria in Hirschsprung Disease and Hirschsprung-Associated Enterocolitis. Microorganisms, 9(11), 2241. https://doi.org/10.3390/microorganisms9112241






