Field Based Assessment of Capsicum annuum Performance with Inoculation of Rhizobacterial Consortia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Procurement and Mass Culturing of Microbial Strains
2.2. Characterization of PGPR Isolates
2.3. Field Experiments to Evaluate Performance of PGPR’s Consortia
2.4. Statistical Analysis
3. Results and Discussion
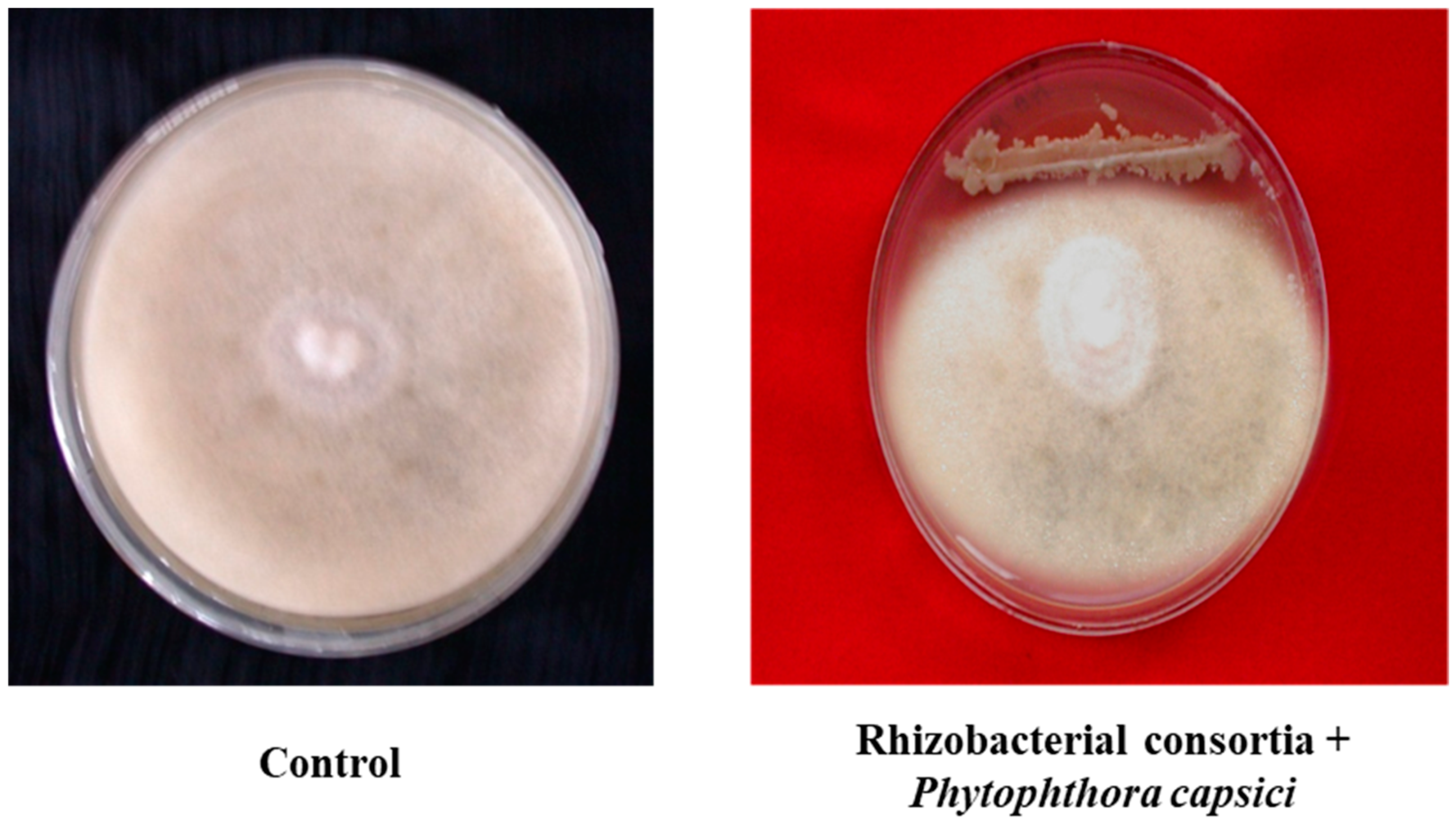
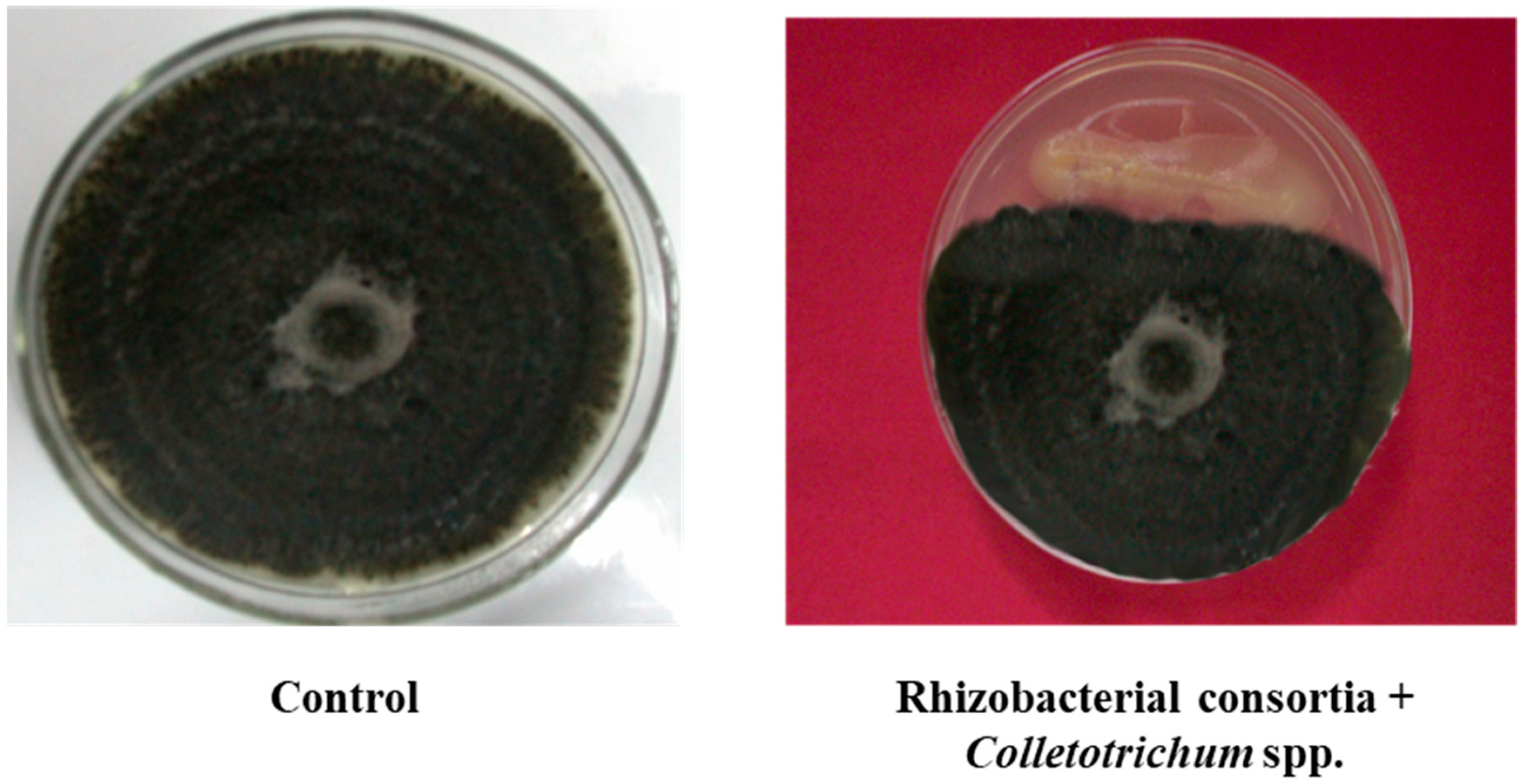
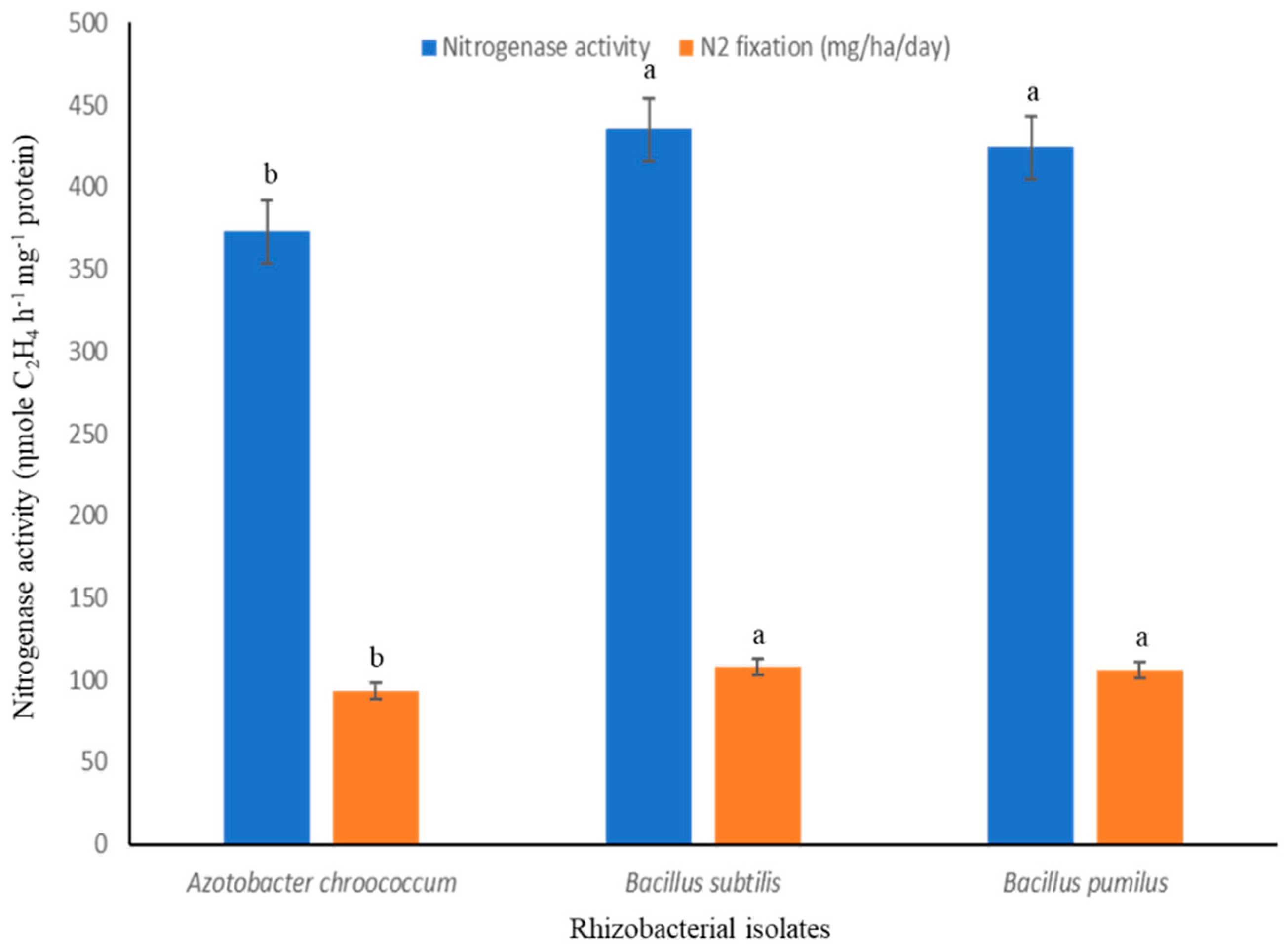
3.1. Characterization of PGPR Isolates
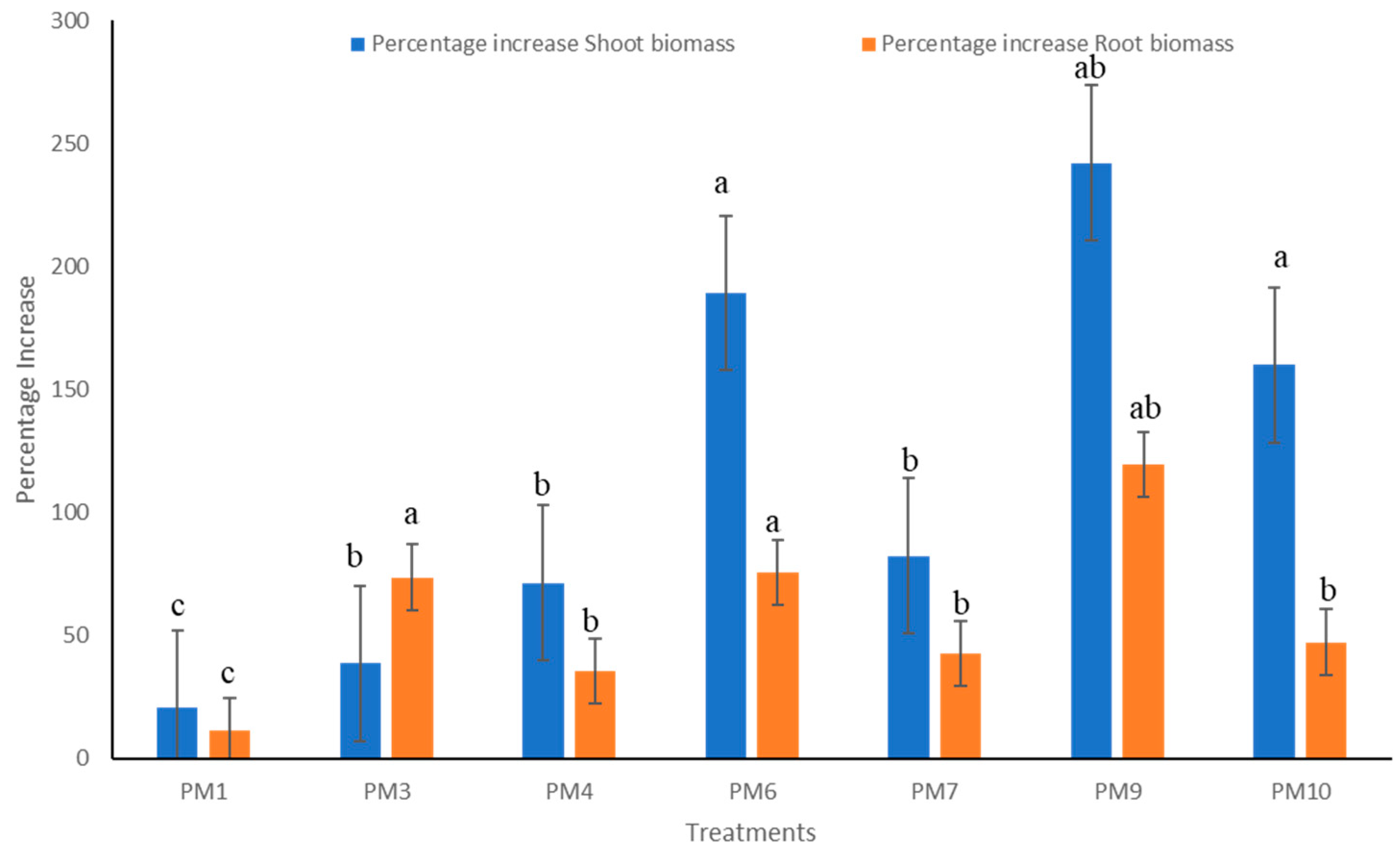
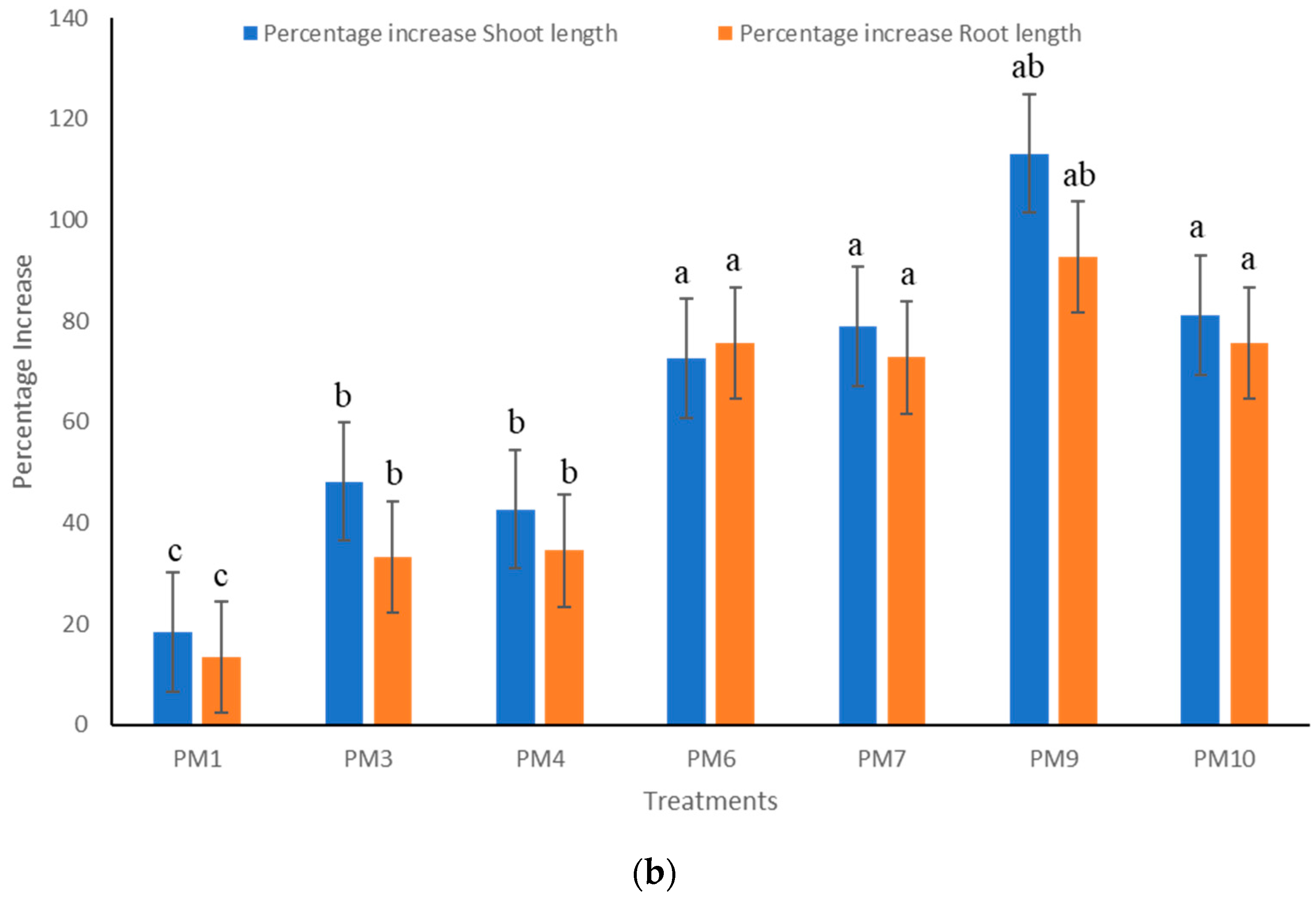
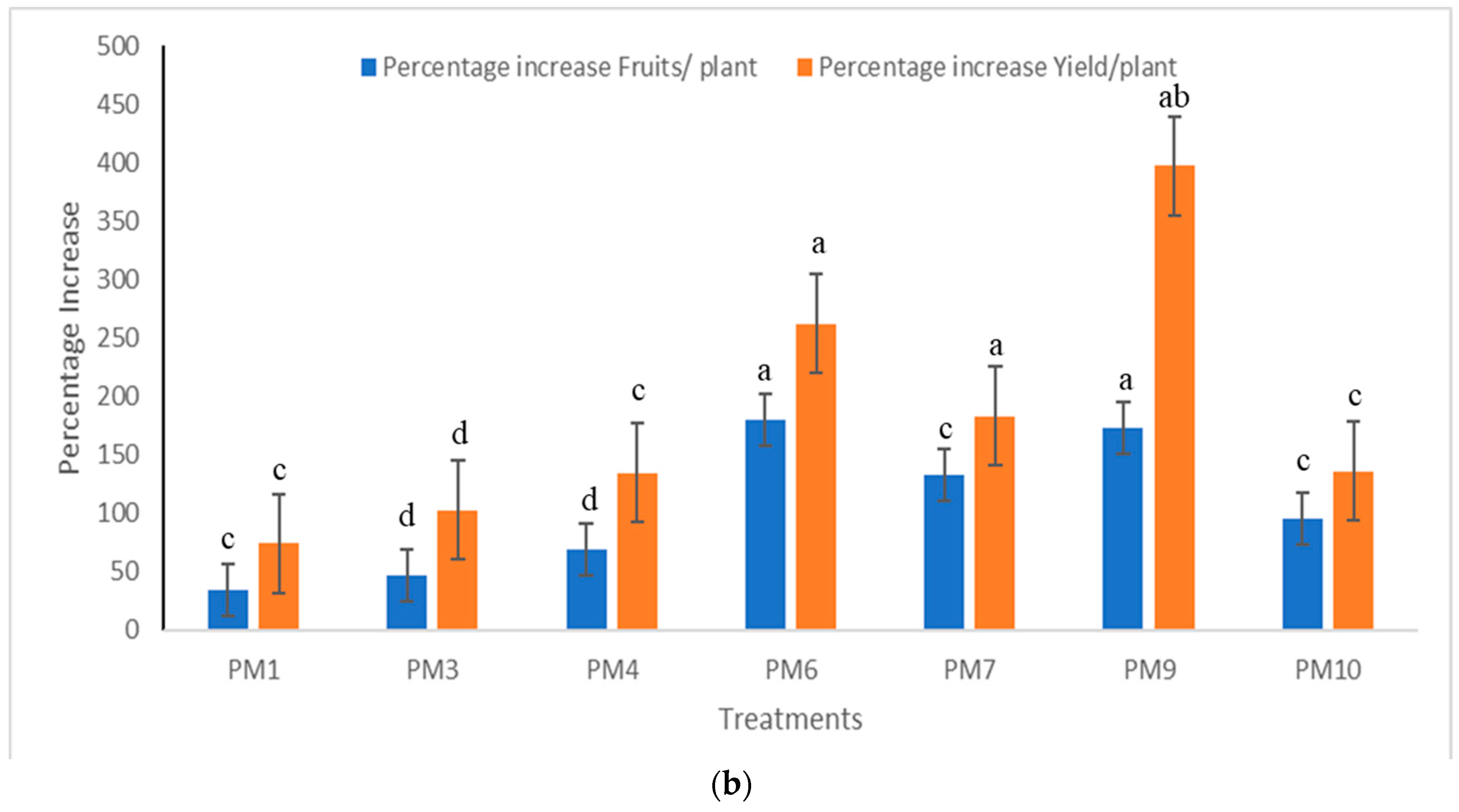
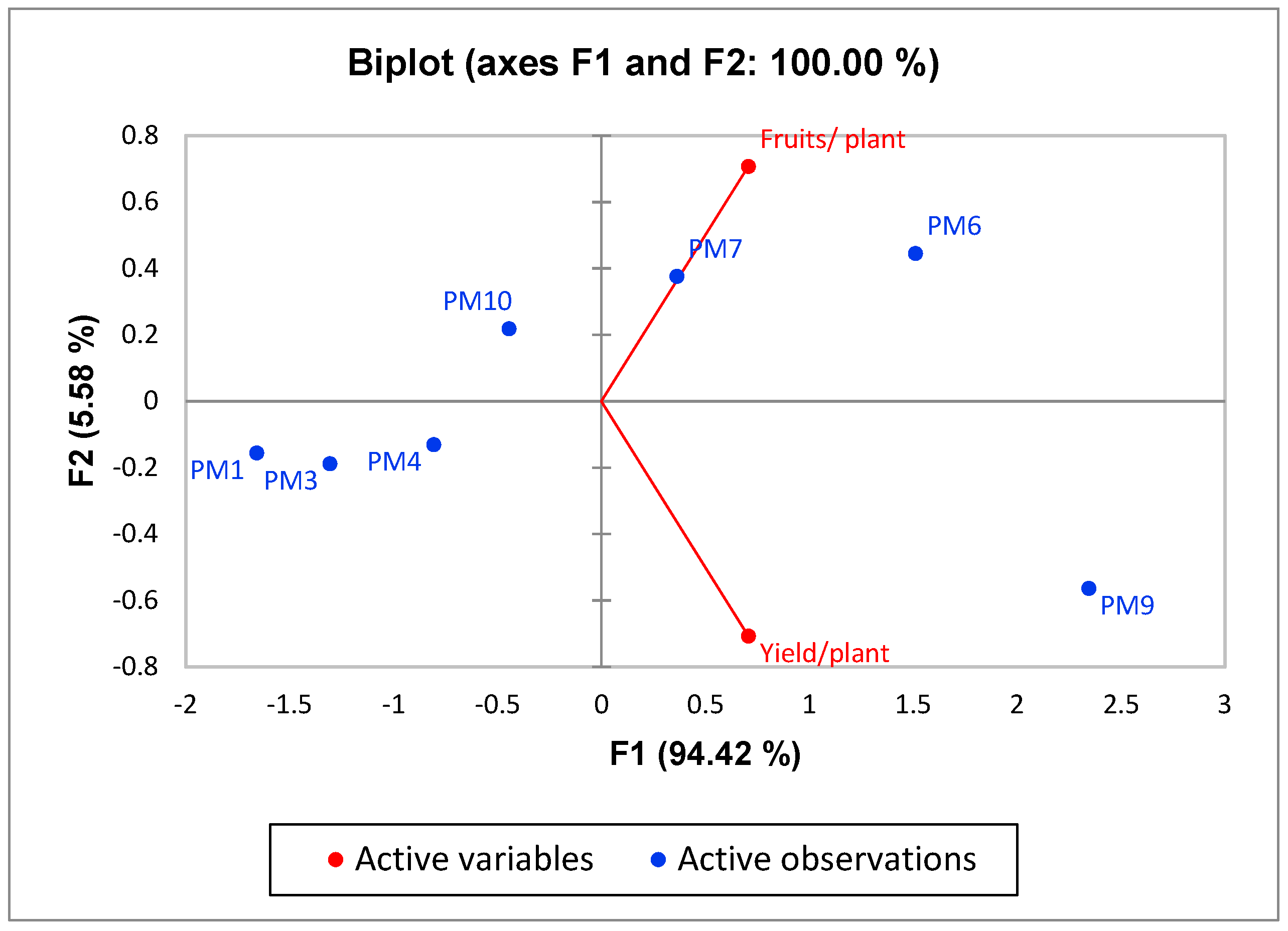
3.2. Field Experiments to Evaluate Performance of PGPR’s Consortia
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Akram, N.A.; Ashraf, M. Regulation in plant stress tolerance by a potential plant growth regulator, 5-aminolevulinic acid (ALA). J. Plant Growth Regul. 2013, 32, 663–679. [Google Scholar] [CrossRef]
- Sahitya, U.L.; Deepthi, R.S.; Kasim, D.P.; Suneetha, P.; Krishna, M.S.R. Anthracnose, a prevalent disease in Capsicum. Res. J. Pharm. Biol. Chem. Sci. 2014, 5, 1583–1604. [Google Scholar]
- Than, P.P.; Prihastuti, H.; Phoulivong, S.; Taylor, P.W.J.; Hyde, K.D. Chilli anthracnose disease caused by Colletotrichum species. J. Zhejiang Univ. Sci. B 2008, 9, 764–778. [Google Scholar] [CrossRef] [PubMed]
- Tariq, M.I.; Afzal, S.; Hussain, I.; Sultana, N. Pesticide Exposure in Pakistan: A Review. Environ Int. 2007, 33, 1107–1122. [Google Scholar] [CrossRef] [PubMed]
- Hassen, W.; Neifar, M.; Cherif, H.; Najjari, A.; Chouchane, H.; Driouich, R.C.; Salah, A.; Naili, F.; Mosbah, A.; Souissi, Y.; et al. Pseudomonas rhizophila S211, a New Plant Growth-Promoting Rhizobacterium with Potential in Pesticide-Bioremediation. Front. Microbiol. 2018, 9, 34. [Google Scholar] [CrossRef] [PubMed]
- Sang, M.K.; Kim, K.D. Plant growth-promoting rhizobacteria suppressive to Phytophthora blight affect microbial activities and communities in the rhizosphere of pepper (Capsicum annuum L.) in the field. Appl. Soil Ecol. 2012, 62, 88–97. [Google Scholar] [CrossRef]
- Etesami, H.; Maheshwari, D.K. Use of plant growth promoting rhizobacteria (PGPRs) with multiple plant growth promoting traits in stress agriculture: Action mechanisms and future prospects. Ecotoxicol. Environ. Saf. 2018, 156, 225–246. [Google Scholar] [CrossRef]
- Kaushal, M.; Wani, S.P. Rhizobacterial-plant interactions: Strategies ensuring plant growth promotion under drought and salinity stress. Agric. Ecosyst. Environ. 2016, 231, 68–78. [Google Scholar] [CrossRef]
- Kaushal, M.; Kumar, A.; Kaushal, R. Bacillus pumilus strain YSPMK11 as plant growth promoter and biocontrol agent against Sclerotinia sclerotiorum. 3 Biotech 2017, 7, 90. [Google Scholar] [CrossRef]
- Son, J.S.; Sumayo, M.; Hwang, Y.J.; Kim, B.S.; Ghim, S.Y. Screening of plant growth promoting rhizobacteria as elicitor of systemic resistance against gray leaf spot disease in pepper. Appl. Soil Ecol. 2014, 73, 1–8. [Google Scholar] [CrossRef]
- Dinesh, R.; Anandaraj, M.; Kumar, A.; Subila, K.P.; Bini, Y.K.; Aravind, A. Native multi-trait rhizobacteria promote growth and suppress Phytophthora capsici in black pepper. J. Spices Aromat. Crops 2014, 23, 156–163. [Google Scholar]
- Kaushal, M.; Kaushal, R. Screening and characterization of rhizobacterial strains of Bacillus spp. isolated from rhizosphere of cauliflower (Brassica oleracea var. botrytis L.). Afr. J. Microbiol. Res. 2013, 7, 1657–1663. [Google Scholar]
- Hardy, R.W.F.; Holsten, R.D.; Jackson, E.K.; Burns, R.C. The acetylene-ethylene assay for Nz fixation: Laboratory and field evaluation. Plant Physiol. 1986, 43, 1185–1207. [Google Scholar] [CrossRef]
- Cappellari, L.; Santoro, M.V.; Reinoso, H.; Travaglia, C.; Giordano, W.; Banchio, E. Anatomical, morphological, and phytochemical effects of inoculation with plant growth- promoting rhizobacteria on peppermint (Mentha piperita). J. Chem. Ecol. 2015, 41, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Raut, V.; Shaikh, I.; Naphade, B.; Prashar, K.; Adhapure, N. Plant growth promotion using microbial IAA producers in conjunction with azolla: A novel approach. Chem. Biol. Technol. Agric. 2017, 4, 1. [Google Scholar] [CrossRef]
- Beneduzi, A.; Ambrosini, A.; Passaglia, L. Plant growth-promoting rhizobacteria (PGPR): Their potential as antagonists and biocontrol agents. Gen. Mol. Biol. 2012, 35, 1044–1051. [Google Scholar] [CrossRef]
- Gupta, G.; Parihar, S.S.; Ahirwar, N.K.; Snehi, S.K.; Singh, V. Plant Growth Promoting Rhizobacteria (PGPR): Current and Future Prospects for Development of Sustainable Agriculture. J. Microb. Biochem. Technol. 2015, 7, 96–102. [Google Scholar]
- Etesami, H.; Beattie, G.A. Mining Halophytes for Plant Growth-Promoting Halotolerant Bacteria to Enhance the Salinity Tolerance of Non-halophytic Crops. Front. Microbiol. 2018, 9, 148. [Google Scholar] [CrossRef] [PubMed]
- Zahid, M.; Abbasi, M.K.; Hameed, S.; Rahim, N. Isolation and identification of indigenous plant growth promoting rhizobacteria from Himalayan region of Kashmir and their effect on improving growth and nutrient contents of maize (Zea mays L.). Front. Microbiol. 2015, 6, 207. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Mukherjee, S.; Tarafdar, J.; Guharoy, S. Biocontrol and plant growth promoting activity of bacterial strain Pseudomonas aeruginosa KUCd1in root rot disease of Chilli (Capsicum sp.) caused by Phytophthora capsici Leonian under in vivo conditions. J. Mycopathol. Res. 2016, 54, 93–100. [Google Scholar]
- Kaushal, M.; Wani, S.P. Plant-growth-promoting rhizobacteria: Drought stress alleviators to ameliorate crop production in drylands. Ann. Microbiol. 2015, 66, 35–42. [Google Scholar] [CrossRef]
- Palazzini, J.; Roncallo, P.; Cantoro, R.; Chiotta, M.; Yerkovich, N.; Palacios, S.; Echenique, V.; Torres, A.; Ramirez, M.; Karlovsky, P.; et al. Biocontrol of Fusarium graminearum sensu stricto, Reduction of Deoxynivalenol Accumulation and Phytohormone Induction by Two Selected Antagonists. Toxins 2018, 10, 88. [Google Scholar] [CrossRef]
- Hariprasad, P.; Chandrashekar, S.; Singh, S.B.; Niranjana, S.R. Mechanisms of plant growth promotion and disease suppression by Pseudomonas aeruginosa strain 2apa. J. Basic Microbiol. 2014, 54, 792–801. [Google Scholar] [CrossRef] [PubMed]
- Mendis, H.C.; Thomas, V.P.; Schwientek, P.; Salamzade, R.; Chien, J.T.; Waidyarathne, P.; Kloepper, J.; Fuente, L.D.L. Strain-specific quantification of root colonization by plant growth promoting rhizobacteria Bacillus firmus I-1582 and Bacillus amyloliquefaciens QST713 in non-sterile soil and field conditions. PLoS ONE 2018, 13, e0193119. [Google Scholar] [CrossRef] [PubMed]
- Prasannakumar, S.P.; Gowtham, H.G.; Hariprasad, P.; Shivaprasad, K.; Niranjana, S.R. Delftia tsuruhatensis WGR–UOM–BT1, a novel rhizobacterium with PGPR properties from Rauwolfia serpentina (L.) Benth. ex Kurz also suppresses fungal phytopathogens by producing a new antibiotic—AMTM. Lett. Appl. Microbiol. 2015, 61, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Fan, X.; Cai, X.; Hu, F. The inhibitory mechanisms by mixtures of two endophytic bacterial strains isolated from Ginkgo biloba against pepper Phytophthora blight. Biol. Control 2015, 85, 59–67. [Google Scholar] [CrossRef]
- Khabbaz, S.E.; Zhahag, L.; Caceres, L.A.; Sumarah, M.; Wang, A.; Abbasi, P.F. Characterization of antagonistic Bacillus and Pseudomonas strains for biocontrol potential and suppression of damping-off and root rot diseases. Ann. Appl. Biol. 2015, 166, 456–471. [Google Scholar] [CrossRef]
- Santiago, T.R.; Grabowski, C.; Rossato, M.; Romeiro, R.S.; Mizubuti, E.S.G. Biological control of eucalyptus bacterial wilt with rhizobacteria. Biol. Control 2015, 80, 14–22. [Google Scholar] [CrossRef]
- Hassan, M.N.; Namood-e-Sahar; Shah, S.Z.; Afgan, S.; Hafeez, F.Y. Suppression of red rot disease by Bacillus sp. based biopesticide formulated in non-sterilized sugarcane filter cake. BioControl 2015, 60, 691–702. [Google Scholar] [CrossRef]


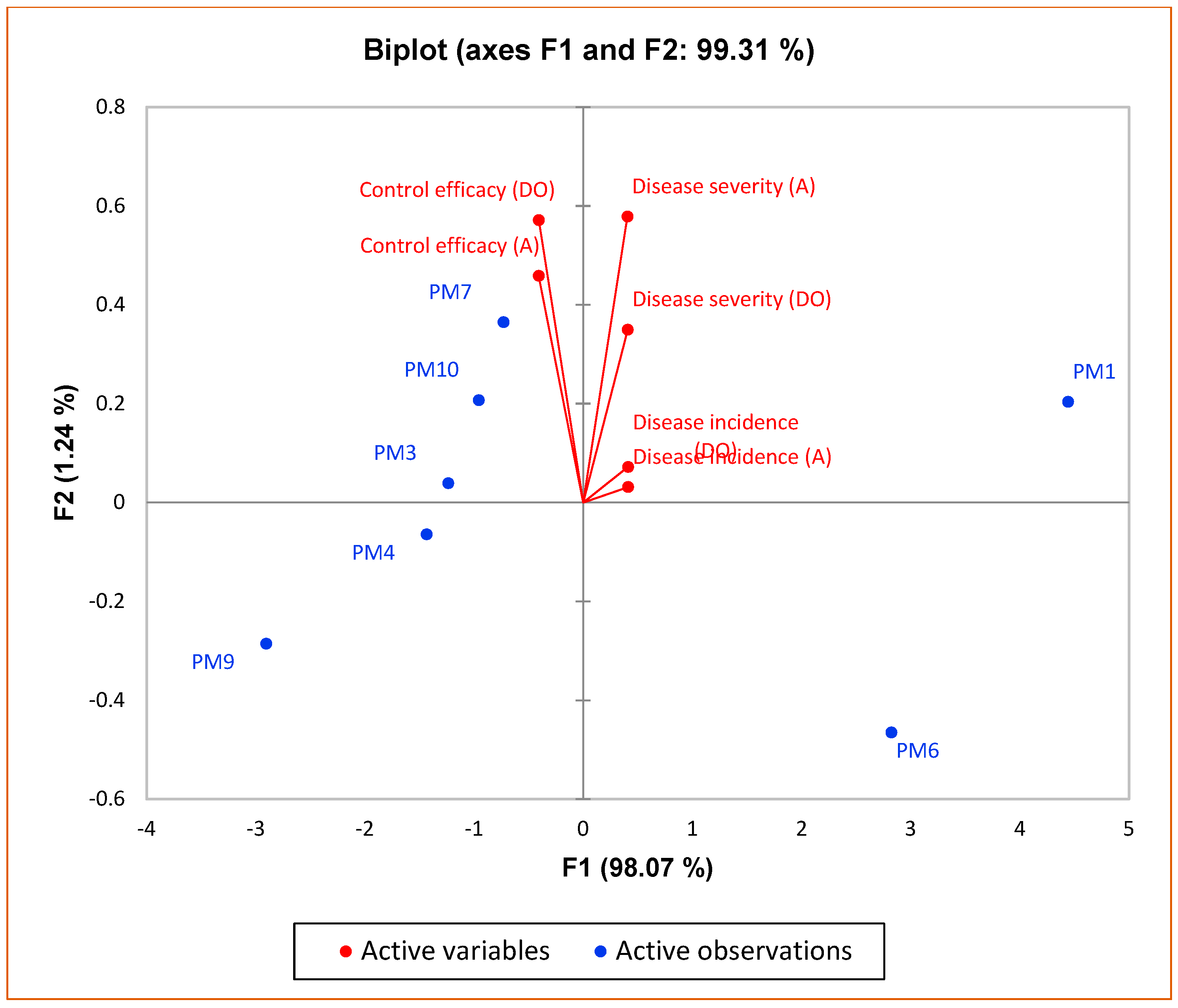
| Treatments | Damping Off | Anthracnose | ||||
|---|---|---|---|---|---|---|
| Disease Incidence (%) | Disease Severity (%) | Control Efficacy | Disease Incidence (%) | Disease Severity (%) | Control Efficacy | |
| PM1 | 7.06 ± 0.38 | 8.75 ± 0.24 | 24.83 ± 1.89 | 8.01 ± 0.41 | 9.70 ± 0.26 | 22.75 ± 2.01 |
| PM3 | 2.96 ± 0.21 | 3.47 ± 0.41 | 78.80 ± 0.46 | 2.70 ± 0.23 | 2.41 ± 0.24 | 80.32 ± 1.25 |
| PM4 | 2.86 ± 0.17 | 3.10 ± 0.03 | 79.26 ± 0.38 | 2.79 ± 0.36 | 1.78 ± 0.28 | 82.65 ± 2.46 |
| PM6 | 5.80 ± 0.34 | 6.44 ± 0.21 | 29.60 ± 1.05 | 6.10 ± 0.37 | 7.20 ± 0.22 | 31.29 ± 1.21 |
| PM7 | 3.44 ± 0.31 | 3.96 ± 0.42 | 77.35 ± 2.51 | 3.22 ± 0.20 | 3.90 ± 0.25 | 81.80 ± 3.48 |
| PM9 | 1.81 ± 0.31 | 0.62 ± 0.01 | 94.50 ± 1.08 | 1.75 ± 0.29 | 0.69 ± 0.10 | 95.64 ± 1.07 |
| PM10 | 2.98 ± 0.19 | 3.55 ± 0.37 | 74.56 ± 1.63 | 3.29 ± 0.67 | 3.41 ± 0.29 | 84.40 ± 2.44 |
| LSD | 0.905 | 0.702 | 7.532 | 0.916 | 0.605 | 8.364 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaushal, M.; Mandyal, P.; Kaushal, R. Field Based Assessment of Capsicum annuum Performance with Inoculation of Rhizobacterial Consortia. Microorganisms 2019, 7, 89. https://doi.org/10.3390/microorganisms7030089
Kaushal M, Mandyal P, Kaushal R. Field Based Assessment of Capsicum annuum Performance with Inoculation of Rhizobacterial Consortia. Microorganisms. 2019; 7(3):89. https://doi.org/10.3390/microorganisms7030089
Chicago/Turabian StyleKaushal, Manoj, Priyanka Mandyal, and Rajesh Kaushal. 2019. "Field Based Assessment of Capsicum annuum Performance with Inoculation of Rhizobacterial Consortia" Microorganisms 7, no. 3: 89. https://doi.org/10.3390/microorganisms7030089
APA StyleKaushal, M., Mandyal, P., & Kaushal, R. (2019). Field Based Assessment of Capsicum annuum Performance with Inoculation of Rhizobacterial Consortia. Microorganisms, 7(3), 89. https://doi.org/10.3390/microorganisms7030089




