Identification of Multiple Bacteriocins in Enterococcus spp. Using an Enterococcus-Specific Bacteriocin PCR Array
Abstract
:1. Introduction
2. Experimental Section
2.1. Bacterial Strains, Storage, and Growth Conditions
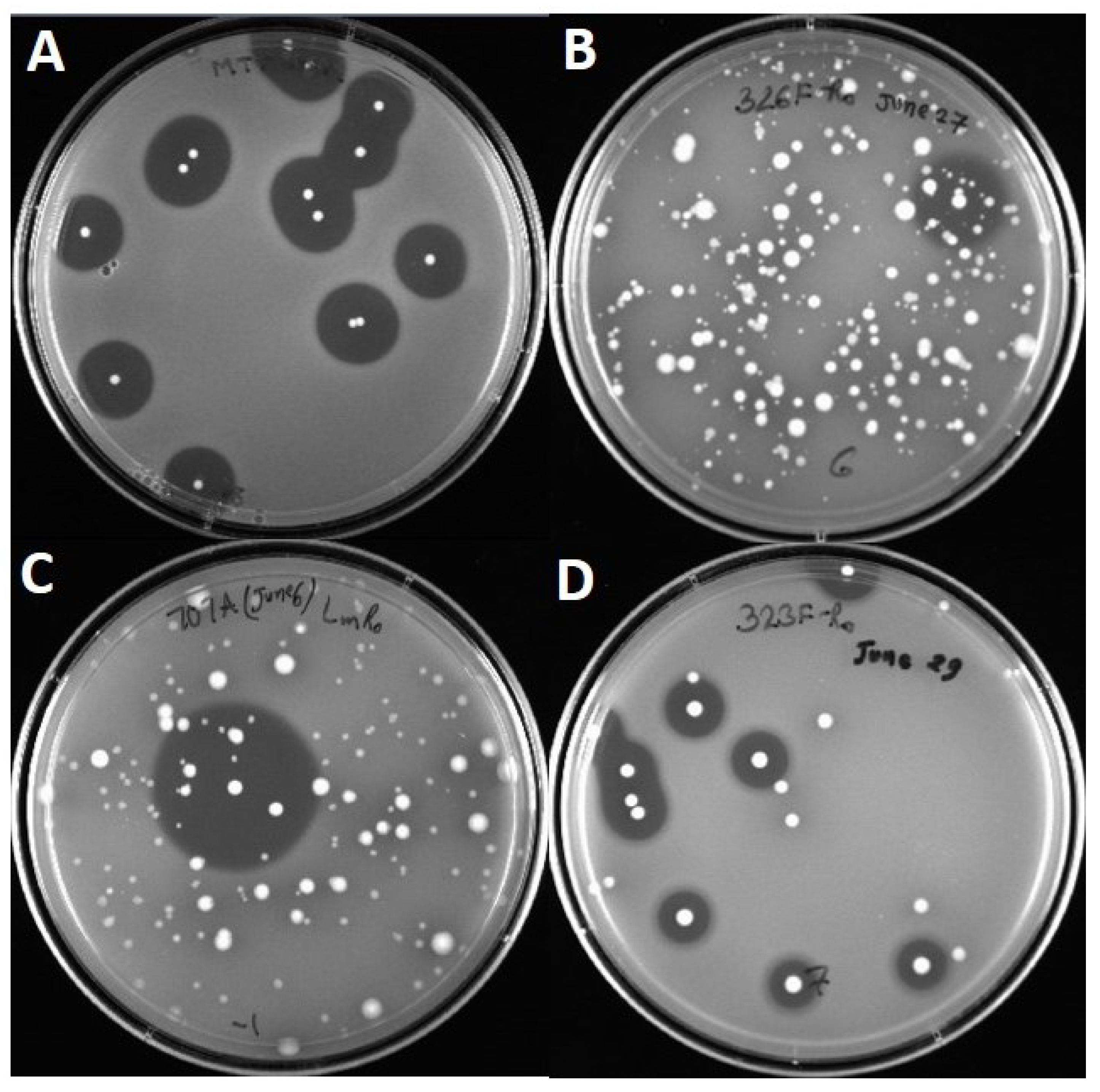
2.2. Activity against Listeria Monocytogenes
2.3. Primer Creation
2.4. PCR Detection of Enterococcal Bacteriocin Genes
3. Results
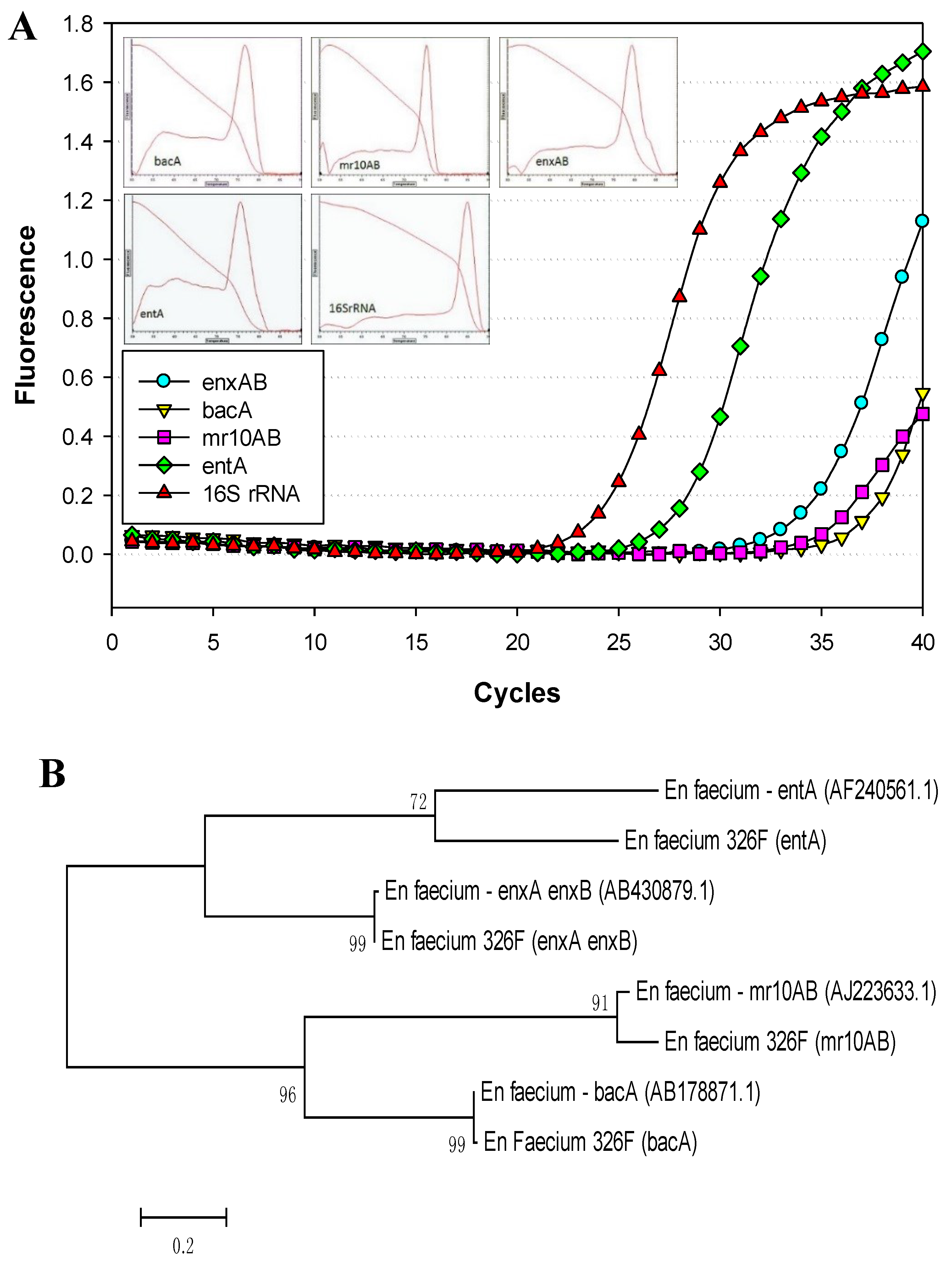
3.1. Primer Design
3.2. PCR Detection of Enterococcal Bacteriocin Genes by Real-Time PCR
3.3. Inhibition of L. monocytogenes 39-2 R0 (Wild-Type) and R1 (curvaticin FS47R)
| Primer | Target Gene | Sequence (5′→3′) | Product Size (bp) | Included Genes | Homologous Genes |
|---|---|---|---|---|---|
| 1 | Avicin A (avcA)—FJ851402.1 | 236 | avicin A precurson (avcA) immunity protein (avcI) divergicin-like bact (avcB) | sakacin X (sakX) | |
| Forward | ACG CGA AAT GAA GAA TGT TG | ||||
| Reverse | TTT CAT TTC CGC CAG AAA AC | ||||
| 2 | Columbicin A (colA)—EF033111.1 | 299 | columbicin A (colA) hypothetical protein (orfB) | bovicin A (bovA) enterocin AS-48 | |
| Forward | TTT TTC TTG GGT TAT TTA CAG GAA | ||||
| Reverse | ATG TGC AAT GGG CAA AAA CT | ||||
| 3 | Durancin Q (duqQ)—AB284369.1 | 384 | immunity protein (duqI) durancin Q (duqQ) inducing peptide (duqF) | durancin TW-49 (durM) | |
| Forward | GCA CTG ATT CCG GCA CTA AT | ||||
| Reverse | CGT AAC TCT AAT GGC GGG AAG | ||||
| 4 | Enterocin 96 (ent96)—FJ769024.1 | 291 | enterocin 96 | - | |
| Forward | GTG GAG AGG ACG AAA GGA GA | ||||
| Reverse | TTG ATT AGT GGA GAG GAC GGT TA | ||||
| 5 | Enterocin mr10A/mr10B (mr10AB) | 247 | enterocin mr10A (mr10A) enterocin mr10B (mr10B) | enterocin JSB (entJSB) enterocin NA (entNA) enterocin NB (entNB) enterocin L50A (entL50A) enterocin L50B (entL50B) enterocin 62-6A (ent626A) enterocin 62-6B (ent626B) enterocin RJ-11 | |
| Forward | ATG GGA GCA ATC GCA AAA T | ||||
| Reverse | CAT CCT TGT CCG ATA AAC TGC | ||||
| 6 | Enterocin C (entC)—FU862242.1 | 506 | enterocin C1 (entC1) enterocin C2 (entC2) enterocin C immunity (entCI) | enterocin 1081A enterocin 1071B | |
| Forward | AGG TCC AGC TGC TTA TTG GA | ||||
| Reverse | CCA TTA GAA TGA ATA CGC TAA AGA AA | ||||
| 7 | Enterocin SE-K4 (entSE-K4)—AB092692.1 | 608 | enterocin SE-K4 (entSE-K4) enterocin precursor (orf7) entSE-K4 homologue (orf8) entSE-K4 immunity (orf9) | bacteriocin II (D78257.1) enterocin TW-21 bacteriocin 31 | |
| Forward | ATG TAG AAG CCG CCA CGT AT | ||||
| Reverse | AAT CCC AAT CAT CCC ACA AA | ||||
| 8 | Enterocin EJ97(ej97a)—AJ490170.1 | 104 | enterocin ej97 | - | |
| Forward | AAA GCG ATG ATT AAG AAG TTT CC | ||||
| Reverse | TCC CAA GGA TAA CGA CCG TA | ||||
| 9 | Enterocin Wα/Wβ (enwAB)—AB600897.1 | 423 | enterocin W alpha (enwA) enterocin W beta (enw B) | - | |
| Forward | GGG GTT GAA TTA TTG TAG AAA GGA | ||||
| Reverse | AAC TAG CCT CTA CCG CCA CA | ||||
| 10 | Enterocin Q (entqA)—DQ832184.1 | 231 | enterocin Q (entqA) | - | |
| Forward | ATC ACA AAG TGA GCC CCT GT | ||||
| Reverse | TGG TAT CGC AAA ATG GAT GA | ||||
| 11 | Enterocin P (entP)—AF005726.1 | 431 | enterocin P (entP) enterocin P immunity (entQ) | - | |
| Forward | TTC CCC GAA GAA TAC AAA TGA | ||||
| Reverse | AAT TTC TGG GGT GGC TAA TG | ||||
| 12 | Enterocin A (entA)—AF240561.1 | 362 | enterocin A (entA) immunity protein (entI) | - | |
| Forward | AAA ATA AAT GTA CGG TCG ATT GG | ||||
| Reverse | CCA GCA GTT CTT CCA ATT TCA | ||||
| 13 | Enterocin B (entB)—U87997,1 | 257 | enterocin B (entB) | enterocin CRL35 | |
| Forward | CAG AGT TCC CAA CTG TTT GCT | ||||
| Reverse | AGC CCA TGC TAG TGG TCCT T | ||||
| 14 | Enterocin Xα/Xβ (enxAB)—AB430879.1 | 321 | enterocin X alpha (enxA) enteorcin X beta (enxB) | - | |
| Forward | GGACAATTTATGGGTAAACAAGC | ||||
| Reverse | TACGTCCACCATTCCAACCT | ||||
| 15 | Bacteriocin T8 (bacA)—AB178871.1 | 469 | bacteriocin precursor (bacA) hypothetical immunity protein (bacB) | hiracin JM79 Bac43 | |
| Forward | TTGTCTAGCTGGCATCGGTA | ||||
| Reverse | CCAATAGAAGCCCATCCTCT | ||||
| 16 | Mundticin KS (munA)—KC291253.1 | 285 | mundticin KS (munA) | mundticin L (munL) enterocin HF | |
| Forward | AAA AGG GTG CAG TGT TGA TTG | ||||
| Reverse | TCC ACT GAA ATC CAT GAA TGA | ||||
| Isolate | Species | entA | mr10AB | enxAB | bacA | entP | entB | munA |
|---|---|---|---|---|---|---|---|---|
| FS707 | En. durans | 99% | ||||||
| BJ-12 | En. faecalis | 100% 2 | ||||||
| BJ-13 | En. faecalis | 99% | ||||||
| BJ-19 | En. faecalis | 100% | ||||||
| BJ-27 | En. faecalis | 100% | 96% | |||||
| 326F | En. faecium | 100% | 92% | 99% | 99% | |||
| FS56-1 | En. faecium | 100% | 97% | |||||
| FS97-2 | En. faecium | 100% | 99% | 100% | ||||
| JCP B-5 | En. faecium | 97% | 100% | 99% | ||||
| JCP M-2 | En. faecium | 95% | 100% | 99% | ||||
| JCP-9 | En. faecium | 98% | 100% | 99% | ||||
| Milk12 | En. faecium | 100% | 99% | 100% | ||||
| Milk5 | En. faecium | 100% | 99% | 100% | 99% | |||
| NP-7 | En. faecium | 100% | ||||||
| Pop4 | En. faecium | 100% | 96% | 100% | 99% | |||
| THYME2 | En. faecium | 100% | 100% | |||||
| THYME3 | En. faecium | 100% | ||||||
| 323F | En. hirae | 100% | 97% | |||||
| 323RL1 | En. hirae | 100% | 96% | 99% | ||||
| 341FA | En. hirae | 100% | 93% | 99% | ||||
| FS92 | En. thailandicus | 100% | 97% | |||||
| RP-1 | En. thailandicus | 100% |
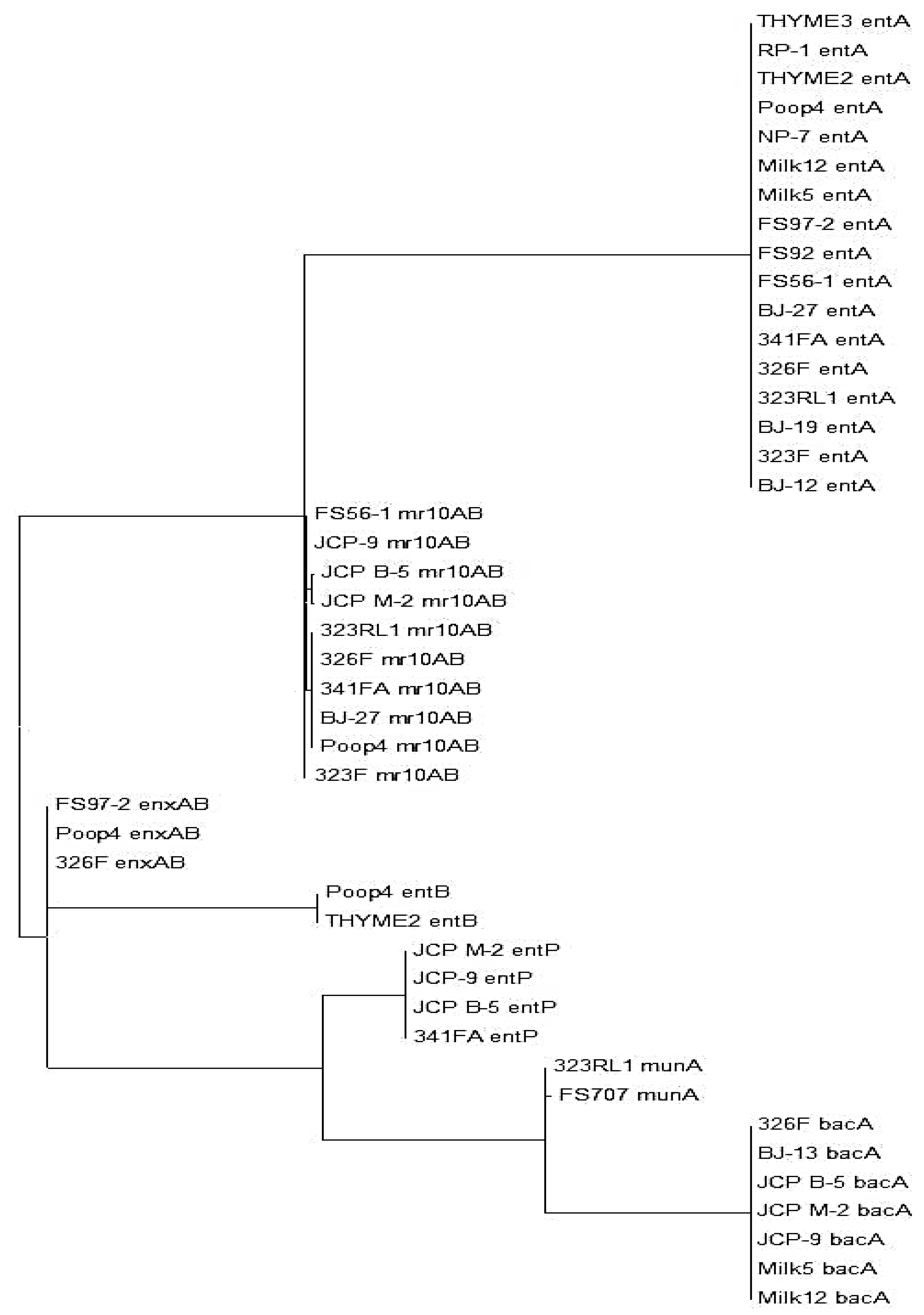
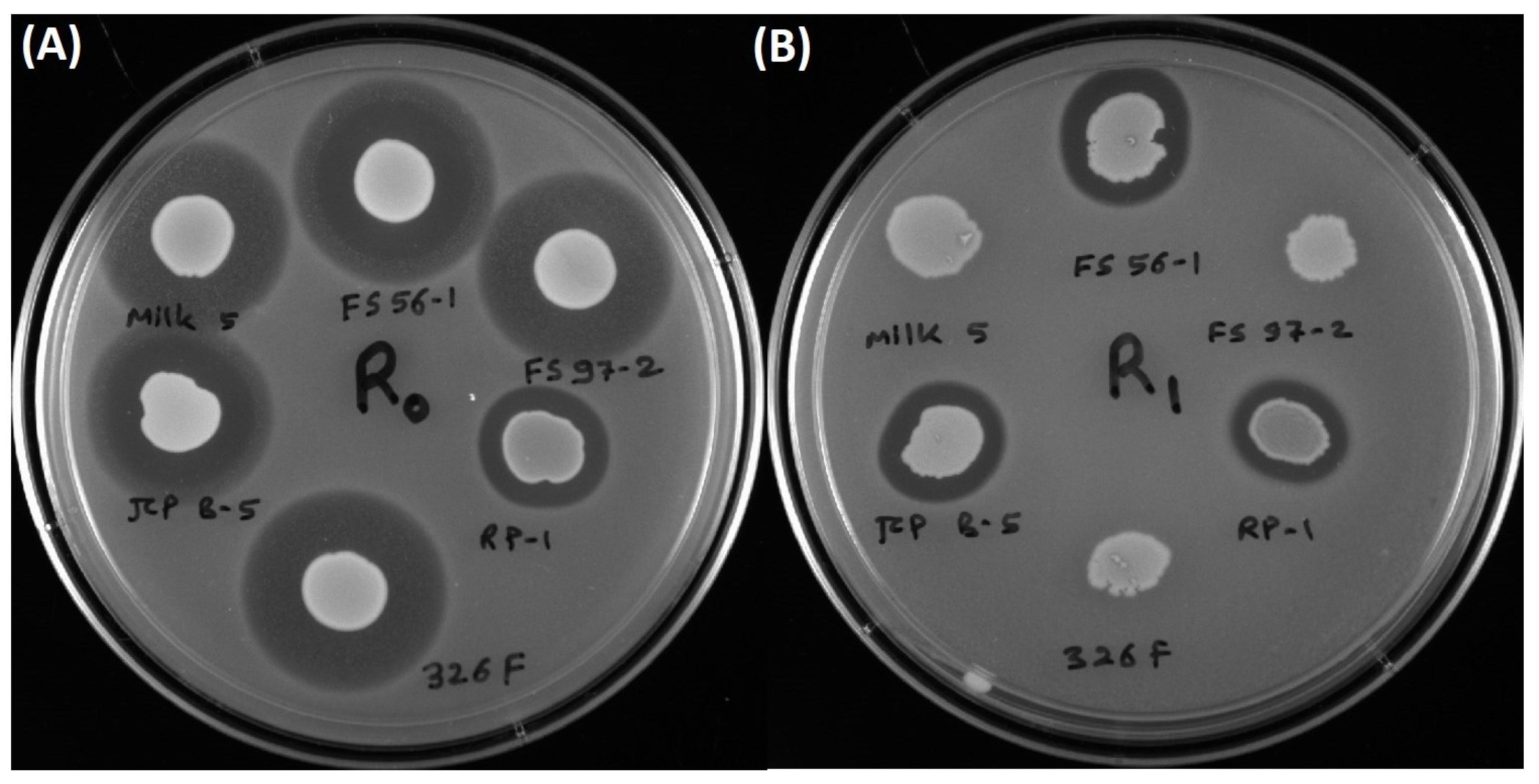
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Devriese, L.A.; Pot, B.; van Damme, L.; Kersters, K.; Haesebrouck, F. Identification of Enterococcus species isolated from foods of animal origin. Int. J. Food Microbiol. 1995, 26, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Pesavento, G.; Calonico, C.; Ducci, B.; Magnanini, A.; Lo Nostro, A. Prevalence and antibiotic resistance of Enterococcus spp. isolated from retail cheese, ready-to-eat salads, ham, and raw meat. Food Microbiol. 2014, 41, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gomes, B.C.; Esteves, C.T.; Palazzo, I.C.V.; Darini, A.L.C.; Felis, G.E.; Sechi, L.A.; Franco, B.D.G.M.; de Martinis, E.C.P. Prevalence and characterization of Enterococcus spp. Isolated from brazilian foods. Food Microbiol. 2008, 25, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Jamet, E.; Akary, E.; Poisson, M.-A.; Chamba, J.-F.; Bertrand, X.; Serror, P. Prevalence and characterization of antibiotic resistant Enterococcus faecalis in french cheeses. Food Microbiol. 2012, 31, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Kearns, A.M.; Freeman, R.; Lightfoot, N.F. Nosocomial enterococci: Resistance to heat and sodium hypochlorite. J. Hosp. Infect. 1995, 30, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Franz, C.M.A.P.; Huch, M.; Abriouel, H.; Holzapfel, W.; Gálvez, A. Enterococci as probiotics and their implications in food safety. Int. J. Food Microbiol. 2011, 151, 125–140. [Google Scholar] [CrossRef] [PubMed]
- Surawicz, C.M. Probiotics, antibiotic-associated diarrhoea and clostridium difficile diarrhoea in humans. Best Pract. Res. Clin. Gastroenterol. 2003, 17, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, P.; Parani, M. First complete genome sequence of a probiotic Enterococcus faecium strain T-110 and its comparative genome analysis with pathogenic and non-pathogenic Enterococcus faecium genomes. J. Genet. Genomics 2014. [Google Scholar] [CrossRef]
- Liu, X.; Vederas, J.C.; Whittal, R.M.; Zheng, J.; Stiles, M.E.; Carlson, D.; Franz, C.M.A.P.; McMullen, L.M.; van Belkum, M.J. Identification of an n-terminal formylated, two-peptide bacteriocin from Enterococcus faecalis 710c. J. Agric. Food Chem. 2011, 59, 5602–5608. [Google Scholar] [CrossRef] [PubMed]
- Amalaradjou, M.A.R.; Bhunia, A.K. Chapter five—Modern approaches in probiotics research to control foodborne pathogens. Adv. Food Nutr. Res. 2012, 67, 185–239. [Google Scholar] [PubMed]
- Vera Pingitore, E.; Todorov, S.D.; Sesma, F.; de Melo Franco, B.D.G. Application of bacteriocinogenic Enterococcus mundtii CRL35 and Enterococcus faecium ST88Ch in the control of Listeria monocytogenes in fresh minas cheese. Food Microbiol. 2012, 32, 38–47. [Google Scholar]
- Abee, T. Pore-forming bacteriocins of gram-positive bacteria and self-protection mechanisms of producer organisms. FEMS Microbiol. Lett. 1995, 129, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Héchard, Y.; Sahl, H.-G. Mode of action of modified and unmodified bacteriocins from gram-positive bacteria. Biochimie 2002, 84, 545–557. [Google Scholar] [CrossRef] [PubMed]
- Zacharof, M.P.; Lovitt, R.W. Bacteriocins produced by lactic acid bacteria a review article. APCBEE Procedia 2012, 2, 50–56. [Google Scholar] [CrossRef]
- Jagannathan, B.; Gautam, D.; Adhikari, R.; Vijayakumar, P.; Henning, C.; Muriana, P. Phylogenetic identification, microplate assay, and fermentor-based growth conditions to optimize bactericidal activity of bacteriocins produced by lactic acid bacteria. In Proceedings of the annual meeting of Institute of Food Technologists, New Orleans, LA, USA, 21–24 June 2014.
- Garver, K.I.; Muriana, P.M. Detection, identification and characterization of bacteriocin-producing lactic acid bacteria from retail food products. Int. J. Food Microbiol. 1993, 19, 241–258. [Google Scholar] [CrossRef] [PubMed]
- Hammami, R.; Zouhir, A.; Ben Hamida, J.; Fliss, I. BACTIBASE: A new web-accessible database for bacteriocin characterization. BMC Microbiol. 2007, 7, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3—New capabilities and interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. Mega5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Macwana, S.J.; Muriana, P.M. A “bacteriocin pcr array” for identification of bacteriocin-related structural genes in lactic acid bacteria. J. Microbiol. Methods 2012, 88, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Nes, I.F.; Diep, D.B.; Holo, H. Bacteriocin diversity in Streptococcus and Enterococcus. J. Bacteriol. 2006, 189, 1189–1198. [Google Scholar] [CrossRef] [PubMed]
- Rehaiem, A.; Belgacem, Z.B.; Edalatian, M.R.; Martínez, B.; Rodríguez, A.; Manai, M.; Guerra, N.P. Assessment of potential probiotic properties and multiple bacteriocin encoding-genes of the technological performing strain Enterococcus faecium MMRA. Food Control 2014, 37, 343–350. [Google Scholar] [CrossRef]
- Kurushima, J.; Hayashi, I.; Sugai, M.; Tomita, H. Bacteriocin protein bacl1 of Enterococcus faecalis is a peptidoglycan d-isoglutamyl-l-lysine endopeptidase. J. Biol. Chem. 2013, 288, 36915–36925. [Google Scholar] [CrossRef] [PubMed]
- Kornacki, J.L. Enterococcus faecium NRRL B-2354: Tempest in a teapot or serious foodborne pathogen? Available online: http://www.foodsafetymagazine.com/magazine-archive1/april-may-2012/enterococcus-faecium-nrrl-b-2354-tempest-in-a-teapot-or-serious-foodborne-pathogen/ (accessed on 29 January 2015).
- Kopit, L.M.; Kim, E.B.; Siezen, R.J.; Harris, L.J.; Marco, M.L. Safety of the surrogate microorganism Enterococcus faecium NRRL B-2354 for use in thermal process validation. Appl. Environ. Microbiol. 2014, 80, 1899–1909. [Google Scholar] [CrossRef] [PubMed]
- Cebrián, R.; Baños, A.; Valdivia, E.; Pérez-Pulido, R.; Martínez-Bueno, M.; Maqueda, M. Characterization of functional, safety, and probiotic properties of Enterococcus faecalis UGRA10, a new AS-48-producer strain. Food Microbiol. 2012, 30, 59–67. [Google Scholar] [CrossRef] [PubMed]
- FAO/WHO. Probiotics in Food: Health and Nutritional Properties and Guidelines for Evaluation. Available online: http://tinyurl.com/8bccc3r (accessed on 29 January 2015).
- Liu, G.; Griffiths, M.W.; Wu, P.; Wang, H.; Zhang, X.; Li, P. Enterococcus faecium LM-2, a multi-bacteriocinogenic strain naturally occurring in “byaslag”, a traditional cheese of inner mongolia in china. Food Control 2011, 22, 283–289. [Google Scholar] [CrossRef]
- Perez, R.H.; Himeno, K.; Ishibashi, N.; Masuda, Y.; Zendo, T.; Fujita, K.; Wilaipun, P.; Leelawatcharamas, V.; Nakayama, J.; Sonomoto, K. Monitoring of the multiple bacteriocin production by Enterococcus faecium NKR-5-3 through a developed liquid chromatography and mass spectrometry-based quantification system. J. Biosc. Bioeng. 2012, 114, 490–496. [Google Scholar] [CrossRef]
- Aymerich, T.; Holo, H.; Håvarstein, L.S.; Hugas, M.; Garriga, M.; Nes, I.F. Biochemical and genetic characterization of enterocin a from Enterococcus faecium, a new antilisterial bacteriocin in the pediocin family of bacteriocins. Appl. Environ. Microbiol. 1996, 62, 1676–1682. [Google Scholar] [PubMed]
- Cleveland, J.; Montville, T.J.; Nes, I.F.; Chikindas, M.L. Bacteriocins: Safe, natural antimicrobials for food preservation. Int. J. Food Microbiol. 2001, 71, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Wieckowicz, M.; Schmidt, M.; Sip, A.; Grajek, W. Development of a pcr-based assay for rapid detection of class iia bacteriocin genes. Lett. Appl. Microbiol. 2011, 52, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Naghmouchi, K.; Belguesmia, Y.; Baah, J.; Teather, R.; Drider, D. Antibacterial activity of class I and IIa bacteriocins combined with polymyxin E against resistant variants of Listeria monocytogenes and Escherichia coli. Res. Microbiol. 2011, 162, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Macwana, S.; Muriana, P.M. Spontaneous bacteriocin resistance in Listeria monocytogenes as a susceptibility screen for identifying different mechanisms of resistance and modes of action by bacteriocins of lactic acid bacteria. J. Microbiol. Methods 2012, 88, 7–13. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Henning, C.; Gautam, D.; Muriana, P. Identification of Multiple Bacteriocins in Enterococcus spp. Using an Enterococcus-Specific Bacteriocin PCR Array. Microorganisms 2015, 3, 1-16. https://doi.org/10.3390/microorganisms3010001
Henning C, Gautam D, Muriana P. Identification of Multiple Bacteriocins in Enterococcus spp. Using an Enterococcus-Specific Bacteriocin PCR Array. Microorganisms. 2015; 3(1):1-16. https://doi.org/10.3390/microorganisms3010001
Chicago/Turabian StyleHenning, Chris, Dhiraj Gautam, and Peter Muriana. 2015. "Identification of Multiple Bacteriocins in Enterococcus spp. Using an Enterococcus-Specific Bacteriocin PCR Array" Microorganisms 3, no. 1: 1-16. https://doi.org/10.3390/microorganisms3010001







