Tetrachloromethane-Degrading Bacterial Enrichment Cultures and Isolates from a Contaminated Aquifer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Sampling of Groundwater and Concentration of Microorganisms
2.3. Cultivation Methods
2.4. Gas Chromatography
2.5. Determination of CCl4 Degradation Rates and Chloride Production
2.6. Bacterial Phenotyping
2.7. DNA Purification
2.8. T-RFLP Analysis
2.9. Denaturing High Performance Liquid Chromatography (D-HPLC) Analysis
2.10. Fluorescence in Situ Hybridization
2.11. 16S rRNA Gene Cloning, Sequencing and Analysis
2.12. Accession Numbers
3. Results
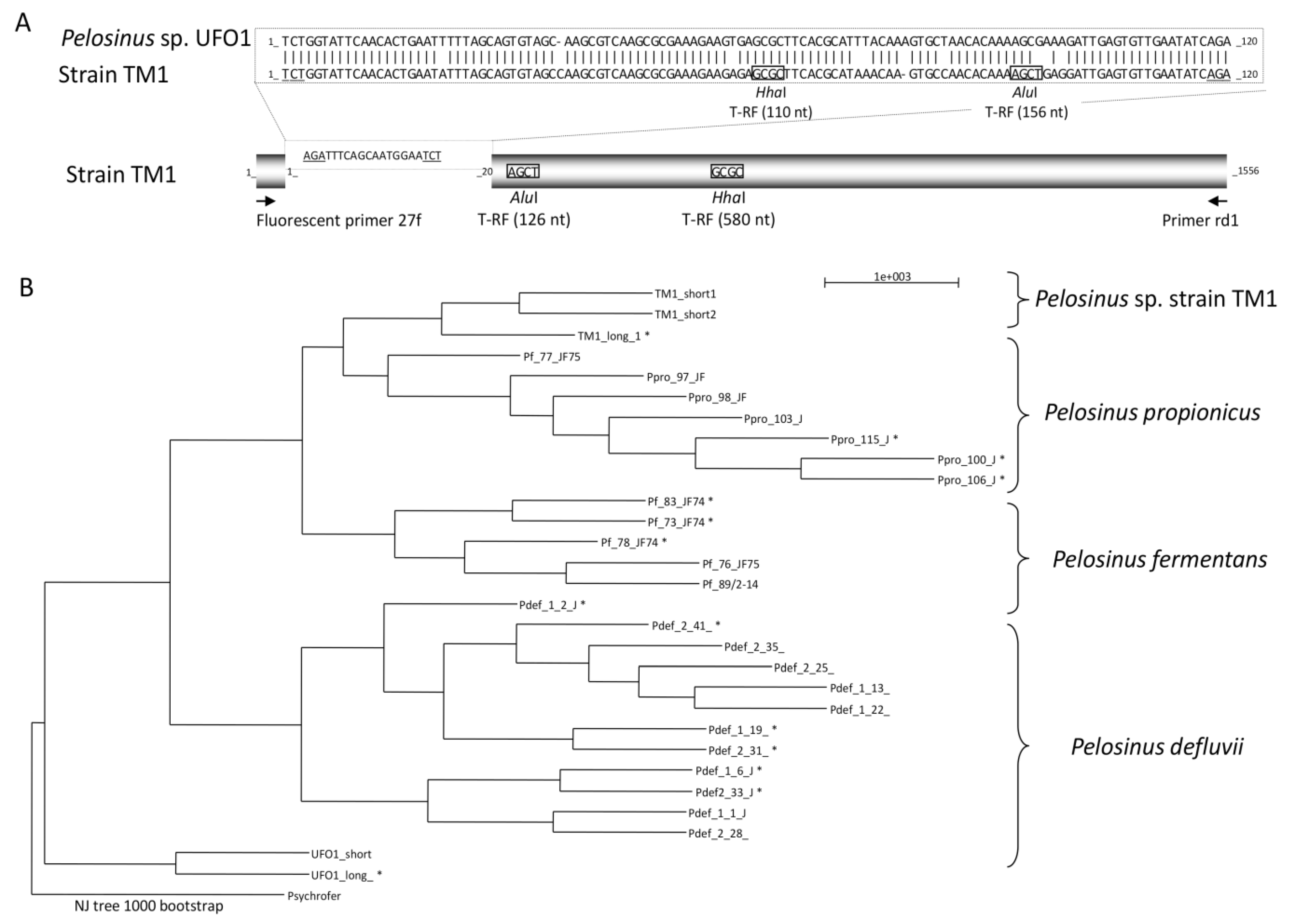
3.1. CCl4-Degrading Enrichments and Isolation of Pelosinus sp. TM1
| Name | Proposed affiliation/Accession Number a | Best Hit a | |
|---|---|---|---|
| Taxon/Accession Number | Identity (%)/Length (bp) | ||
| Cultivated isolates | |||
| Strain TM1 | Pelosinus sp./FN689722,FN68972, KP219716 | Pelosinus propionicus TmPM3/AM258974 | 99/1549 |
| Strain TM2 | Klebsiella sp./FN689724 | Klebsiella oxytoca M1/CP008841 | 98/1437 |
| Strain TM5 | Peptostreptococcaceae bacterium/FN689725 | Clostridium ruminantium Neferana 2/KJ722512 | 99/1382 |
| Strain C5_1 | Staphylococcus sp. | Staphylococcus aureus sub sp. Aureus/BA000017 | 99/535 |
| Uncultivated OTUs | |||
| C1_2, C2_3, C3_3 | No sequence data | No T-RFLP database match | n.a.b |
| C2_1 | No sequence data | Uncultured actinobacteria/AJ575500, AJ575552, AJ575544, AJ575549, AJ575559; Uncultured gamma-proteobacterium/AB294927 | n.a. n.a. |
| C3_2 | Clostridiales/FN689728 | Uncultured Clostridiaceae bacterium B-LO-T0_OTU18/FM204961 | 99/439 |
| C4_1 | Bacteroidetes/FN689727 | Uncultured bacterium ASSO-106/JN391680 | 97/402 |
| C4_2 | Lactobacillales/FN689726 | Uncultured Trichococcus sp. D4R-61/AB331457 | 100/479 |
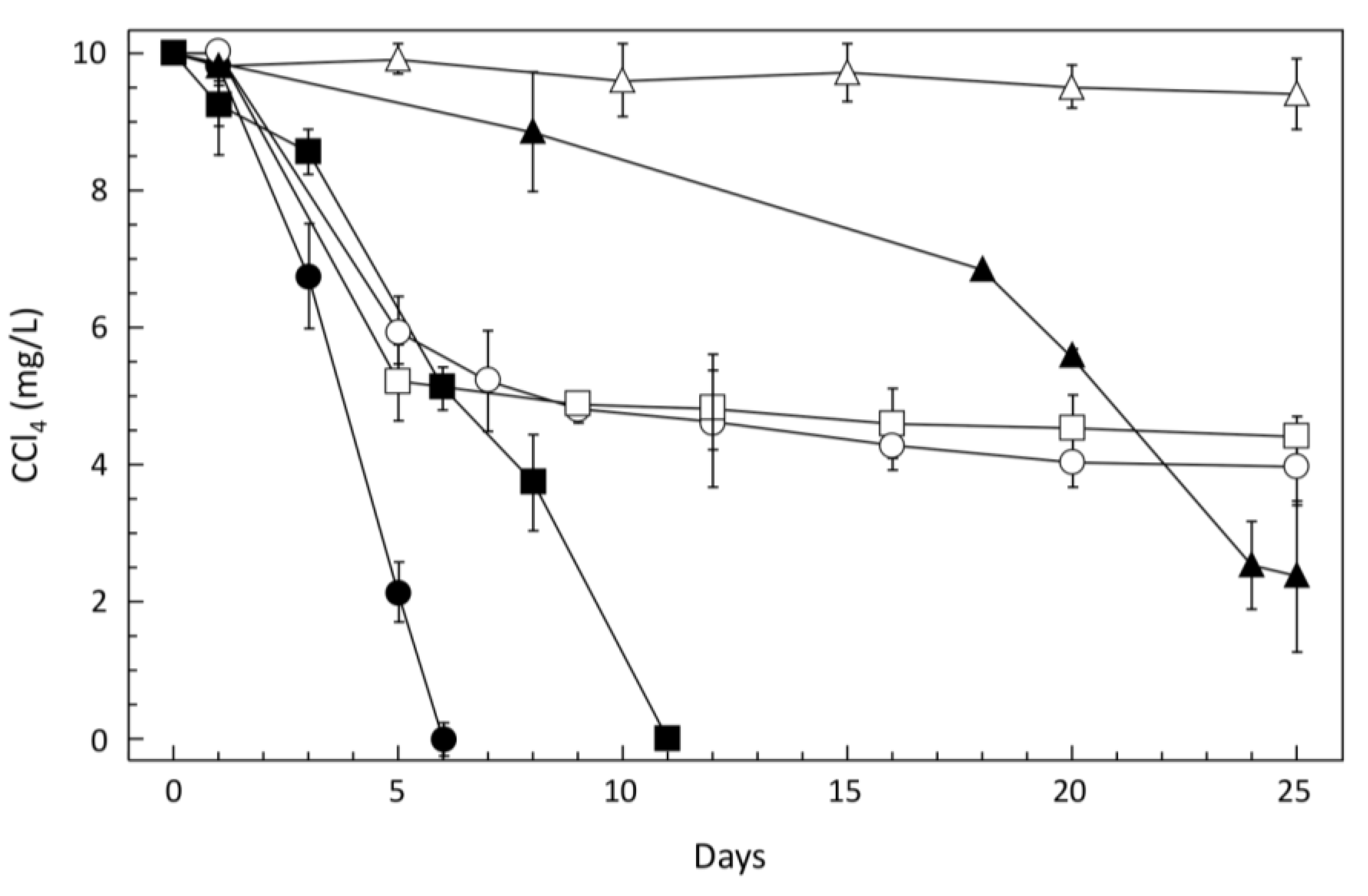
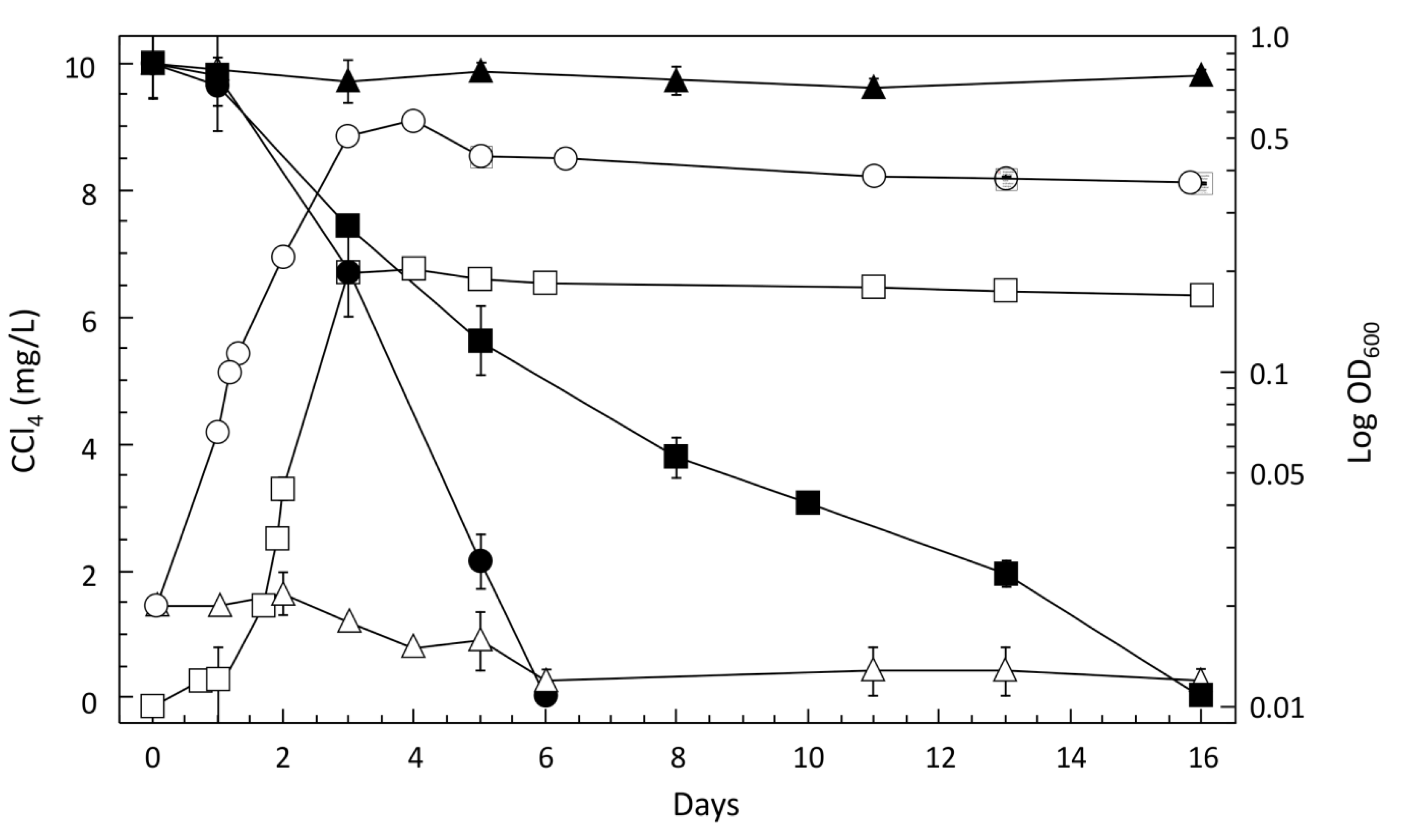
3.2. Characteristics of CCl4-Degrading Pelosinus sp. TM1
| Culture Fraction a | Degradation Rate | ||
|---|---|---|---|
| μg mL (OD 1.0) −1 day−1 | μg mg protein−1 day−1 | ||
| Liquid culture in C medium | 1.9 ± 0.6 | 2.2 ± 0.7 | |
| Resuspended cells | Phosphate buffer b | 9.1 ± 1.1 | 10.6 ± 1.3 |
| Phosphate buffer b, heat-treated | 13.6 ± 0.9 | 15.8 ± 1.0 | |
| Phosphate buffer b, heat-treated, oxygen-exposed | <0.1 | <0.1 | |
| Supernatant | Filter-sterilized | 1.4 ± 0.3 | n.d. c |
| Filter-sterilized, heat-treated | 1.6 ± 0.4 | n.d. | |
| Filter-sterilized, oxygen-exposed | <0.1 | n.d. | |
| Filter-sterilized, oxygen-exposed, cysteine/Na2S-treated | 1.4 ± 0.4 | n.d. | |
4. Discussion
CCl4 Degradation by Pelosinus sp. TM1 is a Co-Metabolic Process
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Doherty, R.E. A history of the production and use of carbon tetrachloride, tetrachloroethylene, trichloroethylene and 1,1,1-trichloroethane in the united states: Part 1—Historical background; carbon tetrachloride and tetrachloroethylene. Environ. Forensics 2000, 1, 69–81. [Google Scholar] [CrossRef]
- Weber, L.W.; Boll, M.; Stampfl, A. Hepatotoxicity and mechanism of action of haloalkanes: Carbon tetrachloride as a toxicological model. Crit. Rev. Toxicol. 2003, 33, 105–136. [Google Scholar] [CrossRef] [PubMed]
- Liang, Q.; Newman, P.A.; Daniel, J.S.; Reimann, S.; Hall, B.D.; Dutton, G.; Kuijpers, L.J.M. Constraining the carbon tetrachloride (CCl4) budget using its global trend and inter-hemispheric gradient. Geophys. Res. Lett. 2014, 41, 5307–5315. [Google Scholar] [CrossRef]
- Carpenter, L.J.; Reimann, S. Update on ozone-depleting substances (ODSs) and other gases of interest to the Montreal protocol. World Meteorological Organization Global Ozone Research and Monitoring Project—Report No. 55. 2014. Chapter 1. p. 101. Available online: http://hdl.handle.net/2268/175647 (accessed on 5 January 2015).
- Vogel, T.M.; Criddle, C.S.; McCarty, P.L. Transformations of halogenated aliphatic compounds. Environ. Sci. Technol. 1987, 21, 722–736. [Google Scholar] [CrossRef] [PubMed]
- Penny, C.; Vuilleumier, S.; Bringel, F. Microbial degradation of tetrachloromethane: Mechanisms and perspectives for bioremediation. FEMS Microbiol. Ecol. 2010, 74, 257–275. [Google Scholar] [CrossRef] [PubMed]
- Hashsham, S.A.; Scholze, R.; Freedman, D.L. Cobalamin-enhanced anaerobic biotransformation of carbon tetrachloride. Environ. Sci. Technol. 1995, 29, 2856–2863. [Google Scholar] [CrossRef] [PubMed]
- Krone, U.E.; Thauer, R.K.; Hogenkamp, H.P.; Steinbach, K. Reductive formation of carbon monoxide from CCl4 and FREONs 11, 12, and 13 catalyzed by corrinoids. Biochemistry 1991, 30, 2713–2719. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.S.; Boonchayaanant, B.; Tang, W.; Trost, B.M.; Criddle, C.S. Simple menaquinones reduce carbon tetrachloride and iron (III). Biodegradation 2009, 20, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Doong, R.-A.; Lee, C.-C.; Lien, C.-M. Enhanced dechlorination of carbon tetrachloride by Geobacter sulfurreducens in the presence of naturally occurring quinones and ferrihydrite. Chemosphere 2014, 97, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, F.J.; Martínez, C.M.; Gonzalez-Estrella, J.; Márquez, A.; Arriaga, S. Kinetics during the redox biotransformation of pollutants mediated by immobilized and soluble humic acids. Appl. Microbiol. Biotechnol. 2013, 97, 2671–2679. [Google Scholar] [CrossRef] [PubMed]
- Picardal, F.W.; Arnold, R.G.; Couch, H.; Little, A.M.; Smith, M.E. Involvement of cytochromes in the anaerobic biotransformation of tetrachloromethane by Shewanella putrefaciens 200. Appl. Environ. Microbiol. 1993, 59, 3763–3770. [Google Scholar] [PubMed]
- Krone, U.E.; Laufer, K.; Thauer, R.K.; Hogenkamp, H.P.C. Coenzyme F430 as a possible catalyst for the reductive dehalogenation of chlorinated C1 hydrocarbons in methanogenic bacteria. Biochemistry 1989, 28, 10061–10065. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Barajas, C.; Field, J.A. Enhancement of anaerobic carbon tetrachloride biotransformation in methanogenic sludge with redox active vitamins. Biodegradation 2005, 16, 215–228. [Google Scholar] [CrossRef] [PubMed]
- Lewis, T.A.; Paszczynski, A.; Gordon-Wylie, S.W.; Jeedigunta, S.; Lee, C.H.; Crawford, R.L. Carbon tetrachloride dechlorination by the bacterial transition metal chelator pyridine-2,6-bis(thiocarboxylic acid). Environ. Sci. Technol. 2001, 35, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Field, J.A.; Sierra-Alvarez, R. Biodegradability of chlorinated solvents and related chlorinated aliphatic compounds. Rev. Environ. Sci. Biotechnol. 2004, 3, 185–254. [Google Scholar] [CrossRef]
- De Best, J.H.; Hunneman, P.; Doddema, H.J.; Janssen, D.B.; Harder, W. Transformation of carbon tetrachloride in an anaerobic packed-bed reactor without addition of another electron donor. Biodegradation 1999, 10, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Egli, C.; Tschan, T.; Scholtz, R.; Cook, A.M.; Leisinger, T. Transformation of tetrachloromethane to dichloromethane and carbon dioxide by Acetobacterium woodii. Appl. Environ. Microbiol. 1988, 54, 2819–2824. [Google Scholar] [PubMed]
- Pandey, J.; Chauhan, A.; Jain, R.K. Integrative approaches for assessing the ecological sustainability of in situ bioremediation. FEMS Microbiol. Rev. 2009, 33, 324–375. [Google Scholar] [CrossRef] [PubMed]
- Penny, C.; Nadalig, T.; Alioua, M.; Gruffaz, C.; Vuilleumier, S.; Bringel, F. Coupling of denaturing high-performance liquid chromatography and terminal restriction fragment length polymorphism with precise fragment size calling for microbial community profiling and characterization. Appl. Environ. Microbiol. 2010, 76, 648–651. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Noble, R.T.; Steele, J.A.; Schwalbach, M.S.; Hewson, I.; Fuhrman, J.A. Virus and prokaryote enumeration from planktonic aquatic environments by epifluorescence microscopy with SYBR Green I. Nat. Protoc. 2007, 2, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Gälli, R.; McCarty, P.L. Biotransformation of 1,1,1-trichloroethane, trichloromethane, and tetrachloromethane by a Clostridium sp. Appl. Environ. Microbiol. 1989, 55, 837–844. [Google Scholar] [PubMed]
- Lee, C.H.; Lewis, T.A.; Paszczynski, A.; Crawford, R.L. Identification of an extracellular catalyst of carbon tetrachloride dehalogenation from Pseudomonas stutzeri strain KC as pyridine-2,6-bis(thiocarboxylate). Biochem. Biophys. Res. Commun. 1999, 261, 562–566. [Google Scholar] [CrossRef] [PubMed]
- Novak, P.J.; Daniels, L.; Parkin, G.F. Enhanced dechlorination of carbon tetrachloride and chloroform in the presence of elemental iron and Methanosarcina barkeri, Methanosarcina thermophila, or Methanosaeta concillii. Environ. Sci. Technol. 1998, 32, 1438–1443. [Google Scholar] [CrossRef]
- Dybas, M.J.; Tatara, G.M.; Criddle, C.S. Localization and characterization of the carbon tetrachloride transformation activity of Pseudomonas sp. strain KC. Appl. Environ. Microbiol. 1995, 61, 758–762. [Google Scholar] [PubMed]
- Vogel, K.; Bluemer, N.; Korthals, M.; Mittelstaedt, J.; Garn, H.; Ege, M.; von Mutius, E.; Gatermann, S.; Bufe, A.; Goldmann, T.; et al. Animal shed Bacillus licheniformis spores possess allergy-protective as well as inflammatory properties. J. Allergy Clin. Immunol. 2008, 122, 307–312. [Google Scholar] [CrossRef] [PubMed]
- DeLong, E.F. Archaea in coastal marine environments. Proc. Natl. Acad. Sci. USA 1992, 89, 5685–5689. [Google Scholar] [CrossRef] [PubMed]
- Shyu, C.; Soule, T.; Bent, S.J.; Foster, J.A.; Forney, L.J. Mica: A web-based tool for the analysis of microbial communities based on terminal-restriction fragment length polymorphisms of 16S and 18S rRNA genes. Microbial Ecol. 2007, 53, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Blackwood, C.B.; Hudleston, D.; Zak, D.R.; Buyer, J.S. Interpreting ecological diversity indices applied to terminal restriction fragment length polymorphism data: Insights from simulated microbial communities. Appl. Environ. Microbiol. 2007, 73, 5276–5283. [Google Scholar] [CrossRef] [PubMed]
- Stahl, D.A.; Amann, R. Development and application of nucleic acid probes. Nucleic Acids Tech. Bact. Syst. 1991, 8, 207–248. [Google Scholar]
- Weisburg, W.G.; Barns, S.M.; Pelletier, D.A.; Lane, D.J. 16S ribosomal DNA amplification for phylogenetic study. J. Bacteriol. 1991, 173, 697–703. [Google Scholar] [PubMed]
- Cole, J.R.; Wang, Q.; Fish, J.A.; Chai, B.; McGarrell, D.M.; Sun, Y.; Brown, C.T.; Porras-Alfaro, A.; Kuske, C.R.; Tiedje, J.M. Ribosomal database project: Data and tools for high throughput rRNA analysis. Nucleic Acids Res. 2014, 42, D633–D642. [Google Scholar] [CrossRef] [PubMed]
- Li, X.M.; Zhou, S.G.; Li, F.B.; Wu, C.Y.; Zhuang, L.; Xu, W.; Liu, L. Fe(iii) oxide reduction and carbon tetrachloride dechlorination by a newly isolated Klebsiella pneumoniae strain l17. J. Appl. Microbiol. 2009, 106, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Stromeyer, S.A.; Stumpf, K.; Cook, A.M.; Leisinger, T. Anaerobic degradation of tetrachloromethane by Acetobacterium woodii: Separation of dechlorinative activities in cell extracts and roles for vitamin B12 and other factors. Biodegradation 1992, 3, 113–123. [Google Scholar] [CrossRef]
- Moe, W.M.; Stebbing, R.E.; Rao, J.U.; Bowman, K.S.; Nobre, M.F.; da Costa, M.S.; Rainey, F.A. Pelosinus defluvii sp. nov., isolated from chlorinated solvent-contaminated groundwater, emended description of the genus Pelosinus and transfer of Sporotalea propionica to Pelosinus propionicus comb. nov. Int. J. Syst. Evol. Microbiol. 2012, 62, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.; Utturkar, S.; Magnuson, T.; Ray, E.; Poole, F.; Lancaster, W.; Thorgersen, M.; Adams, M.; Elias, D. Complete genome sequence of Pelosinus sp. strain UFO1 assembled using single-molecule real-time DNA sequencing technology. Genome Announc. 2014, 2, e00881–e00814. [Google Scholar] [CrossRef] [PubMed]
- Boga, H.I.; Ji, R.; Ludwig, W.; Brune, A. Sporotalea propionica gen. nov. sp. nov., a hydrogen-oxidizing, oxygen-reducing, propionigenic firmicute from the intestinal tract of a soil-feeding termite. Arch. Microbiol. 2007, 187, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Men, Y.; Seth, E.C.; Yi, S.; Allen, R.H.; Taga, M.E.; Alvarez-Cohen, L. Sustainable growth of Dehalococcoides mccartyi 195 by corrinoid salvaging and remodeling in defined lactate-fermenting consortia. Appl. Environ. Microbiol. 2014, 80, 2133–2141. [Google Scholar] [CrossRef] [PubMed]
- Justicia-Leon, S.D.; Higgins, S.; Mack, E.E.; Griffiths, D.R.; Tang, S.; Edwards, E.A.; Löffler, F.E. Bioaugmentation with distinct Dehalobacter strains achieves chloroform detoxification in microcosms. Environ. Sci. Technol. 2014, 48, 1851–1858. [Google Scholar] [CrossRef] [PubMed]
- Men, Y.; Lee, P.K.H.; Harding, K.C.; Alvarez-Cohen, L. Characterization of four TCE-dechlorinating microbial enrichments grown with different cobalamin stress and methanogenic conditions. Appl. Microbiol. Biotechnol. 2013, 97, 6439–6450. [Google Scholar] [CrossRef] [PubMed]
- Boopathy, R. Anaerobic biotransformation of carbon tetrachloride under various electron acceptor conditions. Bioresource Technol. 2002, 84, 69–73. [Google Scholar] [CrossRef]
- Criddle, C.S.; DeWitt, J.T.; McCarty, P.L. Reductive dehalogenation of carbon tetrachloride by Escherichia coli K-12. Appl. Environ. Microbiol. 1990, 56, 3247–3254. [Google Scholar] [PubMed]
- Mazumder, T.K.; Nishio, N.; Fukuzaki, S.; Nagai, S. Production of extracellular vitamin B12 compounds from methanol by Methanosarcina barkeri. Appl. Microbiol. Biotechnol. 1987, 26, 511–516. [Google Scholar] [CrossRef]
- Koons, B.W.; Baeseman, J.L.; Novak, P.J. Investigation of cell exudates active in carbon tetrachloride and chloroform degradation. Biotechnol. Bioeng. 2001, 74, 12–17. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Penny, C.; Gruffaz, C.; Nadalig, T.; Cauchie, H.-M.; Vuilleumier, S.; Bringel, F. Tetrachloromethane-Degrading Bacterial Enrichment Cultures and Isolates from a Contaminated Aquifer. Microorganisms 2015, 3, 327-343. https://doi.org/10.3390/microorganisms3030327
Penny C, Gruffaz C, Nadalig T, Cauchie H-M, Vuilleumier S, Bringel F. Tetrachloromethane-Degrading Bacterial Enrichment Cultures and Isolates from a Contaminated Aquifer. Microorganisms. 2015; 3(3):327-343. https://doi.org/10.3390/microorganisms3030327
Chicago/Turabian StylePenny, Christian, Christelle Gruffaz, Thierry Nadalig, Henry-Michel Cauchie, Stéphane Vuilleumier, and Françoise Bringel. 2015. "Tetrachloromethane-Degrading Bacterial Enrichment Cultures and Isolates from a Contaminated Aquifer" Microorganisms 3, no. 3: 327-343. https://doi.org/10.3390/microorganisms3030327
APA StylePenny, C., Gruffaz, C., Nadalig, T., Cauchie, H.-M., Vuilleumier, S., & Bringel, F. (2015). Tetrachloromethane-Degrading Bacterial Enrichment Cultures and Isolates from a Contaminated Aquifer. Microorganisms, 3(3), 327-343. https://doi.org/10.3390/microorganisms3030327






