Toxic Algal Bloom Recurrence in the Era of Global Change: Lessons from the Chilean Patagonian Fjords
Abstract
1. Introduction
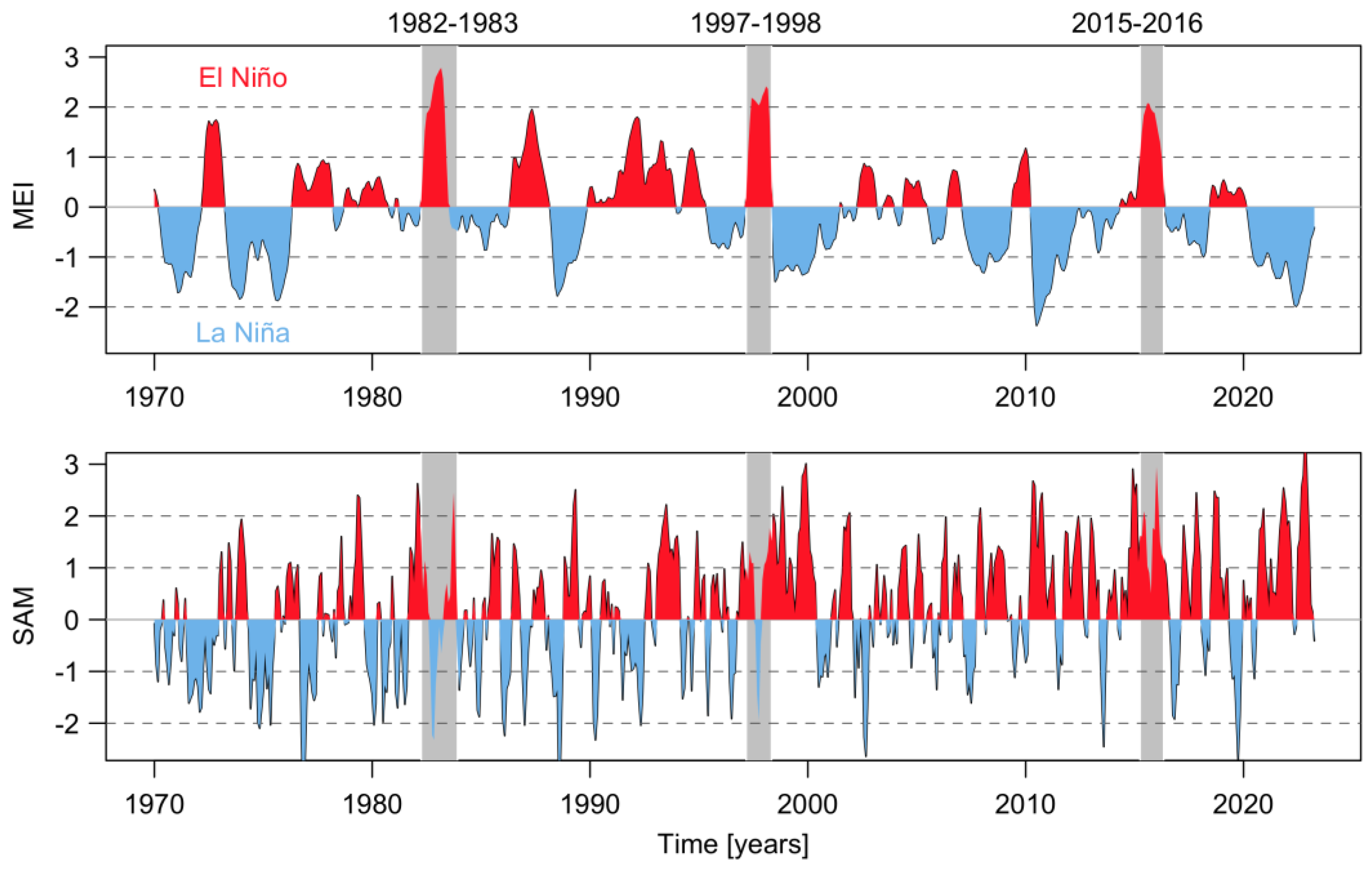
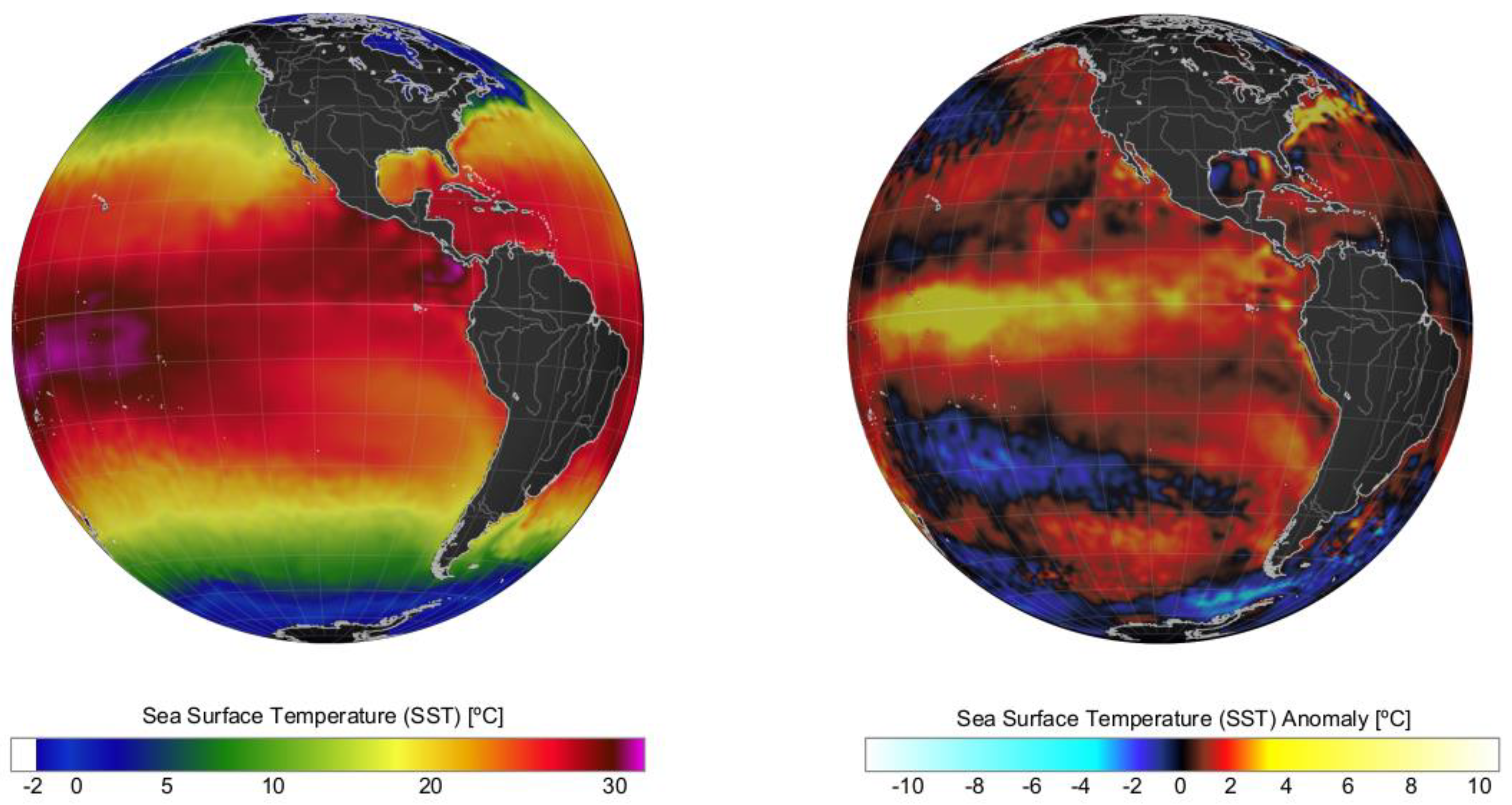
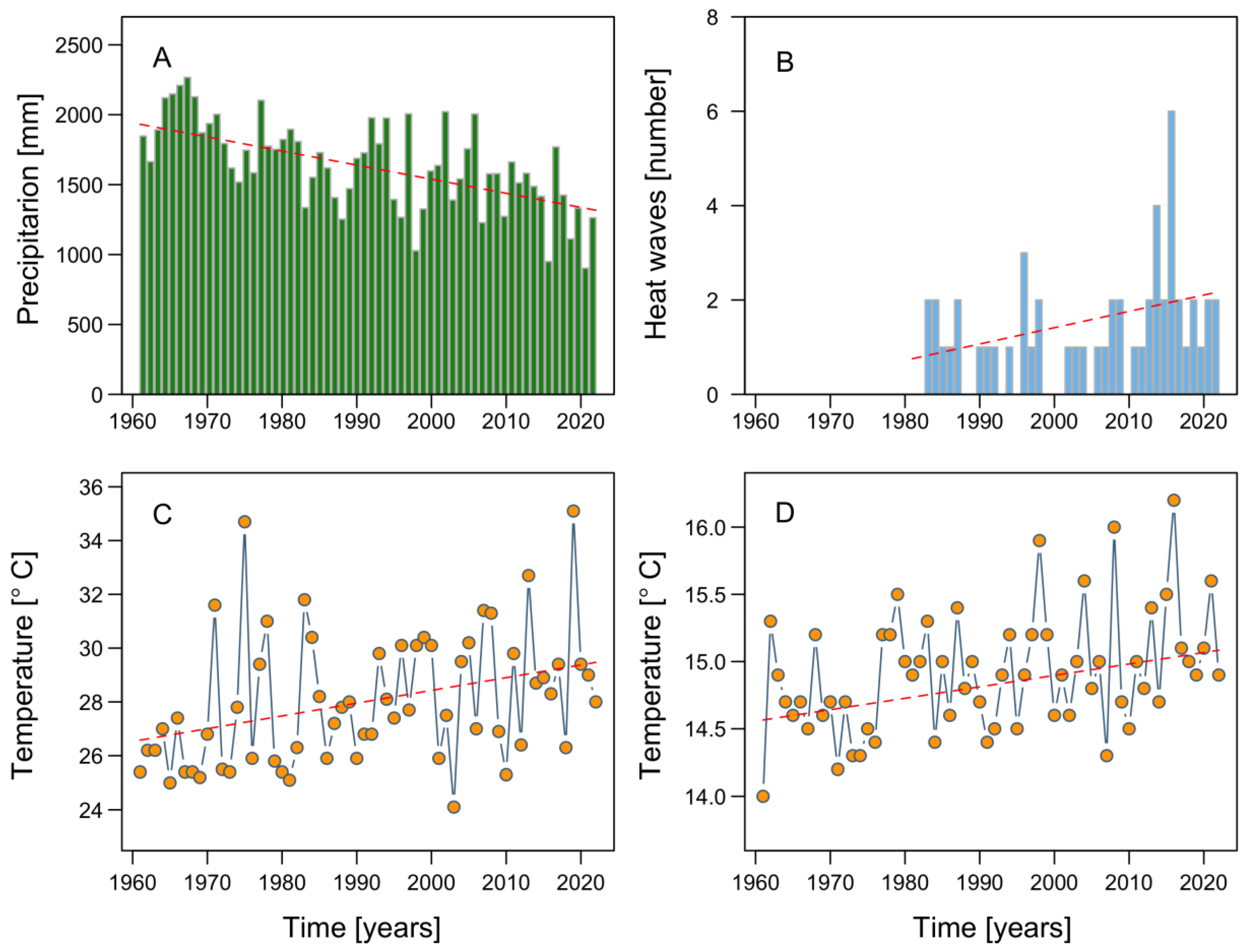
2. Climate Modulation: A Global Phenomenon Affecting Microalgal Local Responses
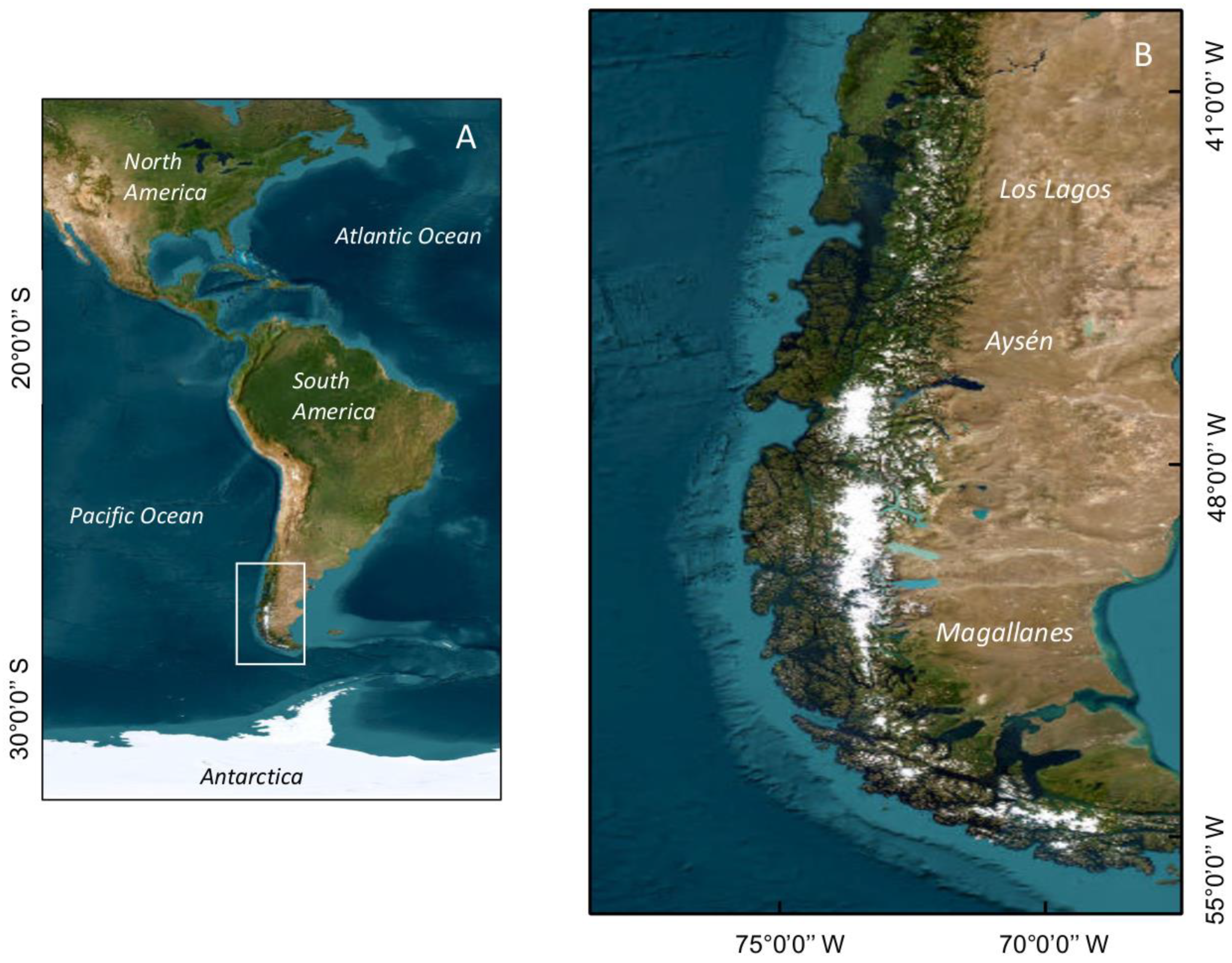
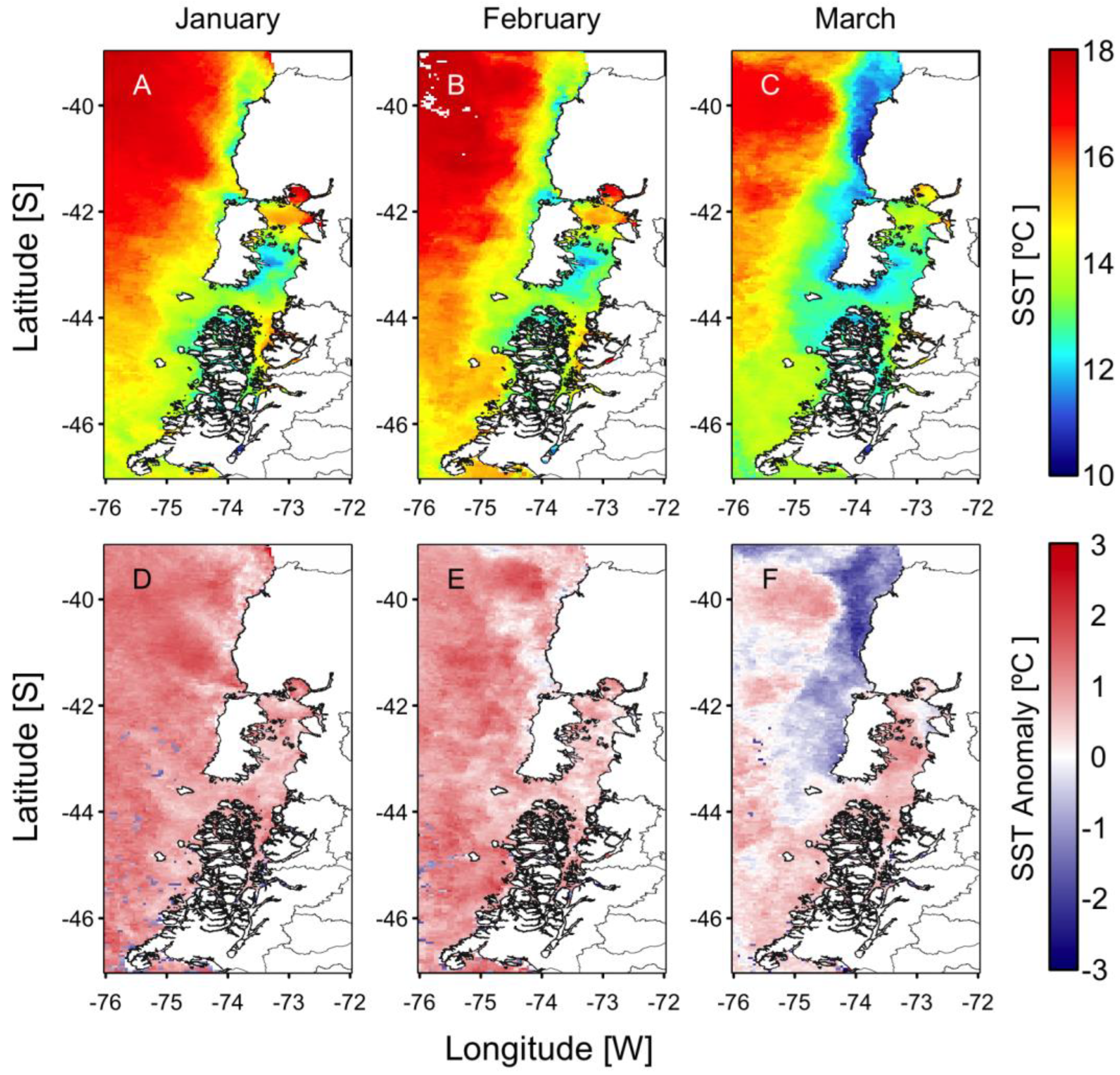
3. Extreme HAB Events in Chilean Patagonia
3.1. HABs of Biotoxin-Producing Microalgae
3.2. Fish-Killing Microalgal Blooms
3.3. High-Biomass Blooms
4. Learning to Live with HAB: Some Lessons
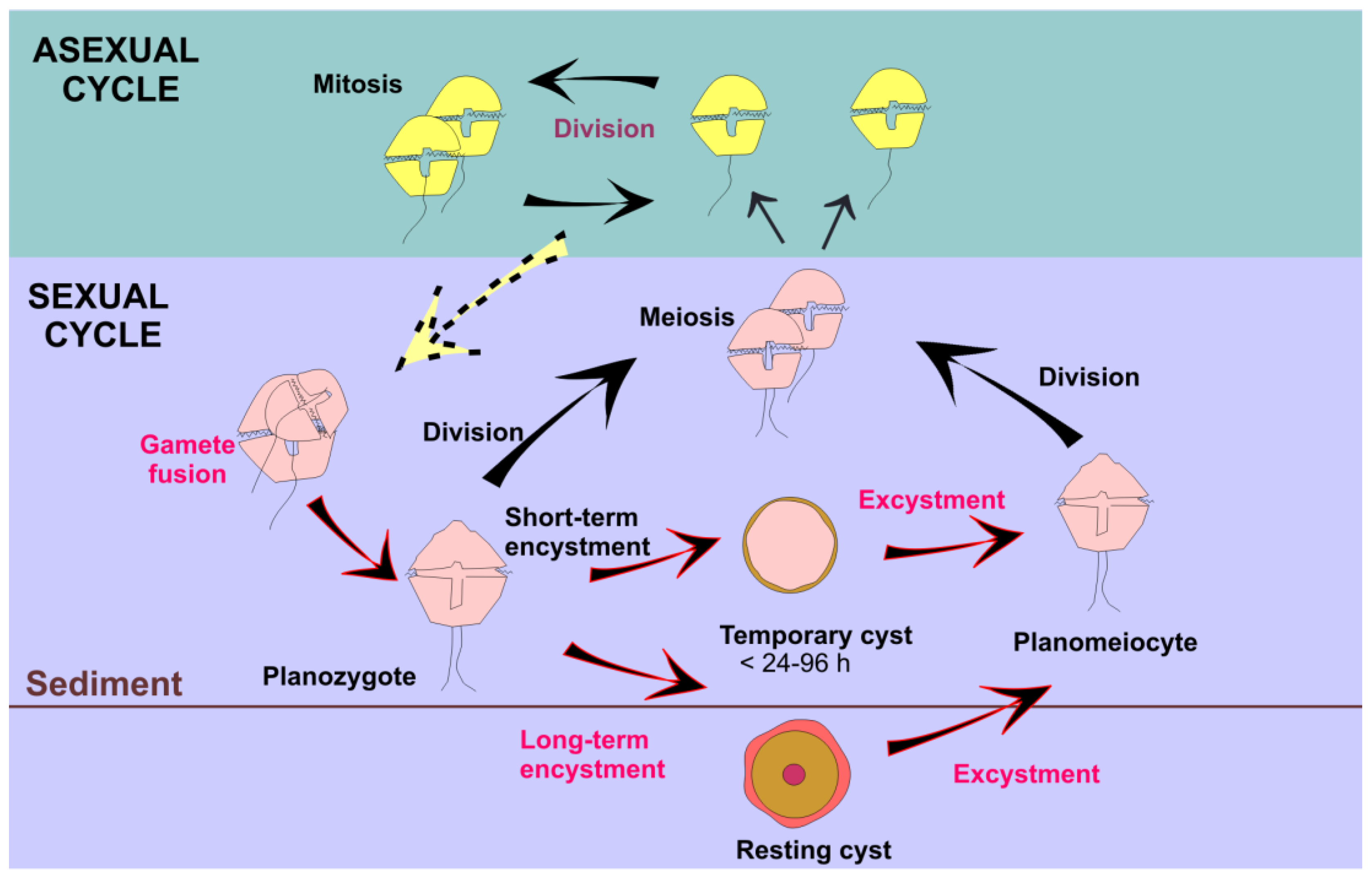
5. Future Perspectives: Potential Species Responses, Trends, and Unresolved Topics
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hallegraeff, G.; Enevoldsen, H.; Zingone, A. Global harmful algal bloom status reporting. Harmful Algae 2021, 102, 101992. [Google Scholar] [CrossRef] [PubMed]
- Marquet, P.A.; Buschmann, A.H.; Corcoran, D.; Díaz, P.A.; Fuentes-Castillo, T.; Garreaud, R.; Pliscoff, P.; Salazar, A. Cambio global y aceleración de las presiones antrópicas en los ecosistemas Patagónicos In Conservación de la Patagonia Chilena: Evaluación del Conocimiento, Oprtunidades y Desafios; Castilla, J.C., Armesto, J.J., Martínez-Harms, M.J., Eds.; Ediciones Universidad Católica: Santiago, Chile, 2021; pp. 65–103. [Google Scholar]
- Díaz, P.A.; Molinet, C.; Seguel, M.; Díaz, M.; Labra, G.; Figueroa, R.I. Species diversity and abundance of dinoflagellate resting cysts seven months after a bloom of Alexandrium catenella in two contrasting coastal systems of the Chilean Inland Sea. Eur. J. Phycol. 2018, 53, 410–421. [Google Scholar] [CrossRef]
- Rodríguez-Villegas, C.; Díaz, P.A.; Salgado, P.; Tomasetti, S.J.; Díaz, M.; Marín, S.L.; Baldich, A.M.; Niklitschek, E.; Pino, L.; Matamala, T.; et al. The role of physico-chemical interactions in the sesonality of toxic dinoflagellate cyst assemblages: The case of NW Patagonian fjords system. Environ. Pollut. 2022, 311, 119901. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Villegas, C.; Lee, M.; Salgado, P.; Figueroa, R.I.; Baldrich, A.; Pérez-Santos, I.; Tomasetti, S.J.; Niklitschek, E.; Díaz, M.; Álvarez, G.; et al. Drivers of dinoflagellate benthic cyst assemblages in the NW Patagonian Fjords System and its adjacent oceanic shelf, with a focus on harmful species. Sci. Total Environ. 2021, 785, 47378. [Google Scholar] [CrossRef]
- Díaz, P.A.; Molinet, C.; Seguel, M.; Díaz, M.; Labra, G.; Figueroa, R. Coupling planktonic and benthic shifts during a bloom of Alexandrium catenella in southern Chile: Implications for bloom dynamics and recurrence. Harmful Algae 2014, 40, 9–22. [Google Scholar] [CrossRef]
- Moore, S.K.; Trainer, V.L.; Mantua, N.J.; Parker, M.S.; Laws, E.A.; Backer, L.C.; Fleming, L.E. Impacts of climate variability and future climate change on harmful algal blooms and human health. Environ. Health 2008, 7, S4–S12. [Google Scholar] [CrossRef]
- Moore, S.; Mantua, N.; Salathe, E. Past trends and future scenarios for environmental conditions favoring the accumulation of paralytic shellfish toxins in Puget Sound shellfish. Hamrful Algae 2011, 10, 521–529. [Google Scholar] [CrossRef]
- León-Muñoz, J.; Urbina, M.A.; Garreaud, R.; Iriarte, J.L. Hydroclimatic conditions trigger record harmful algal bloom in western Patagonia (summer 2016). Sci. Rep. 2018, 8, 1330. [Google Scholar] [CrossRef]
- Trainer, V.L.; Moore, S.K.; Hallegraeff, G.; Kudela, R.M.; Clement, A.; Mardones, J.I.; Cochlan, W.P. Pelagic harmful algal blooms and climate change: Lessons from nature’s experiments with extremes. Harmful Algae 2020, 91, 101591. [Google Scholar] [CrossRef]
- Díaz, P.A.; Pérez-Santos, I.; Basti, L.; Garreaud, R.; Pinilla, E.; Barrera, F.; Tello, A.; Schwerter, C.; Arenas-Uribe, S.; Soto-Riquelme, C.; et al. How local and climate change drivers shaped the formation, dynamics and potential recurrence of a massive fish-killer microalgal bloom in Patagonian fjord. Sci. Total Environ. 2023, 865, 161288. [Google Scholar] [CrossRef]
- Díaz, P.A.; Álvarez, A.; Varela, D.; Pérez-Santos, I.; Díaz, M.; Molinet, C.; Seguel, M.; Aguilera-Belmonte, A.; Guzmán, L.; Uribe, E.; et al. Impacts of harmful algal blooms on the aquaculture industry: Chile as a case study. Perspect. Phycol. 2019, 6, 39–50. [Google Scholar] [CrossRef]
- Mardones, J.I.; Paredes, J.; Godoy, M.; Suarez, R.; Norambuena, L.; Vargas, V.; Fuenzalida, G.; Pinilla, E.; Artal, O.; Rojas, X.; et al. Disentangling the environmental processes responsible for the world’s largest farmed fish-killing harmful algal bloom: Chile, 2016. Sci. Total Environ. 2021, 766, 144383. [Google Scholar] [CrossRef] [PubMed]
- Mardones, J.I.; Paredes-Mella, J.; Flores-Leñero, A.; Yarimuzu, K.; Godoy, M.; Cascales, E.; Espinoza, J.P.; Norambuena, L.; Garreaud, R.; Gonzáles, H.E.; et al. Extreme harmful algal blooms, climate change, and potential risk of eutrophication in Patagonian fjords: Insights from an exceptional Heterosigma akashiwo fish-killing event. Prog. Oceanogr. 2023, 210, 102921. [Google Scholar] [CrossRef]
- Pickard, G.L. Some physical oceanographic features of inlets of Chile. J. Fish. Res. Board Can. 1971, 28, 1077–1106. [Google Scholar] [CrossRef]
- Schneider, W.; Pérez-Santos, I.; Ross, L.; Bravo, L.; Seguel, R.; Hernández, F. On the hydrography of Puyuhuapi Channel, Chilean Patagonia. Prog. Oceanogr. 2014, 128, 8–18. [Google Scholar] [CrossRef]
- Pérez-Santos, I.; Díaz, P.A.; Silva, N.; Garreaud, R.; Montero, P.; Henríquez-Castillo, C.; Barrera, F.; Linford, P.; Amaya, C.; Contreras, S.; et al. Oceanography time series reveals annual asynchrony input between oceanic and estuarine waters in Patagonian fjords. Sci. Total Environ. 2021, 798, 149241. [Google Scholar] [CrossRef]
- Aguirre, C.; Pizarro, Ó.; Strub, P.T.; Garreaud, R.; Barth, J.A. Seasonal dynamics of the near-surface alongshore flow off central Chile. J. Geophys. Res. Ocean. 2012, 117, C01006. [Google Scholar] [CrossRef]
- Saldías, G.S.; Sobarzo, M.; Quiñones, R. Freshwater structure and its seasonal variability off western Patagonia. Prog. Oceanogr. 2018, 174, 143–153. [Google Scholar] [CrossRef]
- Ross, L.; Valle-Levinson, A.; Pérez-Santos, I.; Tapia, F.J.; Schneider, W. Baroclinic annular variability of internal motions in a Patagonian fjord. J. Geophys. Res.-Oceans 2015, 120, 5668–5685. [Google Scholar] [CrossRef]
- Sauter, T. Revisiting extreme precipitation amounts over southern South America and implications for the Patagonian Icefields. Hydrol. Earth Syst. Sci. 2020, 24, 203–2016. [Google Scholar] [CrossRef]
- Viale, M.; Garreaud, R. Orographic effects of the subtropical and extratropical Andes on upwind precipitating clouds. J. Geophys. Res.—Atmos. 2015, 120, 4962–4974. [Google Scholar] [CrossRef]
- Sandoval-Sanhueza, A.; Aguilera-Belmonte, A.; Basti, L.; Figueroa, R.I.; Molinet, C.; Álvarez, G.; Oyadenel, S.; Riobó, P.; Mancilla-Gutiérrez, G.; Díaz, P.A. Interactive effects of temperature and salinity on the growth and cytotoxicity of the sh-killing microalgal species Heterosigma akashiwo and Pseudochattonella verruculosa. Mar. Pollut. Bull. 2022, 174, 113234. [Google Scholar] [CrossRef] [PubMed]
- White, W.B.; Peterson, R.G. An Antarctic circumpolar wave in surface pressure, wind temperature and sea-ice extent. Nature 1996, 380, 699–702. [Google Scholar] [CrossRef]
- Gillett, N.P.; Kell, T.D.; Jones, P.D. Regional climate impacts of the Southern Annular Mode. Geophys. Res. Lett. 2006, 33, L23704. [Google Scholar] [CrossRef]
- Gillett, N.P.; Thompson, D.W.J. Simulation of recent southern hemisphere climate change. Science 2003, 302, 273–275. [Google Scholar] [CrossRef] [PubMed]
- Montecinos, A.; Aceituno, P. Seasonality of the ENSO-related rainfall variability in central Chile and associated circulation anomalies. J. Clim. 2003, 16, 281–296. [Google Scholar] [CrossRef]
- Mantua, N.J.; Hare, S. The Pacific Decadal Oscillation. J. Oceanogr. 2002, 58, 35–44. [Google Scholar] [CrossRef]
- Zhang, Y.; Wallace, J.M.; Battisti, D.S. ENSO-like interdecadal variability. J. Clim. 1997, 10, 1004–1020. [Google Scholar] [CrossRef]
- Fogt, R.L.; Marshall, G.J. The Southern Annular Mode: Variability, trends, and climate impacts across the Southern Hemisphere. WIREs Clim. Change 2020, 11, e652. [Google Scholar] [CrossRef]
- Garreaud, R.; Lopez, P.; Minvielle, M.; Rojas, M. Large-scale control on the Patagonian climate. J. Clim. 2013, 26, 215–230. [Google Scholar] [CrossRef]
- Margalef, R.; Estrada, M.; Blasco, D. Functional morphology of organisms involved in red tides, as adapted to decaying turbulence. In Toxic Dinoflagellate Blooms; Taylor, D., Seliger, H., Eds.; Elsevier: New York, NY, USA, 1979; pp. 89–94. [Google Scholar]
- Wyatt, T. Margalef ’s mandala and phytoplankton bloom strategies. Deep Sea Res. II 2014, 101, 32–49. [Google Scholar] [CrossRef]
- GEOHAB. Global Ecology and Oceanography of Harmful Algal Blooms, GEOHAB Core Research Project: HABs in Fjords and Coastal Embayments; Cembella, A., Guzma, L., Roy, S., Dioge, J., Eds.; IOC: Paris, France; SCOR: Newark, DE, USA, 2010; 57p. [Google Scholar]
- Kudela, R.M.; Berdalet, E.; Enevoldsen, H.; Pitcher, G.; Raine, R.; Urban, E. GEOHAB–The Global Ecology and Oceanography of Harmful Algal Blooms Program: Motivation, goals, and legacy. Oceanography 2017, 30, 12–21. [Google Scholar] [CrossRef]
- Edwards, M.; Richardson, A.J. Impact of climate change on marine pelagic phenology and trophic mismatch. Nature 2004, 430, 881–884. [Google Scholar] [CrossRef]
- Baldrich, A.; Pérez-Santos, I.; Álvarez, G.; Reguera, B.; Fernández-Pena, C.; Rodríguez-Villegas, C.; Araya, M.; Álvarez, F.; Barrera, F.; Karasiewicz, S.; et al. Niche differentiation of Dinophysis acuta and D. acuminata in a stratified fjord. Harmful Algae 2021, 103, 102010. [Google Scholar] [CrossRef]
- Baldich, A.M.; Díaz, P.A.; Álvarez, G.; Pérez-Santos, I.; Schwerter, C.; Díaz, M.; Araya, M.; Nieves, M.G.; Rodríguez-Villegas, C.; Barrera, F.; et al. Dinophysis acuminata or Dinophysis acuta: What Makes the Difference in Highly Stratified Fjords? Mar. Drugs 2023, 21, 64. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M.; Beaugrand, G.; Reid, P.C.; Rowden, A.A.; Jones, M.B. Ocean climate anomalies and the ecology of the North Sea. Mar. Ecol. Prog. Ser. 2002, 239, 1–10. [Google Scholar] [CrossRef]
- Escalera, L.; Reguera, B.; Pazos, Y.; Moroño, A.; Cabanas, J.M. Are different species of Dinophysis selected by climatological conditions? Afr. J. Mar. Sci. 2006, 28, 283–288. [Google Scholar] [CrossRef]
- Hallegraeff, G. Ocean climate change, phytoplankton community responses, and harmful algal blooms: A formidable predictive challenge. J. Phycol. 2010, 46, 220–235. [Google Scholar] [CrossRef]
- Molinet, C.; Lafón, A.; Lembeye, G.; Moreno, C.A. Patrones de distribución espacial y temporal de floraciones de Alexandrium catenella (Whedon & Kofoid) Balech 1985, en aguas interiores de la Patagonia noroccidental de Chile. Rev. Chil. Hist. Nat. 2003, 76, 681–698. [Google Scholar]
- Guzmán, L.; Pacheco, H.; Pizarro, G.; Alárcon, C. Alexandrium catenella y veneno paralizante de los mariscos en Chile. In Floraciones Algales Nocivas en el Cono sur Americano; Sar, E.A., Ferrario, M.E., Reguera, B., Eds.; Instituto Español de Oceanografía: Madrid, Spain, 2002; Volume 11, pp. 235–255. [Google Scholar]
- Álvarez, G.; Díaz, P.A.; Godoy, M.; Araya, M.; Ganuza, I.; Pino, R.; Álvarez, F.; Rengel, J.; Hernández, C.; Uribe, E.; et al. Paralytic Shellfish Toxins in Mesodesma donacium during an exceptional bloom of Alexandrium catenella associated to an intense mass mortality. Toxins 2019, 11, 188. [Google Scholar] [CrossRef]
- Armijo, J.; Oerder, V.; Auger, P.; Bravo, A.; Molina, E. The 2016 red tide crisis in southern Chile: Possible in uence of the mass oceanic dumping of dead salmons. Mar. Pollut. Bull. 2020, 150, 110603. [Google Scholar] [CrossRef]
- Garreaud, R. Record-breaking climate anomalies lead to severe drought and environmental disruption in western Patagonia in 2016. Clim. Res. 2018, 74, 217–229. [Google Scholar] [CrossRef]
- Mardones, J.I.; Fuenzalida, G.; Zenteno, K.; Alves-de-Souza, A.; Astuya, A.; Dorantes-Aranda, J.J. Salinity-growth response and ichthyotoxic potency of the Chilean Pseudochattonella verruculosa. Front. Mar. Sci. 2019, 6, 24. [Google Scholar] [CrossRef]
- Díaz, P.A.; Álvarez, G.; Pizarro, G.; Blanco, J.; Reguera, B. Lipophilic toxins in Chile: History, producers and impacts. Mar. Drugs 2022, 20, 122. [Google Scholar] [CrossRef] [PubMed]
- Mardones, J.I.; Clément, A.; Rojas, X.; Aparicio, C. Alexandrium catenella during 2009 in Chilean waters, and recent expansion to coastal ocean. Harmful Algae News 2010, 41, 8–9. [Google Scholar]
- Buschmann, A.H.; Riquelme, V.A.; Hernández-González, M.A.; Varela, D.; Jiménez, E.; Henríquez, L.A.; Vergara, P.A.; Guíñez, R.; Filúm, L. A review of the impacts of salmonid farming on marine coastal ecosystems in the southeast Pacific. ICES J. Mar. Sci. 2006, 63, 1338–1345. [Google Scholar] [CrossRef]
- Hernández, C.; Díaz, P.A.; Molinet, C.; Seguel, M. Exceptional climate anomalies and northwards expansion of Paralytic Shellfish Poisoning outbreaks in Southern Chile. Harmful Algae News 2016, 54, 1–2. [Google Scholar]
- Lagos, N. Paralytic Shellfish Poisoning Phycotoxins: Occurrence in South America. Comments Toxicol. 2003, 9, 175–193. [Google Scholar] [CrossRef]
- Guzmán, L.; Espinoza-González, O.; Pinilla, E.; Besoain, V.; Calderón, M.J.; Cáceres, J.; Iriarte, L.; Muñoz, V.; Martínez, R.; Hernández, C.; et al. Atmospheric and oceanographic processes on the distribution and abundance of Alexandrium catenella in the North of Chilean fjords. In Proceedings of the 18th International Conference on Harmful Algae, Nantes, France, 21–26 October 2018. [Google Scholar]
- Paredes-Mella, J.; Mardones, J.I.; Norambuena, L.; Fuenzalida, G.; Labra, G.; Espinoza-González, O.; Guzmán, L. Toxic Alexandrium catenella expanding northward along the Chilean coast: New risk of paralytic shell sh poisoning off the Bío-Bío region (36° S). Mar. Pollut. Bull. 2021, 172, 112783. [Google Scholar] [CrossRef]
- Sernapesca. Anuario Estadistico de Pesca; Servicio Nacional de Pesca: Valparaíso, Chile, 2021. [Google Scholar]
- Díaz, P.A.; Álvarez, G.; Seguel, M.; Marín, A.; Krock, B. First detection of pectenotoxin-2 in shellfish associated with an intense spring bloom of Dinophysis acuminata on the central Chilean coast. Mar. Pollut. Bull. 2020, 158, 11414. [Google Scholar] [CrossRef]
- Reguera, B.; Riobó, P.; Rodríguez, F.; Díaz, P.A.; Pizarro, G.; Paz, B.; Franco, J.M.; Blanco, J. Dinophysis toxins: Causative organisms, distribution and fate in shellfish. Mar. Drugs 2014, 12, 394–461. [Google Scholar] [CrossRef] [PubMed]
- Lembeye, G.; Yasumoto, T.; Zhao, J.; Fernández, R. DSP outbreak in Chilean fjords. In Toxic Phytoplankton Blooms in the Sea; Smayda, T.J., Shimizu, Y., Eds.; Elsevier: Ámsterdam, The Netherlands, 1993; pp. 525–529. [Google Scholar]
- Zhao, J.; Lembeye, G.; Cenci, G.; Wall, B.; Yasumoto, T. Determination of okadaic acid and dinophysistoxin-1 in mussels from Chile, Italy and Ireland. In Toxic Phytoplankton Blooms in the Sea; Smayda, T.J., Shimizu, Y., Eds.; Elsevier: Amsterdam, The Netherlands, 1993; pp. 587–592. [Google Scholar]
- Villarroel, O. Detección de toxina paralizante, diarreica y amnésica en mariscos de la XI región por Cromatografía de Alta Resolución (HPLC) y bioensayo de ratón. Cienc. Tecnol. Mar. 2004, 27, 33–42. [Google Scholar]
- Díaz, P.A.; Pérez-Santos, I.; Álvarez, G.; Garreaud, R.; Pinilla, E.; Díaz, M.; Sandoval, A.; Araya, M.; Álvarez, F.; Rengel, J.; et al. Multiscale physical background to an exceptional harmful algal bloom of Dinophysis acuta in a fjord system. Sci. Total Environ. 2021, 773, 145621. [Google Scholar] [CrossRef]
- Diaz, P.A.; Álvarez, G.; Figueroa, R.I.; Garreaud, R.; Pérez-Santos, I.; Schwerter, C.; Díaz, M.; López, L.; Pinto-Torres, M.; Krock, B. From lipophilic to hydrophilic toxin producers: Phytoplankton succession driven by an atmospheric river in western Patagonia. Mar. Pollut. Bull. 2023, 193, 115214. [Google Scholar] [CrossRef]
- Suárez-Isla, B.A.; López-Rivera, A.; Hernández, C.; Clément, A.; Guzmán, L. Impacto económico de las floraciones de microalgas nocivas en Chile y datos recientes sobre la ocurrencia de veneno amnésico de los mariscos. In Floraciones Algales Nocivas en el Cono sur Americano; Sar, E., Ferrario, M.E., Reguera, B., Eds.; Instituto Español de Oceanografía: Madrid, Spain, 2002; pp. 257–268. [Google Scholar]
- Espinoza-González, O.; Guzmán, L.; Opazo, D.; Pinilla, E.; Hernández, C.; Norambuena, L.; Palma, M. Historical blooms of Pseudo-nitzschia and outbreaks of domoic acid in the inner sea of Chiloé island, Southern Chile, 2020–2021. In Proceedings of the 19th International Conference on Harmful Algae, La Paz, Mexico, 11–15 October 2018. [Google Scholar]
- Díaz, P.A.; Pérez-Santos, I.; Schwerter, C.; Arenas, S.; Navarro, P.; Mancilla-Gutiérrez, G.; Barrera, F. Multi-specific Harmful Algal Bloom in a Chilean Fjord: A dangerous phytoplankton cocktail. Harmful Algae News 2022, 70, 8–9. [Google Scholar]
- Buschmann, A.; Farías, L.; Tapia, F.; Varela, D.; Vásquez, M. Scientific Report on the 2016 Southern Chile Red Tide; Chilean Department of Economy: Santiago, Chile, 2016; 66p. [Google Scholar]
- Clément, A.; Lincoqueo, L.; Saldivia, M.; Brito, C.G.; Muñoz, F.; Fernández, C.; Pérez, F.; Maluje, C.P.; Correa, N.; Mondaca, V.; et al. Exceptional summer conditions and HABs of Pseudochattonella in southern Chile create record impacts on salmon farm. Harmful Algae News 2016, 53, 1–3. [Google Scholar]
- Sernapesca. Manual de Implementación de la Normativa Excepcional Aplicable Ante Eventos de Mortalidades Masivas; Sernapesca: Victoria, Chile, 2017; 21p. [Google Scholar]
- Clement, A.; Lembeye, G. Phytoplankton Monitoring Program in the Fish Farming Region of South Chile. In Toxic Phytoplankton Blooms in the Sea; Smayda, T., Shimizu, Y., Eds.; Elsevier Science Publishers B.V.: Amsterdam, The Netherlands, 1993; pp. 223–228. [Google Scholar]
- Edvardsen, B.; Imai, I. The ecology of harmful flagellates within Prymnesiophyceae and Raphidophyceae. In Ecology of Harmful Algae. Ecological Studies (Analysis and Synthesis); Granéli, E., Turner, J.T., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; Volume 189. [Google Scholar]
- Twiner, J.M.; Dixon, S.J.; Trick, C.G. Toxic effects of Heterosigma akashiwo do not appear to be mediated by hydrogen peroxide. Limnol. Oceanogr. 2001, 46, 1400–1405. [Google Scholar] [CrossRef]
- Silke, J.; O’Beirn, F.; Cronin, M. Karenia Mikimotoi: An Exceptional Dinoflagellate Bloom in Western Irish Waters, Summer 2005; Marine Institute: Galway, Ireland, 2005. [Google Scholar]
- Uribe, C.; Ruiz, M. Gymnodinium Brown Tide in the Magellanic Fjords, Southern Chile. Rev. Biol. Mar. Ocenog. 2001, 36, 155–164. [Google Scholar] [CrossRef]
- Villanueva, F.; Cortez, H.; Uribe, C.; Peña, P.; Cassis, D. Mortality of Chilean farmed salmon in wellboats in transit through a Karenia bloom. Harmful Algae News 2017, 57, 4–5. [Google Scholar]
- Mardones, J.; Norambuena, L.; Paredes-Mella, J.; Fuenzalida, G.; Dorantes-Aranda, J.J.; Lee Chang, K.L.; Guzmán, L.; Krock, B.; Hallegraeff, G. Unraveling the Karenia selliformis complex with the description of a non-gymnodimine producing Patagonian phylotype. Harmful Algae 2020, 98, 101892. [Google Scholar] [CrossRef]
- Vellojin, J.P.; Mardones, J.I.; Vargas, V.; Leal, P.P.; Corredor-Acosta, A.; Iriarte, J.L. Potential effects of climate change on the growth response of the toxic dino agellate Karenia selliformis from Patagonian waters of Chile. Prog. Oceanogr. 2023, 211, 102956. [Google Scholar] [CrossRef]
- Iriarte, J.L.; Quiñones, R.; González, R. Relationship between biomass and enzymatic activity of a bloom-forming dinoflagellate (Dynophyceae) in southern Chile(41º S): A field approach. J. Plankton Res. 2005, 27, 159–166. [Google Scholar] [CrossRef]
- Rodríguez-Benito, C.; Navarro, C.; Caballero, I. Using Copernicus Sentinel-2 and Sentinel-3 data to monitor harmful algal blooms in Southern Chile during the COVID-19 lockdown. Mar. Pollut. Bull. 2020, 161, 111722. [Google Scholar] [CrossRef]
- Serre-Fredj, L.; Jacqueline, F.; Navon, M.; Izabel, G.; Chasselin, L.; Jolly, O.; Repecaud, M.; Claquin, P. Coupling high frequency monitoring and bioassay experiments to investigate a harmful algal bloom in the Bay of Seine (French-English Channel). Mar. Pollut. Bull. 2021, 168, 112387. [Google Scholar] [CrossRef] [PubMed]
- Mancilla-Gutiérrez, G.; Díaz, P.A.; Pérez-Santos, I.; Pujol, C.; Schwerter, C.; Altamirano, R.; Arenas-Uribe, S.; Navarro, P.; Saldías, G.S. High-biomass harmful algal blooms (HB-HAB) in the Chilean fjords system: Lepidodinium chlorophorum. Harmful Algae News 2022, 71, 5–6. [Google Scholar]
- GEOHAB. Global Ecology and Oceanography of Harmful Algal Blooms, Science Plan; Glibert, P., Pitcher, G., Eds.; SCOR: Baltimore, MD, USA; IOC: Paris, France, 2001. [Google Scholar]
- Trainer, V.L.; Pitcher, G.C.; Reguera, B.; Smayda, T.J. The distribution and impacts of harmful algal bloom species in eastern boundary upwelling systems. Prog. Oceanogr. 2010, 85, 33–52. [Google Scholar] [CrossRef]
- Smayda, T. Adaptations and selection of harmful and other dinoflagellate species in upwelling systems 1. Morphology and adaptive polymorphism. Prog. Oceanogr. 2010, 85, 53–70. [Google Scholar] [CrossRef]
- Smayda, T. Adaptations and selection of harmful and other dinoflagellate species in upwelling systems. 2. Motility and migratory behaviour. Prog. Oceanogr. 2010, 85, 71–91. [Google Scholar] [CrossRef]
- Soto, D.; León-Muñoz, J.; Garreaud, R.; Quiñones, R.A. Scientific warnings could help to reduce farmed salmon mortality due to harmful algal blooms. Mar. Policy 2021, 133, 104705. [Google Scholar] [CrossRef]
- Linford, P.; Pérez-Santos, I.; Montero, P.; Díaz, P.; Aracena, C.; Pinilla, E.; Barrera, F.; Castillo, M.; Alvera-Azcárate, A.; Alvarado, M.; et al. Oceanographic processes favoring deoxygenation inside Patagonian fjords. EGUsphere 2023, 1–34. [Google Scholar] [CrossRef]
- Pérez-Santos, I.; Seguel, R.; Schneider, W.; Linford, P.; Donoso, D.; Navarro, E.; Amaya-Cárcamo, C.; Pinilla, E.; Daneri, G. Synoptic-scale variability of surface winds and ocean response to atmospheric forcing in the eastern austral Pacific Ocean. Ocean Sci. Discuss. 2019, 15, 1247–1266. [Google Scholar] [CrossRef]
- Narváez, D.; Vargas, C.; Cuevas, A.; García-Loyola, S.; Lara, C.; Segura, C.; Tapia, F.; Broitman, B. Dominant scales of subtidal variability in coastal hydrography of the Northern Chilean Patagonia. J. Marine Syst. 2019, 193, 59–73. [Google Scholar] [CrossRef]
- Mardones, J.I.; Flores-Leñero, A.; Pinto, M.; Vergara, M.; Leal, P.; Paredes-Mella, J. Sistemas de mitigación de microalgas en el sur de Chile: Una tarea pendiente. Salmonxpert 2023, 116, 46–55. [Google Scholar]
- Rodríguez-Villegas, C.; Figueroa, R.I.; Pérez-Santos, I.; Molinet, C.; Saldías, G.S.; Rosales, S.A.; Álvarez, G.; Linford, P.; Díaz, P.A. Continental shelf off northern Chilean Patagonia: A potential risk zone for the onset of Alexandrium catenella toxic bloom? Mar. Pollut. Bull. 2022, 184, 114103. [Google Scholar] [CrossRef]
- Wells, M.; Trainer, V.; Smayda, T.; Karlson, B.; Trick, C.; Kudela, R.; Ishikawa, A.; Bernard, S.; Wulff, A.; Anderson, D.; et al. Harmful algal blooms and climate change: Learning from the past and present to forecast the future. Harmful Algae 2015, 49, 68–93. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, G.E. Population studies-animal ecology and demography-concluding remarks. In Cold Spring Harbor Symposia on Quantitative Biology; Bungtown, R.D., Ed.; Cold Spring Harbor Lab Press: New York, NY, USA, 1957; Volume 22, pp. 415–427. [Google Scholar]
- Aguilera-Belmonte, A.; Inostroza, I.; Saéz Carrillo, K.; Franco, J.M.; Riobo, P.; Gómez, P.I. The combined effect of salinity and temperature on the growth and toxin content of four Chilean strains of Alexandrium catenella (Whedon and Kofoid) Balech 1985 (Dinophyceae) isolated from an outbreak occurring in southern Chile in 2009. Harmful Algae 2013, 23, 55–59. [Google Scholar] [CrossRef]
- Aguayo, R.; León-Muñoz, J.; Vargas-Baecheler, J.; Montecinos, A.; Garreaud, R.; Urbina, M.; Soto, D.; Iriarte, J.L. The glass half-empty: Climate change drives lower freshwater input in the coastal system of the Chilean Northern Patagonia. Clim. Change 2019, 155, 417–435. [Google Scholar] [CrossRef]
- Boisier, J.P.; Alvarez-Garreton, C.; Cordero, R.; Damian, A.; Gallardo, L.; Garreaud, R.; Lambert, F.; Ramallo, C.; Rojas, M.; Rondanelli, R. Anthropogenic drying in central-southern Chile evidenced by long term observations and climate model simulations. Elem. Sci. Anthr. 2018, 6, 74. [Google Scholar] [CrossRef]
- Klais, R.; Tamminen, T.; Kremp, A.; Spilling, K.; Olli, K. Decadal-scale changes of dinoflagellates and diatoms in the anomalous Baltic Sea spring bloom. PLoS ONE 2011, 6, e21567. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, R.I.; Vázquez, J.A.; Massanet, A.; Murado, M.A.; Bravo, I. Interactive effects of salinity and temperature on planozygote and cyst formation on Alexandrium minutum (Dinophyceae). J. Phycol. 2011, 47, 13–24. [Google Scholar] [CrossRef]
- Ramilo, I.; Figueroa, R.I.; Rayón-Viña, F.; Cuadrado, Á.; Bravo, I. Temperature-dependent growth and sexuality of the ciguatoxin producer dinoflagel-late Gambierdiscus spp. in cultures established from the Canary Islands. Harmful Algae 2021, 110, 102130. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, R.I.; Garces, E.; Bravo, I. Comparative study of the life cycles of Alexandrium tamutum and Alexandrium minutum (Gonyaulacales, Dinophyceae) in culture. J. Phycol. 2007, 43, 1039–1053. [Google Scholar] [CrossRef]
- Brosnahan, M.L.; Fischer, A.D.; Lopez, C.B.; Moore, S.K.; Anderson, D.M. Cyst-forming dinoflagellates in a warming climate. Harmful Algae 2020, 91, 101728. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.M. Shellfish toxicity and dormant cysts in toxic dinoflagellates blooms. In Seafood Toxins; Ragelis, E.P., Ed.; American Chemical Society Symposium Series N° 262; American Chemical Society: Washington, DC, USA, 1984; pp. 125–138. [Google Scholar]
- Brosnahan, M.L.; Velo-Suárez, L.; Ralston, D.K.; Fox, S.E.; Sehein, T.R.; Shalapyonok, A.; Sosik, H.K.; Olson, R.J.; Anderson, D.M. Rapid growth and concerted sexual transitions by a bloom of the harmful dinoflagellate Alexandrium fundyense (Dinophyceae). Limnol. Oceanogr. 2015, 60, 2059–2078. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, R.I.; Dapena, C.; Bravo, I.; Cuadrado, A. The hidden sexuality of Alexandrium minutum: An example of overlooked sex in dinoflagellates. PLoS ONE 2015, 10, e0142667. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díaz, P.A.; Figueroa, R.I. Toxic Algal Bloom Recurrence in the Era of Global Change: Lessons from the Chilean Patagonian Fjords. Microorganisms 2023, 11, 1874. https://doi.org/10.3390/microorganisms11081874
Díaz PA, Figueroa RI. Toxic Algal Bloom Recurrence in the Era of Global Change: Lessons from the Chilean Patagonian Fjords. Microorganisms. 2023; 11(8):1874. https://doi.org/10.3390/microorganisms11081874
Chicago/Turabian StyleDíaz, Patricio A., and Rosa I. Figueroa. 2023. "Toxic Algal Bloom Recurrence in the Era of Global Change: Lessons from the Chilean Patagonian Fjords" Microorganisms 11, no. 8: 1874. https://doi.org/10.3390/microorganisms11081874
APA StyleDíaz, P. A., & Figueroa, R. I. (2023). Toxic Algal Bloom Recurrence in the Era of Global Change: Lessons from the Chilean Patagonian Fjords. Microorganisms, 11(8), 1874. https://doi.org/10.3390/microorganisms11081874









